1 Department of Cardiology, Nanfang Hospital, Southern Medical University, 510515 Guangzhou, Guangdong, China
2 Department of Cardiology, The First Affiliated Hospital of Wannan Medical College, 241000 Wuhu, Anhui, China
3 Department of Emergency, The First Affiliated Hospital of Wannan Medical College, 241000 Wuhu, Anhui, China
Abstract
Transcatheter aortic valve implantation (TAVI) is increasingly utilized for patients with pure aortic regurgitation (PAR). A significant clinical challenge in this patient population is the need for permanent pacemaker implantation (PPI), which occurs frequently post-TAVI and can impact cardiac conduction and rhythm management. This study aimed to explore the effects of PPI on short-term mortality, rates of adverse events, and cardiac function in PAR patients following TAVI.
This retrospective study, conducted in a single center, included 69 PAR patients who underwent TAVI from January 2021 to December 2023. Patients were categorized into two groups: those who received a permanent pacemaker (PM) and those who did not (NPM). The outcomes measured included complications such as pacemaker pocket hematoma and infection, changes in postoperative left ventricular ejection fraction (LVEF) and left ventricular end-diastolic diameter (LVEDD) at 6 months, as well as rates of rehospitalization and mortality.
No significant differences were noted in baseline characteristics or complications between the PM and NPM groups (p > 0.05). The types of PPI and associated complications were also comparable. There was no significant disparity in the incidence of all-cause mortality (PM: 12%, NPM: 11.36%, p = 0.755), major bleeding (PM: 4%, NPM: 4.55%, p = 0.612), or cerebral embolism (PM: 12%, NPM: 4.55%, p = 0.506) between the two groups at 6 months post-TAVI. Additionally, readmission rates were similar at 1, 3, and 6 months following the procedure. Multinomial logistic regression analysis revealed that age (p = 0.020), history of cerebral infarction (p = 0.015), and hypertension (p = 0.019) were significant predictors of mortality. The survival curve indicated that fatalities in the NPM group predominantly occurred during the perioperative period. At the 6-month follow-up, there was no significant difference in survival rates between the two groups (p = 0.971). Regarding cardiac function, irrespective of PPI, a decreasing trend in LVEDD (PM: –4.19 mm, NPM: –6.16 mm, p = 0.000) and an increasing trend in LVEF (PM: +2.19%, NPM: +2.74%, p = 0.053) were observed.
This study was the first to investigate the effects of PPI on the short-term mortality, adverse events, and cardiac function of PAR after TAVI. The results indicated that for PAR, advanced age and previous cerebral embolism increase the mortality after TAVI; however, PPI was not associated with mortality and adverse events after 6 months.
Keywords
- transcatheter aortic valve implantation
- pure aortic regurgitation
- permanent pacemaker implantation
- mortality
- readmission rate
- cardiac function
Transcatheter aortic valve implantation (TAVI) is predominantly utilized for patients diagnosed with aortic stenosis (AS). However, with the ongoing advancements in valve technology and stent design, a growing cohort of patients with pure aortic regurgitation (PAR) are receiving TAVI [1]. A common complication associated with TAVI is the need for permanent pacemaker implantation (PPI). The incidence of PPI following atrioventricular block in AS patients undergoing TAVI varies between 3.4% and 25.9% [2]. In contrast to AS, PAR, characterized by regurgitated flow due to aortic valve leaflet dysfunction, leads to left ventricular dilation and volume overload, presenting unique challenges compared to the calcific stenosis in AS. This distinction may impact TAVI procedures and the need for PPI, with PAR patients showing a higher PPI requirement post-TAVI, likely due to the altered left ventricular outflow tract geometry and increased risk of conduction disturbances [3].
Although prior research has indicated that pacemaker implantation, particularly with leads positioned in the right ventricular apex, is associated with an increased risk of heart failure and atrial fibrillation [4, 5], the impact of PPI on cardiac function following TAVI in AS patients remains a subject of debate [6]. A growing body of clinical trials suggests that PPI may increase hospitalization rates and mortality among these individuals [7, 8]. Consequently, this study is designed to investigate the effects of PPI on PAR patients during a six-month follow-up period after TAVI, addressing a gap in the literature where the influence of PPI on cardiac function and prognosis in PAR patients post-TAVI has yet to be documented.
By bridging this knowledge gap, this research will provide valuable insights into the effects of PPI on cardiac function and prognosis, which will be instrumental in refining patient selection, procedural strategies, and post-procedure management. Ultimately, these findings will contribute to the development of guidelines for the management of PAR patients undergoing TAVI, with a particular focus on the decision-making process for PPI.
This single-center, retrospective, and consecutive study encompassed all PAR
patients who underwent TAVI at our institution from January 2021 to December
2023. Patients who successfully received TAVI were categorized into two groups
based on the necessity for PPI post-TAVI: the permanent pacemaker (PM) group and
the group who did not receive a permanent pacemaker (NPM). The study recorded the
incidence of complications such as pacemaker pocket hematoma, infection, changes
in postoperative cardiac function at six months, rehospitalization rates, and
mortality rates. Inclusion criteria for PAR patients included symptomatic severe
PAR and a Society of Thoracic Surgeons (STS) risk score of
The TAVI procedure was performed utilizing the VitaFlow Liberty system via the
femoral artery. The size of the prosthesis was determined based on a computed
tomography scan of the aortic ring area. Aortic valve positioning was guided by
angiography and trans esophageal echocardiography. The valve was deployed at the
level of the coronary sinus under rapid pacing (
All patients underwent temporary pacemaker implantation via the right internal
jugular vein prior to TAVI. PPI was performed if severe bradycardia persisted for
5 to 7 days post-TAVI without resolution. The right ventricular lead was
positioned in the mid to lower septum. Cardiac resynchronization therapy (CRT)
was administered to patients with a left ventricular ejection fraction (LVEF) of
(1) Hypertension is a major risk factor for cardiovascular events and is closely monitored in patients undergoing TAVI [10, 11].
(2) Diabetes mellitus is associated with increased morbidity and mortality in cardiovascular patients, including those undergoing valve interventions [12].
(3) Coronary artery disease confirmed by coronary angiography or computed tomography angiography with coronary artery stenosis of 50% or greater. The presence of coronary artery disease can complicate outcomes following TAVI and is an important comorbidity to consider [13].
(4) Atrial fibrillation is a common arrhythmia that can affect patient management and outcomes post-TAVI [14].
(5) Mitral regurgitation was diagnosed via doppler echocardiography, mitral regurgitation can significantly impact left ventricular function and is a relevant comorbidity in patients with aortic valve disease [15].
(6) The incidence of hematoma and major bleeding in TAVI patients following pacemaker implantation was observed. Major bleeding was defined by the occurrence of one of the following three conditions: fatal bleeding; symptomatic bleeding in critical locations or organs, such as intracranial, spinal, intraocular, peritoneal, intra-articular, pericardial, or intramuscular compartment syndrome; or a decrease in hemoglobin by 20 g/L (1.2 mmol/L) or more, resulting in the transfusion of two or more units of whole blood or red blood cells [16].
Data were analyzed using SPSS Statistics 26.0 (IBM, Armonk, NY, USA). Chi-square
and Fisher’s exact tests were employed to evaluate associations between outcomes
and categorical variables. The t-test was utilized to compare means of
continuous variables between patient groups and the Mann-Whitney U test was used
for abnormally distributed data. Repeated measures ANOVA of PPI on left
ventricular end-diastolic diameter (LVEDD) and LVEF after TAVI, and stepwise
regression analysis of risk factors for mortality after TAVI on PAR patients was
conducted to identify factors associated with six-month mortality, utilizing
hazard ratios. The Kaplan-Meier survival curve, along with the log-rank test, was
employed to compare six-month mortality, with p
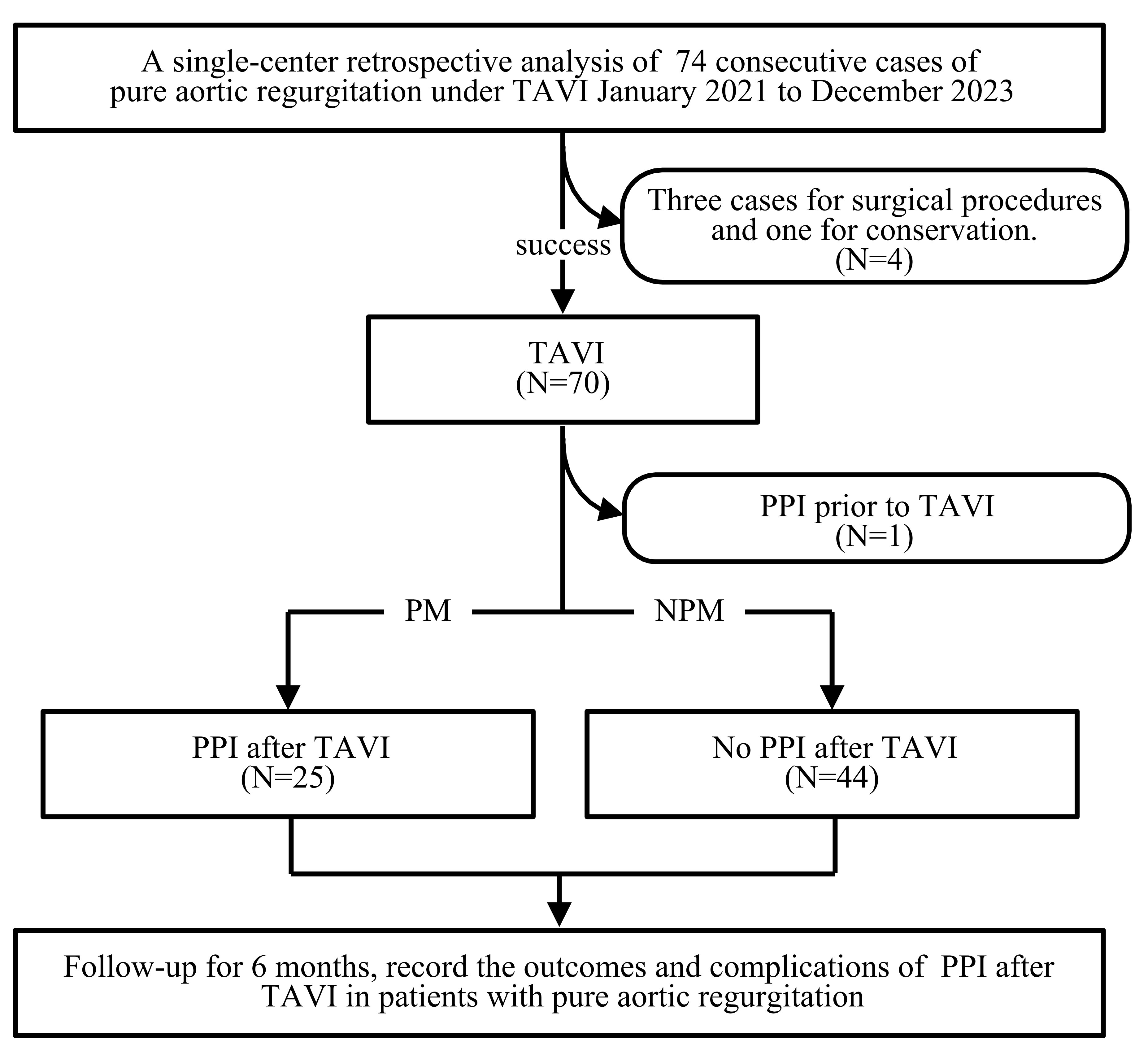 Fig. 1.
Fig. 1.
Study flow diagram. TAVI, transcatheter aortic valve implantation; PPI, permanent pacemaker implantation; PM, permanent pacemaker; NPM, no permanent pacemaker.
In this study, 74 patients with PAR underwent TAVI. After excluding four cases
of failure and one patient with a previously implanted permanent pacemaker, 69
patients with a successfully implanted TAVI were included. Among these, 25
patients required postoperative implantation of a permanent pacemaker and were
classified into the PM group, while 44 patients did not require a pacemaker and
were classified into the NPM group. No significant differences in baseline
characteristics or complications were observed between the two groups (p
| Parameters | NPM (n = 44) | PM (n = 25) | p-value |
| Age (Y) | 73.55 |
73.64 |
0.961 |
| Gender (male, %) | 27 (61.36) | 14 (56.00) | 0.663 |
| BMI (kg/m2) | 23.08 |
22.96 |
0.832 |
| Creatinine (µmol/L) | 111.57 |
98.91 |
0.592 |
| Uric acid (mmol/L) | 315.82 |
344.93 |
0.537 |
| Glucose (mmol/L) | 4.83 |
5.25 |
0.179 |
| Cholesterol (mmol/L) | 3.46 |
3.45 |
0.987 |
| Triglyceride (mmol/L) | 0.98 |
1.10 |
0.517 |
| HDL-c (mmol/L) | 1.29 |
1.26 |
0.758 |
| LDL-c (mmol/L) | 1.80 |
1.86 |
0.787 |
| Lipoprotein(a) (mmol/L) | 140.40 (19.02, 341.10) | 87.00 (13.70, 226.70) | 0.427 |
| Prothrombin time (S) | 14.09 |
12.74 |
0.156 |
| INR | 1.23 |
1.10 |
0.129 |
| APTT (S) | 31.75 |
35.32 |
0.430 |
| Platelet count (109/L) | 134.43 |
134.20 |
0.987 |
| Prosthesis size (mm) | 28.61 |
28.44 |
0.717 |
| NYHA association | 3.18 |
3.00 |
0.316 |
| Hypertension (N, %) | 25 (56.82) | 11 (44.00) | 0.306 |
| Diabetes mellitus (N, %) | 5 (11.36) | 2 (8.00) | 0.976 |
| Cerebral embolism (N, %) | 5 (11.36) | 1 (4.00) | 0.297 |
| CAD (N, %) | 12 (27.91) | 5 (20.00) | 0.468 |
| Atrial fibrillation (N, %) | 16 (36.36) | 6 (24.00) | 0.289 |
| Mitral regurgitation (N, %) | 6 (13.64) | 5 (20.00) | 0.632 |
Y, year; BMI, body mass index; HDL-c, high-density lipoprotein cholesterol; LDL-c,
low-density lipoprotein cholesterol; CAD, coronary artery disease; NYHA, New York
Heart Association; APTT, activated partial thromboplastin time; INR,
international normalized ratio; p
25 patients received single-chamber, dual-chamber, or CRT pacemakers. One
patient was treated with warfarin, three with rivaroxaban, fourteen with aspirin
plus clopidogrel, one with aspirin plus clopidogrel plus rivaroxaban, and five
with aspirin plus clopidogrel plus low molecular weight heparin. There were no
significant differences in various anticoagulation regimens between the two
groups (p
| Parameters | PM (N = 25) | NPM (N = 44) | p-value | |
| Warfarin (N, %) | 1 (4.00) | 2 (4.55) | 0.612 | |
| Rivaroxaban (N, %) | 3 (12.00) | 5 (11.36) | 0.755 | |
| Asprin+Clopidogrel (N, %) | 14 (56.00) | 20 (45.45) | 0.400 | |
| Clopidogrel+Heparin (N, %) | 1 (4.00) | 4 (9.09) | 0.763 | |
| Asprin+Clopidogrel+Rivaroxaban (N, %) | 1 (4.00) | 3 (6.82) | 0.957 | |
| Asprin+Clopidogrel+Heparin (N, %) | 5 (20.00) | 10 (22.73) | 0.792 | |
| Pacemaker | ||||
| VVI (N, %) | 5 (20.00) | - | - | |
| DDD (N, %) | 19 (76.00) | - | - | |
| CRT (N, %) | 1 (4.00) | - | - | |
| Complications | ||||
| Pocket hematoma | 0 | - | - | |
| Pocket infection | 0 | - | - | |
| Electrode displacement | 0 | - | - | |
PAR, pure aortic regurgitation; VVI, ventricular demand pacing; DDD,
dual-chamber pacing; CRT, cardiac resynchronization therapy. p
Patients with PAR who underwent TAVI were followed up for six months. Excluding the eight deceased patients, the remaining 61 were analyzed for changes in baseline echocardiographic parameters, LVEDD and LVEF, at six-month follow-up, and the impact of PPI on these parameters was assessed. Repeated measures ANOVA for changes in LVEDD revealed significant main effects of follow-up time in both PM and NPM groups (p = 0.000), indicating that the LVEDD was significantly reduced after TAVI regardless of PPI (Table 3). LVEF exhibited an upward trend, but repeated measures ANOVA for changes in LVEF revealed no significant main effects of follow-up time in both the PM and NPM groups (p = 0.053), which was also seen in the group effect (p = 0.652) and the interaction effect between group and follow-up time (p = 0.789). This suggests that regardless of PPI, although there was an increase in LVEF during the follow-up period, there were no significant differences between or within the groups (Table 3).
| Parameters | LVEDD (mm) | LVEF (%) | ||
| PM (N = 20) | NPM (N = 41) | PM (N = 20) | NPM (N = 41) | |
| Baseline | 56.50 |
57.23 |
55.81 |
54.84 |
| 3 month follow-up | 52.07 |
53.16 |
56.69 |
54.81 |
| 6 month follow-up | 52.31 |
51.07 |
58.00 |
57.58 |
| Group p value | 0.917 | 0.652 | ||
| Time p value | 0.000* | 0.053 | ||
| Group by time p-value | 0.158 | 0.789 | ||
LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection
fraction; Data are presented as mean
No significant differences in the incidence of all-cause mortality, major
bleeding, or cerebral embolism were observed between the PM and NPM groups six
months post-TAVI (p
| Parameters | PM (N = 25) | NPM (N = 44) | p-value | ||
| Adverse events | 13 (59.09) | 18 (43.18) | 1.486 | 0.223 | |
| Major bleeding | 1 (4.0) | 2 (4.55) | 0.257 | 0.612Δ | |
| Cerebral embolism | 3 (12) | 2 (4.55) | 0.442 | 0.506 | |
| All-cause mortality | 3 (12) | 5 (11.36) | 0.097 | 0.755 | |
| Follow-up | |||||
| one-month readmission | 2 (8) | 3 (6.82) | 0.091 | 0.763 | |
| three-month readmission | 4 (16) | 8 (18.18) | 0.010 | 0.920 | |
| six-month readmission | 6 (24) | 10 (22.73) | 0.014 | 0.904 | |
As depicted in Table 5, a stepwise regression analysis was conducted for all
variables, including patient basic parameters and key observation indicators such
as LVEF, pacemaker implantation, and other variables within the model, with death
as the dependent variable. The findings indicated that age, serum creatinine, and
cerebral infarction significantly increased the risk of mortality, with each unit
increase in cerebral infarction leading to a 47.718-fold increase in the
incidence of death. In contrast, hypertension had a significantly negative impact
on mortality (p = 0.019), whereas pacemaker implantation and LVEF were
not statistically significant (p
| Parameters | Stepwise regression analysis | |||
| coefficient | p-value | odds ratio | 95% CI | |
| Age | 0.238 | 0.020 | 1.268 | 1.038–1.550 |
| Creatinine | 0.010 | 0.032 | 1.010 | 1.001–1.019 |
| Cerebral embolism | 3.865 | 0.015 | 47.718 | 2.091–1088.739 |
| Hypertension | –3.621 | 0.019 | 0.027 | 0.001–0.552 |
McFadden R-squared = 0.378, Cox & Snell R-squared = 0.239, Nagelkerke R-squared = 0.46.
The survival curve indicated that mortality among NPM patients predominantly occurred during the perioperative period. No significant differences in survival rates were observed between the two groups at six months post-TAVI (p = 0.971) (Fig. 2).
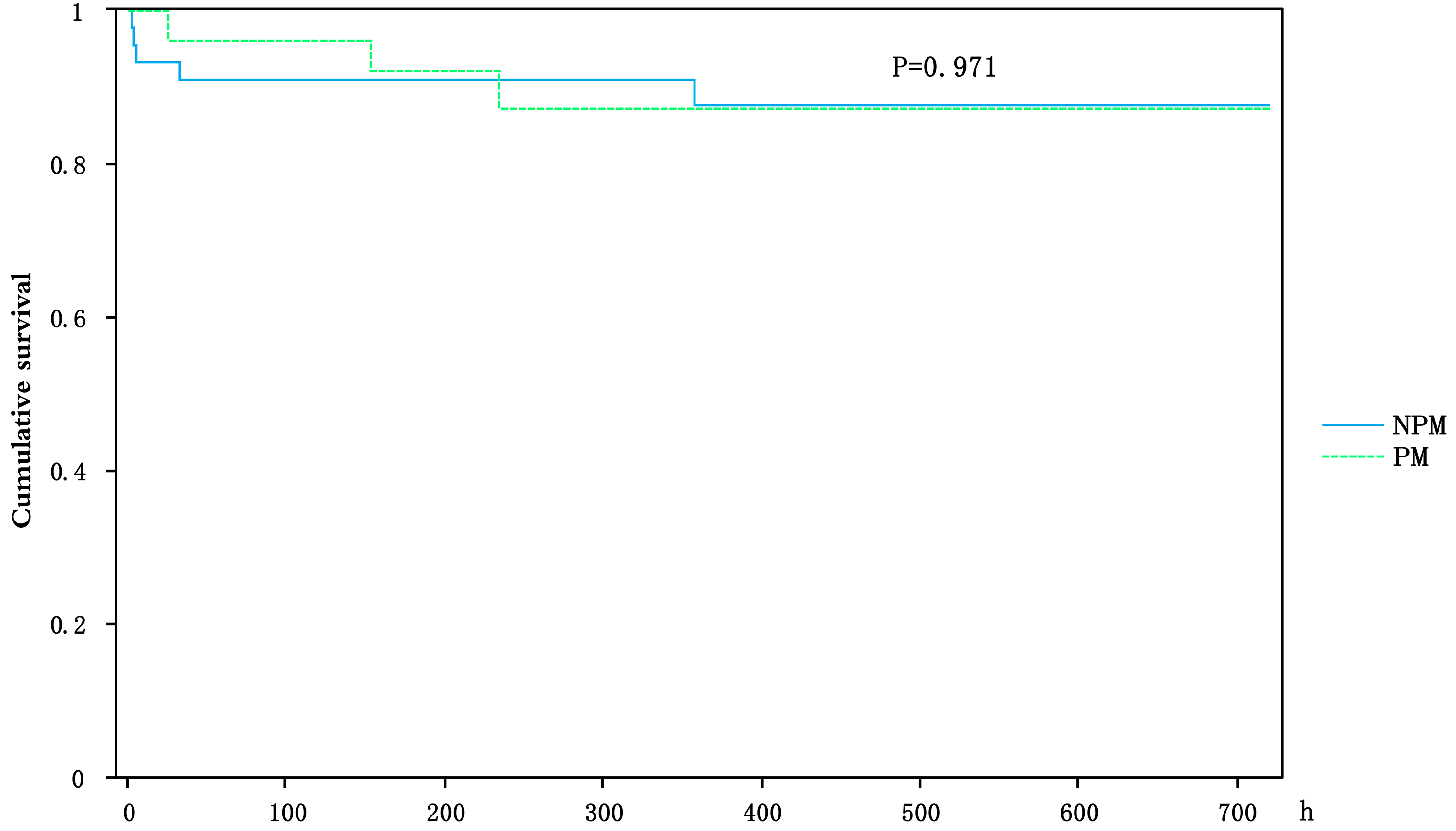 Fig. 2.
Fig. 2.
The impact of PPI or not on survival among pure aortic regurgitation patients after TAVI.
This study represents the first investigation into the effects of PPI on short-term mortality, adverse events, and cardiac function in PAR patients following TAVI. The findings indicate that PPI is not associated with mortality or the incidence of adverse events after six months among PAR patients. Factors such as advanced age, elevated creatinine levels, and prior cerebral embolism were identified as contributors to increased mortality post-TAVI.
Regarding the impact of PPI on cardiac structure and function post-TAVI, the
PACE-TAVI registry revealed that AS patients with a right ventricular pacing
(RVP) ratio of
The relationship between PPI and mortality or rehospitalization rates post-TAVI
remains contentious. Numerous studies have established that PPI correlates with
increased rehospitalization and mortality rates among patients undergoing TAVI
[21, 22]. The PACE-TAVI registry also indicated that patients with an RVP ratio
The debate surrounding whether PPI increases mortality post-TAVI persists, yet
other factors contributing to elevated mortality rates have been identified.
Advanced age and myocardial fibrosis have been shown to increase mortality rates
among AS patients post-TAVI [27]. In a cohort of 500 patients undergoing TAVR,
with a median follow-up of 5.2 years, advanced age, male gender, chronic kidney
disease stage
The small sample size and the short follow-up period constrain the generalizability of our findings and limit our capacity to assess the long-term outcomes effectively. Future research endeavors should aim to incorporate larger cohorts and extended follow-up periods to more accurately determine the effects of PPI on survival and cardiac function in PAR patients post-TAVI. Furthermore, the absence of LVEF stratification in our analysis represents a significant limitation, suggesting a clear need for future studies to explore the nuanced impact of PPI across different LVEF levels. Such larger, longitudinal studies are essential to establish the definitive role of PPI in the management of PAR patients following TAVI, ultimately informing clinical practice and patient care standards.
Our study, the first to examine the short-term impact of PPI on patients with PAR following TAVI, reveals no association between PPI and increased mortality or adverse events within the initial six-month follow-up. Notably, advanced age, elevated creatinine levels, and a history of cerebral infarction emerged as significant predictors of mortality, thereby underscoring their critical role in post-TAVI care. Overall, these findings provide valuable insights into the management strategies for PAR patients in the aftermath of TAVI, highlighting the need for tailored approaches based on individual patient characteristics.
AS, aortic stenosis; CRT, cardiac resynchronization therapy; LBBaP, left bundle branch area pacing; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; PAR, pure aortic regurgitation; PPI, permanent pacemaker implantation; RVP, right ventricular pacing; TAVI, transcatheter aortic valve implantation.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
CJ designed the research, collected data and wrote the manuscript. YZ completed the data analysis, SC and ZG designed the research and revised the manuscript. TG and ST performed the research. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was conducted in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments. All procedures involving human participants were approved by the Scientific Research and New Technology of Wannan Medical College Yijishan Hospital’s Ethics Committee (Grant No. [2024]23), and informed consent was obtained from all participants.
We would like to express their gratitude to EditSprings (https://www.editsprings.cn/) for the expert linguistic services provided.
This study was supported by the Key Specialty Project of Anhui Province Medical and Health (Award Number: KZSJZ008), Anhui Province Quality Engineering Research Project (Award Number: 2023jyxm1238) and Enterprise-commissioned R&D projects (Award Number: KY21610724).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


