1 School of Life Sciences, Anhui Medical University, 230601 Hefei, Anhui, China
2 Central Laboratory, School of Medicine, The Second Affiliated Hospital, The Chinese University of Hong Kong, Shenzhen & Longgang District People’s Hospital of Shenzhen, 518172 Shenzhen, Guangdong, China
3 Department of Cardiology, School of Medicine, The Second Affiliated Hospital, The Chinese University of Hong Kong, Shenzhen & Longgang District People’s Hospital of Shenzhen, 518172 Shenzhen, Guangdong, China
†These authors contributed equally.
§These authors contributed equally.
Abstract
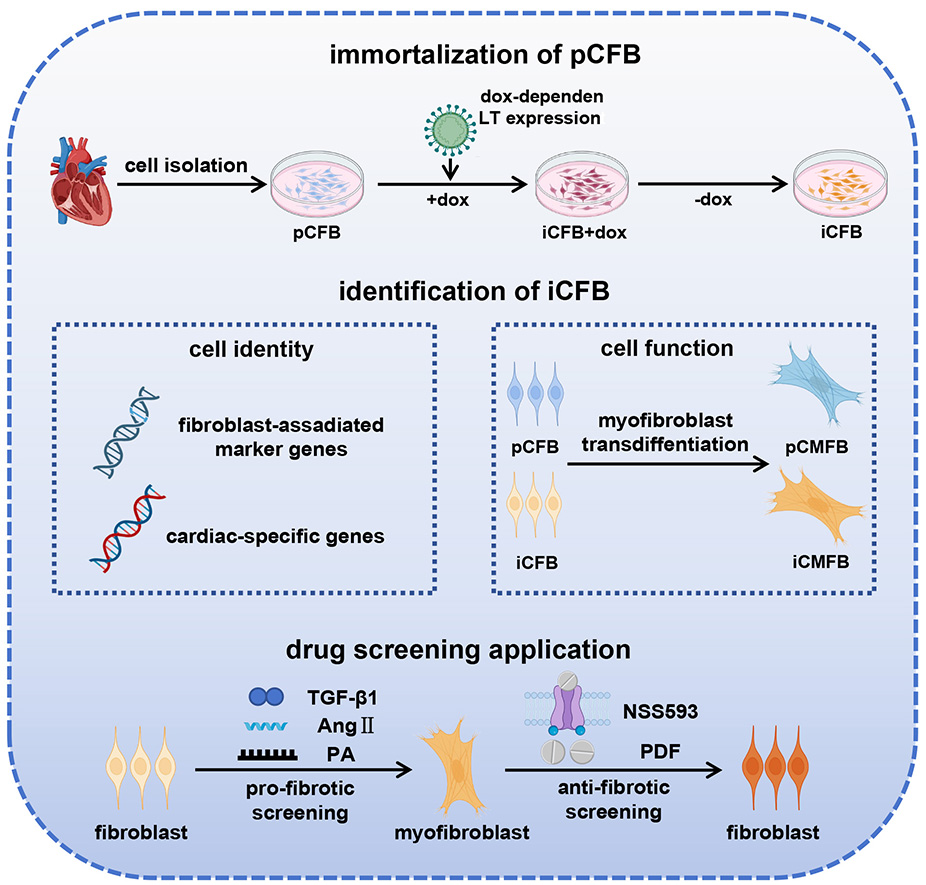
The development of anti-fibrotic therapies for heart failure is hindered by the absence of phenotypically stable and scalable human cardiac fibroblast (CFB) models suitable for pre-clinical drug screening. This study aimed to establish and validate a conditionally immortalized CFB (iCFB) cell line for screening cardiac fibrosis drugs.
An iCFB line was generated using a doxycycline (dox)-inducible system. The cellular characteristics were evaluated using molecular, protein, and functional analyses. The utility of the model was assessed by exposure to multiple pro-fibrotic stimuli (transforming growth factor-β1 (TGF-β1), angiotensin II, and palmitic acid) and two anti-fibrotic compounds (N-[(1R)-1,2,3,4-tetrahydro-1-naphthalenyl]-1H-benzimidazol-2-amine [NS8593] and pirfenidone).
iCFB proliferation was tightly regulated by dox. Upon dox withdrawal, the iCFBs reverted to a quiescent state and exhibited a molecular expression profile (collagen type I alpha 1 chain [COL1A1], periostin [POSTN], gap junction protein alpha 1 [GJA1], and T-box transcription factor 20 [TBX20]) comparable to that of primary CFBs. This phenotypic fidelity, along with a robust capacity for TGF-β1-induced myofibroblast differentiation, was maintained during long-term culture up to population doubling 60. The model responded consistently to diverse pro-fibrotic stimuli, confirming the anti-fibrotic efficacy of the transient receptor potential melastatin 7 (TRPM7) inhibitor, NS8593, and demonstrating the therapeutic potential of the repurposed drug pirfenidone.
We developed a novel iCFB model that integrates long-term expandability, high biological fidelity, and broad responsiveness to fibrotic signaling. This robust platform is well-suited for mechanistic studies and drug screening, thereby facilitating and accelerating the discovery of anti-fibrotic therapeutics.
Graphical Abstract
Keywords
- cell immortalization
- fibroblasts
- fibrosis
- myofibroblasts
- cell transdifferentiation
- drug evaluation
Cardiac fibroblasts (CFBs) are the principal extracellular matrix (ECM) producers in the heart and exhibit considerable plasticity. Under physiological conditions, they play a critical role in maintaining normal cardiac structure and function [1, 2, 3]. However, in response to pathological stimuli such as myocardial injury or pressure overload, they become activated and differentiate into cardiac myofibroblasts (CMFBs) [4, 5, 6]. Activated CMFBs exhibit sustained proliferative activity and secrete excess collagen and other ECM components [7, 8, 9, 10]. Although this response is initially adaptive-for example, by preventing cardiac rupture following myocardial infarction-excess or persistent fibrotic activation increases myocardial stiffness, reduces ventricular compliance, impairs cardiac function, and ultimately contributes to heart failure progression [11, 12, 13, 14, 15].
At present, cardiac fibrosis has no specific and effective therapies, underscoring an urgent need for anti-fibrotic drug development [16, 17, 18, 19]. Progress in this field is constrained by the limitations of available experimental models. Current assessments of fibrosis primarily rely on animal models and in vitro assays employing fibroblasts [20]. However, animal-derived cellular models exhibit substantial species-specific differences compared to human cells, and primary human CFBs (pCFBs) are difficult to obtain in sufficient quantities owing to limited tissue availability, restricted proliferative capacity, and ethical considerations. This creates a critical knowledge gap: the lack of a scalable, phenotypically stable, and physiologically relevant human CFB in vitro model that can bridge the translational gap between pre-clinical research and clinical anti-fibrotic drug development and is suitable for high-throughput pro-fibrotic and anti-fibrotic drug screening. As an innovative optimization strategy, conditional immortalization technology directly addresses this long-standing technical bottleneck via a drug-controlled reversible gene expression system [21, 22]. This strategy enables flexible switching between an immortalized proliferative state for large-scale cell production and a quiescent, primary-cell-like state for functional experiments, thus achieving the unification of long-term scalability and high biological fidelity, which is difficult to realize with existing models. Consequently, the development of a stable, reproducible, and physiologically relevant human-based in vitro CFB model using this conditional immortalization approach is essential to accelerate the discovery and evaluation of anti-fibrotic therapeutics.
In this study, we aimed to establish a conditionally immortalized human CFB cell
line (iCFB) based on a doxycycline (dox)-inducible system that supports long-term
expansion while retaining the key biological characteristics of pCFB. CMFB
activation was induced in vitro using pro-fibrotic mediators, including
transforming growth factor-
The dox-inducible plasmid, pLVET-tTR-KRAB, was obtained from Addgene (Cambridge, MA, USA; plasmid #11644). Based on the gene sequences retrieved from GenBank, the SV40 large T antigen (LT) fragment was synthesized and used to construct a lentiviral shuttle recombinant plasmid. Specifically, the human EEF1A1 promoter and enhanced green fluorescent protein (EGFP)-coding sequence in pLVET-tTR-KRAB were replaced with the collagen type I alpha 2 chain (COL1A2) promoter and LT gene, respectively. All plasmid construction and lentiviral packaging procedures were performed using VectorBuilder (Guangzhou, Guangdong, China).
pCFBs were isolated from ventricular tissue of a fetus at 14 weeks of gestation. The tissue was donated by the fetal parents with written informed consent. This study was approved by the Ethics Committee of Longgang District People’s Hospital of Shenzhen (approval number: 2025101). Cells were cultured in high-glucose Dulbecco’s modified Eagle medium (H-DMEM; Gibco, Thermo Fisher Scientific, Waltham, MA, USA; Catalog No. 11965118) supplemented with 10% fetal bovine serum (FBS; Gibco, Catalog No. A5256901), 1% penicillin-streptomycin (Gibco, Catalog No. 15140122), and 10 ng/mL recombinant human basic fibroblast growth factor (bFGF; R&D Systems, Minneapolis, MN, USA; Catalog No. 233-FB) and maintained at 37 °C in a humidified incubator with 5% CO2. The culture medium was replaced every three days. Upon reaching confluence, pCFBs were passaged at a 1:4 ratio and cells at population doubling 4 (PD4) were used for subsequent experiments.
The pCFBs at approximately PD4 were transduced with lentiviral vectors carrying the dox-inducible LT expression system. After 24 h of initial culture in the primary cell medium, the medium was replaced with proliferation medium supplemented with 100 ng/mL dox (Sigma-Aldrich, St. Louis, MO, USA; Catalog No. D9891) to induce LT expression. Successfully transduced cells were designated conditional iCFBs and maintained in freshly prepared dox-containing proliferation medium, which was replaced every alternate day. When the cultures reached approximately 90% confluence, they were passaged in a 1:8 ratio.
For growth curve analysis, iCFBs and pCFBs were passaged at ratios of 1:8 and 1:4, respectively, upon reaching confluency. To evaluate the proliferation rates and their dependence on LT expression, the cells were seeded at a low density and cultured in medium supplemented with 100 ng/mL dox (iCFBs) or standard culture medium (pCFBs). The cell numbers were quantified at the indicated time points using a cytometer (BodBoge, Guangzhou, Guangdong, China).
Primary human foreskin fibroblasts (FFBs) were purchased from Jennio Biotech Co., Ltd. (Guangzhou, Guangdong, China; Catalog no. JNO-H0612) and cultured in H-DMEM supplemented with 10% FBS in a humidified incubator at 37 °C with 5% CO2. The culture medium was replaced every three days. The cells were routinely passaged at a 1:4 ratio when the cultures reached approximately 80% confluence.
All primary cells were routinely tested for mycoplasma contamination using the
BeyoDirect™ Mycoplasma qPCR Detection Kit (Beyotime, Shanghai,
China; Catalog No. C0303S) and consistently tested negative, as defined by the
kit’s criteria (sample Ct
The cells were fixed with 4% paraformaldehyde in phosphate-buffered saline
(PBS), permeabilized with 0.5% Tween-20 in PBS, and blocked with PBS containing
1% bovine serum albumin (Sigma-Aldrich, Catalog No. A8022), and 0.05% Tween-20
(Sangon Biotech, Shanghai, China; CAS No. 9005-64-5). pCFB, iCFB, and FFB
monolayers were incubated overnight at 4 °C with the following primary
antibodies: mouse anti-SV40 LT (1:50; Santa Cruz Biotechnology, Dallas, TX, USA;
Catalog No. sc-147), anti-Collagen type I (anti-COL-1) (1:200; Proteintech,
Wuhan, Hubei, China; Catalog No. 66761-1-Ig), anti-
After three washes with PBS containing 0.05% Tween-20, the bound primary antibodies were detected using Alexa Fluor 594-conjugated goat anti-mouse IgG (H+L) (1:200; Thermo Fisher Scientific, Catalog No. A-11005). Nuclei were counterstained with Hoechst 33342 (10 µg/mL; Thermo Fisher Scientific, Catalog No. H3570). Fluorescence images were acquired using a Leica STELLARIS 5 confocal laser-scanning microscope (Leica Microsystems).
Protein extracts were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred on 0.22 or 0.45 µm polyvinylidene difluoride (PVDF) membranes (Millipore, Billerica, MA, USA; Catalog No. IPVH00010). After blocking with 5% skim milk in Tris-buffered saline containing Tween-20 (TBST), the membranes were incubated overnight at 4 °C with the following primary antibodies: anti-SV40 LT (1:1000; Santa Cruz Biotechnology, Catalog No. sc-147) and anti-glyceraldehyde-3-phosphate dehydrogenase (anti-GAPDH) (1:5000; Proteintech, Catalog No. 60004-1-Ig).
The membranes were subsequently incubated with horseradish peroxidase (HRP)-conjugated secondary antibody (1:5000; Proteintech, Catalog No. SA00001-0) for 1 h at room temperature. Protein bands were visualized using the Thermo iBright CL1000 imaging system (Thermo Fisher Scientific) with Pierce™ ECL Western Blotting Substrate (Thermo Fisher Scientific, Catalog No. 32209). Band intensities from at least three independent experiments were quantified using the ImageJ software (version 1.46r, National Institutes of Health, Bethesda, MD, USA).
Total RNA was extracted from the cultured cells using AG RNAex Pro Reagent (Accurate Biotechnology, Ningbo, Zhejiang, China) according to the manufacturer’s instructions. The isolated RNA was reverse transcribed into complementary DNA (cDNA) using Evo M-MLV RT Premix for qPCR (Accurate Biotechnology). Gene expression levels were determined by RT-qPCR using the SYBR® Green Premix Pro Taq HS qPCR Kit (Accurate Biotechnology). PCR amplification and fluorescence detection were performed using the QuantStudio 3 Real-Time PCR System (Thermo Fisher Scientific). Relative gene expression was calculated using the 2-ΔΔCt method. The primer sequences are listed below. 5′-CAGCCACTAGCCATTGTGGA-3′ (F, gap junction protein alpha 1 [GJA1]); 5′-GGCTGTTGAGTACCACCTCC-3′ (R, GJA1); 5′-TGACGAGACCAAGAACTGCC-3′ (F, collagen type I alpha 1 chain [COL1A1]); 5′-GCACCATCATTTCCACGAGC-3′ (R, COL1A1); 5′-GGAGGCAAACAGCTCAGAGT-3′ (F, periostin [POSTN]); 5′-GGTGTGTCTCCCTGAAGCAG-3′ (R, POSTN); 5′-GTTCCGCTCCTCTCTCCAAC-3′ (F, actin alpha 2 [ACTA2]); 5′-TAGTCCCGGGGATAGGCAAA-3′ (R, ACTA2); 5′-ACCATCCGGGTGTCCTTTTC-3′ (F, T-box transcription factor 20 [TBX20]); 5′-TGACCCTCGATTTGGGGTTG-3′ (R, TBX20); 5′-TTGCATCAGCCTGTGGATGT-3′ (F, discoidin domain receptor tyrosine kinase 2 [DDR2]); 5′-GAGTCCAGCCAAAGGTCTCC-3′ (R, DDR2); 5′-CTCTGGCACGTCTTGACCTT-3′ (F, vimentin [VIM]); 5′-TTGCGCTCCTGAAAAACTGC-3′ (R, VIM); 5′-AATGGGCAGCCGTTAGGAAA-3′ (F, GAPDH); 5′-GCGCCCAATACGACCAAATC-3′ (R, GAPDH).
The collagen-based cell contraction assay was performed using a commercial kit
(Cell Biolabs, San Diego, CA, USA; Catalog No. CBA-201) according to the
manufacturer’s instructions. iCFBs and pCFBs (2.0
The iCFBs were cultured in doxycycline-free DMEM for 4 days to suppress LT
expression. Thereafter, pCFBs and iCFBs were treated with 10 ng/mL TGF-
To evaluate the anti-fibrotic response of iCFBs, cells were treated for 48 h
with medium containing 10 ng/mL TGF-
Data are presented as mean
To establish an iCFB line, pCFBs were transduced with lentiviral particles encoding a dox-inducible LT expression cassette under the control of the COL1A2 promoter (Fig. 1A). Successful immortalization was confirmed by LT expression, which was robustly expressed in transduced cells (iCFBs) cultured in dox-containing medium, whereas no LT signal was detected in pCFBs (Fig. 1B,C).
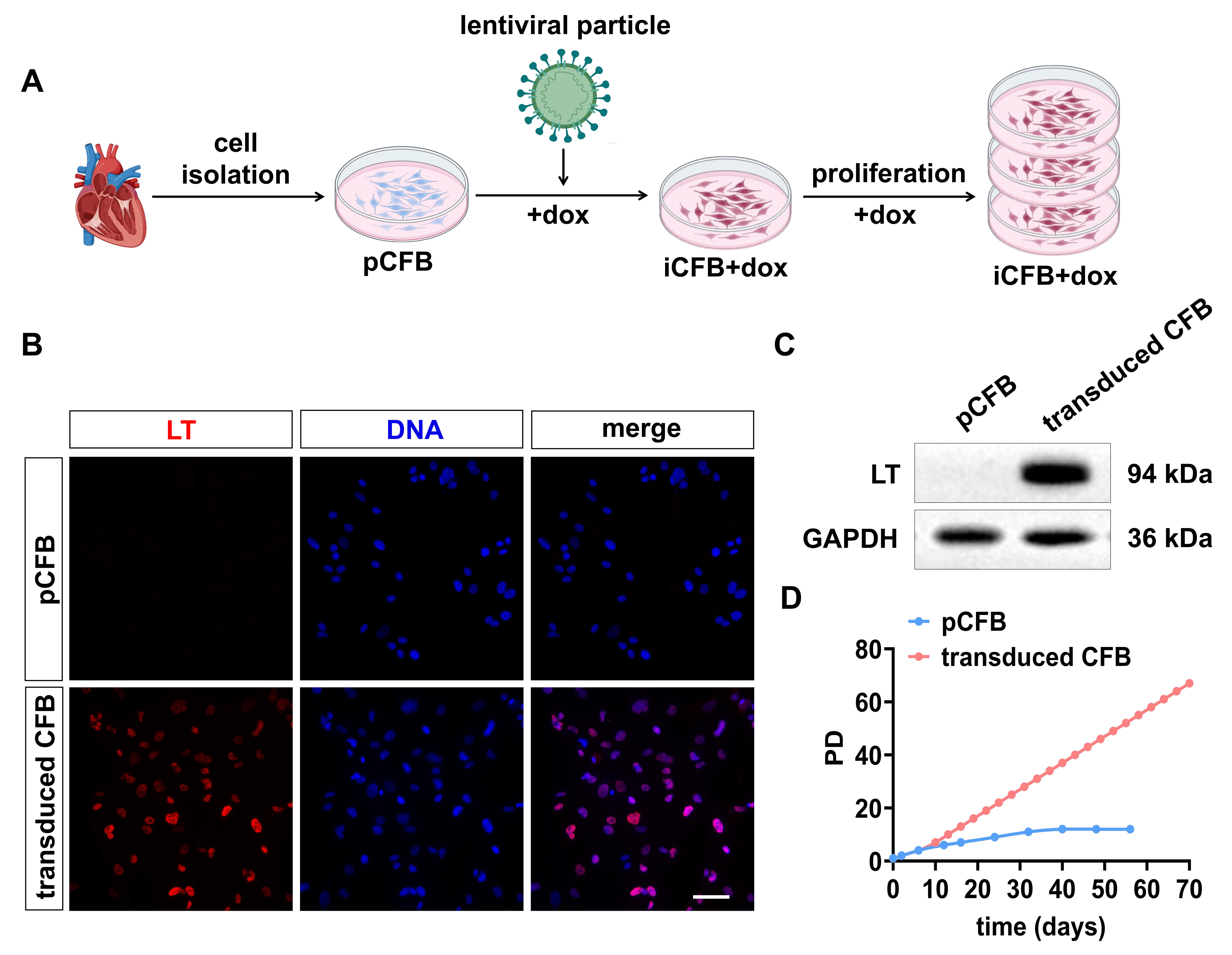 Fig. 1.
Fig. 1.
Generation of conditionally immortalized cardiac fibroblasts (iCFBs). (A) Schematic overview of the conditional immortalization strategy. (B) Immunofluorescence staining confirming LT expression in transduced cardiac fibroblasts cultured in the presence of dox. Scale bar, 50 µm. (C) Western blot analysis confirming LT expression in transduced CFB cultured in the presence of dox. (D) Growth curves of pCFBs and transduced CFBs cultured in the presence of dox. CFB, cardiac fibroblast; pCFB, primary cardiac fibroblast; LT, SV40 large T antigen; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; dox, doxycycline; PD, population doubling; iCFB+dox, iCFB cultured in the presence of dox; DNA, deoxyribonucleic acid. Created in BioRender. Liu, J. (2026) https://BioRender.com/iuxys2w.
Growth curve analysis demonstrated that pCFBs ceased to proliferate after approximately 9 population doublings (PDs). In contrast, iCFBs cultured in the presence of dox maintained sustained proliferation for over 70 PDs without entering senescence, with an average population doubling period of approximately 24 h (Fig. 1D). Taken together, these results confirmed the successful establishment of an iCFB line.
To determine whether LT expression and the associated immortalization phenotype in iCFBs were strictly dox-dependent, they were cultured in a medium switched from dox-containing to dox-free conditions (Fig. 2A). Western blot analysis confirmed LT expression in iCFBs maintained in dox-containing medium (day 0), with a time-dependent decline to undetectable levels by day 6 after dox withdrawal (Fig. 2B,C), which was further validated by immunofluorescence staining (Supplementary Fig. 1). Consistent with the changes in LT expression, Ki-67 antigen (Ki-67) positivity was abundant in dox-cultured iCFBs, but decreased substantially after dox withdrawal (Fig. 2D). Accordingly, iCFBs proliferated continuously after dox treatment. In contrast, following dox removal, cell numbers plateaued after a modest initial increase within days 0–2 (Fig. 2E). Taken together, these findings demonstrate that dox tightly regulates both LT expression and proliferative capacity of iCFBs, enabling a reversible switch between an immortalized proliferative state and a non-immortalized phenotype.
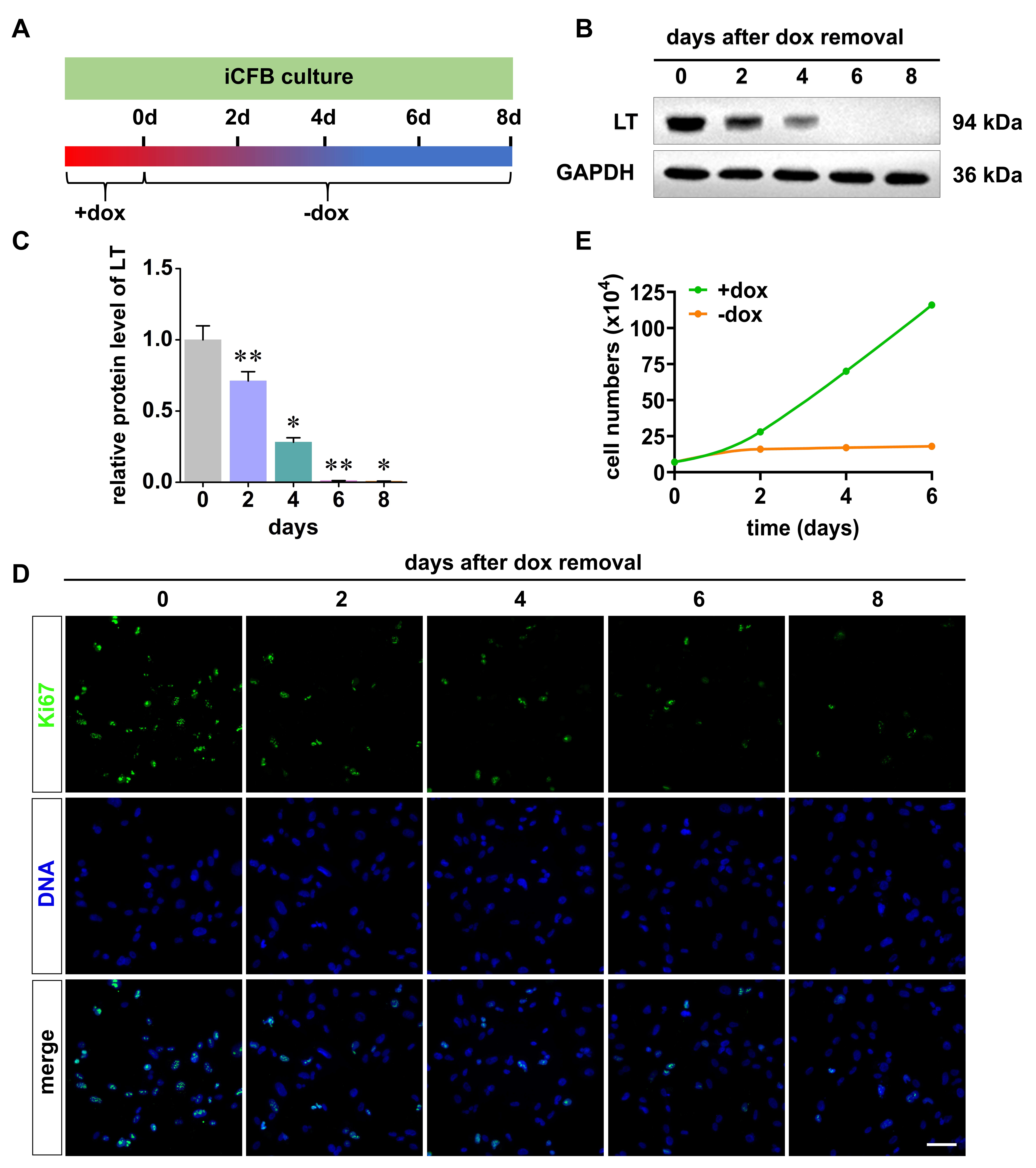 Fig. 2.
Fig. 2.
Conditional proliferative capacity of conditionally immortalized
cardiac fibroblasts (iCFBs). (A) Schematic illustration of iCFB preparation and
dox withdrawal process. (B,C) Western blot analysis and corresponding
quantification of LT protein expression in iCFBs before (day 0) and after (days
2, 4, 6, and 8) dox withdrawal. (D) Immunofluorescence staining of the
proliferation marker Ki-67 in iCFBs before (day 0) and after (days 2, 4, 6, and
8) dox withdrawal. Scale bar, 50 µm. (E) Quantitative analysis of iCFB cell
counts after culture in media with or without dox. iCFB, conditionally
immortalized cardiac fibroblast; LT, SV40 large T antigen; GAPDH,
glyceraldehyde-3-phosphate dehydrogenase; dox, doxycycline; Ki-67, Ki-67 antigen;
DNA, deoxyribonucleic acid. Data were presented as mean
Next, we examined whether the iCFBs retained their phenotypic characteristics. We compared the expression of canonical CFB markers in iCFBs maintained under proliferative conditions with dox (iCFB+dox), iCFBs cultured without dox for 4 days to revert to a non-immortalized state (iCFB), and pCFBs. No significant differences were observed in the expression levels of key CFB marker genes, including the fibroblast-associated markers COL1A1, DDR2, VIM, and POSTN, as well as cardiac markers GJA1 and TBX20 between pCFBs and iCFBs (Fig. 3A). Notably, sustained exposure to dox (iCFB+dox) reduced the expression of these CFB-associated markers compared with that in both pCFBs and iCFBs (Fig. 3A).
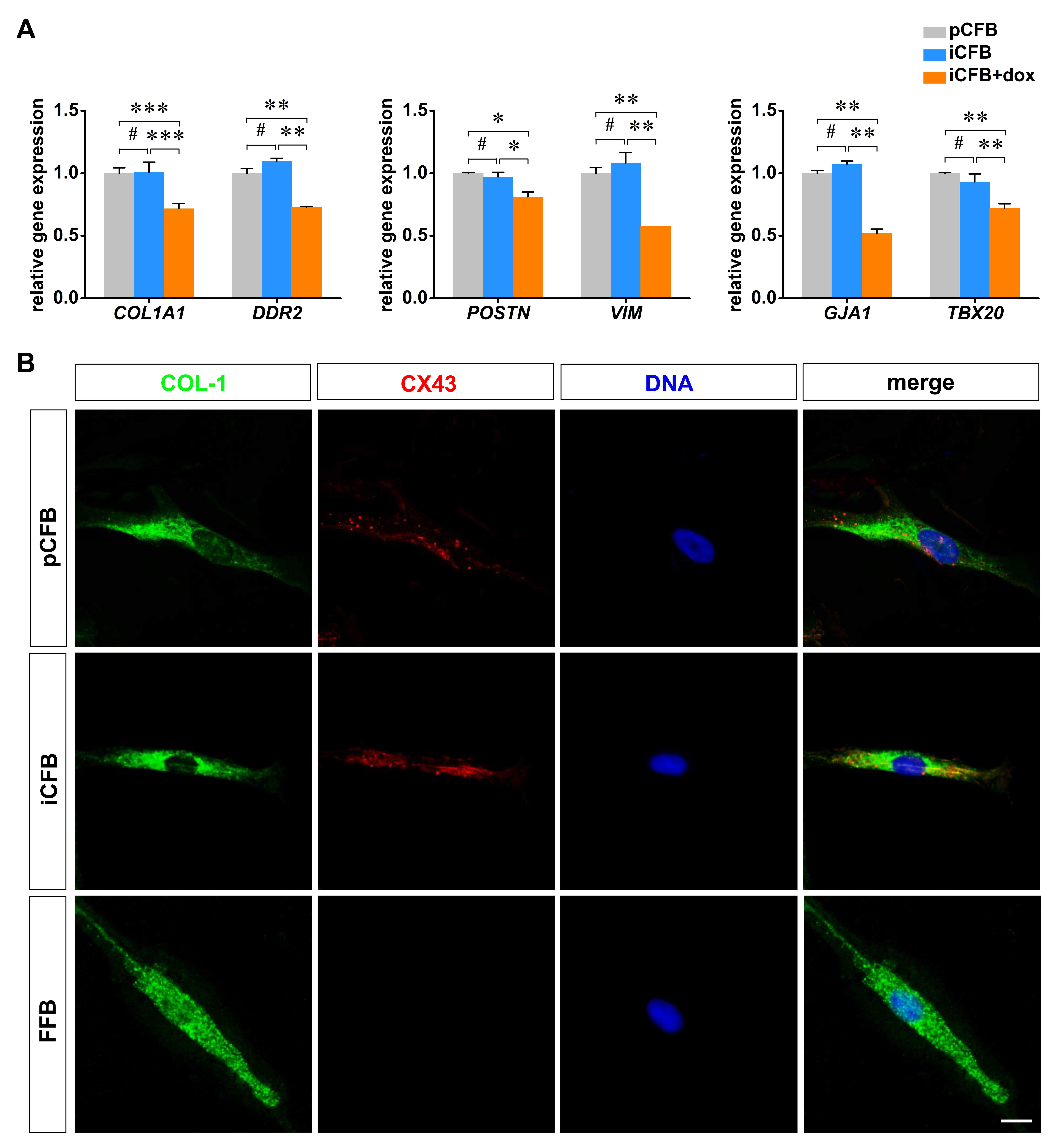 Fig. 3.
Fig. 3.
Conditionally immortalized cardiac fibroblasts (iCFBs) retain
primary cardiac fibroblast (pCFB) identity. (A) RT-qPCR analysis of
fibroblast-associated marker genes (COL1A1, DDR2,
POSTN, and VIM) and cardiac-specific genes (GJA1 and
TBX20) in pCFBs and iCFBs cultured in the presence or absence of dox.
(B) Representative immunofluorescence images showing the expression of COL-1
(green) and CX43 (red) in iCFBs, pCFBs, and FFBs. Nuclei were counterstained with
Hoechst 33342 (blue). Scale bar, 10 µm. RT-qPCR, reverse
transcription-quantitative polymerase chain reaction; COL1A1, collagen
type I alpha 1 chain; DDR2, discoidin domain receptor tyrosine kinase 2;
POSTN, periostin; VIM, vimentin; GJA1, gap junction
protein alpha 1; TBX20, T-box transcription factor 20; COL-1, Collagen
type I; CX43, connexin 43; pCFB, primary cardiac fibroblast; iCFB, conditionally
immortalized cardiac fibroblast; iCFB+dox, iCFB cultured in the presence of dox;
FFB, foreskin fibroblast; DNA, deoxyribonucleic acid. Data were presented as mean
Consistent with the transcriptional data, immunofluorescence staining verified comparable COL-1 and CX43 protein expression in pCFBs and dox-withdrawn iCFBs, confirming restoration of the pCFB molecular profile in iCFBs (Fig. 3B). FFBs, as non-cardiac fibroblast controls, expressed COL-1, but not CX43, further supporting the cardiac-specific identity of dox-withdrawn iCFBs (Fig. 3B). Taken together, these results indicate that, in the absence of dox-induced proliferative pressure, the molecular identity of iCFBs closely resemble that of pCFBs. Accordingly, iCFBs cultured for 4 d without dox were used for all subsequent functional experiments.
A series of functional assays was performed to compare the biological behaviors
of iCFBs and pCFBs. TGF-
Immunofluorescence analysis showed that TGF-
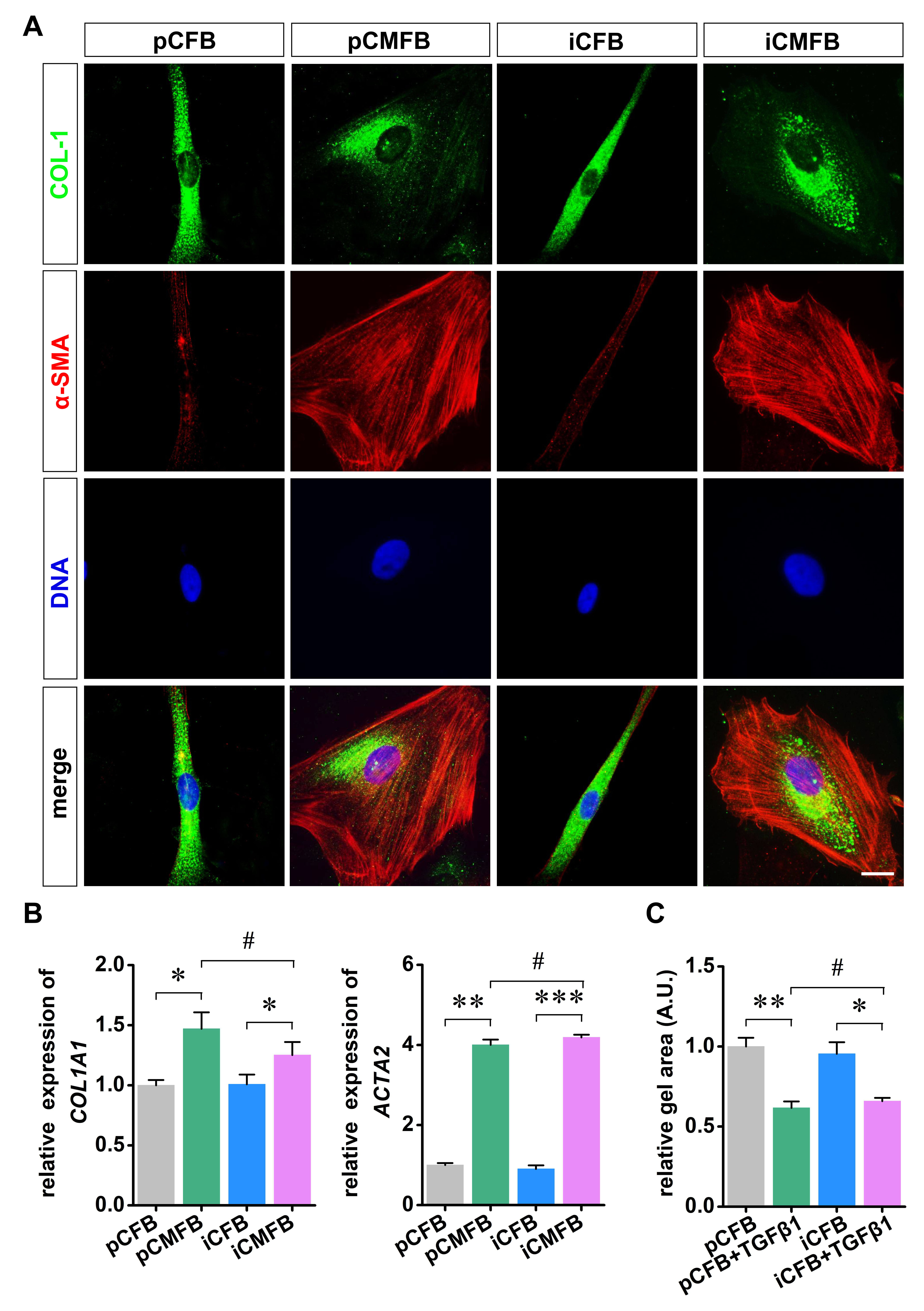 Fig. 4.
Fig. 4.
Conditionally immortalized cardiac fibroblasts (iCFBs) retain
key biological functions of primary cardiac fibroblasts (pCFBs). (A)
Representative immunofluorescence images showing the expression of COL-1 (green)
and
Enhanced contractile activity is a hallmark of myofibroblast differentiation. In
line with this, collagen gel contraction assays revealed that TGF-
To assess the scalability of iCFBs as in vitro models, we examined
their phenotypic stability and functional capacity during long-term subculture.
Marker gene expression and TGF-
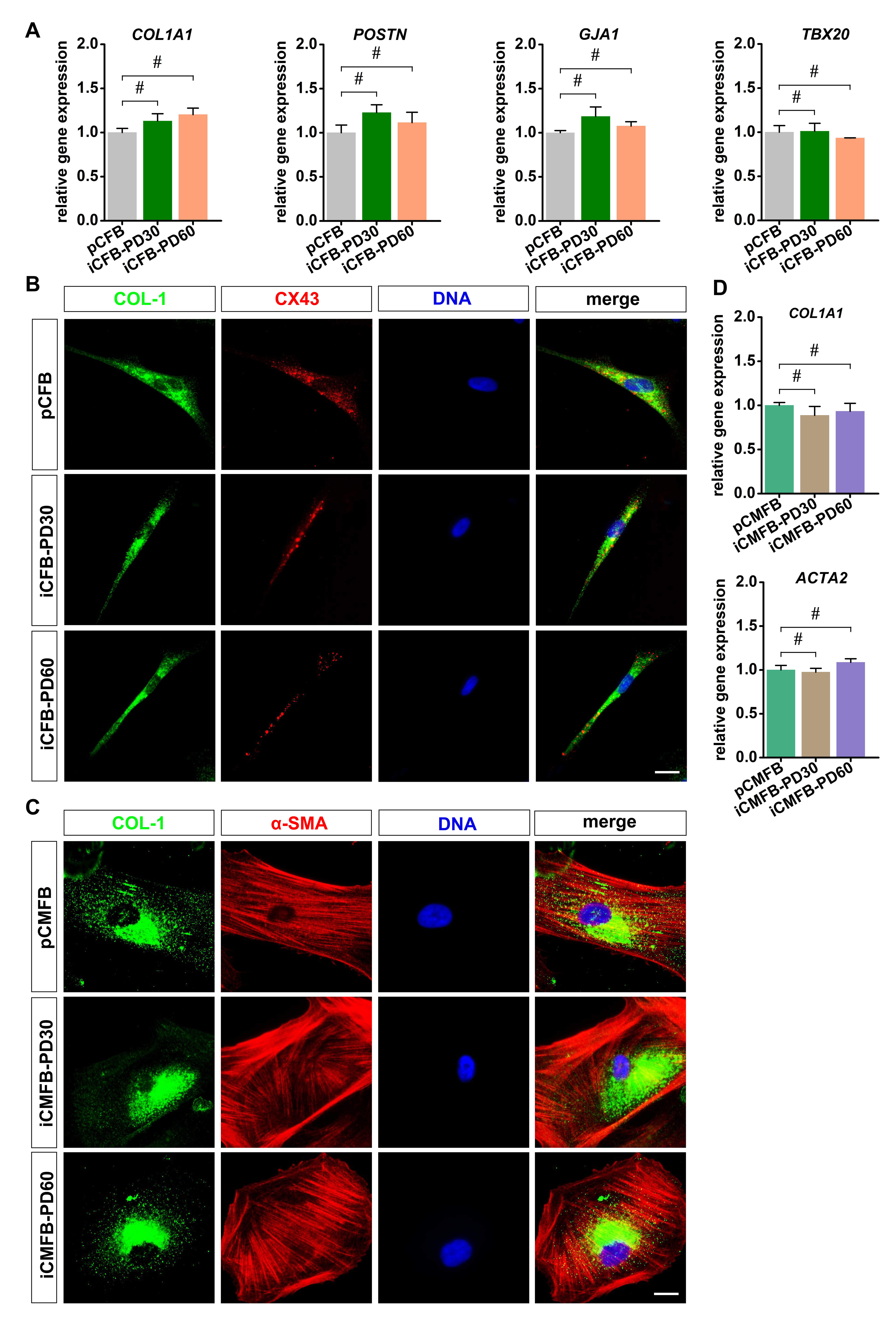 Fig. 5.
Fig. 5.
Conditionally immortalized cardiac fibroblasts (iCFBs) preserve
phenotypic and functional properties during long-term expansion. (A) RT-qPCR
analysis of COL1A1, POSTN, GJA1, and TBX20
expression levels in pCFBs and iCFBs at PD30 and PD60. (B) Representative
immunofluorescence images showing COL-1 (green) and CX43 (red) expression in
pCFBs and iCFBs at PD30 and PD60. Nuclei were counterstained with Hoechst 33342
(blue). Scale bar, 10 µm. (C) Representative immunofluorescence images
showing COL-1 (green) and
Immunofluorescence staining further demonstrated that iCFBs at both PD30 and PD60 displayed cellular morphology and expression patterns of COL-1 and CX43 that were highly similar to those observed in pCFBs, with no spontaneous differentiation (Fig. 5B).
After TGF-
Taken together, these results demonstrate that iCFBs maintain their core molecular identity and myofibroblast transdifferentiation potential after prolonged in vitro expansion.
To evaluate the suitability of iCFB model for drug screening, we assessed its
responsiveness to multiple pro-fibrotic stimuli. After 4 days of culture in
dox-free medium, iCFBs were treated with TGF-
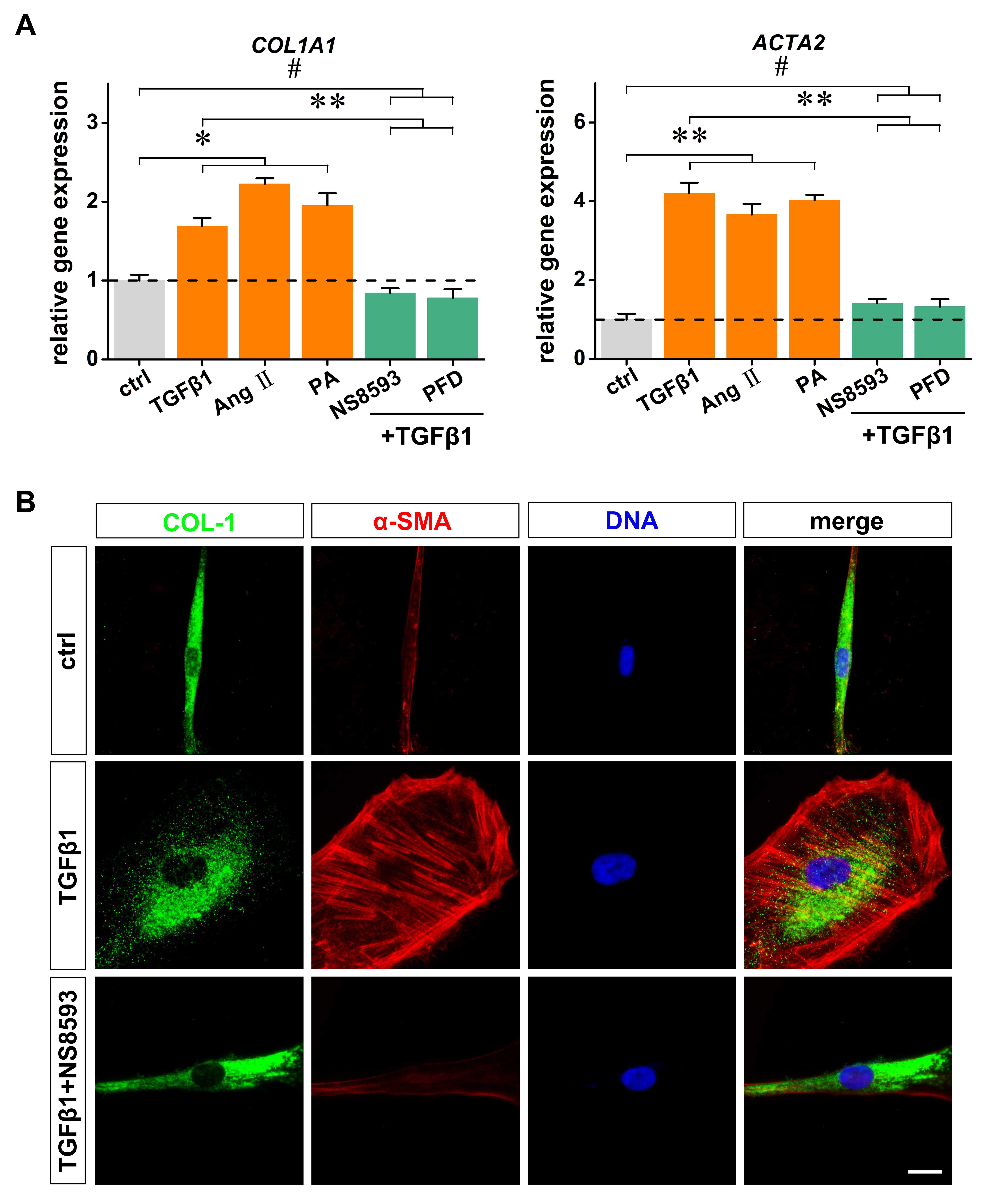 Fig. 6.
Fig. 6.
Conditionally immortalized cardiac fibroblasts (iCFBs) serve as
a platform for pro- and anti-fibrotic drug screening. (A) RT-qPCR analysis of
myofibroblast marker gene expression (COL1A1 and ACTA2) in
iCFBs treated with pro-fibrotic stimuli (TGF-
Having established the broad pro-fibrotic responsiveness of the model, we
examined its utility for evaluating anti-fibrotic compounds. Specifically, we
tested whether the transient receptor potential melastatin 7 (TRPM7) channel
antagonist NS8593 and the clinically approved anti-fibrotic drug PFD could
suppress TGF-
Consistent with these molecular findings, immunofluorescence analysis showed
that TGF-
In this study, we established and characterized an iCFB cell line as a scalable
and phenotypically stable platform for studying fibrosis. A key feature of this
model is its ability to respond to multiple pro-fibrotic stimuli, including
TGF-
Using this platform, we confirmed the anti-fibrotic effect of the TRPM7
inhibitor NS8593 [28], which attenuated TGF-
We also observed that PFD significantly suppressed TGF-
Conditional immortalization offers a practical advantage over permanent methods. Proliferation is tightly controlled by dox. Upon withdrawal, iCFBs exit the cell cycle and re-express key cardiac fibroblast markers (COL1A1, POSTN, GJA1, TBX20) at levels comparable to those in primary cells. This reversible system allows scalable expansion while enabling experiments to be performed on cells in a quiescent, primary-like state, thereby avoiding the phenotypic drift often observed in constitutively immortalized lines.
This model also demonstrates long-term stability. The iCFBs retained their molecular identity and differentiation capacity even at high passage numbers (up to PD60). This consistency addresses the limitation of primary cultures, namely senescence and batch-to-batch variability, and supports the use of iCFBs in reproducible large-scale screening applications.
This study has several limitations. First, as a monocellular in vitro model, iCFBs cannot fully recapitulate the complex in vivo microenvironment of the fibrotic myocardium, which involves dynamic interactions between multiple cell types (such as cardiomyocytes, immune cells, and endothelial cells), biomechanical cues, and paracrine signaling networks.
Second, iCFBs are derived from fetal cardiac ventricular tissue and therefore may not fully reflect the phenotypic and functional characteristics of adult CFBs, which primarily mediate fibrotic remodeling in adult heart failure and myocardial injury. Fetal and adult cardiac fibroblasts exhibit distinct molecular profiles, proliferative capacities, and responsiveness to pro-fibrotic stimuli. This difference may limit the direct translational relevance of the findings obtained using this fetus-derived model in adult cardiac fibrosis.
Third, our drug screening assessments were based on short-term in vitro exposure and did not evaluate long-term functional outcomes or potential off-target effects. Therefore, the findings of this model require further validation in more physiologically relevant systems, including ex vivo models, in vivo animal studies, and clinical investigations. Future studies using adult-derived iCFBs and co-culture systems incorporating multiple cardiac cell types will enhance the translational relevance of this platform.
In conclusion, we successfully established a conditionally immortalized human CFB line as a novel and superior in vitro model for cardiac fibrosis research. The core innovation lies in dox-controlled reversible immortalization, which provides unlimited scalability and allows cells to revert to a primary-like quiescent state for functional assays, a critical advantage over permanently immortalized lines that often suffer from phenotypic drift. Comprehensive characterization confirmed that iCFBs maintained the key molecular identity, long-term stability, and differentiation potential of primary fibroblasts. Furthermore, we validated the broad applicability of the model by demonstrating its robust activation by multiple pathogenic stimuli and its utility for evaluating both novel target (NS8593) and repurposed (PFD) anti-fibrotic compounds. This work not only provides a reliable and standardized cellular platform to overcome the limitations of primary cell sourcing, but also opens efficient pathways for both de novo drug discovery and drug repurposing for treating cardiac fibrosis. Therefore, the iCFB model holds substantial promise for enhancing the precision and throughput of pre-clinical development of much-needed anti-fibrotic therapies.
All data and materials are available from the corresponding author upon reasonable request.
SW and RH designed the experiments. SW, RG, and CW performed the experiments. SW, CW, and WK analyzed the data. RH, JL, and CL contributed to data interpretation and manuscript revision. RH, JL, and CL wrote the paper. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Ethics Committee of Longgang District People’s Hospital of Shenzhen, with the approval number: 2025101. The study was carried out in accordance with the guidelines of the Declaration of Helsinki. And obtained the corresponding written informed consent from the fetal parents.
We thank Mrs. Lei Zhang from Longgang District People’s Hospital of Shenzhen for her administrative assistance.
This work was granted by the Shenzhen Science and Technology Innovation Program (No. JCYJ20230807141801002 and JCYJ20240813160259020) and the Longgang District Science and Technology Innovation Bureau Project (LGKCYLWS2022025). This work was supported by the Longgang District Key Specialty Program.
The authors declare no conflicts of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL49465.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






