1 Department of Pharmacy, Shizuoka Cancer Center, 411-8777 Shizuoka, Japan
2 Faculty of Pharmaceutical Sciences, Shonan University of Medical Sciences, 244-0806 Kanagawa, Japan
3 Graduate School of Pharmacy and Pharmaceutical Science, Juntendo University, 279-0013 Chiba, Japan
4 Faculty of Pharmacy, Laboratory of Clinical Pharmacology, Juntendo University, 279-0013 Chiba, Japan
5 Department of Pharmacy, Showa University Hospital, 142-8666 Tokyo, Japan
6 Department of Pharmacy, Kyushu University Hospital, 812-8582 Fukuoka, Japan
7 Department of Pharmacy, Juntendo University Urayasu Hospital, 279-0021 Chiba, Japan
Abstract
Prophylactic opioid rescue therapy is often used to manage cancer-related pain. However, the effect of prophylactic rescue therapy in suppressing increases in breakthrough pain due to body movements has not yet been investigated. Therefore, this study aimed to compare the efficacy and safety of prophylactic rescue therapy before and after administration.
This multicenter, retrospective survey was conducted in Japan. Participants were patients with cancer who received prophylactic strong opioid rescue therapy for the first time. The primary endpoint was the suppression of an increase in pain, assessed using a Numerical Rating Scale (NRS), before and after administration of prophylactic rescue due to body movement.
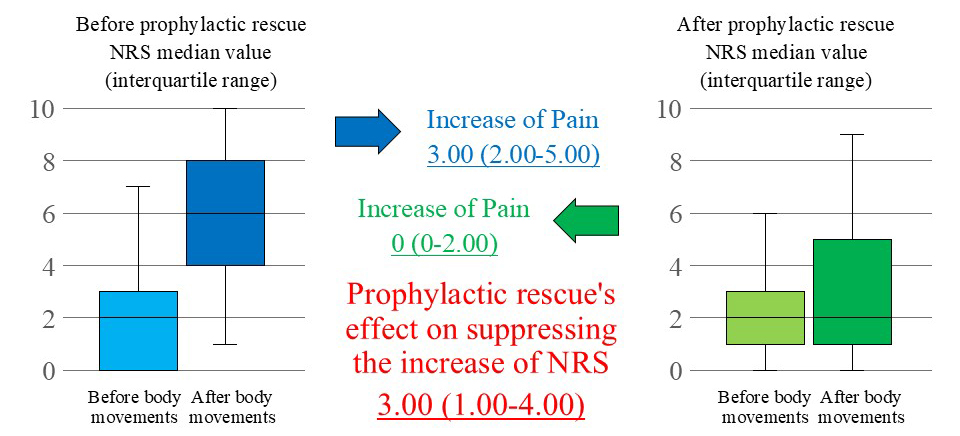
The overall analysis (193 cases) showed that prophylactic rescue suppressed the increase in the median NRS value by 3.00 (interquartile range: 1.00–4.00, p < 0.001). In addition, subgroup analysis by purpose (meals/bathing/rehabilitation/radiation therapy/others) also showed a significant reduction in the increase in NRS, with all categories exhibiting a decrease of 2.00 or more.
Prophylactic opioid rescue with appropriate patient selection can effectively suppress breakthrough pain caused by body movement.
Graphical Abstract
Keywords
- analgesics, opioid
- pain management
- breakthrough pain
- cancer pain
Pain experienced by patients with cancer can be classified into two types: persistent pain, which lasts for most of the day, and breakthrough pain, which is a transient exacerbation triggered by body movement or other factors. As a basic method of using opioid analgesics for these two types of pain, it is recommended that a daily opioid (sustained-release oral preparation or continuous injection) be used regularly to control persistent pain. In contrast, rescue medications (immediate-release oral preparation or additional injection) should be used to control breakthrough pain [1]. However, even with fast-acting formulations developed specifically for rescue, a time lag of 30–60 min is observed before the drug takes effect [2], requiring patients to endure pain during that period.
In clinical practice, if a patient has a history of conditions likely to cause sharp pain during rehabilitation, prophylactic opioid rescue may be administered beforehand [3]. Mercadante [4] also reported that prophylactic opioid rescue to prevent breakthrough pain impacts nutritional status and quality of life. However, for prophylactic rescue to be effectively used in clinical practice, patients must receive proper education regarding the appropriate timing of drug administration.
When prophylactic rescue is administered, potential issues include the risk of inducing somnolence or nausea, which may negatively affect the intended activity, as well as an increase in the total opioid dosage, potentially leading to dose-dependent adverse events such as constipation or delirium [5, 6, 7]. Additionally, opioid dependence and use disorders have become serious global concerns, necessitating careful research into prophylactic rescue [8, 9, 10]. Regarding safety, a single-center retrospective observational study in Japan showed no significant differences in the incidence of adverse events between prophylactic rescue and rescue administered after breakthrough pain [11]. However, the efficacy of prophylactic rescue in suppressing breakthrough pain and its effectiveness in targeting body movements have not yet been investigated. Therefore, we conducted a real-world analysis of the efficacy and safety of prophylactic rescue in a Japanese multicenter retrospective observational study.
This multicenter, retrospective survey was conducted at 12 facilities in Japan. The 12 facilities’ locations were as follows: 2 facilities in the north of Japan (Hokkaido region and Tohoku region), 8 facilities in the center of Japan (Kanto region, Chubu region and Kinki region), and 2 facilities in the south of Japan (Chugoku region, Shikoku region and Kyushu region). Pharmacists affiliated with the Japanese Society for Pharmaceutical Palliative Care and Sciences collected electronic medical record data from their respective facilities, which were then integrated and analyzed at the Shizuoka Cancer Center.
This study included patients with cancer who were administered strong opioid prophylactic rescue agents (morphine tablets, powder, oral solution, and injection, and oxycodone powder, oral solution, and injection, hydromorphone tablets, hydromorphone injection, and fentanyl injection) for the first time between June 2017 and September 2023. The following patients were excluded from this study: ① non-cancer patients; ② patients with an interval of more than one week between the pre- and post-prophylactic rescue assessments; ③ patients who received prophylactic rescue for postoperative pain; ④ patients who received rescue treatment for conditions other than pain, such as dyspnea; ⑤ patients who received prophylactic rescue for end-of-dose failure; ⑥ patients whose base opioid dose was increased within 3 days before prophylactic rescue; ⑦ patients whose base opioid dose was increased before and after prophylactic rescue; ⑧ patients whose change in pain before and after the prophylactic rescue was not evaluated using the Numerical Rating Scale (NRS).
The primary endpoint was defined as the suppression of an increase in pain using the NRS before and after the implementation of prophylactic rescue due to body movement (meals/bathing/rehabilitation/radiation therapy/other). This NRS was rated on an 11-point scale, where 0 represents no pain and 10 represents the worst possible pain [12]. We investigated the change in pain due to body movement before and after prophylactic rescue (both assessments were conducted within one week) and calculated the difference between the fluctuation values before and after prophylactic rescue. If multiple assessments were conducted, the assessment closest to the time when the prophylactic rescue was conducted was adopted.
As secondary evaluation items, the occurrence and severity of adverse events were investigated using common terminology criteria for adverse events Ver. 5.0. The adverse events that occurred before prophylactic rescue and those that occurred or changed in severity after prophylactic rescue were recorded separately. In addition, the effect on body movements requiring prophylactic rescue (meals/bathing/rehabilitation/radiation therapy/others) was evaluated on a three-level scale: effective, no change (ineffective), and worsening. The case collectors (pharmacists affiliated with the Japanese Society for Pharmaceutical Palliative Care and Sciences) at each facility conducted a composite assessment based on patient-reported outcomes, nursing notes, and physician judgment in electronic medical records. If the body movements were more actively pursued after prophylactic rescue, they were evaluated as effective; if they were pursued passively or discontinued after prophylactic rescue, they were evaluated as worsening. If the severity of adverse events or their impact on body movements was not recorded in the electronic medical records, they were classified as unknown.
The following patient background data were collected: age, sex, height, weight, type of cancer, stage and presence or absence of metastasis, medical history, general condition, presence or absence of renal impairment, presence or absence of hepatic impairment, components and dosage of prophylactic rescue, components, and dosage of base opioid, purpose of movement requiring prophylactic rescue, time from administration of prophylactic rescue to movement, concomitant medications, and blood test values (immediately before administration of prophylactic rescue), including blood urea nitrogen (BUN), serum creatinine (Cr), aspartate aminotransferase (AST), and alanine aminotransferase (ALT). Additionally, drugs used in combination with the prophylactic rescue were investigated. The conversion between morphine oral: morphine injection, oxycodone oral: oxycodone injection: hydromorphone oral: hydromorphone injection, fentanyl patch: fentanyl injection: tapentadol oral was performed at a ratio of 30:15:20:15:6:1.2:1:0.3:100.
We conducted the Wilcoxon signed-rank test on the same patients to investigate the effect of prophylactic rescue on suppressing the increase in NRS scores. Before conducting the Wilcoxon signed-rank test, we checked that the data did not follow a normal distribution. Additionally, we compared the incidence of adverse events before and after the prophylactic rescue using Fisher’s exact test. For patient background factors, items for which a mean value or median value was calculated were subjected to the Shapiro-Wilk test; where the data followed a normal distribution, the mean was reported, and where it did not, the median was reported. All statistical tests were performed using the Bell Curve for Excel (Social Survey Research Information Co., Ltd. Shinjuku, Tokyo, Japan), with the level of statistical significance set at 5%.
A total of 5385 patients were enrolled at participating facilities, and 5192 patients were excluded; therefore, data from 193 patients were analyzed (Fig. 1).
 Fig. 1.
Fig. 1.
Flow diagram of patient selection. The enrolled patients and the exclusion criteria are described.
The background factors of the analyzed patients are shown in Table 1. As
p
| Background factors | n = 193 |
| Age; median value (interquartile range) | 68 (61–74) |
| Sex (male/female) | 117/76 |
| Cancer type (gastrointestinal/lung/breast/head and neck/hematological malignancy/other) | 61/52/19/22/9/30 |
| Performance status (0/1/2/3/4/unknown) | 25/57/52/46/12/1 |
| Body mass index; median value (interquartile range) | 20.4 (18.3–23.2) |
| Blood urea nitrogen; median value (interquartile range) | 15.0 (11.6–19.7) |
| Serum creatinine; median value (interquartile range) | 0.69 (0.54–0.86) |
| Aspartate aminotransferase; median value (interquartile range) | 24 (17–34) |
| Alanine aminotransferase; median value (interquartile range) | 18 (12–30) |
| Body movements that require prophylactic rescue (meals/bathing/rehabilitation/radiation therapy/other) | 62/16/19/74/22 |
| Time (minutes) from administration of prophylactic rescue to movement (5–15/16–30/31–60/unknown) | 31/130/28/4 |
| Daily opioid preparation type (morphine oral/morphine injection/oxycodone oral/oxycodone injection/hydromorphone oral/hydromorphone injection/fentanyl patch/fentanyl injection/tapentadol oral/none) | 22/6/73/17/5/1/6/2/8/53 |
| Dosage of daily opioid (morphine oral equivalent) mg/day; median value (interquartile range) | 15.0 (0–30.0) |
| Prophylactic rescue opioid preparation type (morphine oral/morphine injection/oxycodone oral/oxycodone injection/hydromorphone oral/hydromorphone injection/fentanyl injection) | 50/7/103/17/13/1/2 |
| Dosage of prophylactic rescue opioid (morphine oral equivalent) mg/dose; median value (interquartile range) | 3.8 (3.8–5.0) |
Table 2 shows the overall analysis of prophylactic rescue’s effect on
suppressing the increase of NRS due to body movement median value by 3.00
(interquartile range: 1.00–4.00, p
| Subgroup | n | Before prophylactic rescue | After prophylactic rescue | Prophylactic rescue’s effect on suppressing the increase of NRS due to body movement median value (interquartile range) | p | |||||
| NRS median value (interquartile range) | NRS median value (interquartile range) | |||||||||
| Before body movements | After body movements | Increase of NRS | Before body movements | After body movements | Increase of NRS | |||||
| All | 193 | 2.00 (0–3.00) | 6.00 (4.00–8.00) | 3.00 (2.00–5.00) | 2.00 (1.00–3.00) | 2.00 (1.00–5.00) | 0 (0–2.00) | 3.00 (1.00–4.00) | ||
| Cancer type | ||||||||||
| Gastrointestinal | 61 | 2.00 (1.00–3.00) | 6.00 (4.00–7.00) | 3.00 (1.00–5.00) | 2.00 (1.00–3.00) | 2.00 (1.00–4.00) | 0 (−1.00–1.00) | 3.00 (1.00–4.00) | ||
| Lung | 52 | 2.00 (0–4.00) | 7.00 (4.00–8.00) | 3.00 (2.00–5.00) | 2.00 (0–3.00) | 2.00 (0–4.00) | 0 (0–2.00) | 3.50 (2.00–5.00) | ||
| Breast | 19 | 1.00 (0.50–3.00) | 5.00 (5.00–8.50) | 5.00 (2.50–6.00) | 1.00 (1.00–3.00) | 3.00 (2.00–6.00) | 1.00 (0.50–4.00) | 2.00 (0.50–4.50) | 0.001* | |
| Head and neck | 22 | 3.00 (1.25–4.00) | 7.00 (4.25–8.00) | 3.00 (2.00–4.75) | 2.50 (1.25–3.75) | 3.00 (2.00–5.00) | 0 (0–1.75) | 2.00 (2.00–3.00) | ||
| Hematological malignancy | 9 | 1.00 (0–3.00) | 5.00 (3.00–5.00) | 3.00 (2.00–5.00) | 1.00 (1.00–2.00) | 1.00 (1.00–2.00) | 1.00 (0–1.00) | 3.00 (2.00–4.00) | 0.007* | |
| Other | 30 | 2.00 (0–3.00) | 7.00 (5.00–8.00) | 4.00 (2.25–6.75) | 2.00 (0–3.00) | 3.00 (2.00–4.00) | 1.00 (0–2.75) | 3.00 (1.00–5.00) | ||
| Body movements that require prophylactic rescue | ||||||||||
| Meals | 62 | 2.00 (0–4.00) | 7.00 (5.00–7.75) | 3.00 (2.00–5.00) | 2.00 (0.25–3.75) | 3.00 (1.00–5.00) | 1.00 (0–2.75) | 3.00 (2.00–4.00) | ||
| Bathing | 16 | 1.50 (0–3.00) | 4.50 (2.00–6.00) | 2.00 (1.75–3.25) | 1.50 (0.75–2.25) | 1.00 (0.75–2.00) | 0 (−1.00–0) | 2.50 (1.00–4.00) | 0.002* | |
| Rehabilitation | 19 | 2.00 (0–3.00) | 5.00 (4.00–7.00) | 3.00 (2.50–5.00) | 2.00 (0–3.00) | 2.00 (1.00–3.00) | 1.00 (0–1.50) | 3.00 (2.00–5.00) | ||
| Radiation therapy | 74 | 2.00 (1.00–3.00) | 7.00 (4.00–8.00) | 4.00 (2.00–6.00) | 2.00 (1.00–3.00) | 2.00 (1.00–4.00) | 0 (−1.00–0.75) | 3.00 (1.25–5.00) | ||
| Other | 22 | 2.50 (1.00–4.00) | 5.50 (4.00–8.00) | 2.50 (2.00–4.75) | 1.00 (1.00–2.00) | 2.50 (1.00–4.75) | 0 (−0.75–2.00) | 2.00 (0.25–3.75) | ||
| Time from administration of prophylactic rescue to movement | ||||||||||
| 5–15 minutes | 31 | 2.00 (1.00–3.00) | 5.00 (2.50–6.50) | 2.00 (1.00–3.50) | 1.00 (0.50–2.00) | 2.00 (0.50–4.00) | 0 (0–2.00) | 1.00 (0–3.00) | ||
| 16–30 minutes | 130 | 2.00 (0–3.00) | 6.00 (4.00–8.00) | 3.50 (2.00–5.00) | 2.00 (0.25–3.00) | 2.00 (1.00–4.75) | 0 (−1.00–2.00) | 3.00 (2.00–5.00) | ||
| 31–60 minutes | 28 | 2.00 (1.00–4.00) | 7.00 (5.00–8.00) | 4.50 (2.00–6.00) | 2.00 (1.00–3.00) | 2.50 (2.00–3.25) | 1.00 (−0.25–2.00) | 4.00 (1.00–5.00) | ||
| Prophylactic rescue opioid ingredient | ||||||||||
| Morphine | 57 | 2.00 (0–4.00) | 7.00 (5.00–8.00) | 3.00 (2.00–5.00) | 2.00 (0–4.00) | 2.00 (1.00–5.00) | 0 (0–2.00) | 3.00 (1.00–4.00) | ||
| Oxycodone | 120 | 2.00 (1.00–3.00) | 6.00 (4.00–8.00) | 3.00 (2.00–5.00) | 2.00 (1.00–3.00) | 2.00 (1.00–5.00) | 0 (0–2.00) | 2.00 (1.00–4.00) | ||
| Hydromorphone | 14 | 1.00 (0.25–1.75) | 5.00 (4.25–7.75) | 4.50 (3.25–6.00) | 1.50 (1.00–3.00) | 1.00 (1.00–3.50) | 0 (−0.75–0.75) | 4.00 (2.00–6.75) | 0.001* | |
| Fentanyl | 2 | 3.00 (2.50–3.50) | 6.00 (5.50–6.50) | 3.00 (3.00–3.00) | 2.50 (2.25–2.75) | 1.00 (0.50–1.50) | −1.50 (−1.75–−1.25) | 4.50 (4.25–4.75) | 0.180* | |
*Wilcoxon signed-rank test; Since there are only two cases in the fentanyl subgroup, no meaningful clinical interpretation should be drawn. NRS, Numerical Rating Scale.
The impact on body movements requiring prophylactic rescue was as follows: 166 patients (86.0%) responded that it was effective, 21 patients (10.9%) responded that there was no change (ineffective), four patients (2.1%) responded that it worsened, and two patients (1.0%) responded that it was unknown.
As shown in Table 3, the incidence of somnolence increased significantly after
prophylactic rescue (4.7%
| Adverse event | Before prophylactic rescue n (%) | After prophylactic rescue n (%) | p |
| Somnolence (All/Gr1/Gr2) | 9/6/3 (4.7/3.1/1.6) | 33/29/4 (17.1/15.0/2.1) | |
| Nausea (All/Gr1/Gr2) | 20/14/6 (10.3/7.3/3.1) | 28/19/9 (14.5/9.8/4.7) | 0.280* |
| Delirium (All/Gr1/Gr2) | 5/4/1 (2.6/2.1/0.5) | 5/4/1 (2.6/2.1/0.5) | 1.000* |
| Constipation (All/Gr1/Gr2/Gr3) | 88/46/41/1 (45.6/23.8/21.2/0.5) | 95/42/52/1/0 (49.2/21.8/26.9/0.5) | 0.541* |
| Dizziness (All/Gr1) | 0/0 (0/0) | 1/1 (0.5/0.5) | 1.000* |
| Restlessness (All/Gr1) | 0/0 (0/0) | 1/1 (0.5/0.5) | 1.000* |
| Muscle weakness lower limb (All/Gr1) | 0/0 (0/0) | 1/1 (0.5/0.5) | 1.000* |
| Urinary retention (All/Gr unknown) | 0/0 (0/0) | 1/1 (0.5/0.5) | 1.000* |
| Pruritus (All/Gr unknown) | 0/0 (0/0) | 1/1 (0.5/0.5) | 1.000* |
*Fisher’s exact test.
Our results showed that prophylactic rescue significantly suppressed breakthrough pain caused by body movements. On the other hand, the incidence of somnolence increased after prophylactic rescue. The potential risks of falls and functional impairment associated with somnolence are points to be considered, especially in older or frail patients. The effectiveness of analgesics has been previously reported as a minimal clinically important difference, defined as a reduction in pain of NRS: 1 or more [13, 14]. In the overall analysis, a reduction of 3.00 was observed, and the subgroup analysis of body movements that require prophylactic rescue showed a difference of at least 2.00, which effectively suppressed the increase in pain. Therefore, prophylactic rescue is applicable for breakthrough pain caused by a wide range of body movements. The subgroup analysis of cancer type also suggested that prophylactic rescue is effective for a wide range of cancer pain. The subgroup analysis of time from administration suggested that while prophylactic rescue within 5–15 minutes was effective, prophylactic rescue within 16–30 minutes or 31–60 minutes was more effective. The reason no significant difference in the efficacy of fentanyl was observed is that only two cases were included, resulting in insufficient statistical power. Furthermore, although hydromorphone demonstrated high efficacy, the number of cases was considerably smaller compared to morphine and oxycodone, potentially introducing bias into the results. Further studies with larger sample sizes are needed for these opioid ingredients.
The ratio of prophylactic rescue to daily opioids was approximately 1/4 in the calculation of the median value. Even in regular pain management, 1/4–1/8 is commonly used in clinical practice [15], suggesting that the same rescue dose should be sufficient for prophylactic rescue. Additionally, the impact on body movements requiring prophylactic rescue showed a high efficacy rate of 86.0%, indicating that appropriate prophylactic rescue can improve the quality of life in addition to pain control.
Only the rate of grade 1 to 2 somnolence significantly increased after prophylactic rescue administration. This is because some side effects of opioids, such as constipation or nausea, occur even at doses lower than those required for analgesia [16, 17]. If an adequate amount of opioids for an analgesic effect had been administered, constipation and nausea would have already occurred. Therefore, even if the total dose of opioids was increased due to prophylactic rescue, there was no increase in these side effects. The ED50 values for analgesic and sedative effects were very similar [18]; therefore, it is thought that the sedative effect was confirmed as somnolence due to little prophylactic rescue. In addition, the overall prophylactic rescue and baseline morphine equivalent doses were small, and the proportion of patients with hepatic and renal dysfunction was also low; therefore, it can be predicted that no serious adverse events occurred. However, when prophylactic rescue is applied in clinical practice, somnolence and the associated risk of falls and functional impairment should be explained to patients when providing medication guidance. In a previous study on the safety of prophylactic rescue [11], the incidence of adverse events of Gr2 or higher was reported to be higher than that in the present study, with 20.6% for somnolence and 22.1% for nausea. A possible reason for this difference may be the period of investigation for adverse events; while this study evaluated adverse events within 1 week of performing prophylactic rescue, previous studies examined the entire period in which prophylactic rescue was used, which may have led to a higher incidence rate. Among the concerns regarding prophylactic rescue, it encourages the use of disorders, dependence, and chemical coping strategies [8, 19]. However, none of these were observed in this study, likely because of appropriate patient selection for prophylactic rescue. Furthermore, in Japan, there is a high level of awareness among patients and healthcare professionals regarding the dangers of opioids, which may explain the low levels of both opioid consumption and disorders [20, 21]. The decision to administer prophylactic rescue should be carefully considered for each patient, considering the opioid addiction situation in each country.
One of the major strengths of this study is its design as a multicenter, collaborative research project involving 12 institutions across Japan. While facilities were slightly more concentrated in the center of Japan, this multicenter approach enabled the inclusion of diverse patient populations, thereby enhancing the external validity and generalizability of the findings. Data aggregated from multiple sites helped to reduce institutional variability, providing robust and reliable evidence of the efficacy and safety of prophylactic rescue therapy. Additionally, the nationwide scope of the study allowed for the consideration of regional differences and variations in therapeutic practices, ensuring that the results reflected real-world clinical settings. Consequently, this study contributes valuable findings regarding the appropriate use and effects of prophylactic rescue therapy, with multiple implications for clinical practice.
One of the limitations of this study is that it was a retrospective survey; therefore, the application of prophylactic rescue was left to the discretion of the attending physicians. There is a possibility of bias because many patients with a high probability of efficacy and a low incidence of adverse events were included in the analysis based on their clinical judgment. Second, this study only analyzed cases in which NRS evaluations before and after prophylactic rescue were sufficiently recorded. As shown in Fig. 1, several cases were excluded. Third, the efficacy and safety of electronic medical records depend on the skills of the attending physician, nurse, and pharmacist who implement and record them (e.g., frequency of implementation, years of employment), which may affect the results. Particularly regarding opioid use disorder, while the relevant terms were not recorded within the electronic medical records in this study, changes in the number and frequency of rescue doses were not implemented, meaning such cases may not have been detected. Long-term studies focusing specifically on opioid use and psychological effects are required going forward. Finally, because there is no standardized method for evaluating the impact of body movements requiring prophylactic rescue, we used a three-level evaluation: effective/no change (ineffective)/worsening. In this survey, the case collectors at each facility conducted composite assessment of whether the body movements were more actively from electronic medical record entries; therefore, there is a possibility of significant bias. Furthermore, as this study is not a placebo-controlled trial, it is difficult to exclude the possibility that the observed improvement in pain may be partly attributable to the use of rescue medication itself, rather than the timing of its administration. Future prospective randomized placebo-controlled trials are needed to eliminate these biases. However, this study is significant because it is the first to demonstrate, using real-world data from Japan, that prophylactic rescue is an effective method.
This study showed that prophylactic opioid rescue with appropriate patient selection can suppress breakthrough pain caused by body movements.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; Cr, serum creatinine; NRS, numerical rating scale; SD, standard deviation.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
RyT collected and analyzed the data and edited the manuscript. ReT designed this original concept and wrote the manuscript. EK and SH collected the data and edited the manuscript. MU performed the data validation and edited the manuscript. HT designed this original concept, performed the data validation and edited the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki. The present study complied with the “ethical guidance for a study in medicine-targeted humans” and was approved by the Institutional Review Board of Shizuoka Cancer Center (approval number: T2023-42-2024-1-3). Information about study inclusion criteria was posted on the hospital’s bulletin, and consent was obtained via the opt-out method.
We would like to thank the following pharmacists for their cooperation in collecting the cases for this study: Shinya Kobayashi (Kondo Hospital), Takuma Matsumoto, Yoshika Takechi, Naoko Mukai, Tsubasa Wada, Akari Beniya, Koki Kimura (Shikoku Cancer Center), Ayuko Kusakabe (Otaru General Hospital), Yoshiaki Takashina (Seirei Mikatahara General Hospital), Hiroki Sugiyama (Shizuoka City Shimizu Hospital), Saori Takahashi (Tokyo Nishi Tokushukai Hospital), Makoto Akao (Yonezawa City Hospital), Ai Suzuki (JR Tokyo General Hospital), and Daisuke Ebitani (Kamagaya Branch, Ebitani Pharmacy).
This study was supported by the Japanese Society for Pharmaceutical Palliative Care and Science.
The authors declare no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

