Frontiers in Bioscience-Elite (FBE) is published by IMR Press from Volume 13 Issue 2 (2021). Previous articles were published by another publisher on a subscription basis, and they are hosted by IMR Press on imrpress.com as a courtesy and upon agreement with Frontiers in Bioscience.
1 Department of Anatomy, All India Institute of Medical Sciences, New Delhi, India
2 Department of Rheumatology, All India Institute of Medical Sciences, New Delhi, India
Abstract
Rheumatoid arthritis (RA) is one of the most common chronic inflammatory autoimmune diseases, which adversely affects the quality of life. RA is a disease of unknown etiology, however, both genetic and environmental factors appear to contribute to the susceptibility to this disease. The severity and progression of the disease are attributable to the release of a host of inflammatory cytokines, cytotoxic and immune regulatory factors. The treatments of RA are primarily limited to symptomatic alleviation of pain or other symptoms or to the use of cytotoxic drug treatment in severe forms of the disease which is commonly associated with significant side effects. Despite lack of a cure, the disease may be controlled by mind-body interventions. Holistic treatments such as Yoga significantly improve and reduce the psycho-somatic symptoms, pain perception, disability quotient, joint flexibility, range of motion, posture, muscle strength, coordination, and disease activity. Here, we discuss the features of RA and address how Yoga can be used as a therapeutic regimen to improve the quality of life of patients with RA.
Keywords
- Rheumatoid arthritis
- Yoga
- Inflammation
- Oxidative stress
- Immune-ageing
- Immune-dysregulation
- Review
Rheumatoid arthritis (RA) is a complex progressive multisystem autoimmune disease characterized by chronic pain and swelling, affecting primarily the peripheral joints (1). RA is associated with symmetric arthritis characterized by synovial hyperplasia, cartilage damage, and joint deformities, which lead to progressive functional disability, reduced quality of life, early death, and increased socioeconomic burden (2).Various environmental and lifestyle-associated factors are known to induce and perpetuate autoimmunity. In RA, the autoimmunity is linked with an aberrant immune system in which the distinction between foreign and self-antigen is not absolute. The well-established symptomatic disease is preceded by a period of an asymptomatic low physiological level of self-reactivity, which is characterized by the presence of circulating autoantibodies (3). The etiology of RA is still unknown, but various factors predispose an individual to develop the disease. The interaction of multiple factors, including genetic, hormonal, and environmental, leads to the initiation and propagation of autoimmune diseases like RA (Table 1). Also, psychological stress and depression are known to precipitate the state of auto-inflammation and autoimmunity (4). There is a bi-directional association of comorbid depression with the pathophysiology of RA due to dysfunctional neuroendocrine system, systemic inflammation, accelerated immune aging, oxidative DNA damage, and abnormal DNA damage sensing and repair machinery. An asymptomatic pre-clinical stage precedes the establishment of symptomatic RA, which is characterized by synovial inflammation leading to progressive joint erosion (5). RA affects both articular and extra-articular systems, including skin, eye, heart, lung, kidneys, nervous, and gastrointestinal systems, making it a systemic disease (6). RA is associated with joint damage causing progressive disability, systemic complications leading to increased morbidity, mortality, and socioeconomic costs, which further aggravate psychological stress. For the clinical remission of the disease and prevention of associated disabilities, an aggressive therapy targeting the symptomatic relief as well as disease modification is necessary. Soon after the diagnosis of the disease, an early intervention including disease-modifying anti-rheumatic drugs (DMARDs) and biologic disease-modifying therapies sometimes fail to produce the desired response. RA is typically diagnosed when the joint manifestations have begun. The period of the pre-clinical stage lapsed, a time when the window for optimal treatment is likely to be missed. Furthermore, significant proportions of RA patients don’t respond to any given therapeutic agent and hence become prone to multiple disabilities and organ involvement. Hence, the ultimate goal of RA management should be the prevention of inflammatory joint condition and prolongation of remission periods, thereby, prevention of associated depression and disability (7).
| Genetic factors | Susceptibility HLA genes within the MHC region |
|---|---|
| • HLA–DRB1 | |
| Non-HLA genes within the MHC region | |
| • TNF-alpha | |
| Genetic risk factors loci outside the MHC region | |
| • PADI4 |
|
| Immunological factors | • Dysregulation of immune homeostasis |
| Hormonal factors | • Estrogen |
| Environmental factors | • Infections (Bacterial, Viral. Parasitic) |
Studies are citing the positive effects of mind-body interventions improving RA symptoms, patients’ overall well-being and quality of life, reducing their psychological distress and creating a positive mental state, and decreasing their pain intensity (8–13). Yoga is a profound science of a mind-body technique that acts via a well-defined psycho-neuro-endocrine pathway, aims to rejuvenate mind by counteracting the stress response, restores endocrine health and relaxes the body by maintaining flexibility and improving joint mobility. Majority of complex diseases like cardiovascular diseases, depression, cancers, and autoimmune diseases arising due to interaction of several genes and environmental factors and our unhealthy social habits have common underlying etiology like persistent activation of the hypothalamic-pituitary-adrenal axis, oxidative stress (OS), depression, anxiety, stress, shorter telomeres, chronic inflammation, gut microbiome dysbiosis and dysregulated immune system. Yoga targets each of these factors and corrects the dysregulated systems and brings about homeostasis. Hence, it can be used as an adjunct therapy in the management of these disorders. Adoption of a simple lifestyle-based intervention like yoga and meditation may help to break the surge of triggering factors as it reduces inflammatory state by regulating OS induced cellular damage, maintains cortisol melatonin rhythms, elevates endorphin levels and maintains a balance of pro-inflammatory and anti-inflammatory cytokines. It also normalizes the levels of dysregulated transcripts, especially of genes regulating free radical levels and maintaining DNA integrity (14–16). Yoga also aids in reducing the rate of cellular aging by upregulation of telomerase enzyme activity and genes responsible for DNA repair and cell-cycle control. Yoga improves mitochondrial integrity by altering the expression levels of transcripts which maintain mitochondrial integrity (AMPK, IGF1R, PRC-1, TFAM, SIRT-1, TIMP-1, and CLOTHO) which aids in the maintenance of optimal free radical levels, improves energy and insulin metabolism and thus reduces the rate of telomere attrition by supra-physiological ROS levels (9, 10). In a recent study in our laboratory, we found an increase in mitochondrial biogenesis post-yoga intervention.
Combination of DMARDs with yoga intervention and adoption of other healthy lifestyle habits like quitting smoking, increased intake of fruits and vegetables, maintaining optimal weight, improving sleep patterns/ adequate sleep can bring about reversal of altered metabolic, genetic and epigenetic bioprocesses which provide not only amelioration of symptoms but also fewer chances of relapses. Infiltration of this ancient practice, yoga, in the era of western medicine and growing patients’ demands for an inexpensive, natural and safe alternative therapy has paved the road for relieving the stressors of this busy life and ensuring overall health and wellness. Yoga is one of the commonest integrative medicine modalities practiced worldwide. Yoga is a scientific technique for achieving optimal physical and mental well-being by increasing joint range of motion, flexibility, strength, coordination, posture, and improving the quality of life of patients as well as the caregivers (10, 15, 17–19).Previous studies have shown that yoga may improve wellness in healthy people and have benefits for patients, especially with chronic diseases and improve their quality of life and coping mechanism (15, 20–22). Psychological and health-restorative benefits of yoga and meditation have been investigated, but their impact on the immune system and the associated molecular mechanism remains poorly defined. A meta-analysis on the effect of mind-body therapies on the immune system concluded that yoga has anti-inflammatory and immunomodulatory effects (23).The recent growth trends and keen diversion towards mind-body therapies, including yoga-based lifestyle intervention (YBLI), mindfulness-based stress reduction (MBSR), Tai-chi, Qigong, etc. have emphasized the need to discover the molecular mechanisms behind their therapeutic actions. Short term yoga and related practices result in rapid global gene expression profile changes in the peripheral blood mononuclear cells, which may be the basis for their long term cell biological and higher-level health effects (24). Recent studies from our laboratory on various complex lifestyle diseases like unexplained male factor infertility, idiopathic recurrent pregnancy losses, depression and glaucoma found that YBLI may have important roles in slowing the rate of cellular aging and improving cellular longevity thus promoting health and lifespan (8, 14, 15, 18, 25).Studies from our laboratory on unexplained infertility suggested that simple lifestyle interventions like yoga and meditation can reduce testicular inflammation by optimizing OS markers, thereby reducing oxidative DNA damage and therefore reduce the incidence of infertility and future consequences in the next generations. It is due to the impact of yoga on the sperm genome and epigenome (16, 26, 27). Our studies also suggested an increase in levels of beta-endorphins, brain-derived neurotrophic factor (BDNF), dehydroepiandrosterone (DHEA), melatonin and sirtuin-1 (SIRT-1) and decrease in levels of cortisol and Interleukin (IL)-6 indicating an improvement in stress and inflammatory response after YBLI with regulatory changes in the brain through hypothalamic-pituitary-adrenal (HPA) axis in patients with major depressive disorder (9, 10). It was the first study to show that yoga increases levels of sirtuins independent of intake of resveratrol or caloric restriction. A recent study from our laboratory on active RA patients has shown to reduce pain perception, functional disability, and disease severity, followed by 8-weeks of YBLI (8). This study also documented a post-yoga upregulation in levels of an immune-modulatory molecule, soluble human leukocyte antigen - G (sHLA-G), and a decrease in levels of IL-6 (extra-articular symptoms) and IL-17A and tumor necrosis factor (TNF)-alpha (articular symptoms) (8). Thus yoga may aid in establishing immunological tolerance and molecular remission and hence can be beneficial as an adjunct therapy in the management of this severe chronic progressive autoimmune arthritis. These studies from our laboratory served as the evidence base, which proved the beneficial and therapeutic roles of YBLI in healthy and diseased individuals; further insights may help us provide the exact underlying mechanism of action.
RA is a most common heterogeneous systemic inflammatory autoimmune disease characterized by chronic joint inflammation leading to cartilage destruction, bony erosions, joint damage, and eventually, deformity. The concept of horror autotoxicus was first proposed by Paul Ehrlich (1854-1915), a German bacteriologist and immunologist, to describe the body's innate aversion to mount an immunological response against self (28). Today, we know that autoimmunity results from the breakdown of maintaining tolerance to self-antigens, which leads to immune-mediated destruction to own cells and tissues. More than 80 autoimmune diseases have been identified, including RA, which is one of the leading causes of death worldwide. The first recognized description of RA was made in 1800 by Dr. Augustin Jacob Landré-Beauvais (1772-1840), who was a French resident physician at Salpêtrière Hospital. The term "Rheumatoid arthritis" was coined by British rheumatologist Dr. Alfred Baring Garrod in 1859. Later in 1957, Charles Short described RA as a distinct clinical entity, setting it apart from many other spondyloarthropathies (29). Symmetrical inflammatory polyarthritis is the primary clinical manifestation. The inflamed synovium lining the joint extends and then erodes the articular cartilage and bone, causing joint deformity and progressive physical disability. Additional signs and symptoms which accompany may include chronic fatigue, depression, loss of appetite, malaise, low-grade fever, weight loss, and anemia etc. (30). The disease course includes episodes of symptoms/flares (relapses) followed by periods with no symptoms (remissions). In severely affected cases, continuous health problems may precipitate for many years. The abnormal inflammation can lead to severe joint damage along with a variety of systemic manifestations, which limits movement and can cause significant disability affecting the quality of life. Lack of early diagnosis and non-specific therapeutics are substantial challenges in the management of RA (31). Serum auto-antibodies like Rheumatoid factor (RF) and antibodies to citrullinated protein antigens (ACPAs) are now recognized to be a valuable biomarker of diagnostic and prognostic significance (32). Advances in radiographic investigations like ultrasound and magnetic resonance imaging have improved the ability to detect joint inflammation and destruction in RA. The science of RA has taken a significant leap forward with the identification of new disease-related genes and further deciphering of the molecular pathways of disease pathogenesis. The relative importance of these different molecular mechanisms has been highlighted by the effects of modern, highly targeted biologic therapies. Despite extensive research, the incomplete understanding of triggering and initiation of pathogenic pathways of RA remains a barrier to its cure and prevention.
Around 0.5-1.0% of the adult population is affected by RA worldwide, which accounts for around 250,000 hospitalizations and 9 million doctor visits per year (33). According to world health organization (WHO), RA is more prevalent in developed countries and female sex, and around 50% of patients in developed countries are unable to carry a full-time job within ten years of disease onset (34). Not only the prevalence of the disease, which differs among different continents, races, ages, and socioeconomic levels but also the disease pattern. The overall incidence of RA has been decreasing in recent decades. But, the prevalence has remained the same because individuals with RA are living longer. RA is associated with a higher mortality rate over other inflammatory disorders, especially autoimmune inflammatory disorders. There is a strong association of rheumatic diseases (RA, systemic lupus erythematosus, systemic sclerosis, vasculitis, or spondyloarthritis) with the increased risk of death (35). According to Indian council of medical research (ICMR) 2012, the prevalence rate of RA in India accounts for 0.55% and ranges from 0.49% to 0.71%. The prevalence of musculoskeletal diseases in Delhi is 7.08%, of which 0.17% is contributed by RA (36).
RA falls under the category of inflammatory rheumatic disease, which presents articular and extra-articular manifestations resulting in pain, disability, and mortality (37). The clinical picture of RA among patients varies concerning the type, number, and the pattern of joint involvement. Various endophenotypes of RA exist according to the presence or absence of several variables, including genetic background, environmental exposures, and epigenetic insults. The presenting features of early RA are challenging to distinguish from other inflammatory arthritides. Hence patients with early RA are categorized under the umbrella term “undifferentiated arthritis” before the definitive diagnosis (38). The presence of auto-antibodies characterizes the asymptomatic phase of RA precedes much before the development of an established RA.
The clinical manifestations of symmetrical joint involvement include pain, swelling, redness, tenderness to palpitation, morning stiffness, and severe motion restriction in the involved joints. The clinical presentation of RA varies, the most frequent finding is an insidious onset of pain with swollen small joints of hand and feet, including metacarpophalangeal, proximal interphalangeal, and metatarsophalangeal joints, as well as in the wrists and knee. The patient’s disease activity as measured by Disease Activity Score 28 (DAS28) which is based on four factors: (i) number of tender joints (0-28), i.e., tender joint count (TJC), (ii) number of swollen joints (0-28), i.e., swollen joint count (SJC), (iii) patient’s assessment of global disease activity by visual analog scale (VAS) and (iv) acute phase reactant, i.e., either erythrocyte sedimentation rate (ESR) or C- reactive protein (CRP) (39). The duration of early morning stiffness is also related to disease activity. The typical manifestations of RA include polyarthritis, symmetric arthritis, hand arthritis, and early morning stiffness. RA may also show atypical presentations like polymyalgic onset (usually late onset RA with acute stiffness around the shoulder and pelvic girdle), palindromic onset (recurrent episodes of joint inflammation lasting for a day may be experienced), systemic-onset (disease onset occurs with extra-articular manifestations like fatigue, depression, fever, vasculitis, etc. followed by arthritis at a later stage) and persistent monoarthritis (disease onset with a single joint involvement like knee, ankle, wrist or shoulder). Progressive pain, swelling, and loss of function lead to soft tissue damage and joint destruction, which may result in irreversible deformities (40). The most common deformities include ulnar deviation, swan-neck deformity, boutonnière deformity, and Z-line deformity, etc. (41).
Extra-articular manifestations of RA occur in about 40% of patients. These manifestations become evident either in the beginning or during the disease course (42). The extra-articular organ system includes skin, gastrointestinal, cardiac, renal, nervous, and pulmonary involvement (Table 2). Patients with high titers of RF, i.e., autoantibodies to the Fc component of immunoglobulin G, are most likely to have extra-articular manifestations of their disease, including rheumatoid nodules, rheumatoid vasculitis, and pleura-pulmonary, neurologic, digestive, cardiovascular, cutaneous, hematologic, and ocular complications (43).
| Sites of involvement | Signs and symptoms |
|---|---|
| Physical involvement | |
| Skin | Rheumatoid nodules, Livedo reticularis, Atopic dermatitis, Pyoderma gangrenosum, Purpura |
| Eye | Keratoconjunctivitis sicca (Secondary Sjögren's syndrome), Episcleritis, Scleritis, Peripheral ulcerative keratitis |
| Oral cavity | Periodontitis |
| Gastro-intestinal tract | Mesenteric vasculitis, Liver cirrhosis |
| Respiratory tract | Asthma, Pleural effusions, pulmonary nodules, interstitial lung disease, pulmonary vasculitis, associated pneumonia |
| Cardiovascular system | Hypertension, Pericarditis, Ischemic heart disease, myocarditis, cardiomyopathy, congestive heart failure, endocarditis, Arterial stiffness |
| Urinary tract | Chronic Kidney Disease, mesangial glomerulonephritis, secondary amyloidosis |
| Nervous system | Cervical myelopathy, peripheral neuropathy |
| Blood and bone marrow | Anemia, neutropenia, thrombocytopenia, thrombocytosis, eosinophilia, splenomegaly, hematological malignancies |
| Skeletal system | Osteoporosis |
| Endocrine system | Hypoandrogenism, Hypothyroidism |
| Psychological involvement | |
| Emotional symptoms | Depression, Anxiety |
The pathogenic mechanisms of synovial inflammation are likely to result from a complex interplay of genetic and environmental factors that produce dysregulation of the immune system and a breakdown in self-tolerance. A schematic diagram shows the progression of the disease from triggering to targeting (Figure 1). The contribution of genetic factors in susceptibility to RA is around 60% with the human leukocyte antigen (HLA) locus accounting for at least 30% of overall genetic risk (Table 3) (44). Non-HLA genes lying within the major histocompatibility complex (MHC) region, like TNF-alpha, have also been investigated for association with RA (45). A considerable number of confirmed associations with RA and other autoimmune disease susceptibility loci including peptidyl arginine deiminase type 4 (PADI4), protein tyrosine phosphatase non-receptor type 22 (PTPN22), signal transducer and activator of transcription (STAT4), cluster of differentiation 244 (CD244) and cytotoxic T lymphocyte-associated antigen 4 (CTLA4), located outside the MHC have been reported recently (45–48). Autoimmunity and systemic inflammation may be triggered as a result of environmental stimuli acting together with the genotype at mucosal surfaces like lungs, oral and gastrointestinal mucosa years before the onset of joint symptoms. The presence of autoantibodies indicates ACPAs and RF, which can be present in the blood long before the first signs of joint inflammation (29, 32). The maturation of this autoimmune response results in elevated titers of autoantibodies. Epitope spreading occurs with the development of an immune response to endogenous epitopes secondary to the release of self-antigens in the regional lymph nodes and other peripheral lymphoid organs (secondary sites of immune response after mucosal surfaces). Various possibilities drive the transition of an autoimmune and inflammatory reaction at a mucosal site followed by an invasion of lymph nodes, then to attack multiple organs and synovial joints to develop into visible rheumatoid ‘arthritis.’ The development of a novel biomarker to detect the silent predisposition of healthy individuals into deforming and depressing RA is the need of the hour. Interactions between the T cell receptor (TCR) and MHC class II-peptide antigen with co-stimulation through CD28-CD80/86 pathway leads to the activation of CD4+ T cells via antigen-presenting cells (APCs). In early RA, a range of inflammatory processes may be responsible for the generation of immune responses against specific proteins that have undergone particular forms of post-translational modifications (49). Smoking is linked with the increased expression of peptidyl arginine deiminase (PAD) isoform 2 in respiratory mucosa, which triggers the conversion of arginine into citrulline (32). Local hyper-citrullination thus provides a source of autoantigens driving the development of ACPAs. Another cause for citrullinated autoantigens in the formation of neutrophil extracellular traps (NET) which contain citrullinated proteins, e.g., keratins, fibrinogen, actin, fillagrins, vimentin, histone H2A, H3, and H4, etc., due to the action of different isoforms of PAD enzyme (50). ACPAs can target the products of NETosis; also, ACPAs can stimulate NET formation, hence may contribute to the induction and expansion of autoimmunity in a positive feedback loop (51). The binding of ACPAs to osteoclasts induces the production of the chemokine CXCL8, which functions as an autocrine growth factor for osteoclasts. CXCL8 also can bind to its receptors CXCR1 and CXCR2 on nociceptive nerves and thereby cause pain. The circulating immune complexes containing citrullinated fibrinogen activate macrophages through the synergistic engagement of toll-like receptor 4 and Fc receptors and possibly can enter the joint and deposit on cartilage and in synovial tissue (32). Synovial CD4+ T cells differentiate into T helper 1 (Th1) and T helper 17 (Th17) cells and secrete pro-inflammatory mediators like TNF-alpha, IL‑6, and matrix metalloproteinases. TNF-alpha is a major cytokine in the pathogenesis of RA as it upregulates adhesion molecule on endothelial cells, promoting polymorphonuclear leukocyte influx into the joint, stimulates the production of other inflammatory mediators, such as IL-1, IL-6, and granulocyte-macrophage colony-stimulating factor (GM-CSF). The disproportion between pro- and anti-inflammatory cytokine levels favors the formation of autoimmune complexes, chronic inflammation, and, thereby, joint damage (1, 52).
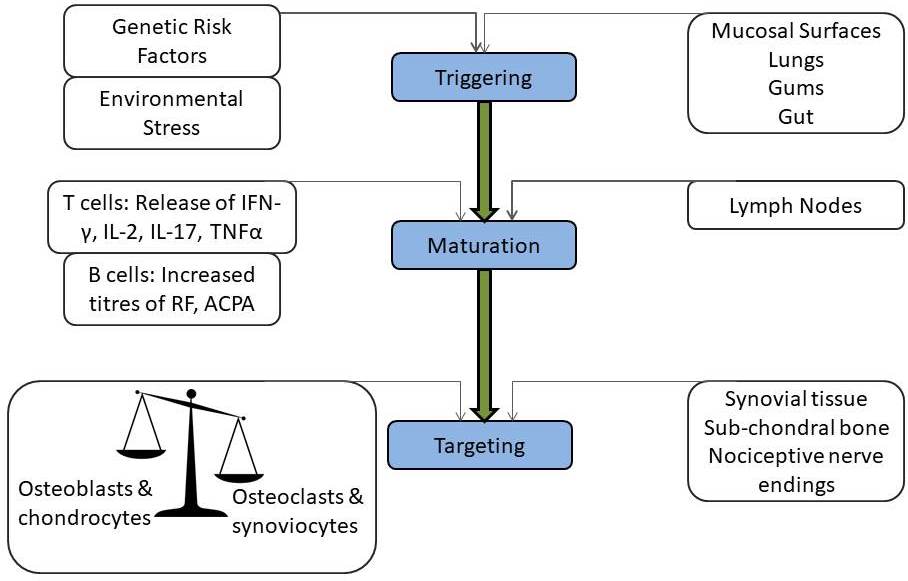 Figure 1
Figure 1Stages of RA: From triggering to targeting.
| Gene | Chromosomal location | Function | Variations observed | References |
|---|---|---|---|---|
| HLA-DRB1 | 6p21.3 | Encodes cell surface antigens that present proteins to the T lymphocytes | rs660895 | (46, 165) |
| TNF-alpha | 6p21.3 | Plays a central role in the inflammatory cascade in affected joints and the striking efficacy of TNF-alpha antagonists as therapeutic agents | rs1800629; |
(47, 166) |
| PADI4 | 1p36.13 | Encodes enzymes responsible for the conversion of arginine residues to citrulline residues. This gene may play a role in granulocyte and macrophage development leading to inflammation and immune response | rs1748033 | (45, 46, 48, 167) |
| PTPN22 | 1p13.2 | Encodes a protein tyrosine phosphatase, which is expressed primarily in lymphoid tissues. This enzyme is involved in several signaling pathways associated with the immune response | rs2476601 | (44–46, 168, 169) |
| STAT4 | 2q32.2–q32.3 | Provides instructions for a protein that acts as a transcription factor, which means that it attaches (binds) to specific regions of DNA and helps control the activity of certain genes. Encodes a transcription factor for signals from certain cytokines | rs7574865 | (46, 48, 170) |
| CTLA4 | 2q33 |
Member of the immunoglobulin superfamily and encodes a protein which transmits an inhibitory signal to T-cells | A49G polymorphism | (46, 48) |
| CD244 | 1q23.3 | One of the molecules that activates or inhibits natural killer cells have indicated that they play critical roles in the immune system and in autoimmune diseases | rs3766379 rs6682654 | (45, 46, 48) |
| TRAF1-C5 | 9q33–34 | Involved in signaling pathways that play a role in cell proliferation and differentiation, apoptosis, bone remodeling and activation or inhibition of cytokines | rs10818488 | (46, 48) |
| TNFAIP3 | 6q23 | Encodes a cytoplasmatic zinc-finger protein that inhibits NFKB activation and TNF-mediated apoptosis | rs2230926 | (48, 166) |
| SPRED2 | 2p14 | Involved in regulating CD45 positive cells via the Ras-MAP kinase pathway | - | (46) |
Various other environmental risk factors like diet, infections, obesity, drugs, hormone supplements, and exposure to tobacco smoke are associated with an increased risk for RA (53–55). Though the prevalence of the disease is irrespective of age and sex, clinical studies have shown more chances of occurrence in aging subjects and females. Various theories have proposed the possible role of estrogen in disease pathogenesis, and most of the theories center on the role of estrogens and X-linked genetic factors in enhancing the immune response (56). Some experimental studies have shown that estrogen can stimulate the production of TNF-alpha, a major cytokine in the pathogenesis of RA. Perhaps drastic changes in the immune system after menopause in females and old age in males might be the reason. The high pollution levels, particulate matter, occupational dust, lower temperature influence rheumatic pains and worsen the RA symptoms significantly among middle-aged patients (57, 58). High dietary salt intake has been linked as a risk factor for the development of autoimmune diseases by the polarization of T cells to pathogenic Th17 cells via upregulation of serum/glucocorticoid-regulated kinase-1 activity (59). The association of tobacco smoke has been studied extensively and found strongly associated with seropositive RA in genetically predisposed individuals (32, 58).
The era of super-specialization and fast-paced life has led to the discovery of new technologies, industries, inventions, chemicals, drugs, foods and dietary habits, and a sedentary disease prone lifestyle. Previous investigations have found that a regular regimented vegetarian diet after a period of fasting shows a decrease in disease activity (53). In contrast, increased red meat and protein intake were associated with an increased risk of developing RA (60). However, a subsequent study showed no association between the amount of protein, red meat, poultry, and fish consumption and modification of RA risk (61). Studies suggest that the use of Vitamin D and alcohol may decrease the risk of RA. Amelioration of RA is observed during pregnancy with exacerbation in the postpartum period. All these factors together lead to a dysregulated immune homeostasis and hence affect the body’s delicate immune recognition system leading to triggering auto-immune diseases in pre-disposed individuals. The triggers of immune-dysregulation and autoimmune reactivity are essential for a better understanding of disease pathogenesis and the development of new therapies (62). The environmental stimulus may program the immune system in such a way that results in epigenetic modifications, acceleration of posttranslational modification of proteins, which can cause the emergence of arthritis inducing immunoreactivity (58).
The full collaboration of both the innate and adaptive arms of the immune system plays a crucial role in the promotion or inhibition of autoimmune disease. Generally, to clear infections, the innate immune cells can upregulate costimulatory molecules and produce a mixture of pro and anti-inflammatory cytokines such as IL-1 beta, IL-12, transforming growth factor (TGF)-beta, IL-23, TNF-alpha, and IL-6 that regulate the adaptive arm of the immune system. As in RA patients, there is altered homeostasis between regulatory T cells (Tregs) and effector Th17 cells functions (Figure 2). Dysfunction of Tregs fails to maintain peripheral tolerance and results in autoimmunity (63, 64). Tregs secrete anti-inflammatory cytokines like TGF-beta, IL-10, IL-35, and also sHLA-G molecule. The altered immune dysregulation in RA is attributed to increased secretion of inflammatory cytokines by effector Th17 cells and a loss of Tregs suppressor function. Tregs by secreting immune-regulatory cytokines prevent aberrant immune response and prevent autoimmunity. In RA, Tregs also show immune-plasticity by producing IL-17 pro-inflammatory cytokine but manage to retain their suppressive functions resulting in milder disease forms (65). An immune-regulatory and anti-inflammatory molecule, sHLA-G, is secreted by cells of the immune system, such as monocytes, T cells, especially Treg cells and dendritic cells. There are various suppressive mechanisms of Tregs which form a basis of the immune-surveillance system and utilized for Treg cells based therapeutic adventures to reach complete clinical remission.
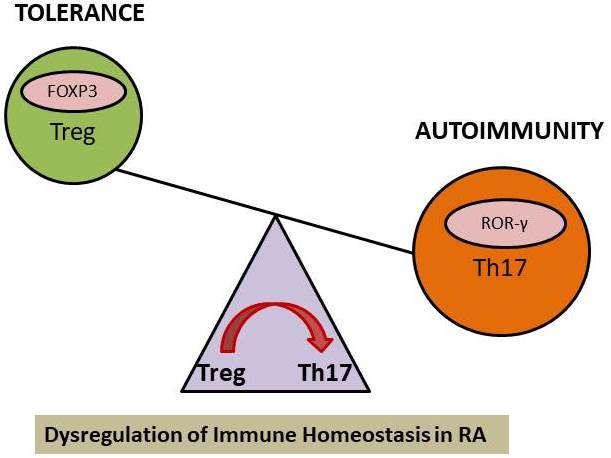 Figure 2
Figure 2Immune dysregulation in RA: Inefficient/Loss of suppressor mechanism of Tregs.
The innate immune system is the first line of defense against microbial pathogens (bacteria, viruses, fungi, and parasites). There is a release of inflammatory cytokines when the foreign antigen is encountered by macrophages and dendritic cells via membrane-bound or intracellular pattern recognition receptors. When the sentinels of innate immunity fail to overcome the pathogenic antigen alone, then an adaptive response is initiated in lymphoid organs. Then, the processed antigens are presented by MHC molecules to naïve T-cells, thus starting an adaptive immune response with lasting immunological memory. In RA, there is a regular expression of macrophage-derived cytokines such as TNF-alpha, IL-1, and IL-6, which shows the evidence of innate immune system activation (66). The effector cells of innate immunity are found in the rheumatoid synovium, and an increasing body of evidence indicates that they are directly involved in joint inflammation and the destruction of the joint cartilage and bone (67).
Adaptive immunity involves B and T cells mediated specific immune response characterized by the recognition of foreign antigens and the development of immunological memory. B cells play an important role in regulating immune responses, and dysregulated behavior of B cells leads to aberrant immune response and contribute to the development of autoimmune diseases (68). B cells are precursors of autoantibodies-secreting plasma cells, which give rise to specific autoantibodies like RF, ACPA, anti-keratin, anti-perinuclear factor providing evidence for humoral disturbances in RA. Autoantibodies form immune complexes activating antigen-presenting cells like B cells, macrophages, and follicular dendritic cells, thus enhancing local inflammatory processes by the release of TNF-alpha, IL-6, etc. (69). The presence of self-antigens like carbamylated or citrullinated antigens leads to the activation and differentiation of naïve T cells into effector CD8+ (cytotoxic T cells) and CD4+ cells (helper T cells) including Th1, Th2, Th17 and Tregs (70). Upon stimulation, the classical T cell subsets give rise to their respective cytokines like Th1 cells (CD3+CD4+T-bet+) secrete interferon (IFN)-gamma and IL-12, Th2 cells (CD3+CD4+Gata-3+) secrete IL-4, Th17 cells (CD3+, CD4+, CD8-, IL-17A+, ROR gamma t+) secrete IL-17 and T reg cells (CD3+, CD4+, CD25+, Foxp3+) secrete TGF-beta, IL-10 etc. All these cell types, inflammatory cytokines, and chemokine receptors are involved in the development of RA and infiltrate the synovium and are responsible for creating an inflammatory environment (52).
Stress, both physical and psychological, can be detrimental to immune health. Various unknown trigger factors have implications in the development of autoimmune diseases (71). Stress triggers the release of neuroendocrine hormones like epinephrine, norepinephrine, acetylcholine, substance P, vasoactive intestinal peptide, glucagon, insulin, cytokines, growth factors, and numerous other mediators may lead to aberrant immune regulation, decreased host defense resulting in autoimmune disease. The activation of the stress response, release of stress-related hormones, cytokine imbalance, influences the close relationship between the HPA axis, sympathetic nervous system (SNS), and immune system. Cortisol functions to reduce inflammation in the body, but over time elevated cortisol levels as a result of chronic stress may lead to the suppression of the immune system (72). The active RA possess typical symptoms like joint swelling, stiffness, and pain show a diurnal variation which coincides with the circadian difference in plasma concentrations of cortisol, melatonin, and pro-inflammatory cytokines (73, 74). There is a late-night load of pro-inflammatory cytokines (TNF-alpha, IL-6) and hormones (melatonin, prolactin), whereas reduction in endogenous anti-inflammatory hormone (cortisol) may explain nocturnal inflammation. Chronobiology has a prominent function in RA (73). Melatonin is a pro-inflammatory hormone and a potent antioxidant with the highest subcellular concentration in mitochondria released 2 hours earlier in RA patients as compared to healthy controls; it has higher plasma levels in RA than healthy controls. It regulates circadian cytokine production, stimulating the production of IL-1, IL-2, IFN-gamma, and IL-12 in lymphocytes, as well as the production of IL-6 in monocytes (75). Cortisol and melatonin play a significant role in the stress pathway. A study on the effects of Hatha yoga and Omkar meditation suggested that an increase in endogenous secretion of melatonin might be responsible for an improved sense of well-being (76). Also, diaphragmatic breathing induces relaxation via a reduction in the production of cortisol, elevation in melatonin levels, correlated with lowering of OS (77). Monteleone et al. found that there exists a relationship between cortisol and melatonin rhythms as an exercise-induced increase in plasma cortisol precedes night’s lowering of melatonin secretion (78). But how switching on the relaxed mode in the brain by yoga and meditation practice might induce an increase in melatonin levels remains elusive, or whether this is simply due to a decrease in cortisol remains to be clarified.
OS plays a vital role in the pathogenesis of RA due to polymorphonuclear leukocyte and lymphocyte production of reactive oxygen species (ROS) affecting cell-signaling events (79). OS results in free radical-mediated damage to bio-membranes, especially lipid bilayer and essential cellular components. OS disrupts the plasma membrane by lipid peroxidation, which generates a variety of reactive electrophiles like malondialdehydes (MDA), propanal, hexanal,4-hydroxy-2-non-enal (4-HNE), isoprostane, etc. The most toxic product of lipid peroxidation is 4-HNE which plays an essential role as a signaling molecule stimulating gene expression with protective functions that can enhance cellular antioxidant capacity (80). Cells can survive when 4-HNE levels are low, but there is damage to organelle and protein, leading to the induction of autophagy, senescence, and cell cycle arrest when 4-HNE levels increase. At high levels of 4-HNE, there is the induction of apoptosis/necrosis programmed cell death, which eventually leads to molecular cell damage (81). Such mutagenic and toxic secondary lipid peroxidation products can also react with proteins and DNA to form adducts resulting in a variety of cytotoxic and genotoxic consequences. Severe OS results in DNA damage, both single-strand and double-strand breaks and nucleotide modifications in the DNA. The oxidizing capacity of ROS results in the accumulation of mutagenic base, 8-hydroxy-2’-deoxyguanosine (8-OHdG), and also causes dysregulation in levels of mRNAs/transcripts (8, 26). These highly reactive molecules can be counteracted by a continuously available system of antioxidants and replenished externally by dietary means. ROS are also generated via an inflammatory response following a tissue injury or damage. In the synovial joint, neutrophils release myeloperoxidase, which catalyzes the formation of hypochlorous acid, a potent oxidant, disrupts protein tertiary structure and activity. Neutrophil-derived ROS can also influence immune responses by activating signaling pathways like the Nuclear factor kappa-light-chain-enhancer of activated B cells (NFKB) and the extracellular-signal-regulated kinase (ERK) pathway. ROS controls T cell activation, modulates T cell receptor signaling pathways, suppresses T cell proliferation, an inducer of apoptosis, and downregulates their responsiveness. In RA synovial joint, ROS act as inflammatory mediators and contribute to tissue destruction by the degradation of collagen, apoptosis of chondrocytes, breakdown of the extracellular matrix, loss of self-repair mechanisms (82).
Immunosenescence is a natural process that leads to gradual deterioration of the immune system by loss of protective abilities and gain of pro-inflammatory functions. In RA, the rate of immunosenescence is accelerated (5, 83). T cells become susceptible to aging as there is enormous proliferative stress due to antigenic exposure, homeostatic proliferation due to thymic involution and their long lifespan as they serve as the carriers of immune memory. The proliferation of peripheral T-cells and telomere attrition reaches the “Hayflick limit” after which cell division stops and a senescent associated secretory phenotype (SASP) is adopted (84). Senescent cells release SASP proteins, which induce tissue inflammation by its action on microvessels, osteoclasts, fibroblasts, vascular smooth muscle cells, neutrophils, dendritic cells, and naïve T cells. Molecular mechanisms associated with T-cell aging in RA include leakiness of DNA repair systems like ATM (ataxia-telangiectasia mutated) and MRN complex (Meiotic recombination 11 homolog A (MRE11A), RAD50, Nijmegen breakage syndrome 1 (NBS1) and inefficiency of telomeric maintenance and repair which leads to accumulation of damaged DNA causing proliferative arrest leading towards apoptosis (85). Deficient activity of the double-strand break repair protein MRE11A in patients with RA promotes T-cell aging and destructive inflammation in synovial tissue (86). Hence, a cascade of cellular events gets initiated due to environmental factors like poor lifestyle, smoking, high pollution levels in genetically predisposed individuals precipitating psychological stress and which may cause OS induced accelerated immune aging or vice-versa (Figure 3). The inefficient DNA damage sensing and repair machinery of RA T cells are linked to premature T cell aging and arthritogenic effector functions (87). The peculiar features of prematurely aged RA T cells include a loss of CD28 expression, shrinking naïve and expanding memory repertoire, shortened telomeres, a gain of cytotoxicity, DNA damage accumulation, lack of an irreversible cell cycle arrest, more vulnerability to apoptosis, altered tissue trafficking, excess production of pro-inflammatory cytokines, etc (86). Abnormalities in the MRN complex, composed of MRE11A, RAD50, and NBS1, fails to sense DNA double-strand breaks to amplify DNA repair (86–88). In RA T cells, the accelerated telomere attrition is attributed to an increased proliferative frequency of naive T cells in RA and a reduced human telomerase reverse transcriptase expression in T cells, which leads to failure of telomerase activity after TCR stimulation. Telomerase insufficiency is reported in T cells of RA patients, which accelerate telomere shortening (83). Also, telomeric shortening could be a consequence of deficits in the DNA repair system. A report by Li et al. states that reduced activity of MRE11A, a double-strand break repair nuclease, induces cellular senescence by promoting telomere damage and upregulation of cellular senescence markers, such as p16, p21, and the unraveling of heterochromatin (86).
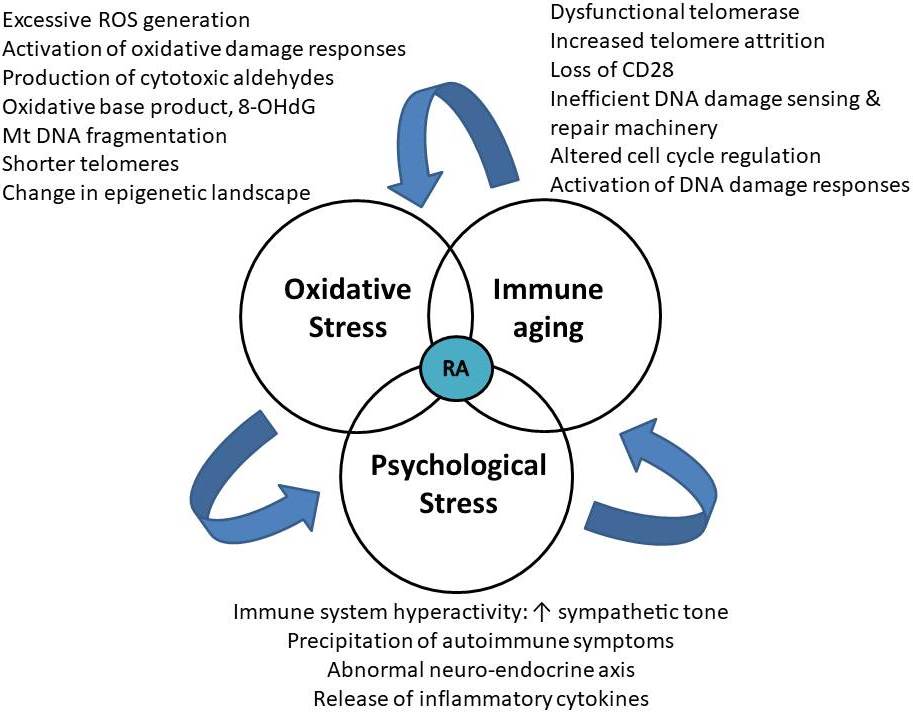 Figure 3
Figure 3Bermuda triangle of OS, Psychological stress and immune aging in RA.
The diagnosis of RA is based mainly on clinical signs and symptoms of chronic inflammatory arthritis, with laboratory and radiographic results providing crucial supplemental information. In 2010, a collaborative effort between the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) revised the 1987 ACR classification criteria for RA to improve early diagnosis to identify patients who would benefit from early introduction of disease-modifying therapy (89). Application of the newly revised standards yields a score of 0–10, with a score of ≥6 fulfilling the requirements for definite RA. The goals of RA management are to control pain and swelling, delay disease progression, prolong remissions, minimize disability, and improve quality of life. The absence of new appropriate management of RA can lead to severe joint damage and disability (37). Initiation of treatment, particularly by a combination of DMARDs concurrent with short duration of corticosteroid, is expected to prevent progressive course and even change the natural course of RA (90). DMARDs are so named because of their ability to slow or prevent structural progression of RA. The conventional DMARDs include hydroxychloroquine, sulfasalazine, methotrexate, and leflunomide; they exhibit a delayed onset of action of approximately 6–12 weeks. Methotrexate is the DMARD of choice for the treatment of RA and is the anchor drug for most combination therapies (91). Biologic response modifiers are therapeutics designed mostly to target cytokines and cell-surface molecules. TNF inhibitors, Anakinra, Abatacept, rituximab, and tocilizumab are few drugs of this class (92). The successful management of RA needs a combination of prescription medications with exercises and physical activity. Dynamic strength training, community-based comprehensive physical therapy, and physical-activity coaching have all been shown to improve muscle strength and perceived health status. Surgical approaches are best suited for the correction of deformities like synovectomy, tenosynovectomy, tendon realignment, reconstructive surgery or arthroplasty, and arthrodesis. Hence, the new approach towards the management of RA focuses on the following goals: early, aggressive therapy to prevent joint damage and disability; frequent modification of treatment with the utilization of combination therapy where appropriate; individualization of treatment in an attempt to maximize response and minimize side effects; achieving, whenever possible, remission of clinical disease activity. A considerable amount of evidence supports this intensive treatment approach (93).
The prevention of RA has become a global challenge despite significant research to predict the trajectory of RA development right from the discovery of genetic markers and triggering risk factors. If managed at an early stage, RA progression and course can be modified by prolonging periods of remission and minimizing frequent relapses (94). Some risk factors like female sex and advancing age can’t be changed, but certain triggering factors like tobacco smoking, limited exposure to pollutants, and maintaining adequate weight can be avoided. Implementation of prevention strategies would certainly be beneficial at the patient, caregiver, and health care system level (3). Apart from the usage of routine drug therapy, the adoption of YBLI has proved to be beneficial for active RA patients at health, economic, and social level (8). The interaction of genetic and environmental factors triggers the initiation of RA. Among the genetic risk factors, HLA-G polymorphism regulates the expression of an immunomodulatory molecule, HLA-G, which plays a vital role in disease pathogenesis, course, and response to treatment. A recent study from our lab documented that HLA-G +3142G>C polymorphism (rs1063320) might decrease the risk of occurrence of RA in the North-Indian population. The sHLA-G levels were found to be significantly lower in +3142GG and +3142GC RA patients as compared to healthy controls, which could favor the triggering and worsening of RA (95). The practice of YBLI may upregulate sHLA-G levels in RA patients with +3142GG and +3142GC genotypes who are low sHLA-G producers and thus delay the onset or may even prevent the beginning of this disease or experience mild illness.
The interest for mind-body interventions is rapidly growing intending to integrate physical, mental, and spiritual dimensions by focusing interactions among brain, mind, body, and behavior. Various studies have documented the association of stress with immunological defects, especially auto-immune diseases via activation of the HPA axis and the sympathetic–adrenal medullary axis (96). RA affects the psychological health of an individual resulting in a life of dissatisfaction, psychological distress, and poor quality of life with more worsening of RA outcome. This co-existence of depression, along with RA symptoms, poses a significant healthcare burden at an individual, care-provider, and social level. High disease activity is associated with elevated depression scores in RA patients, which hampers their quality of life and also the treatment response rate (4). Multiple organ system involvements, along with numerous extra-articular symptoms, may also occur, slowly progressing towards physical disability and psychiatric comorbidity (6). A study from our laboratory documented that yoga intervention can significantly alter the biomarkers, which are associated with neuroplasticity, systemic inflammation, cellular health, and aging in RA patients, which can substantially reduce the disease activity and severity (8). The science of psychoneuroimmunology has paved the way for the establishment and utilization of these therapies as an adjunct. Various studies have evaluated the impact of brain-body interactions concerning clinical symptoms, psychological assessment, and clinical health outcomes (8–10, 18). Further progress will arise from studies for the exploration of the mechanism of action of these therapies at a molecular level.
Yoga, an Indian origin mind-body intervention, is an ancient science dating back over several thousand years. It aims to bring positivity by promoting optimal physical and mental health and alleviation of the suffering (97). This contemplative practice is being used by people all over the world for disease prevention, maintenance of health, or peace of mind. The literal meaning of the word “yoga” means “to yoke” or “to unite” (98). According to Maharishi Patanjali, rightly called “the father of yoga,” yoga is defined as the discipline to develop one's inherent power in a balanced manner and suppression of modifications of one’s mind. The science and technology of yoga and meditation has gained worldwide awakening in the last few decades, and now 21st June, day of the summer solstice, is celebrated as International Day of Yoga since its inception in United Nations General Assembly.
The historical development of yoga can be divided into three periods, namely: Pre-Patanjali period (Before 500 BC), Patanjali period (500 BC to 800 AC), and Post Patanjali period (800 AC onward). Maharishi Patanjali gave the systematic eightfold path of yoga popularly known as “Ashtanga Yoga” via which one can attain enlightenment (Figure 4) (97, 98). The practice of yoga includes maintenance of physical postures (Asanas) associated with synchronous deep breathing exercises (Pranayama) followed by meditation (Dhyana), which aids in increasing blood flow to the muscles, hence improves tissue oxygenation. It develops muscle strength, provides balance and equilibrium, and maintains joint mobility by keeping the muscles in isometric contraction (98, 99). Yoga has the potential to prevent psychosomatic disorders and provides the ability to endure stressful situations. The Ministry of Ayurveda, Yoga and Naturopathy, Unani, Siddha, and Homoeopathy (AYUSH) lists the salient features of yoga as a universal practical discipline, evolutionary, and act as a soul therapy. Various types of yoga are practiced like Japa yoga, Karma yoga, Gyana yoga, Bhakti yoga, Raja yoga, Swarayoga, Kundalini, and Nadi. Each school has its principles and practices leading to the fulfillment of the ultimate goal of yoga, which is the attainment of inner well-being. Yoga is essentially a spiritual discipline that leads to the union of individual consciousness with that of the universe, bringing a perfect harmony between mind and body (22, 23, 97, 99).
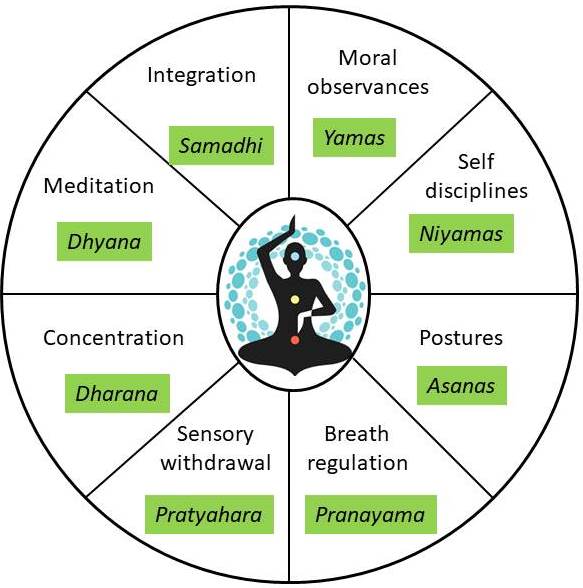 Figure 4
Figure 4Components of Eight-fold path of Patanjali’s Raja yoga.
In the Yoga Sutras, Yoga is defined as “union” of mind, body, and spirit. Classically, yoga is understood as the science of the mind (100). These days it is assuming importance in improving mental health and quality of life in the treatment of several disorders (21). Quality of life means an excellent physical and mental condition, consisting of two elements: the ability to cope with everyday tasks and the patient’s satisfaction from his activities at all levels as well as control over the disease and symptoms connected with the treatment method being applied. (101). Despite several studies on yoga, the molecular and cell biological mechanisms of the effects of yoga and related practices remain mostly unknown. It is now well established that perturbations in the environment give rise to distinct changes in gene expression. Recent work has shown that not only physical changes in the environment, but also psychological, social, and cultural components can induce gene expression changes, studied by the emerging field of psychosocial genomics (102). In a previous study from our lab, we have shown that patients with a chronic disorder like glaucoma are 6.9 times likely to have depression and even assessed the magnitude of caregivers burden and found that primary care-givers have significant emotional and psychological strain and moderate depression in 1/3rd of individuals giving primary care (15, 18, 103). Adverse life experiences have been suggested to give rise to significant changes in gene expression in circulating immune cells (104). Consistent with this framework, the first set of studies on long-term (months to years) yogic practitioners have found that these practices may positively affect gene expression profiles in immune cells in the circulation (24, 99).
RA has no permanent cure to date, but the disease can be partially conquered by the minimization of risk factors responsible for the beginning and the progression of the disease. Restoring defective mechanisms causing psychological and OS and reduction in the rate of immune aging emerges as a therapeutic target in resetting immune abnormalities in RA. Like other complex lifestyle disorders, RA has been identified as a stress-aggravated illness that often can be dealt with through a program of stress management (105). Yoga is a comprehensive philosophical system that seeks to develop one’s inherent power of mind, body, and personality in a balanced manner. Yoga system comprises a collection of asanas (postures), pranayama (breathing exercises), and dhyana (meditation). Yoga is practiced to reduce stress and to treat anxiety, high blood pressure, and musculoskeletal conditions. In a previous study from our laboratory on a set of healthy individuals, we have observed a decline in levels of cortisol and IL-6, a decrease in free radical concentrations, and up-regulation of telomerase levels after 12 weeks of yoga and meditation practice (9). These practices have shown positive changes in gene expression profiles in immune cells of long-term yogic practitioners (24, 106).
Despite several studies on yoga, the exact molecular mechanisms underlying the benefits of yoga and related practices remain largely unknown. It is now well established that not only physical changes in the environment but also psychological, social, and cultural components give rise to distinct changes in gene expression. A recently published study from our lab has shown a decrease in OS in RA patients after 8-weeks of yoga and meditation practice (yoga protocol is shown in Table 4) which is seen by reduced ROS, reduced 8-OHdG and upregulation of total antioxidant capacity levels and also the upregulation of various anti-inflammatory markers and downregulation of pro-inflammatory markers (8).The yoga protocol used in our study was framed in such a way that it did not cause any further damage to already inflamed joints, and the main purpose was to achieve optimal range of motion, improve muscle flexibility and reduce the inflammation (8).The yoga asanas were modified and adapted so that the patients could perform it without any difficulty in the initial stages of the intervention. The anatomical and physiological aspects related to each yogic practice were kept in mind. The formulation of the yoga protocol specific to RA patients as each yoga practice is responsible for the activation of specific muscles associated with it (100, 107, 108). Yogic sukshmavyayama and sthoolvyama help to sequentially warm up all joints of the body with a special focus on small joints of hand and feet and helps to maintain balance and coordination, respectively (100). This system has a strong purifying effect on the energy body of a human (100).Asanas (postures) were taught to maintain in different positions like sitting, standing, supine, and prone. Trikoṇasana (the triangle pose) maintains flexibility of the spine, strengthens the calf, thigh, and waist muscles and improves lung capacity (109). Katichakrasana (spinal twists pose) helps to improve the waist and spinal mobility and leads to stretching of different groups of abdominal muscles. Tadasana (palm tree posture) stability in the body helps to clear up congestion of the spinal nerves and corrects faulty posture (100, 107, 109).Virabhadrasana (warrior pose) helps in stretching and strengthening of the pectoral, shoulder, neck, pelvic, psoas muscles. It also strengthens and stretches the thighs, calves, and ankles and leads to the stretching of the entire front of the body while strengthening the thighs, ankles, and back. Gomukhasana (cow face pose) stretching of gluteal muscles, relieves chronic knee pain, strengthens spinal, and abdominal muscles (107, 109). Paschimottanasana (seated forward bend) stretches the back extensor muscles, stretches the body from head to ankle, and contracts the anterior abdominal wall muscles. It also creates pressure on the thorax and abdomen, improving the process of respiration and the functioning of the intra-abdominal glands, especially the secretions and provides a soothing effect on the mind, removing anxiety, anger, and irritability (107, 109). Shashankasana (the hare pose) induces relaxation, relieves depression, and provides a good upper body stretch. Vakrasana (simple spinal twist) massages the abdominal organs, helps facilitate digestion, regulates the secretion of digestive juices, and reduces constipation and stomach diseases (11, 100, 109). Ekpadashalabhasana (single leg locust pose) and poornashalabhasana (complete locust pose) improves flexibility and coordination, exercises the back muscles, and increases strength and stamina (100, 109). Bhujangasana (cobra pose) relieves stress, reduces abdominal fat and relieves constipation, and helps to relieve backache and bronchial problems (109). Makarasana (crocodile pose) promotes relaxation of the whole body, helps in recovery of back problems and counterbalances stress and anxiety levels (109, 110). Uttanpadasana (raised leg pose) strengthens the abdominal muscles, tones all the abdominal organs, and improves the functioning of reproductive organs. Setubandhasana (bridge pose) helps relieves depression, anxiety and strengthens lower back muscles and stretches abdominal organs, improves digestion, and helps to relieve constipation (100, 109). Pawanmukatasana (wind liberating pose) removes constipation, gives relief from flatulence, decreases the bloating sensation in the abdomen, aids digestion, generates deep internal pressure, and improves stretching of muscles, ligaments, and tendons in the pelvis and waist region and tones up the back muscles and spinal nerves (100, 109).Matsyasana (fish pose) stimulates spinal muscles, cervical muscles, thorax, rib cage, and the lungs, thus relieving your fatigue almost instantly. Pranayama is a practice of breath control and helps in developing awareness of one's mind and helps to establish control over the mind (109, 111).Supraphysiological levels of ROS give rise to the progression of the disease characterized by elevated ESR levels, a marker for inflammation. Regular practice of yoga and meditation lowers OS, ongoing inflammation, and disease activity (16, 27, 112). In concordance with the results of other studies, we also found statistically significant improvement in disease activity (measured by DAS28-ESR), pain acuity scale (measured by VAS) and disability quotient (measured by health assessment questionnaire -HAQ) in patients who underwent 8-weeks of YBLI (8, 113–116). Thus this simple mind-body intervention can reduce disease severity and associated comorbid depression and thereby results in lowering the dose of DMARDs used in RA treatment.
| S.No. | Practice to be done | Duration | ||
|---|---|---|---|---|
| 1 | Session preparation instructions | 2 min | ||
| Starting Prayer | 5 min | |||
| Sukshmavyayama | Finger loosening, Wrist loosening, Elbow loosening, Shoulder loosening, Toe bending, Ankle bending, Knee cap tightening, patella movement, Knee bending & Hip rotation | 5 min | ||
| Sthoolvyama | Rekhagati, Sarvaangpushti | 5 min | ||
| 2 | Asana | Standing | Trikonasana, Katichakrasana, Tadasana, Virabhadrasana | 5 min |
| Sitting | Gomukhasana, Paschimottanasana, Shashankasana, Vakrasana | 5 min | ||
| Prone | Ekpadashalabhasana, Bhujangasana, Poornashalabhasana, Makarasana | 5 min | ||
| Supine | Uttanpadasana, Setubandhasana, Pawanmukatasana, Matsyasana | 5 min | ||
| 3 | Relaxation | Savasana | 10 min | |
| 4 | Pranayama | Kapalbhati | 20 min | |
| Ujjayi | ||||
| Nadishodhana | ||||
| Bhramari | ||||
| 5 | Nada anusandhana, AUM- Aumkar recitation | 3 min | ||
| 6 | Dhyana (meditation) | 15 min | ||
| 7 | Shanti mantra- Closing prayer | 5 min | ||
| 8 | Interactive Session/ Self-directed learning | 30 min | ||
| Total | 120 min | |||
RA serves as a model of disrupted intracellular signaling cytokine-mediated pathways like Janus Kinase/Signal Transducers and Activators of Transcription (JAK/STAT), stress-activated protein kinase/mitogen-activated protein kinase (SAPK/MAPK) or Phosphatidylinositide-3-Kinase/AKT/mammalian Target of Rapamycin (PI-3K/AKT/mTOR) which can alter the activity of various immune cells and stimulate inflammation (117). Improved understanding of the pathogenesis of RA has led to the development of biologic agents that are designed to target specific cellular interactions and cytokines that mediate RA associated tissue damage like TNF-alpha antagonist, IL-1 inhibitors, B cell-targeted therapy, etc. (1, 118). There may be multiple serious side-effects reported on various organ systems of the body by the drugs used in the management of RA, which may lead to hospitalization (118–120). Inflammatory stress stimulates the HPA axis and leads to glucocorticoid overexposure, which is associated with the ‘resetting’ of the HPA axis sensitivity (121, 122). Glucocorticoid overexposure, hypoxia or over-expression of hypoxia-inducible factor, increased ROS generation causes up-regulation of glucocorticoid receptor at the transcriptional level. It creates an epigenetic alteration in specific genes related to the re-programming of the HPA axis (123). The bidirectional dysregulation of immune response and triggering of stress precipitates autoimmune conditions (124). Current treatment mainly focusses on treating symptoms rather than to cure the root cause as a whole. Yoga has a buffering action on stress-mediated immune response and maintains the cardio-vagal tone. It maintains homeostasis between the parasympathetic and sympathoadrenal nervous system and downregulates the HPA axis response (1, 99, 125–127). An 8-weeks meditation practice program has led to the elicitation of the relaxation response in healthy subjects showed downregulation of transcripts associated with regulation of apoptosis, nuclear transport, metabolic processes, JAK-STAT cascade, T and B cell activation, regulation of cell cycle, insulin sensitivity, glucose transport, DNA replication, chemokine signaling and stress response (106). Yoga intervention also upregulates the expression of DNA repair genes and anti-inflammatory cytokines such as IL-2 and IL-4 and decreases the levels of oxidative DNA damage marker, 8-OHdG, inflammatory cytokines such as IL-6, TNF-alpha (9, 15). Our recent study on active RA patients documented the suppression of acute-phase responses by lowering the levels of ESR and CRP, alteration of inflammatory markers and destruction of the hyperactive immune system by downregulation of pro-inflammatory cytokines (IL-6, IL-17A, and TNF-alpha), whereas upregulation of anti-inflammatory cytokines (TGF-beta) followed by an 8-weeks of yoga-based mind body intervention (8). Yoga normalizes the levels of transcripts involved in DNA repair, DNA damage, genome stability, cell cycle checkpoint control, and causes downregulation of pro-inflammatory genes (8, 10, 14, 25). Microarray studies on the effect of YBLI from our lab showed downregulation in expression levels of various pro-inflammatory cytokine and chemokine receptor-ligand transcripts an upregulation in expression levels of various anti-inflammatory, cell cycle control, DNA repair genes. Alteration of the hyperactive immune system by upregulation of an immune-modulatory and anti-inflammatory molecule, i.e., sHLA-G by yoga significantly reduced disease activity in RA patients following yoga regimen along with DMARDs as compared to RA patients only on routine DMARDs (8). Non-classical HLA class Ib molecule, sHLA-G, possesses immune-modulatory and anti-inflammatory properties. Since its reduced levels are associated with a severe form of the disease and high disease activity, it becomes a vital reference marker while assessing the severity of the disease. HLA-G is a tolerogenic molecule, and its upregulation is beneficial in autoimmune disorders as it prevents breach in self-tolerance and progression of auto-immunity (128). Therefore, yoga as a lifestyle intervention possesses an immune-modulatory potential which maintains immunological tolerance and remission at a molecular level.
Cellular health integrity is essential for the maintenance of life. Yoga aids in the maintenance of cellular health and longevity by optimization of redox homeostasis, checking genomic damage, and telomere length maintenance by upregulation of telomerase. HPA hyperactivity is associated with the generation of more free radicals leading to OS (129), causing altered methylation of DNA repair genes and modulation of base-excision repair activity increasing oxidative DNA damage susceptibility (130). OS is a hallmark of inflammation in RA, as evidenced by a fivefold increase in mitochondrial ROS production in whole blood and monocytes of RA patients as compared with healthy subjects (79). T-cell exposure to increased OS perpetuates the abnormal immune response and mutation of p53 in RA-derived fibroblast-like synoviocytes (131, 132). Citrullination of peptides in RA is linked to perturbation in mitochondrial homeostasis leading to excessive ROS production, impaired mitochondrial dynamics, electron transport chain defects, bioenergetics imbalance, and increased AMPK activity, decreased mitochondrial NAD+ and altered metabolism, and mitochondrial calcium accumulation (50). Such mitochondrial signals activate p53/p21 and p16/pRb pathways resulting in cellular senescence (133). Low levels of antioxidants are associated with RA, like GSH, tocopherols, beta-carotene, and retinol, and low activities of GR and SOD have been associated (79). This redox imbalance impairs cellular integrity and causes wide-spread damage to nuclear DNA, cell membranes, and even mitochondrial DNA (mtDNA).Within the CNS, in comparison to glial cells, neurons are more vulnerable to OS due to the relatively poor expression of endogenous antioxidants. 8-OHdG, a major product of DNA oxidation, leads to DNA breaks, base modifications, DNA mutations, genomic instability in nuclear, and mtDNA (134). Products of lipid peroxidation like conjugated diene hydroperoxides and unstable substances, which disintegrate into various bioactive aldehydes such as MDA and 4-HNE, are known to alter protein structures and render them antigenic (135). ROS can influence the methylome through the formation of oxidized DNA lesions formed by hydroxylation of pyrimidines and 5-methylcytosine, which can interfere due to structural similarities with epigenetic signals related to 5-hydroxy-methylcytosine (136). ROS also affects DNA demethylation by DNA oxidation and TET-mediated hydroxymethylation (137). ROS can indirectly modulate the activity of the epigenetic machinery since histone-modifying enzymes depend on intracellular levels of essential metabolites, such as Acetyl-CoA, Fe, ketoglutarate, NAD+, and S-adenosylmethionine, indicating that epigenetic changes are tightly linked to global cellular metabolism and energy levels of the cell (138). Therefore, OS can globally influence the cell on multiple levels, from DNA and histones to histone modifiers, which directly affects the epigenetic landscape of the cell (26, 27). Hence, maintaining and achieving optimum oxidative eustress, even under extremes of stress associated with lifestyle and environmental challenges, is a highly challenging task and needs a regulated redressal. Although OS could be reversed by supplementation of various synthetic anti-oxidants, however, their indiscriminate usage may cause a cell to undergo “reductive stress” (9). Physical exercises also help in reducing the production of free radical and upregulation of beta-endorphin, an endogenous opioid produced in the pituitary gland (19, 139, 140). Previous studies from our laboratory have documented that YBLI reduces OS, maintains mitochondrial integrity by elevating cytochrome c oxidase (COX) activity, upregulates total anti-oxidant capacity, and telomerase enzyme. Hence, YBLI aids in the maintenance of telomere length and genomic stability in the healthy population (9), thus reducing and delaying the onset of age-related chronic diseases and complex lifestyle disorders (8, 16, 27, 112, 141). Yoga also increases plasma melatonin levels hence maintains sleep-wake cycles and also aids in the upregulation of SIRT1, which promotes longevity by deactivation of p53 mediated pathways. RA patients have insufficient amounts of telomerase reverse transcriptase enzyme and undergo accelerated immune aging, which is evident by telomere shortening (83, 142). Even the cases with a positive familial history of RA, i.e., with a strong genetic component, showed a marked improvement in the symptoms of RA after 8-weeks of YBLI. There is an upregulation in telomerase enzyme levels with 8-weeks YBLI, which maintains the pace of cellular aging and telomere length in RA patients. Hence, yoga not only delays the premature early aging of the immune system in RA but also reduces the disease activity, severity, and co-morbid functional disability by the maintenance of cellular health and longevity (8). Although aging is natural and inevitable, however, with the adoption of yoga, one can age gracefully.
Studies have shown that complementary and alternative medicine therapies like yoga are popularly used as an adjunct to modern medicine and help in the reduction of depressive symptoms (8-10, 145–148). Yoga includes a set of physical exercise and emotional focus, which aims to bring overall relaxation and harmony of mind. The co-morbid depression can be reduced by acquiring attentional control over internal thoughts and emotions with the help of behavioral approaches like the gold standard cognitive-behavioral therapy (CBT), which helps curb anxiety and maladaptive thinking. Still, many CBT trials left patients symptomatic and non-respondents (143, 147, 148). Psychological components associated with RA have been ineffectively dealt with by DMARDs, which leads to an exaggeration of the disease symptomatology (149). Yoga promotes neurogenesis as it upregulates BDNF, which is a marker of neuroplasticity (8-10, 152). Also, yoga results in significant fold change by an upregulation in levels of CX3CL1, GPR50, and RB gene; CX3CL1 is a cytokine found in the brain, especially in neurons with receptors in microglia with an essential role in microglial migration. Its levels are upregulated with spatial learning and facilitate neurotransmitter tone and maintain protective plasticity of synaptic or homeostatic scaling. Also, there was an upregulation in levels of GPR50 and expression of the RB gene, a tumor suppressor gene. Depression is associated with an increased risk of cancer and also in cases like RA, which is a chronic progressive inflammatory arthritis. Thus, the increased expression of the tumor suppressor gene aids in regulating cell proliferation. A study from our lab reported a significant reduction in depressive symptoms of RA patients who practiced yoga along with the routine medications, as indicated by their time-dependent decreased BDI-II (Beck Depression Inventory-II) scores (8). Although other forms of yoga, such as Iyengar yoga, performed twice weekly for 6-weeks did not produce significant changes in disease activity or pain, whereas the quality of life, mood, and fatigue improved significantly (148). Duration, frequency, and intensity of yoga intervention according to the severity of patients’ symptomatology are a critical factor in producing a desirable clinical response. Both long term and short term yoga and meditation practitioners exhibited gene expression changes associated with the upregulation of genomic stability pathways like telomere packaging, telomeres maintenance, whereas downregulation of inflammatory and apoptotic pathways (106). Data from randomized trials suggest that yoga reduces symptoms of anxiety and depression along with positive benefits for inflammation, reductions in SNS tone, and increases in vagal activity (126, 151–155). Deficiency of neurotransmitters like dopamine, serotonin, and norepinephrine are associated with anxiety, social phobia, and depression (156–158). Studies suggested that yoga is found to significantly elevate the markers related to neuroplasticity like BDNF, serotonin, and beta-endorphins, neuregulins, neurotrophins post 8-week YBLI along with the reduction of depression measuring scale, i.e., BDI-II (144, 159–161).
Yoga, an integrative health strategy, helps to alleviate RA symptoms, reduction of disease activity by regressing inflammatory pathways and contributes as an adjunct in the management of RA (8, 11). Yoga focusses on both the psychological and physical aspects of the disease and helps to maintain immune homeostasis between various T cell subset populations, especially Th17 and Tregs. Yoga also aids in amelioration of depression associated with RA and disease activity along with improvement in systemic markers of inflammation – acute phase reactants (ESR and CRP), pro-inflammatory (IL-6, IL-17A, TNF-alpha), anti-inflammatory cytokines (TGF-beta) and immune-modulatory molecule (sHLA-G); markers of neuro-plasticity (BDNF, DHEA, serotonin, beta-endorphins, melatonin); markers of OS (ROS, TAC and 8-OHdG); and cellular health and longevity (SIRT1), cellular aging (telomerase activity and maintenance of telomere length) (Figure 5) (8, 10). Increase in levels of BDNF, DHEA is both cytoprotective and promote synaptogenesis and are the underlying mechanism of the increase in hippocampal, prefrontal cortex, and cerebellar volume. Yoga most probably acts via the psycho-neuro-immunological axis, which creates a homeostatic balance between sympathetic and parasympathetic limbs of the autonomic nervous system during the aggressive symptomatic phase and helps to normalize the flare and achieve remission (97, 157). Yoga helps to improve the overall quality of life by reducing pain perception, disability quotient, and disease activity in active RA, which is seen by a significant reduction in VAS, HAQ-DI, and DAS28-ESR scores respectively (8, 12). Yoga is a cost-effective emerging health discipline that, unlike drugs, has no side-effects and aid in prolonging the periods of remissions with fewer relapses. Yoga not only reduces disease severity, optimizes OS levels, increases periods of remission, but also minimizes the usage of drugs with minimum side effects hence can be beneficial as an adjunct therapy.
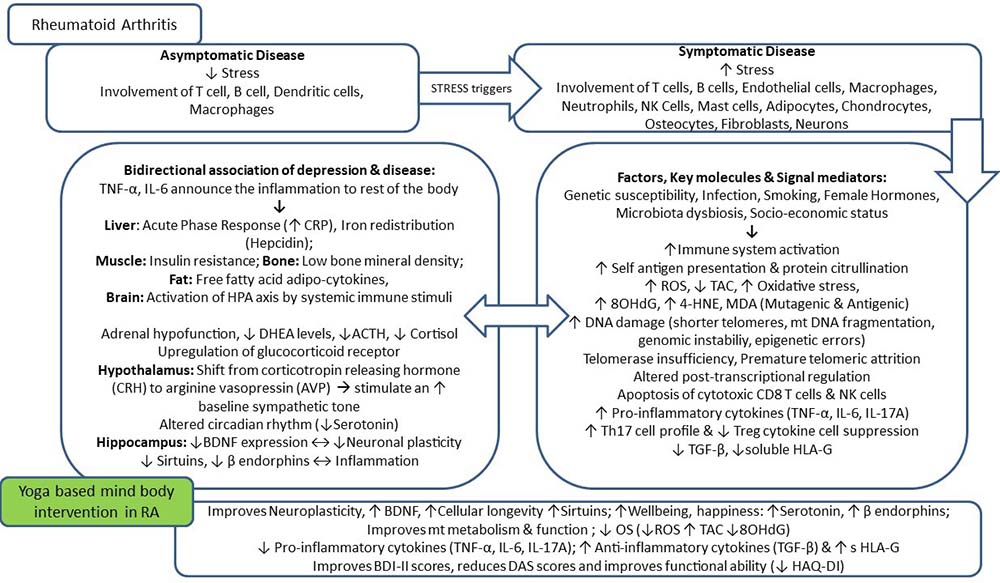 Figure 5
Figure 5A comprehensive summary highlighting the biological markers associated with RA and the impact of YBLI on these biomarkers.
Over the last two decades, yoga has received increasing awareness and attention from the scientific community seeking to understand the safety and efficacy of these widely accepted practices (23). A large number of studies suggest mindfulness-based interventions, including MBSR, mindfulness-based cognitive therapy, and integrated mindfulness yoga practices are effective psychological interventions to reduce depression and anxiety in clinical and non-clinical populations (162, 163). Mindfulness can be described as a process of sustained attention and awareness toward present-moment sensations without concentrating on any image, mantra, or thought. The MBSR approach includes daily mindfulness activities related to the recognition of mind and body while focusing on the breath. The mindfulness-based cognitive therapy approach consists of an integration of principles of cognitive-behavioral therapy with MBSR, designed to reduce relapse and recurrence of major depressive disorders (162, 163). Yoga creates a balance between the two limbs of the autonomic nervous system, i.e., induction of parasympathetic nervous system (PNS) activation via the release of neurotransmitters responsible for relaxation response, improving cardio-vagal tone, decreasing heart rate and blood pressure; and activation of limbic structures of the brain and suppression of SNS activation, which is responsible for the stress response including the release of cortisol (108). With long term practice of yoga, PNS activation dominates over SNS activation, even during stressful situations (164). Yoga can be a useful add-on therapy for RA patients to improve the physical and mental health of patients. A definitive role of yoga in RA improvement has been documented in various studies as it aids in reducing pain, improving function, and creating a positive mental state. Still, the underlying mechanism behind its observed mechanisms has not been explored fully (11, 100). An account of existing literature consisting of various clinical trials on the effect of yoga on RA is shown in Table 5. Pranayama Yoga, Zen, Transcendental meditation, and other meditation program has diaphragmatic breathing as a mainstay procedure. Meditation proves to reduce anxiety, alleviate chronic pain, improve cognitive functions, and lowers OS levels (lower cortisol levels and higher melatonin levels) (22, 77). These results show that practices like yoga and meditation are beneficial for RA patients with active disease and can become adjunct therapy for controlling RA symptoms, prolonging periods of remissions, and preventing relapses.
| Study Design; participants | Number of participants | Duration | Intervention |
Control Description | Biomarkers | Results | References |
|---|---|---|---|---|---|---|---|
| RCT; RA patients | n=72; |
8-weeks | Yoga group: yoga-based mind-body intervention based on Patanjali’s Raja yoga: 120 min per day and 5 sessions per week for 8 weeks by yoga instructors + prescribed DMARDs | Non-yoga control group: to follow normal day to day physical activities + prescribed DMARDs | Markers of Systemic inflammation: ESR, CRP, IL-6, IL-17A, TNF-alpha and TGF-beta, soluble HLA-G; Markers of neuroplasticity: BDNF, serotonin, beta-endorphins; Markers of oxidative stress and DNA damage: ROS, TAC, 8-OHdG Markers of longevity and cellular aging: SIRT-1, telomerase activity and telomere length |
ESR, CRP, IL-6, IL-17A, and TNF-alpha showed a significant (p < 0.05) decline, whereas TGF-beta and soluble HLA-G (p < 0.05) showed a significant increase in yoga group as compared to control. A significant decrease in OS as there was a decrease in ROS, 8-OHDG (p < 0.05) and increase in TAC (p < 0.05); SIRT1, telomerase activity showed a significant increase in yoga group (p < 0.05) and change in telomere length was not significant in either group. A significant decline in HAQ-DI, DAS28ESR and BDI-II scores of yoga group as compared to the control group (all p < 0.05) | Gautam S et al., 2019 (8) |
| Pilot RCT RA patients | n=26; |
13 weeks | Yoga group: 8-week yoga group practice once-weekly for 75-min by yoga therapists and followed by a 5 week home yoga practice thrice a week for 20-min guided with the help of a CD. Yoga group continued with their usual medical care | Usual care group: Participants continued with their usual medical care provided by their rheumatologist. | Adverse events: musculoskeletal pain, nausea, RA flare up, Flu, Infection, Neuralgia etc. |
Yoga practice was feasible and safe for the RA population experiencing mild pain, mild to moderate functional disability, and moderate disease activity without any significant adverse events and significant improvements in the outcome measures. | Ward L et al., 2018 (171) |
| RCT (RA (n=37)+OA (n=38)) patients | n=75; yoga group (n=35); waitlist group (n=40) | 8-weeks; post-yoga follow-up at 9-months also | Yoga group: 60-minute Integral-based hatha yoga class held twice weekly for 8-weeks | Waitlist group: to maintain current levels of physical activity for 8-weeks, and then they were invited to participate in upcoming yoga classes. | Assessment of physical health: SF-36 Physical Component Summary and Fitness; Assessment of psychological function: SF-36 Mental Component Summary, CES-D, PANAS; |
Clinically significant improvements in physical and mental health, fitness, psychological function, and HRQOL with no adverse outcomes. | Moonaz SH et al. 2015 (172) |
| Single-arm pilot study; (RA+OA) patients in minority communities | n =20 (yoga group) | 8-weeks; post-yoga follow up at 3-months also | 8-week program of yoga classes consisting of 60-minute sessions, twice a week | - | Assessment of Health literacy: REALM-SF, SAHLSA-50 |
A number of challenges in recruiting and retaining participants from a community clinic serving minority populations. | Middleton KR et al., 2013 (173) |
| RCT; RA patients | n=80; Experimental group (n==40); Control group (n==40) | 7-weeks | Experimental group: Yogic package of 1.5 hours per day for 6-days a week for 7-weeks | Control group: they were wait-listed to participate in upcoming yoga classes. | Outcome measures: Pain intensity, Inflamed joints, Morning Stiffness, Pulse rate, Systolic BP, Diastolic BP, Lymphocyte count, CRP Level, Serum uric acid | Yoga significantly reduced pain intensity, joint inflammation, early morning stiffness, pulse rate, blood pressure, lymphocyte count, C –reactive protein and uric acid towards normalcy in participants as compared to controls | Singh VK et al., 2011 (174) |
| RCT; RA patients | n =30 (Iyenger yoga group, n=14; Wait-list control group, n=16) | 6-weeks | Iyengar yoga group: twice a week for 1.5 hours duration per session for 6-weeks | Wait-list control group: continued with their ongoing medical treatment | Assessment of HRQOL, pain, and functioning: |
No significant changes in disease activity or pain in yoga group as compared to the control group, but markers of well-being- including quality of life, mood and fatigue-improved in the yoga group compared to the control group | Evans S et al., 2013 (175) |
| Exercise intervention; post-menopausal RA women | n =16 (yoga group, n= 9; control group, n=7) | 10-weeks | Yoga group: Three 75 minutes of yoga classes per week for 10-weeks | Control group: Wait-list participants | Outcome measures: Diurnal cortisol patterns, resting heart rate, Breg balance test, HAQ-DI, VAS, BDI | Yoga resulted in a significantly decreased HAQ-DI, VAS, BDI, and increased balance. No significant change in awakening or diurnal cortisol patterns in the yoga group | Bosch PR et al.,2009 (176) |
| RCT; RA patients | n = 47; yoga group, n=26; control group, n =21) | 8-weeks | Yoga group: 1 hour each twice a week for 8-weeks | Control group: patients who were interested in yoga but unable to commit to schedule at the present time due to work or personal priorities | Outcome measures: Tender joint count, swollen joint count, global patient assessment, E SR, DAS 28, HAQ, Fatigue, Change in DMARD usage, SF-36 | The yoga group showed statistically significant improvements in DAS28 and HAQ, but not QOL | Badsha H et al., 2009 (115) |
Yoga is a comprehensive philosophical system that seeks to bring balance into all aspects of body, mind, and soul. Yoga system comprises a collection of asanas (postures), pranayama (breathing exercises), and dhyana (meditation). Combined treatment with yoga or a long term adoption of yoga as a lifestyle behavior in combination with other healthy lifestyle behaviors can bring about a reversal of adverse epigenetic changes and provide not only amelioration of RA symptoms and may decline the chances of relapses (8, 10, 160, 164). There are limited numbers of studies that have explored the molecular aspect behind yoga therapy to date. The results from our laboratory study highlight the positive impact of 8 weeks of yoga-based mind-body intervention on depression associated with RA, which further influences the disease sequelae via changes in biomarkers at a systemic level (8). Overall, RA outcome is dependent on disease activity, which relates to functional abilities, ongoing systemic inflammation, and depression severity. Yoga facilitates mind’s capacity to overcome disease and improve physical symptoms via a variety of downstream pathways like – maintenance of immune-homeostasis via an optimized balance between Tregs and Th17 cells, the balance between pro and anti-inflammatory markers, OS eustress, psychological stress regulation, harmonization of sleep-wake cycles and circadian rhythms, stress and anxiety management by the improvement of neuroplasticity markers and promotes neurogenesis, maintenance of pace of cellular aging via telomere metabolism, attaining genomic stability, maintenance of optimum levels of neurotransmitters, and upregulation of immune regulatory proteins (8–10, 99). These underlying mechanisms significantly aid in the improvement of overall health, provide long-term clinical remission in RA and reduced episodes of relapses, and bring about natural tolerance. Hence, yoga holds the potential to positively modify the pathobiology and disease course of autoimmune diseases like RA by approaching mind-body interactions.
YBLI results in achieving optimal levels of OS, reducing the rate of cellular aging and psychological stress by a gradual decline in free radical levels in the blood, upregulation of telomerase activity, reduction in cortisoland inflammatory cytokines and upregulation in the expression of various anti-inflammatory and cell cycle control genes, improving mitochondrial integrity and upregulating expressions levels of multiple genes which code for antioxidants and thereby increase the total antioxidant capacity. The Bermuda triangle of OS, immune aging, and psychological stress has a definitive role in the pathogenesis of RA. Regular practice of yoga provides a buffering effect on accelerated immune aging in susceptible individuals hence a key to healthy senescence. Adoption of a healthy lifestyle by quitting smoking, increased intake of fruits and vegetables, maintaining optimal weight, improving sleep patterns/ adequate sleep, and integrating yoga and meditation into our life will not only minimize OS, prevent autoimmunity but also significantly improve quality of life and promote physical and mental health. Thus yoga targets the common underlying etiology of most complex lifestyle diseases. Yoga not only promotes health but can prevent/delay the onset of such diseases even in cases with positive family history and has immense preventive and rehabilitative potential. Thus this mind-body intervention is the need of the hour in the age of super specialization. A simple lifestyle based mind-body intervention like yoga significantly reduces disease activity, chronic disease pain, swelling, and associated stress and disability, hence holds the potential to be an adjunct therapeutic regimen in RA patients.
The authors sincerely thank the yoga therapists, Ms. Richa Mishra and Mr. Sandeep Kumar, for providing yoga instructions and counseling to RA patients. Financial assistance by the Department of Science and Technology (DST), India (SR/SATYAM/55/2016) and Indian Council of Medical Research (ICMR), New Delhi, India, in the form of a Senior Research Fellowship (45/2/2019-ANA/BMS) to Ms. Surabhi Gautam is thankfully acknowledged.
RA
Rheumatoid arthritis, DMARDs: Disease-modifying anti-rheumatic drugs
Yoga-based lifestyle intervention
Mindfulness-based stress reduction
Brain-derived neurotrophic factor
Dehydroepiandrosterone
sirtuin-1
Interleukin
hypothalamic-pituitary-adrenal
Soluble human leukocyte antigen
Tumor necrosis factor-alpha
Rheumatoid factor
Antibodies to citrullinated protein antigens
World health organization
Indian council of medical research
Disease activity score 28
Tender joint count
Swollen joint count
Visual analog scale
Erythrocyte sedimentation rate
C- reactive protein
Human leukocyte antigen
Major histocompatibility complex
peptidyl arginine deiminase type 4
Protein tyrosine phosphatase non-receptor type 22
signal transducer and activator of transcription
cluster of differentiation 244
cytotoxic T lymphocyte-associated antigen 4
T-cell receptor
Antigen presenting cells: PAD: Peptidyl arginine deiminase
Neutrophil extracellular traps
Granulocyte-macrophage colony-stimulating factor
Transforming growth factor-beta
regulatory T cells
T helper 17 cells
T helper 1 cells
Interferon: SNS: Sympathetic nervous system
Parasympathetic nervous system
Reactive oxygen species
Oxidative stress
malondialdehyde
hexanal,4-hydroxy-2-non-enal
8-hydroxy-2’-deoxyguanosine
Senescent associated secretory phenotype
Ataxia-telangiectasia mutated
Meiotic recombination 11 homolog A
Nijmegen breakage syndrome 1
American College of Rheumatology
European League Against Rheumatism
Ayurveda, Yoga and Naturopathy, Unani, Siddha, and Homoeopathy
Health assessment questionnaire
Nuclear factor kappa-light-chain-enhancer of activated B cells
Extracellular-signal-regulated kinase
Janus Kinase/Signal Transducers and Activators of Transcription
stress-activated protein kinase/mitogen-activated protein kinase
Phosphatidylinositide-3-Kinase/AKT/mammalian Target of Rapamycin
cytochrome c oxidase
Cognitive-behavioral therapy
Beck Depression Inventory-II.





