1 Faculty of Pharmacy, Middle East University, 11610 Amman, Jordan
Abstract
Reactive oxygen species (ROS) have been identified as one of the critical factors in cancer development. ROS have been linked to cancer at all stages, and their applications in cancer treatment have gained attention due to their concentration-dependent implications: (1) low to moderate levels as fundamental signaling molecules, and (2) higher levels in cancer cells as a unique characteristic of cancer and cytotoxic agents. However, resistance and off-target effects are the main barriers that can hinder and limit the therapeutic efficacy of chemotherapies. The main reason for that is the complex tumor microenvironment such as hypoxia. Developing drug nanocarriers that can target ROS represents a potential delivery platform to overcome these barriers. For instance, doxorubicin-encapsulated ROS (nitric oxide) micelles accumulated 6.7-fold more drug in PC3-Luc cancer cells than when using this drug alone. Regrettably, the past studies have merely discussed the micelle alone as a nanocarrier for the delivery of ROS-based therapy in cancer without exploring dendrimers. Instead, this review examines the structural design of dendrimers tailored for oxygen transport, their conjugation with ROS-generating therapies, and therapeutic applications in photodynamic therapy, radiotherapy, and chemotherapy. Besides, it also discusses the translational challenges and future perspectives for ROS-based dendrimers. For the first time, this work also critically compares various dendrimer types and generations, oxygen-delivery strategies, drug loading properties, in vitro/in vivo outcomes, and toxicity data. A dedicated section discussing biodistribution, clearance, biocompatibility, and regulatory considerations of dendrimers was also explored in this study. Finally, this review concludes that the dendrimers can be engineered to carry and deliver active oxygen by using the following delivery strategies: (1) addition of oxygen carriers, (2) enzyme functionalization, (3) the incorporation of photosensitizers and metal ions, and (4) surface alterations.
Keywords
- reactive oxygen species
- dendrimers
- oxygen-delivery strategies
- cancer treatment
- comparative study
- tumor microenvironment
Cancer is one of the main causes of death worldwide which characterized by dysregulation of cellular proliferation, cellular invasion into surrounding tissues, and metastasis to distant organs. Off-target toxicity and treatment resistance are common challenges with current conventional therapies such as surgery, chemotherapy, and radiotherapy [1]. Tumor microenvironment (TME) is an interaction of cancer cells, stromal cells, immune cells, extracellular matrix components, and malfunctioning vasculature that plays an important role in resistance. Hypoxia is one of the distinctive characteristics of the TME and can be defined as locations having low quantities of oxygen [2, 3, 4]. The TME has also unique biochemical characteristics such as acidic pH, increased amounts of reactive oxygen species (ROS), and high interstitial fluid pressure. ROS are defined as chemically active, smaller molecules consisting of oxygen. Superoxide (O2•–), nitric oxide (NO•), and hydroxyl (•OH) radicals as well as nonradical ROS such as hydrogen peroxide (H2O2) and singlet oxygen (1O2) play significant roles in cancer biology [5, 6]. For example, low to moderate ROS levels endorse tumor growth and survival, but higher levels cause oxidative damage to DNA, lipids, and proteins resulting in apoptosis or necrosis. Thus, this fact inspired researchers to develop targeted drug delivery [7, 8, 9, 10]. Nanoparticles can be formulated to respond to these triggers and preferentially release drugs within tumors leading to lower systemic side effects [11, 12, 13, 14, 15].
Photodynamic therapy (PDT), radiation, chemotherapies, and chemodynamic therapies are currently being investigated as therapeutic options that increase ROS levels over toxic thresholds. Chemodynamic therapies are treatments that work by converting endogenous hydrogen peroxide of metal drugs into potent cytotoxic hydroxyl radicals. PDT involves phototherapies that incorporate light and photosensitizing chemical ingredients along with molecular oxygen to trigger cellular death. However, hypoxia reduces ROS generation which represents a significant obstacle for these treatments [16, 17, 18, 19]. To address this issue, ROS-based micelles were widely developed and tested to selectively deliver chemotherapy to tumor cells. Nonetheless, only limited studies have examined other promising nanocarriers such as ROS-based dendrimers [20, 21, 22, 23]. The term “dendrimer” is derived from the Greek phrase “dendron”, which refers to a tree or branch. Dendrimers are nanosized and highly branched polymers with variable surface chemistry that have distinct benefits such as oxygen transporters and ROS modulators. For example, their multivalency and multifunctional platforms enable coupling with oxygen-binding moieties, catalytic enzymes, and photosensitizers. As a result, dendrimers could be used to overcome hypoxia and improve therapeutic effects [24, 25, 26]. However, the most recent advances in dendrimers for active oxygen delivery and ROS control in cancer therapies were not extensively investigated. Consequently, this review mainly explores and focuses on the chemistry and functionalization methods of ROS-based dendrimers, the impact of the tumor microenvironment on nanoparticle design, therapeutic applications, translational challenges, and future prospects. Besides, it also highlights and analyzes the key differences between the various types and generations of dendrimers as well as oxygen delivery strategies including drug loading properties, pharmacokinetic profiles, and toxicity data.
The information regarding ROS-based micelles and dendrimer-based drug delivery was largely reiterated in the previous reviews. Instead, this review critically compares and explores the old and novel dendrimer types, oxygen-delivery methods, and translational challenges. Besides, it also involves comparative tables summarizing key parameters (such as dendrimer generations, oxygen carriers, drug loading, in vitro/in vivo outcomes, and toxicity data) and provides a critical assessment of the strengths and limitations of each approach. Moreover, dedicated sections discussing biodistribution, clearance, biocompatibility, and regulatory considerations are also included in this work.
The systematic literature search strategy such as databases, keywords, and inclusion/exclusion criteria was discussed in this section to ensure transparency, reproducibility, and avoid narrative selection of references. Furthermore, this initiates with a transparent search strategy utilizing major databases such as Scopus, ScienceDirect, PubMed, Embase, and Web of Science, coupled with comprehensive and broad keywords such as “Dendrimers Types and Generations”; “Reactive Oxygen Species”; “Tumor Microenvironment”; “Oxygen-Delivery Strategies”; “Translational Challenges of Dendrimers”; “Toxicity Profiles of Dendrimers”; “Oxygen Carriers”; “Biodistribution, Clearance, Biocompatibility, and Regulatory Considerations of Dendrimers”. Besides, comparative MeSH phrases such as systematic comparison, systematic evaluation of in vitro/in vivo outcomes, and critical appraisal of strengths and limitations were also implemented. The selection process was governed by predefined inclusion and exclusion criteria. Furthermore, the inclusion criteria include: English-language publications, no publication date restrictions (with preference for the last 5 years), any study design or related original research (particularly that include quantitative comparison of in vitro, in vivo, clinical trials, safety profiles, and computational/modeling outcomes studies), and intervention-based studies should include dendrimers as a carrier or oxygen-delivering strategies. However, irrelevant studies conducted on non-dendritic nanocarriers, retracted articles, and non-English language publications were excluded from this search strategy.
The environment of TME significantly differs from normal tissue that may involve hypoxia, acidic pH (~5.5–6.5 vs. ~7.4 in normal tissues), and high ROS characteristic of tumors [27, 28]. These properties offer valuable triggers for designing responsive nanoparticles. For instance, Redox-responsive groups can react to elevated glutathione or ROS levels, whereas pH-sensitive linkers may selectively promote payload release in acidic tumors [29]. Since the formation of ROS requires molecular oxygen as a substrate, hypoxia significantly reduces the release of ROS-based cytotoxic species during treatments such as PDT and radiation [30, 31]. Thus, hypoxia can be reversed and cancers become more susceptible to ROS-mediated therapy by using nanoparticles that can deliver and generate oxygen within the tumor. It is crucial to note that these altered tumor vasculatures can also improve the enhanced permeability and retention (EPR), or passive drug accumulation towards tumor cells. Furthermore, the EPR effect promotes nanoparticles whose size ranges from 10 to 100 nm to preferentially concentrate in tumor tissue due to leaky vasculature and limited lymphatic drainage [32]. For example, dendrimers which are typically 1–10 nm in size can easily penetrate deeply within tumors. Besides, their surfaces can also be optimized to improve circulation time, biodistribution, and cellular absorption of loaded drugs [33, 34]. In line, treatment with NO•-loaded nanoparticles led to 1.6- and 6.7-fold increases in doxorubicin (DOX) accumulation in PC3-Luc cancer cells when compared to control nanoparticles and free DOX [35]. This suggests that NO•-loaded nanoparticles efficiently reduced tumor growth with no obvious toxic effect to normal tissue. In addition to increasing EPR-based DOX delivery through the vasodilatory effect of NO•, the biological functions of NO• can also lead to tumor regression. NO• has recently been found to reduce drug efflux through P-glycoprotein and reverse drug resistance. However, excessive NO• levels can induce cytotoxicity by upregulating p53 genes and promoting cell apoptosis. As a result, a combination of NO•-based nanoparticle treatments with the previously mentioned biological characteristics of NO• can lead to an increase the antitumor efficacy [35, 36]. However, the potential risks of NO•-loaded nanoparticles for off-target vasodilation such as hypotensive crises or long-term systemic toxicity to normal organs still need to be evaluated. In cancer therapy, NO• may boost the effect of anticancer drugs by enhancing the drug delivery, reducing of drug resistance, and inhibiting tumor metastasis. Consequently, these characteristics will make them promising candidates for combination therapies with chemodynamic and radiotherapy [36, 37].
The first “cascade” and “nonskid-chain-like” molecules with cavity topologies were developed and described by Buhleir and colleagues as early as 1978. These compounds were subsequently identified as the initial forms of dendritic polymers [33]. They were then further developed by Donald A. Tomalia and his colleagues to include a central, hollow core and tendrils that branched outward, one from another, precisely and predictably. Nowadays, the major differences between the old (conventional) such as Poly(amidoamine) (PAMAM) and Poly(propylene imine) (PPI) versus new (novel or specialized) dendrimers is based on their chemical structural complexity, functionality, and specific uses [38, 39]. The primary structure of old dendrimers involves an initiator core, repeating branched units (generations), and terminal end-groups. These types were widely used for the basic drug encapsulation, gene delivery (due to their cationic charge), and diagnostic imaging [39]. Despite the prior therapeutic applications, there are still certain issues that need to be resolved. The main challenges may involve the nonspecific adsorptions of plasma proteins, accelerated elimination by the reticuloendothelial system (RES), high cost of production, toxicity and biodistribution which are significantly correlated with their size and surface chemistry [39, 40]. To overcome these issues, the new or specialized dendrimers have unique advantages such as biocompatibility, excellent biorecognition, selective binding and minimal toxicity, and targeted delivery [40]. The previous and novel types of dendrimers were critically compared in Table 1 (Ref. [33, 38, 39]), Table 2 (Ref. [39, 40, 41]).
| Feature | Description | Key examples | References |
| Structure | Highly symmetric, spherical, and nearly monodisperse (uniform size). Consist of an initiator core, repeating branched units (generations), and terminal end-groups. | PAMAM (Poly(amidoamine)) and PPI (Poly(propylene imine)). | [33] |
| Key properties | Defined size (nanoscale), high surface functionality (exponentially increasing end-groups with generation), and internal cavities for encapsulation. | [33] | |
| Limitations | -Significant cytotoxicity or offsite release for higher generations of cationic dendrimers (like PAMAM G5 and up) due to the positive charge of their terminal primary amine groups interacting with cell membranes. | [38] | |
| -Expensive and difficulties to synthesize beyond a certain generation due to steric hindrance. | |||
| Applications | Primarily used for basic drug encapsulation, gene delivery (due to cationic charge), and diagnostic imaging. | [38, 39] |
| Feature | Description | Key examples | References |
| Structural complexity | Often non-symmetric, hybrid, or built with specialized monomers/cores to achieve unique properties. For examples, their periphery and core containing sugar or carbohydrate, amino acid, and amphiphilic building units. | Janus, Dendritic-Linear Hybrid, Supramolecular, Glycodendrimers, Shape-Persistent, Tecto-dendrimers. | [39] |
| Functional improvements | -Self-Assembling Properties: Formation of stable supramolecular aggregates (micelles) in water. | [40] | |
| -High Drug Loading for poorly soluble drugs via core encapsulation. | |||
| -Reduced Cytotoxicity: Through surface modification such as PEGylation (adding polyethylene glycol) or neutralizing cationic groups with anionic ones (e.g., carboxylate-terminated PAMAM). | |||
| -Enhanced Targeting: Attachment of specific targeting ligands (e.g., antibodies, sugars) to the surface and direct targeting of tumor microenvironment (e.g., acidic pH of cancer). | |||
| -Biodegradability: Incorporation of cleavable bonds (e.g., ester, amide) in the backbone for controlled breakdown in vivo. | |||
| Design principles | Focus on optimizing a specific property (e.g., making one side hydrophobic and the other hydrophilic in Janus dendrimers) or combining the properties of two different materials (e.g., dendritic-linear hybrids). | [40, 41] | |
| Applications | Targeted drug/gene delivery (via active targeting), high-fidelity MRI contrast agents, molecular scaffolds for vaccines, and advanced materials science applications. | [41] | |
| Possible challenges | -Complex, multi-step synthesis to ensure perfect valency. | [41] | |
| -Potential for high in vivo clearance for untargeted dendrimers. | |||
| -High cost and difficulty of large-scale, high-generation synthesis (due to peptide coupling yields). | |||
| -Potential for immunogenicity if the peptide sequence is non-human. |
Dendrimer generations are the number of branching cycles in their synthesis with
each subsequent generation roughly doubling the molecular weight. As the
generation progresses, the size of dendrimer increases and its shape changes from
an asymmetric open disc to a more spherical structure [41]. Furthermore, the
number of surface functional groups rises with each generation allowing for
greater customization. Synthetic processes may affect the size, surface charge,
peripheral functional groups, and solubility of dendrimers. For example,
higher-generation dendrimers have greater sizes, inner cavities, and more
terminal functional groups. Jackson and colleagues revealed that the nanoscopic
features of individual PAMAM dendrimers could be found for generations 5 to 10
(G5 to G10) [33]. For PAMAM G5, the mean diameter is 4.3 nm, while it is 14.7 nm
for PAMAM G10 [33, 42]. In addition to its nanoscopic property, the multivalency
of dendrimers enables additional modification or conjugation with chemical groups
such as fluorophores, targeting ligands, drugs and genes. Low-generation
dendrimers have limited drug loading capability due to their narrow internal
area. PEGylated dendrimers have higher drug loading capacity because of the
aggregation of molecules [42, 43]. PEGylated dendrimers may improve the EPR effect
of loaded drugs. Size limitation is a crucial factor. Furthermore, PAMAM
dendrimers of generation 5 or below can be removed through glomerular filtration
in the kidney, but generation 6 and above require hepatic clearance. Dendrimers
ranging from 4–10 nm can interact with nanometric cellular components and
overcome endocytosis barriers [33, 44]. PAMAM dendrimers of generation 6 and
higher have high costs and severe toxicity; thus they are rarely used [44]. In
line, Zinselmeyer et al. (2002) [45] have found that the generation 2
PPI dendrimer retains optimal in vitro gene transfer activity by
combining high DNA binding and minimal cell toxicity. Furthermore, the
cytotoxicity of PPI dendrimers increases with generations, as the following
trend: PPI G8
| Feature | Generation 0 (G0) | Generation 1 (G1) | Generations 2 & 3 (G2 & G3) | Generations 4–6 (G4–G6) | High Generations (G7+) | References |
| Number of repeating units | Very Low | Low | Moderate | High | Very High (Geometric Growth) | [33, 41] |
| Surface functional groups | Low | Low | Moderate | High | Very High | [41] |
| Molecular weight | Lowest | Low ( |
Moderate ( |
High ( |
Highest (Exponential Increase) | [42, 43] |
| Molecular shape/conformation | Open, Flexible, Ellipsoidal | Flexible, Ellipsoidal | More Compact, Flexible | Globular/Spherical, more rigid | Highly Dense, Globular/Solid-like | [41, 44] |
| Internal cavities/space | Minimal/Open | Small/Developing | Developing/Medium-sized internal space | Well-defined internal cavities/voids | Highly dense internal structure, surface crowding | [41, 43] |
| Size/Diameter | Smallest (Nanoscale) | Small (Linear increase with G) | Medium | Larger ( |
Largest (Size increase slows down) | [33, 44] |
| Application suitability | Surface modification, scaffolds | Drug/gene delivery (smaller payload), scaffolds | Drug/gene delivery (medium payload), imaging agents, catalysts | Optimal for Drug/Gene Delivery (large payload, excellent targeting/solubility), imaging | Nanoparticle-like behaviour, surface chemistry, solid-like properties | [44, 45] |
The main feature of dendrimers is their ability to exert control over size, shape, and surface chemistry. They also have a central core, several terminal functional groups, and repeating branching units [46]. These architectural features of dendrimers enable the encapsulation or conjugation of oxygen carriers, catalytic enzymes, and drugs. These nanocarriers can be developed to provide oxygen using various delivery mechanisms including the addition of oxygen carriers, enzyme functionalization, the incorporation of photosensitizers and metal ions, and surface alterations.
Perfluorocarbons (PFCs) are organofluorine compounds with the formula CxFy, which indicates that they solely include carbon and fluorine. They frequently have unique features such as increased stability, volatility, and hydrophobicity. Nowadays, a variety of fluorocarbons and their derivatives have been used as commercial polymers, drugs, anesthetics, and refrigerants [47]. Many organic compounds containing fluorine can also be called fluorocarbons. The carbon–fluorine bond is one of the strongest linkages in organic chemistry which makes perfluoroalkanes extremely stable. Its strength arises from the partial ionic nature (electronegativity) of fluorine imparted to the carbon and fluorine atoms through partial charges. This reduces the length and strengthens this bond (in contrast to carbon-hydrogen bonds) through covalent interactions. Fluorocarbons are colorless molecules with densities greater than two times those of water. They are insoluble with most of the organic solvents (e.g., ethanol, acetone, ethyl acetate, and chloroform); however, they are only solubilized with certain hydrocarbons. These compounds are not miscible in water (about 10 ppm). Fluorocarbons are slightly vulnerable to the transient dipoles that serve as the foundation for the London dispersion force. Besides, the high electronegativity of fluorine leads to a decrease in the polarizability of the atom. As a result, fluorocarbons are not only hydrophobic substances, but also they have minimal intermolecular attractive forces. These compounds could be incorporated into dendrimers to serve as oxygen reservoirs because of their high oxygen solubility and biocompatibility [48, 49]. PFCs might be covalently bonded to terminal groups or physically confined inside dendrimer interiors. During treatments, these dendrimer-PFC complexes may increase the formation of ROS by releasing oxygen in hypoxic tumor areas [50, 51]. Fig. 1 simulates the main aspects of treating cancer and generating ROS using dendrimers coupled with perfluorocarbons.
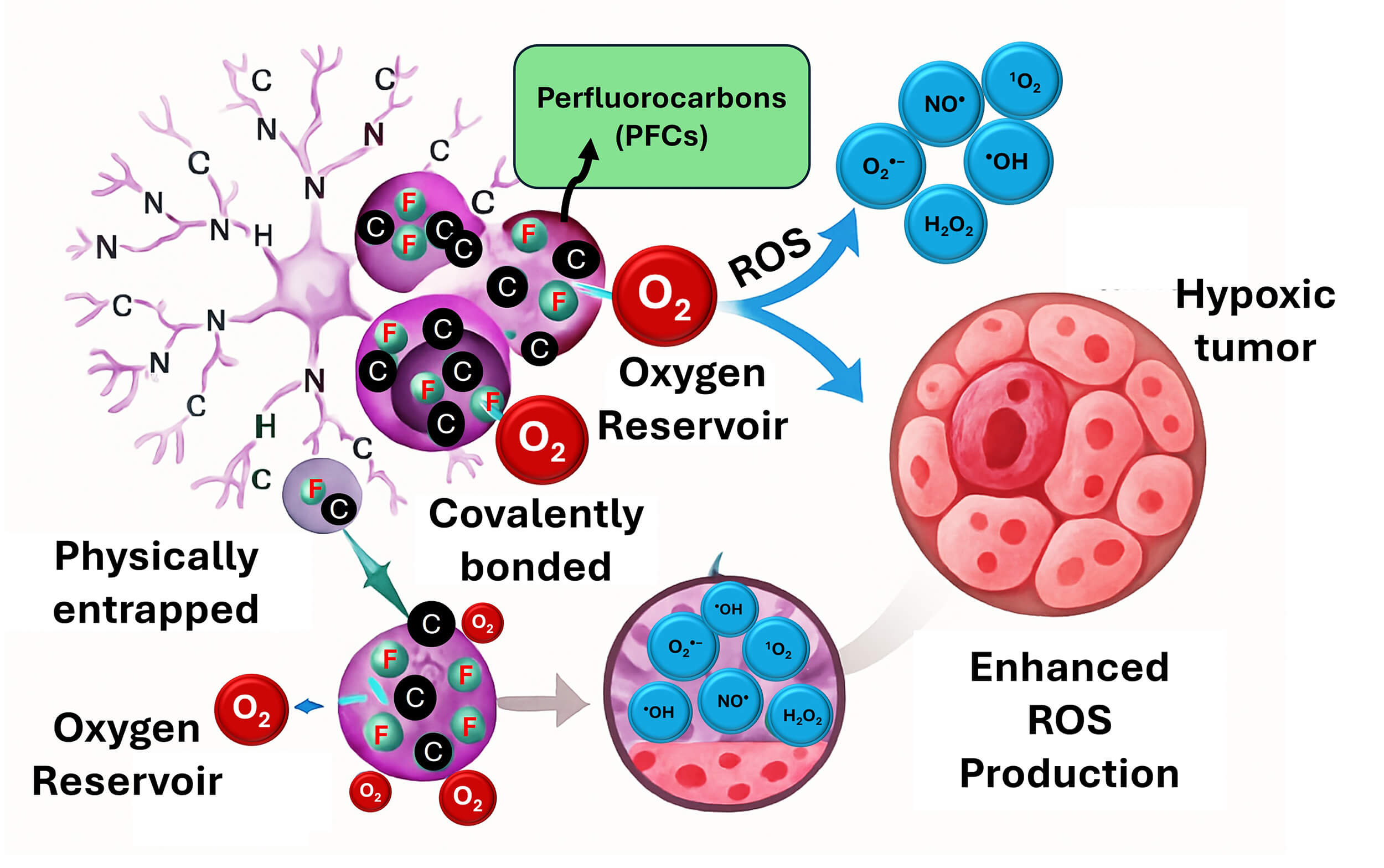 Fig. 1.
Fig. 1.
Illustration the simulated mechanism of perfluorocarbons-loaded dendrimers for cancer treatment and enhanced ROS production. Furthermore, coupling these compounds with dendrimers via covalent bonds or physical attachment can selectively generate oxygen in hypoxic tumors by creating oxygen self-enriching nanomedicines, extending 1O2 lifetime, increasing ROS production, and preserving oxygen. ROS, reactive oxygen species.
The past studies have revealed that the main causes of multiple drug resistance
(MDR) in cancer is intratumoral hypoxia. It can result in a poor prognosis for
patients taking chemotherapy. The development of an MDR inhibitor that reduces
the hypoxic environment is critical for cancer management and treatment [52]. It
has been demonstrated that the hypoxic TME can receive oxygen via a biocompatible
and biodegradable catalase-conjugated iron oxide nanoparticle (Cat-IONP) which
can transform reactive oxygen species into molecular oxygen [52, 53]. Cat-IONP has
initial enzymatic activity equivalent to free catalase, but it provides nearly
three times more long-term enzymatic activity. Cat-IONP has been shown to
drastically inhibit the in vitro expression of hypoxia-inducible factors
in a breast cancer cell line. In hypoxic-cultured cells, dual therapy with
paclitaxel and Cat-IONP enhances drug sensitivity. Cell viability is reduced by
more than two-fold and five-fold compared to cells treated with 80 and 120
In line, catalases are enzymes that catalyze the breakdown of hydrogen peroxide into water and oxygen. These enzymes could be conjugated to dendrimer surfaces to generate oxygen in situ from the high hydrogen peroxide levels found in cancers [53, 54]. This method decreases oxidative stress while increasing oxygen availability for ROS-based cancer therapies, as illustrated in Fig. 2 [54, 55].
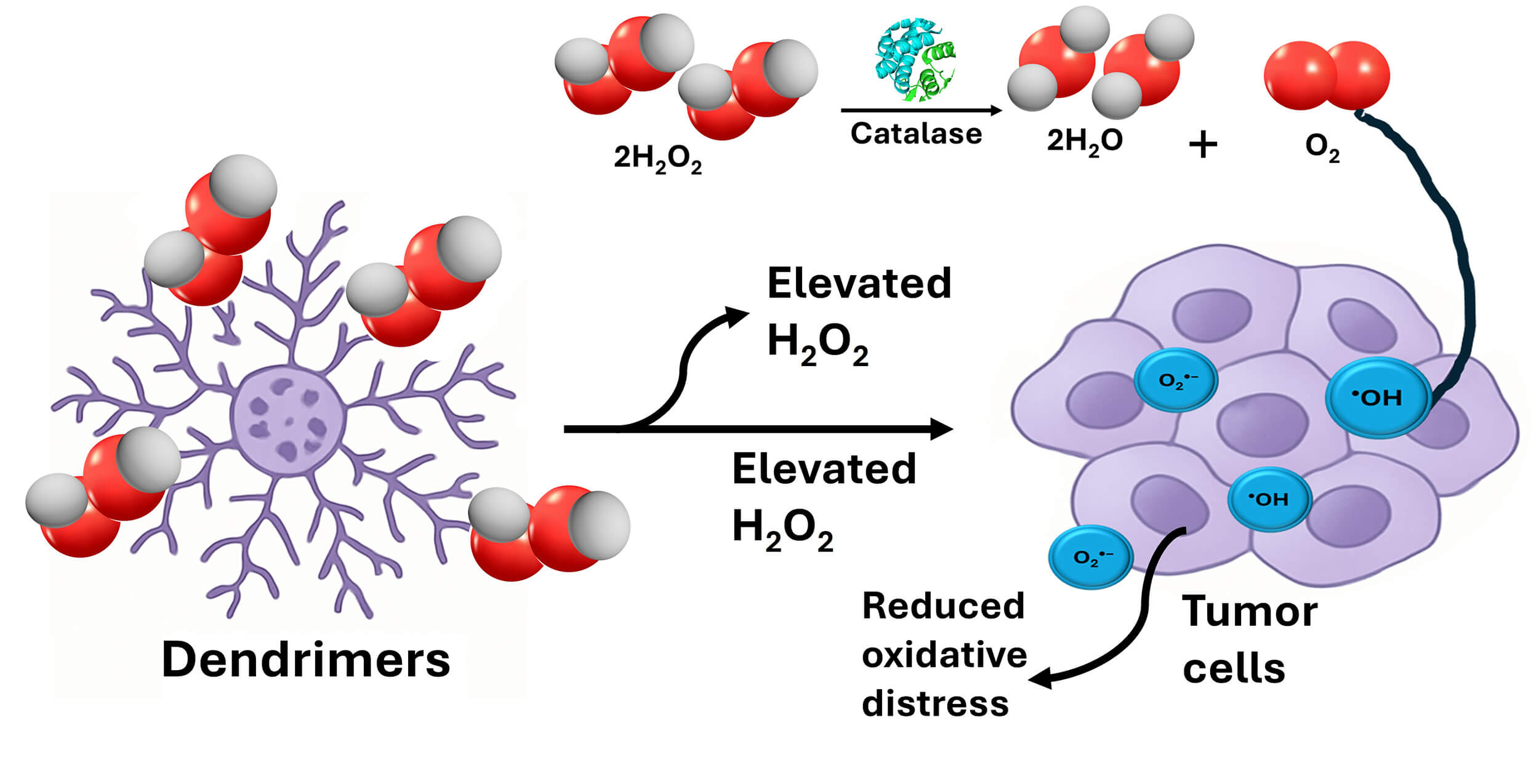 Fig. 2.
Fig. 2.
Schematic diagram showing the hypothesized mechanism of catalase-functionalized dendrimers for reversing hypoxia-induced chemotherapeutic resistance and enhanced ROS production. Furthermore, this approach can overcome these barriers by converting the excessive hydrogen peroxide (H2O2) of TME into molecular oxygen (O2). This in situ oxygen synthesis reduces tumor hypoxia by initiating the degradation of hypoxia-inducible factors (HIF-1) which are essential drivers of treatment resistance. As a result, enzyme functionalization can supply the essential oxygen to enhance ROS-dependent therapies.
PEGylation increases the solubility and circulatory time of dendrimers while decreasing their immunogenicity [56, 57]. Targeting ligands including folic acid, peptides, and antibodies are coupled to dendrimers to enhance their tumor selectivity by identifying overexpressed receptors on cancer cells [58, 59].
Stimuli-responsive linkages that are sensitive to pH, redox conditions, or enzymes allow for the sustained release of oxygen and drugs inside the TME [60]. Among these bonds, diselenide crosslinks have been shown to act as a diffusion barrier for decreasing the premature drug release and enabling on-demand intracellular drug release. As a result, this can lead to larger tumor accumulation than non-cross-linked counterparts. Furthermore, selenium itself can act as an adjuvant for cancer therapy by triggering cell apoptosis, sensitizing cancer cells to chemotherapies, and increasing the selectivity of cancer drugs towards cancer cells [35, 61]. The primary concepts of the surface modification strategy for dendrimers to improve cancer treatment are illustrated in Fig. 3. The two primary dendrimers-based oxygen delivery methods and regulatory issues are summarized in Table 4 (Ref. [62, 63]).
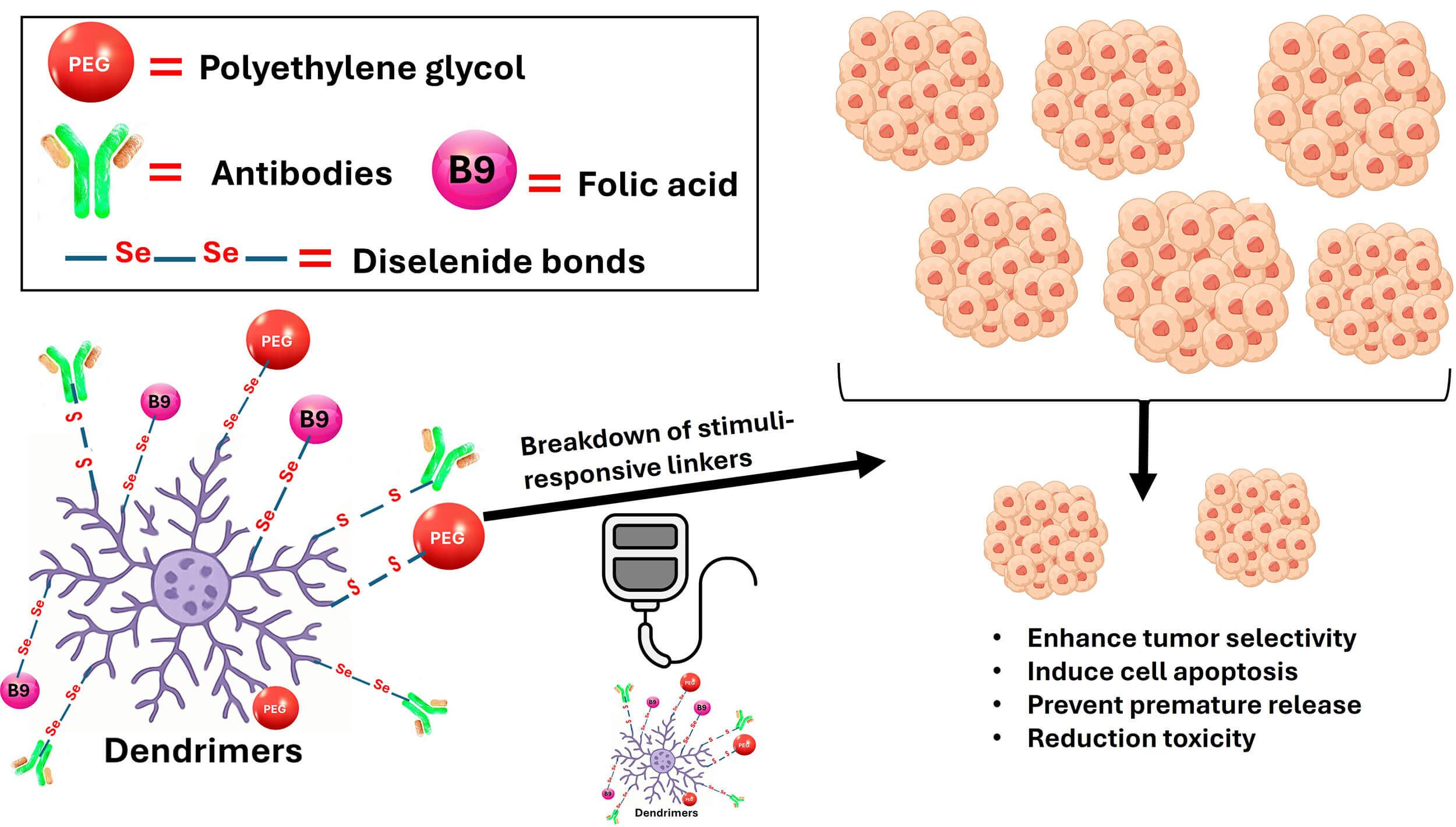 Fig. 3.
Fig. 3.
Suggested surface modification approach of dendrimers to enhance delivery of cancer therapy. PEGylation and active targeting of dendrimers using stimuli-responsive bonds can improve tumor selectivity, reduce rapid drug release, and induce growth inhibition of cancer cells. Key principles may involve reversal surface charge to increase tumor penetration, coating them with targeting ligands (e.g., folic acid) for cell-specific delivery, and introducing catalysts (e.g., iron, copper) to release ROS within the acidic TME. TME, tumor microenvironment.
| Oxygen delivery strategy | Mechanism of action | Critical translational challenge | Regulatory/Mitigation strategy | Reference |
| Perfluorocarbon (PFC) emulsions | Physical dissolution of high volumes; delivered intravenously. | -Rapid clearance by Reticuloendothelial System (RES) (such as liver/spleen). | Nanoscale Engineering: Reducing droplet size and using PFC compounds with shorter biological half-lives. | [62] |
| -Retention and Long Half-life: Long-term visceral toxicity concerns. | ||||
| Enzyme Functionalization/Catalase system | In situ generation at the target site via enzymatic breakdown. | Gas Emboli Risk: Uncontrolled or rapid generation leading to micro-embolisms in the vasculature. | Precise control over catalyst (catalase) activity through robust nanocapsule encapsulation. | [62, 63] |
ROS may have both positive and negative impacts on cancer biology. For example, they control homeostasis and signaling at low to moderate levels. However, they result in fatal oxidative damage at high concentrations [64, 65]. The high ROS baseline in cancer cells makes them vulnerable to further oxidative damage. This strategy has been applied in PDT, chemodynamic therapy, radiotherapy, and oxidative therapy.
Since 1976, the PDT widely used to treat patients with bladder cancer, several photosensitizers have been developed for cancer treatments at a specific light wavelength. When light is absorbed, the photosensitizer becomes excited and produces cytotoxic ROS through type I (e.g., radical and radical anion species such as H2O2, O2•–, and •OH) or type II (nonradical 1O2) photochemical reactions [35, 66]. These reactions involve electron transfer or energy transfer from excited photosensitizers to molecular oxygen. Nanomedicines have been extensively studied to optimize PDT because they can efficiently transfer photosensitizers to tumor sites via the EPR effect, followed by site-specific activation of the photosensitizer using light. Furthermore, various efforts have been made to increase depth limitation and ROS generation capability by employing multifunctional nanoparticles such as two-photon excitation nanoparticles, up-conversion nanoparticles, and organic/inorganic hybrid nanoparticles [67, 68, 69]. The oxygen reliance of PDT has resulted in another well-known issue with its applicability to hypoxic tumoral areas, as tissue oxygen is an essential substrate in type II reactions. Because oxygen is not necessary for photothermal therapy (PTT), combining PDT with PTT could serve as an effective method to solve this challenge [70, 71].
PDT generates singlet oxygen and other ROS to cause cancer cell death by administering photosensitizers and then irradiating the area with particular light wavelengths [71]. Its effectiveness is largely dependent on oxygen availability, which is frequently impaired in hypoxic tumors [72].
In preclinical models, dendrimer-based co-delivery of photosensitizers and oxygen carriers overcomes hypoxia-induced PDT resistance by improving ROS production and cancer cell apoptosis [72, 73]. The potential function of dendrimers as nanocarriers for oxygen and photosensitizers is depicted in Fig. 4.
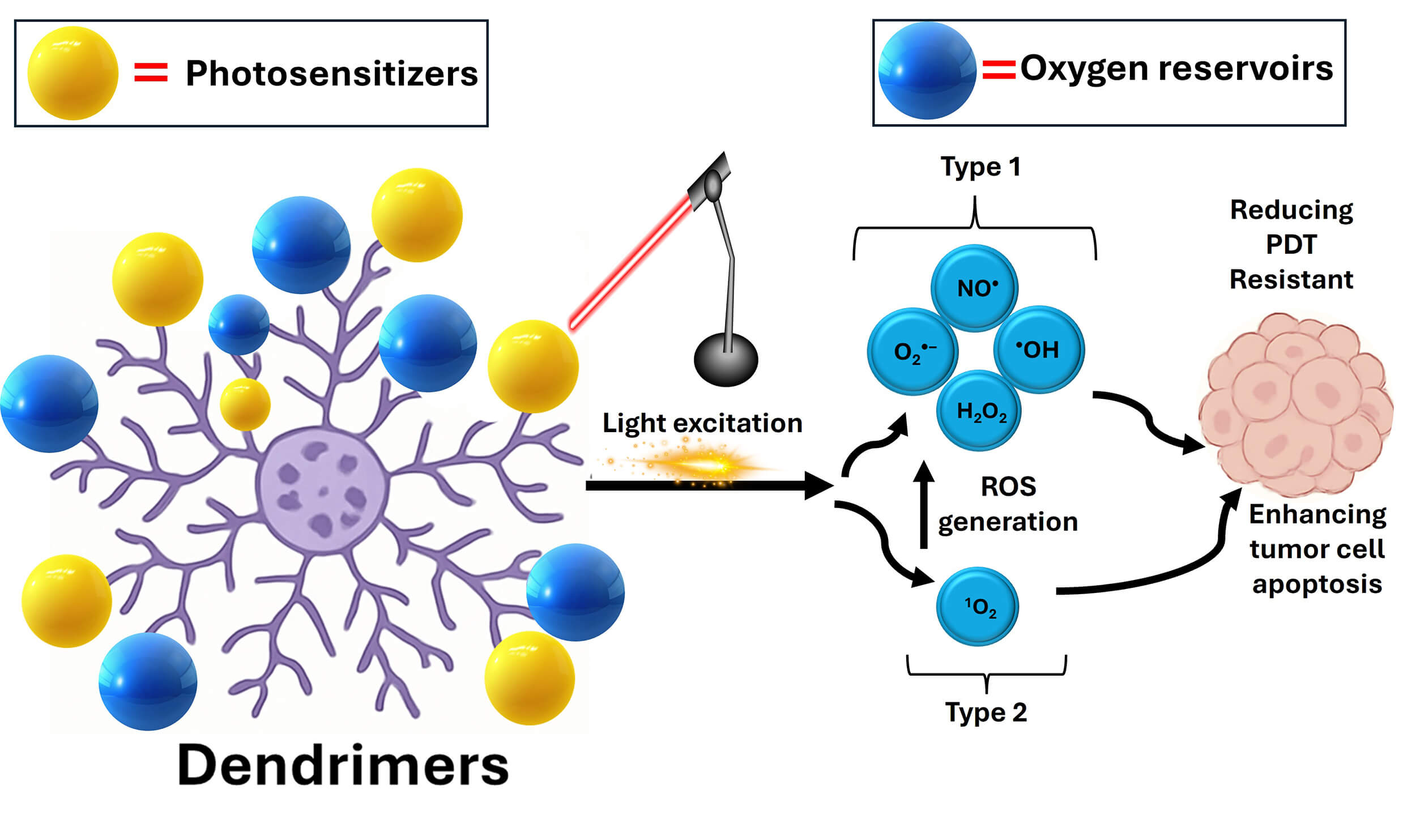 Fig. 4.
Fig. 4.
The possible beneficial roles of dendrimers as nanocarriers for photosensitizers and oxygen carriers. These nanomedicines could be used to mitigate hypoxia-induced PDT resistance by increasing ROS production and oxygen supply, lowering oxygen consumption by suppressing the mitochondrial respiration, suppression the activity of reducing agents such as glutathione levels to prevent the neutralization of ROS, blocking the efflux pumps such as P-glycoprotein, inhibition the HIF-1 expression, and promoting cancer cell apoptosis. PDT, photodynamic therapy.
CDT uses transition metal ions to induce Fenton or Fenton-like reactions which transform endogenous hydrogen peroxide into highly cytotoxic hydroxyl radicals [74, 75, 76]. This treatment is tumor-specific due to the acidic and hydrogen peroxide-rich environment of cancers [77]. Metal ion-loaded dendrimers catalyze intratumoral Fenton reactions which generate cytotoxic ROS selectively in tumors while exhibiting smaller off-target effects. Dendrimers act as carriers for photosensitizers (such as porphyrins and chlorins) which produce singlet oxygen when activated by light of PDT as represented in Fig. 4 [78, 79]. Furthermore, dendrimers can carry metal ions (Fe2+, Cu+, Mn2+) and induce Fenton or Fenton-like reactions within tumors. (as illustrated in Fig. 5) For example, core-shell tecto dendrimers (CSTD) were developed by supramolecular assembly of G5 core dendrimers and G3 shell dendrimers that had been modified with pyridine for Cu (II) complexation, arginine-glycine-aspartic acid (RGD) peptide for glioma targeting, and dermorphin for blood-brain barrier (BBB) crossing. The obtained modularly designed Cu (II)-complexed CSTDs with acetyl termini complexes could induce a Fenton-like reaction inside C6 cells (a Rattus norvegicus glioma cell line) to generate cytotoxic •OH via H2O2 decomposition and increase the intracellular ROS level at higher concentrations of Cu. In vivo intravenous injection of these complexes effectively reduced orthotopic glioblastoma indicating effective CDT across the BBB [80, 81, 82, 83].
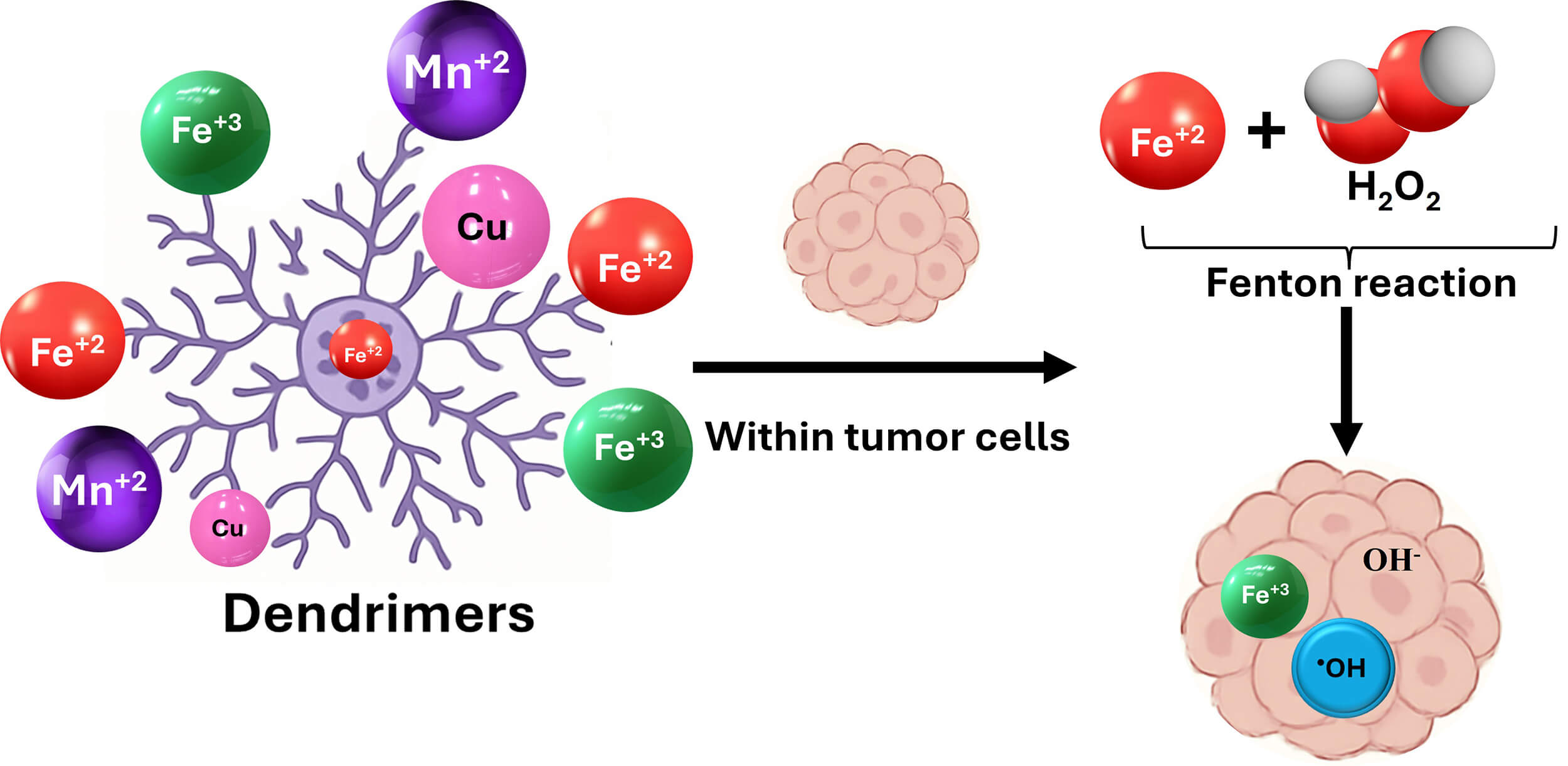 Fig. 5.
Fig. 5.
Schematic representation of suggested ROS-mediated strategy for CDT-based dendrimers. The main mechanisms for this approach may involve the enhancement production of ROS for CDT by selectively delivering Fenton-active metals (Fe2+, Cu+, Mn2+) to tumors which utilizing the acidic TME to convert endogenous hydrogen peroxide into highly potent hydroxyl radicals, as well as depleting antioxidants such as glutathione to promote oxidative stress. CDT, chemodynamic therapy.
Oxidation therapy is a novel method of treating cancer by specifically increasing the oxidative stress of cancer cells above the fatal threshold. To disrupt the redox balance of cancer cells, several therapeutic agents have been investigated to stimulate oxidative stress by generating ROS (e.g., piperlongumine, cinnamaldehyde, and xanthine oxidase) or blocking antioxidant pathways [35, 84]. However, since low to moderate levels of ROS promote cell development, oxidative stress should be increased to a level that kills tumor cells. Thus, there were great expectations for merging the methodologies of increasing ROS levels and suppressing antioxidant ability to ensure tumor eradication. To do this, a polymeric nanoreactor (NR) containing glucose oxidase (GOD) was developed to produce H2O2 by glucose catalysis. Therapeutic NRs (theraNRs) with a vesicular structure were used to stimulate the enzymatic process of producing oxidative stress locally in response to the tumor microenvironment. TheraNRs based on diblock copolymers contain PEG copolymerized with phenyl boronic ester or piperidine-functionalized methacrylate (P (PBEM-co-PEM)). Hydrophobic PPEM and PPBEM segments were added to induce sensitivity to extracellular tumor pH (pH 6.5–6.8) and H2O2 level, respectively. When theraNRs reach the cancer tissues via the passive targeting approach, pH-responsive PPEM segments transform into hydrophilic by protonation, enabling the infiltration of small molecular molecules (glucose and oxygen) for enzymatic catalysis via the permeable membrane of theraNRs within cancer tissue [39, 85]. Therefore, H2O2 is then produced by GOD-mediated glucose oxidation and causes theraNRs to break down resulting in the generation of quinone methide (QM) byproducts by PPBEM segment destruction. QM is known to decrease GSH, one of the most prevalent antioxidants in the intracellular tumor environment. Thus, theraNRs can successfully produce oxidative stress not only by generating H2O2 during consuming nutrients, but also by depleting GSH-mediated ROS scavenging resulting in cancer cell death and tumor eradication in vivo. Oxidation therapy can result in synergistic therapeutic effects when combined with chemotherapy, photodynamic therapy, and radiotherapy. This is due to the fact that the photolysis of iron-containing materials may convert the H2O2 molecule to form reactive hydroxyl radicals. Ionizing radiation produces ROS by water radiolysis which destroys DNA resulting in cancer cell death [39]. The stabilization of radiation-induced DNA damage that mediated by oxygen can increase the potency of treatment. Hypoxia impairs this stabilization resulting in radioresistance. Oxygen-generating dendrimers can improve radiosensitivity by reversing hypoxia, inducing DNA damage, and tumor suppression in vivo [39, 85, 86].
The selection of drug loading strategy determines release kinetics that profoundly affects the clinical performance and regulatory pathway of dendrimers as shown in Table 5 (Ref. [40, 87]).
| Loading mechanism | Drug-Carrier interaction | Release kinetics (impact) | Translational advantage | References |
| Non-Covalent encapsulation | Physical entrapment within internal void spaces; electrostatic interactions. | Fast/Burst Release: Release dictated by diffusion. Poor control suitable for systemic or rapid-onset drugs. | Significant improvement in the solubility and bioavailability of highly hydrophobic active pharmaceutical ingredients. | [40, 87] |
| Covalent conjugation | Chemical bond (e.g., ester, hydrazone) between drug and surface groups (prodrug). | Controlled/Triggered Release: Release relaying on the cleavage rate of the linker (e.g., pH, enzyme, reduction sensitivity). | Enabling highly specific targeting and minimizing systemic toxicity by only releasing the active drug at the pathological site. | [40, 87] |
In-vitro and in-vivo comparative studies are critical in biomedical research, especially for drug discovery and development. They provide a full understanding of a drug effects by combining the distinct capabilities of both methods. Furthermore, they can bridge the gap in understanding which is critical for validating and translating to a clinical context, as well as mandatory for regulatory and bioequivalence requirements [88, 89, 90, 91, 92]. Table 6 (Ref. [88, 89, 90, 91, 92]) summarizes key variables and outcomes, based on data from recent drugs and oxygen delivery studies.
| Parameter | Dendrimer type/generation | Oxygen carrier/drug encapsulated | Loading/Conjugation strategy | The main in vitro/in vivo outcomes | References |
| Generation | G2, G3 PAMAM | Curcumin (Drug) | Encapsulation/Conjugation (e.g., PEG-citrate dendrimers) | In Vitro: Significant HIV-1 inhibition (50% up to 90% reduction of HIV proliferation at 12 µM and 60 µM, respectively) for curcumin loaded in G2 and G3 PEG-citrate dendrimers. | [88] |
| Oxygen carrier | G4–6 PAMAM | ROS | PAMAM dendrimers contain a 2-carbon ethylenediamine core and primary amino groups on the surface | In Vitro: Enhanced the time of: (1) release of ROS (highest at |
[89] |
| Drug loading | G4 PAMAM (Folate-conjugated) | Curcumin (Drug) | Encapsulation, often -responsive | In Vitro: Drastic improvement of growth inhibitory effect (more than 67%) in cancer cell lines (MG-63 and HT-29) for Curcumin-based-G4 PAMAM complex. Increased the cellular uptake (up to 63.7%) in Folate Receptor-expressing cells. | [90] |
| Toxicity | G2 vs G3 Citric Acid Dendrimers | N/A (Carrier Only) | Chemical Synthesis | In Vitro: Reduction in the toxicity to the PC12 cell line (derived from rat adrenal medulla) for G2 dendrimers compared with G3, with CC50 values of 1243 and 784 µg/mL, respectively. In the Vero cell line (derived from kidney epithelial cells), no significant variation in toxicity between G2 and G3. | [91] |
| Toxicity | Cationic vs Anionic/PEGylated Dendrimers | N/A (Carrier Only) | Surface Modification (e.g., PEGylation) | In Vivo: Cationic dendrimers exerting more cytotoxic effect and haemolytic reaction than anionic or PEGylated dendrimers. In vivo doses up to 2.56 g/kg and 1.28 g/kg iv resulting in no toxicity, death, or abnormal blood chemistry including blood urea nitrogen levels and alanine transaminase activity. | [92] |
ROS-based (or ROS-responsive) dendrimers are a sort of smart nanocarrier that releases its cargo only in settings (such as tumors or inflamed tissues) with excessively high ROS levels [93, 94, 95]. The toxicity profile of these systems is a balance between the inherent toxicity of the core dendrimer (typically cationic PAMAM) and the favorable reduction in off-target effects induced by the ROS-sensitive release mechanism. While the biodistribution and clearance profiles are vital to verify targeting, enable passive targeting (EPR effect), control blood circulation time, and optimize dosing schedule [96, 97]. Table 7 (Ref. [93, 94, 95, 96, 97]) represents a comprehensive evaluation of the toxicity, biodistribution, and clearance characteristics of ROS-based dendrimers.
| Parameter | Cationic dendrimers (precursors) | ROS-Responsive dendrimers (modified) | Rationale for change | Reference |
| Biodistribution | Rapid accumulation in RES (Liver, Spleen) and non-target organs; short half-life. | Extended circulation time (reduced RES uptake) due to surface masking (e.g., PEGylation or charge-reversal moieties). | Modification (e.g., PEGylation or neutral surface), preventing opsonization, and promoting the EPR effect for tumor targeting. | [93] |
| Toxicity (General) | High cytotoxicity (hemolysisand cell death) in a generation-dependent manner (G4+) due to membrane disruption. | Low systemic toxicity; site-specific and restricted cytotoxicity (only at high ROS sites). | The ROS-sensitive linker (e.g., thioketal, boronic ester) shields the toxic core and releasing the payload only at pathologically high ROS concentration (times normal). | [94, 95] |
| Clearance | Primarily renal excretion for low generations; hepatic for larger aggregates. | Designed to degrade into smaller, non-toxic, renally-clearable fragments upon ROS cleavage in the target tissue. | Biodegradability decreasing long-term organ accumulation of the entire dendrimer structure and addressing a key safety concern. | [96] |
| Biocompatibility | Low due to membrane disruption, pro-inflammatory cytokine induction, and non-specific cell binding. | Improved biocompatibility due to prevention of non-specific cell binding by charge neutralization/shielding and confining activity. | Surface modification (e.g., acetylation, PEGylation) of terminal amines significantly reducing the cytotoxicity by masking positive charge. | [97] |
Biocompatibility is the ability of a drug to complete its intended function with a suitable host reaction in a specific application. Thus, it is crucial to prevent immune rejection, avoid adverse reactions, manage degradation products, and maintain stability of ROS-based dendrimers [97, 98]. Regulatory concerns refer to the legal and administrative standards established by authorities such as the Food and Drug Administration (FDA) in order to obtain approval for clinical trials and market access [99]. That is essential to ensure the quality control, classify the pharmaceutical products (drug, a biologic, a medical device, and cosmetics or a combination product), ensure public safety and maintain trust in novel nanomedicine such as ROS-based dendrimers as described in Table 8 (Ref. [94, 96, 97, 98, 99]).
| Aspect | Current Status/observation | Regulatory considerations & challenges | Reference |
| ROS-Cleavage mechanism | Relying on highly selective linkers (e.g., thioketal, aryl boronic esters) that considered stable at physiological ROS and efficiently removed at pathological ROS levels. | Premature Release: Requirement for extensive stability and release studies to prevent in vivo drug leakage in systemic circulation. | [94, 98] |
| Biodegradability & metabolites | Designing intentionally biodegradable ROS-based dendrimers (e.g., dendrons cleave off upon ROS exposure) to limit long-term bioaccumulation. | Toxic Metabolites: Ensuring non-toxicity, non-immunogenicity, and rapidly elimination via the renal or hepatic pathways of all degradation products (metabolites). | [96, 98] |
| Batch-to-Batch consistency | Stimuli-responsive moieties adding synthetic complexity and making consistent production of identical batch materials challenging. | Quality Control (QC): Creating standardized Good Manufacturing Practice (GMP) protocols to ensure highly reproducible size, surface charge, and ROS-response kinetics for every batch. | [97] |
| Long-Term effects | Limited data on the chronic safety profile of dendrimer degradation fragments after accumulated in the RES over time. | Chronic Toxicity Studies: Requirement for comprehensive long-term toxicity and carcinogenicity studies before approval for chronic diseases like cancer, especially with novel linkers. | [99] |
Future research should focus on combining multimodal imaging for real-time monitoring and developing dendrimers that can be used to deliver combination therapies involving immunomodulation. Dendrimer-based nanocarriers formulated for active oxygen delivery and ROS regulation offer a promising method to overcome tumor hypoxia and improve cancer therapy effectiveness [99, 100]. Their adaptable architecture enables multi-functionalization with oxygen carriers, catalytic enzymes, photosensitizers, and targeting ligands. Ongoing multidisciplinary research involving nanotechnology, oncology, and pharmacology will be required for translating these advances into therapeutic medicines [101, 102, 103]. As a result, this review focuses on the latest developments in dendrimers for cancer treatment including active oxygen delivery and ROS regulation. This study also investigates the dendrimer chemistry and functionalization techniques, how the tumor microenvironment affects nanoparticle design, therapeutic applications, obstacles, and potential futures.
Despite the results of the above preclinical study are promising, this discipline is still in its early stages and is therefore inadequate for real practical use. Furthermore, the ROS-based techniques presented in this research are not yet included in the appropriate therapeutic modalities for US Food and Drug Administration-approved nanomedicines. As a result, considerable further development and investigation are planned within the context of bench-to-bedside translation. To maximize the translational potential of dendrimers, multidisciplinary attempts to enhance the reproducibility of their preparation and evaluation in preclinical studies are required. The obstacles in the preparation of ROS-based dendrimers include scaling up to produce them with uniform physicochemical properties such as size, surface property, and morphology [104]. While seeking to avoid complex synthetic strategies for developing ROS-based dendrimers during the design phase, using high-precision microfluidic and nano-molding techniques may be worthwhile to reduce batch-to-batch variability [105]. To avoid potentially contradicting results from the same nanocarriers, cell lines and experimental procedures from cell culture to therapeutic efficacy and safety assessment used in research of ROS-based dendrimers must be standardized. The current primary focus of preclinical research is to assess the therapeutic efficacy of ROS-based dendrimers for proof-of-concept; however, detailed in vivo evaluations including long-term toxicity cannot be ignored. When harnessing ROS generated by dendrimers, it is critical to accurately evaluate the amount of ROS generated, their long-term systemic effect, and any off-target side effects of ROS-generating nanomaterials and sensitizers in PDT [105]. The better in vitro arrangements such as organ-on-a-chip appear to be advantageous for accelerating the clinical translation of dendrimer-based nanomedicines by predicting biological activities and selecting the best candidates for subsequent in vivo studies. To obtain more valuable clinical in vivo data, the widely used human xenograft tumor models (derived from cultured cancer cell lines and inoculated in immune-compromised mice) should be replaced by models that are more closely related to the actual disease in patients such as patient-derived xenograft tumors implanted in genetically modified or similar humanized mice [105, 106]. For developing ROS-mediated methods for practical medicinal purposes, it is essential to consider potential cancer heterogeneity in redox state such as higher intracellular glutathione and ROS levels in cancer cells, as this may provide future barriers. Overall histological complexity and variety of cancer have already been identified in preclinical investigations and clinics emphasizing the significance of tailored therapy choices [106]. Given increasing confirmation of immune-boosting effects in cytotoxic ROS generated during PDT, integrating this approach with immunotherapy which had been thought to be an internally targeted cancer treatment modality is interesting. To guarantee future success in this field, additional effort should be directed to determining the role of specific types of ROS that act normally or under pathophysiological condition. Since the umbrella term ROS for active small molecule oxidants is commonly accepted for identifying their similarities, it has been critiqued for being generic and ambiguous. Furthermore, it is uncommon for relevant quantitative data to be reported in the literature [106]. Thus, technological improvements in developing molecular probes for each oxidant, as well as reliable in vivo detection procedures for each ROS are critical for better shaping the novel paradigm for cancer treatment based on dendrimers for the ROS-mediated strategy. The most common challenges (scale-up, toxicity, and target efficiency) were well investigated in the literature. While Table 9 (Ref. [107, 108, 109, 110, 111]) discusses less systematically addressed barriers that have a substantial impact on the clinical and commercial success of dendrimer-based therapies.
| Translational challenge | Description/Mechanism | Unique impact on dendrimers translation | Overlooked solution/mitigation strategy | Reference |
| Excipient-induced toxicity | Toxicity stemming originating from necessary formulation components (surfactants, co-solvents, lipids, etc.). | Mimicking nanocarrier toxicity, leading to misinterpretation of safety data and delaying in development, as well as complicating regulatory approval. | “Stealth Excipient” Development: Focusing on using high-purity, de novo designed excipients with established toxicity profiles in humans or GRAS (Generally Recognized as Safe) status. | [107, 108] |
| Clinical trial end-point mismatch | Clinical trials that mainly focusing on enhanced efficacy without safety, leading to failure of drug approval. | Creation an issue for approval due to limited patient cohorts. | Developing of “Hybrid” Clinical Trials: Trials explicitly designed to show non-inferior efficacy and significantly improved safety/quality of life, with appropriate endpoints. | [108, 109] |
| Post-Market intellectual property (IP) hurdles | The IP landscape for dendrimers includes layers of patents (drug, formulation, manufacturing process, and even excipients), leading to “patent thickets”. | Making the entry of generic/biosimilar extremely difficult, limiting patient access and competition post-approval. | Strategic Patent Pooling/Licensing: Encouraging collaborative IP strategies and clear regulatory pathways for nanomedicine generics based on robust in vitro and in vivo characterization. | [110, 111] |
Conceptualization, methodology, validation, formal analysis, investigation, writing original draft preparation, writing review and editing, supervision, project administration, and funding acquisition were all perfomed by MA.
Not applicable.
Not applicable.
This research received no external funding.
The author declares no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





