1 Department of Cardiology, West China Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
†These authors contributed equally.
Abstract
Background: The impact of cardiac arrest (CA) at admission on the prognosis of patients with acute myocardial infarction (AMI) complicated by cardiogenic shock (CS) remains a subject of debate. Methods: We conducted a retrospective study at West China Hospital from 2018 to 2021, enrolling 247 patients with AMI complicated by CS (AMI-CS). Patients were categorized into CA and non-CA groups based on their admission status. Univariate and multivariate Cox regression analyses were performed, with 30-day and 1-year mortality as the primary endpoints. Kaplan–Meier plots were constructed, and concordance (C)-indices of the Global Registry of Acute Coronary Event (GRACE) score, Intra-aortic Balloon Pump in Cardiogenic Shock (IABP-SHOCK) II score, and IABP-SHOCK II score with CA were calculated. Results: Among the enrolled patients, 39 experienced CA and received cardiopulmonary resuscitation at admission. The 30-day and 1-year mortality rates were 40.9% and 47.0%, respectively. Neither univariate nor multivariate Cox regression analyses identified CA as a significant risk factor for 30-day and 1-year mortality. In C-statistics, the GRACE score exhibited a moderate effect (C-indices were 0.69 and 0.67, respectively), while the IABP-SHOCK II score had a better predictive performance (C-indices were 0.79 and 0.76, respectively) for the 30-day and 1-year mortality. Furthermore, CA did not enhance the predictive value of the IABP-SHOCK II score for 30-day (p = 0.864) and 1-year mortality (p = 0.888). Conclusions: Cardiac arrest at admission did not influence the survival of patients with AMI-CS. Active resuscitation should be prioritized for patients with AMI-CS, regardless of the presence of cardiac arrest.
Keywords
- cardiac arrest
- cardiogenic shock
- acute myocardial infarction
- mortality
Acute myocardial infarction (AMI) complicated by cardiogenic shock (CS) presents a formidable challenge in clinical practice, possessing a mortality rate of approximately 40–50% [1]. Notably, AMI patients with cardiac arrest (CA) are more prone to developing CS. In the subset of AMI complicated by CS (AMI-CS), the incidence of CA ranges from 28.5 to 51% [2, 3, 4, 5], and CA is frequently inferred as the primary cause of death in cases of AMI complicated by CS [6].
While previous studies have established CA as a significant risk factor for mortality in patients with acute coronary syndrome (ACS) [7, 8, 9, 10], this association exhibits inconsistency within AMI-CS patients [2, 3, 11]. Some studies suggest that CA may not independently influence all-cause in-hospital or long-term mortality among patients hospitalized with AMI-CS [2, 3]. Notably, these studies reported low proportions of coronary angiography (61.3%) and revascularization (53.3%), potentially compromising the survival probability of CA patients [2, 3]. In contrast, a comprehensive registry study demonstrated higher rates of coronary angiography (88.8%) and revascularization (77.2%) in AMI-CS patients [12].
Recent advancements in acute percutaneous coronary intervention (PCI) and circulatory support devices have significantly improved the short-term prognosis of AMI patients, particularly through early revascularization [1]. Consequently, in the context of active revascularization, the association between CA and the prognosis of patients with AMI-CS has become a subject of debate. In this study, we aimed to assess the impact of CA on AMI-CS outcomes by analyzing data collected from September 2018 to November 2021. Notably, the study cohort exhibited high proportions of coronary angiography (89.5%) and revascularization (80.2%), providing valuable insights into the contemporary landscape of AMI-CS management.
This retrospective study involved 247 patients diagnosed with AMI-CS treated at
the West China Hospital of Sichuan University between September 2018 and November
2021. The inclusion criteria encompassed patients within the Society for
Cardiovascular Angiography and Interventions (SCAI) shock stages B to E [12, 13].
Cardiac shock was defined as a primary cardiac disorder leading to hypotension
(systolic blood pressure
Demographic characteristics such as body mass index (BMI), blood pressure, heart rate, and respiration status were obtained from nursing records at admission. Medical history (history of hypertension, diabetes, stroke, dyslipidemia, coronary artery disease, peripheral artery disease, and chronic kidney disease), mechanically supported therapies (including circulatory and mechanical respiratory data), and type of AMI were obtained from hospital records. Laboratory parameters were routinely measured after each patient’s admission, and baseline biochemical tests and blood gas results were collected. Coronary artery lesions and intervention information were obtained from in-hospital coronary angiography images and reports. At admission, the left ventricular ejection fraction (LVEF) was evaluated by echocardiography. The Global Registry of Acute Coronary Event (GRACE) and Intra-aortic Balloon Pump in Cardiogenic Shock (IABP-SHOCK) II scores were calculated according to the baseline data of the patients at admission [4, 10]. The GRACE scores of the patients were calculated using the following variables: age, systolic blood pressure, heart rate, serum creatinine level, ST-segment deviation of the electrocardiogram, and cardiac arrest at admission [10]. The IABP-SHOCK Ⅱ scores were calculated by variables such as age, arterial lactate, serum glucose, creatine, history of stroke, and a thrombolysis in myocardial infarction (TIMI) flow grade reaching three after PCI [4].
The primary endpoint was death from any cause within 30 days and 1 year. Moreover, we followed the brain injury of patients with CA, while the severity of hypoxic brain damage was evaluated using cerebral performance categories (CPC) [15]. Follow-up information was obtained through telephonic interviews, medical charts, and outpatient visits. Hospital records were used to corroborate all data.
The Kolmogorov–Smirnov test was used to assess data distribution. Continuous
variables are presented as the mean or median with each range, and we used
independent t-tests or the Kruskal–Wallis test to compare variables
between groups. Qualitative variables are presented as frequencies and
corresponding percentages, and chi-squared or Fisher’s exact tests were used to
compare variables between groups. Associations between the clinical variables and
endpoints were first assessed using univariate Cox regression analysis. Variables
significantly associated with 30-day and 1-year mortality in univariate testing
(p
Since the mortality rate of AMI-CS was about 40% [1], prior estimates of power
were conducted assuming a hazard ratio (HR) of 1.3 for CA; this power analysis
showed that a sample size of 247 participants would result in a power of 0.88
(
Table 1 summarizes the baseline and clinical characteristics of both the CA and
non-CA groups within the cohort of 247 patients experiencing AMI-CS. Among these patients, 39
(15.8%) experienced CA. Most of the cohort was male (74.5%), with no
significant difference in sex distribution between the CA and non-CA groups. The
average age of all patients was 68 years, with the CA group showing a slightly
younger profile (66
| Total (n = 247) | CA group (n = 39) | Non-CA group (n = 208) | p-value | ||
| Demographic data | |||||
| Age, yrs | 68 (30–89) | 66 (30–89) | 69 (32–89) | 0.281 | |
| Male | 184/247 (74.5) | 29/39 (74.4) | 155/208 (74.6) | 0.983 | |
| BMI, kg/m |
23.4 (13.9–35.6) | 23.7 (16.8–31.3) | 23.4 (13.9–35.6) | 0.613 | |
| SBP, mmHg | 91 (51–135) | 87 (52–122) | 92 (51–135) | 0.044 | |
| MAP, mmHg | 70.7 (40.7–105) | 65.6 (40.7–100) | 71.7 (41–105) | 0.002 | |
| Heart rate, bpm | 97 (39–180) | 100 (52–180) | 96 (39–151) | 0.305 | |
| Cardiovascular risk factors/CVD (%) | |||||
| Smoking | 115/247 (46.6) | 20/39 (51.3) | 95/208 (45.7) | 0.519 | |
| Arterial hypertension | 119/247 (48.2) | 13/39 (33.3) | 106/208 (50.9) | 0.043 | |
| Diabetes mellitus | 77/247 (31.2) | 9/39 (23.1) | 68/208 (32.7) | 0.234 | |
| History of CAD | 48/247 (19.4) | 7/39 (17.9) | 41/208 (19.7) | 0.798 | |
| History of Stroke | 19/247 (7.7) | 2/39 (5.1) | 17/208 (8.2) | 0.513 | |
| Dyslipidemia | 40/247 (16.3) | 3/39 (7.7) | 37/208 (17.8) | 0.116 | |
| Known PAD | 7/247 (2.8) | 0/39 (0) | 7/208 (3.4) | 0.245 | |
| CKD | 19/247 (7.7) | 2/39 (5.1) | 17/214 (8.2) | 0.513 | |
| Laboratory results | |||||
| Arterial lactate, mmol/L | 4.7 (1.0–20) | 6.9 (1.1–20) | 4.3 (1.0–20) | 0.004 | |
| Glucose, mmol/L | 12.06 (3.17–37.47) | 13.34 (3.17–37.47) | 11.82 (3.59–35.91) | 0.176 | |
| Serum creatinine, µmol/L | 149.9 (51–1014) | 177.4 (57–665) | 144.7 (51–1014) | 0.094 | |
| TnT, ng/L | 7295.6 (97.3–10,000) | 7346.7 (699–10,000) | 7285.9 (97.3–10,000) | 0.906 | |
| STEMI (%) | 175/247 (70.9) | 30/39 (76.9) | 145/208 (69.7) | 0.363 | |
| Anterior STEMI (%) | 112/247 (45.3) | 25/39 (64.1) | 87/208 (41.8) | 0.010 | |
| Ventilation (%) | 186/247 (75.3) | 36/39 (92.3) | 150/208 (72.1) | 0.007 | |
| CAG (%) | 221/247 (89.5) | 34/39 (87.2) | 187/208 (89.9) | 0.611 | |
| Coronary lesions (%) | 0.069 | ||||
| Mono-vessel disease | 55/219 (25.1) | 12/34 (35.3) | 43/185 (23.2) | ||
| Bi-vessel disease | 61/219 (27.9) | 9/34 (26.5) | 52/185 (28.1) | ||
| Multi-vessel disease | 103/219 (47.0) | 13/34 (38.2) | 90/185 (48.7) | ||
| Revascularization (%) | 198/247 (80.2) | 34/39 (87.2) | 164/208 (79.4) | 0.231 | |
| Complete revascularization (%) | 94/247 (38.1) | 18/39 (46.1) | 76/208 (36.5) | 0.256 | |
| TIMI flow grade 3 after PCI (%) | 184/221 (83.3) | 29/34 (85.2) | 155/187 (82.9) | 0.730 | |
| IABP (%) | 104/247 (42.1) | 23/39 (59.0) | 81/208 (38.9) | 0.020 | |
| ECMO (%) | 6/247 (2.4) | 2/39 (5.1) | 4/208 (1.9) | 0.233 | |
| GRACE score | 193 (122–265) | 215 (157–265) | 189 (122–237) | 0.000 | |
| IABP-SHOCK II score (%) | 0.130 | ||||
| 0–2 | 137/219 (62.6) | 17/35 (48.6) | 120/184 (65.2) | ||
| 3–4 | 54/219 (24.6) | 9/35 (25.7) | 45/184 (24.5) | ||
| 5–9 | 28/219 (12.8) | 9/35 (25.7) | 19/184 (10.3) | ||
| LVEF, % | 40 (15–75) | 41 (16–75) | 40 (15–70) | 0.856 | |
Abbreviations: n, number; AMI, acute myocardial infarction; CS, cardiogenic shock; CA, cardiac arrest; BMI, body mass index; SBP, systolic blood pressure; MAP, mean arterial pressure; bpm, beats per minute; CVD, coronary vascular disease; CAD, coronary artery disease; PAD, peripheral artery disease; CKD, chronic kidney disease; TnT, troponin T; STEMI, ST-segment elevation myocardial infarction; CAG, coronary angiography; TIMI, thrombolysis in myocardial infarction; PCI, percutaneous coronary intervention; IABP, intra-aortic balloon pump; ECMO, extracorporeal membrane oxygenation; GRACE, Global Registry of Acute Coronary Event; IABP-SHOCK, Intra-aortic Balloon Pump in Cardiogenic Shock; LVEF, left ventricular ejection fraction.
Within the AMI-CS cohort, 101 patients succumbed within 30 days and 116 patients within 1 year, resulting in 30-day and 1-year mortality rates of 40.9% and 47.0%, respectively. In both timeframes, the majority of deaths were attributed to cardiac causes, encompassing sudden cardiac death, ventricular tachycardia or fibrillation, refractory heart failure, or cardiogenic shock, with 93.1% at 30 days and 91.4% at 1 year (Table 2). Survival analysis, as depicted in Fig. 1, did not reveal a significant difference between the groups with and without CA. The overall rate of hypoxic brain damage during the hospital stay was 4.9%. Notably, the CA group exhibited a higher incidence of brain injury (25.6% vs. 1.0%), with 58.3% of these patients succumbing within 30 days and 75% within 1 year. For those surviving cardiac arrest with hypoxic brain injury, the extent of damage was generally mild, as indicated by CPC scores ranging from 1 to 3.
| Total | CA group | Non-CA group | ||
| Death within 30 days (%) | 101/247 (40.9) | 21/39 (53.9) | 80/208 (38.5) | |
| Cardiac death | 94/101 (93.1) | 20/21 (95.2) | 74/80 (92.5) | |
| Non-cardiac death | 7/101 (6.9) | 1/21 (4.8) | 6/80 (7.5) | |
| Death within 1 year (%) | 116/247 (47.0) | 23/39 (59.0) | 93/208 (44.7) | |
| Cardiac death | 106/116 (91.4) | 20/23 (87.0) | 86/93 (92.5) | |
| Non-cardiac death | 10/116 (8.6) | 3/23 (13.0) | 7/93 (7.5) | |
| Brain injury (%) | 12/247 (4.9) | 10/39 (25.6) | 2/208 (1.0) | |
| CPC score (1–3) | 3/247 (1.2) | 3/39 (7.7) | 0/208 (0) | |
| CPC score (4–5) | 9/247 (3.7) | 7/39 (17.9) | 2/208 (1.0) | |
Abbreviations: AMI, acute myocardial infarction; CS, cardiogenic shock; CA, cardiac arrest; CPC, cerebral performance category.
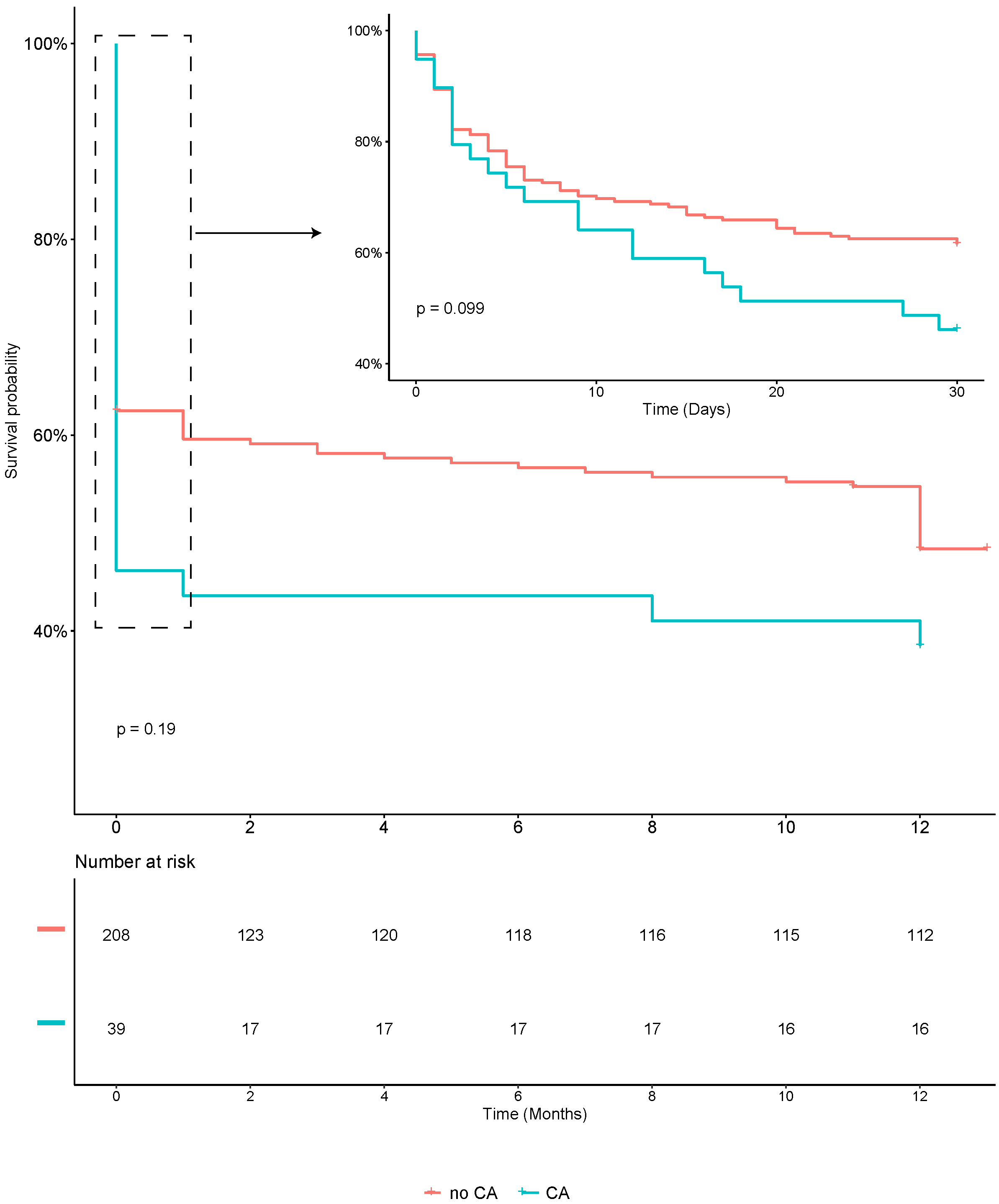 Fig. 1.
Fig. 1.Kaplan–Meier plot of 30-day and 1-year mortality in AMI-related CS patients with and without CA. Abbreviations: AMI, acute myocardial infarction; CS, cardiogenic shock; CA, cardiac arrest.
In the univariate Cox regression analysis, CA did not exhibit a significant
association with either short- or long-term mortality, with hazard ratios (HRs)
of 1.48 (p = 0.108) and 1.43 (p = 0.127), respectively (Tables 3,4). After adjustment by multivariate analysis, CA continued to lack a
significant association with both short- and long-term mortality (HR = 0.88,
p = 0.669; HR = 1.04, p = 0.881, respectively; Tables 3,4 and
Supplementary Table 1). Exploring the univariate analysis, older age, female
gender, higher heart rate, lower SBP and MAP, elevated baseline creatinine,
arterial lactate, and glucose levels, presence of hypertension and CKD, TIMI flow
grade
| Univariable analysis of 30-day mortality | Multivariable analysis of 30-day mortality | |||
| HR (95% CI) | p-value | HR (95% CI) | p-value | |
| Male | 0.51 (0.34–0.77) | 0.001 | ||
| Age, yrs | 1.04 (1.02–1.06) | 0.000 | 1.04 (1.02–1.06) | 0.001 |
| BMI, kg/m |
0.97 (0.91–1.03) | 0.293 | ||
| SBP, mmHg | 0.98 (0.97–1.00) | 0.011 | ||
| MAP, mmHg | 0.96 (0.95–0.98) | 0.003 | ||
| Heart rate, bpm | 1.01 (1.00–1.02) | 0.009 | ||
| CA | 1.48 (0.92–2.40) | 0.108 | 0.88 (0.48–1.60) | 0.669 |
| Ventilation | 14.4 (4.58–45.6) | 0.000 | 9.84 (2.38–40.7) | 0.002 |
| Arterial hypertension | 2.36 (1.57–3.55) | 0.000 | ||
| Diabetes mellitus | 1.38 (0.92–2.06) | 0.122 | ||
| History of CAD | 1.05 (0.65–1.72) | 0.835 | ||
| History of stroke | 1.07 (0.52–2.19) | 0.862 | ||
| Dyslipidemia | 0.98 (0.58–1.68) | 0.952 | ||
| Known PAD | 1.50 (0.55–4.08) | 0.427 | ||
| CKD | 2.28 (1.27–4.08) | 0.006 | ||
| Arterial lactate, mmol/L | 1.15 (1.11–1.19) | 0.000 | 1.15 (1.09–1.20) | 0.000 |
| Glucose, mmol/L | 1.06 (1.03–1.08) | 0.000 | ||
| Serum creatinine, µmol/L | 3.18 (2.37–4.26) | 0.000 | 2.24 (1.43–3.52) | 0.000 |
| TnT, ng/L | 1.000 (0.999–1.001) | 0.612 | ||
| STEMI | 0.76 (0.50–1.14) | 0.178 | ||
| Anterior STEMI | 0.98 (0.66–1.45) | 0.930 | ||
| Revascularization | 0.26 (0.17–0.39) | 0.000 | ||
| Complete revascularization | 0.45 (0.29–0.70) | 0.000 | ||
| TIMI flow grade 3 after PCI | 0.17 (0.11–0.28) | 0.000 | 0.20 (0.11–0.31) | 0.000 |
| IABP | 1.17 (0.79–1.73) | 0.428 | ||
| ECMO | 3.77 (1.65–8.33) | 0.002 | ||
| LVEF, % | 0.96 (0.94–0.98) | 0.000 | ||
| GRACE score | 1.03 (1.02–1.04) | 0.000 | ||
| IABP-SHOCK II score | 1.64 (1.47–1.83) | 0.000 | ||
Abbreviations: AMI, acute myocardial infarction; CS, cardiogenic shock; HR, hazard ratio; CI, confidence interval; BMI, body mass index; SBP, systolic blood pressure; MAP, mean arterial pressure; CA, cardiac arrest; CAD, coronary artery disease; PAD, peripheral artery disease; CKD, chronic kidney disease; TnT, troponin T; STEMI, ST-segment elevation myocardial infarction; TIMI, thrombolysis in myocardial infarction; PCI, percutaneous coronary intervention; IABP, intra-aortic balloon pump; ECMO, extracorporeal membrane oxygenation; GRACE, Global Registry of Acute Coronary Event; IABP-SHOCK, Intra-aortic Balloon Pump in Cardiogenic Shock; LVEF, left ventricular ejection fraction.
| Univariable analysis of 1 year mortality | Multivariable analysis of 1 year mortality | |||
| HR (95% CI) | p-value | HR (95% CI) | p-value | |
| Male | 0.55 (0.37–0.81) | 0.002 | ||
| Age, yrs | 1.03 (1.02–1.05) | 0.000 | 1.03 (1.01–1.05) | 0.010 |
| BMI, kg/m |
0.95 (0.90–1.00) | 0.066 | ||
| SBP, mmHg | 0.99 (0.97–1.00) | 0.07 | ||
| MAP, mmHg | 0.97 (0.96–0.99) | 0.001 | ||
| Heart rate, bpm | 1.01 (1.00–1.02) | 0.005 | ||
| CA | 1.43 (0.90–2.25) | 0.127 | 1.04 (0.59–1.85) | 0.881 |
| Ventilation | 6.42 (3.13–13.2) | 0.000 | 3.56 (1.60–7.91) | 0.002 |
| Arterial hypertension | 2.39 (1.64–3.50) | 0.000 | 1.60 (1.01–2.52) | 0.045 |
| Diabetes mellitus | 1.42 (0.98–2.08) | 0.06 | ||
| History of CAD | 1.05 (0.67–1.66) | 0.829 | ||
| History of Stroke | 1.04 (0.53–2.05) | 0.915 | ||
| Dyslipidemia | 0.95 (0.57–1.57) | 0.837 | ||
| Known PAD | 1.33 (0.49–3.60) | 0.579 | ||
| CKD | 2.01 (1.13–3.59) | 0.018 | ||
| Baseline arterial lactate, mmol/L | 1.15 (1.11–1.19) | 0.000 | 1.12 (1.07–1.18) | 0.000 |
| Baseline glucose, mmol/L | 1.06 (1.03–1.08) | 0.000 | ||
| Baseline serum creatinine, µmol/L | 2.97 (2.21–4.00) | 0.000 | 2.08 (1.37–3.15) | 0.001 |
| TnT, ng/L | 1.11 (0.85–1.44) | 0.435 | ||
| STEMI | 0.75 (0.51–1.10) | 0.144 | ||
| Anterior STEMI | 0.91 (0.63–1.32) | 0.622 | ||
| Revascularization | 0.29 (0.19–0.43) | 0.000 | ||
| Complete revascularization | 0.42 (0.28–0.65) | 0.000 | ||
| TIMI flow grade 3 after PCI | 0.21 (0.13–0.33) | 0.000 | 0.23 (0.14–0.37) | 0.000 |
| IABP | 1.21 (0.84–1.74) | 0.317 | ||
| ECMO | 3.65 (1.60–8.35) | 0.002 | ||
| LVEF, % | 0.96 (0.94–0.98) | 0.000 | ||
| GRACE score | 1.03 (1.02–1.03) | 0.000 | ||
| IABP-SHOCK II score | 1.59 (1.43–1.76) | 0.000 | ||
Abbreviations: AMI, acute myocardial infarction; CS, cardiogenic shock; HR, hazard ratio; CI, confidence interval; BMI, body mass index; SBP, systolic blood pressure; MAP, mean arterial pressure; CA, cardiac arrest; CAD, coronary artery disease; PAD, peripheral artery disease; CKD, chronic kidney disease; TnT, troponin T; STEMI, ST-segment elevation myocardial infarction; TIMI, thrombolysis in myocardial infarction; PCI, percutaneous coronary intervention; IABP, intra-aortic balloon pump; ECMO, extracorporeal membrane oxygenation; GRACE, Global Registry of Acute Coronary Event; IABP-SHOCK, Intra-aortic Balloon Pump in Cardiogenic Shock; LVEF, left ventricular ejection fraction.
In the context of C-statistics, the GRACE score exhibited a moderate predictive value for both 30-day and 1-year mortality, with an area under the curve (AUC) of 0.69 [95% confidence interval (CI): 0.64 to 0.75, p = 0.000] and 0.67 (95% CI: 0.62 to 0.72, p = 0.000), respectively. Conversely, the IABP-SHOCK Ⅱ score demonstrated a high predictive value for both short- and long-term mortality, recording AUC values of 0.79 (95% CI: 0.74 to 0.84, p = 0.000) and 0.76 (95% CI: 0.71 to 0.81, p = 0.000), respectively. Comparatively, the GRACE score exhibited lower predictive performance than the IABP-SHOCK II score in forecasting short- and long-term mortality among AMI-related CS patients (p = 0.003). Additionally, incorporating CA into the IABP-SHOCK II score did not improve the predictive power for either short- or long-term mortality (p = 0.863 and 0.888, respectively; Table 5).
| Models to predict 30-day death | Models to predict 1 year death | |||
| AUC (95% CI) | p-value | AUC (95% CI) | p-value | |
| GRACE score | 0.69 (0.64–0.75) | 0.000 | 0.67 (0.62–0.72) | 0.000 |
| IABP-SHOCK II score | 0.79 (0.74–0.84) | 0.000 | 0.76 (0.71–0.81) | 0.000 |
| IABP-SHOCK II score + CA | 0.79 (0.74–0.84) | 0.000 | 0.76 (0.71–0.81) | 0.000 |
Abbreviations: AMI, acute myocardial infarction; CS, cardiogenic shock; AUC, area under the curve; CI, confidence interval; GRACE, Global Registry of Acute Coronary Event; IABP-SHOCK, Intra-aortic Balloon Pump in Cardiogenic Shock; CA, cardiac arrest.
The primary finding of this study was that CA did not emerge as an independent risk factor for either short- or long-term mortality in AMI-CS patients. Despite this, it is noteworthy that CA showed a significant association with the occurrence of brain injury in this patient population. While CA is acknowledged as a crucial element in the GRACE scoring system, our results indicate that the GRACE risk score provides only a moderate predictive value for individuals with AMI-CS. Furthermore, including CA as a risk factor in the IABP-SHOCK II scoring system did not improve its predictive accuracy for either the short- or long-term mortality assessment among AMI-CS patients. This suggests that the predictive value of CA within established risk models may not be as impactful as initially anticipated in shaping the prognosis of individuals with AMI-CS.
Cardiogenic shock after AMI poses a significant threat to life, with an overall
30-day mortality rate of approximately 40–50% [17]. Our study aligns with
existing literature, revealing a comparable 30-day mortality rate of 40.7% in
patients with AMI-CS. Historically, CA has been identified as a substantial risk
factor for mortality in patients with ACS [3, 4, 5, 6]. While the GRACE score, a widely
utilized tool for assessing death risk in ACS patients, includes CA as an
important predictive factor [18], limited data exists on the correlation between
CA and mortality, specifically in AMI-CS patients [11]. Current risk prediction
models for CS, such as the IABP-SHOCK Ⅱ score, CARD-SHOCK risk score, SAVE, and
ENCOURAGE score, notably omit CA as a risk factor [17]. Our study underscores
that CA is not a significant prognostic factor for short- and long-term mortality
in AMI-CS patients. Instead, age, lactate level, creatinine level, a TIMI grade
This study holds significant clinical implications on multiple fronts. Firstly, it contributes valuable data to the ongoing exploration of whether CA is a prognostic factor for mortality in patients with AMI-CS. This finding emphasizes the importance of proactive rescue efforts for such patients, urging the public to engage in life-saving interventions actively. In China, where awareness regarding cardiac arrest and CPR remains limited [27, 28], our study underscores the critical need for heightened public education on CA and CPR. Importantly, our results suggest that surviving CA does not independently jeopardize the prognosis of AMI-CS patients. Thus, advocating for effective CPR becomes even more imperative, particularly in regions where a substantial number of patients grapple with AMI and cardiogenic shock. Secondly, our study poses a challenge to the GRACE score in predicting outcomes in AMI-CS patients, revealing its limited predictive capability. This observation prompts a reassessment of the GRACE score’s applicability, suggesting that it may not be universally suitable for all patients with ACS. This insight calls for a critical examination of existing risk assessment tools and the potential development of more tailored predictive models for specific subsets of ACS patients, particularly those confronting the complexities of AMI-CS.
While providing valuable insights, this study has limitations. Firstly, the observed rate of CA in our study was 15.8%, lower than previously reported rates [2, 3, 4, 5, 29]. This discrepancy might be attributed to excluding patients who did not survive until hospital admission, potentially leading to an underestimation of the true incidence of CA. The exclusion criteria, particularly limiting CPR time to 30 minutes, aimed to ensure a focus on genuine cases of AMI-CS. Secondly, despite not reaching statistical significance, the 30-day and 1-year mortality rates appeared higher in the CA group (53.9% vs. 38.5% and 59% vs. 44.7%, respectively). While not statistically conclusive, these findings may still bear clinical significance. Previous studies noted comparable outcomes, with one reporting a 62.7% mortality rate at 7 days for the cardiogenic shock population with CA, surpassing that of the non-CA group [3]. Another study, encompassing 1573 patients, identified a relatively higher short-term mortality risk in the CA group, though long-term mortality risk did not reach statistical significance (HR = 1.19, p = 0.055) [30]. Indeed, it is essential to acknowledge these trends, even without statistical significance. Thirdly, our study is limited by its retrospective, single-center design, which may limit the generalizability of findings to the broader Chinese population. However, the study population adhered to the criteria established by large prospective randomized controlled trials of AMI-CS [26, 29], and patients spanned various SCAI stages. Fourthly, the lack of exact timing and detailed information on the cardiac arrest events constitutes another limitation. Nevertheless, the meticulous selection of comprehensive clinical data and a robust sample size, pre-determined through careful sample size estimation, mitigate the likelihood of results occurring by chance. Despite these limitations, the study’s findings contribute valuable insights into the complex interplay of cardiac arrest and mortality in the context of AMI-CS.
In the context of AMI-CS in China, cardiac arrest has emerged as a non-contributory factor in predicting both short- and long-term mortality. Contrary to its significant impact on other cardiac conditions, CA does not independently pose an elevated risk in AMI-CS patients. This finding underscores the importance of active CPR efforts for patients facing AMI-CS, as survival from CA does not inherently compromise their overall prognosis. These results advocate for a proactive and vigorous approach to CPR in this specific patient cohort, emphasizing the need for continued efforts to enhance public awareness and responses to cardiac emergencies in the Chinese population.
AMI, acute myocardial infarction; CS, cardiogenic shock; AMI-CS, AMI complicated by CS; CA, cardiac arrest; GRACE, Global Registry of Acute Coronary Event; IABP-SHOCK, Intra-aortic Balloon Pump in Cardiogenic Shock; ACS, acute coronary syndrome; PCI, percutaneous coronary intervention; SCAI, cardiovascular angiography, and intervention; VT, ventricular tachycardia; CPR, cardiopulmonary resuscitation; ROSC, return of spontaneous circulation; BMI, body mass index; LVEF, left ventricular ejection fraction; TIMI, thrombolysis in myocardial infarction; CPC, cerebral performance categories; HR, hazard ratio; SBP, systolic blood pressure; MAP, mean arterial pressure; CAD, coronary artery disease; PAD, peripheral artery disease; CKD, chronic kidney disease; STEMI, ST-segment elevation myocardial infarction; IABP, intra-aortic balloon pump; AUC, area under the curve.
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
QFX: Conceptualization, Methodology, Software, Writing-Original draft preparation. XW: Conceptualization, revise-original draft preparation. SW: Data collection, Writing-Original draft preparation, Funding acquisition. YX and YY: Data collection, Writing-Original draft preparation. FYH: Conceptualization, Software, Writing-Reviewing, Funding acquisition. MC: Conceptualization, Supervision, Funding acquisition, Writing-Reviewing and Editing critically. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
We declare that the study protocol received approval from the Ethics Committee of West China Hospital, Sichuan University (Chengdu, China; approval number: 2021–1770). All participants provided written informed consent.
Not applicable.
This work was supported by the National Natural Science Foundation of China
(81970325, and 82170375); Key Research and Development Project of Science &
Technology Department of Sichuan Province (2022ZDZX0020); Chinese Medical
Association Cardiovascular Branch (CSC) Clinical Research Special Fund Project
(CSCF2020B04); “1
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

