1 Department of Ultrasound, Beijing Tiantan Hospital, Capital Medical University, 100070 Beijing, China
2 Department of Ultrasound, The Affiliated Hospital of Southwest Medical University, 646000 Luzhou, Sichuan, China
Abstract
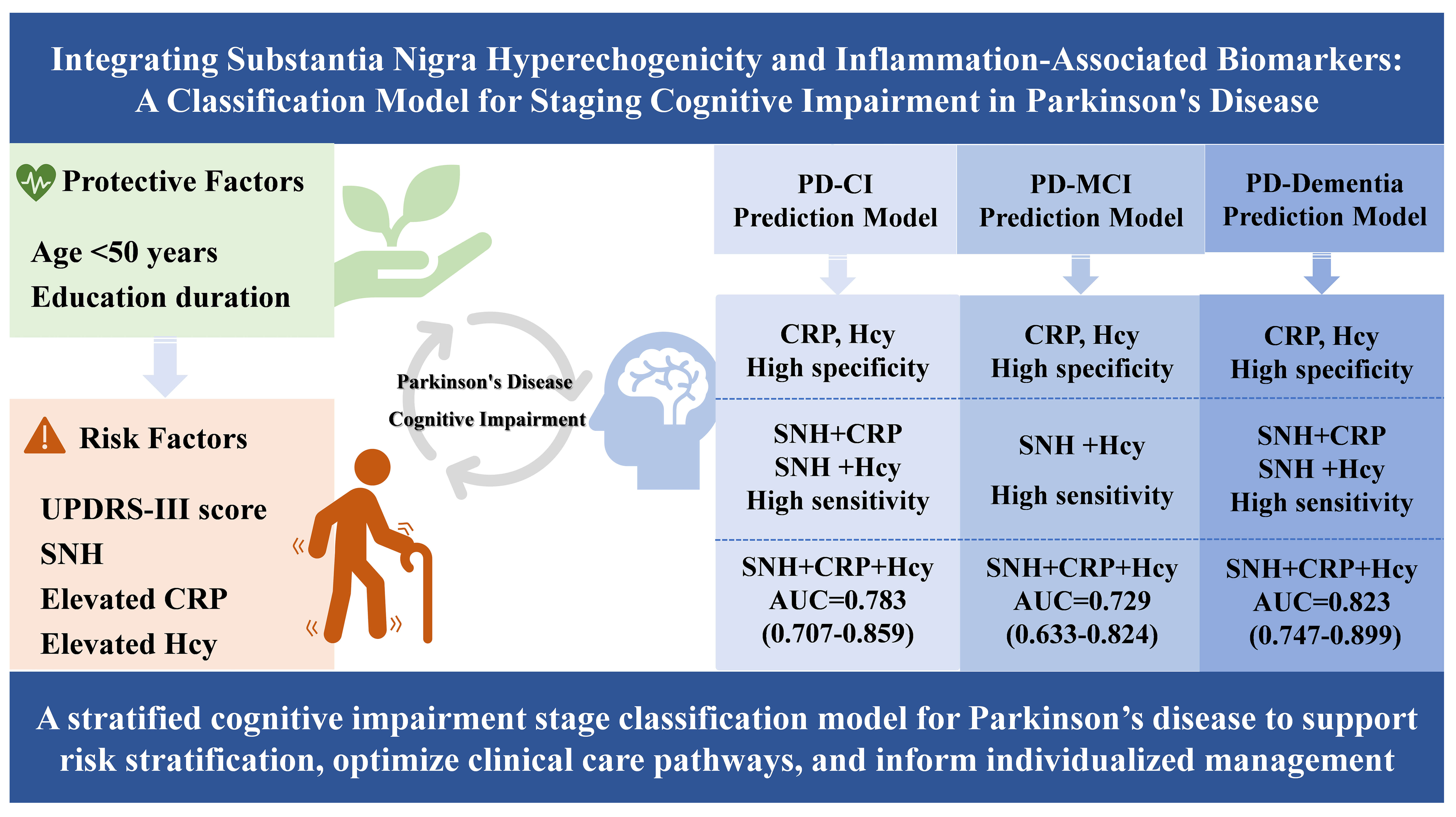
Cognitive impairment (CI) is recognized as a debilitating complication of Parkinson’s disease (PD). This study was designed to develop a diagnostic classification model by integrating substantia nigra hyperechogenicity (SNH) and inflammationassociated biomarkers to evaluate its diagnostic performance in distinguishing PD CI stages.
Between January, 2023 and May, 2024, 184 patients with PD who underwent transcranial sonography were prospectively enrolled. Based on Montreal Cognitive Assessment (MoCA) scores, participants were categorized into three groups: cognitive impairment (PD-CI, MoCA <26), mild cognitive impairment (PD-MCI, MoCA 22–25), and dementia (PD-dementia, MoCA ≤21). Ultrasound features and inflammationassociated biomarkers were screened with univariate analyses. Multivariate logistic regression was used to identify independent diagnostic factors, and receiver operating characteristic (ROC) curve analysis was used to assess model discrimination.
Multivariate regression analysis indicated that age <50 years and more years of education were significantly associated factors for CI (OR = 0.170, p = 0.0350; OR = 0.8780, p = 0.0020, respectively), whereas Unified Parkinson’s Disease Rating Scale Part III (UPDRSIII) score (OR = 1.024, p = 0.0270), SNH (OR = 2.550, p = 0.0030), elevated C-reactive protein (CRP) (OR = 2.038, p = 0.0350), and elevated homocysteine (Hcy) (OR = 2.830, p = 0.0020) were independent risk factors. The area uinder the curves (AUCs) for the combined SNH+CRP+Hcy model in predicting PD-CI, PD-MCI, and PD-dementia were 0.783, 0.729, and 0.823, respectively; these values were significantly superior to those for single or dual marker combinations (p < 0.05), with the strongest performance for distinguishing PD-dementia.
An SNH and inflammationassociated biomarkerbased model was developed for predicting the stage of cognitive impairment in PD. Clinical targets for individualized intervention can be provided, and clinical risk stratification and care pathways can be optimized. Furthermore, the model supports the iron deposition-neuroinflammation-CI pathway hypothesis, providing a mechanistic rationale for ultrasoundbased PD-CI diagnosis.
Graphical Abstract
Keywords
- Parkinson’s disease
- cognitive impairment
- substantia nigra
- ultrasonography
- inflammation
Parkinson’s disease (PD) is recognized as the most prevalent movement disorder worldwide and as the second most common neurodegenerative disease after Alzheimer’s disease (AD) [1, 2]. Beyond the cardinal motor symptoms (bradykinesia, rigidity, resting tremor), PD is characterized by heterogeneous non-motor sequelae, and cognitive impairment (CI) shows a sixfold higher prevalence than in age-matched controls [2]. CI substantially diminishes functional independence and quality-of-life measures in patients with PD, imposing socioeconomic burdens that exceed those of motor disability even during prodromal phases [3, 4]. Accordingly, early diagnostic and identification of CI are essential for PD management.
Neuroinflammation is recognized as a core pathological mechanism in PD. It affects not only the substantia nigra (SN) and striatum but also cognition-related regions such as the frontal lobe and hippocampus, thereby contributing to CI [5, 6, 7]. This process is amplified by peripheral metabolic disturbances, in which homocysteine (Hcy) accumulation—mediated by deficiencies of the cofactors folate (FA) and vitamin B12 (VB12)—induces excitotoxicity via N-methyl-D-aspartate (NMDA) receptor overactivation and impairs DNA repair in cognitive circuits [8, 9, 10]. Concurrently, iron dyshomeostasis, reflected by elevated ferritin (Fer), potentiates microglial activation and lipid peroxidation, thereby exacerbating neuronal vulnerability through ferroptotic pathways. The resulting oxidative milieu depletes endogenous antioxidants such as uric acid (UA), further diminishing neuroprotection and accelerating cognitive decline [11, 12, 13]. Systemic inflammation is evidenced by elevated C-reactive protein (CRP), which correlates with blood-brain barrier disruption and microglial priming [14], whereas neuron-specific enolase (NSE) serves as a surrogate marker of neuronal damage in cortico-limbic networks [15, 16]. These peripheral biomarkers appear mechanistically interlinked: elevations in Hcy (due to FA/VB12 deficiency) synergize with iron overload (high Fer) to amplify mitochondrial reactive oxygen species (ROS) generation, which in turn depletes UA and upregulates CRP, creating a pathological feed-forward loop that drives neuroinflammation and CI. Therefore, neuroinflammation-related peripheral biomarkers may help in the diagnosis of CI in PD.
Transcranial sonography (TCS) has emerged as a noninvasive neuroimaging tool, and substantia nigra hyperechogenicity (SNH) has been validated as an imaging biomarker of nigrostriatal pathology in PD [17]. SNH can be detected in prodromal PD and persists throughout disease progression. Although current TCS research predominantly examines associations between SNH and measures of PD duration or severity, its utility for predicting cognitive impairment has been underexplored [18, 19].
Given the neuroinflammatory heterogeneity underlying CI in PD and the diagnostic advantages of TCS-identified high echogenicity in the SN, SNH, and multisource inflammation markers were integrated to identify individuals at increased risk of early, rapid cognitive decline. This approach is intended to enhance diagnostic stratification and to support the implementation of precision-prevention protocols.
Data on patients with PD treated in the Movement Disorders Department who underwent TCS between January 2023 and May 2024 were prospectively collected. The diagnosis of PD was made according to the International Parkinson’s and Movement Disorder Society criteria [20]. Antiparkinsonian medications were withheld for at least 12 hours before clinical assessment. Exclusion criteria included atypical Parkinsonian syndromes, secondary Parkinsonism, other non-Parkinsonian disorders, incomplete clinical records, an inadequate temporal bone window for TCS, indeterminate ultrasound findings, or a history of brain surgery or traumatic brain injury.
Neurological evaluations were performed by two board-certified neurologists,
each with more than five years of experience in movement disorders, using the
Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS).
In instances of discordant evaluations, the final diagnosis was adjudicated by a
senior specialist with more than ten years of experience in movement disorders.
Global cognitive function was assessed with the Montreal Cognitive Assessment
(MoCA) [21], following guidelines from the International Parkinson and Movement
Disorder Society; total scores range from 0 to 30, with values
Hyperechoic areas of the SN were identified and measured by two experienced sonographers, each with more than ten years of neuroimaging experience, who were blinded to the patients’ clinical diagnoses. A Canon Aplio i900 system (Canon Medical Systems Corporation, Ōta, Tokyo, Japan) equipped with a 2.0–3.0 MHz phased-array transducer (I6SX1) was used. Standardized parameters included a scan depth of 14–16 cm and a dynamic range of 45–55 dB, with real-time adjustments to gain compensation and brightness tailored to individual anatomy.
With the patient in the supine position and the temporal acoustic window fully exposed, the phased-array transducer was placed over the temporal region. Systematic midbrain exploration was performed by incrementally angling the ultrasound beam along the orbitomeatal line (connecting the lateral canthus and external auditory meatus). Once the characteristic butterfly-shaped midbrain morphology adjacent to the midline was identified, peripheral and internal nuclei were assessed when bone-window penetration permitted adequate visualization. SNH was documented when present in the ipsilateral or contralateral SN. SN echogenicity was graded according to the standardized Bartova classification [24], and interrater reliability for SNH area quantification and grading was assessed using intraclass correlation coefficients (ICCs).
Comprehensive baseline clinical data were collected for all participants,
including sex, age, disease duration, years of formal education, and disease
severity assessed by the modified Hoehn and Yahr (H-Y) stage. Motor symptom
severity was evaluated using the Movement Disorder Society-Unified Parkinson’s
Disease Rating Scale Part III (UPDRS-III), and global cognitive function was
screened with the MoCA. Comorbidity status was documented for hypertension,
hyperlipidemia, and diabetes mellitus. Anthropometric data included body mass
index (BMI), along with self-reported smoking status and alcohol consumption
patterns. Age was categorized into
Serum biomarkers related to neuroinflammation and metabolic pathways were
quantified from venous blood collected under standardized fasting conditions
using validated immunoassay techniques. The panel included FA, VB12, Fer, CRP, NSE, UA, and Hcy. For analysis, each biomarker was
dichotomized into normal or abnormal based on established clinical reference
ranges. Normal ranges were defined as: FA
Statistical analyses were conducted using SPSS v21.0 (IBM Corp., Armonk, NY,
USA). Continuous variables were expressed as mean
Missing data management: Variables with
Between January 2023 and May 2024, 647 patients were enrolled in this study.
Following screening, 463 patients were excluded: Parkinsonism (n = 230),
non-Parkinsonism (n = 139), ambiguous SN findings on
TCS examination (n = 90), history of Traumatic Brain
Injury (n = 1), and history of deep brain stimulation (DBS) surgery (n = 3); a
detailed flowchart of the patient selection process is presented in Fig. 1.
Consequently, 184 patients met the inclusion criteria and were analyzed in this
study. Among the 184 patients analyzed, 16 had
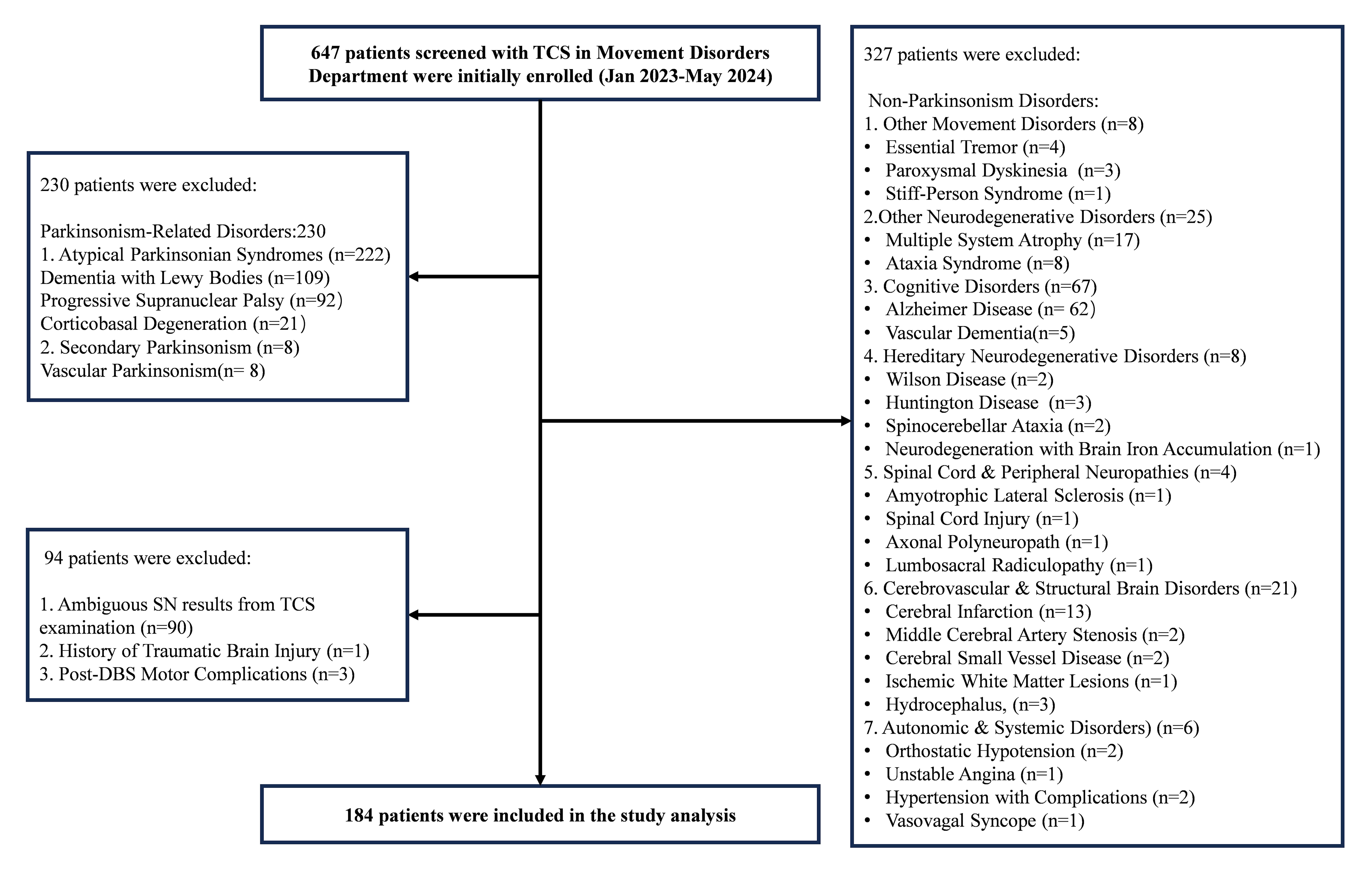 Fig. 1.
Fig. 1.
Study flow chart. DBS, deep brain stimulation; SN, substantia nigra; TCS, transcranial sonography.
Based on MoCA scores, the participants were stratified as follows: 42 subjects
demonstrated normal cognitive function (MoCA score
| Variables | PD-NC (n = 42) | PD-MCI (n = 60) | PD-dementia (n = 82) | p | ||
| Age | 11.916 | 0.064 | ||||
| 6 | 5 | 2 | ||||
| 50–69 | 24 | 47 | 56 | |||
| 70–74 | 9 | 5 | 16 | |||
| 3 | 3 | 8 | ||||
| Sex (male) | 0.390 | 0.823 | ||||
| Male | 27 | 41 | 52 | |||
| Female | 15 | 19 | 30 | |||
| BMI (kg/m2) | 24.88 |
24.15 |
24.54 |
6.815 | 0.575 | |
| Duration of disease (years) | 5.82 |
6.94 |
6.68 |
2.084 | 0.353 | |
| Education level (years) | 11.95 |
11.60 |
9.17 |
16.684 | ||
| UPDRS-III | 32.31 |
38.10 |
41.87 |
5.687 | 0.004 | |
| H-Y stage | 1.95 |
1.98 |
2.32 |
9.230 | 0.010 | |
| Smoking | 0.277 | 0.871 | ||||
| Yes | 9 | 15 | 21 | |||
| No | 33 | 45 | 61 | |||
| Drinking alcohol | 2.599 | 0.273 | ||||
| Yes | 9 | 21 | 21 | |||
| No | 33 | 39 | 61 | |||
| Hypertension | 4.585 | 0.101 | ||||
| Yes | 17 | 15 | 34 | |||
| No | 25 | 45 | 48 | |||
| Dyslipidemia | 0.167 | 0.920 | ||||
| Yes | 6 | 10 | 14 | |||
| No | 36 | 50 | 68 | |||
| Diabetes mellitus | 0.292 | 0.864 | ||||
| Yes | 14 | 17 | 25 | |||
| No | 28 | 43 | 57 | |||
H-Y stage, Hoehn and Yahr stage; PD, Parkinson’s disease; PD-dementia, PD patients with dementia; PD-MCI, PD patients with mild cognitive impairment; PD-NC, PD patients with normal cognitive function; UPDRS-III, Unified Parkinson’s Disease Rating Scale Part III; BMI, body mass index.
TCS assessment of the SN revealed statistically significant findings (p
= 0.0020). Among inflammatory-associated biomarkers, significant differences
were observed for CRP and Hcy (p = 0.0001 and p = 0.0030,
respectively). In contrast, no statistically significant differences were found
for FA, VB12, Fer, NSE, or UA (all p
| Variables | PD-NC (n = 42) | PD-MCI (n = 60) | PD-dementia (n = 82) | p | ||
| SNH | 13.001 | 0.002 | ||||
| Present | 14 | 31 | 55 | |||
| Absent | 28 | 29 | 27 | |||
| FA | 4.935 | 0.085 | ||||
| Decreased | 9 | 24 | 33 | |||
| Normal | 33 | 36 | 49 | |||
| VB12 | 3.763 | 0.439 | ||||
| Increased | 4 | 5 | 7 | |||
| Decreased | 2 | 9 | 6 | |||
| Normal | 36 | 46 | 69 | |||
| Fer | 2.097 | 0.351 | ||||
| Increased | 0 | 2 | 4 | |||
| Normal | 42 | 58 | 78 | |||
| CRP | 15.375 | |||||
| Increased | 5 | 22 | 39 | |||
| Normal | 37 | 38 | 43 | |||
| NSE | 4.239 | 0.120 | ||||
| Increased | 4 | 9 | 4 | |||
| Normal | 38 | 51 | 78 | |||
| UA | 1.688 | 0.430 | ||||
| Increased | 4 | 2 | 5 | |||
| Normal | 38 | 58 | 77 | |||
| Hcy | 11.790 | 0.003 | ||||
| Increased | 7 | 26 | 39 | |||
| Normal | 35 | 34 | 43 | |||
CRP, C-reactive protein; FA, folic acid; Fer, ferritin; Hcy, homocysteine; NSE, neuron-specific enolase; UA, uric acid; VB12, vitamin B12.
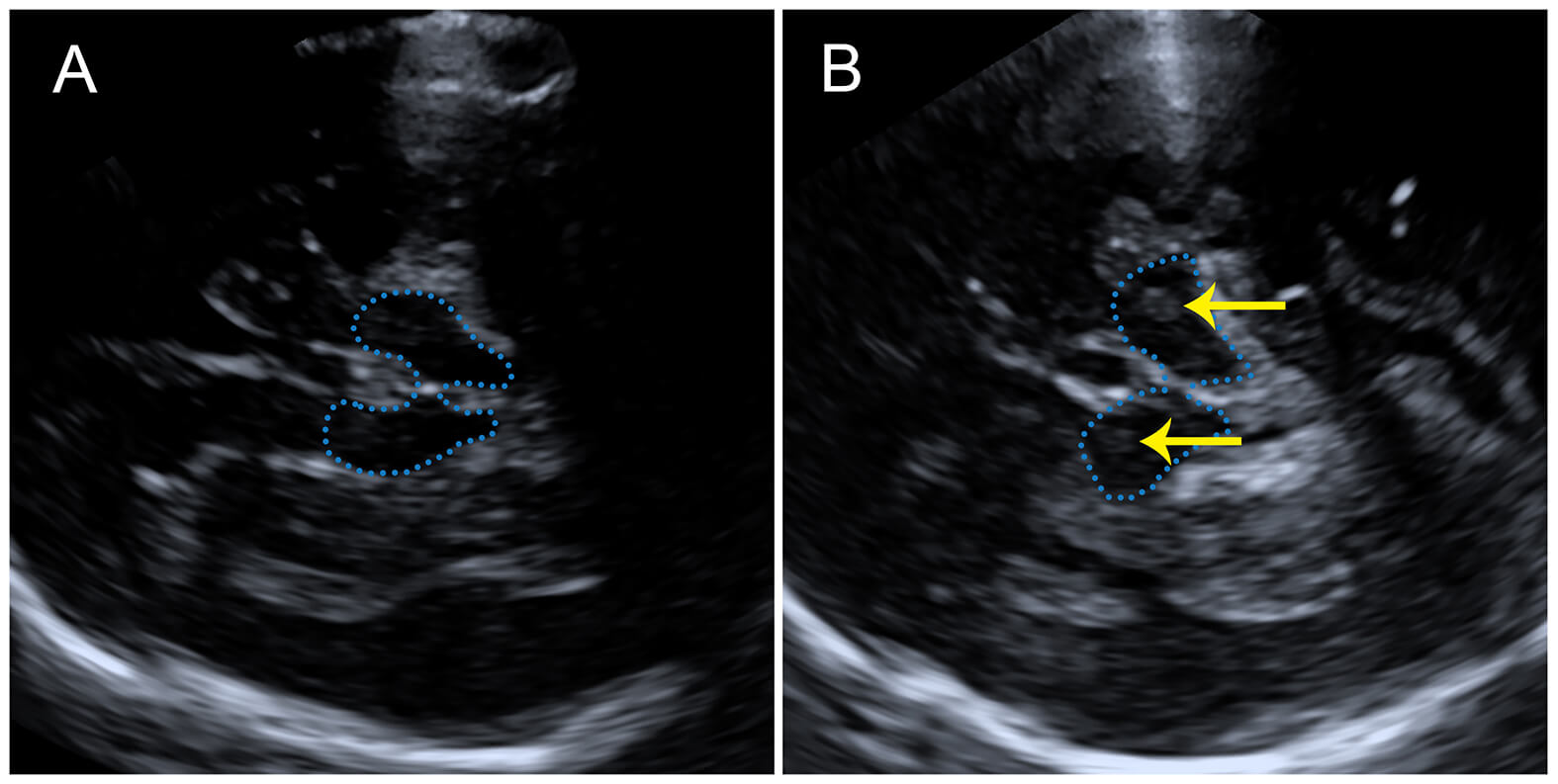 Fig. 2.
Fig. 2.
Transcranial ultrasonography showing butterfly-shaped midbrain and internal sonographic features. (A) No significant substantia nigra hyperechogenicity (SNH) is detected within the midbrain. (B) Focal SNH is visualized within the midbrain (yellow arrow). The blue dashed line outlines the midbrain region.
Variables exhibiting a significant association (p
Participants younger than 50 years exhibited an 83.0% reduced risk of CI
compared to those aged
| Variables | B | SE | Wald test | OR (95% CI) | p | |
| Age (years) | ||||||
| –1.775 | 0.840 | 4.469 | 0.170 (0.033–0.879) | 0.035 | ||
| 50–69 | –0.442 | 0.604 | 0.537 | 0.642 (0.197–2.099) | 0.464 | |
| 70–74 | –0.864 | 0.687 | 1.581 | 0.422 (0.110–1.621) | 0.209 | |
| - | - | - | - | - | ||
| Education level (years) | –0.130 | 0.043 | 9.299 | 0.878 (0.808–0.955) | 0.002 | |
| H-Y stage | –0.027 | 0.200 | 0.018 | 0.973 (0.658–1.441) | 0.893 | |
| UPDRS-III | 0.024 | 0.011 | 4.921 | 1.024 (1.003–1.046) | 0.027 | |
| SNH | 0.936 | 0.314 | 8.885 | 2.550 (1.378–4.721) | 0.003 | |
| CRP | 0.712 | 0.337 | 4.460 | 2.038 (1.052–3.945) | 0.035 | |
| Hcy | 1.040 | 0.335 | 9.660 | 2.830 (1.469–5.453) | 0.002 | |
| FA | 0.268 | 0.341 | 0.620 | 1.308 (0.671– 2.550) | 0.431 | |
B, regression coefficient; SE, standard error.
Elevated CRP independently predicted CI (OR = 2.038, p = 0.0350), conferring a 2.04 times increased risk. Hcy elevation was associated with a highly significant risk (OR = 2.830, p = 0.0020), indicating a 2.83 times higher CI risk. Further, SNH was an independent risk factor for CI, with positive cases having a 2.55-fold higher risk than negative cases (OR = 2.550, p = 0.0030) (Table 3).
3.3.2.1 PD-NC (MoCA score
To evaluate the ability of the three independent predictors to identify PD-CI,
we performed ROC curve analysis. The AUC for the single
predictors SNH, CRP, and Hcy was 0.636, 0.655, and 0.646, respectively. Pairwise
comparison analysis revealed no statistically significant differences in AUC
values among these individual biomarkers (all p
| Variables | Sensitivity | Specificity | AUC (95% CI) | †p | ‡p | §p |
| SNH | 60.56 | 66.67 | 0.636 (0.553–0.719) | - | - | - |
| CRP | 42.96 | 88.10 | 0.655 (0.591–0.719) | 0.728 | - | - |
| Hcy | 45.77 | 83.33 | 0.646 (0.575–0.716) | 0.862 | 0.840 | - |
| SNH+CRP | 76.76 | 57.14 | 0.727 (0.651–0.803) | 0.018 | 0.004 | - |
| SNH+Hcy | 80.28 | 57.14 | 0.724 (0.640–0.807) | 0.028 | - | 0.001 |
| CRP+Hcy | 69.01 | 73.81 | 0.736 (0.662–0.810) | - | 0.010 | |
| SNH+CRP+Hcy | 69.01 | 73.81 | 0.783 (0.707–0.859) | 0.001 |
†p, p-value obtained by comparing SNH with row headers; ‡p, p-value obtained by comparing CRP with row headers; §p, p-value obtained by comparing Hcy with row headers.
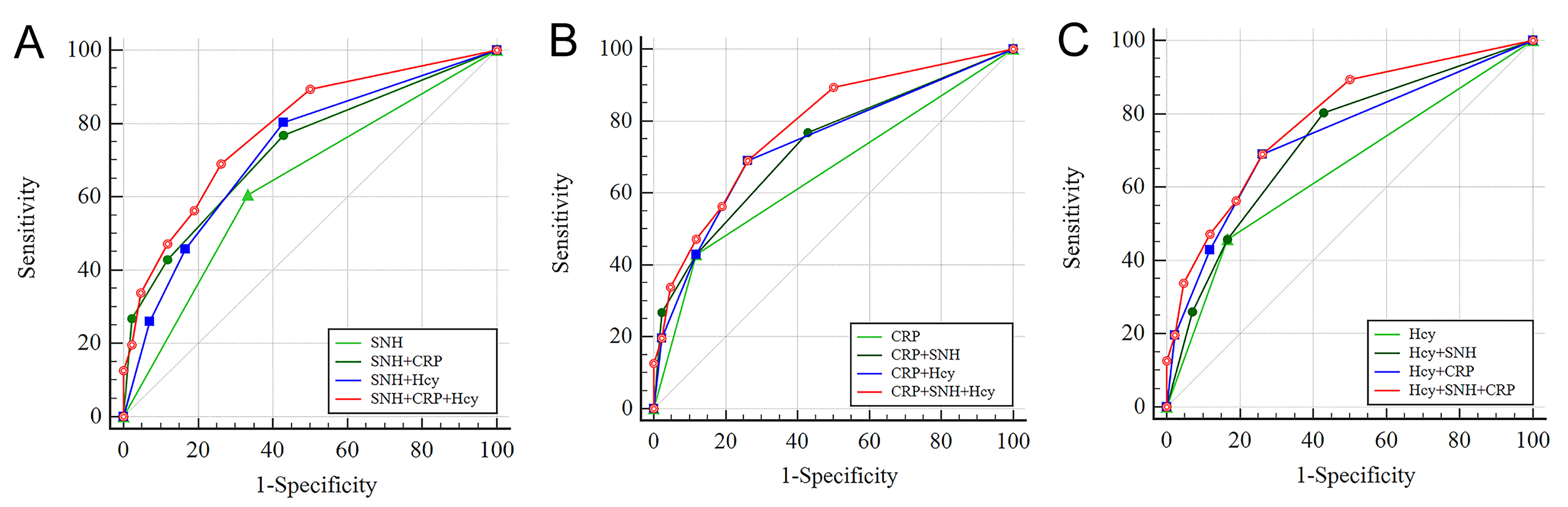 Fig. 3.
Fig. 3.
Receiver operating characteristic curve of models predicting the CI of PD. (A) SNH-related diagnostic classification model. (B) CRP-related diagnostic classification model. (C) Hcy-related diagnostic classification model.
Given the limitations of single predictors, we explored the diagnostic performance of different indicator combinations. SNH+CRP model and SNH+Hcy model resulted in increased sensitivity 76.76%, 80.28%, respectively) compared to any single-indicator model, with AUCs also improving to an upper-moderate level (0.727, 0.724, respectively). Conversely, the CRP+Hcy model exhibited a different trend, achieving an AUC of 0.736, a sensitivity of 69.01%, and a specificity increased to 73.81%. The comprehensive model combining all three indicators (SNH+CRP+Hcy) showed the highest AUC among the models evaluated for PD-CI. This model’s AUC improved significantly to 0.783, surpassing the performance of all single- and dual-indicator models. Compared to the dual-marker models, its specificity was significantly enhanced, reaching 73.81%. The lower limit of the model’s 95% confidence interval (CI: 0.707–0.859) was above the chance level (0.5), and the narrow 95%CI suggests stable estimates, indicating good discriminatory ability for distinguishing PD-CI from PD patients with normal cognitive function (PD-NC) (Table 4, Fig. 3).
3.3.2.2 PD-NC (MoCA score
The diagnostic performance of the indicators was evaluated using ROC curve
analysis. The AUC values for the individual indicators SNH, CRP, and Hcy were
0.592, 0.624, and 0.633, respectively. Pairwise comparisons indicated no
statistically significant differences in the AUCs of these indicators (all
p
| Variables | Sensitivity | Specificity | AUC (95% CI) | †p | ‡p | §p |
| SNH | 51.67 | 66.67 | 0.592 (0.495–0.688) | - | - | - |
| CRP | 36.67 | 88.10 | 0.624 (0.545–0.703) | 0.611 | - | - |
| Hcy | 43.33 | 83.33 | 0.633 (0.548–0.718) | 0.526 | 0.867 | - |
| SNH+CRP | 36.67 | 57.14 | 0.666 (0.569–0.764) | 0.089 | 0.185 | - |
| SNH+Hcy | 75.00 | 57.14 | 0.692 (0.593–0.791) | 0.037 | - | 0.047 |
| CRP+Hcy | 61.67 | 73.81 | 0.695 (0.604–0.787) | - | 0.014 | 0.123 |
| SNH+CRP+Hcy | 61.67 | 73.81 | 0.729 (0.633–0.824) | 0.011 | 0.005 | 0.019 |
†p, p-value obtained by comparing SNH with row headers; ‡p, p-value obtained by comparing CRP with row headers; §p, p-value obtained by comparing Hcy with row headers.
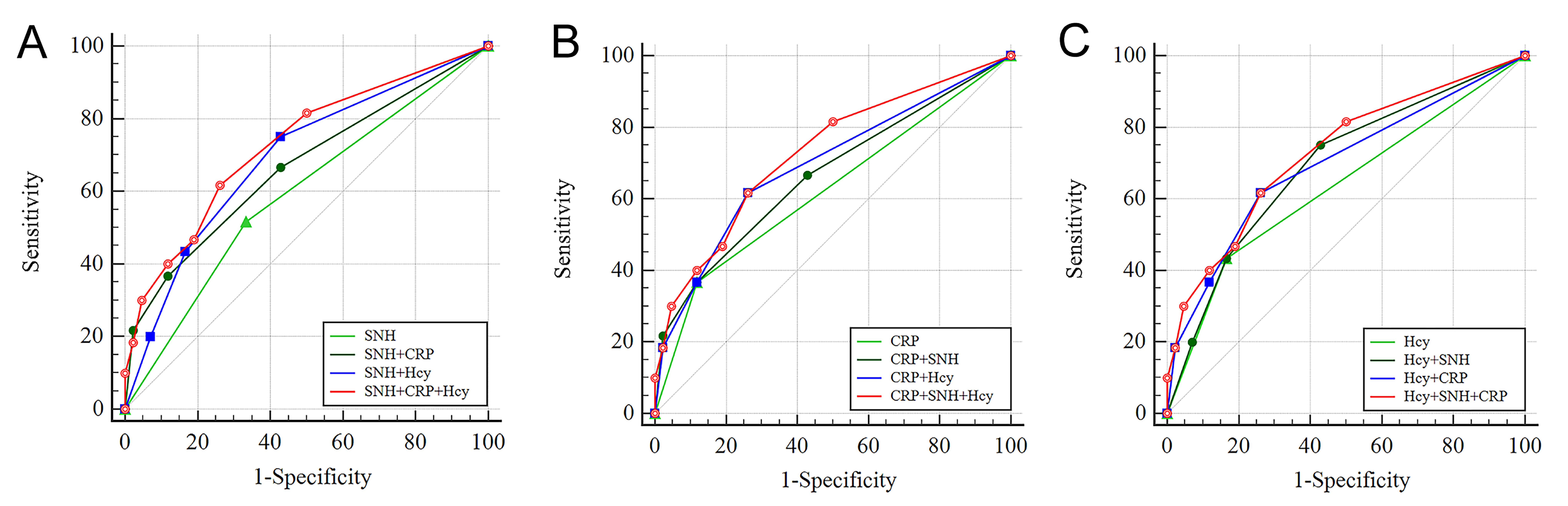 Fig. 4.
Fig. 4.
Receiver operating characteristic curve of models predicting the MCI of PD. (A) SNH-related diagnostic classification model. (B) CRP-related diagnostic classification model. (C) Hcy-related diagnostic classification model.
Combination models significantly enhanced diagnostic performance compared to single-indicator models. Among dual-indicator models, the SNH+Hcy model yielded the highest sensitivity (75.00%), albeit with lower specificity (57.14%). In contrast, the CRP+Hcy model maintained relatively high specificity (73.81%) while providing a more balanced sensitivity (61.67%), achieving the higher AUC among dual-indicator models (0.695). Notably, the integrated SNH+CRP+Hcy model demonstrated superior overall diagnostic performance (AUC = 0.729), exceeding those of single-indicator and several dual-indicator models. It achieved balanced performance with a sensitivity of 61.67% and a specificity of 73.81%. The lower limit of its 95% confidence interval (0.633) was notably above the chance level (0.5), further validating the better diagnostic performance of this SNH+CRP+Hcy model for differentiating between PD-NC and PD-MCI (Table 5, Fig. 4).
3.3.2.3 PD-NC (MoCA
ROC curve analysis revealed AUC values for the single indicators SNH, CRP, and Hcy of 0.669, 0.678, and 0.654, respectively, with no statistically significant differences between them. Among the single-indicator tests, SNH demonstrated the highest sensitivity (67.07%), while CRP and Hcy exhibited superior specificity (88.10% and 83.33%, respectively) (Table 6, Fig. 5).
| Variables | Sensitivity | Specificity | AUC (95% CI) | †p | ‡p | §p |
| SNH | 67.07 | 66.67 | 0.669 (0.580–0.757) | - | - | - |
| CRP | 47.56 | 88.10 | 0.678 (0.605–0.752) | 0.875 | - | - |
| Hcy | 47.56 | 83.33 | 0.654 (0.576–0.733) | 0.811 | 0.669 | - |
| SNH+CRP | 84.15 | 57.14 | 0.771 (0.691–0.851) | 0.019 | - | |
| SNH+Hcy | 84.15 | 57.14 | 0.747 (0.659–0.835) | 0.069 | - | |
| CRP+Hcy | 74.39 | 73.81 | 0.766 (0.685–0.846) | - | 0.006 | |
| SNH+CRP+Hcy | 74.39 | 73.81 | 0.823 (0.747–0.899) | 0.002 |
†p, p-value obtained by comparing SNH with row headers; ‡p, p-value obtained by comparing CRP with row headers; §p, p-value obtained by comparing Hcy with row headers.
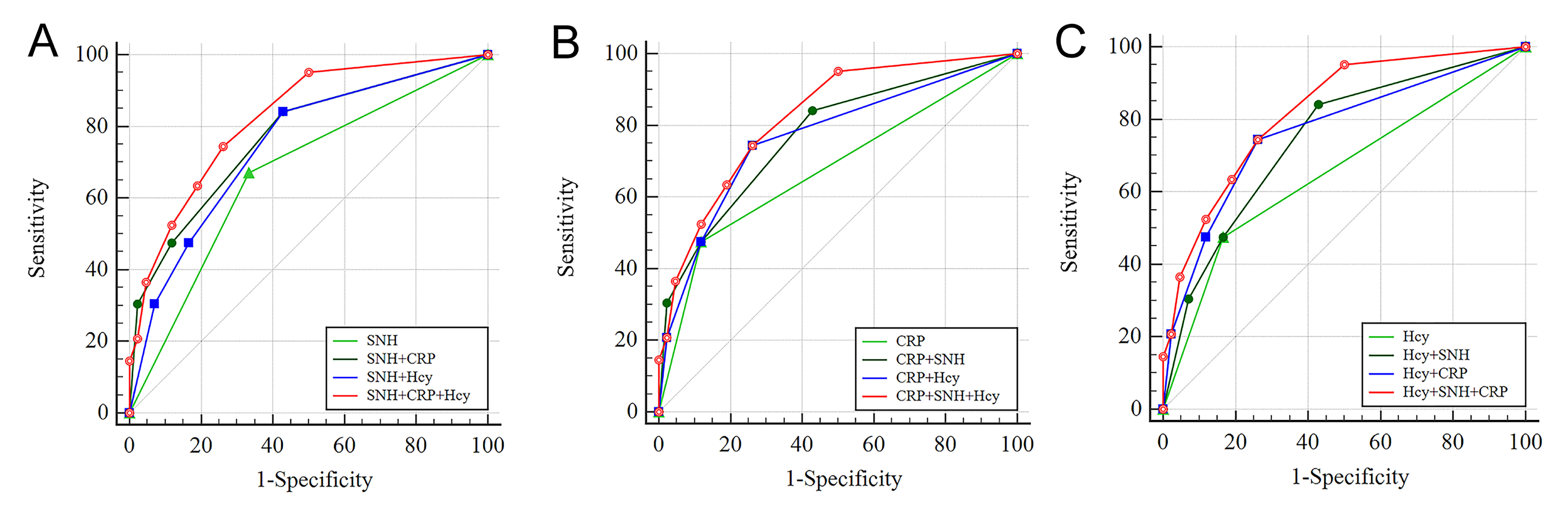 Fig. 5.
Fig. 5.
Receiver operating characteristic curve of models predicting the dementia of PD. (A) SNH-related diagnostic classification model. (B) CRP-related diagnostic classification model. (C) Hcy-related diagnostic classification model.
Notably, the SNH+CRP model and the SNH+Hcy model achieved substantially higher sensitivity (84.15% for both), though with reduced specificity (57.14% for both). The AUCs for these dual-indicator models were significantly higher than those of the single-indicator model (SNH+CRP: AUC = 0.771; SNH+Hcy: AUC = 0.747). The CRP+Hcy model maintained relatively high specificity (73.81%) while providing reasonable sensitivity (74.39%), yielding an AUC of 0.766 (Table 6, Fig. 5).
Most importantly, the SNH+CRP+Hcy model demonstrated the best diagnostic efficacy. Its AUC increased significantly to 0.823 while maintaining sensitivity of 74.39% and specificity of 73.81%, demonstrating a balance of performance characteristics and solid discriminatory power. The lower limit of this model’s 95% confidence interval (CI: 0.747–0.899) substantially exceeded the chance level (0.5), further confirming its reliable diagnostic performance for distinguishing PD-NC from PD-dementia (Table 6, Fig. 5).
This study developed a diagnostic classification model by combining SNH with serum inflammation-associated biomarkers CRP and Hcy, significantly improving the diagnostic performance of PD-CI risk stratification. This diagnostic classification model not only provides clinical targets for individualized intervention but also optimizes CI risk stratification and clinical pathways. Additionally, this study provides some research support for the mechanism gap in ultrasound technology for PD-CI diagnosis, as SNH-reflected substantia nigra iron deposition and abnormal inflammatory markers may support the hypothesis of the “iron deposition-neuroinflammation-CI” circuit.
This study indicates that patients
Each additional year of formal education was associated with a 12.2% reduction in the odds of PD-CI. These results are consistent with a prior study indicating that higher educational attainment is associated with better cognitive function and reduced dementia risk, even in the presence of neurodegenerative disease [27]. Educational attainment influences cognitive trajectories, with higher levels associated with better working memory and subcortical task performance, greater compensatory capacity of the frontostriatal circuit, enhanced dopaminergic efficiency, and greater cognitive reserve [26]. In the Proceedings of the National Academy of Sciences, Schwarzacher and colleagues showed that repetitive neural activity induced by learning expands synapses on dendritic spines, strengthening neuronal connections. This synaptic plasticity helps stabilize neuronal activity and supports cognitive function [28].
Additionally, each one-point increase in the UPDRS-III score corresponded to a 2.4% increase in the odds of PD-CI. From an effect-size perspective, the impact may be modest at low UPDRS-III scores, but cumulative effects at higher scores can meaningfully increase the odds of PD-CI (e.g., ~48% higher odds for a 20-point increase from 30 to 50). These cumulative effects have clear implications for clinical intervention. Furthermore, as the gold standard for assessing motor symptoms, an increase in UPDRS-III scores reflects worsening degeneration of the cortical-basal ganglia-thalamic circuit (CBTC) [29]. The observed association between PD-CI and UPDRS-III deepens understanding of the scale’s diagnostic performance, as score changes mark motor deterioration and may also indicate cognitive decline via motor-cognitive neural circuits [30].
SNH serves as an imaging marker for SN iron deposition, with a sensitivity of 60.56% and specificity of 66.67%, consistent with a previous TCS study [15]. The high specificity of CRP and Hcy (88.10% and 83.33%, respectively) further supports the significant role of neuroinflammation in PD-CI [31, 32, 33]. Additionally, the SNH+CRP+Hcy diagnostic classification model demonstrated improved AUCs (PD-CI: 0.783; PD-MCI: 0.729; PD-dementia: 0.823), with the most significant gain observed for PD-dementia. This pattern may reflect synergistic effects of multiple pathway biomarkers amplified by widespread corticolimbic degeneration [34]. This mechanism aligns with proteomics findings of abnormal plasma H3C15 (histone-associated neuroinflammation) and PSAP (lysosomal dysfunction) in PD dementia patients, suggesting that multimodal biomarkers reflect interplay between neurodegenerative and inflammatory pathways [35].
Notably, SNH, CRP, and Hcy were independent risk factors for PD-CI, with a
gradient in effect sizes: SNH-positive cases had 2.55-fold higher odds (OR =
2.550), elevated CRP conferred 2.04-fold higher odds (OR = 2.038), and elevated
Hcy had the strongest association (OR = 2.830; 2.83-fold higher odds). These
factors may collectively participate in an “iron
deposition-neuroinflammation-CI” circuit: SNH reflects iron deposition in the
SN, which activates microglia and promotes release of inflammatory mediators
(e.g., IL-1
Quantitative susceptibility mapping (QSM) on MRI provides supporting evidence for links between iron deposition and CI-related circuits. Voxel-based QSM study has shown that, relative to PD with normal cognition, PD-MCI exhibits higher susceptibility in the cuneus, precuneus, caudate head, fusiform gyrus, and orbitofrontal cortex, and susceptibility in these regions is inversely associated with MoCA scores, indicating structural and functional links between iron burden and cognitive deficits [39]. A subsequent study has linked QSM-derived iron metrics to the severity and progression of cognitive impairment in PD, suggesting a pathological connection between iron dyshomeostasis and dysfunction of corticostriatal networks [40]. These observations align with the present framework: SNH primarily reflects nigral iron deposition and glial activation, providing a PD-specific anchor for peripheral inflammation-related signals, whereas QSM quantifies iron burden in key extranigral circuits (including cortical and striatal regions), complementing ultrasound by addressing limitations in spatial resolution and quantification for mechanistic interpretation.
The core clinical insight of this study is the development of a CI-oriented diagnostic framework that integrates SNH measured by transcranial sonography with the inflammatory biomarkers CRP and Hcy, providing a practical avenue for early screening and more precise intervention in PD. In PD patients at lower risk of CI, the SNH+CRP/Hcy combination achieves a sensitivity of 76.76%–80.28% in the PD-CI group. When all three markers are combined, the AUC increases to 0.783, enabling efficient preliminary screening. For high-risk PD populations (such as those with RBD or olfactory dysfunction), the SNH+CRP/Hcy combination achieves a sensitivity of 84.15% in the PD-dementia group. When all three are combined, the AUC reaches 0.823, maximizing the identification of early cognitive decline signals. In specialized clinical settings, the SNH+CRP+Hcy model demonstrates balanced performance, supporting the accurate identification of PD-dementia. Because transcranial sonography is simple, safe, and cost-effective, combining it with readily available blood markers yields a screening protocol suitable for primary care and may reduce costs compared with strategies relying on cerebrospinal fluid testing or advanced imaging. This panel is positioned as a complement to the Montreal Cognitive Assessment, allowing integration into standardized PD assessment pathways and enabling prioritization of neuropsychological testing and shorter follow-up intervals when MoCA results are normal or borderline but clinical concern persists. This approach may help address limitations of single neuropsychological assessments and enable earlier identification of cognitive impairment in PD, thereby guiding clinical decisions.
This study has limitations. First, follow-up data were not available. However, as a cross-sectional study, the research focused on evaluating the diagnostic performance of the SNH and inflammation-associated biomarker model rather than disease-progression trajectories. Multivariable logistic regression controlled for time-related variables (e.g., disease duration and age), and the model maintained significant diagnostic power across CI stages, supporting the robustness of the conclusions. Future work will employ longitudinal cohorts, combining MoCA subscale scores with dynamic biomarker changes to construct a temporal risk model for cognitive decline. Second, cognitive status was defined using MoCA cutoffs for pragmatic, reproducible multicenter evaluation of model discrimination, rather than diagnostic confirmation by comprehensive neuropsychological assessment. Because MoCA may be influenced by education, we interpret education-related findings as screening-based associations rather than a causal “protective” effect. Third, the sample size was modest, increasing the risk of overfitting. Nevertheless, credibility is supported by a parsimonious, clinically prespecified set of covariates. Effect directions and magnitudes were consistent across cognitive strata. Evidence converged across independent modalities, including SNH and inflammation-related biomarkers. AUC estimates and their confidence intervals were clearly above chance. Findings support the model’s potential to guide early risk stratification and clinical decision-making, while underscoring the need for confirmation in larger prospectively validated cohorts and external validation studies. Future studies will expand sample size through multicenter collaboration to enhance generalizability.
In conclusion, a diagnostic classification model based on ultrasound-derived SNH and the inflammation-associated biomarkers CRP and Hcy was proposed, establishing a framework for stratified diagnosis of CI stage in PD. This framework may provide clinical targets for individualized intervention and help optimize PD risk stratification and clinical pathways.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
WH and WZ conceived and designed the study, provided study materials/patients, and provided administrative support. JZ conceived and designed the study and provided study materials/patients. JZ, YL, SW and LX collected and assembled the data. JZ and CH analyzed and interpreted the data. All authors drafted the manuscript or revised it critically for important intellectual content. All authors read and approved the final version to be published. All authors participated sufficiently in the work and agreed to be accountable for all aspects of the work, ensuring that any questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The study protocol was approved by the Institutional Board of Beijing Tiantan Hospital, Capital Medical University (approval No. KY2022-015-04). Informed consent was obtained from all participants. All procedures involving human participants were conducted in accordance with the ethical standards of the institutional research committee and the Declaration of Helsinki (1964) and its later amendments or comparable ethical standards.
Not applicable.
This work was supported by the National Natural Science Foundation of China (No. 82271995 and 81730050).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN46585.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





