1 Department of Obstetrics, Paulista School of Medicine, Federal University of São Paulo (EPM-UNIFESP), 04021-001 São Paulo, Brazil
2 Discipline of Woman Health, Municipal University of São Caetano do Sul (USCS), 09550-051 São Caetano do Sul, Brazil
3 Department of Pediatrics, Pediatric Cardiology, Federal University of Rio de Janeiro (UFRJ), 21941-912 Rio de Janeiro, Brazil
4 Department of Biomedical and Dental Sciences and Morphofunctional Imaging, “G. Martino” University Hospital, 98100 Messina, Italy
Abstract
This review aims to improve the accuracy and efficacy of ultrasound diagnosis of fetal bradyarrhythmias. Fetal arrhythmias represent 10% to 20% of referrals to specialized clinics for the evaluation of fetal heart rhythm abnormalities
Various methods are available for assessing fetal heart rhythm through ultrasound and fetal echocardiography, which facilitate accurate diagnosis and help determine the optimal management strategies. Despite advancements in diagnostic techniques, the absence of studies with a substantial number of cases has resulted in significant variability in treatment approaches, influenced by local expertise and available resources.
Fetal bradycardias can be transient or persistent conditions. Differentiating between them is critical for determining appropriate management. Second-trimester physiological transient bradycardia is characterized by temporary decelerations in fetal heart rate that resolve spontaneously to normal rhythm without requiring treatment or follow-up. In contrast, isolated or immune-mediated complete atrioventricular block may progress rapidly to hydrops, requiring close monitoring, medical intervention, and, in some cases, early delivery.
In this review, we provide a comprehensive overview of all types of fetal bradycardias and the intrauterine management strategies used for their treatment.
Keywords
- fetal bradycardia
- complete congenital heart block
- maternal autoantibodies
- prenatal diagnosis
- prevention
- intrauterine treatment
- terbutaline
In general, all fetal cardiac arrhythmias cause significant concern when diagnosed during fetal heart evaluations. Irregularities in fetal heart rhythm affect approximately 0.5% to 2% of pregnancies and are responsible for 10% to 20% of referrals for intrauterine cardiac evaluations [1]. Fetal cardiac arrhythmias may be associated with structural heart problems or conduction system abnormalities, or they may result from transplacental passage of substances ingested by the mother or be immune-mediated due to an active pre-existing maternal disease. Clinically significant fetal and neonatal arrhythmias occur in approximately 1 in 4000 live births and are important causes of morbidity and mortality [2].
The arrhythmia diagnosis depends on the identification of mechanical consequences visualized through two-dimensional fetal echocardiography, M-mode, and spectral Doppler. The determination of heart rates for atrial and ventricular contractions can further define the arrhythmia mechanism [3]. Although there have been advances in diagnosis, there are not enough high-quality studies or evidence-based algorithms for managing these various disorders [4]. As a result, there is considerable variability in treatments based on local experience and available resources [5].
The objective of this scientific review is to describe the best practices (based on available data), indications, and types of treatments for fetal bradyarrhythmias.
The components of the cardiac electrical conduction system are: (1) the sinoatrial (SA) node, (2) the atrioventricular (AV) node, and (3) the His-Purkinje system. The role of this system is to generate and propagate electrical impulses through the myocardium to produce each heartbeat. The conduction system of the fetal heart becomes functionally mature around the 16th week of gestation. The AV node forms in the 10th week, separately from the His bundle, and their connection occurs secondarily [3].
The SA node is a group of autonomous cells that function as the heart’s pacemaker, located in the upper wall of the right atrium (RA). The electrical impulse generated by the SA node is conducted through the atrial myocardium to the AV node, where it is briefly delayed before being transmitted to the ventricles. From the AV node, the electrical impulse is propagated to the ventricles through the His bundles and Purkinje fibers [6].
The electrical events of the cardiac cycle can be assessed using ultrasound, by observing the movements or blood flows caused by atrial and ventricular contractions. During atrial systole, the atria contract and pump blood into the ventricles (active ventricular filling), corresponding to the A-wave in the Doppler recording of the AV valves, either via conventional or tissue Doppler, and in the venous Doppler (pulmonary vein, vena cava, and ductus venosus), as well as in M-mode through the contraction of its wall. The A-wave corresponds to the P-wave in the electrocardiogram (ECG) [6, 7]. Ventricular systole is noticeable by the Doppler of aortic or pulmonary flows by ultrasound and corresponds to the QRS complex on the ECG. It can also be captured using M-mode, where we are able to observe mechanical contractions of the ventricular wall. Heart rhythm is determined using simultaneous recordings of the mechanical movements of the atrial and ventricular walls in M-mode. Although widely used, M-mode assessment is limited during the first trimester and in adverse situations such as unfavorable fetal position, in hydropic fetuses with cardiac contractile dysfunction, and in obese pregnant women [6]. Moreover, in the initial trimester, prior to 10 weeks’ gestation, the conventional Doppler assessment of fetal heart rate (HR) based on the mitral A-wave (ventricular filling) is also a limitation, as it is typically not feasible to discern the E and A waves.
Therefore, in the presence of arrhythmia, several important characteristics should be evaluated: (1) HR, (2) rhythm regularity, (3) AV relationship, and (4) AV and ventriculoatrial (VA) intervals. Fig. 1 shows the methods for evaluating fetal HR. One of the main causes of fetal heart failure (FHF) is arrhythmia, particularly fetal tachycardia and complete atrioventricular block [7]. It is important to remember that the fetal myocardium has some differences that make it more susceptible to developing FHF in response to stress, such as reduced contractile capacity due to myocardial immaturity, lower compliance, greater dependence of cardiac output on fetal HR and reduced sympathetic innervation [8, 9].
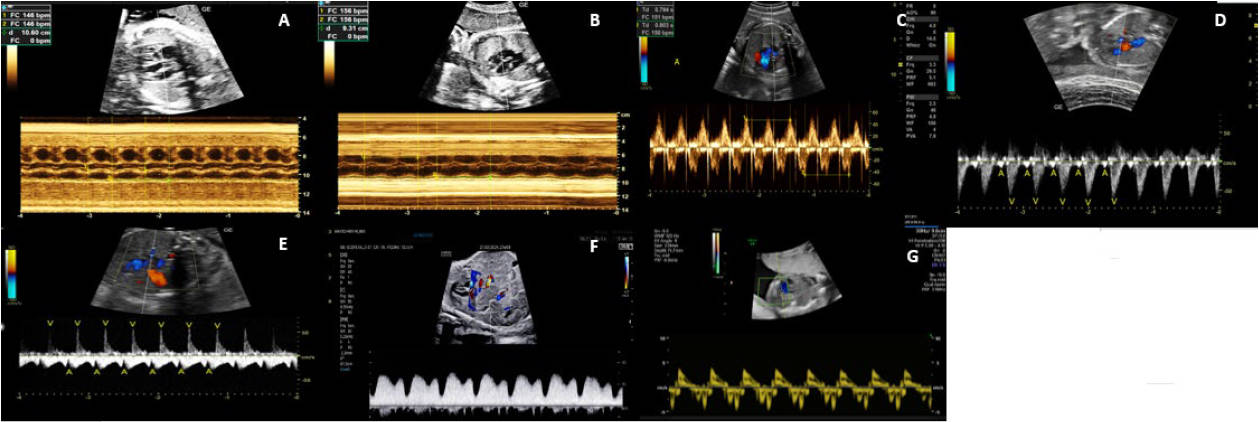 Fig. 1.
Fig. 1.
Methods for evaluating fetal heart rate. Mid-trimester imaging demonstrating how to evaluate fetal heart rhythm using M-mode and Doppler ultrasound. (A) M-mode of the parasternal long-axis view in the fetus, showing the movement of the aortic valve and atrial contraction motion. (B) M-mode of the perpendicular four-chamber view, showing the motion of the contraction of the right atrial wall and the movement of the lateral wall of the left ventricle. (C) Doppler of mitral-aortic flow in the five-chamber view, with the sample between the inflow and outflow of the left ventricle. (D) Doppler of the aorta and superior vena cava in the three-vessel view, marked by two well-defined flows: one low-velocity flow marking atrial contraction and another systolic flow with higher velocity. (E) Doppler of the right pulmonary vein and right pulmonary artery in the lung parenchyma, showing a low-velocity venous flow from the pulmonary vein with a notch marking atrial contraction and another positive systolic flow. (F) Ductus venosus Doppler characterized by two peaks and a valley: the first peak is ventricular systole, the second peak is ventricular diastole, and the valley represents atrial contraction. (G) Tissue Doppler with a positive peak representing ventricular systole and the first negative peak representing ventricular filling and the second representing atrial contraction.
Some conditions may improve spontaneously or respond to intrauterine treatment
or after birth; others respond poorly or are not amenable to therapy and may
progress to heart failure, with hydrops, multiple organ failure, and fetal death
[8]. A score for the echocardiographic evaluation of fetal cardiac function is a
system proposed to grade and monitor the severity of FHF using five
echocardiographic parameters: (1) presence of fetal effusions (hydrops), (2)
venous Doppler, (3) heart size, (4) cardiac function, and (5) arterial Doppler.
The evaluation and quantification of FHF are critical steps that allow for
counseling in high-risk pregnancies and decision-making both during the
intrauterine period and in the neonatal stage [8]. A score of 10 is considered
normal, 8–9 indicate mild FHF, 6–7 indicate moderate FHF, and
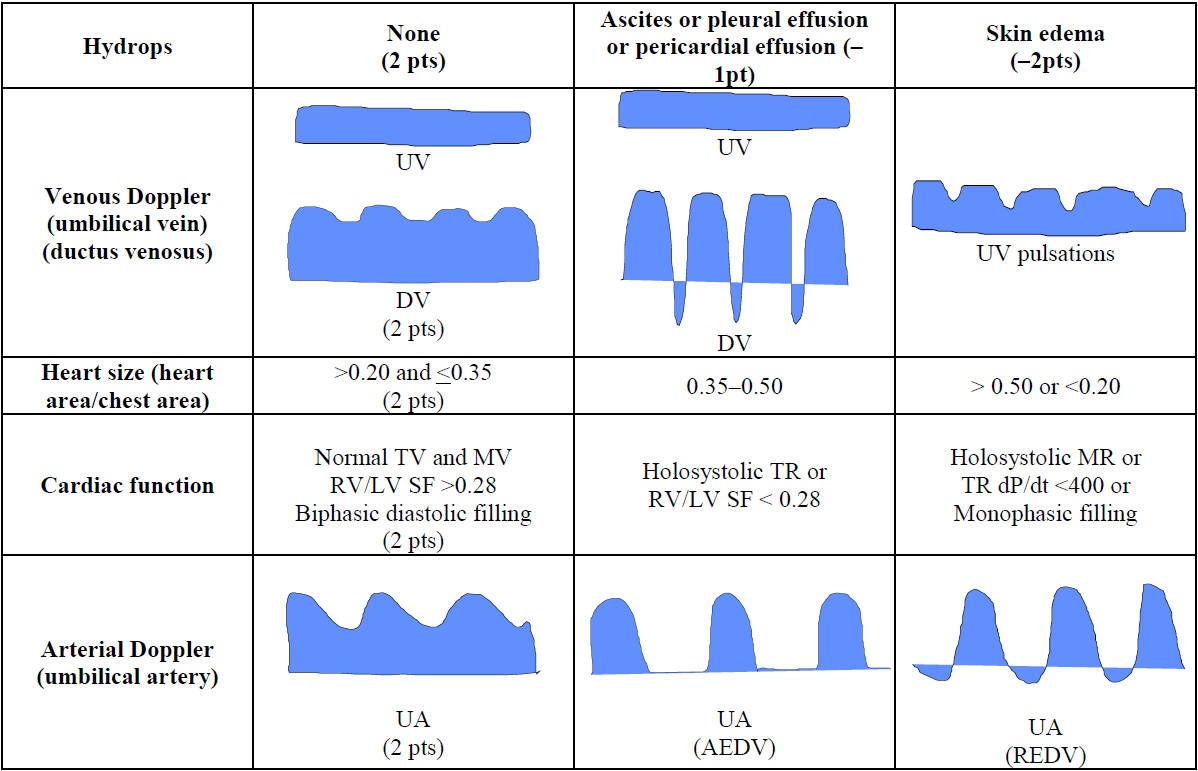 Fig. 2.
Fig. 2.
The cardiovascular profile (CVP) score. The normal value of CVP score is if there are no abnormal signs and reflects 2 points for each of 5 categories: hydrops, venous Doppler, heart size, cardiac function, and arterial Doppler. AEDV, absent end diastolic velocity; dP/dt, change in pressure over time of tricuspid regurgitation (TR) jet; DV, ductus venosus; LV, left ventricle; MR, mitral valve regurgitation; MV, mitral valve; pts, points; pt, point; SF, ventricular shortening fraction; TR, tricuspid valve regurgitation; TV, tricuspid valve; REDV, reversed end diastolic velocity; RV, right ventricle; UV, umbilical vein; UA, umbilical artery.
The normal fetal heart rhythm is regular, with atrial and ventricular heart
rates being similar (AV ratio 1:1) and varying with gestational age. Some authors
consider the normal HR range to be 110 bpm to 180 bpm [5], while more recent
studies show a normal range between 120 bpm and 180 bpm [1]. At any gestational
age, a mean, sustained HR
Fetal bradyarrhythmia can be classified as follows: (1) sinus bradycardia, (2) ectopic atrial rhythm such as a low atrial bradycardia; (3) blocked atrial bigeminism (BAB); (4) incomplete AV block such as second-degree AV block and (5) complete AV block (CAVB). In the case of persistent fetal bradycardia with sinus rhythm characteristics (AV 1:1), the presence of maternal anti-Sjogren’s-syndrome-related antigen A (anti-Ro/SSA) antibodies and maternal viral infection should be investigate. Important differential diagnosis of persistent sinus bradycardia includes ectopic atrial rhythm, representing a diagnostic challenge in utero. The presence of structural heart disease, such as left isomerism, should draw attention to ectopic atrial rhythm in such cases; nevertheless, it is impossible to confirm this diagnosis in utero. Furthermore, although rare, long QT syndrome (LQTS) is a diagnosis that should be considered in cases with a family history of sudden death, syncope and congenital deafness. Fetal magnetocardiography is a diagnostic modality that enables the diagnosis of fetal arrhythmias with repolarization abnormalities, such as LQTS. It is currently only available in a few specialized centers. However, advances in technology may allow this diagnostic modality to become widely available in clinical practice in the future.
Second-degree AV block can be distinguished from blocked atrial bigeminy through the measurement of atrial intervals. In the latter condition, the atrial rate is irregular, with variations exceeding 20 milliseconds. Conversely, in second-degree AV block, the atrial rate is regular, with premature atrial events conducted through the refractory atrioventricular node. Table 1 illustrates the main types of fetal bradycardia [10, 12].
| Type of bradycardia | Rhythm | HR | AV conduction and other features | Therapeutic management |
| Sinus bradycardia | Regular with transient low-HR | In general: |
1:1, sinus bradycardia due to long QT: IVRT above the normal range | -No specific treatment |
| Low-atrial | Regular with transient low-HR | In general: 80–110 bpm | 1:1, short AV | -No specific treatment |
| -Investigate potential causes (maternal use of sedatives or beta-blockers, maternal anti-Ro/anti-La antibodies or viral myocarditis) | ||||
| Blocked atrial bigeminism (BAB) | Irregular ventricular HR | In general: 75–110 bpm | A rate |
-No specific treatment |
| -Follow-up: weekly evaluation of fetal HR by echocardiogram or sonar | ||||
| First-AV block | Regular | In general: 65–90 bpm | 1:1, prolongation of AV time interval | -May benefit from in utero treatment with fluorinated steroids* if maternal anti-Ro/anti-La antibodies+ |
| -Follow-up: 1 to 2 weekly AV interval from weeks 18 to 26; | ||||
| Second-AV block | Regular or not | In general: 60–75 bpm | 2:1 or higher AV ratio | -May benefit from in utero treatment with fluorinated steroids* if maternal anti-Ro/anti-La antibodies+ |
| -Follow-up: 1 to 2 weekly AV interval from weeks 18 to 26; | ||||
| Complete-AV block | Regular ventricular HR | AV completely dissociated | -Close fetal myocardial function monitoring (CV score) | |
| -Fetal HR | ||||
| -Fetal endocardial fibroelastosis or systolic dysfunction: consider maternal intravenous immunoglobulin*** |
HR, heart rate; AV, atrioventricular interval; bpm, beats per minute; IVRT, isovolumetric relaxation time; ICT, isovolumetric contraction time; w, weeks of gestation; CV score, cardiovascular score. * Fluorinated steroid: maternal dexamethasone 4–8 mg/day (oral use), reduced to 2 mg/day after 30 w. ** Maternal beta-agonist: salbutamol 10 mg/day (oral use bis in die (b.i.d)). *** Maternal intravenous immunoglobulin: 1–2 g/kg (maximum: 70 g) every 3–4 weeks.
These are transient decelerations of the fetal HR which can drop as low as 40 bpm and resolve within one minute, returning to a normal heart rhythm. This type of bradycardia is observed in the second trimester and is considered benign, meaning clinically insignificant. It is believed to be caused by exaggerated vagal tone, which is common during this period. This type of bradycardia does not require treatment (Table 1).
Characterized by a 1:1 AV conduction with a regular fetal HR
Unlike sinus bradycardia, low atrial rhythm mechanisms include congenital displacement of atrial activation or absence of the sinoatrial node [5], meaning the electrical stimulus can originate from any focus within the atria. Ectopic atrial rhythm can appear in association with congenital heart defects such as left atrial isomerism or hereditary arrhythmias like LQTS. Low atrial rhythm is characterized by regular low fetal HR ranging from 80 bpm to 110 bpm with 1:1 AV conduction with short AV interval. It does not require antiarrhythmic treatment in utero [1] (Table 1).
LQTS is a very important and currently well-studied condition. It is inherited
in an autosomal dominant manner and can present as mild isolated bradycardia,
supraventricular extrasystole with 2:1 blocked bigeminy, second-degree
atrioventricular block (AVB) [12], Torsades de Pointes (TdP), or ventricular
tachycardia (VT) [7]. While TdP and second-degree AV block are generally easily
recognized and highly specific, they occur infrequently. It is known that 40% of
fetuses with bradycardia have associated LQTS. According to Mitchell et
al. [12], this association can reach 44% to 66% between the 26th and 40th
weeks of gestation. Previous publications have described fetal HR varying from
100 bpm to 130 bpm in fetuses with LQTS. Other reports indicate that a fetal HR
of 110 bpm at any gestational age could exclude potential carriers of this
syndrome. Thus, using fetal HR
When LQTS is suspected, fetal management includes careful observation and a detailed family history of LQTS. Fetal magnetocardiography is an examination used in the diagnosis of such arrhythmias, enabling the detection of fetal repolarization abnormalities, especially in LQTS. In the future, it may become a more accessible diagnostic tool, though it is currently only available in specialized centers [4]. Regardless of family history, a postpartum ECG should be performed with measurement of the corrected QT (QTc) interval. If there is a positive family history or the fetus presents with complex rhythms, first-degree relatives should be screened by ECG, even if they are asymptomatic. First-degree relatives with ECG showing evidence of LQTS may justify genetic testing. If the postnatal genetic test is positive in a fetus suspected of having LQTS without clinical or genetic manifestations in first-degree relatives, parental mosaicism could be a possibility, especially for monitoring future pregnancies [12].
Maternal electrolyte imbalances, such as hypomagnesemia and hypocalcemia, as
well as certain drugs and anesthetic agents that can prolong the QT interval,
should be avoided [5]. Fetal treatment is not recommended for bradycardia, but
tachycardias such as TdP and VT do require intervention. Measuring isovolumetric
time intervals derived from spectral Doppler can help distinguish challenging
rhythms. In fetuses with blocked bigeminy or antibody-mediated AV block who
exhibit an isovolumetric relaxation time (IVRT)
Blocked supraventricular extrasystoles present with a 2:1 AV conduction and a ventricular rate that varies from 75 bpm to 110 bpm [1]. Some authors suggest that the fetal HR may vary between 70 bpm and 90 bpm [5] and usually do not require treatment, as their resolution is often spontaneous before delivery [13]. Weekly monitoring is necessary to detect cases that may develop into supraventricular tachycardia (SVT), as approximately 10% to 13% can progress to SVT [1].
Blocked atrial bigeminy (BAB) frequently exhibits a typical atrial rhythm with
alternating shorter and longer intervals between atrial contractions (AA
interval), while in second-degree AVB, the interatrial frequency is regular
(i.e., the AA interval remains consistent). Furthermore, the IVCT is
shorter, which can help with the differential diagnosis from 2:1 AVB [5].
Sonesson et al. [13] found IVCT values of 13.6
It is important to ensure that this is indeed a BAB, as AV block is a differentiation diagnosis. BAB requires no specific treatment but may progress with spontaneous resolution, whereas second-degree BAB may evolve to complete resolution, which is usually irreversible. It is advisable to follow up BAB with weekly assessment of fetal HR by echocardiogram or sonar.
AV block are electrical conduction disturbances that occur between atrial depolarization and ventricular depolarization. Bradycardia and electromechanical dissociation cause the fetal heart to accommodate a larger stroke volume to maintain adequate cardiac output.
All cardiac impulses are conducted from the atria to the ventricles with a
prolonged AV conduction time, marked by a PR interval
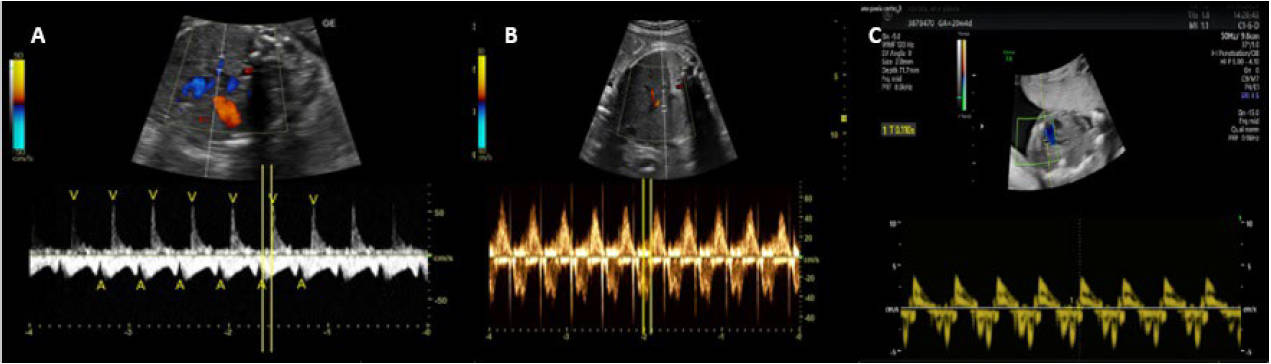 Fig. 3.
Fig. 3.
Evaluation of the mechanical PR interval. (A) Doppler of the pulmonary vein and the right pulmonary artery with the mechanical PR interval measured from atrial contraction to the beginning of pulmonary systolic flow. (B) Mitral-aortic Doppler in the five-chamber view, with the PR interval measured between the E and A waves of the mitral flow to the beginning of aortic systolic flow. (C) Tissue Doppler of the left ventricle’s lateral wall, where the PR interval should be measured between the E’ and A’ waves to the start of the S’ wave.
In pregnant women with anti-Ro (anti-SSA) and or anti-La (anti-Sjogren’s-syndrome-related antigen B (anti-SSB)) antibodies
such as in Systemic Lupus Erythematosus (SLE) or Sjögren’s Syndrome, without
established CAVB, it is recommended to monitor the mechanical PR interval 1 to 2
weekly between 18 and 26 weeks of gestation. After 26 weeks of gestation, it is
recommended to continue monitoring until birth, as follows: (1) if the mechanical
PR interval remains stable and
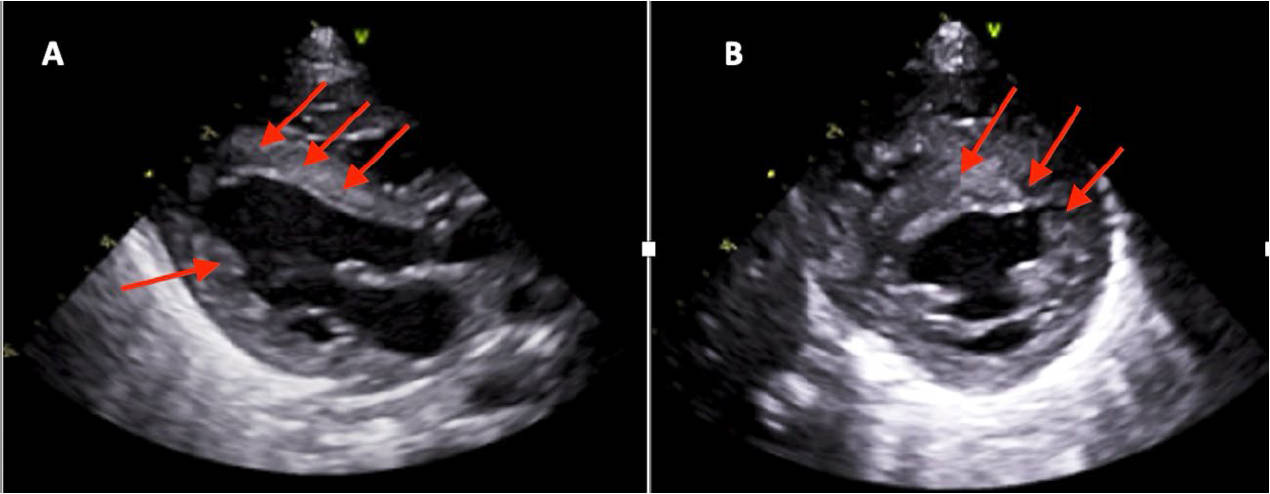 Fig. 4.
Fig. 4.
Newborn of a mother with Sjögren’s Syndrome without congenital complete atrioventricular block, showing signs of fibroelastosis, due to the proliferation of collagen and elastic fibers caused by the transplacental passage of maternal antibodies. (A) Transthoracic echocardiogram, parasternal long-axis view showing thickening of the ventricular endocardium in the septal and left ventricular posterior wall regions (red arrows). (B) Parasternal short-axis view showing endocardial thickening in the septal region (red arrows) with more echogenic points.
There is a regular or not low HR with progressive increase in the PR interval until an atrial electrical impulse is not conducted to the ventricle. The assessment of the IVCT helps differentiate BAB, which usually resolves spontaneously, from second-degree AV block [13].
Ciardulli et al. [20] demonstrated that there was progression to CAVB in 52% of fetuses treated with corticosteroids and 73% of those not treated. Once second-degree AV block is diagnosed, the use of corticosteroids may be benefit to prevent progression to third-degree AV block (until more robust evidence-based studies such meta-analyses are available). Furthermore, it has been suggested that the corticosteroids treatment improves myocardial performance and interferes with the inflammatory process in fetal conduction tissue, in addition to accelerating lung maturity when there is a risk of premature birth.
CAVB is caused by an electrical disorder leading to electromechanical dissociation, with interruption of electrical impulses from the atria to the ventricles (Fig. 5). The incidence of AV block is 1 in every 15,000–20,000 live births. Of these cases, 50% to 55% of the cases are associated with structural congenital heart defects, with 60% associated with left atrial isomerism and about 40% associated with positive maternal SSA/Ro or SSB/La antibodies [21, 22, 23, 24, 25]. Immune-mediated CAVB typically presents with a normal structural heart.
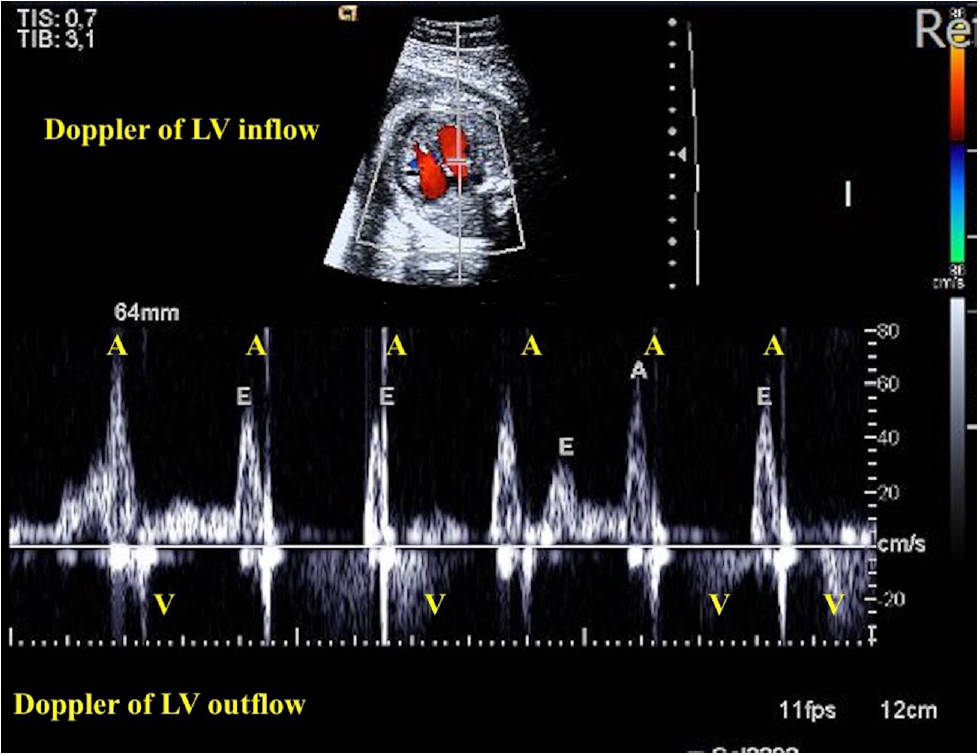 Fig. 5.
Fig. 5.
Complete atrioventricular block (CAVB) in a case with maternal high anti-SSA and anti-SSB antibody titers. Doppler of mitral and aortic flow recordings showing the A-V dissociation. Note that the atrial HR (number of A waves) higher than ventricular HR (number of V waves). A, atrial contraction; V, ventricular contraction; anti-SSA, anti-Sjogren’s-syndrome-related antigen A; anti-SSB, anti-Sjogren’s-syndrome-related antigen B.
Immune-mediated AV block without cardiac structural anomaly appears to have the best long-term prognosis. Lopes et al. [26] reported that spontaneous regression of AV block in utero is possible in fetuses whose mothers remained seronegative throughout pregnancy. Baruteau and Schleich [27] reported an interim analysis of a multicenter study, showing that 37% of these seronegative fetuses with AV block progressed to third-degree AV block during childhood.
The pathophysiology of immune-mediated AV block is not fully understood in susceptible fetuses. The fetuses of pregnant woman with anti-SSA/Ro and/or anti-SSB/La antibodies are at risk of cardiac damage due to transplacental crossing of these antibodies. The presence of anti-SSA (anti-Ro) and anti-SSB (anti-La) occurs in autoimmune diseases such as Sjögren’s syndrome and systemic lupus erythematosus. The “Ro” autoantigen consists of two proteins (weighing 60 kDa and 50 kDa) which can be detected using separate solid-phase immunoassays. Ribonucleoproteins from the antibodies bind to autoantibodies, which enter the fetal circulation in the middle of the second trimester, being expressed in the cardiomyocytes of the fetal myocardium, particularly: 52 kD Ro/SSA (Ro52), 60 kD Ro/SSA (Ro60), and 48 kD La/SSB (La48) [6]. This immune process results in fetal conduction tissue inflammation of the fetal conduction tissue and myocardium with progressive and irreversible fibrosis [21]. This can lead to functional abnormalities including cardiomegaly, ventricular hypertrophy, impaired ventricular function, and atrioventricular valve regurgitation, contributing to pericardial effusions and, in severe cases, FHF and hydrops fetalis [21]. Furthermore, a study described the presence of ventricular fibroelastosis [24] (Fig. 6).
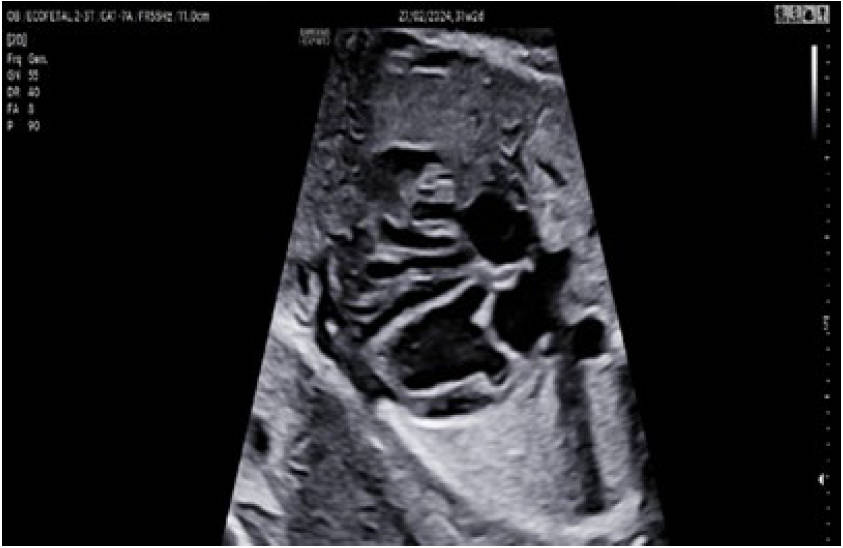 Fig. 6.
Fig. 6.
Fetus at 31 weeks of gestation with congenital complete atrioventricular showing signs of fibroelastosis. The fetal echocardiogram, in a four-chamber view, shows thickening of the left ventricular endocardium in the septal and lateral regions, along with left ventricular hypertrabeculation and increased echogenicity.
Fetal congenital immune-mediated CAVB occurs in 2% to 6% of anti-Ro–positive mothers with a recurrence rate of 18% in subsequent pregnancies, and it carries a mortality rate of approximately 30% [4]. The outcome can vary depending on the management and monitoring of these pregnancies [22, 23]. It is known that around 50% of mothers with anti-Ro/SSA antibodies are asymptomatic at the time of delivery or diagnosis [21] (Fig. 7).
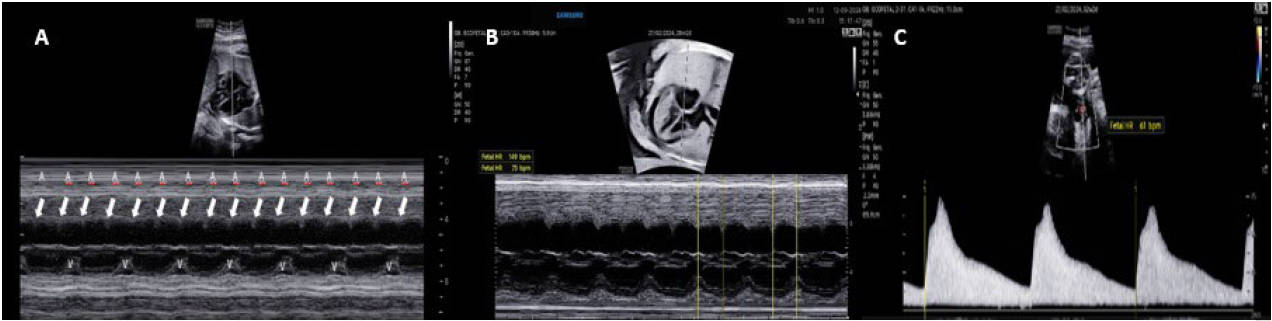 Fig. 7.
Fig. 7.
Fetal heart rate evaluation using M-mode showing right atrial and left ventricular contraction, and spectral Doppler of umbilical artery flow. (A) M-mode with a sample cutting through the right atrium and left ventricle, in an oblique four-chamber view, where arrows indicate atrial contraction (A) and ventricular contraction (V). (B) M-mode demonstrating mechanical movement of atrial contraction with a heart rate of 149 bpm and ventricular contraction with a heart rate of 75 bpm, indicating 2:1 complete atrioventricular block. (C) Spectral Doppler of the umbilical artery showing a ventricular heart rate of 61 bpm.
Several treatments have been attempted for fetal CAVB, but no consistent study
has been achieved. Some authors have shown that there is no evidence supporting
the use of corticosteroids in already established immune-mediated CAVB due to
irreversible fibrosis in the cardiac conduction tissue, as well as the side
effects caused in pregnant women [24, 28, 29]. Lopes et al. [26] also
demonstrated that in fetuses with a HR
Jaeggi et al. [31] proposed a protocol where: for a fetal HR
Regarding treatment with
The follow-up protocol for such cases is not well-established [1, 29]. Weekly or biweekly monitoring of the AV interval is recommended from weeks 18 to 26 weeks of gestation (Table 1). Delivery should be planned in a tertiary center, with either cesarean section or vaginal delivery at 37 weeks of gestation, if the course is satisfactory. In cases of fetal hydrops progression, a cesarean section is recommended, with pacemaker implantation immediately after birth [27].
Although controversial because it does not appear to reverse congenital heart
block and may have potential adverse effects on both the developing fetus and the
mother, the use of immunoglobulin can improve fetal and postnatal survival in
fetuses with systolic cardiac dysfunction and/or signs of endocardial
fibroelastosis and/or myocarditis [1]. Dexamethasone may also be used to reduce
the prevalence of dilated cardiomyopathy in stablished immune-mediated CAVB with
signs of myocardial inflammation, however, it was not classically recommended for
CAVB due to the significant side effects that can occur in the mother or fetus.
Therefore, the use of this medication should be discontinued if they occur
[32, 33, 34]. Histological evidence shows a
Indeed, studies comprehensive studies are currently underway with the objective of evaluating the efficacy of treatment with steroids and intravenous immunoglobulin in the context of immune-mediated incomplete fetal heart block. Additionally, the possibility of restoring the fetal heart rhythm to a normal state or preventing the development of complete heart block is being explored in such trials [Surveillance and Treatment to Prevent Fetal Atrioventricular Block Likely to Occur Quickly; ClinicalTrials.gov: NCT04474223]. These investigations are conducted in a stratified fashion, taking into account the titer of anti-Ro/SSA antibody present in each pregnancy [Slow Heart Registry of Fetal Immune-mediated High Degree Heart Block; ClinicalTrials.gov: NCT04559425].
The maternal use of beta-sympathomimetics (salbutamol 30–40 mg/d or terbutaline5–30 mg/d) is indicated when fetal HR is bellowing 50 bpm to 55 bpm and/or in the presence of ventricular dysfunction. Maternal extrasystoles and sinus tachycardia may occur, but these drugs are generally well tolerated. An increase in fetal heart rate of about 10%–15% of baseline is expected, which may prolong pregnancy to term or near term [1, 24, 29, 32].
Regarding the use of hydroxychloroquine, while it is not a medication for reversing antibody-mediated atrioventricular block, it has been shown to reduce the recurrence rate by more than 50% in future pregnancies [4]. As a result, the prescription of hydroxychloroquine to pregnant women with ant-Ro/SSA antibodies is supported by European League Against Rheumatism (EULAR) since 2016 [35, 36].
The estimated fetal weight is critically important, especially for planning the postnatal management. This is particularly relevant because the permanent pacemaker implantation typically occurs when the infant’s weight exceeds 2800 grams. Additionally, the need for a temporary pacemaker significantly increases the risk of neonatal mortality [33]. Table 1 describes the main characteristics and management of the CAVB and the main types of fetal bradyarrhythmia.
The fetal heart rhythm can be assessed using fetal ultrasound or echocardiography. This can aid in the correct diagnosis and help determine the optimal approach to be adopted. In cases of sinus bradycardia, the identification of underlying causative factors is imperative, as such conditions can be indicative of fetal hypoxia and acidemia during or prior to labor, particularly in instances of placental insufficiency. In the context of autoimmune AVB, the maternal administration of hydroxychloroquine, while not a pharmacological agent capable of reversing fetal atrioventricular block, has been shown to reduce the likelihood of recurrence by over 50% in subsequent pregnancies. Weekly or biweekly fetal echocardiography will therefore detect emerging congenital heart block only by chance. Telemedicine with home monitoring of fetal heart rate may be a useful tool in this context. Despite advances in fetal diagnosis, there is a lack of studies on how to predict the time of establishment of CAVB, leading to considerable variability in the therapeutic management of autoimmune AV block. In cases of CAVB, it is crucial to monitor fetal heart function with regularity and, when the fetal heart rate is markedly depressed, to plan for immediate pacemaker implantation.
MEMA: conceptualization, methodology, investigation; LKdAT: data curation, investigation, resources; EAJ: writing, editing, supervision, validation; NJBV: writing draft, methodology, formal analysis; RG: project administration, validation, resources. All authors have read and agreed to the published version of the manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Roberta Granese is serving as one of the Editorial Board members of this journal. We declare that Roberta Granese had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Paolo Ivo Cavoretto.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







