1 Department of Obstetrics and Gynecology, Wu Yi Maternal & Child Care Hospital, 321200 Jinhua, Zhejiang, China
2 Physical Examination Center, Traditional Chinese Medicine Hospital of Wuyi, 321200 Jinhua, Zhejiang, China
†These authors contributed equally.
Abstract
Low-grade squamous intraepithelial lesions (LSIL) are early pathological changes associated with cervical cancer, exhibiting varying progression rates. The risk factors for progression from LSIL to high-grade squamous intraepithelial lesions (HSIL) and the development of a predictive model for this progression have not been thoroughly investigated.
This single-center, retrospective study followed 122 patients diagnosed with LSIL, who were monitored without treatment, over a 3-year period. Clinical and pathological data were collected, and univariate and multivariate logistic regression analyses were performed to identify independent risk factors for progression to HSIL. A predictive model was developed based on these factors and validated using both training and validation cohorts.
Of the 122 patients, 11 (9.01%) progressed to HSIL within 3 years. Univariate analysis revealed that persistent high-risk human papillomavirus (HPV) infection, HPV16/18 genotype, and persistent or recurrent symptoms significantly increased the risk of progression to HSIL. Multivariate analysis confirmed that persistent high-risk HPV infection (odds ratio (OR) = 7.67, p = 0.027) and persistent or recurrent symptoms (OR = 21.07, p < 0.001) were independent risk factors for progression to HSIL. A nomogram incorporating these factors demonstrated excellent predictive performance in both the training cohort (area under the curve (AUC) = 0.92) and the validation cohort (AUC = 0.88). Calibration and decision curve analysis further validated the model’s reliability and clinical applicability.
Persistent high-risk HPV infection and recurrent symptoms are key predictors of LSIL progression to HSIL. The developed predictive model can assist clinicians in identifying high-risk patients for early intervention, thus improving clinical management and optimizing healthcare resource utilization.
Keywords
- low-grade squamous intraepithelial lesions (LSIL)
- high-grade squamous intraepithelial lesions (HSIL)
- HPV infection
- persistent infection
- recurrent symptoms
- predictive model
Cervical squamous intraepithelial lesions (SIL) are identified as early pathological changes that may develop into cervical cancer [1]. According to the 2014 World Health Organization (WHO) classification, these lesions are categorized into low-grade (LSIL) and high-grade (HSIL) [2, 3]. Globally, LSIL accounts for approximately 1.7% of all cervical cytology results, while HSIL comprises about 0.3% [4, 5]. Although many LSIL cases may regress spontaneously, a subset can progress to HSIL and eventually to cervical cancer. The progression from LSIL to carcinoma in situ or invasive cervical cancer typically spans 5 to 10 years [6]. Once progression to cervical cancer occurs, it poses a significant threat to women’s health. Cervical cancer ranks as the fourth most common cancer among women worldwide, leading to substantial morbidity and mortality [7]. It can result in compromised fertility and diminished quality of life. What’s more, epidemiological data indicate an annual incidence of approximately 1.39 cases of cervical precancerous lesions per 1000 women [8]. LSIL is more prevalent among women under 30, whereas the incidence of HSIL increases with age, particularly in those over 40 [9]. These patterns underscore the critical importance of early screening and monitoring to identify high-risk individuals, thereby reducing the incidence of cervical cancer [10].
Currently, there is a significant difference in the treatment strategies for LSIL and HSIL. LSIL patients are typically associated with human papillomavirus (HPV) infection, which may resolve spontaneously. Therefore, conservative management, primarily involving regular follow-up and monitoring, is generally employed [11]. In contrast, HSIL patients have a higher risk of progressing to cervical cancer and require more aggressive interventions, such as conization, cryotherapy, or laser treatment [12]. Clinically, it has been observed that some LSIL patients may be overtreated, leading to unnecessary medical interventions and potential complications [13]. Over-treatment not only increases the psychological and financial burden on patients but may also have adverse effects on fertility. Given that LSIL can resolve over time, monitoring rather than invasive interventions is usually recommended, especially for patients who wish to conceive. On the other hand, monitoring of HSIL relies on cytological screening, and the success of early detection depends on the effectiveness of screening techniques and management of high-risk populations [14]. However, some high-risk women may not receive adequate follow-up after screening, allowing the lesions to progress [15]. Therefore, accurately identifying individuals at high risk for progression and providing preventive advice and lifestyle modification strategies are crucial for improving patient outcomes.
Based on this, the present study focused on patients diagnosed with LSIL at our institution who underwent monitoring only, without receiving treatment. These patients were observed over a 3-year period to assess whether they progressed to HSIL or malignant tumors. Clinical and pathological data were collected and analyzed to develop a predictive model for evaluating the risk of progression. This model aims to assist clinicians in identifying high-risk patients and implementing timely interventions. For low-risk patients, enhanced monitoring can be adopted to avoid unnecessary treatment, thereby facilitating individualized clinical management and optimizing the utilization of healthcare resources.
This study is a single-center, retrospective study aimed at observing the progression of patients with LSIL who received monitoring only and no treatment, from an initial diagnosis to high-grade lesions or malignant tumors within 3 years. The study participants were sourced from Wu Yi Maternal & Child Care Hospital, with the initial diagnosis occurring from January 1, 2015, to October 31, 2021, and the observation period lasted for 3 years, until October 31, 2024. The study was approved by the Wu Yi Maternal & Child Care Hospital Ethics Committee, with ethics approval number 2024112201. All procedures followed the principles outlined in the Declaration of Helsinki. Informed consent was obtained from all participants, and the research data were rigorously reviewed twice before use to ensure the protection of patient privacy.
Diagnostic Criteria: Various grades of cervical squamous epithelial lesions, such as HSIL or LSIL, were diagnosed based on the diagnostic criteria specified in the 2014 WHO Classification of Tumors of the Female Reproductive Organs [16]: (1) LSIL: Identified by mild abnormalities in cervical epithelial cells during cytological or histological examinations, including slightly enlarged nuclei, a mild increase in the nucleus-to-cytoplasm ratio, increased nuclear chromatin, and possible vacuolization (clear cells). The lesion is usually confined to the lower third of the epithelium. (2) HSIL: Significant abnormalities in cells, including markedly enlarged nuclei, a significantly increased nucleus-to-cytoplasm ratio, irregular nuclear morphology, highly condensed chromatin, and possible abnormal mitotic figures. The lesion involves the middle to full thickness of the epithelium. (3) Cervical cancer: Clinically, patients may present with abnormal vaginal bleeding (e.g., postcoital, intermenstrual, or postmenopausal), unusual vaginal discharge, or pelvic pain. Cytological screening, such as abnormal Pap test results, often indicates squamous cell carcinoma or glandular abnormalities. Diagnosis is confirmed by histopathological examination of biopsy specimens, with squamous cell carcinoma and adenocarcinoma being the most common subtypes. Imaging techniques like magnetic resonance imaging (MRI) or computed tomography (CT) are used to assess tumor size, local invasion, and distant metastases, while high-risk HPV DNA or RNA testing provides additional confirmation of malignancy [17].
Inclusion Criteria: (1) Initial diagnosis of LSIL through liquid-based cytology, colposcopy, or biopsy; (2) Diagnosis date between January 1, 2015, and October 31, 2021; (3) Female patients; (4) No treatment received after diagnosis (including cryotherapy, laser therapy, conization, or drug treatment); (5) Regular cervical cytology and HPV testing after initial LSIL diagnosis; (6) Complete medical records and follow-up data; (7) Patients signed informed consent allowing the use of their medical data for research purposes.
Exclusion Criteria: (1) Any treatment for LSIL during follow-up; (2) A history of HSIL or cervical cancer; (3) Coexisting immune-related conditions, such as immunodeficiency, autoimmune diseases, or long-term use of immunosuppressive medications; (4) Missing critical data during follow-up, such as missing test results, making outcome determination impossible; (5) Coexisting reproductive tract infections or diseases, such as pelvic inflammatory disease or malignancies; (6) Patients who refused to allow the use of their data for research.
The clinical and pathological factors considered in this study included age
(16–30 years, 31–45 years,
Among the eligible participants in the hospital’s records, the majority had documentation of HPV testing and cervical cytology examination every 3–6 months during the 3-year observation period. The time of HPV progression was defined as the duration from the initial diagnosis to the date of the positive HPV test. Patients who tested negative for HPV during months 10–14 of the third year were recorded as having a negative HPV test result. HPV genotyping was performed using PCR-reverse dot blot (PCR-RDB) method. This technique detects 17 high-risk and 6 low-risk HPV subtypes, including types like HPV16 and HPV18. The specific HPV subtype is determined based on the location of color spots on the membrane. An internal control (IC) is included on the membrane; if only the IC shows a color signal, the result is negative. If any HPV subtype site other than the IC shows a color signal, the result is considered positive. Patients with persistent high-risk HPV infection or abnormal colposcopy findings were subjected to further biopsy evaluations. These biopsies were conducted to confirm whether the lesions had progressed to a higher grade or cervical cancer, serving as the study’s primary endpoint. If any screening during the 3-year follow-up period identified progression to a higher-grade lesion or cervical cancer, the outcome for that patient was recorded as “progressed”. The follow-up outcomes for all participants were determined exclusively based on the diagnostic results obtained from the institution conducting the study. This standardized follow-up protocol ensured consistency in data collection and the reliability of the outcome measures.
Prior to conducting statistical analyses, the normality of the data was assessed
using the Shapiro-Wilk test. For normally distributed quantitative data, the
results are presented as mean
A predictive model was constructed using significant factors identified from multivariate logistic regression analysis. To assess the model’s prognostic accuracy, the concordance index (C-index) and calibration curve were used. Additionally, the receiver operating characteristic (ROC) curve was employed to evaluate the predictive performance of the nomogram for progression within 3 years. The nomogram, C-index, calibration curve, and ROC curve were constructed using R version 4.2.1 (https://www.r-project.org/) in R Studio. The ROC curve was used to describe the sensitivity and specificity of the constructed nomogram, while the calibration curve was used to assess and validate its accuracy. The dataset was split into training and validation sets in a 6:4 ratio. The predictive model was validated using 500 bootstrap samples to prevent overfitting and obtain relatively unbiased estimates.
In this study, 122 LSIL patients who only underwent monitoring were followed up
for 3 years. The results showed that 11 patients (9.01%) progressed to HSIL
within 3 years (Table 1). Notably, in our study, we did not observe any patients
who progressed to malignancy within the 3-year follow-up period. Chi-square test
results revealed that patients infected with HPV16/18, those with persistent
high-risk HPV infection, and those with persistent or recurrent symptoms were
more likely to progress to HSIL within 3 years (p
| Variables | Total (n = 122) | Progression to HSIL within 3 years | Statistic | p | ||
| No (n = 111) | Yes (n = 11) | |||||
| Age (years), Mean |
33.75 |
33.80 |
33.27 |
t = 0.13 | 0.901 | |
| Number of Lesions, Mean |
2.61 |
2.60 |
2.64 |
t = –0.10 | 0.917 | |
| Lesion Location, n (%) | χ2 = 0.05 | 0.830 | ||||
| Cervical Os | 31 (25.41) | 29 (93.55) | 2 (6.45) | |||
| Cervical Canal | 91 (74.59) | 82 (90.11) | 9 (9.89) | |||
| HPV16/18 Infection, n (%) | χ2 = 5.42 | 0.020 | ||||
| Positive | 59 (48.36) | 50 (84.75) | 9 (15.25) | |||
| Negative | 63 (51.64) | 61 (96.83) | 2 (3.17) | |||
| P16, n (%) | χ2 = 0.29 | 0.591 | ||||
| No | 81 (66.39) | 75 (92.59) | 6 (7.41) | |||
| Yes | 41 (33.61) | 36 (87.80) | 5 (12.20) | |||
| Ki-67, n (%) | χ2 = 0.05 | 0.818 | ||||
| Positive | 46 (37.70) | 41 (89.13) | 5 (10.87) | |||
| Negative | 76 (62.30) | 70 (92.11) | 6 (7.89) | |||
| HPV Infection Type, n (%) | Fisher exact | 0.092 | ||||
| Single Infection | 65 (53.28) | 62 (95.38) | 3 (4.62) | |||
| No Infection | 15 (12.30) | 14 (93.33) | 1 (6.67) | |||
| Mixed Infection | 42 (34.43) | 35 (83.33) | 7 (16.67) | |||
| Abnormal Uterine Bleeding, n (%) | χ2 = 3.53 | 0.060 | ||||
| No | 90 (73.77) | 85 (94.44) | 5 (5.56) | |||
| Yes | 32 (26.23) | 26 (81.25) | 6 (18.75) | |||
| Marital Status, n (%) | Fisher exact | 0.609 | ||||
| Single/Unmarried | 58 (47.54) | 52 (89.66) | 6 (10.34) | |||
| Married | 38 (31.15) | 36 (94.74) | 2 (5.26) | |||
| Unknown/Other | 19 (15.57) | 17 (89.47) | 2 (10.53) | |||
| Divorced | 7 (5.74) | 6 (85.71) | 1 (14.29) | |||
| Residence, n (%) | χ2 = 0.00 | 1.000 | ||||
| Urban | 82 (67.21) | 75 (91.46) | 7 (8.54) | |||
| Rural | 40 (32.79) | 36 (90.00) | 4 (10.00) | |||
| Family History of Cancer, n (%) | χ2 = 1.22 | 0.269 | ||||
| No | 107 (87.70) | 99 (92.52) | 8 (7.48) | |||
| Yes | 15 (12.30) | 12 (80.00) | 3 (20.00) | |||
| Vaginal Discharge, n (%) | Fisher exact | 1.000 | ||||
| Increased | 40 (32.79) | 36 (90.00) | 4 (10.00) | |||
| Decreased | 17 (13.93) | 16 (94.12) | 1 (5.88) | |||
| Normal | 65 (53.28) | 59 (90.77) | 6 (9.23) | |||
| Transformation Zone Typing, n (%) | χ2 = 1.46 | 0.228 | ||||
| Type I+II | 81 (66.39) | 76 (93.83) | 5 (6.17) | |||
| Type III | 41 (33.61) | 35 (85.37) | 6 (14.63) | |||
| Number of Pregnancies, n (%) | χ2 = 0.00 | 1.000 | ||||
| 48 (39.34) | 44 (91.67) | 4 (8.33) | ||||
| 74 (60.66) | 67 (90.54) | 7 (9.46) | ||||
| Menopausal Status, n (%) | Fisher exact | 0.259 | ||||
| No | 111 (90.98) | 102 (91.89) | 9 (8.11) | |||
| Yes | 11 (9.02) | 9 (81.82) | 2 (18.18) | |||
| Use of Intrauterine Device, n (%) | χ2 = 0.26 | 0.612 | ||||
| No | 101 (82.79) | 93 (92.08) | 8 (7.92) | |||
| Yes | 21 (17.21) | 18 (85.71) | 3 (14.29) | |||
| Persistent High-Risk HPV Infection, n (%) | χ2 = 6.25 | 0.012 | ||||
| No | 71 (58.20) | 69 (97.18) | 2 (2.82) | |||
| Yes | 51 (41.80) | 42 (82.35) | 9 (17.65) | |||
| Persistent or Recurrent Symptoms, n (%) | χ2 = 26.98 | |||||
| No | 108 (88.52) | 104 (96.30) | 4 (3.70) | |||
| Yes | 14 (11.48) | 7 (50.00) | 7 (50.00) | |||
LSIL, low-grade squamous intraepithelial lesions; HSIL, high-grade squamous intraepithelial lesions; HPV, human papillomavirus.
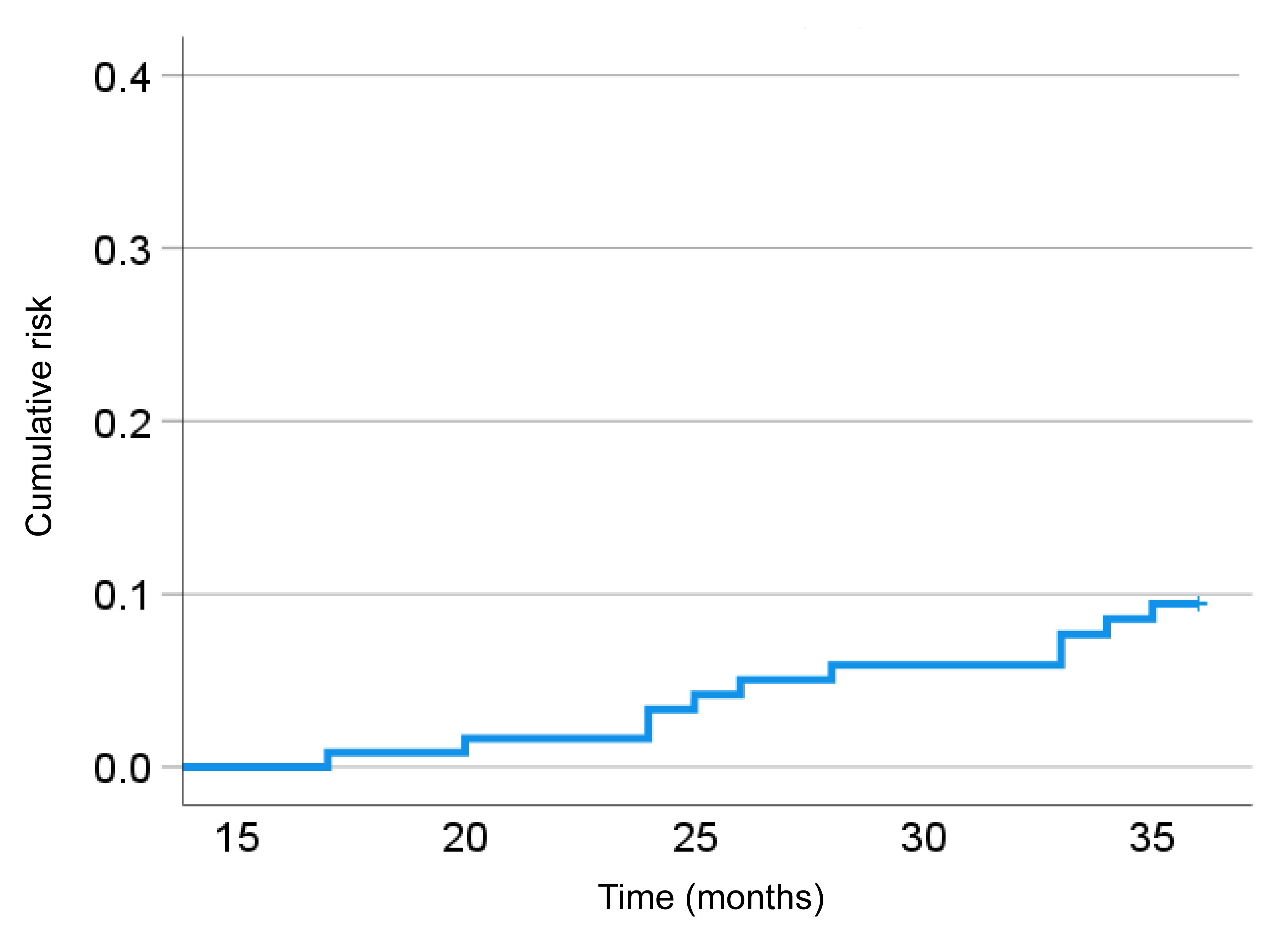 Fig. 1.
Fig. 1.
The cumulative risk curve for progression to HSIL within 3 years in all patients.
As previously described, we randomly divided the complete dataset of 122
patients into a training set and a validation set at a 6:4 ratio. The general
characteristics and clinicopathological factors of the training set are presented
in Table 2, which revealed statistically significant differences in factors such
as HPV16/18 infection type, presence of persistent HPV infection, and occurrence
of recurrent persistent symptoms. Additionally, there were no statistically
significant differences between the training set and the validation set at
baseline, indicating comparability of the results, which was consistent across
both the overall population dataset and the dataset of patients developed into
HSIL, as shown in Table 3. Univariate analysis revealed that the risk of
progression to HSIL was significantly lower in patients infected with
non-HPV16/18 genotypes (odds ratio (OR) = 0.18, 95% CI: 0.04~0.88, p
= 0.034) (see Table 4). Conversely, patients with persistent high-risk HPV
infection (OR = 7.39, 95% CI: 1.52~35.87, p = 0.013)
and persistent or recurrent symptoms (OR = 26.00, 95% CI:
6.11~110.55, p
| Variables | Total (n = 73) | Progression to HSIL within 3 years | Statistic | p | ||
| No (n = 67) | Yes (n = 6) | |||||
| Age (years), Mean |
33.75 |
33.69 |
34.36 |
t = –0.22 | 0.827 | |
| Number of Lesions, Mean |
2.61 |
2.60 |
2.64 |
t = –0.10 | 0.917 | |
| Lesion Location, n (%) | χ2 = 0.82 | 0.366 | ||||
| Cervical Os | 56 (76.71) | 50 (74.63) | 6 (100.00) | |||
| Cervical Canal | 17 (23.29) | 17 (25.37) | 0 (0.00) | |||
| HPV Types, n (%) | χ2 = 5.01 | 0.025 | ||||
| HPV16/18 | 35 (47.95) | 29 (43.28) | 6 (100.00) | |||
| Non-HPV16/18 | 38 (52.05) | 38 (56.72) | 0 (0.00) | |||
| P16, n (%) | χ2 = 1.47 | 0.225 | ||||
| No | 47 (64.38) | 45 (67.16) | 2 (33.33) | |||
| Yes | 26 (35.62) | 22 (32.84) | 4 (66.67) | |||
| Ki-67, n (%) | χ2 = 0.01 | 0.919 | ||||
| Positive | 29 (39.73) | 26 (38.81) | 3 (50.00) | |||
| Negative | 44 (60.27) | 41 (61.19) | 3 (50.00) | |||
| HPV Infection Type, n (%) | - | 0.270 | ||||
| Single Infection | 42 (57.53) | 40 (59.70) | 2 (33.33) | |||
| No Infection | 23 (31.51) | 19 (28.36) | 4 (66.67) | |||
| Mixed Infection | 8 (10.96) | 8 (11.94) | 0 (0.00) | |||
| Abnormal Uterine Bleeding, n (%) | χ2 = 3.15 | 0.076 | ||||
| No | 53 (72.60) | 51 (76.12) | 2 (33.33) | |||
| Yes | 20 (27.40) | 16 (23.88) | 4 (66.67) | |||
| Marital Status, n (%) | - | 0.501 | ||||
| Single/Unmarried | 33 (45.21) | 30 (44.78) | 3 (50.00) | |||
| Married | 5 (6.85) | 4 (5.97) | 1 (16.67) | |||
| Unknown/Other | 11 (15.07) | 10 (14.93) | 1 (16.67) | |||
| Divorced | 24 (32.88) | 23 (34.33) | 1 (16.67) | |||
| Residence, n (%) | χ2 = 0.01 | 0.911 | ||||
| Urban | 41 (56.16) | 37 (55.22) | 4 (66.67) | |||
| Rural | 32 (43.84) | 30 (44.78) | 2 (33.33) | |||
| Family History of Cancer, n (%) | - | 0.073 | ||||
| No | 67 (91.78) | 63 (94.03) | 4 (66.67) | |||
| Yes | 6 (8.22) | 4 (5.97) | 2 (33.33) | |||
| Vaginal Discharge, n (%) | - | 0.737 | ||||
| Increased | 24 (32.88) | 22 (32.84) | 2 (33.33) | |||
| Decreased | 12 (16.44) | 12 (17.91) | 0 (0.00) | |||
| Normal | 37 (50.68) | 33 (49.25) | 4 (66.67) | |||
| Transformation Zone Typing, n (%) | χ2 = 0.00 | 1.000 | ||||
| Type I+II | 49 (67.12) | 45 (67.16) | 4 (66.67) | |||
| Type III | 24 (32.88) | 22 (32.84) | 2 (33.33) | |||
| Number of Pregnancies, n (%) | χ2 = 0.00 | 1.000 | ||||
| 26 (35.62) | 24 (35.82) | 2 (33.33) | ||||
| 47 (64.38) | 43 (64.18) | 4 (66.67) | ||||
| Menopausal Status, n (%) | - | 0.357 | ||||
| No | 68 (93.15) | 63 (94.03) | 5 (83.33) | |||
| Yes | 5 (6.85) | 4 (5.97) | 1 (16.67) | |||
| Use of Intrauterine Device (IUD), n (%) | χ2 = 0.23 | 0.631 | ||||
| No | 60 (82.19) | 56 (83.58) | 4 (66.67) | |||
| Yes | 13 (17.81) | 11 (16.42) | 2 (33.33) | |||
| Persistent HPV Infection, n (%) | χ2 = 6.48 | 0.011 | ||||
| No | 42 (57.53) | 42 (62.69) | 0 (0.00) | |||
| Yes | 31 (42.47) | 25 (37.31) | 6 (100.00) | |||
| Persistent or Recurrent Symptoms, n (%) | - | 0.015 | ||||
| No | 65 (89.04) | 62 (92.54) | 3 (50.00) | |||
| Yes | 8 (10.96) | 5 (7.46) | 3 (50.00) | |||
t, t-test;
SD, standard deviation.
| Variables | Overall population dataset | Dataset of patients developed HSIL | |||||||||
| Total (n = 122) | Test (n = 49) | Train (n = 73) | Statistic | p | Total (n = 11) | Test (n = 5) | Train (n = 6) | Statistic | p | ||
| Age (years), Mean |
33.75 |
34.90 |
32.99 |
t = 1.08 | 0.284 | 32.00 (23.50, 42.00) | 41.00 (32.00, 43.00) | 23.50 (18.25, 33.25) | W = 7.001 | 0.177 | |
| Number of Lesions, Mean |
2.61 |
2.49 |
2.68 |
t = –1.07 | 0.285 | 2.00 (2.00, 3.00) | 2.00 (2.00, 2.00) | 3.00 (2.25, 3.00) | Z = 1.261 | 0.209 | |
| Lesion Location, n (%) | χ2 = 0.43 | 0.511 | - | 0.182 | |||||||
| Cervical Os | 91 (74.59) | 35 (71.43) | 56 (76.71) | 9 (81.82) | 3 (60.00) | 6 (100.00) | |||||
| Cervical Canal | 31 (25.41) | 14 (28.57) | 17 (23.29) | 2 (18.18) | 2 (40.00) | 0 (0.00) | |||||
| HPV Types, n (%) | χ2 = 0.01 | 0.911 | - | 0.182 | |||||||
| HPV16/18 | 59 (48.36) | 24 (48.98) | 35 (47.95) | 9 (81.82) | 3 (60.00) | 6 (100.00) | |||||
| Non-HPV16/18 | 63 (51.64) | 25 (51.02) | 38 (52.05) | 2 (18.18) | 2 (40.00) | 0 (0.00) | |||||
| P16, n (%) | χ2 = 0.33 | 0.566 | - | 0.242 | |||||||
| No | 81 (66.39) | 34 (69.39) | 47 (64.38) | 6 (54.55) | 4 (80.00) | 2 (33.33) | |||||
| Yes | 41 (33.61) | 15 (30.61) | 26 (35.62) | 5 (45.45) | 1 (20.00) | 4 (66.67) | |||||
| Ki-67, n (%) | χ2 = 0.32 | 0.574 | - | 1.000 | |||||||
| Positive | 46 (37.70) | 17 (34.69) | 29 (39.73) | 5 (45.45) | 2 (40.00) | 3 (50.00) | |||||
| Negative | 76 (62.30) | 32 (65.31) | 44 (60.27) | 6 (54.55) | 3 (60.00) | 3 (50.00) | |||||
| HPV Infection Type, n (%) | χ2 = 1.33 | 0.514 | - | 1.000 | |||||||
| Single Infection | 65 (53.28) | 23 (46.94) | 42 (57.53) | 3 (27.27) | 1 (20.00) | 2 (33.33) | |||||
| No Infection | 42 (34.43) | 19 (38.78) | 23 (31.51) | 7 (63.64) | 3 (60.00) | 4 (66.67) | |||||
| Mixed Infection | 15 (12.30) | 7 (14.29) | 8 (10.96) | 1 (9.09) | 1 (20.00) | 0 (0.00) | |||||
| Abnormal Uterine Bleeding, n (%) | χ2 = 0.02 | 0.887 | - | 1.000 | |||||||
| No | 88 (72.13) | 35 (71.43) | 53 (72.60) | 3 (27.27) | 1 (20.00) | 2 (33.33) | |||||
| Yes | 34 (27.87) | 14 (28.57) | 20 (27.40) | 8 (72.73) | 4 (80.00) | 4 (66.67) | |||||
| Marital Status, n (%) | - | 0.873 | - | 1.000 | |||||||
| Single/Unmarried | 58 (47.54) | 25 (51.02) | 33 (45.21) | 6 (54.55) | 3 (60.00) | 3 (50.00) | |||||
| Married | 7 (5.74) | 2 (4.08) | 5 (6.85) | 1 (9.09) | 0 (0.00) | 1 (16.67) | |||||
| Unknown/Other | 19 (15.57) | 8 (16.33) | 11 (15.07) | 2 (18.18) | 1 (20.00) | 1 (16.67) | |||||
| Divorced | 38 (31.15) | 14 (28.57) | 24 (32.88) | 2 (18.18) | 1 (20.00) | 1 (16.67) | |||||
| Residence, n (%) | χ2 = 0.14 | 0.713 | - | 1.000 | |||||||
| Urban | 82 (67.21) | 32 (65.31) | 50 (68.49) | 7 (63.64) | 3 (60.00) | 4 (66.67) | |||||
| Rural | 40 (32.79) | 17 (34.69) | 23 (31.51) | 4 (36.36) | 2 (40.00) | 2 (33.33) | |||||
| Family History of Cancer, n (%) | χ2 = 0.28 | 0.595 | - | 1.000 | |||||||
| No | 114 (93.44) | 47 (95.92) | 67 (91.78) | 8 (72.73) | 4 (80.00) | 4 (66.67) | |||||
| Yes | 8 (6.56) | 2 (4.08) | 6 (8.22) | 3 (27.27) | 1 (20.00) | 2 (33.33) | |||||
| Vaginal Discharge, n (%) | χ2 = 1.05 | 0.592 | - | 0.740 | |||||||
| Increased | 40 (32.79) | 16 (32.65) | 24 (32.88) | 4 (36.36) | 2 (40.00) | 2 (33.33) | |||||
| Decreased | 17 (13.93) | 5 (10.20) | 12 (16.44) | 1 (9.09) | 1 (20.00) | 0 (0.00) | |||||
| Normal | 65 (53.28) | 28 (57.14) | 37 (50.68) | 6 (54.55) | 2 (40.00) | 4 (66.67) | |||||
| Transformation Zone Typing, n (%) | χ2 = 0.04 | 0.835 | - | 0.242 | |||||||
| Type I+II | 81 (66.39) | 32 (65.31) | 49 (67.12) | 5 (45.45) | 1 (20.00) | 4 (66.67) | |||||
| Type III | 41 (33.61) | 17 (34.69) | 24 (32.88) | 6 (54.55) | 4 (80.00) | 2 (33.33) | |||||
| Number of Pregnancies, n (%) | χ2 = 1.06 | 0.304 | - | 1.000 | |||||||
| 48 (39.34) | 22 (44.90) | 26 (35.62) | 4 (36.36) | 2 (40.00) | 2 (33.33) | ||||||
| 74 (60.66) | 27 (55.10) | 47 (64.38) | 7 (63.64) | 3 (60.00) | 4 (66.67) | ||||||
| Menopausal Status, n (%) | χ2 = 0.49 | 0.485 | - | 1.000 | |||||||
| No | 111 (90.98) | 43 (87.76) | 68 (93.15) | 9 (81.82) | 4 (80.00) | 5 (83.33) | |||||
| Yes | 11 (9.02) | 6 (12.24) | 5 (6.85) | 2 (18.18) | 1 (20.00) | 1 (16.67) | |||||
| Use of Intrauterine Device (IUD), n (%) | χ2 = 0.05 | 0.832 | - | 1.000 | |||||||
| No | 101 (82.79) | 41 (83.67) | 60 (82.19) | 8 (72.73) | 4 (80.00) | 4 (66.67) | |||||
| Yes | 21 (17.21) | 8 (16.33) | 13 (17.81) | 3 (27.27) | 1 (20.00) | 2 (33.33) | |||||
| Persistent HPV Infection, n (%) | χ2 = 0.03 | 0.856 | - | 0.182 | |||||||
| No | 71 (58.20) | 29 (59.18) | 42 (57.53) | 2 (18.18) | 2 (40.00) | 0 (0.00) | |||||
| Yes | 51 (41.80) | 20 (40.82) | 31 (42.47) | 9 (81.82) | 3 (60.00) | 6 (100.00) | |||||
| Persistent or Recurrent Symptoms, n (%) | χ2 = 0.05 | 0.827 | - | 0.545 | |||||||
| No | 108 (88.52) | 43 (87.76) | 65 (89.04) | 4 (36.36) | 1 (20.00) | 3 (50.00) | |||||
| Yes | 14 (11.48) | 6 (12.24) | 8 (10.96) | 7 (63.64) | 4 (80.00) | 3 (50.00) | |||||
t, t-test; χ2, Chi-square test; -, Fisher exact; W, Mann-Whitney U test; Z, Z-test.
SD, standard deviation.
| Variables | Univariate Logistic Regression Analysis | Multivariate Logistic Regression Analysis | |||||||||
| S.E | Z | p | Odds ratio (OR) (95% CI) | S.E | Z | p | OR (95% CI) | ||||
| HPV16/18 Infection | |||||||||||
| Positive | 1.00 (Reference) | 1.00 (Reference) | |||||||||
| Negative | –1.7 | 0.8 | –2.12 | 0.034 | 0.18 (0.04~0.88) | –0.71 | 0.94 | –0.75 | 0.454 | 0.49 (0.08~3.13) | |
| Persistent High-Risk HPV Infection | |||||||||||
| No | 1.00 (Reference) | 1.00 (Reference) | |||||||||
| Yes | 2 | 0.81 | 2.48 | 0.013 | 7.39 (1.52~35.87) | 2.04 | 0.92 | 2.22 | 0.027 | 7.67 (1.27~46.36) | |
| Persistent or Recurrent Symptoms | |||||||||||
| No | 1.00 (Reference) | 1.00 (Reference) | |||||||||
| Yes | 3.26 | 0.74 | 4.41 | 26.00 (6.11~110.55) | 3.05 | 0.89 | 3.41 | 21.07 (3.66~121.30) | |||
Based on the results of multivariate logistic regression analysis, a nomogram was developed to predict the progression of LSIL patients to high-grade lesions within 3 years, incorporating two independent risk factors: persistent high-risk HPV infection and persistent or recurrent symptoms (see Fig. 2). To evaluate the model’s performance, we plotted the ROC curve. In the training cohort, the model’s area under the curve (AUC) was 0.92 (95% CI: 0.84–0.99), demonstrating excellent predictive ability. In the validation cohort, the AUC was 0.88 (95% CI: 0.70–1.00), further confirming the model’s reliability (Fig. 2B). The calibration curve (Fig. 2C) showed good consistency between predicted probabilities and actual outcomes, with a Hosmer-Lemeshow test p-value of 0.704, indicating a well-fitting model. Decision curve analysis (DCA) (Fig. 2D) showed that at most high-risk thresholds, the net benefit of using the predictive model for intervention was significantly higher than that of the “all” or “none” strategies.
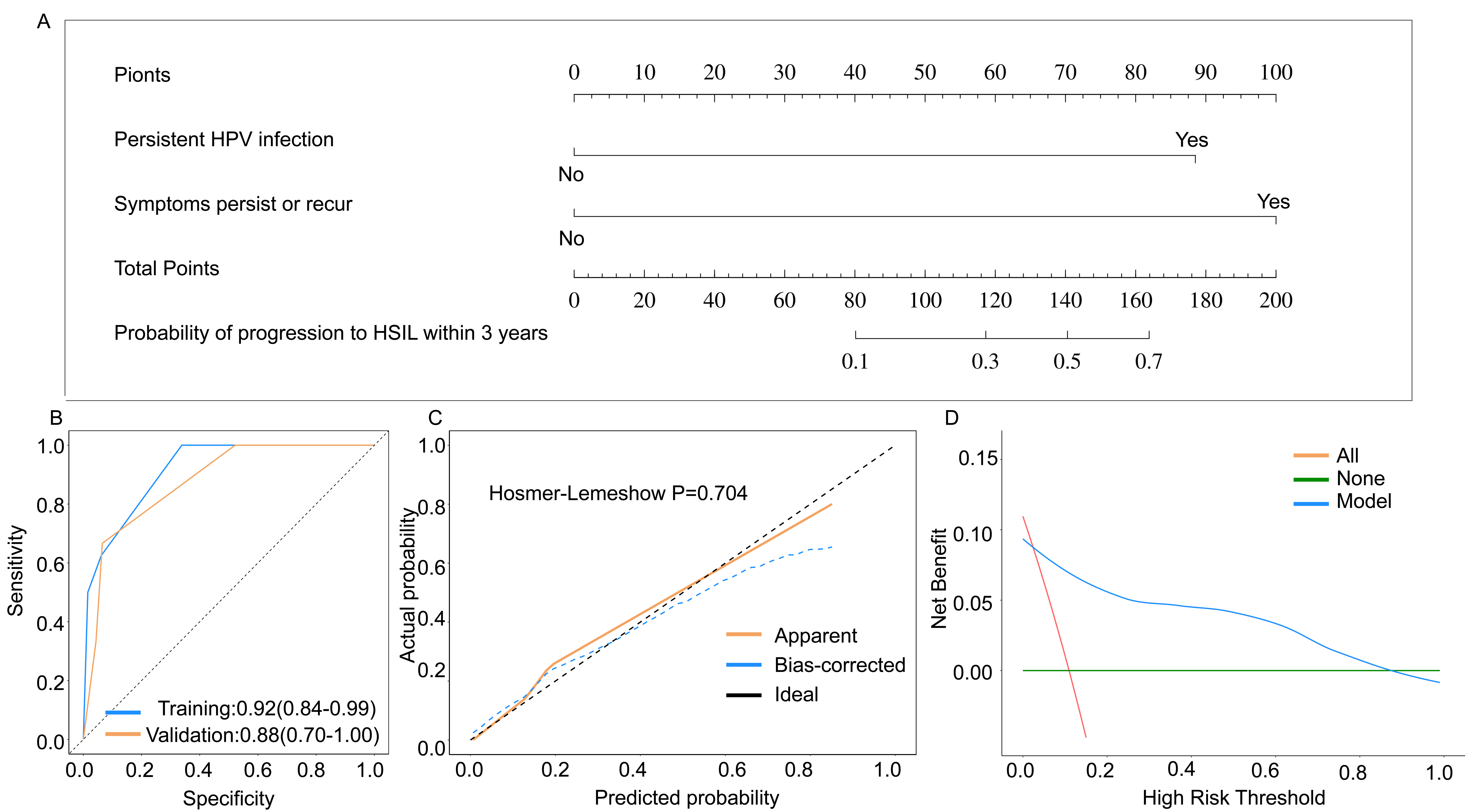 Fig. 2.
Fig. 2.
Prediction model for progression to HSIL within 3 years and associated evaluation parameters. (A) Prediction model. (B) Receiver operating characteristic (ROC) curves for the training and validation sets. (C) Calibration plot for the training set. (D) Decision Curve Analysis for the training set.
LSIL represents early cervical changes that may precede the development of
cervical cancer, with their progression influenced by the persistence or
recurrence of cervical symptoms such as abnormal bleeding, increased discharge,
and postcoital spotting [18]. These symptoms often reflect chronic cervical
inflammation or persistent high-risk HPV infection, both of which can disrupt the
cervical microenvironment. Chronic inflammation, driven by cytokines like
interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-
This study found that persistent high-risk HPV infection is an independent risk factor for the progression of LSIL to HSIL and significantly increases the risk of progression (OR = 7.67, p = 0.027). This result is consistent with several existing studies [29, 30], which have shown that persistent high-risk HPV infection is a critical factor in the progression of LSIL to HSIL or cancer. Zhao et al. [31] noted that persistent high-risk HPV infection not only increases the incidence of cervical intraepithelial neoplasia (CIN) but is also closely associated with the development of cervical cancer. Furthermore, the antiretroviral therapy (ART) and HPV Review Group (2017) reported that immune dysfunction, such as in HIV-infected individuals, exacerbates the persistence of HPV infection and accelerates the progression of lesions. This is particularly evident in immunocompromised populations, where persistent high-risk HPV infection is the primary driver of cervical lesion progression [32]. Related studies also highlight the decisive role of persistent high-risk HPV infection and local inflammatory responses in the deterioration of LSIL [33, 34]. These findings further support the results of this study, emphasizing the critical role of persistent high-risk HPV infection in the progression of LSIL to HSIL.
In previous studies, age, socioeconomic status (SES), and sexual history have also been identified as significant risk factors for the progression of LSIL to HSIL [35, 36, 37]. According to research by Evered [38], younger women, particularly those under 30, often experience a higher prevalence of LSIL due to the transient nature of HPV infections in this age group; however, their risk of progression to HSIL is relatively low. In contrast, women with lower SES often face barriers to screening and treatment, leading to more advanced lesions at the time of diagnosis [39]. The number of sexual partners and engagement in high-risk sexual behaviors increase the likelihood of HPV exposure and the persistence of infection [32]. These social and behavioral factors are closely linked to the persistence of HPV infection, which is the fundamental cause of lesion progression. Ultimately, factors such as lower socioeconomic status or having multiple sexual partners increase the likelihood of sustained HPV exposure, which in turn raises the risk of lesion progression.
This study also found that the persistence or recurrence of symptoms is an
independent risk factor for the progression of LSIL to HSIL (OR = 21.07,
p
Based on the results of the multivariate analysis in this study, we developed a predictive model that includes persistent high-risk HPV infection and the persistence or recurrence of symptoms. This model demonstrated good predictive ability in both the training cohort (AUC = 0.92) and the validation cohort (AUC = 0.88). Using this model, clinicians can identify high-risk patients and implement early interventions to prevent further progression of the lesions. This model not only showed excellent performance in this study but also aligns with the potential application of existing data-driven predictive tools in clinical practice [41]. Such a personalized risk prediction tool helps improve the precision of clinical decision-making, particularly in resource-limited settings, where it can effectively identify high-risk patients for early intervention.
Despite the valuable clinical insights provided by this study, several limitations remain. First, the sample size is relatively small, which may affect the generalizability of the results, particularly in the evaluation of rare factors [42]. Second, the data for this study were sourced from a single center, which may introduce limitations due to regional and ethnic factors, potentially affecting the external validity of the results. The retrospective design may lead to information bias, and the small number of events for some predictive factors could impact the stability of the model [43]. Additionally, handling of missing data may introduce bias into the analysis. Furthermore, the 3-year observation period may have missed cases where progression occurred after this time frame, potentially underestimating the true rate of progression. Nevertheless, this study explored early trends in the progression of cervical dysplasia and provides preliminary data for future research. We believe these findings are of exploratory significance and offer valuable insights for guiding clinical practice and subsequent studies. Therefore, future studies should aim to increase the sample size and incorporate multi-center data for validation, extend the follow-up period to capture long-term progression, and explore other potential risk factors, such as host immune status, to further refine the predictive model and enhance its ability to predict the risk of progression in LSIL patients.
This study confirms that persistent high-risk HPV infection and the persistence or recurrence of symptoms are independent risk factors for the progression of LSIL to HSIL, and it has developed an effective predictive model to assist clinicians in identifying high-risk patients. The model demonstrated good predictive ability in both the training and validation cohorts, showing strong clinical applicability. persistent high-risk HPV infection, as the core driver of progression, highlights the importance of effective monitoring and intervention for high-risk HPV infections. Additionally, the persistence or recurrence of symptoms may reflect chronic inflammation or changes in the immune microenvironment, which further accelerate the progression of lesions. Therefore, for patients exhibiting these clinical signs, enhanced follow-up and early intervention are crucial.
LSIL, low-grade squamous intraepithelial lesions; HSIL, high-grade squamous intraepithelial lesions; HPV, human papillomavirus; OR, odds ratio; AUC, area under the curve; SIL, cervical squamous intraepithelial lesions; ROC, the receiver operating characteristic curve; IUD, intrauterine device; CI, confidence interval; DCA, decision curve analysis; CIN, cervical intraepithelial neoplasia; ART, antiretroviral therapy; IC, internal control; MRI, magnetic resonance imaging; CT, computed tomography.
The data supporting the findings of this study are available upon reasonable request from the corresponding author.
HZ contributed to the design, data collection, supervision, compilation, and writing of the manuscript. YF was responsible for the organization, conceptualization, methodology, and revision of the study. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This research was carried out in accordance with the guidelines of the Declaration of Helsinki and was approved by the Wu Yi Maternal & Child Care Hospital Ethics Committee, with ethics approval number 2024112201. Throughout the study, the privacy of participants was safeguarded, and all data were subjected to anonymization processes to ensure confidentiality. All procedures were conducted in accordance with the participants’ signed informed consent.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/CEOG36364.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


