1 Graduate School of Pharmaceutical Sciences, The University of Osaka, 565-0871 Suita, Osaka, Japan
2 School of Pharmaceutical Sciences, The University of Osaka, 565-0871 Suita, Osaka, Japan
3 Institute for Advanced Co-Creation Studies, The University of Osaka, 565-0871 Suita, Osaka, Japan
4 Department of Endocrine Pharmacology, Tokyo University of Pharmacy and Life Sciences, 192-0392 Hachioji, Tokyo, Japan
5 Graduate School of Medicine, The University of Osaka, 565-0871 Suita, Osaka, Japan
6 Global Center for Medical Engineering and Informatics, The University of Osaka, 565-0871 Suita, Osaka, Japan
7 Institute for Open and Transdisciplinary Research Initiatives, The University of Osaka, 565-0871 Suita, Osaka, Japan
†These authors contributed equally.
Abstract
The placenta plays important roles in pregnancy maintenance and fetal development, and chemical-induced functional or structural abnormalities can lead to adverse pregnancy outcomes. However, information on the placental effects of chemicals remains limited. To help address this gap, this study aimed to investigate the effects of two model chemicals, phenytoin and phenobarbital, on syncytialization (the fusion of cytotrophoblasts into multinucleated syncytiotrophoblasts), a critical process in placental development, using the human choriocarcinoma cell line BeWo.
Phenytoin and phenobarbital, anticonvulsant drugs known to cause major congenital malformations, were each co-treated with forskolin, which promotes syncytialization in BeWo cells, for 48 h.
Evaluation of cell fusion showed that phenytoin significantly suppressed forskolin-induced luciferase activity, whereas phenobarbital did not. Enzyme-linked immunosorbent assay showed that the concentration of human chorionic gonadotropin beta (hCGβ) in the cell culture supernatant was decreased in phenytoin-treated syncytialized BeWo cells but increased in phenobarbital-treated cells. Western blotting also showed a similar pattern in the hCG protein expression level.
Collectively, these results indicate that phenytoin suppresses the process of syncytialization, whereas phenobarbital does not affect cell fusion and instead enhances hCG production.
Keywords
- anticonvulsant drug
- phenytoin
- phenobarbital
- placenta
- syncytialization
Pregnant women and fetuses are vulnerable to chemicals, as exposure can cause adverse pregnancy outcomes, such as miscarriage and congenital abnormalities [1, 2]. However, there are limitations on women avoiding exposure to chemicals when pregnant; for example, as many as 19 chemicals, including benzophenone, pesticides, and phthalates, were detected in about 90% or more of pregnant women [3]. Thus, it is important to understand the effects of chemicals on pregnancy for the maintenance of a normal pregnancy and for the growth and development of the fetus. However, reproductive and developmental toxicity assessments mostly focus on signs of fetal toxicity, and those focusing on the placenta are limited in scope [4].
The placenta develops between 3 and 12 weeks of pregnancy [5] and plays essential roles, including protecting the fetus from external substances via the placental barrier, exchanging gas and nutrients between fetus and mother, and producing placental hormones necessary for pregnancy maintenance [6]. During placentation, placental syncytialization (the fusion of cytotrophoblasts into multinucleated syncytiotrophoblasts) is a critical process for placental development [7]. Normal progression of syncytialization allows the placenta to form a layered structure and to acquire its hormone-producing capacity [6]. However, abnormalities in the progression of syncytialization are reported to be associated with the development of pregnancy-specific diseases such as preeclampsia [8]. Moreover, considering that the development of preeclampsia has been reported to negatively affect fetal neurodevelopment [9], syncytialization is essential not only in the structural and functional maturation of the placenta but also in fetal development. Thus, reproductive and developmental toxicity caused by chemicals needs to be assessed with a focus on its effects on the process of placental development.
Phenytoin and phenobarbital are used as anticonvulsant drugs and are first-line drugs in developing countries, primarily because they are inexpensive [10]. It has been reported that the rate of births with major congenital malformations was about 5–9% in pregnant women taking phenytoin and about 6–9% in those taking phenobarbital, compared to about 3% in unexposed pregnancies [11]. The incidence of major congenital malformations in pregnant women treated with levetiracetam, oxcarbazepine, carbamazepine, topiramate, phenytoin, phenobarbital, and valproic acid was also reported to be higher for phenytoin and phenobarbital after valproic acid [11]. While the effects of valproic acid on placental formation and function have been reported [12, 13], the effects of phenytoin and phenobarbital on the placenta and the mechanism of toxicity remain unclear. To understand how the fetal toxicity of phenytoin and phenobarbital results in adverse outcomes, we investigated their effect on the process of syncytialization by using the human choriocarcinoma cell line BeWo, a widely used model of the trophoblast syncytialization, in which BeWo cells, which have cytotrophoblastic properties, are induced to cell fusion by using forskolin, an activator of the cAMP signaling pathway [14, 15].
The human choriocarcinoma cell line BeWo (JCRB9111; Osaka, Japan) was cultured
in 10% heat inactivated fetal bovine serum (S00CO; Biosera, Nuaille, France) and
1% (v/v) penicillin-streptomycin-amphotericin B suspension (161-23181; Fujifilm
Wako Pure Chemical, Osaka, Japan) in Ham’s F-12 nutrient mixture (087-08335;
Fujifilm Wako Pure Chemical). The cell was authenticated by morphological
observation. Mycoplasma contamination tests were confirmed to be negative. The
cells were maintained at 37 °C and
BeWo cells were seeded at 1.5
Cell fusion was evaluated by using the split luciferase based HiBiT system [16].
NanoLuc, a shrimp-derived, small, bright luciferase [17], was divided into a
C-terminal 11-amino acid peptide (HiBiT) and an 18-kDa polypeptide (LgBiT) [18].
HiBiT binds to LgBiT and recovers luciferase activity [18].
mCherry-labeled-HiBiT- and GFP-labeled-LgBiT-expressing BeWo cells were kindly
provided by Dr. Mikihiro Yoshie, Tokyo University of Pharmacy and Life Sciences
[16]. These were authenticated by morphological observation. Mycoplasma
contamination tests were confirmed to be negative. When mCherry-HiBiT- and
GFP-LgBiT-expressing BeWo cells are fused in response to forskolin, the
luciferase activity is recovered. Both types of BeWo cells were cultured and
maintained under standard cell-culture conditions. Cells were seeded at 1.0
BeWo cells were seeded at 1.5
Proteins were extracted using RIPA (50 mM Tris–HCl; pH 7.5, 150 mM NaCl, 1%
NP40, 0.1% sodium dodecyl sulfate (SDS), 0.5% sodium deoxycholate, and 1 mM
EDTA) with protease and phosphatase inhibitor (78442; Thermo Fisher Scientific,
Waltham, MA, USA). Proteins were separated by 12.5% sodium dodecyl sulfate
polyacrylamide gel electrophoresis and electro-transferred onto a polyvinylidene
difluoride membrane (IPVH00010; Millipore, Billerica, MA, USA) using Protein
Ladder One Plus (19593-54; Nacalai Tesque, Kyoto, Japan) as the standard. The
membranes were incubated with primary antibodies against hCG (1:500; ab54410;
Abcam, Cambridge, UK) and
Statistical analyses were conducted in GraphPad Prism version 10.2.3 (GraphPad
Software, Boston, MA, USA). Data are expressed as means
First, we assessed the effect of phenytoin and phenobarbital on placental syncytialization in BeWo cells. The cell viability assay showed that the concentration of phenytoin 250 µM or less in cells treated with forskolin was not cytotoxic to BeWo cells (Fig. 1A) and that none of the concentrations of phenobarbital used were cytotoxic to BeWo cells (Fig. 1B). Therefore, phenytoin was used with 250 µM as the highest concentration in the subsequent experiments. The cell fusion assay revealed that treatment with forskolin increased the luciferase activity, and co-treatment with a protein kinase A inhibitor H-89 downregulated the forskolin-induced luciferase activity (Fig. 2). Phenytoin significantly suppressed the forskolin-induced luciferase activity in a concentration-dependent manner (Fig. 2). In contrast, co-treatment with forskolin and phenobarbital showed no significant changes in the forskolin-induced luciferase activity of the co-culture model, indicating that phenytoin inhibited forskolin-induced BeWo cell fusion, but phenobarbital did not.
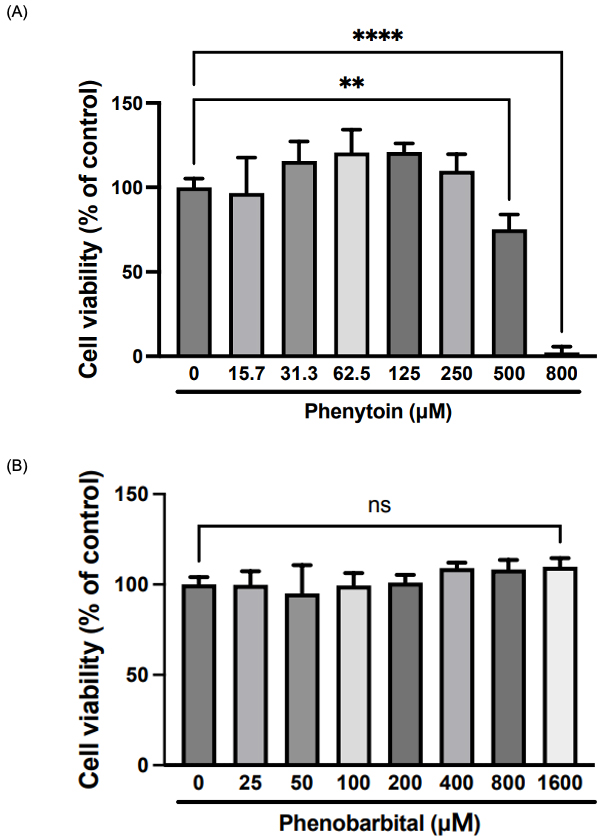 Fig. 1.
Fig. 1.
Cytotoxicity of phenytoin and phenobarbital in human
choriocarcinoma BeWo cells. Cells were treated with forskolin (50 µM)
containing different concentrations of (A) phenytoin or (B) phenobarbital for 48
h. Cell viability was evaluated by colorimetric dye assay. Data are expressed as
mean
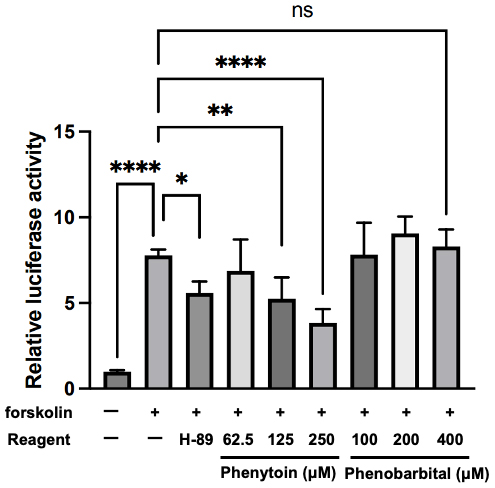 Fig. 2.
Fig. 2.
Effects of phenytoin and phenobarbital on cell fusion of
mCherry-HiBiT– and GFP-LgBiT–expressing BeWo cells. Cells were treated with
forskolin (50 µM) together with phenytoin or phenobarbital for 48 h.
Luciferase activity was measured to evaluate cell fusion. The protein kinase A
inhibitor H-89 (10 µM) was used as a positive control. Data are expressed
as mean
hCG is a hormone that is produced by the placenta in the first trimester of
pregnancy and is upregulated in BeWo cells in which syncytialization has been
induced by forskolin [20]. Therefore, we analyzed concentrations of hCG
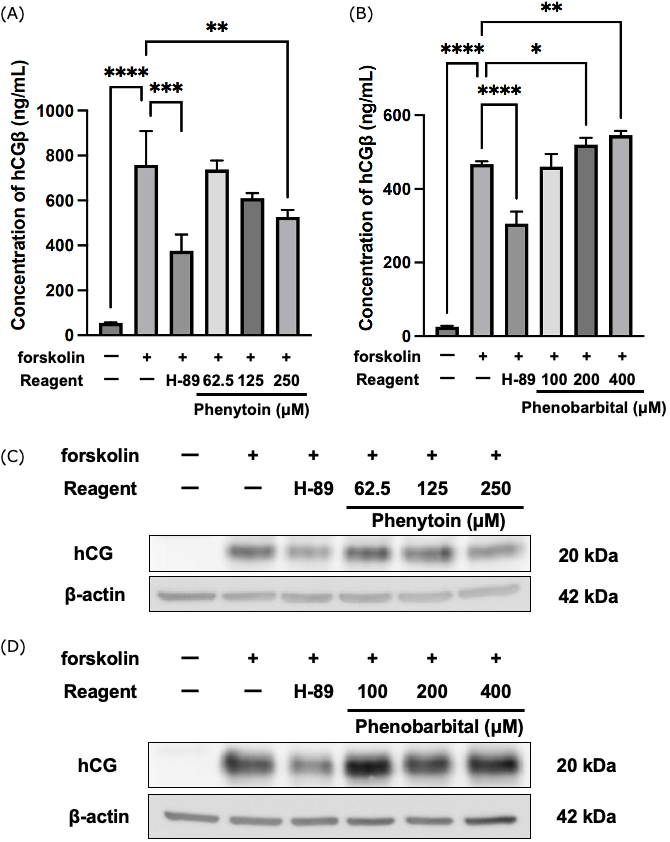 Fig. 3.
Fig. 3.
Effects of phenytoin and phenobarbital on human chorionic
gonadotropin (hCG) production and secretion. BeWo cells were treated with
forskolin (50 µM) together with phenytoin or phenobarbital for 48 h. The
protein kinase A inhibitor H-89 (10 µM) was used as a positive control.
(A,B) Concentration of hCG
Although both phenytoin and phenobarbital are anticonvulsant drugs, they each have a different pharmacological mechanism of action. We consider that this difference might reflect the difference in the effects on syncytialization observed in the phenytoin- and phenobarbital-treated BeWo cells. Phenytoin acts as a voltage-gated sodium ion channel blocker [21] but has also been reported to inhibit calcium transport mechanisms in Caco-2 human colorectal cancer cells [22], suggesting that phenytoin inhibits calcium transport in a membrane potential–independent manner. Placental cell fusion requires calcium signaling, as evidenced by the fact that transmembrane protein 16 activated by calcium signaling was found to regulate trophoblast cell fusion [23]. Therefore, phenytoin may inhibit calcium transport and reduce intracellular calcium ion concentrations, leading to the suppression of BeWo syncytialization. There is a need for further research to determine whether phenytoin inhibits calcium transport in BeWo cells via a membrane potential–independent mechanism.
It is known that hCG secretion is modulated by gamma-aminobutyric acid (GABA)
acting via GABAA receptors [24]. Given that the antiepileptic effects of
phenobarbital work via GABAA receptors [21], it can be hypothesized that
phenobarbital agonism at the GABAA receptor may cause enhanced hCG
The limitation of this study is that we assessed syncytialization only by evaluating hCG production and secretion. Various signaling pathways and molecules like syncytin-1 and syncytin-2 are involved in syncytialization [26]. We should also evaluate the expression changes of these various signaling pathways and molecules to provide stronger evidence of the effects of phenytoin and phenobarbital on syncytialization. In addition to that, the evaluation of the impact of these agents on not only BeWo but also human trophoblast stem cells syncytialization should be done. Moreover, the effects of these agents on placental structure and function also need to be evaluated in vivo. With these validations, this study will help us understand the adverse outcome pathways of phenytoin- and phenobarbital-induced fetal toxicity from a placental perspective.
Collectively, we showed that while phenytoin suppressed the process of syncytialization, phenobarbital had no effect on fusion and increased hCG production. These findings suggest that phenytoin and phenobarbital disrupt the process of placental development. Although further research is needed, our data help to understand the effects of anticonvulsant drugs on placenta.
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
MS: Data curation, Investigation, Validation, Visualization, Writing – original draft; and Writing – review & editing. WO: Investigation, Writing – review & editing. KH: Conceptualization, Funding acquisition, Project administration, Supervision, Writing – review & editing. MY: Methodology, Writing – review & editing. KT: Methodology, Writing – review & editing. YH: Conceptualization, Supervision, Writing – review & editing. YT: Conceptualization, Funding acquisition, Project administration, Supervision, Writing – review & editing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
The authors thank the Platform Project (JP25ama121054) for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)) from the Japan Agency for Medical Research and Development (AMED) for providing access to research equipment.
This study was supported by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (no. 23K18520 to KH and no. 23H02651 to YT) and by The Mochida Memorial Foundation for Medical and Pharmaceutical Research (to KH).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



