1. Introduction
Axons are the elongated portion of the neuron that transport various molecules
between the soma and axon terminals and transmit signals between neurons or
between neurons and effector cells by forming synapses [1]. Axonogenesis,
referring to the formation and outgrowth of axons during neuronal development and
nerve regeneration, is essential for the establishment and maintenance of
neuronal polarity [2]. Progressive axon degeneration is a dynamic process
resulting in the loss of communication between neurons, which has been recognized
as a common pathological feature of neurodegenerative diseases, such as
Alzheimer’s disease and Parkinson’s disease [3]. A better understanding of the
physiological mechanisms of axonogenesis may facilitate the development of
therapeutic strategies to block axon degeneration in these diseases.
Cdc42 is a member of the Rho GTPase family that plays important roles in
multiple cellular processes, such as cytoskeletal organization, cell cycle
progression, vesicle transport, and cell survival [4]. Cdc42 is widely expressed
in the cell body and neurites of neurons, participating in the establishment of
neuronal polarity and the formation of axons, dendrites, and dendritic spines
[5]. Previous evidence has shown that knockout of cdc42 inhibited axonogenesis of
primary murine hippocampal neurons [6]. Collapsin response mediator protein-2
(CRMP-2) is a cytosolic protein that acts as a key regulator of axonogenesis [7].
Overexpression of full-length CRMP-2 was shown to promote axon elongation and
induced the formation of multiple axons, while transfection of CRMP-2 dominant
negative mutants inhibited axonogenesis [8]. CRMP-2 can bind directly to the
tubulin heterodimers, promoting microtubule assembly and axon growth [9].
Moreover, we previously demonstrated that cdc42 promotes axonogenesis of rat
hippocampal neurons by reducing CRMP-2 phosphorylation at Thr514 and stabilizing
microtubules [10].
Glycogen synthase kinase-3 (GSK-3) is a serine/threonine
kinase originally identified as a key regulator of glucose metabolism. Increasing
evidence has suggested that GSK-3 is highly implicated in neuronal
development and neurodegeneration [11]. Phosphorylation of GSK-3 has
been found to inhibit phosphorylation of CRMP-2 at Thr514 and promote
axonogenesis [12]. Our previous findings revealed that cdc42 promoted CRMP-2
dephosphorylation in rat hippocampal neurons by enhancing GSK-3
phosphorylation [10]. However, whether GSK-3 is involved in
cdc42-mediated axonogenesis remains uncertain.
In this study, primary rat hippocampal neurons were used to investigate whether
cdc42 promotes axonogenesis by inhibiting GSK-3 activity. Specifically,
we sought to determine the effects of increased GSK3 activity and
GSK3 inhibition on axonogenesis.
2. Materials and Methods
2.1 Isolation of Rat Hippocampal Neurons
The use of animals was approved by the Animal Care and Use Committee of the
Southern Medical University and all protocols were carried out in accordance with
the NIH Guidelines for the Care and Use of Laboratory Animals. Primary
hippocampal neurons were isolated from neonatal Sprague-Dawley rats as previously
described [13]. Briefly, rats (male:female = 1:1) on postnatal day 1 were
obtained from the Experimental Animal Center of Southern Medical University. The pups were euthanized by decapitation and the head was separated from the
body. The intact brain was immediately removed and quickly placed into dissection
medium. The hippocampus was carefully separated under a microscope and placed in
pre-chilled D-Hank’s solution (8 g NaCl, 0.4 g KCl, 0.06 g KHPO, 0.35
g NaHCO, 0.12 g NaHPO•12HO in 1 L ddHO;
Guangdong Guanghua Sci-Tech Co., Ltd., Guangzhou, China). Subsequently, the
hippocampus was cut into small pieces and digested with 0.25% trypsin-EDTA (1:5
v/v; Gibco, Grand Island, NY, USA) at 37 C for 10 min. DMEM/F12 medium
(Gibco) containing 10% fetal bovine serum (FBS; Gibco) and 10% newborn calf
serum (NBCS; Gibco) were added to stop digestion. Cells were centrifuged at
80 g for 5 min at room temperature. The supernatant was discarded, and
cell pellets were resuspended in Neurobasal-A medium (Gibco, Grand Island, NY,
USA) supplemented with 2% B27 (Gibco), 1% 100 GlutaMAX-I (Gibco), 25
M Glutamate (Genview, League City, TX, USA), and 0.5 g/L Plasmocin
(InvivoGen, San Diego, CA, USA). All cells were cultured in a 5% CO
incubator at 37 C.
2.2 Cell Transfection
Primary rat hippocampal neurons were transfected with designated plasmid vectors
using the Amaxa™ Rat Neuron Nucleofector Kit (Cat. # VPG-1003,
Lonza, Basel, Switzerland) as previously reported [14]. Plasmid vectors carrying
a constitutively active cdc42 mutant (pIRES-EGFP-cdc42F28L) were gifts from Prof.
Andreas Püschel (Westfälische Wilhelms-Universität Münster,
Germany). Plasmid vectors carrying a dominant negative cdc42 mutant
(pIRES-EGFP-cdc42N17) were gifts from Prof. Lin Zhang (Southern Medical
University, China). Plasmid vectors carrying a wild-type GSK-3
(pcDNA3.1-GSK-3 wild type) or a constitutively active GSK3
mutant (pcDNA3.1-GSK-3 S9A) were gifts from Prof. James R. Woodgett
(University of Toronto, Canada). These plasmids were validated in our previous
study [10]. Cells transfected with empty vectors (pIRES-EGFP or pcDNA3.1) were
used as the controls. The culture medium was replaced with Neurobasal-A medium
containing 2% B27, 1% 100 GlutaMAX-I, 25 M Glutamate, and 0.5
g/L Plasmocin at 4 and 24 h after transfection.
2.3 Drug Treatment
At 24-h post-transfection, rat hippocampal neurons were treated with or without
phosphate-buffered saline (PBS), 2 mmol/L NaCl (diluted in cultured medium), or 2
mmol/L LiCl (diluted in cultured medium) for an additional 48 h.
2.4 Measurement of GSK-3 Activity
GSK-3 activity in primary rat hippocampal neurons following different
treatments was measured using the GENMED kit (Genmed Scientifics Inc.,
Wilmington, DE, USA) as reported previously [15]. In brief, cells were harvested
and centrifuged at 16,000 g for 5 min at 4 C. The supernatant
was collected, and the protein concentration was determined using the BCA assay
(Pierce, USA). Protein samples in triplicates (10 g each) were added to a
96-well plate and incubated with the reagents from the kit. Optical density was
measured at 340 nm using the SpectraMax 190 microplate reader (Molecular Devices,
San Jose,CA, USA). GSK-3 activity in the control group (transfected with
empty pIRES-EGFP vectors) was set as 1.
2.5 Immunofluorescence
After treatment, rat hippocampal neurons were washed with PBS, fixed in 4%
paraformaldehyde (Sigma-Aldrich, St. Louis, MO, USA) for 90 min, and then
permeabilized in 1% Triton X-100 (Sigma-Aldrich) for 30 min. After blocking with
3% bovine serum albumin (Genview, League City, TX, USA) in Tris-buffered saline
(Sigma-Aldrich) at room temperature for 1 h, neurons were probed with anti-Tau-1
antibody (1:200 dilution; cat no. MAB3420, Merck Millipore, Billerica, MA, USA)
and anti-microtubule-associated protein 2 (MAP2; 1:1000 dilution; cat no. AB5622,
Merck Millipore, USA) at 4 C overnight. Subsequently, cells were
incubated with rhodamine Red™-X goat anti-mouse IgG (1:700
dilution; Invitrogen, USA) and goat polyclonal secondary antibody to rabbit
IgG-H&L (Cy5), pre-adsorbed (1:1000 dilution; cat no. ab6564, Abcam, New
Temtories, HK, China) for 2 h. The nuclei were stained with Hoechst 33258 (0.5
g/mL in PBS, Sigma-Aldrich) for 3 min at room temperature. The fluorescent
signal was observed under a confocal microscope (FV500; Olympus, Japan).
Processes with Tau-1 immunoreactivity in their distal segments were counted as
axons. The longest axon of individual neurons in each group (100 GFP-positive
neurons per group) was traced to determine axon length. The number of axons per
cell and the percentages of neurons with no axon, a single axon, or multiple
axons in individual groups were calculated. The longest axon of each neuron was
used to determine the axon length using the Image-Proplus software (Version 6.0;
Media Cybernetics, Silver Springs, MD, USA). One hundred GFP-positive neurons per
group were analyzed.
2.6 Statistical Analysis
Data were analyzed using the SPSS software (Version 19.0; IBM, Armonk, NY, USA)
and presented as mean standard deviation (SD) from three independent
experiments. One-way ANOVA followed by Tukey’s test was used to compare data
among groups. A p-value of less than 0.05 was defined as statistically
significant.
3. Results
3.1 Transfection with GSK-3 wt or GSK-3 S9A
Restored GSK-3 Activity in Rat Hippocampal Neurons Overexpressing
Cdc42F28L
Hippocampal neurons were isolated from neonatal rats and transfected with
vectors expressing cdc42F28L (a constitutively active cdc42 mutant) alone or
together with vectors carrying GSK-3 wt, GSK-3 S9A (a
constitutively active GSK3 mutant), or empty vectors (GSK-3
vector). Cells transfected with empty pIRES-EGFP vectors served as the controls
(the Vector group). After 72 h, GSK-3 activity in different groups of
cells was measured. Overexpression of cdc42F28L significantly decreased
GSK-3 activity in hippocampal neurons compared with the Vector group
(p 0.01). Transfection with vectors carrying either GSK-3
wt or GSK-3 S9A significantly increased GSK-3 activity in
neurons overexpressing cdc42F28L (p 0.01). The delivery of empty
GSK-3 vector did not change GSK-3 activity in
cdc42F28L-overexpressing neurons (Fig. 1). These findings suggest that
transfection of GSK-3 wt or GSK-3 S9A restored GSK-3
activity in hippocampal neurons overexpressing cdc42F28L.
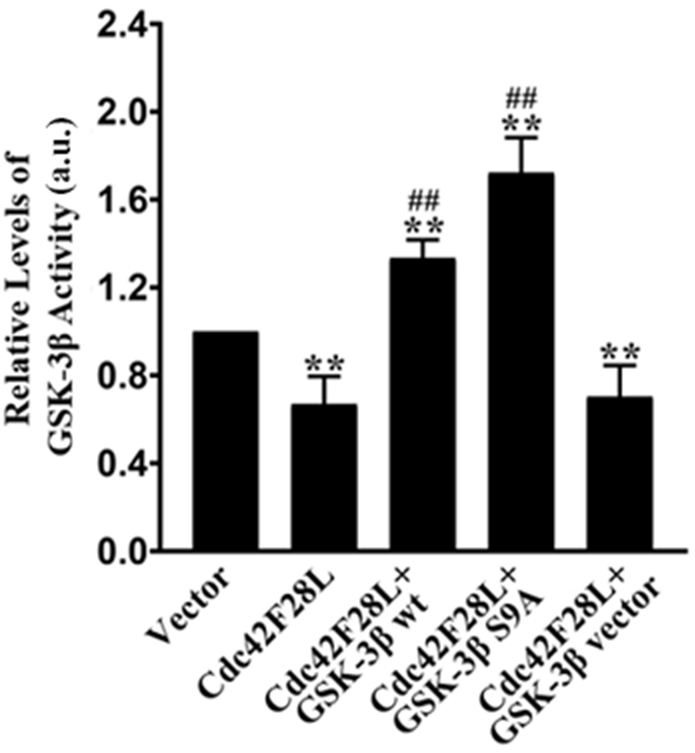 Fig. 1.
Fig. 1.
Effects of GSK-3 wt or GSK-3 S9A on
GSK-3 activity in rat hippocampal neurons overexpressing cdc42F28L.
Primary rat hippocampal neurons were transfected with pIRES-EGFP vectors
expressing cdc42F28L alone or together with pcDNA3.1 vectors carrying
GSK-3 wt, GSK-3 S9A, or empty pcDNA3.1 vectors (GSK-3
vector). Cells transfected with empty pIRES-EGFP vectors served as the controls
(Vector). At 72-h post-transfection, GSK-3 activity was measured. The
result of the Vector group was set as 1. Data are shown as mean SD.
**p 0.01 vs. the Vector group; ##p 0.01 vs. the
Cdc42F28L group. a.u., absorbance unit.
3.2 Transfection with GSK-3 wt or GSK-3 S9A
Attenuated Axonogenesis of Rat Hippocampal Neurons with Excessive Cdc42 Activity
To investigate whether transfection of GSK-3 wt or GSK-3 S9A
could affect axonogenesis in hippocampal neurons with excessive expression of
cdc42F28L, we quantified the number and length of neuronal axons using
immunofluorescence. At 72 h after transfection, different groups of neurons were
stained with Tau-1 for axons, MAP2 for dendrites, and Hoechst 33258 for nuclei.
Cells were then observed under a laser confocal microscope. The distal of the
axons and the proximal segments of the dendrites were positively stained with
Tau-1 (red). The dendrites and the proximal segments of the axons were stained
with MAP2 (purple). As neurons were cultured for only 3 days, the dendrites were
relatively short (Fig. 2A). Compared with the Vector group, neurons
overexpressing cdc42F28L showed significantly lower numbers of neurons with no
axon (14.33% vs. 5.66%; p 0.01) or a single axon (74.55% vs.
51.67%; p 0.01) but had a higher number of neurons with multiple
axons (11.67% vs. 42.67%; p 0.01) (Fig. 2B). The number of axons
per neuron and the mean axon length of the Vector group were significantly lower
than those of the Cdc42F28L group (0.98 vs. 1.58 and 105.47 m vs. 159.80
m, respectively; both p 0.01) (Fig. 2C,D). In neurons
overexpressing cdc42F28L, transfection with vectors carrying GSK-3 wt or
GSK-3 S9A significantly increased the number of neurons with no axon
(21.00% and 45.67%, respectively; p 0.01 vs. the Cdc42F28L group)
but decreased the number of neurons with multiple axons (6.33% and 2.66%,
respectively; p 0.01 vs. the Cdc42F28L group) (Fig. 2B).
GSK-3 wt or GSK-3 S9A also significantly decreased the number
of axons per neuron (0.92 and 0.57, respectively; p 0.01 vs. the
Cdc42F28L group) and the mean axon length (66.88 m and 38.03
m, respectively; p 0.01 vs. the Cdc42F28L group) compared
with the Cdc42F28L group (Fig. 2C,D). There was no significant difference in the
percentage of different subtypes of neurons, the number of axons per cell, or
mean axon length between the Cdc42F28 and Cdc42F28 + GSK-3 vector groups
(p 0.05) (Fig. 2B–D). These results support that GSK-3 wt
or GSK-3 S9A attenuated Cdc42F28L overexpression-induced axonogenesis of
rat hippocampal neurons.
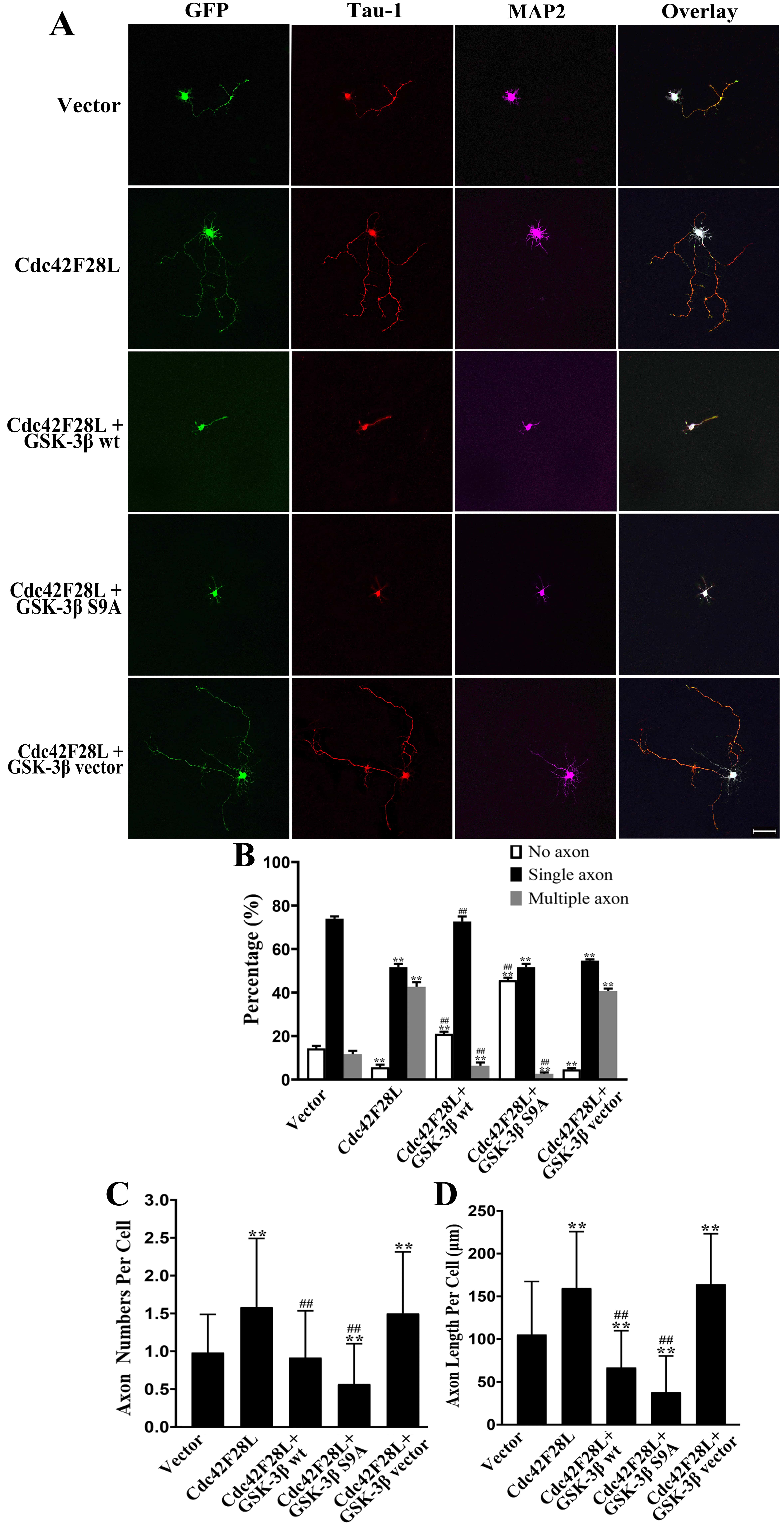 Fig. 2.
Fig. 2.
Effects of GSK-3 wt or GSK-3 S9A on
axonogenesis of rat hippocampal neurons with excessive cdc42 activity. Primary
rat hippocampal neurons were transfected with pIRES-EGFP vectors expressing
cdc42F28L alone or together with pcDNA3.1 vectors carrying GSK-3 wt,
GSK-3 S9A, or empty pcDNA3.1 vectors (GSK-3 vector) at a ratio
of 1:3 [24]. Cells transfected with empty pIRES-EGFP vectors served as the
controls (Vector). (A) Immunofluorescence analysis of neurons. Different groups
of cultured neurons were stained with Tau-1, MAP2, and fluorescent secondary
antibodies, followed by nuclei staining with Hoechst 33258. Images were captured
under a laser confocal microscope. Green, GFP; red, Tau-1; purple, MAP2; blue,
Hoechst. Bar = 50 m. (B) Percentage of neurons with no axon, a single
axon, and multiple axons. (C) Number of axons per cell. (D) Mean axon length.
Data are shown as mean SD. **p 0.01 vs. the Vector group;
##p 0.01 vs. the Cdc42F28L group.
3.3 Treatment with LiCl Decreased GSK-3 Activity in Rat
Hippocampal Neurons Overexpressing Cdc42N17
To determine if cdc42 promoted axonogenesis by inhibiting GSK-3, we
transfected primary rat hippocampal neurons with vectors expressing cdc42N17 (a
dominant negative cdc42 mutant) or empty control vectors, and then treated the
cells with or without PBS, NaCl, or LiCl (a GSK-3 inhibitor) for 48 h.
GSK-3 activity assays showed that transfection with cdc42N17
significantly induced the kinase activity in hippocampal neurons. Treatment with
LiCl, however, significantly decreased GSK-3 activity in neurons
overexpressing cdc42N17 (p 0.01). No significant difference was
observed in the Cdc42N17, Cdc42N17 + PBS, and Cdc42N17 + NaCl groups (p
0.05) (Fig. 3).
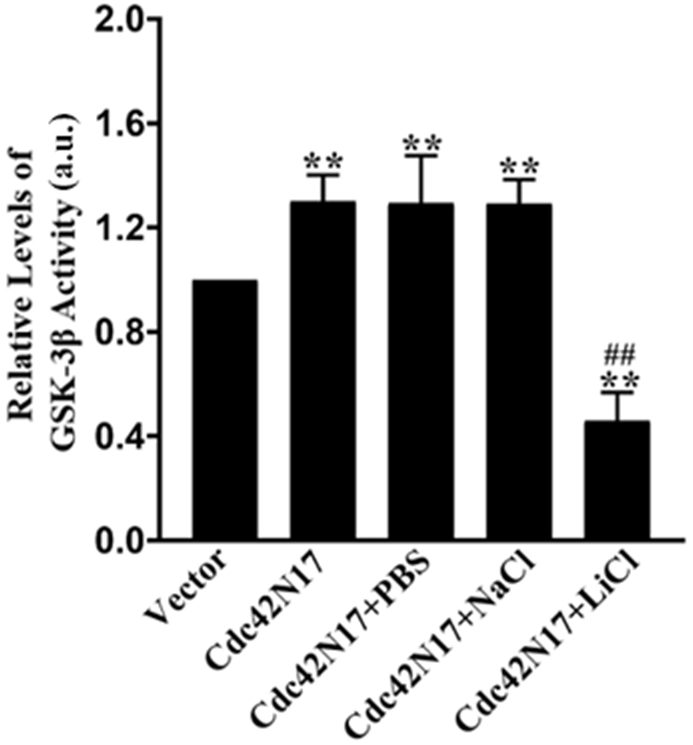 Fig. 3.
Fig. 3.
Effect of LiCl treatment on GSK-3 activity in rat
hippocampal neurons overexpressing cdc42N17. Primary rat hippocampal neurons
were transfected with pIRES-EGFP vectors expressing cdc42N17 or empty pIRES-EGFP
vectors (Vector) for 24 h, followed by treatment with or without PBS, NaCl, or
LiCl for 48 h. GSK-3 activity in different groups of neurons was
measured. Data are shown as mean SD. **p 0.01 vs. the Vector
group; ##p 0.01 vs. the Cdc42N17 group. a.u., absorbance unit.
3.4 Treatment with LiCl Abolished the Inhibitory Effect of Cdc42n17
Overexpression on Axonogenesis
We further analyzed the effects of GSK-3 inhibition on the axonogenesis
of rat hippocampal neurons. After transfection with vectors carrying cdc42N17 or
empty control vectors, neurons were treated with or without PBS, NaCl, or LiCl
for 48 h. Immunofluorescence staining of neurons with Tau-1 (for axons), MAP2
(for dendrites), and Hoechst (for nuclei) is shown in Fig. 4A. Compared with the
Vector group, transfection with cdc42N17 significantly increased the number of
neurons with no axon (14.00% vs. 35.34 %; p 0.01) and decreased
the number of neurons with a single axon (71.00% vs. 60.33%; p
0.01) or multiple axons (15.00% vs. 4.33%; p 0.01) (Fig. 4B). The
number of axons per neuron and the mean axon length of the Cdc42N17 group were
significantly lower compared to the controls (0.68 vs. 1.05 and 40.81
m vs. 113.33 m, respectively; both p 0.01) (Fig. 4C,D). Inhibition of GSK-3 by LiCl treatment effectively reduced the
number of axon-free neurons (2.00%) and neurons with a single axon (46.00%) but
increased the number of neurons with multiple axons (52.00%) compared with the
Cdc42N17 group (all p 0.01) (Fig. 4B). Neurons exposed to LiCl also
exhibited a significantly higher number of axons per cell (1.60) and mean axon
length (147.10 m) compared to the Cdc42N17 group (both p
0.01) (Fig. 4C,D). There was no significant difference in the percentage of
different subtypes of neurons, the number of axons per cell, or mean axon length
between the Cdc42N17, Cdc42N17 + PBS, and Cdc42N17 + NaCl groups (p
0.05) (Fig. 4B–D). The above data demonstrate that inhibition of GSK-3
by LiCl abolished the inhibitory effect of cdc42N17 overexpression on
axonogenesis of rat hippocampal neurons.
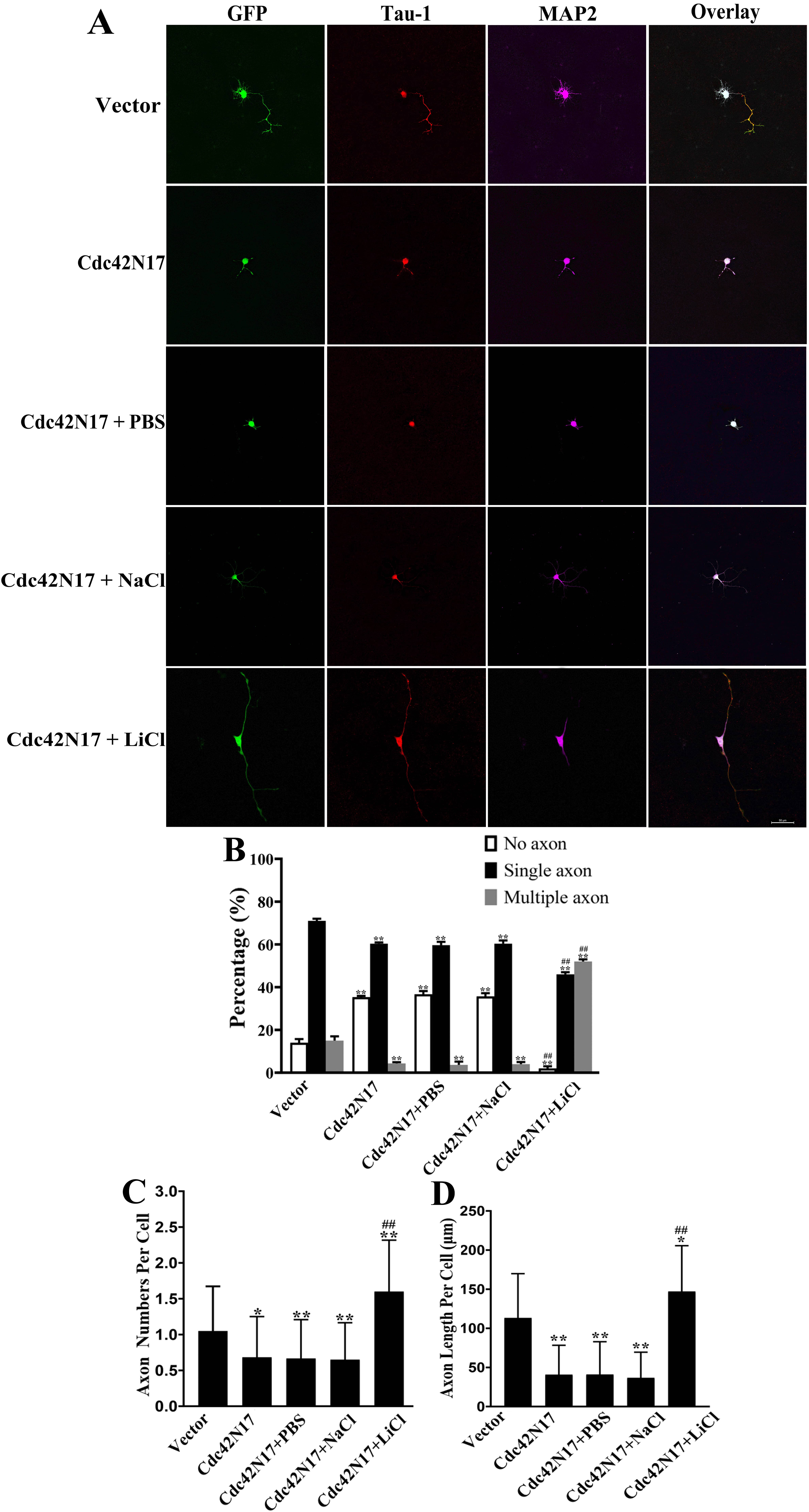 Fig. 4.
Fig. 4.
Effect of LiCl treatment on the inhibitory effect of cdc42N17
overexpression on axonogenesis. Primary rat hippocampal neurons were transfected
with pIRES-EGFP vectors expressing cdc42N17 or empty pIRES-EGFP vectors (Vector)
for 24 h, and then treated with or without PBS, NaCl, or LiCl for 48 h. (A)
Immunofluorescence analysis of neurons. Different groups of cultured neurons were
stained with Tau-1, MAP2, and fluorescent secondary antibodies, followed by
nuclei staining with Hoechst 33258. Images were captured under a laser confocal
microscope. Green, GFP; red, Tau-1; purple, MAP2; blue, Hoechst. Bar = 50
m. (B) Percentage of neurons with no axon, a single axon, and multiple
axons. (C) Number of axons per cell. (D) Mean axon length. Data are shown as mean
SD. *p 0.05 vs. the Vector group;
**p 0.01 vs. the Vector group; ##p 0.01
vs. the Cdc42N17 group.
4. Discussion
Progressive loss of neuronal polarity is a major pathological event in
neurodegenerative diseases [16]. The morphological development and functional
polarization of axons are key steps in the development and maintenance of
neuronal polarity [17]. A better understanding of axonogenesis-related pathways
may contribute to the development of novel therapeutic interventions for
neurodegenerative disorders. Our previous work has shown that cdc42 promoted
axonogenesis of rat hippocampal neurons by enhancing microtubule stabilization
and reducing CRMP-2 phosphorylation [10]. Moreover, dephosphorylation of CRMP-2
by cdc42 was mediated through GSK-3 phosphorylation. In the present
study, we showed that cdc42 promoted axonogenesis of rat hippocampal neurons by
inhibiting GSK-3.
Axonogenesis is a dynamic process mediated by various effectors and signaling
pathways, which ultimately act on the actin and microtubule cytoskeleton [18].
Cdc42 regulates axon specification, elongation, and guidance via interacting with
effector proteins, such as cofilin, a key factor for sustaining axon outgrowth
[19]. Cdc42 ablation increased phosphorylation (inactivation) of cofilin in the
axonal growth cones of mouse hippocampal neurons, while the active
(non-phosphorylated) form was enriched in the wild-type neurons [6]. Schwamborn
et al. [20] reported that transfection of a cdc42 active mutant induced
the formation of multiple axons in rat hippocampal neurons, whereas knockdown of
cdc42 by RNA interference led to complete loss of neuronal polarity.
Consistently, our data showed that increasing the activity of cdc42 by
transfecting cells with an active cdc42 mutant induced axonogenesis in primary
rat hippocampal neurons, while transfection of a negative cdc42 mutant exerted an
opposite effect.
Emerging evidence has suggested that GSK-3 plays a key regulatory role
in multiple neurodevelopmental events, including neurogenesis, neuronal
polarization, axon growth and guidance, and neuronal migration [21].
Phosphorylation of GSK-3 at Ser9 inactivates GSK-3 and
dephosphorylates CRMP-2, accelerating microtubule polymerization and
stabilization, thereby promoting the formation and elongation of axons [22]. In
this study, we observed that GSK-3 attenuated axonogenesis of rat
hippocampal neurons with excessive cdc42 activity, whereas inhibition of
GSK-3 by LiCl abolished the inhibitory effect of the negative cdc42
mutant on axonogenesis, suggesting that cdc42 induced axonogenesis via inhibiting
GSK-3.
Protein kinase B (Akt) is considered the major regulator of GSK-3
phosphorylation. Constitutive activation of GSK3 has been found to
reduce Akt-induced axon regeneration [23]. Co-transfection of hippocampal neurons
with an active Akt mutant (Myr-Akt) and an active GSK-3 mutant
(GSK-3 S9A) partially inhibited Akt-induced formation of multiple axons
[22]. However, a study by Gärtner et al. [24] suggested that
GSK-3 inhibition promotes the development of neuronal polarity in mice
independent of Akt pathway activation, implying that other factors may regulate
GSK-3 activity. The Par complex, comprising Par6, Par3, and an atypical
PKC (aPKC), is a key factor in cdc42 signaling and plays an essential role in
axonogenesis [20]. In migrating astrocytes, cdc42 was shown to regulate the
direction of cell protrusion by inducing phosphorylation of GSK-3 at
Ser9 via the Par6-aPKC complex [25]. In fibroblasts, cdc42 inactivated
GSK-3 (phosphorylation at Ser9) via activating the Par6/aPKC complex,
thereby promoting cell polarization. These findings suggest that cdc42 inhibits
GSK-3 activation through the Par6/aPKC complex and subsequently inhibits
CRMP-2 phosphorylation to promote axonogenesis. Further investigation into the
mechanisms of cdc42-mediated axonogenesis is needed.
5. Conclusions
In conclusion, our results suggest that cdc42 induces axonogenesis of rat
hippocampal neurons via inhibiting GSK-3 activity. These findings
support future exploration of cdc42/GSK-3-mediated axonogenesis
in vivo.
Abbreviations
CRMP-2, collapsin response mediator protein-2; GSK-3, Glycogen synthase
kinase-3; FBS, fetal bovine serum; NBCS, newborn calf serum; PBS,
phosphate-buffered saline.
Author Contributions
YTL, FZC, WC and HMZ contributed equally to this work; HHW designed the
research; YTL, FZC, WC and HMZ performed the experiments; YC, ZLL, FY, ZYL, WRD,
LZ, and HHW analyzed and interpreted the data; and HHW wrote and reviewed the
manuscript.
Ethics Approval and Consent to Participate
The use of animals was approved by the Animal Care and Use Committee of Southern
Medical University (The approval date was 26 March 2020) and all protocols were carried out in accordance with the NIH
Guidelines for the Care and Use of Laboratory Animals.
Acknowledgment
Not applicable.
Funding
This work is supported in part by grants from the National Natural Science
Foundation of China (81572222), Guangdong Basic and Applied Basic Research
Foundation (2021A1515010529), Science and Technology Projects in Guangzhou
(202102080091) and Innovation Training Program for College Students
(S202012121131 and 202112121315).
Conflict of Interest
The authors declare no conflict of interest.
 Fig. 1.
Fig. 1. Fig. 2.
Fig. 2. Fig. 3.
Fig. 3. Fig. 4.
Fig. 4.



