1 Health Sciences North Research Institute, Sudbury, ON P3E 2H2, Canada
2 Flosonics Medical, Toronto, ON M5V 2Y1, Canada
3 Northern Ontario School of Medicine, Sudbury, ON P3E 2C6, Canada
Abstract
Background: The change in the corrected flow time of the common carotid
artery (ccFT
Keywords
- carotid artery
- corrected flow time
- fluid responsiveness
- passive leg raise
- Frank-Starling
A fundamental tenet of cardiac physiology is that of stretch-induced myofibrillar calcium sensitivity [1, 2]. That is, when cardiac muscle fibers are lengthened, their subsequent contraction is more forceful because muscle-shortening hinges upon intracellular calcium. Practically, this mechanism is exploited whenever a clinician administers a patient intravenous (IV) fluid to increase stroke volume (SV) [3, 4]. In other words, increasing the volume of blood returning to the heart with IV fluid stretches cardiac myocytes and augments blood flow from the heart to the arteries; this is typically done to rectify diminished organ perfusion [3].
However, the relationship between cardiac stretch and its output does not increase linearly without change. As described by the Frank-Starling-Sarnoff curve, there is normally a steep increase in SV for each increment in cardiac filling until a plateau is reached, beyond which further cardiac filling has little effect on SV [5, 6, 7]. As well, during acute and chronic illnesses, the slope of this curve can flatten such that cardiac filling (i.e., myocardial stretch) has little effect on SV [6]. In this pathophysiological situation, providing IV fluid is physiologically ineffective and, arguably, harmful [8, 9, 10, 11]; this has led clinicians in the intensive care unit (ICU) [8], emergency department (ED) [12, 13] and operating room (OR) [14] to test patients for their ability to respond to IV fluid and mitigate the risk of volume overload [8, 9].
One commonly-employed maneuver to determine if a patient will increase SV in response to IV fluid, or not, is the passive leg raise (PLR) [15, 16, 17]. The PLR involves measuring baseline SV in the semi-recumbent position and then again with the patient supine and legs raised. This positional change mobilizes 200–300 mL of blood towards the heart; a 10–15% increase in SV indicates that the patient will most-likely respond appropriately to IV fluid. While much has been reported on the PLR in critically-ill patients [17, 18, 19], relatively little has been described about this maneuver and its time-course in healthy volunteers [20]. Studying healthy subjects is important because roughly 50% of critically-ill patients are preload unresponsive when a PLR maneuver is performed [16, 18]. However, the fraction of preload unresponsive patients is much lower early in illness [21, 22] suggesting that preload unresponsiveness is a pathological state that evolves during the disease arc. Consistent with this are our observations wherein no healthy subject was unresponsive to preload when performing a squat maneuver [23, 24] or undergoing simulated blood transfusion following moderate-to-severe central hypovolemia [25, 26]. Furthermore, Søndergaard recently observed that 82% of healthy volunteers were preload responsive by stroke volume as evoked by the passive leg raise [27]. Nevertheless, we have not fully-described the fraction of preload unresponsive, ambulatory volunteers during PLR. Finally, our group [23, 25, 26, 28] and others [29] have successfully used the common carotid artery corrected flow time (ccFT) to assess preload reserve. The ccFT is easily obtained from the Doppler spectrogram; it is the duration of mechanical systole (in milliseconds) corrected for heart rate and the direct relationship between the duration of systole and venous return (i.e., cardiac output) has been known for nearly a century [23, 30]. Given the above, in this ambulatory volunteer study, we report on the change in SV and ccFT during a PLR maneuver. We hypothesized that in ambulatory volunteers, preload responsiveness would be nearly ubiquitous and also that the change in ccFT would be an accurate surrogate to detect this state.
We included ambulatory, volunteers at least 18 years of age who were non-fasted
and clinically euvolemic. Euvolemia was determined by normal resting vital signs,
the absence of medical comorbidities known to change blood volume (e.g.,
congestive heart failure, chronic kidney disease), the absence of peripheral
edema or dyspnea and no change in baseline body weight by
The Clearsight (Edwards Lifesciences; Irvine, CA, USA) was applied to the subject in
the semi-recumbent (i.e., semi-Fowler) position. Clearsight (CS) is a
non-invasive pulse contour analysis SV monitor that uses the ‘volume clamp’
method to provide SV every 20 seconds [31]. CS can track changes in cardiac
output and is most accurate in ambulatory volunteers without problems of
peripheral perfusion [31]. The protocol did not begin until there was adequate CS
signal as measured by the Physiocal calibration metric (i.e.,
A U.S. FDA-cleared, wearable carotid Doppler patch (Flosonics Medical, Sudbury, ON, Canada) was placed over the carotid artery below the angle of the jaw to ensure Doppler sampling below the bifurcation of the common carotid artery (Fig. 1). When an audible Doppler shift was heard and a Doppler spectrum consistent with the common carotid artery visualized, the Doppler patch was adhered in place. As previously described [32], the wearable ultrasound is a 4 MHz, continuous wave (CW) Doppler that does not image the carotid artery. The insonation angle is 60 degrees and generated by a physical ‘wedge’ on the face of the transducer. Given that it is CW, the Doppler sample volume is the entirety of the vessel lumen which makes uniform insonation [33, 34] more likely. The automated maximum velocity trace was used to determine the duration of systole from the systolic velocity upstroke to the dicrotic notch (i.e., flow time); the duration of systole in seconds was used to calculate the ccFT per the equation of Wodey, as previously described [23, 24, 25, 26]. Heart rate was used to align the non-invasive pulse contour device and Doppler spectral signals; the calculation of heart rate was standard across participants. Heart rate from the pulse contour device was outputted every 20 seconds and that data was aligned to a 20 second moving average heart rate from the wearable Doppler device. Implemented in Python, the cross correlation of the heart rate signals evaluated the signal similarity for each 20 second time lag. The maximum correlation value between signals was found and the corresponding time lag was used to align signals.
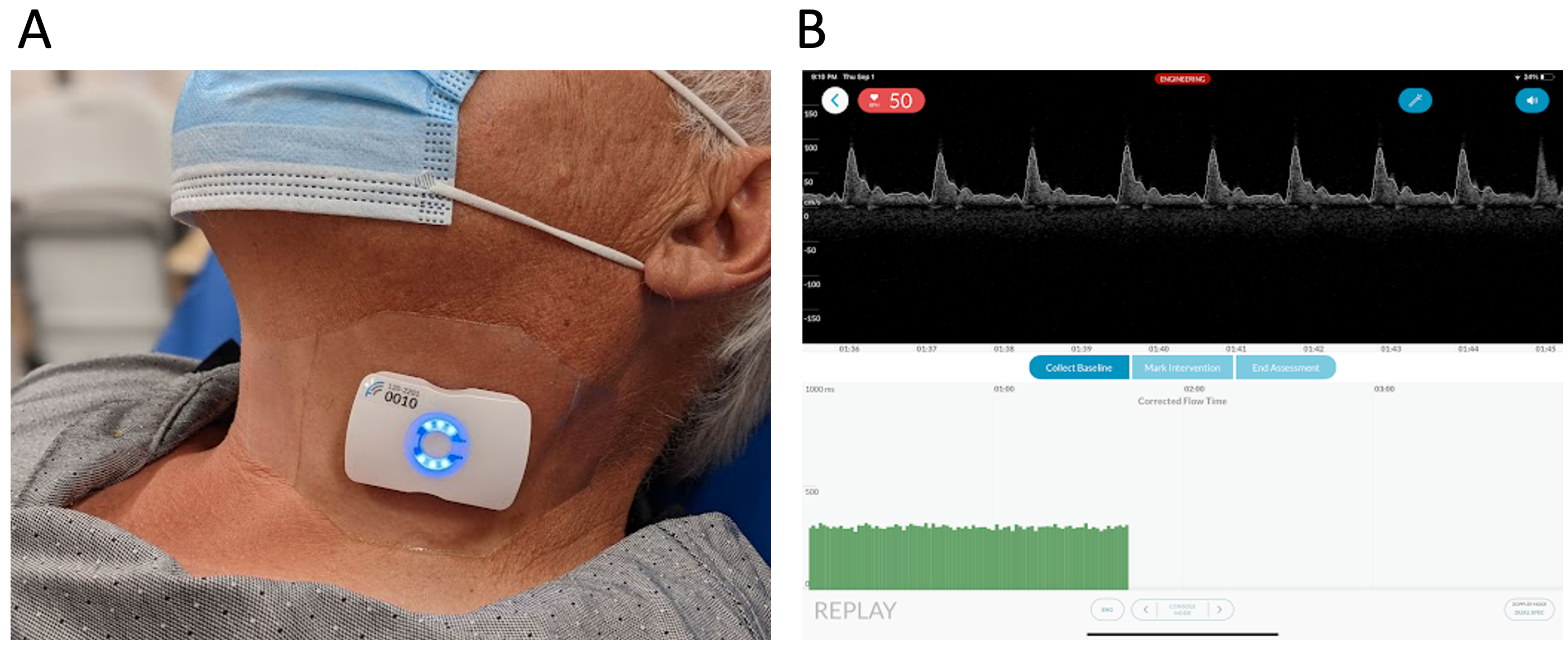 Fig. 1.
Fig. 1.The wireless, wearable Doppler ultrasound system. (A) The device on the neck of a volunteer. (B) The graphical user interface of the ultrasound system displayed on an iOS device.
Each protocol consisted of a 60 second resting baseline followed by passive torso lowering to the supine position with legs elevated to no more than 45 degrees, for an additional 3 minutes (i.e., the PLR) [17, 18]; thus, each protocol was 4 minutes in total duration. Each volunteer performed 2 PLR maneuvers separated by at least 5 minutes of ‘washout’ where they remained at resting baseline prior to the second maneuver.
Baseline values for all measures were calculated from the time window at which SV was the lowest during the baseline period and percent change for each 20 second measure was calculated by reference to this resting baseline; this was done to ascertain the greatest hemodynamic effect of the PLR.
As the non-invasive pulse contour device updates and displays hemodynamic data
every 20 seconds, we analyzed the change during the PLR every 20 seconds.
Therefore, each 3-minute PLR consisted of 9 data points. To evaluate the ability
of the ccFT to detect a 10% change in SV (SV
We used a Willcoxon signed-rank two-sided test, comparing baseline stroke volume and heart rate of the first and second passive leg raises to determine if the 5-minute washout period was adequate. We also used Willcoxon signed-rank two-sided test to determine if there was a difference in all measured hemodynamic variables during the passive leg raise. Finally, test-retest reliability for the two passive leg raise maneuvers was assessed by interclass correlation (ICC).
21 adult volunteers were studied; 11 were women and 10 were men. One subject was entirely excluded for poor carotid Doppler placement with unusable signal; a second subject was entirely excluded because of low Physiocal on the CS device (i.e., less than 10). A single PLR was excluded as there was a significant Doppler angle change noted during the PLR; another PLR was excluded because of low Physiocal noted during the maneuver. The baseline characteristics of the volunteers included in the final analysis are listed in Table 1.
| n = 19 | Mean | std |
|---|---|---|
| Patient Age | 30.95 | |
| Patient Height (m) | 1.71 | |
| Patient Weight (kg) | 82.84 | |
| BMI (kg/m |
27.48 | |
| MAP (mmHg) | 88.37 | |
| HR (bpm) | 70.21 | |
| SV (mL/b) | 87.87 | |
| Systolic Blood Pressure (mmHg) | 118.37 | |
| ccFT (ms) | 310.69 | |
| The average between sessions for each subject was used to calculate the mean and
standard deviation. M is meters, kg/m | ||
Effect of PLR on Hemodynamics: There were 36 PLRs performed across 19
subjects. The % SV
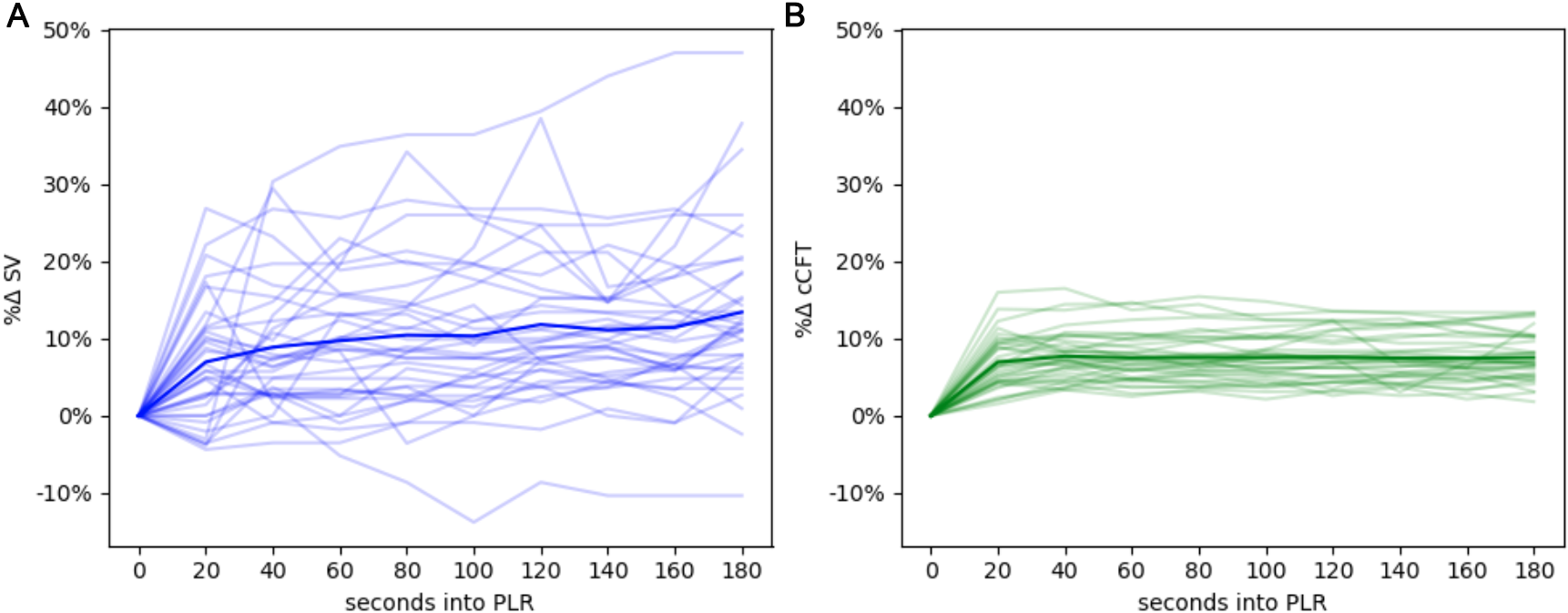 Fig. 2.
Fig. 2.The hemodynamic evolution of the PLR. (A) The change in stroke volume across the PLR, each faint line represents a single maneuver, the emboldened line is the average of all maneuvers over time. (B) The change in ccFT across the PLR.
Willcoxon signed-rank two-sided test comparing HR and SV between the baseline periods of the first and second PLR showed no significant difference; that is, p = 0.17 and p = 0.55, respectively.
Wilcoxon signed-rank test was also used to compare baseline, maximal
intervention pairs for each PLR across participants. Statistically, all values
except heart rate showed a statistically-significant difference in ranked means,
though only ccFT was clinically significant. Mean arterial pressure fell by 3.2
mmHg (p = 0.008), heart rate fell by 1.8 beats per minute (p =
0.1), stroke volume increased by 8.23 mL (p
Test-retest reliability for the two passive leg raise maneuvers was moderate for both percent change SV (ICC = 0.54) and ccft (ICC = 0.54).
Diagnostic Characteristics of Carotid Artery Corrected Flow Time: In
total 8856 carotid beats were considered in this analysis. Fig. 3 shows the
diagnostic characteristics of % ccFT
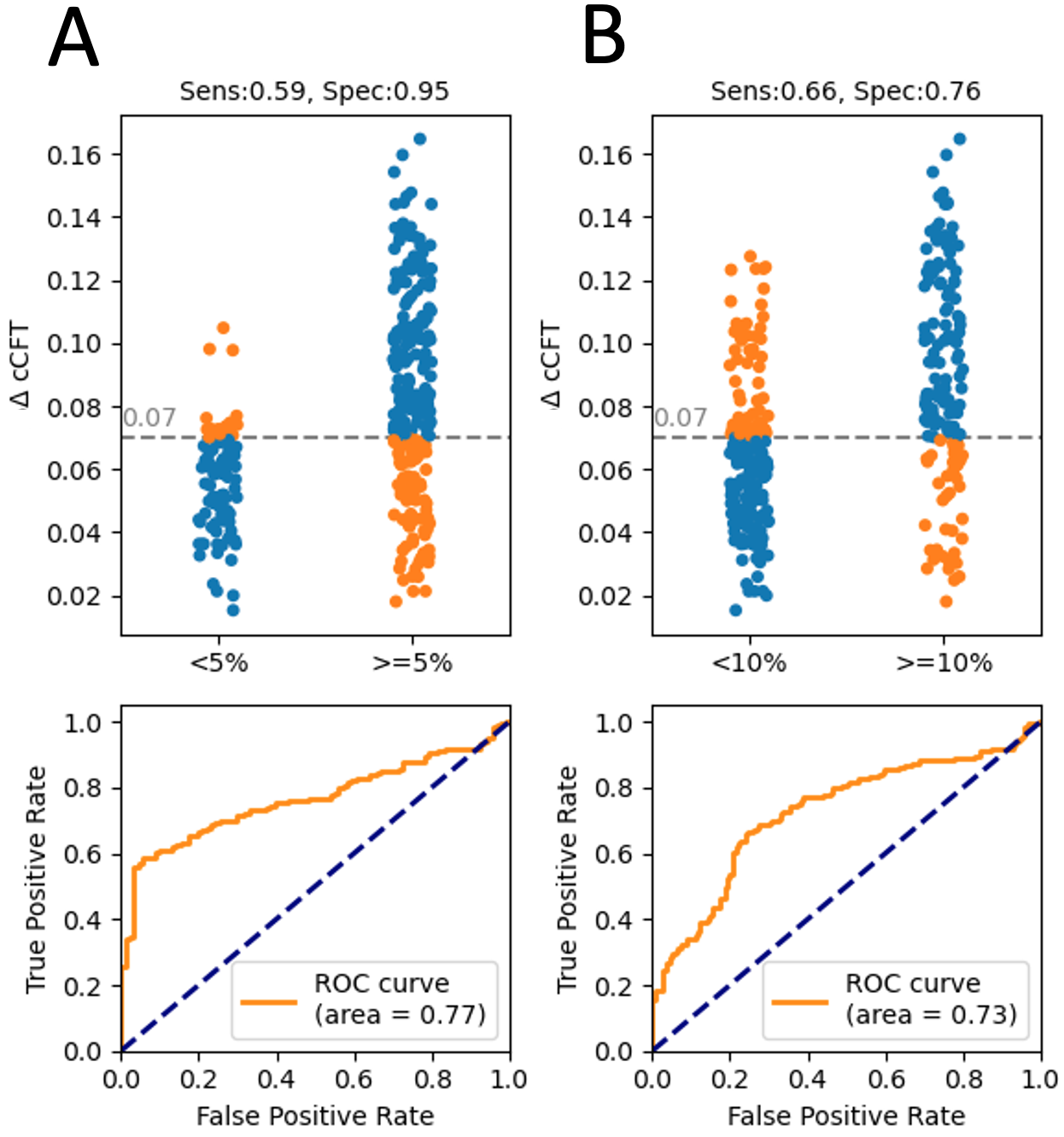 Fig. 3.
Fig. 3.The diagnostic accuracy of changing ccFT at detecting changing CS SV. (A) shows the characteristics for a +5% change in CS SV. (B) for detecting a +10% change in CS SV.
Our results are clinically-important for reasons that flow from the hypotheses
described at the outset; that is, that the ccFT
First, previous investigators have reported 40% error in the CS device when
tracking SV
Second, we observed that the change in the ccFT
Third, that such a large fraction of our volunteers was ‘preload responsive’ provides an important lesson in basic, clinical hemodynamics: the non-fasted, euvolemic, individual is normally preload responsive when assessed by a PLR. In other words, being ‘preload responsive’ does not mean that a patient has a fluid deficit (i.e., is in a state of hypovolemia), nor does it mean that a patient necessarily requires volume replacement [16]. Being ‘preload responsive’ simply means that an individual has recruitable stroke volume, which is evolutionarily advantageous during times of stress. This observation, that ambulatory volunteers normally have preload reserve, calls into question resuscitation protocols that encourage IV fluid until ‘fluid responsiveness’ disappears—which is likely an abnormal state [45, 46]. This could explain why some studies that dictated IV fluid administration until fluid responsiveness was extinguished demonstrated no benefit and excessive IV fluid provision [12, 46, 47]. Our results are also in contradistinction with the finding of Godfrey and colleagues who observed that healthy volunteers were largely fluid unresponsive upon PLR [48]. We note that their study employed transthoracic echocardiography as a gold standard, which is subject to human factors [49], including measurement variability and statistically-limited cardiac cycle sample size [16]. Our study assessed the hemodynamic evolution of the PLR across 4-minutes—comprising thousands of cardiac cycles; our findings are in-line with a more recent investigation by Søndergaard where 82% of healthy subjects were preload responders during a 7-minute passive leg raise [27].
There are limitations to our study. First, we studied ambulatory volunteers, so whether this data can be generalized to hospitalized patients and especially those in the intensive care unit is limited. Nevertheless, it is important to qualify normal physiology so as to better understand departures from it during sickness. As well, other investigators have found that changing carotid Doppler measures can be used in critically-ill populations; this was echoed in a recent systematic review [29, 36, 37, 38, 39, 40]. As a second limitation, we had to exclude a small minority of our sample due to poor signal from the wearable Doppler and the CS. It is possible that inherent differences in these excluded subjects may skew the data and limit its generalizability. Third, we did not compare our technique to photoplethysmography during PLR, which is also known to detect preload responsiveness [16]. Changes in the perfusion index from wearable photoplethysmography is, therefore, another technology comparable to the wearable Doppler system described above [16]. A potential advantage of the carotid Doppler is that it could be hardy to signal disruption induced by peripheral vasoconstriction, which has been a problem observed in both photoplethysmography and uncalibrated pulse contour analysis transduced via finger-cuff [31]. Fourth, there was a wide distribution of body habitus in this study. We recruited volunteers taking no cardiovascular medications. The diversity in body habitus allowed for testing the Doppler device on a wide range of neck sizes. While this BMI distribution may limit the generalizability of our results, we note that the pre-post paradigm of the PLR should also hold true in the obese population. Fifth, we did not account for exercise training [50] nor did we account for other potential confounds such as caffeine, herbal supplement or dietary intake in this study. It is unclear how these confounds may affect the results of the PLR in our study population and this is not a hypothesis we set out to test, though the response to PLR is related to exercise capacity [50]. We note that studies of critically-ill patients in whom preload responsiveness is measured also do not account for exercise training or other dietary vasoactives such as caffeine. Sixth, we note that the test-retest reliability between the two passive leg raises was only moderate. This might be expected given that the reliability of multiple, successive PLRs has been questioned [20]. It is possible that emptying and redistribution of venous blood from certain capacitance beds (e.g., splanchnic) on the first PLR alters subsequent maneuvers.
In this study of non-fasted, clinically-euvolemic, ambulatory volunteers, a
clinically-significant % SV
Available upon reasonable request.
JÉSK—conception, primary drafting, analysis, critical revisions; CH—analysis, critical revisions; ME—analysis, critical revisions; AME—conception, analysis, enrollment, critical revisions; JKE—conception, analysis, enrollment, critical revisions. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The procedures followed were in accord with the ethical standards of the committee on human experimentation at our institution. Written and informed consent was obtained for all subjects, and the study was approved by the Research Ethics Board of Health Sciences North (#19-011).
Not applicable.
This research received no external funding.
JESK, CH, ME, AME, JKE work for Flosonics Medical, the start-up building the wearable Doppler ultrasound.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



