1 Department of Psychiatry, The First Affiliated Hospital, Zhejiang University School of Medicine, 310003 Hangzhou, Zhejiang, China
2 Nanhu Brain-Computer Interface Institute, 310003 Hangzhou, Zhejiang, China
3 The Zhejiang Key Laboratory of Precision Psychiatry, 310003 Hangzhou, Zhejiang, China
4 MOE Frontier Science Center for Brain Science and Brain-Machine Integration, Zhejiang University School of Medicine, 310003 Hangzhou, Zhejiang, China
5 Brain Research Institute of Zhejiang University, 310003 Hangzhou, Zhejiang, China
6 Department of Psychology and Behavioral Sciences, Zhejiang University, 310003 Hangzhou, Zhejiang, China
7 Zhejiang Engineering Center for Mathematical Mental Health, 310003 Hangzhou, Zhejiang, China
†These authors contributed equally.
Abstract
Accumulating evidence highlights the role of Vitamin B12 (VitB12) in the pathophysiology of affective disorders. However, its influence on brain function and the underlying mechanisms remain incompletely understood. In humans, VitB12 is obtained solely from dietary sources, primarily animal-based foods. VitB12 deficiency leads to the accumulation of homocysteine, a known contributor to emotional and behavioral dysregulation. VitB12 plays a critical role in maintaining neuron stability, synapsis plasticity, and regulating neuroinflammation by modulating key bioactive factors. These processes help to alleviate hippocampal damage, mitigate blood-brain barrier disruption, reduce oxidative stress, and enhance both structural and functional connectivity—collectively contributing to resilience against affective disorders. VitB12 from both diet and microbial sources is essential to gut homeostasis. Within the gut lumen, it stabilizes gut microbial communities, promotes short-chain fatty acid (SCFA) production, and supports neurotransmitter metabolism (e.g., serotonin and dopamine) via its role in S-adenosyl-l-methionine biosynthesis. Crucially, VitB12, gut microbiota, SCFAs, intestinal mucosa, and vagal nerve signaling interact synergistically within the gut-brain axis (GBA) to maintain gut microenvironment stability, protect the gut-blood barrier, and suppress neuroinflammatory cascades, eventually reducing the susceptibility to affective disorders. This review synthesizes current evidence on the involvement of VitB12 in the GBA, its association with mood regulation, and its potential as a nutritional adjunct in managing affective disorders. By elucidating this integrative mechanism, we provide new insights into targeting the GBA to improve clinical outcomes in affective disorders.
Keywords
- vitamin B12
- bipolar disorder
- depressive disorder
- gastrointestinal microbiome
• Draw a map of vitamin B12 in the gut-brain axis with the latest findings.
• Link vitamin B12 to affective disorders via systematic circulation and the gut-brain axis.
• Probe the potential of vitamin B12 to influence resilience to affective disorders.
Affective disorders, mainly including major depressive disorder (MDD) and bipolar disorder (BD), remain significant contributors to the substantial global health burden due to their high prevalence, recurrence, and suboptimal clinical outcomes in the absence of timely interventions. According to global health data, MDD and BD ranked 13th and 67th, respectively, in disability-adjusted life years across all age groups. Strikingly, among individuals aged 15–24 years, they rose to 4th and 32nd, underscoring their disproportionate impact on youth population [1]. Despite extensive research efforts, the etiology and pathophysiological mechanisms underlying affective disorders remain incompletely understood, posing a major challenge for the development of targeted therapies.
Vitamin B12 (VitB12), also known as cobalamin (Fig. 1, Ref. [2, 3, 4]), plays a critical role in modulating neurodevelopment [5] and the modulation of psychiatric symptoms, including depression, irritability, hallucinations, and agitation [6]. Within the body, VitB12 exists in two distinct physiological pools: (1) a water-soluble form in the systemic circulation, derived from dietary sources; and (2) components in the gut-brain axis (GBA), which originate from both diet and gut microbiota. Systemic circulation serves as the primary pathway for transporting VitB12 from ileal enterocytes (the site of absorption) to target cells. In target cells, VitB12 is essential for the conversion of homocysteine (Hcy) to methionine, methylmalonyl-CoA to succinyl-CoA [7], and intimately links to the metabolism of one-carbon units, fatty acids [2, 8], amino acids, neurotransmitters [9], myelin synthesis, and DNA/RNA production [10]. Beyond systemic effects, the GBA is a bidirectional communication network connecting gut microbiota and brain function through neuronal, endocrine, and immune pathways [11]. This axis has garnered increasing attention as both a pathophylogical contributor and a therapeutic target for neuropsychiatric disorders, including MDD, BD, schizophrenia, and anxiety [12].
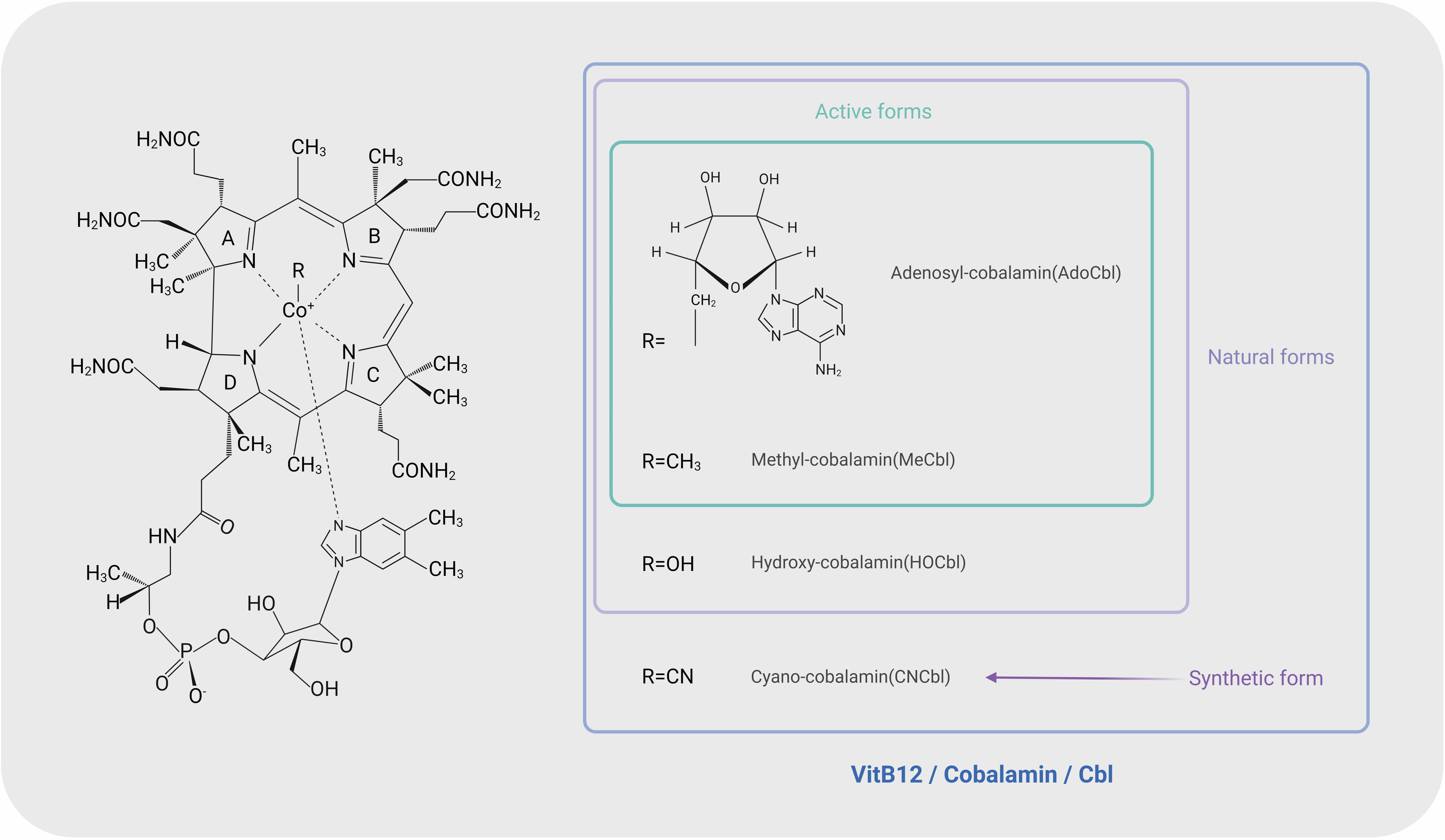 Fig. 1.
Fig. 1.
Different forms of Vitamin B12 (VitB12) and conceptual relationships. VitB12, also called cobalamin, is a family of structurally related vitamers, including adenosyl-cobalamin (AdoCbl), methyl-cobalamin (MeCbl), hydroxyl-cobalamin (HOCbl), and cyano-cobalamin (CNCbl). Among these compounds, AdoCbl, MeCbl, and HOCbl are natural forms, while CNCbl is a synthetic form and will be transformed into other three forms in the human body. AdoCbl and MeCbl are biologically active coenzyme forms for human metabolic processes [2, 3]. In contrast, pseudo-cobalamin or cobalamin analogs refer to molecules that retain the corrin ring structure but lack bioactivity for humans. These analogs may nevertheless interact within the gut microenvironment, influencing gut microbiota composition and short-chain fatty acids metabolism [4]. The figure was created using BioRender (https://www.biorender.com/).
A range of GBA-targeted interventions has been proposed for managing psychiatric disorders, including affective disorders [12]. For instance, probiotic and prebiotic supplementation may restore gut microbiota homeostasis, modulate microbial-derived metabolites (e.g., short-chain fatty acids), and enhance gut barrier integrity [13]. Transcutaneous vagus nerve stimulation has demonstrated potential to improve depressive symptoms by attenuating sympathetic nervous system hyperactivity and reducing systemic inflammatory mediators, thereby enhancing treatment outcomes [14]. Furthermore, emerging evidence suggests that nutritional interventions, particularly VitB12 supplementation, may confer benefits within the GBA framework, potentially addressing neuroinflammation and neurotransmitter dysregulation implicated in mood disorders [15]. However, several issues should be addressed before the clinical application of VitB12 supplementation for affective disorders. For example, the precise mechanisms by which VitB12 improves mood status, the dosage and frequency of VitB12 supplementation, and the potential adverse events caused by VitB12 supplementation. In this review, we explore the multifaceted roles of VitB12 within GBA and discuss its potential as therapeutic adjunct in the management of affective disorders.
VitB12 is synthesized exclusively by certain bacteria and archaea [2, 16]. While non-human mammals acquire VitB12 through coprophagic behavior or via fermentation processes of ruminant digestive systems [17], human rely entirely on animal-derived foods for VitB12 intake, such as meat, eggs, and dairy [18]. The absorption of VitB12 in humans occurs in a multi-step pathway in the gastrointestinal tract (Fig. 2). In the upper gastrointestinal tract, VitB12 is enzymatically released from dietary proteins and binds to haptocorrin (HC), a glycoprotein presents in saliva and gastric secretions. Upon reaching the duodenum, pancreatic proteases degrade HC, allowing VitB12 to bind to intrinsic factor (IF) and forming the IF-VitB12 complex, which is subsequently internalized by enterocytes in the terminal ileum via the cubilin-amnionless receptor complex (cubam) [19]. Once inside ileal enterocytes, the IF-VitB12 complex dissociates, and free VitB12 is released into systemic circulation through the ATP-binding cassette transporter multidrug resistance protein 1 (MRP1) [7]. In circulation, VitB12 is transported by two binding proteins, HC and transcobalamin (TC). Approximately 80–90% of VitB12 is bound to HC and serves as a storage reservoir, while the remaining 10–20% is bound to TC and is delivered to target tissues, including the liver, via receptor-mediated endocytosis [20]. The TC-VitB12 complex is recognized by receptors such as megalin, cubilin, and the CD320 molecule (CD320) [21, 22, 23]. CD320, in particular, is critical for mediating the uptake of TC-VitB12 by central nervous cells, ensuring its role in neurological function [24].
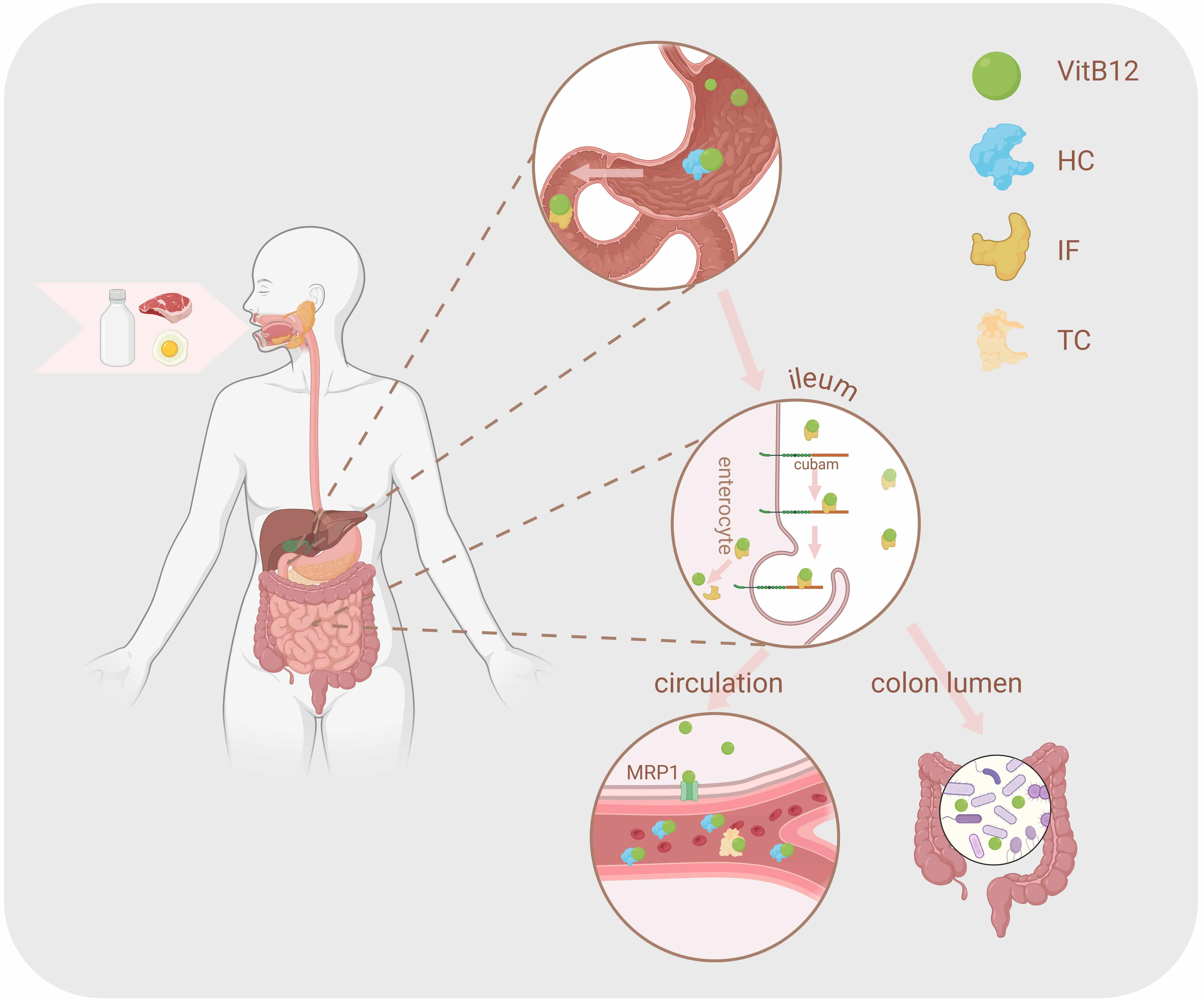 Fig. 2.
Fig. 2.
Absorption and utilization of VitB12. Food-derived VitB12 is absorbed in the small instestine, with partially entering systemic circulation at the end of the ileum, while the remainder contributes to the gut luminal environment. HC, haptocorrin; IF, intrinsic factor; TC, transcobalamin; MRP1, multidrug resistance protein 1. The figure was created using BioRender (https://www.biorender.com/).
The human body maintains sufficient VitB12 stores to meet physiological needs for 3 to 5 years [25]. Nearly 50% of this reserve is localized in the liver and maintained through enterohepatic circulation [26]. Additionally, the kidneys also contribute to VitB12 homeostasis through a dual role by secreting excess VitB12 via urine and simultaneously serving as a secondary storage site [27, 28]. This balance is achieved via a filtration-reabsorption mechanism in the renal tubules. In high-income countries, VitB12 deficiency is relatively uncommon in the general adult population due to efficient physiological regulation and widespread nutritional adequacy [29]. However, specific risk factors persist, including food-cobalamin malabsorption [30], inadequate dietary intake (especially in strict vegetarians), elevated physiological demands (pregnant or lactating women), and medications such as metformin and proton pump inhibitors [31].
Several clinical studies have suggested that low serum VitB12 levels and high
Hcy concentrations are associated with affective disorders in both adolescents
and adults [32, 33, 34, 35, 36, 37, 38, 39], supporting the hypothesis that VitB12 deficiency may lead to
Hcy accumulation [7]. Individuals with depression have been found to exhibit
significantly higher serum Hcy levels compared to healthy controls, and
hyperhomocysteinemia (defined as serum Hcy level
Despite growing evidence, the causal relationship between serum VitB12 levels and affective disorders remains unclear. It has been hypothesized that low VitB12 and elevated Hcy levels may contribute to the onset of affective symptoms. Alternatively, the progress of affective disorder and associated pharmacological treatments may negatively impact dietary intake or intestinal absorption of VitB12, leading to secondary deficiency [35]. Sex- and age-related variations further complicate this relationship. For instance, men generally present with higher Hcy levels than women, possibly due to estrogen’s regulatory effects on Hcy metabolism [40, 41]. Observational studies also indicate that deficiencies in both VitB12 and folate contribute to elevated Hcy levels, suggesting that VitB12 insufficiency alone may not fully explain the risk of mood disorders. Mechanistically, VitB12 serves as a coenzyme for methionine synthase, an enzyme essential for the remethylation of Hcy to methionine using 5-methyltetrahydrofolate as a methyl donor. Thus, both VitB12 and folate deficiencies would hamper this process, leading to the accumulation of Hcy and subsequent disruptions in neurotransmitter synthesis, DNA methylation, myelin maintenance, and other processes critical to neurological and mental health [42].
Epidemiological data suggest that approximately 5% of general psychiatric inpatients present with low serum VitB12 levels, with this figure rising to 10–20% among elderly individuals. Notably, chronic VitB12 deficiency spanning over 40 years may contribute to the development of Alzheimer’s disease or multiple sclerosis, suggesting that the neurological consequences of VitB12 deficiency may develop progressively over long-term exposure [43]. Furthermore, subclinical VitB12 deficiency (blood level less than 200 µmol/L), defined as a state of inadequate VitB12 in the absence of overt clinical symptoms or with only mild symptoms [44], has also been implicated in affective disorders. These findings suggest that even mild or asymptomatic deficiencies could induce subtle physiological impairments, potentially influencing mental health outcomes. Nevertheless, the mechanisms by which subclinical VitB12 deficiency contributes to affective disorders, as well as its broader systemic effects, call for more comprehensive investigation.
Emerging evidence reveals neuroinflammation as a critical mediator of neural
impairment in mood disorders [45]. Chronic neuroinflammatory processes, driven by
dysregulated immune signaling, disrupt synaptic plasticity, impair neurogenesis,
and damage neuronal networks, particularly in brain regions like the prefrontal
cortex, hippocampus, and amygdala, which govern emotional regulation and
cognitive function [45]. Activated microglia,as well as pro-inflammatory
cytokines (e.g., IL-6, TNF-
VitB12 exerts neuroprotective effects on neural tissues through modulating DNA methylation and bioactive factors metabolism [48, 49, 50]. For instance, de Queiroz et al. [48] revealed that VitB12 counteracted hippocampal neuronal damage in a rat model of bacterial meningitis by modulating DNA methylation, stabilizing genomic integrity, and upregulating anti-inflammatory gene expression. S-adenosyl-l-methionine (SAMe), a methyl donor engaging in DNA methylation for genomic instability [48], is not only synthesized systemically through VitB12-engaged one-carbon metabolism [51] but also is derived from gut microbiota in the intestinal lumen [52]. Notably, SAMe itself exhibits antidepressant-like properties via promoting serotonin synthesis and activating the serotonin 1A receptor activation [53]. Similarly, Suryavanshi et al. [50] reported that VitB12 supplementation in diabetic models reduced neuronal apoptosis and restored levels of brain-derived neurotrophic factor (BDNF), a key mediator of nerve development, survival, and synaptic plasticity [54]. Reduced levels of BDNF were strongly linked to depression, and its upregulation was a recognized mechanism of antidepressant treatment [54, 55]. Collectively, this evidence highlights the potential of VitB12 in enhancing resilience against affective disorders by preserving neuronal stability through epigenetic, anti-inflammatory, and neurotrophic pathways.
VitB12 alleviates neuroinflammation through multiple mechanisms. For example, Jonnalagadda et al. [22] reported that neuroinflammation suppressed the VitB12-transcobalamin 2-CD320 pathway in an animal model of multiple sclerosis, and fingolimod, a therapeutic agent for multiple sclerosis, exerted its anti-inflammatory effects via restoring this pathway. Cassiano et al. [56] uncovered that VitB12 attenuated inflammatory infiltrate in the central nervous system, thereby mitigating hippocampal damage and preventing neurological dysfunction in pneumococcal meningitis [57]. These findings collectively implied that the VitB12-mediated modulation of neuroinflammatory processes in the central nervous system was intricately linked to the pathophysiology of neuropsychiatric diseases. In addition, peripheral inflammation alone can trigger functional and structural dysconnectivity in frontostriatal, amygdala-prefrontal, and interoceptive circuits. Connectomic analysis showed that dysregulation within these neuronal dynamic networks underlay mood fluctuations, a core clinical manifestation of BD [58]. Systemic inflammation further disrupted the blood-brain barrier (BBB) and up-regulated reactive oxygen species (ROS), thus creating a neurotoxic milieu that exacerbates affective disorders [57, 59, 60, 61]. This also implied the potential of peripheral inflammatory profiles as predictive biomarkers for affective disorders.
Taken together, VitB12 deficiency leads to the accumulation of ROS, neuroinflammation, and demyelination, contributing to a vicious circle in the central nervous system [57] and potentially involving in the pathogenesis of neuropsychiatric disorders. Conversely, VitB12 supplementation demonstrates neuroprotective properties by mitigating neuronal damage, stabilizing neural integrity, and suppressing inflammatory pathways. However, clinical evidence is lacking to validate this assumption, especially in patients with affective disorders.
The impact of dietary derived VitB12 on the gut microbiota remains a subject of
debate, owing to inconsistent findings across in vitro and in
vivo studies. In vitro experiments suggested that VitB12
supplementation enhanced
Within the gut microenvironment, VitB12 serves as an essential nutrient for microbial communities. About 20% of gut bacteria synthesize VitB12 de novo, while 80% possess receptors for absorbing VitB12, highlighting its important role in microbial survival and cooperation [66]. There are two major routes (aerobic and anaerobic) by certain gut microbes to synthesize VitB12 de novo. The two routes diverge in their cobalt chelation mechanisms and oxygen dependencies, as the aerobic pathway requires oxygen to catalyze ring-contraction, whereas the anaerobic pathway proceeds independently of oxygen [67]. VitB12 is involved in cross-feeding interactions in microbe-microbe and microbe-host patterns [68], and microbe-derived VitB12 is self-sufficient in healthy adult gut [69]. For Escherichia coli , the presence of VitB12 suppresses the expression of BtuB, a gene encoding a transportor responsible for active VitB12 intake [70]. This feedback mechanism illustrates how commensal bacteria dynamically regulate VitB12 utilization to adapt to its availability, optimizing metabolic efficiency within the gut ecosystem.
VitB12 supplementation has limited effects on the gut environment in both in vitro and in vivo studies [8, 69], suggesting the existence of regulatory mechanisms that stabilize VitB12 levels in the gut lumen. For example, Kelly et al. [71] research indicated that high-dose VitB12 supplementation suppressed the endogenous VitB12 production by gut microbiota, suggesting that gut microbiota can sense VitB12 concentrations and regulate its production. Similarly, Li et al. [72] confirmed that in Propionibacterium strain UF1, a high dose (750 µM) of VitB12 completely inhibited gene expression responsible for VitB12 biosynthesis. These findings highlight how VitB12-dependent microbiota and their metabolic activities are integrated into a homeostatic system. Notably, while VitB12 supplementation depleted Bacteroides populations, it failed to change SCFAs concentration or microbial diversity in the distal gut, despite depleting, implying the intrinsic resilience in the gut lumen [71]. Further complexity arises from interactions between gut microbiota and corrinoids, a class of compounds including cobalamin and cobalamin analogs. High-dose (exceeding the recommended levels by 1000-fold) VitB12 supplementation temporarily disrupted corrinoid profiles, but it returned to the baseline level within 10 days [4, 73]. Degnan et al. [73] even proposed an inspiring idea to use non-metabolizable corrinoids that can evade microbial remodelling mechanisms to manipulate the composition and function of gut microbiota. However, major questions remain regarding how microbes discriminate among corrinoids, the dynamics of cobalamin-to-analog conversion, and the precise molecular mechanisms that govern microbial remodeling processes. Altogether, these findings challenge the assumption that dietary VitB12 supplementation has a straightforward or broadly beneficial impact on gut microbiota. Instead, they underscore the complexity and resilience of microbial ecosystems and highlight the need for deeper investigation into how VitB12 and related compounds functionally reshape gut microbial ecology and, by extension, influence mental health outcomes.
Clinical evidence highlights the role of VitB12 in mediating the interactions
between GBA and affective disorders. Both dietary and gut microbiota-derived
VitB12 help to keep a complex, balanced, and stable gut microbial network [9, 16]. Studies consistently indicated discrepancies in gut microbiota composition,
stability index, and disrupted metabolite profiles in individuals with affective
disorders compared to healthy controls. For example, in patients with MDD, fecal
samples mostly showed decreased
VitB12 plays a significant role in modulating inflammation implicated in MDD and BD [82, 83]. It activates gene expression in ileal epithelial cells by sustaining cellular methylation programs, thereby stimulating cell proliferation, enhancing SCFAs metabolism, and suppressing pro-inflammatory pathways [51]. SCFAs (e.g., acetate, propionate, and butyrate) are produced by gut microbiota through the fermentation of dietary fibres [84]. These metabolites can strengthen gut mucosal barrier [85], exert anti-inflammatory effects [12, 51], maintain enteric serotonin production [86], up-regulate BDNF [87], and reduce ROS accumulation [88]. In addition to dietary VitB12 [69], microbe-derived pseudo-VitB12 can promote propionate production independent of diet [89]. Meanwhile, butyrate, propionate, as well as VitB12 itself, serve as protectors to the gut barrier [16, 51, 89]. Food-derived SCFAs can normalize the immunodeficiency of germ-free mice via promoting microglia maturation, suggesting their role in maintaining immune homeostasis [90, 91]. Furthermore, SCFAs can cross the gut-blood barrier and exert an anti-inflammatory effect via binding to G-protein-coupled receptor 43 in systematic circulation [92]. Microbe-derived propionate protects BBB against ROS-induced damage [93]. Moreover, emerging research found that the vagus nerve acted as a key regulator in the gut-brain axis, bidirectionally mediating inflammatory signals between the brain and peripheral immune response [94]. The vagus nerve also transmits brain stress signals to regulate gut microbiota and immune activity [95]. This intricate crosstalk positions VitB12 and SCFAs as critical regulators of neuroimmune interactions in affective disorders.
Under pathological conditions of affective disorders, gut microbial composition exhibits a transdiagnostic pattern, characterized by a reduction in the abundance of butyrate-producing bacteria with anti-inflammatory properties and an increase in pro-inflammatory genera [12, 96]. Disturbance of the gut microbiome disrupts the permeability of the intestinal barrier, allowing the translocation of inflammatory molecules, bacterial endotoxins, and neuroactive metabolites into systemic circulation. Through bidirectional gut-brain crosstalk [81], these mediators propagate chronic systemic inflammation, which compromises the BBB and drives neuroinflammation [97, 98]. Collectively, VitB12, gut microbiota, SCFAs, gut mucosal defenses, and vagal neurotransmission interact synergistically within the GBA to maintain gut and neuroimmune homeostasis and enhance resilience to affective disorders (Fig. 3).
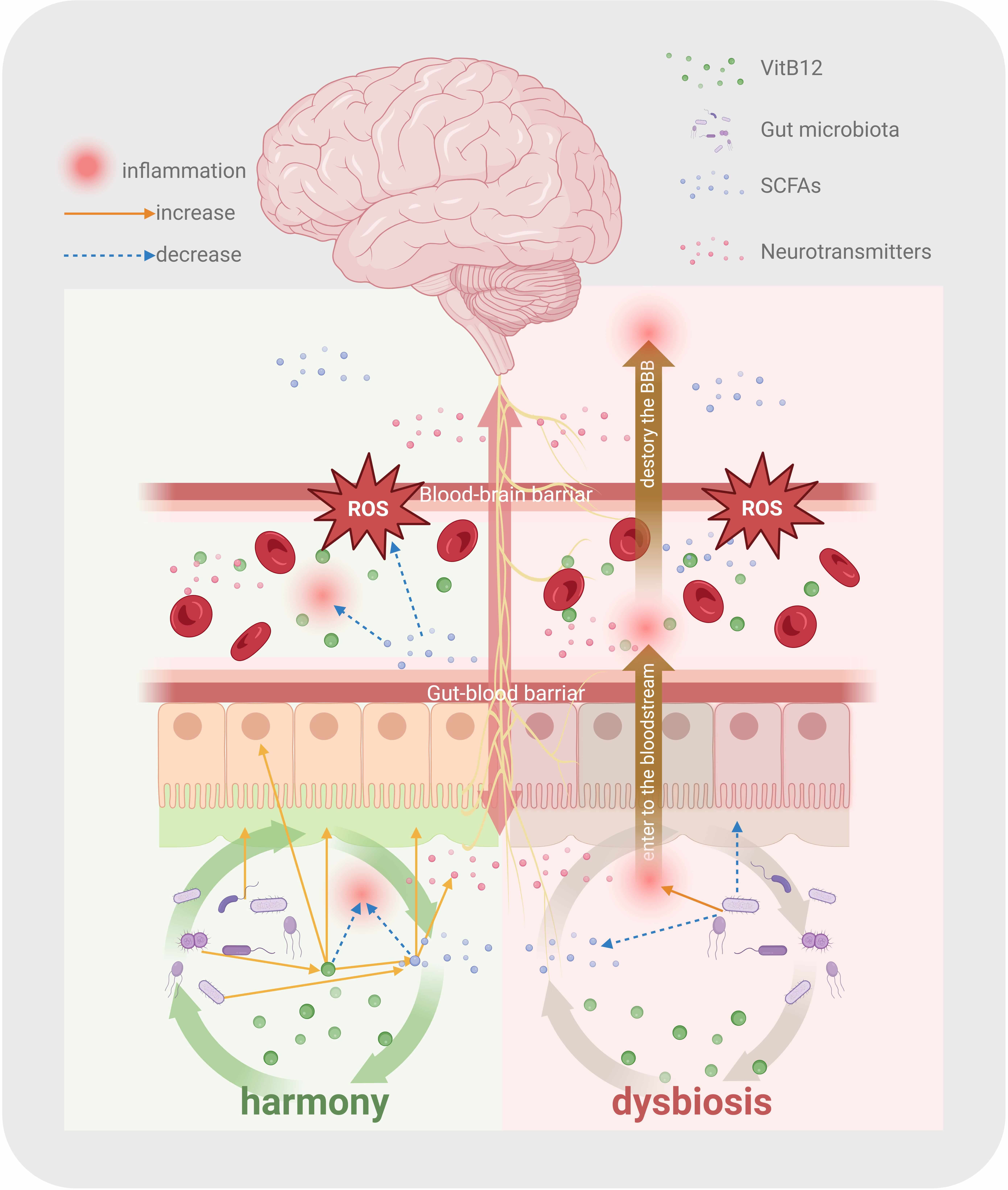 Fig. 3.
Fig. 3.
VitB12 modulates the functional equilibrium of the gut-brain axis. Under homeostatic conditions, VitB12 interacts with gut mucosa, microbiota, and microbial metabolites, synergistically suppressing inflammation and maintaining homeostasis of the gut-brain axis. Under dysbiosis states, these complex interactions are disrupted, triggering inflammatory cascades in both peripheral circulation and central nervous system, thereby increasing susceptibility to affective disorders. SCFAs, short-chain fatty acids; BBB, the blood-brain barrier; ROS, reactive oxygen species. The figure was created using BioRender (https://www.biorender.com/).
For patients diagnosed with VitB12 deficiency, treatment recommendations vary based on symptom severity and absorption capacity. Those presenting with acute symptoms or malabsorption are recommended to receive intramuscular VitB12 injections, while those with no or mild symptoms and without absorption or compliance concerns may be better suited for oral supplementation [29]. Oral supplements showed equal efficacy to intramuscular injection in VitB12 deficiency related to gastrointestinal disorders [99, 100], supporting the feasibility of oral administration in such cases. As for VitB12 forms, Xu et al. [101] found that despite methylcobalamin (MeCbl) and hydroxylcobalamin (HOCbl) exhibited equivalent bioavailability, MeCbl was more favorable for gut microbiome and microbial metabolism, suggesting MeCbl is the preferred choice for oral supplementation. As a water-soluble vitamin, excess VitB12 can be excreted renally, rendering it generally non-toxic even in cases of overdose. Nonetheless, it is preferred that VitB12 should be supplied within an appropriate dosage. Intestinal absorption efficacy declines sharply with higher doses, dropping from approximately 50% for dose below 50 µg to just 1% at 500 µg [3]. Similarly, cellular absorption dose not improve with supraphysiological concentrations [102]. A case report documented VitB12 overdose triggered mixed episodes of BD in a susceptible individual [103]. This also enlightens that VitB12 supplementation should be within a reasonable dosage to mitigate potential risks.
To date, no consensus has been reached regarding the use of VitB12 supplementation in patients with affective disorders. Higher VitB12 level has been proven to relate to better ketamine efficacy [104]. Further studies and clinical trials are therefore needed to explore whether VitB12 supplementation contributes to clinical drug efficacy and disease prognosis.
Whether microbe-derived VitB12 can be absorbed into the bloodstream via the colon requires further exploration. In traditional perspective, microbe-derived VitB12 is unavailable for human consumption, as absorption primarily occurs in the ileum, a site upstream of the predominant colonization of gut microbiota. However, Sun et al. [105] reported an intriguing exception that microbe-derived VitB12 can enter systemic circulation, evidenced by elevated serum VitB12 levels correlating with fecal concentrations. Nevertheless, this interpretation may be confounded by the fact that the liver was the primary storage organ for VitB12, and Silymarin’s effects on liver lipid metabolism may result in alterations to plasma VitB12 concentrations. Previous evidence indicated that small intestinal bacterial overgrowth may lead to VitB12 loss due to host-microbe competition in the small intestine [73, 106]. Whereas bacterial density in the healthy small intestine remains below than 105 CFU/mL [107], the composition and function of these intestinal bacteria are ongoingly being investigated.
Although VitB12 is the focus of this review, a variety of nutritional supplementation is increasingly used as adjunct treatments in patients with affective disorders [108]. The interplay and collaborative effects of B vitamins warrant further investigations. For instance, Rakić et al. [109] proved that a vitamin B complex (VBC, composed of B1, B2, B3, B5, B6, and B12) exerted neuroprotective effects by alleviating neuroinflammation. The same team also found that VBC can repair peripheral nerve cells and improve gut microbiota dysbiosis due to autoimmune encephalomyelitis [110]. Notably, synergistic effects of vitamin B1, B6, and B12 surpass the efficacy of any single B vitamin, implying the potential of VBC in treating peripheral neuropathy [10, 111]. Moreover, vitamin B (folate, B2, B6, and B12) may contribute to the production of butyrate, as vitamin B supplementation has been linked to the increased relative abundance of butyrate-producing commensal bacteria. This suggests a collaborative role of B vitamins in enhancing microenvironmental homeostasis [112].
In summary, VitB12 functions not as an isolated regulator but as an integral component of the GBA system, influencing gut microbial ecology, neuroimmune homeostasis, and host metabolic signalling. Rather than acting independently, VitB12 interacts with microbial communities, short-chain fatty acids, and neuroinflammatory pathways to shape affective outcomes. This positions VitB12 as a promising avenue for advancing our understanding of the pathophysiology of affective disorders and for identifying novel adjunctive therapeutic strategies. To establish its clinical relevance, further human-based studies are needed to elucidate the underlying mechanism of VitB12 to gut-brain communication and to evaluate its clinical applicability as a complementary intervention within the GBA framework.
VitB12, Vitamin B12; GBA, the gut-brain axis; MDD, major depressive disorder; BD, bipolar disorder; Hcy, homocysteine; AdoCbl, adenosyl-cobalamin; MeCbl, methyl-cobalamin; HOCbl, hydroxyl-cobalamin; CNCbl, cyano-cobalamin; SCFAs, short-chain fatty acids; HC, haptocorrin; IF, intrinsic factor; MRP1, multidrug resistance protein 1; CD320, CD320 molecule; TC, transcobalamin; SAMe, S-adenosyl-l-methionine; BDNF, brain-derived neurotrophic factor; BBB, the blood-brain barrier; ROS, reactive oxygen species; VBC, vitamin B complex.
CX and LK contributed equally to this review’s conceptualization, original draft writing, review, editing, and visualization. TM and AT contributed to the review, editing, and visualization. JL led the conceptualization, supervision, and writing editing. SH contributed to conceptualization, supervision, and writing editing. All authors read and approved the final version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We acknowledge Dr. Yi Chen from McGill University for her help in polishing the language of the manuscript. All figures were created with BioRender (https://www.biorender.com/).
This work was supported by funding from the National Natural Science Foundation [82201676, 82471542].
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



