1 Department of Pharmacy, The Cancer Institute Hospital of Japanese Foundation for Cancer Research, 135-8550 Tokyo, Japan
2 Department of Clinical Pharmacology, School of Pharmacy, Tokyo University of Pharmacy and Life Sciences, 192-0392 Tokyo, Japan
3 Department of Breast Medical Oncology, The Cancer Institute Hospital of Japanese Foundation for Cancer Research, 135-8550 Tokyo, Japan
Abstract
Dexamethasone is typically included in the anti-emetic regimens during the administration of anticancer drugs. However, the incidence and severity of nausea and vomiting in patients receiving anticancer therapy, for whom dexamethasone must be avoided to prevent the recurrence of diabetes mellitus or hepatitis, remain unknown.
This retrospective, observational study evaluated nausea and vomiting in patients with breast cancer who underwent highly emetogenic chemotherapy, including anthracycline and cyclophosphamide, for breast cancer. In all patients, dexamethasone was completely omitted from the standard antiemetic regimen for reasons such as hepatitis, and only palonosetron and aprepitant were administered.
For the 82 evaluated cases, the incidence of nausea was 84.1%, vomiting was 14.6%, and the complete response (CR) rate was 8.5%. In addition, the incidence rate of grade 2 or higher nausea (CTCAE ver. 4) was 47.6%, and the proportion of cases in which anticancer drug doses were reduced in the subsequent course due to nausea and vomiting was 2.4%. Factor analysis showed that treatment regimens, age, drinking history, history of prior chemotherapy, and reasons for omitting dexamethasone had no significant effects on the incidence of chemotherapy-induced nausea and vomiting.
This study confirmed that the antiemetic effect of only administering palonosetron and aprepitant is insufficient for patients receiving highly emetogenic chemotherapy in whom dexamethasone cannot be administered. Prophylactic administration of other antiemetic drugs is necessary to effectively manage nausea and vomiting in patients receiving anticancer therapy who cannot receive dexamethasone.
Keywords
- breast neoplasms
- chemotherapy-induced nausea and vomiting
- dexamethasone
- highly emetogenic chemotherapy
- antiemetic agents
Chemotherapy-induced nausea and vomiting (CINV) is one of the most troublesome side effects of anticancer drugs. Persistent nausea and vomiting can cause dehydration, electrolyte imbalance and malnutrition, and it might also decrease patient motivation for treatment. It has also been suggested that even if acute nausea and vomiting have a delayed onset, patients often experience anticipatory symptoms [1], making appropriate control of CINV extremely important for maintaining patient quality of life and general health, as well as for allowing treatment continuation. In the 1990s, nausea and vomiting were the most distressing side effects of treatment in cancer patients, but in the 2000s, a long-acting 5-hydroxytryptamine 3 receptor antagonist (5HT3RA), palonosetron (PALO), was introduced in Japan. Further, aprepitant (APR), a neurokinin-1 receptor antagonist (NK1RA), has also become available, making it possible to administer the global standard of antiemetic therapy. This drug combination has resulted in a dramatic reduction in the incidence of emesis [2]. Sasaki H conducted a survey of patients’ top concerns during cancer chemotherapy, and reported that more patients were concerned about nausea than vomiting [3]. These results suggest that, whereas advances in antiemetic drugs have reduced the incidence of emesis, the management of nausea remains inadequate and significantly affects the quality of life of patients undergoing chemotherapy.
Chemotherapy including anthracycline and cyclophosphamide is a standard treatment that plays an important role in breast cancer chemotherapy. At the time this study was planned in 2018, the antiemetic guidelines of the American Society of Clinical Oncology (ASCO) and other guidelines stated that anthracycline/cyclophosphamide (AC) or cyclophosphamide/epirubicin/5-fluorouracil (CEF) therapy is considered highly emetogenic chemotherapy (HEC), and recommended prophylactic antiemetic therapy with a combination of 5HT3RA, NK1RA and dexamethasone (DEX) for its treatment [4]. Ito et al. [5] conducted a phase III study of this drug combination in Japan and found that complete response (CR) (defined as no vomiting episodes and no use of rescue medication) was achieved within 120 hours after AC therapy. In that study, the CR rate within 120 h after AC therapy was 46.9%, and the total control (TC) rate (defined as no vomiting, no use of rescue medications, and no nausea) was 29.6% [5]. Most patients with breast cancer are female and younger than those with other types of cancer. Since female sex and young age are risk factors for CINV, patients undergoing chemotherapy for breast cancer are generally at a high risk for CINV [6]. However, even temporary use of DEX in antiemetic therapy is associated with the risks of decreased bone density and diabetes mellitus [7, 8]. In addition, use of DEX to treat the side effects of immune checkpoint inhibitors might increase the risk of infections [9]. Therefore, adverse events caused by DEX used for antiemetic purposes are a problem in cancer patients. Saito et al. [10] reported an observational study of a 4-day course of DEX in patients being treated with AC and other therapies, and found that a reduction in the DEX dose on the first day was associated with an increased incidence of nausea, but a 2- to 4-day dose reduction did not affect nausea in the delayed phase. In clinical practice, we often encounter cases where DEX cannot be used for antiemesis, such as in cases with diabetes or hepatitis B. In Japan, Hashimoto et al. [11] suggested that a four-drug combination therapy, including 5HT3RA, NK1RA, DEX and olanzapine (OLN), is effective as standard antiemetic therapy in patients receiving HEC. Consequently, OLN has become available in Japan as the standard antiemetic therapy for HEC since October 2023. However, similar to DEX, it is contraindicated in diabetic patients and therefore cannot be used in these patients. Although dexamethasone constitutes a key component of standard prophylactic antiemetic regimens for HEC, its use is also contraindicated or avoided in a subset of patients due to comorbid conditions, such as diabetes mellitus, hepatitis, or psychiatric disorders. In such populations, clinicians were often forced to rely on nonstandard, steroidfree regimens without sufficient clinical evidence regarding their efficacy.
This paper presents valuable data evaluating the management of CINV in breast cancer patients undergoing HEC, along with the two-drug anti-emetic regimen of PALO and APR, under conditions where DEX or OLN cannot be used for CINV caused by HEC regimens containing AC and CEF. At the time this study was conducted (2014–2018), evidence guiding antiemetic management for patients in whom dexamethasone could not be administered was scarce. Additionally, realworld data describing nausea and vomiting outcomes under these circumstances were particularly limited. Therefore, the present study aimed to evaluate the incidence and severity of CINV in breast cancer patients receiving HEC who were managed with a dual antiemetic regimen consisting of a 5HT3RA and an NK1RA, in the absence of DEX. Although antiemetic strategies have evolved since then, including the introduction of OLNcontaining regimens, we believe that these findings provide important baseline data that underscore the unmet clinical needs in steroidineligible patients, and support the rationale for subsequent development of alternative antiemetic approaches.
In recent years, antibody–drug conjugates (ADCs) have become an important component of breast cancer treatment, with trastuzumab deruxtecan (T‑DXd) now widely used across multiple disease settings. Unlike conventional anthracycline- and cyclophosphamide-based regimens, T‑DXd is associated with a distinct safety profile, and persistent or delayed gastrointestinal toxicities, including prolonged nausea, have been increasingly recognized as a clinical challenge. Recent clinical studies and systematic analyses have reported delayed and sustained nausea during T‑DXd therapy, highlighting its negative impact on patient quality of life and treatment continuity [12, 13].
Several reports have demonstrated that the addition of olanzapine to standard antiemetic prophylaxis improves nausea control in patients receiving T‑DXd [14]. However, both dexamethasone and olanzapine may be contraindicated or difficult to use in patients with comorbidities such as diabetes mellitus because of metabolic adverse effects. Although our study population did not include patients treated with ADC-based therapies, our findings provide important baseline real-world evidence illustrating the limitations of dual antiemetic therapy consisting of a 5‑HT3 receptor antagonist and an NK1 receptor antagonist in high-risk settings when corticosteroids cannot be administered. In the current ADC era, these results underscore the unmet need for effective steroid- and olanzapine-sparing antiemetic strategies, which may become increasingly important for patients receiving newer ADC-based treatments.
A retrospective evaluation was conducted on the antiemetic efficacy of a two-drug regimen consisting solely of PALO and NK1RA, excluding DEX from the standard antiemetic therapy, in patients with breast cancer who received HEC regimens at the the Cancer Institute Hospital of Japanese Foundation for Cancer Research between January 1, 2014, and August 31, 2018, and who were excluded from receiving the standard antiemetic therapy due to conditions such as diabetes mellitus or hepatitis.
We retrospectively evaluated the following factors for the target patients using their electronic medical records: age, gender, performance status (PS), clinical stage, prior chemotherapy history, use of medications affecting CINV, reasons for not using dexamethasone, and anticancer drug dosage.
We investigated the incidence and severity of nausea and vomiting during the first chemotherapy course, as well as CR and TC rates. We also examined the proportion of patients who experienced a course delay or dose reduction of anticancer drugs in subsequent courses due to CINV, and the impact of treatment- and patient-related factors on CINV. The definitions of CR rate and TC rate were as follows.
CR: No vomiting, no dry heaving, no use of additional antiemetic medication;
TC: No nausea, no vomiting, no dry heaving, no use of additional antiemetic medication.
This trial was a single-arm observational study; no intergroup comparisons were
performed. Fisher’s exact probability test was used for the factor analysis of
CINV, with the significance level set at p
This study included patients with breast cancer who received AC or CEF therapy as the initial chemotherapy regimen between January 2014 and August 2018. All patients were outpatients who received dual antiemetic therapy, excluding DEX from the standard triple combination therapy consisting of a 5HT3RA, an NK1RA and DEX, due to comorbidities such as diabetes or hepatitis. Exclusion criteria were patients whose chemotherapy dose was reduced due to CINV, and patients who experienced nausea or vomiting before chemotherapy initiation (Table 1). The overall patient selection process, including inclusion and exclusion criteria, is summarized in Fig. 1. In most cases, rescue antiemetics were pre-prescribed for home use, except for 14 cases without a prescription and one case where the status was unknown whether the patient was hospitalized or an outpatient. Details of the rescue antiemetics are shown in Table 2.
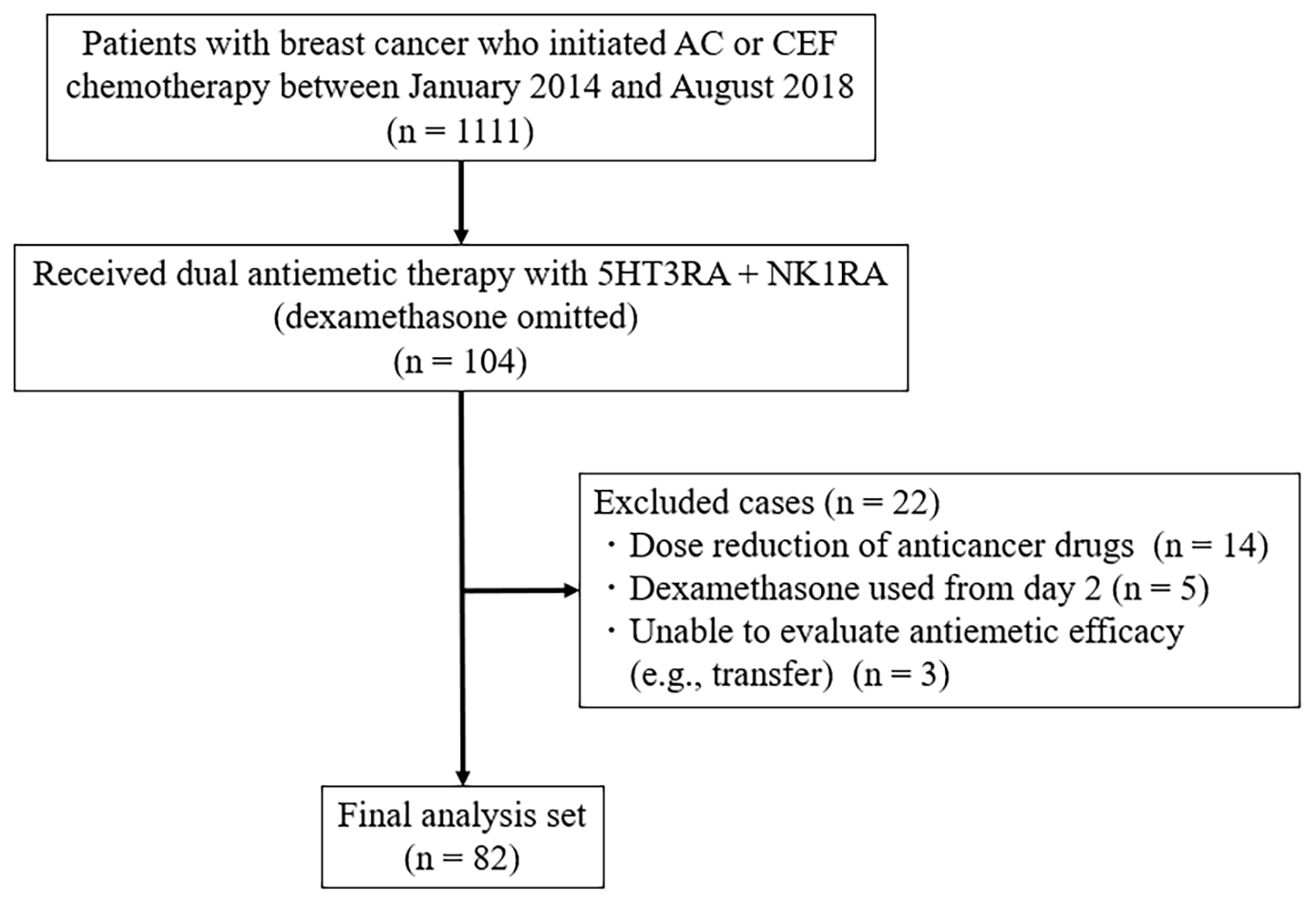 Fig. 1.
Fig. 1.
Flowchart of patient selection. The flowchart shows the inclusion and exclusion of patients with breast cancer who received highly emetogenic chemotherapy (anthracycline- or cyclophosphamide-based regimens) and dual antiemetic therapy with a 5‑HT3 receptor antagonist and an NK1 receptor antagonist without dexamethasone. AC, anthracycline/cyclophosphamide; CEF, cyclophosphamide/epirubicin/5-fluorouracil.
| n (%) | All patients (n = 82) | |
| Age, (y) median (range) | 62 (32–72) | |
| Sex | Female/Male | 80 (97.6)/2 (2.4) |
| Clinical stage | I | 20 (24.4) |
| II | 50 (61.0) | |
| III | 12 (14.6) | |
| IV | 0 (0) | |
| PS | 0 | 76 (92.7) |
| 1 | 3 (3.7) | |
| 2 | 0 (0) | |
| Unknown | 3 (3.7) | |
| Stage | Preoperative | 20 (24.4) |
| Postoperative | 61 (74.4) | |
| Recurrence | 1 (1.2) | |
| Regimen | AC/CEF | 23 (28.0)/59 (72.0) |
| Drinking history | Yes | 51 (62.2) |
| No | 29 (35.4) | |
| Unknown | 2 (2.4) | |
| Prior chemotherapy history | Yes/No | 5 (6.1)/77 (94.0) |
| Concomitant opioid | Yes/No | 0 (0)/82 (100.0) |
| Reason for not using DEX | HBV infection | 66 (80.5) |
| Diabetes mellitus | 12 (14.6) | |
| Mental disorder | 3 (3.7) | |
| DEX allergy | 1 (1.2) |
PS, Performance status; AC, Anthracycline, cyclophosphamide; CEF, Cyclophosphamide, epirubicin and 5-fluorouracil; DEX, Dexamethasone; HBV, Hepatitis B virus.
| Rescue medication | n (%) |
| Metoclopramide tablets 5 mg | 48 (58.5) |
| Domperidone tablets 10 mg | 16 (19.5) |
| Bromazepam suppositories 3 mg | 4 (4.8) |
| Domperidone suppositories 60 mg | 3 (3.6) |
| Lorazepam tablets 0.5 mg | 2 (2.4) |
| None | 14 (17.0) |
| Unknown | 1 (1.2) |
Some of the patients received multiple rescue anti-emetics.
This retrospective observational study was derived from electronic medical record data. It evaluated nausea and vomiting in patients with breast cancer who received highly emetogenic chemotherapy containing anthracyclines or cyclophosphamide. In all patients, dexamethasone was completely excluded from standard antiemetic therapy due to conditions such as hepatitis, and only palonosetron and aprepitant were used. Data on age, sex, performance status (PS), clinical stage, drinking history, chemotherapy history, reasons for omitting DEX, additional medications used for CINV, history of delays in starting the second chemotherapy course due to CINV, and chemotherapy dose reductions were collected from the patients’ electronic medical records. Medications (such as metoclopramide, DEX, OLN, etc.) used during the study period and additional antiemetics, as required, during the second course were also investigated. Nausea and vomiting were evaluated according to the Japanese translation (JCOG version, Japan Clinical Oncology Group, Tokyo, Japan; https://www.jcog.jp) of the Common Terminology Criteria for Adverse Events version 4.0 (CTCAE v 4.0, National Cancer Institute, Bethesda, MD, USA). Since the inclusion criteria for study participation included inability to use DEX, we evaluated the efficacy of a two-drug antiemetic regimen consisting solely of PALO and NK1RA. The total control (TC) rate, which was defined as the percentage of patients without vomiting, no additional antiemetics, and no nausea, was also evaluated.
Patients who received both PALO and APR and underwent AC/CEF therapy at our
institution after 2010, when these drugs became available in Japan, were
included. Patients with incomplete medical records regarding CINV were excluded.
This was a single-arm, observational study without intergroup comparisons. Age
was dichotomized at 60 years for exploratory analysis, as this cutoff is commonly
used in clinical studies and allowed for a practical balance of sample sizes
between groups. For categorical variables, Fisher’s exact test was used to
analyze factors associated with CINV, and two-sided exact p-values were reported,
with a significance level of p
The patients’ background characteristics are shown in Table 1. A total of 82
cases, with a median age of 62 years (range 32 to 72 years), were evaluated. AC
and CEF were used in 23 and 59 patients, respectively. The reasons for not using
DEX were diabetes mellitus (12 patients), HBV infection (66 patients), mental
disorders (three patients), and a history of DEX allergy in one patient. The
incidences of nausea and other symptoms during the first course of anticancer
treatment are shown in Fig. 2. The incidence rate of all-grade nausea was 84.1%,
and that of grade
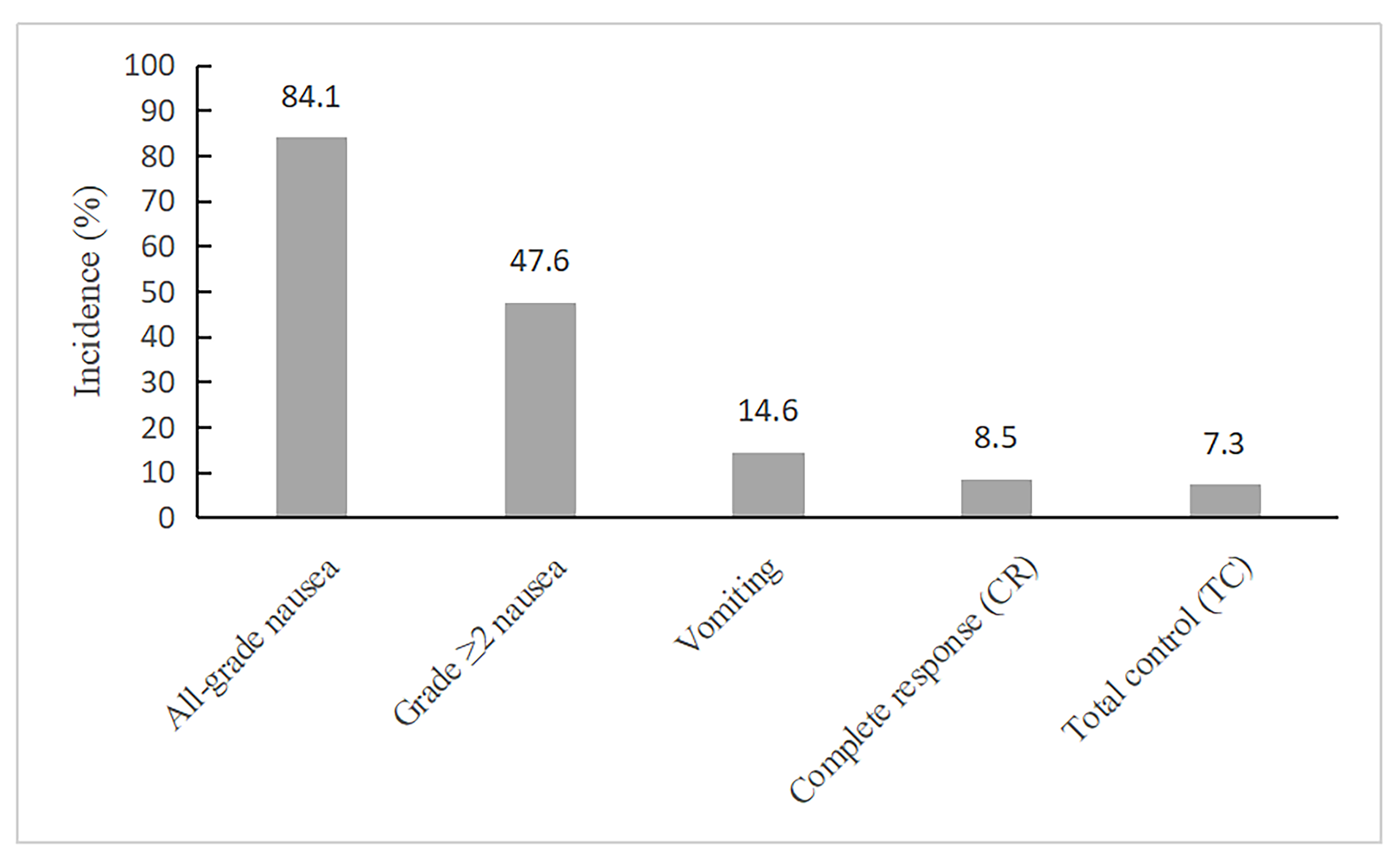 Fig. 2.
Fig. 2.
Incidence of chemotherapy-induced nausea and vomiting outcomes
during the first chemotherapy course (n = 82). The bar chart illustrates the
incidence of all-grade nausea, grade
| Regimen | AC (%), n = 23 | CEF (%), n = 59 | p-value | |
| Nausea (grade |
11 (47.8) | 28 (47.5) | 1.000 | |
| CR | 3 (13.0) | 4 (6.8) | 0.394 | |
| TC | 3 (13.0) | 3 (5.1) | 0.342 | |
| Age (y) | p-value | |||
| Nausea (grade |
23 (43.4) | 16 (55.2) | 0.359 | |
| CR | 5 (9.4) | 2 (6.9) | 1.000 | |
| TC | 4 (7.5) | 2 (6.9) | 1.000 | |
| Drinking history | Yes (%), n = 51 | No (%), n = 29 | p-value | |
| Nausea (grade |
25 (49.0) | 13 (44.8) | 0.817 | |
| CR | 5 (9.8) | 2 (6.9) | 1.000 | |
| TC | 4 (7.8) | 2 (6.9) | 1.000 | |
| History of chemotherapy administration | Yes (%), n = 5 | No (%), n = 77 | p-value | |
| Nausea (grade |
4 (80.0) | 35 (45.5) | 0.186 | |
| CR | 0 (0) | 7 (9.1) | 1.000 | |
| TC | 0 (0) | 6 (7.8) | 1.000 | |
| Reasons for non-use of DEX | Diabetes mellitus (%), n = 12 | Hepatitis-B (%), n = 66 | p-value | |
| Nausea (grade |
3 (25.0) | 34 (51.5) | 0.121 | |
| CR | 1 (8.3) | 6 (9.1) | 1.000 | |
| TC | 0 (0) | 6 (9.1) | 0.582 | |
p-values were calculated using Fisher’s exact test (two-sided exact significance).
Recently, guideline-recommended antiemetic therapy has been widely adopted in
patients receiving HEC, and, in principle, the recommended therapies are
implemented unless the patients have specific restrictions, such as hepatitis or
diabetes mellitus. However, information on antiemetic efficacy when standard
therapy cannot be administered is extremely scarce. The present study evaluated
the antiemetic efficacy of 5-HT3 and NK1 receptor antagonists during HEC. A phase
II trial comparing 3-day DEX administration with 1-day administration as a part
of standard antiemetic therapy for AC showed no significant differences in
overall CR rates (82.9% and 82.1%, respectively) and complete control (CC)
(defined as no vomiting episodes, no use of rescue medication, and no more than
mild nausea) rates between the two groups (48.8% and 61.5%, respectively) [15].
In addition, an observational study in Japan reported a significantly greater
incidence of nausea with 1-day DEX administration (overall nausea incidence:
92%, grade
In the present study, CINV was evaluated in patients in whom DEX was omitted to
avoid increases in blood glucose levels or reactivation of hepatitis B during
chemotherapy. The overall incidences of nausea were 84.1% (all grades) and
47.6% (grade
Furthermore, 2.4% of patients required dose reduction in the subsequent chemotherapy course due to nausea and vomiting. It has been suggested that maintaining a treatment intensity of 85% or higher has beneficial effects on the prognosis of perioperative breast cancer treatment [2], indicating that sufficient alternative measures are needed to avoid dose reductions when standard antiemetic therapy cannot be implemented. Furthermore, severe CINV can lead to anticipatory nausea and vomiting, complicating subsequent treatment [18], which highlights the importance of controlling CINV.
In future, when performing AC therapy, three-drug combination therapy including OLN should be implemented in patients in whom DEX is contraindicated due to hepatitis or other reasons. Saito et al. [10] demonstrated the efficacy of a four-drug combination therapy, including 5 mg OLN, in a Phase III trial for triple-drug antiemetic therapy in AC. Their results suggested that when DEX cannot be used, OLN can be one of the treatment options. However, in patients with diabetes mellitus, OLN is contraindicated, and DEX should be avoided due to the risk of hyperglycemia. Therefore, an alternative combination of antiemetic drugs other than OLN and DEX should be considered. For example, metoclopramide, which is recommended by various guidelines for breakthrough nausea and vomiting, lorazepam [19] or alprazolam [20], which are recommended for anticipatory nausea and vomiting, are other options. In addition, mirtazapine, which has antagonistic effects on receptors such as 5-HT3 and H1 that are related to emesis, and can be used in patients with concomitant diabetes mellitus or hepatitis, has been suggested to have antiemetic effects in observational studies and may be one of the options for CINV in these patients [21, 22].
In this study, analysis was performed for factors such as treatment regimens, age, gender, history of alcohol consumption, prior chemotherapy, and reasons for not using DEX; however, no significant correlations were observed between these factors and CINV. The following limitations of this study should be noted. Although factors such as nausea, motion sickness, and insomnia have also been suggested as risk factors for CINV [23], these factors could not be evaluated in this study. Other limitations include the retrospective, single-center nature of the study, the fact that rescue antiemetics were pre-prescribed in 81.7% of cases (which may have influenced CINV occurrence or severity), and the lack of patient-reported outcomes (PROs), potentially limiting the accuracy of CINV assessment. Further, since the sample size was small, insufficient power cannot be ruled out. Further studies with larger sample sizes are needed to confirm our results.
In breast cancer patients receiving HEC, a two-drug antiemetic regimen consisting of PALO and APR without DEX might be insufficient for control of CINV. This highlights the need for alternative prophylactic antiemetic strategies in patients who are ineligible for DEX, and supports the development and optimization of steroid-sparing antiemetic approaches in clinical practice.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Study conception: WS, TT, MY; study design: WS, KS, TY, KKaw, TA; data acquisition: WS, TS, NS, MH, KKob, KS. WS drafted and revised the manuscript. All authors have revised and approved the final version. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Medical Ethics Review Board of the Japanese Foundation for Cancer Research, Cancer Institute Hospital (IRB approval number: 2018-GA-1113, approval date: October 1, 2018). This study was also approved by the Cancer Research Institute Medical Research Ethics Committee. The study complied with the Declaration of Helsinki, and patient informed consent was obtained through an opt-out method on our hospital’s website.
The authors would like to acknowledge the contributions of all those who contributed to this article and all the participants. The authors would also like to thank FORTE Science Communications for English language editing.
This research received no external funding.
All authors declare that the following competing interests are outside the submitted work. WS reports personal fees from AstraZeneca and Eli Lilly Japan K.K. outside the submitted work. KS reports personal fees from Taiho, Daiichi-Sankyo, Bristol Myers Squibb, Takata Pharmaceutical, Nippon Kayaku, and Nipro Pharma outside the submitted work. TY reports personal fees from AstraZeneca, Astellas Pharma Inc., Janssen Pharmaceutical K.K., and Ono Pharmaceuticals outside the submitted work. TA reports personal fees from Ono Pharmaceuticals, Yakult Pharmaceutical Industry Co., Ltd., and Taiho Pharmaceutical Co., Ltd. outside the submitted work. NS reports personal fees from AbbVie GK, Astellas Pharma Inc., and Nippon Shinyaku Co., Ltd. outside the submitted work. MH reports personal fees from Bayer and Merck & Company, Inc. outside the submitted work. KK reports personal fees from Ono Pharmaceuticals, Astellas Pharma Inc., and Daiichi Sankyo Company, Ltd. outside the submitted work. TT reports personal fees from Chugai, Daiichi-Sankyo, and Eli Lilly outside the submitted work. MY reports personal fees from Daiichi Sankyo Company, Ltd., Takata Pharmaceutical Co., Ltd., Yakult Honsha Co., Ltd, Eisai Co., Ltd., and AbbVie GK outside the submitted work.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


