1 Department of Cardiovascular Medicine, Second Xiangya Hospital, Central South University, 410011 Changsha, Hunan, China
2 Department of Cardiovascular Medicine, Sun Yat-Sen Memorial Hospital, Sun Yat-Sen University, 510120 Guangzhou, Guangdong, China
Abstract
Helicobacter pylori (H. pylori) infection and atrial fibrillation (AF) are prevalent global health concerns that significantly impact societal and economic well-being. This study explored the potential associations between H. pylori infection and the incidence and progression of AF. Emerging research suggests that H. pylori may influence AF through various pathways, including systemic inflammation, metabolic disturbances, immune responses, and changes in the gut microbiota. These pathways provide a novel perspective on the etiology of AF, suggesting that chronic H. pylori infection could exacerbate or even initiate the arrhythmic events typical of AF. Current evidence, while preliminary, points to significant correlations, particularly through changes in markers such as C-reactive protein (CRP) and lipid metabolism, which are heightened in individuals with active H. pylori infection. However, the exact mechanisms and causal nature of this relationship remain elusive, with studies showing conflicting results. This inconsistency underscores the need for more comprehensive and rigorously designed clinical and experimental research to elucidate fully the interactions between H. pylori infection and AF. Understanding these connections is crucial for developing innovative treatments and management strategies targeting microbial influences in AF patients. Future research should focus on defining the role of H. pylori eradication in the clinical management of AF assessing its impact on disease progression and patient outcomes.
Keywords
- Helicobacter pylori
- atrial fibrillation
- inflammation
- gut microbiota
Helicobacter pylori (H. pylori), a flagellated, spiral-shaped, Gram-negative, microaerophilic bacterium, represents the most prevalent chronic bacterial infection globally [1]. The unique morphology and physiological capabilities of this bacterium allow it to penetrate the gastric mucosa and colonize the interstitial space between the mucinous sodium carbonate barrier, thereby allowing it to survive the direct effects of gastric acid. By producing urease to break down urea, H. pylori generates CO2 and ammonia to neutralize stomach acid and create a slightly alkaline environment suitable for survival [1, 2]. Statistically, approximately 43.1% of the global population is infected with H. pylori, with prevalence rates varying significantly across different regions—reaching as high as 56.1% in the Eastern Mediterranean and 53.3% in Africa [3, 4]. In addition, some studies have reported that the infection rate of H. pylori is approximately 30% in developed countries and up to 80% in some developing countries. The disease incidence in adults is significantly greater than in children [5]. H. pylori infection often presents asymptomatically but can lead to upper gastrointestinal diseases, such as gastritis and gastric ulcers. Furthermore, chronic H. pylori infection can result in serious complications, including gastric cancer and mucosa-associated lymphoid tissue (MALT) lymphoma, if not properly treated [6, 7, 8]. The high infection rate and pathogenicity have placed a great burden on society.
Atrial fibrillation (AF), the most common persistent cardiac arrhythmia encountered clinically, is predominantly diagnosed via electrocardiogram and is characterized by rapid and irregular atrial rhythms [9]. According to a study on the 2019 Global Burden of Disease Database, the total number of global patients with atrial fibrillation/atrial flutter (AF/AFL) reached 59.7 million in 2019, with 315,000 deaths due to AF and 8.39 million disability-adjusted life years (DALYs) lost, highlighting the significant harm that AF promotes as a global public health problem [10]. The incidence of AF varies across age groups; for example, it affects up to 9% of the population over 65 years of age and nearly one in five individuals over 85 [11, 12]. Major risk factors for AF include age, hypertension, diabetes, and obesity, all of which are closely linked with severe complications such as stroke, heart failure, and premature death [13].
In recent years, an increasing body of research has explored the potential link between H. pylori infection and AF. Given that both H. pylori and AF are significant global public health concerns, understanding their interplay is crucial for revealing the mechanisms of disease onset and developing novel preventive and therapeutic strategies [1, 9, 12]. Although primarily affecting gastric health, H. pylori and cardiac-affecting AF may seem unrelated, recent studies suggest a possible connection. However, the exact mechanisms of this potential link are not fully understood [14, 15, 16, 17]. The preliminary findings indicate common pathways involving inflammation, metabolic disorders, immune responses, and changes in the gut microbiota [17, 18]. This review aims to systematically evaluate the relationship between H. pylori and AF, explore the potential pathogenic mechanisms, assess current diagnostic and therapeutic approaches, and discuss the implications of this relationship for patient prognosis. We hope to pave the way for future research through an in-depth analysis, particularly in developing targeted treatment and prevention strategies. Thus, this study elaborates on the relationship between H. pylori and AF, aiming to provide deeper insights and guidance for clinical practice.
H. pylori is commonly believed to be closely associated with stomach-related diseases. Meanwhile, further investigations into H. pylori have revealed associations with additional gastric disorders, including neurological, hematological, respiratory, and cardiovascular diseases [19, 20, 21, 22, 23, 24]. The cardiovascular diseases related to H. pylori include AF, stroke, coronary heart disease, hypertension, and atherosclerosis [25]. The relationship between H. pylori and AF has always been a research focus, as shown in Table 1 (Ref. [14, 16, 17, 18, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36]).
| Author (year) | Brief description of study | Conclusions and results |
| Montenero et al. [17] (2005) | The study recruited 30 patients with paroxysmal AF and 29 patients with persistent AF to explore the relationship between H. pylori and AF. | A highly significant correlation was found between AF and H. pylori, which was especially strong in patients with persistent AF. |
| Badran and Mahfouz [30] (2007) | A study recruited 185 patients with coronary heart disease to investigate the relationship between H. pylori and AF among these patients. | A significant link was found between AF and H. pylori Cag A-positive strains in patients with coronary heart disease. |
| Bunch et al. [29] (2008) | The study involved 943 patients with AF. | An association between H. pylori and AF was observed, but this association weakened after adjusting for other factors, especially age. |
| Platonov et al. [31] (2008) | The study recruited 72 permanent AF patients and 72 healthy individuals from the same region to explore the effects of Chlamydia pneumoniae and H. pylori on idiopathic permanent AF. | Permanent AF was associated with elevated CRP levels, but this elevation was unrelated to early infection with Chlamydia pneumoniae and H. pylori. |
| Lunetta et al. [35] (2009) | A 7-year prospective study included 120 H. pylori-positive patients and 60 H. pylori serum-negative patients. | No significant difference was found in the incidence of AF between the two groups, and inflammation caused by H. pylori was ruled out as a cause of AF. |
| Ki et al. [32] (2010) | The study recruited 60 patients with AF (including 27 patients with paroxysmal AF and 33 with persistent AF) while using 36 patients with other rapid arrhythmias as controls. | H. pylori infection was associated with a decrease in serum TGF- |
| Schimke et al. [36] (2010) | The study involved 1179 patients with type 2 diabetes to explore whether the Cag A of H. pylori is related to the pathological changes in major blood vessels. | The Cag A gene in H. pylori was not associated with coronary heart disease, peripheral vascular disease, cerebrovascular disease, and AF. |
| Xie et al. [33] (2014) | In total, 600 patients were recruited to study the risk factors and corresponding intervention measures for H. pylori infection in AF patients. | Ages over 65 years and not receiving H. Pylori treatment are risk factors for H. pylori infection in patients with AF. |
| Geng et al. [26] (2014) | Recruited 45 AF patients and 45 health inspectors to investigate H. pylori in AF patients and the correlation between H. pylori infection and blood–lipid indicators. | H. pylori infection is not associated with AF, but H. pylori infection can promote the occurrence and development of AF by affecting lipid levels. |
| Wang et al. [16] (2015) | In total, 585 patients with AF were recruited to explore the relationship between H. pylori infection and different types of AF. | The values of H. pylori in patients with long-standing AF were significantly higher than those in short-standing AF and control groups. H. pylori δ value |
| Yan et al. [27] (2016) | A meta-analysis was conducted by searching PubMed, Embase, Web of Science, and the Cochrane Library databases for research on the relationship between H. pylori and arrhythmia. | H. pylori infection is one of the risk factors for AF in Asia and Africa. |
| Zhang et al. [18] (2018) | In total, 86 AF patients and 65 healthy examinees were selected to study H. pylori. The correlation between H. pylori infection and AF, CRP, and lipid metabolism. | H. pylori infection may affect the occurrence and development of AF through lipid metabolism and inflammatory response pathways. |
| Tetta et al. [34] (2019) | The review included six retrospective studies exploring the correlation between AF and H. pylori, including 335 H. pylori-positive AF patients, 621 H. pylori-negative AF patients, 643 H. pylori-positive normal individuals, and 1322 H. pylori negative normal individuals. | According to data analysis, no significant correlation was found between H. pylori infection and AF. |
| Rivington and Twohig [28] (2020) | A retrospective study based on IBM Explories (1999–2019) quantified the risk of AF-related diseases. | The likelihood of developing AF after infection with H. pylori increases, but the magnitude of the increase is minimal. |
| Farah et al. [14] (2024) | In total, 180 patients with H. pylori were recruited for the study. | AF is associated with H. pylori and is more significantly associated with age and elevated CRP levels in H. pylori patients. |
AF, atrial fibrillation; H. pylori, Helicobacter pylori; CRP,
C-reactive protein; Cag A, cytotoxin-associated gene A; TGF-
The association between H. pylori and AF was first proposed by Montenero et al. in 2005 [17], who suggested that inflammation mediated by H. pylori might play a role. Further studies have highlighted a stronger correlation between AF and H. pylori, especially in individuals harboring cytotoxin-associated gene A (Cag A)-positive strains of the bacterium. This relationship suggests a potential underlying mechanism that may involve the generation of autoantibodies [17]. Additionally, H. pylori can influence the progression of AF by affecting lipid metabolism pathways [26]. Indeed, H. pylori has been identified as an independent contributor in chronic and enduring cases of AF and is acknowledged as a significant risk factor for AF, particularly in regions such as Asia and Africa [27].
However, most studies utilize H. pylori IgG (which represents previous H. pylori infections). Meanwhile, few studies have investigated the associations between AF and different types of H. pylori, which limits the effectiveness of the support for a correlation between the two [17]. A study in China aimed to address these limitations by utilizing Hp values instead of Hp IgG values and investigated the correlation between different types of AF and H. pylori [16]. The correlation between different types of AF and H. pylori was stronger in long-term persistent AF and permanent AF, which also supports the influence of chronic inflammation caused by H. pylori on the occurrence and development of AF [16]. However, these studies were mostly case‒control studies; thus, they lacked more convincing randomized controlled trials and ignored the impact of proton pump inhibitors [37].
Although many studies have confirmed the correlation between H. pylori and AF, the validity of this conclusion still needs to be questioned owing to experimental design and regional differences; meanwhile, some studies have also reported a lack of correlation between H. pylori and AF. The conclusion that H. pylori is an independent factor for AF in Europe and North America is not significant and may be influenced by socioeconomic level [27]. A large retrospective study exploring the risk factors for AF, regarding noninflammatory diseases, hyperthyroidism, chronic obstructive pulmonary disease, alcohol, pulmonary embolism, inflammatory diseases, chronic kidney disease, inflammatory bowel disease, and H. pylori, revealed that H. pylori had the smallest contribution to AF. However, owing to the retrospective nature of the research data and the inability to obtain individualized data on diagnosis and risk factors, more research is still required to distinguish the correlation between H. pylori and AF [28]. Some studies suggest that the relationship becomes less apparent when adjusting for confounders such as age, while others have highlighted that age is a more significant factor in AF [14, 29]. The body of research is still in its exploratory phase, with inconsistent results both domestically and internationally, reflecting the impact of study design, sample selection, and geographical and ethnic differences on research outcomes [16, 18, 28, 29, 30, 31, 32, 33, 35, 36]. Establishing a causal relationship between H. pylori and AF, rather than just an association, remains challenging and requires further high-quality clinical research. Thus, conducting multiregional research, expanding sample sizes, conducting randomized controlled trials, and prospective studies in the future can elucidate this relationship and increase the persuasiveness of conclusions.
The new guidelines divide AF into four stages: Stage 1 refers to a state that poses a risk of causing AF; Stage 2 is the pre-stage of AF, during which patients are prone to AF; Phase 3 includes paroxysmal AF (3A), persistent atrial fibrillation (3B), long-term persistent atrial fibrillation (3C), and successful atrial fibrillation ablation (3D); Phase 4 is permanent AF. The guidelines define AF as a persistent state of the disease [38, 39, 40]. The triggering factors and maintenance mechanisms are the basis for the occurrence and development of different types of AF. Among them, the triggering factors of AF include abnormal electrical activity of pulmonary veins and other cardiac structures, autonomic nervous system disorders, and inflammation. The maintenance mechanism of AF includes multiple episodes, focal triggering theory, inflammation, and atrial remodeling. Early atrial remodeling manifests as electrical remodeling, such as changes in ion channels, whereas late-stage remodeling manifests as structural remodeling, such as tissue fibrosis [41, 42, 43].
Although there is currently no research directly elucidating the specific mechanism through which H. pylori affects the occurrence and development of different types of AF, with continued research on the relationship between H. pylori and AF, many scholars have proposed hypotheses on how H. pylori is involved in the onset and progression of AF. Research has shown that H. pylori may directly or indirectly affect AF through inflammation, metabolic disorders, and immune pathways [14, 17, 18, 44], as illustrated in Fig. 1.
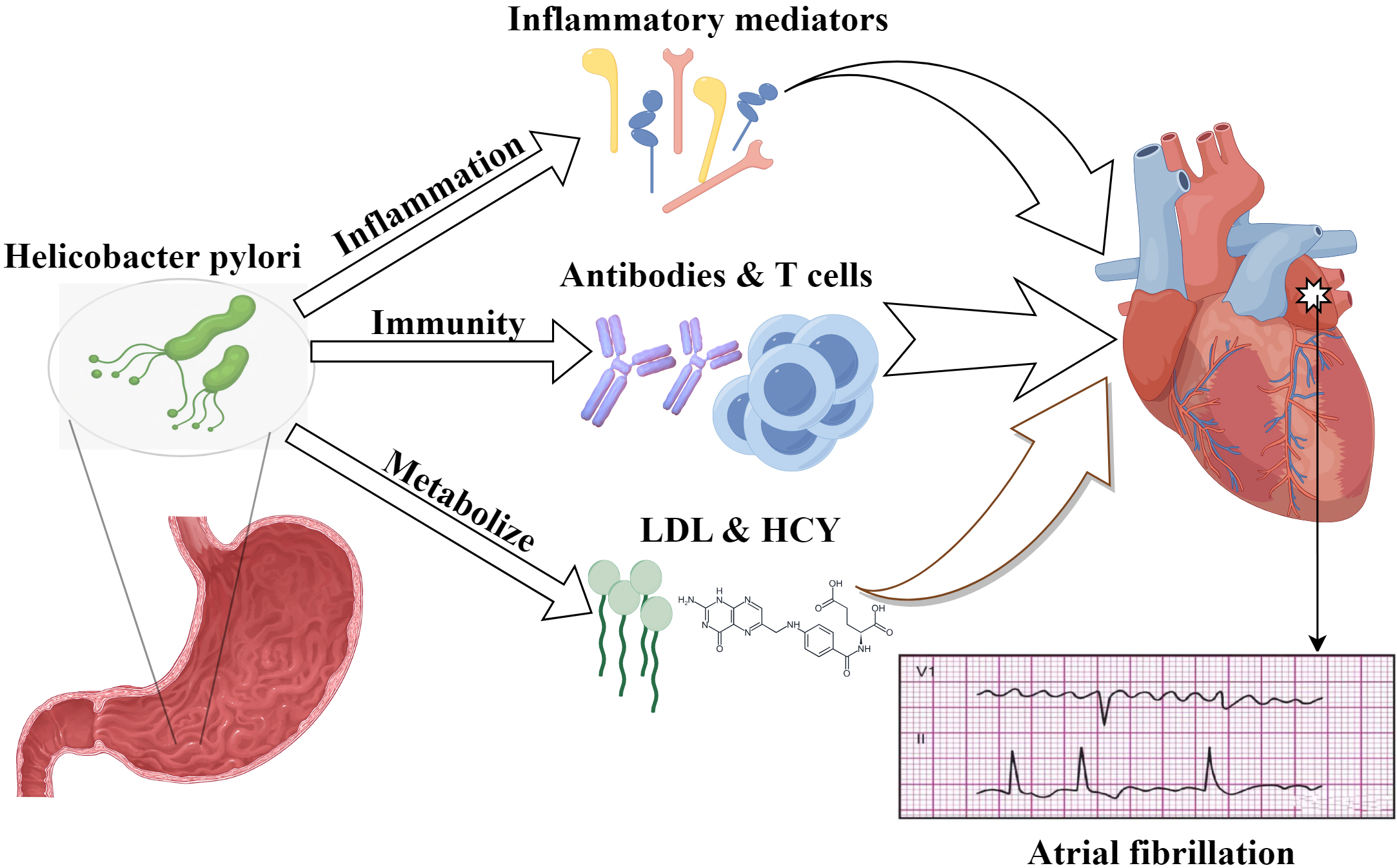 Fig. 1.
Fig. 1.
Possible mechanisms of H. pylori-induced AF. This diagram illustrates the complex interplay between LDL, HCY, and other factors in the context of H. pylori infection, which may culminate in AF. LDL, low density lipoprotein; HCY, homocysteine. The figure is created by Figdraw.
Numerous recent studies have identified a strong association between AF and inflammation, as evidenced by significant correlations between this arrhythmia and various inflammatory biomarkers, including C-reactive protein (CRP), tumor necrosis factor (TNF), and interleukins, interleukin (IL)-1, IL-2, IL-6, and IL-8 [45, 46]. This linkage has led researchers to search for factors that initiate inflammation, with chronic bacterial infection emerging as a likely candidate to trigger and sustain the inflammatory process. Notably, bacterial infections have been increasingly implicated in the pathogenesis of AF, with H. pylori garnering particular interest [46].
Studies have shown that H. pylori infection can induce gastric and esophageal inflammation and systemic and vascular inflammation, potentially increasing the risk of AF through increased CRP levels [14]. This result is achieved through the activation of the classical complement pathway and the binding of CRP and phosphatidylcholine, the latter of which can cause membrane dysfunction, leading to abnormal sodium and calcium processing. For example, CRP increases the risk of AF by increasing calcium influx through the inward L-type calcium channel in the atrial muscle [47, 48]. Moreover, the Cag A protein produced by H. pylori stimulates gastric epithelial cells to secrete the inflammatory mediator IL-8, inducing neutrophil infiltration and consequently causing atrial muscle damage. The extent of the myocardial damage is directly proportional to the duration of infection [16, 28]. Furthermore, research indicates that H. pylori immunoglobulin G (representing past infection) is not associated with the duration of AF, suggesting that long-term chronic inflammation caused by H. pylori may underlie the development of persistent and permanent AF [16]. Therefore, inflammation may be the basis for triggering and maintaining short-term AF. In contrast, long-term inflammation and the cardiac structural and electrical remodeling caused by inflammation may be among the causes of long-term AF [48].
Additionally, H. pylori infection
can also cause an increase in other proinflammatory factors, whereby elevated
levels of IL-6 and TNF-
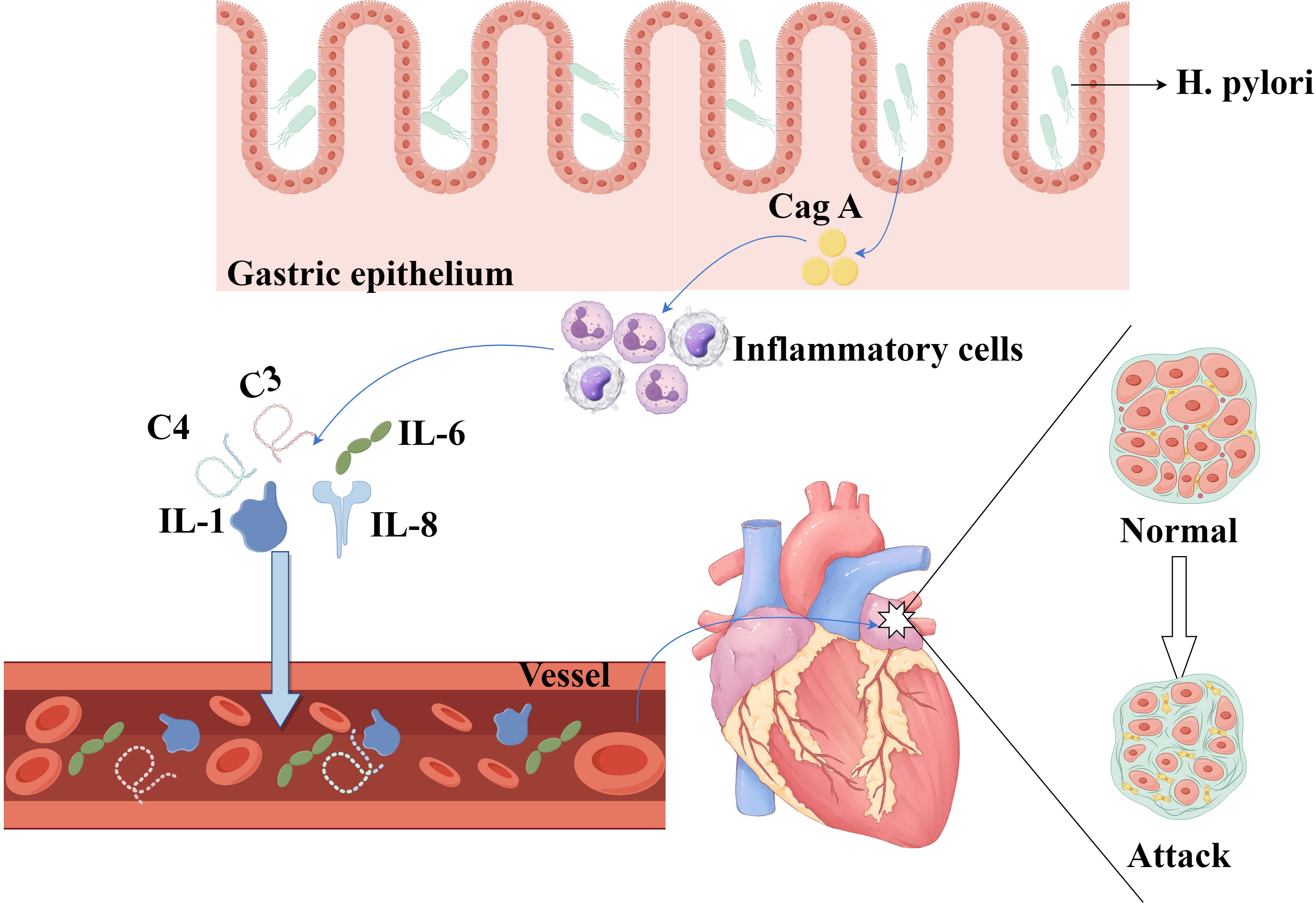 Fig. 2.
Fig. 2.
Inflammatory mechanism of H. pylori-induced AF. The complex inflammatory pathways implicated in H. pylori-induced AF highlight the roles of Cag A and inflammatory mediators, such as IL-1, IL-6, IL-8, and complement system components. IL, interleukin. The figure is created by Figdraw.
H. pylori infection can disrupt lipid metabolism by generating lipid
peroxides and low-density lipoprotein (LDL), which may initiate and accelerate
the development of atherosclerosis, thereby contributing to the pathogenesis of
AF [18, 55]. Notably, as we will explore further, the gut microbiota modulates
these processes and may act as a mediator in establishing the dyslipidemic
profile observed in patients with H. pylori infection. In support of
this hypothesis, antibiotic treatment aimed at eradicating H. pylori has
been shown to reduce total and LDL cholesterol levels and increase high-density lipoprotein cholesterol
(HDL-C) levels [46]. In addition, studies have
shown that this process is initiated by chronic inflammation caused by H.
pylori and is mediated by the inflammatory mediators IL-1, IL-6, and
TNF-
Studies have suggested that H. pylori infection may disrupt the absorption of essential nutrients such as vitamin B6, vitamin B12, and folic acid, leading to their deficiency and affecting the normal methylation of homocysteine. This disruption can result in elevated serum homocysteine levels, which may subsequently cause endothelial damage, thrombosis, and atherosclerosis, thus promoting the development and progression of AF [18, 58, 59]. Furthermore, H. pylori infection might lead to metabolic syndrome, triggering AF [49, 60, 61], a process that could be associated with the action of galectin-3 [15].
H. pylori acts both as an inflammatory agent and an immune regulator. H. pylori infection changes antibody profiles and immune system responses. One notable immune response involves the creation of antibodies against gastric epithelial hydrogen potassium ATPase, which shares structural similarity with the sodium‒potassium ATPase found in cardiac cells, both of which feature a 35 kDa glycoprotein component. Antibodies produced against the hydrogen potassium ATPase during H. pylori infection can inadvertently target the cardiac sodium–potassium ATPase, leading to cardiac cell damage and excessive intracellular sodium accumulation [17].
This increase in intracellular sodium can increase the sarcoplasmic reticulum (SR) Ca2+ load due to impaired Ca2+ extrusion via Na+/Ca2+ exchanger type-1 (NCX1), which is typically inhibited in conditions such as those induced by cardiac glycosides. An overload of SR Ca2+ can also trigger L-type Ca2+-current-dependent triggered Ca2+ waves (TCW), increased SR Ca2+ leakage, and spontaneous SR Ca2+-release events (SCaEs). These SCaEs and TCWs activate a transient inward current mediated by the Na+/Ca2+ exchanger (NCX), resulting in delayed after-depolarizations (DADs) and early after-depolarizations (EADs). Such ectopic activities mediated by DADs and EADs lay an essential groundwork for the occurrence and development of AF [28, 62, 63, 64, 65], as illustrated in Fig. 3.
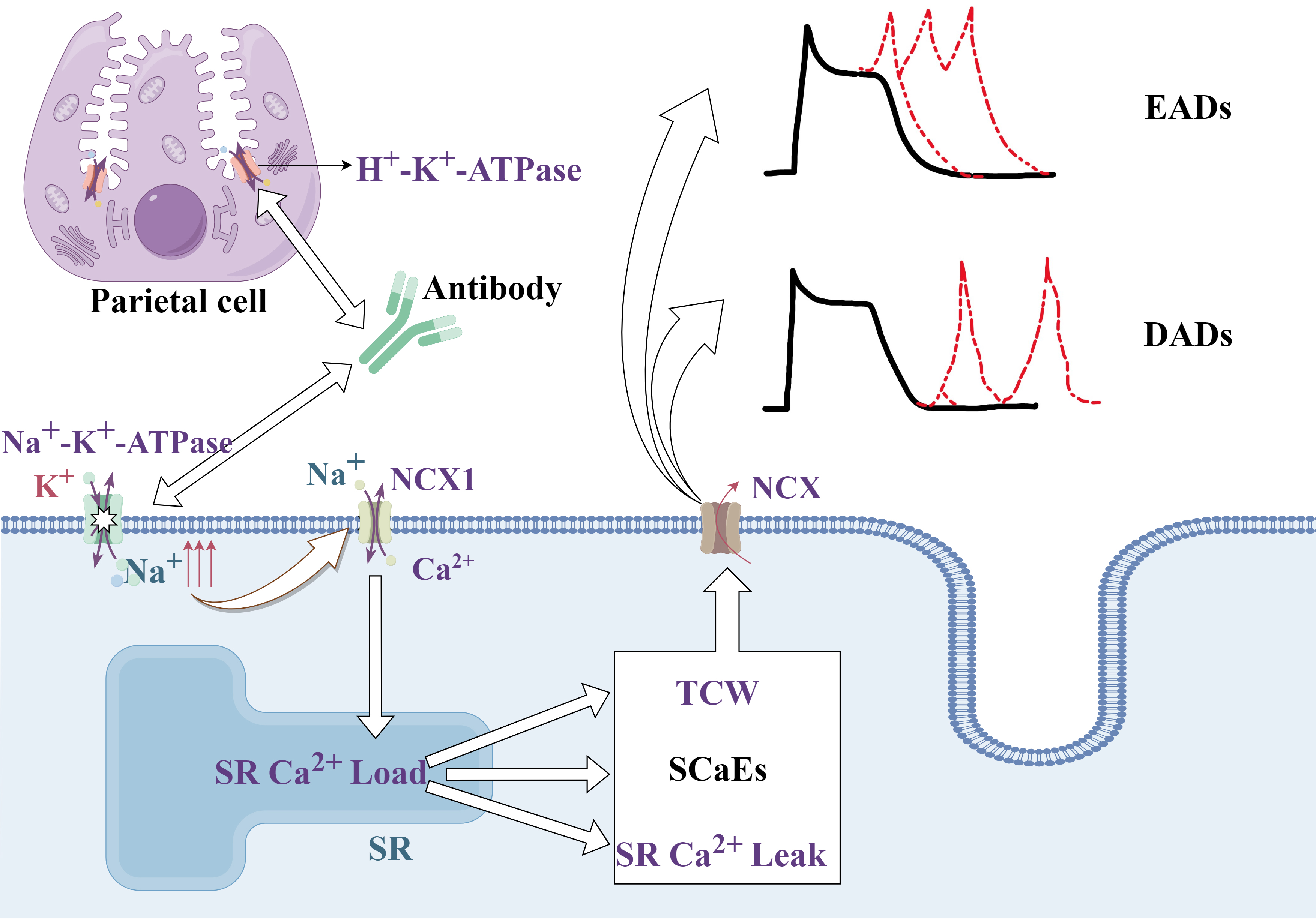 Fig. 3.
Fig. 3.
Mechanism through which antibodies produced by H. pylori lead to AF. The pathway through which antibodies related to H. pylori infection can lead to atrial fibrillation, emphasizing the role of altered ion handling and ectopic activity in cardiac cells, which contributes to arrhythmogenesis. EADs, early afterdepolarizations; DADs, delayed after-depolarizations; NCX, Na+/Ca2+ exchanger; TCW, triggered Ca2+ waves; SCaEs, SR Ca2+-release events; SR, sarcoplasmic reticulum. The figure is created by Figdraw.
In the immediate aftermath of cardiac injury, T helper 1 (Th1) cells predominate
within the myocardium and are known for their antifibrotic functions by releasing
mediators that counteract the profibrotic effects of transforming growth
factor-beta (TGF-
By further compounding this dynamic, the virulence factor vacuolating cytotoxin
A (Vac A) from H. pylori has been shown to block T cell proliferation
effectively by inducing G1/S cell cycle arrest. This disruption can shift the Th
cell balance from Th1 toward Th2, thereby suppressing Th1 activity and
potentially impairing the function of regulatory T cells. Such immunological
shifts could increase the risk of AF [27], as illustrated in Fig. 4.
Additionally, the increase in TNF-
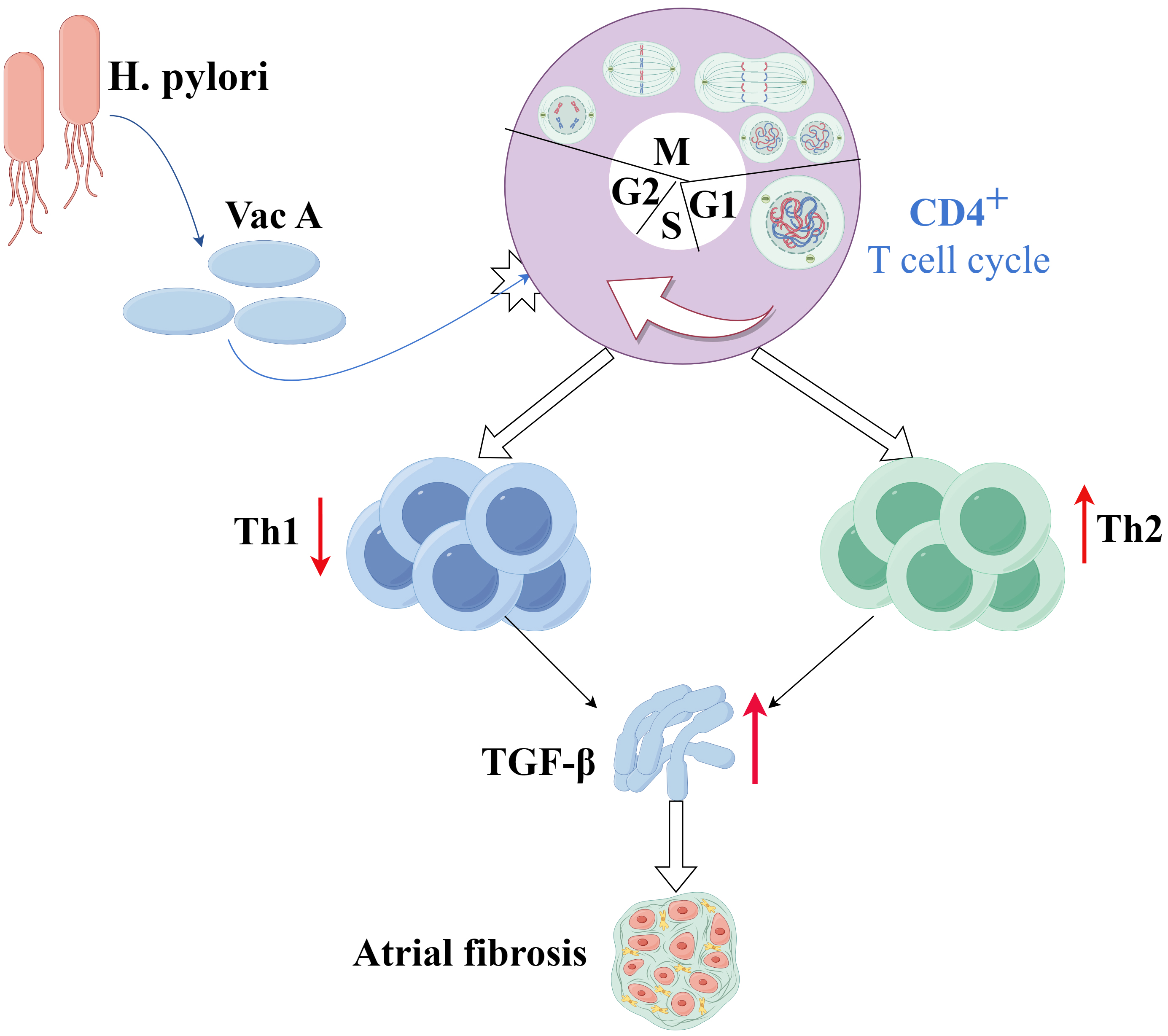 Fig. 4.
Fig. 4.
Mechanism through which H. pylori affects the Th1/Th2 balance that leads to AF. Details of the complex interplay between H. pylori infection and immune cell dynamics within the myocardium highlight how changes in the Th1/Th2 balance due to Vac A may lead to increased fibrosis and subsequent atrial fibrillation. Th1, T helper 1 cell; Th2, T helper 2 cell; Vac A, vacuolating cytotoxin A. The figure is created by Figdraw.
The gut‒brain axis (GBA) forms a multifaceted network in which the central nervous system (CNS) and the enteric nervous system (ENS) engage in two-way communication via neural, hormonal, metabolic, and immune pathways [67, 68, 69]. The concept of this axis has been expanded to include the microbiota‒gut‒brain axis, recognizing the vital influence of the gut microbiota on functional brain‒gut interactions and their contribution to the onset of diverse diseases [70].
The brain is a pivotal hub in this dynamic network, managing and disseminating information across the enteric, sympathetic, and autonomic nervous systems [22, 68]. H. pylori modifies the gut microbial landscape by altering factors such as gastric acidity, immune responses, and the production of antimicrobial peptides and virulence factors. These changes are crucial in shaping gastric and systemic health conditions [71].
H. pylori may disrupt the gut microbiota, thereby causing imbalances in the gut‒brain axis, which could lead to dysregulation of the autonomic nervous system. This dysregulation is implicated in the onset and progression of atrial fibrillation, suggesting a novel pathway through which H. pylori influences cardiac function indirectly via its effects on the gut microbiota [34, 71, 72, 73, 74].
Electrocardiograms (ECGs) and echocardiograms form the primary diagnostic tools for AF and are essential for visualizing the electrical and structural activity of the heart [65]. Although the specific mechanisms underlying AF are not fully understood, and the pathways through which H. pylori might lead to AF are still under investigation, current research suggests potential biomarkers that could assist in diagnosing and predicting AF progression [14].
A study revealed a causal relationship between IL-6 and the occurrence of postoperative AF, which is caused by effects on calcium currents and the stimulation of atrial fibrosis [75, 76, 77]. Another study revealed that IL-17A can represent changes in cardiac fibrosis and left heart function, and it can serve as an independent diagnostic factor to determine the probability of AF recurrence within the first 3 months after catheter ablation [78].
Inflammation plays a central role in the mechanisms through which H. pylori is hypothesized to influence AF. These findings suggest the utility of inflammatory markers such as CRP, IL-1, and IL-6 in exploring the etiology and progression of AF and distinguishing between different AF types. However, these markers are not specific to AF and are primarily indicative rather than diagnostic [79].
Additionally, the serum level of homocysteine, an inflammatory marker, may serve as a valuable prognostic indicator for patients with AF [58]. Furthermore, research has indicated that galectin-3, which is related to H. pylori, might serve as a potential auxiliary diagnostic marker for AF [15]. Moreover, although highly specific, antibodies such as anti-Cag A and anti-Vac A have an uncertain correlation with AF, limiting their clinical diagnostic value [1, 80]. While these biomarkers offer new perspectives for the diagnosis and management of AF, their application must be considered within the overall clinical context of the patient, and their effectiveness and reliability need further validation in future studies.
The fundamental principles of AF treatment aim to eliminate predisposing
factors, restore and maintain sinus rhythm, control the ventricular rate, and
prevent embolic complications and the recurrence of AF. Treatment modalities are
diverse and include pharmacological, electrical, and surgical interventions.
Meanwhile, although there is no direct evidence that eradicating H.
pylori can treat AF, the potential link between the two cannot be overlooked in
managing this type of arrhythmia.
Furthermore, although no clinical trials have
directly studied the benefits and possible risks of eradicating H.
pylori in the treatment of AF, the potential link between the two cannot be
ignored when treating this type of arrhythmia. Theoretically, strategies that
suppress inflammation, reduce the production of autoantibodies, adjust metabolic
pathways, and restore balance to the gut microbiota might improve the progression
of AF [16, 55, 79, 80]. For example, a study suggested that landiolol can reduce
the recurrence of AF after esophageal surgery by lowering IL-6 levels [81]. Other
studies have shown that statins can reduce CRP levels and independently decrease
the risk of AF recurrence. Moreover, statins can control blood lipids and
inflammatory factors such as IL-1, IL-6, and TNF-
Nonetheless, these issues warrant additional investigation and research. The precise effects of H. pylori infection on the risk of gastrointestinal bleeding in patients receiving anticoagulation therapy remain unclear, necessitating further studies to fully comprehend the interactions between these two factors [91, 92].
Additionally, H. pylori infection and anticoagulant treatment may increase the incidence of iron deficiency anemia in female patients with AF, possibly because H. pylori affects iron absorption and anticoagulation therapy, leading to increased menstrual flow [93, 94, 95, 96]. Notably, the use of antibiotics such as clarithromycin could lead to digoxin toxicity, thereby affecting the AF treatment outcome; thus, medication interactions should be carefully managed to avoid exacerbating the condition [97, 98].
Despite studies suggesting that eradicating H. pylori may benefit the treatment of AF, given the potential drug interactions, treatment outcomes, and economic factors, there remains insufficient evidence to support the necessity of H. pylori eradication therapy in all AF patients. However, when AF patients infected with H. pylori experience high levels of inflammatory factors, abnormal blood lipids, and corresponding antibodies, it is beneficial to correct the possible sources of these risk factors and eradicate H. pylori. Therefore, extensive clinical research is required to explore this topic further.
Chronic inflammation induced by H. pylori is closely linked to persistent and permanent AF onset and progression. Therefore, monitoring inflammation-related biomarkers, such as homocysteine levels, may be instrumental in predicting the severity and treatment outcomes of AF patients [58]. Some studies predict that adding the inflammatory factors IL-6 and CRP to the CHA2DS2-VASc risk score can improve its prognostic accuracy; however, excessive use of these inflammatory indicators in risk assessment can lead to incorrect anticoagulant use [39].
Additionally, using proton pump inhibitors may help reduce the recurrence of AF
[37]. In contrast, low TGF-
Moreover, H. pylori infection is considered a potential trigger for thrombogenesis, which could accelerate the formation of AF-related thrombi and even lead to myocardial infarction after catheter ablation. Among the antibodies produced against H. pylori by the human body, antibodies targeting H. pylori Vac A are recognized as the only causal factor linked to an increased risk of stroke, with CRP possibly facilitating this relationship. Therefore, targeting the CRP signaling pathway could lower the risk of stroke in patients infected with Vac A-positive strains of H. pylori [99, 100, 101]. Meanwhile, further research has indicated that eradicating H. pylori might decrease stroke risk in AF patients [99].
Additionally, the association of H. pylori with various cardiovascular diseases, such as coronary artery disease, heart failure, and other arrhythmias, could also influence the prognosis of AF patients, potentially leading to severe adverse outcomes [27, 102, 103, 104]. Therefore, the prognosis of AF patients is influenced not only by traditional cardiovascular risk factors but also by the potential effects of H. pylori infection. These findings suggest that considering the impact of H. pylori could be beneficial in managing AF patients, especially when assessing long-term prognosis and recurrence risk. However, controversy and limitations remain regarding the study of H. pylori in the prognosis of AF, meaning targeted research is needed to verify its true role.
Research has revealed a potential association between these two factors, suggesting that H. pylori may influence the onset and progression of AF through multiple mechanisms, including inflammation, metabolic dysregulation, immune responses, and changes in the gut microbiota. These mechanisms provide crucial insights into the treatment and prognosis of AF.
However, the causal relationship between H. pylori and AF remains unclear, and some studies have even failed to identify a definitive correlation. This inconsistency in research findings indicates that our current understanding remains incomplete and necessitates further clinical and experimental studies to explore the precise relationship between H. pylori and AF in depth. Hence, future research should focus on expanding sample sizes, increasing the number of randomized controlled trials, and conducting prospective studies to explore the interaction mechanism between H. pylori and AF. In addition, clinical studies can be performed to evaluate the potential benefits of H. pylori in improving the health outcomes of patients with AF.
In conclusion, while current evidence suggests a possible link between H. pylori and AF, research in this area is still in its infancy. Thus, comprehensive assessment and understanding of this complex relationship are crucial for developing effective treatment strategies and enhancing patient quality of life. Overall, exploring this topic promises to yield insights that could lead to significant advancements in managing both conditions.
AF, atrial fibrillation; C3, complement 3; C4, complement 4; CNS, central nervous system; DADs, delayed after-depolarizations; EADs, early after-depolarizations; ECGs, electrocardiograms; ENS, enteric nervous system; GBA, Gut-brain axis; H. pylori, Helicobacter pylori; HCY, homocysteine; HDL-C, high-density lipoprotein cholesterol; IL, interleukin; LDL, low density lipoprotein; MALT, mucosa-associated lymphoid tissue; NCX1, Na+/Ca2+ exchanger type-1; NCX, Na+/Ca2+ exchanger; SR, Sarcoplasmic reticulum; SCaEs, spontaneous SR Ca2+-release events; Th1, T helper 1 cell; Th2, T helper 2 cell; TCWs, Triggered Ca2+ Waves; TGF-
WTF, YCX and MXC participated in the conception, writing, and revision of the review. QML and SHZ made substantial contributions to the conception and design of the work, provided critical feedback, and assisted in the revision process. All authors have contributed to the editing and revision of the manuscript. All authors have read and approved the final manuscript. All authors fully participated in this work and agreed to take responsibility for all aspects of the work.
Not applicable.
We would like to express our gratitude to all those who helped us during the writing of the manuscript, as well as to all peer reviewers and their valuable opinions and suggestions.
This paper was supported by the Natural Science Foundation of Hunan Province, China (2023JJ30791).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




