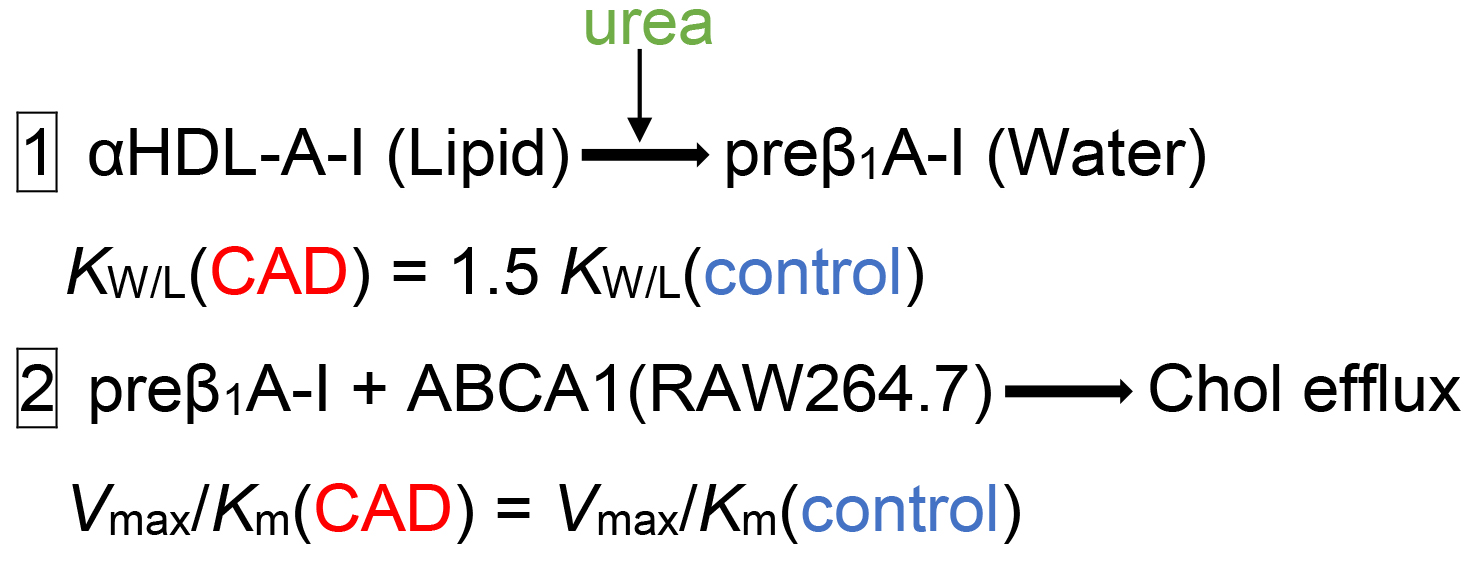
1 Laboratory of Structural Fundamentals of Lipoprotein Metabolism, National Medical Research Center for Therapy and Preventive Medicine, 101990 Moscow, Russia
2 Cardiosurgery Department, M.F. Vladimirsky Moscow Regional Research and Clinical Institute MONIKI, 129090 Moscow, Russia
Abstract
The significance of cholesterol efflux as a predictor of coronary artery disease (CAD) remains controversial. The intracellular cholesterol export via the ABCA1 transporter involves the acceptance of cholesterol by both lipid-free apolipoprotein A-I and high-density lipoproteins (HDL). An estimate of the efficiencies of two reactions is thus required.
HDL from the plasma of 63 control and 76 male CAD patients was obtained by the precipitation of apoB-containing lipoproteins and denatured by urea. We measured apoA-I dissociation concomitant with HDL denaturation by agarose gel electrophoresis followed by immunodetection and the expression of 65 preselected genes in blood mononuclear cells by real-time PCR. The total cholesterol efflux capacity (CEC) of ATP-binding cassette transporter A1 (ABCA1)-mediated cholesterol efflux from RAW 264.7 macrophages, when preβ-HDL and α-HDL act as competitive inhibitors of each other for the binding to ABCA1 transporter, was measured with intact HDL and pre-denatured HDL as a source of lipid-free apoA-I.
The phospholipid:apoA-I and cholesterol:apoA-I ratios in HDL from CAD patients were higher than those for control patients across the full range of plasma HDL-cholesterol levels. ApoA-I partitioned 1.5-fold higher into the water phase for HDL from CAD patients relative to controls. In CAD patients, the dissociation parameter D was inversely correlated with absolute and normalized per apoA-I phospholipid and cholesterol levels in HDL. For control patients, the D parameter was positively correlated with ABCA1 gene expression. For CAD patients, the D parameter was positively correlated with PLTP and inversely with CUBN and ALB gene expression. ApoA-I functionality in ABCA1-mediated cholesterol efflux from RAW 264.7 macrophages to lipid-free apoA-I generated from urea-induced HDL denaturation was similar for HDL from control and CAD groups. The retained CEC of lipid-free apoA-I in CAD may be masked by competition with α-HDL, which has a lower CEC, for ABCA1 binding to preβ-HDL.
The enrichment of HDL with cholesterol and phospholipids may contribute to the increased apoA-I dissociation from HDL in CAD. Estimates of both lipid-free apoA-I and intact HDL may be a prerequisites for a detailed study of ABCA1-mediated cholesterol efflux, which could allow these apoA-I forms to be identified as CAD predictors.
Graphical Abstract
Keywords
- ABCA1
- apolipoprotein A
- cholesterol
- coronary artery disease
- gene expression
- high-density lipoprotein
- lipid-bound
- lipid-free
Human plasma high-density lipoproteins (HDL) possess an atheroprotective effect
through the reverse cholesterol transport from arterial-wall macrophages to the
liver [1], with cholesterol efflux as a first step. The efflux includes active
cholesterol transport by ABCA1 and ABCG1 transporters, facilitated diffusion with
SR-B1, and passive diffusion [2]. ApoA-I is a major HDL apolipoprotein that
exists in lipid-bound and lipid-free forms. The lipid-free apoA-I is a primary
cholesterol acceptor in the ABCA1-mediated efflux. However, small HDL particles
are efficient cholesterol acceptors as well [3]. The HDL heterogeneity by density
and charge results in the existence of light HDL2 and dense HDL3 and
particles with pre
A strong inverse relationship between HDL cholesterol efflux capacity (CEC) and
coronary artery disease (CAD) signifies HDL functionality more than
HDL-cholesterol level in the HDL atheroprotective effect [9]. HDL functionality
includes HDL particle number and heterogeneity and particle ability to accept
effluxed cholesterol. The significance of ABCA1-mediated efflux and HDL
functionality in CAD are inconsistent [3, 10, 11, 12, 13, 14, 15]. CEC from macrophages had a
strong inverse association with morphological and prognostic markers of
angiographic CAD [16]. The higher concentration of pre
The major goals of our study were to quantitatively follow urea-induced denaturation of HDL from control and CAD patients by apoA-I dissociation and to use generated lipid-free apoA-I to measure the efficacy of cholesterol efflux by a single ABCA1 transporter to two competing substrates—lipid-free apoA-I and mature HDL. We describe also the relations between apoA-I dissociation and the expression of selected genes controlling HDL metabolism and atherogenesis in control and CAD patients.
Patient selection with widely varied HDL-C levels (63 control patients and 76 patients with CAD confirmed by coronary angiography) without lipid-lowering therapy, anthropometric data, and laboratory tests was described previously [20]. Only male patients were included in control and CAD groups. The crude HDL preparations (pHDL) were prepared by the precipitation of apoB-containing lipoproteins with PEG 7000–9000 [6]. The concentrations of choline-containing phospholipids and total cholesterol in HDL preparations were measured by enzyme methods using Sentinel CH. SpA (lot 90398; ref: 17320, Milano, Italy) and HUMAN GmbH (lot 23005, ref 10028, Wiesbaden, Germany) kits, respectively, and apoA-I was measured by immunonephelometry with AU 480 (Beckman Coulter Inc, Brea, CA, USA).
ApoA-I content in pre
Urea-induced pHDL denaturation after incubation for 6 h at 25 °C was
followed by the accumulation of lipid-free pre
where
where F and B are the numbers of molecules of lipid-free apoA-I in water and bound to pHDL phospholipids, respectively [21]. The F/B ratio was calculated from the D parameter measured at 4.25 M urea (Eqn. 3):
Cholesterol efflux was measured with the murine macrophage cell line RAW 264.7, which is a standard cell culture for the measurement of ABCA1-mediated cholesterol efflux [15, 22]. The cell line was obtained from the European Collection of Cell Cultures and validated by speciation by DNA-bar sequencing of the COX subunit 1 gene and tested negative for mycoplasma. Efflux from cells to pHDL as a cholesterol acceptor was measured with a fluorescent probe, BODIPY-cholesterol. The basal and cAMP-stimulated effluxes were measured, and ABCA1-mediated efflux was calculated as the difference between stimulated and basal effluxes and expressed as a percent of effluxed cholesterol [6].
To separate the contributions of both pre
where Vi, Ki, and [Si] are the maximal
velocities, Michaelis constants and substrate concentrations for pre
For a series of lines in double reciprocal plots for a first substrate with various R values, the common intersection point does not depend on R, and abscissa and ordinate values are determined by Eqns. 6,7, respectively:
A common maximal velocity Vm at the constant R is determined by Eqn. 8:
In turn, the K2 value is derived from Eqn. 8 (Eqn. 9):
Vm varies within low V2 and high V1 borders. Generally, Vm non-linearly depends on R. The linear plots according to Eqn. 5 were constructed for fully denatured and intact pHDL sets, and the data were subsequently treated with Eqns. 6,7,8,9 to derive kinetic parameters. The V1 and K1 values were measured without S2 interference with pHDL fully denatured with 7.5 M urea. Before mixing, predenatured HDL was diluted 1:100 to lower urea concentration. The residual urea concentration not exceeding 75 mM did not influence efflux.
The expression of 65 genes involved in HDL metabolism and atherogenesis measured by us earlier by real-time PCR in peripheral blood mononuclear cells [20, 24] was compared to apoA-I dissociation from pHDL in the present study. Housekeeping genes included Glyceraldehyde-3-phosphate dehydrogenase (GAPDH), Lactate dehydrogenase A (LDHA), and Ribosomal protein L3 (RPL3) genes. The relative expression of all target genes was normalized by the geometric mean of three reference genes.
Statistica version 13 (TIBCO Software Inc. (http://tibco.com)) software was used
for statistical analysis. The associations between variables were analyzed by the
Spearman rank order correlation coefficient. The statistical significance limit
was accepted as p
The dissociation of apoA-I from pHDL induced by urea treatment and measured by
the D parameter significantly decreased with the increase of absolute
and apoA-I-normalized cholesterol and phospholipid (PL) levels in pHDL from CAD
patients (Table 1). The fit of dissociation data, passed through zero, to the
linear model of apoA-I distribution between lipid-free (F) and lipid-bound (B)
states (Eqn. 2) results in the values of the partition coefficient between
aqueous and lipid phases adjusted for water molarity K = 0.239
| Variable | Control | CAD |
| pHDL level | ||
| pHDL-C | –0.256 (0.043) | –0.366 (0.001) |
| pHDL-PL | n.s. | –0.336 (0.003) |
| pHDL-PL:apoA-I | n.s. | –0.329 (0.004) |
| pHDL-C:apoA-I | n.s. | –0.344 (0.002) |
| Gene expression | ||
| ABCA1 | 0.278 (0.029) | n.s. |
| APOA1 | –0.357 (0.005) | n.s. |
| TLR8 | 0.352 (0.005) | n.s. |
| PRKACB | n.s. | 0.250 (0.031) |
| CUBN | n.s. | –0.305 (0.008) |
| HMGCR | n.s. | 0.274 (0.017) |
| PLTP | n.s. | 0.242 (0.036) |
| ALB | n.s. | –0.280 (0.015) |
| CD36 | n.s. | 0.259 (0.025) |
| SLPI | n.s. | 0.294 (0.011) |
| OLR1 | n.s. | 0.328 (0.004) |
| IL1R1 | n.s. | –0.260 (0.024) |
| HDL function | ||
| Chol efflux | –0.305 (0.015) | –0.300 (0.009) |
Spearman rank order correlation coefficients R with significance level (p) are given for control (n = 63) and CAD (n = 75) patients. n.s. – nonsignificant. CAD, coronary artery disease; HDL, high-density lipoproteins; pHDL, HDL preparations.
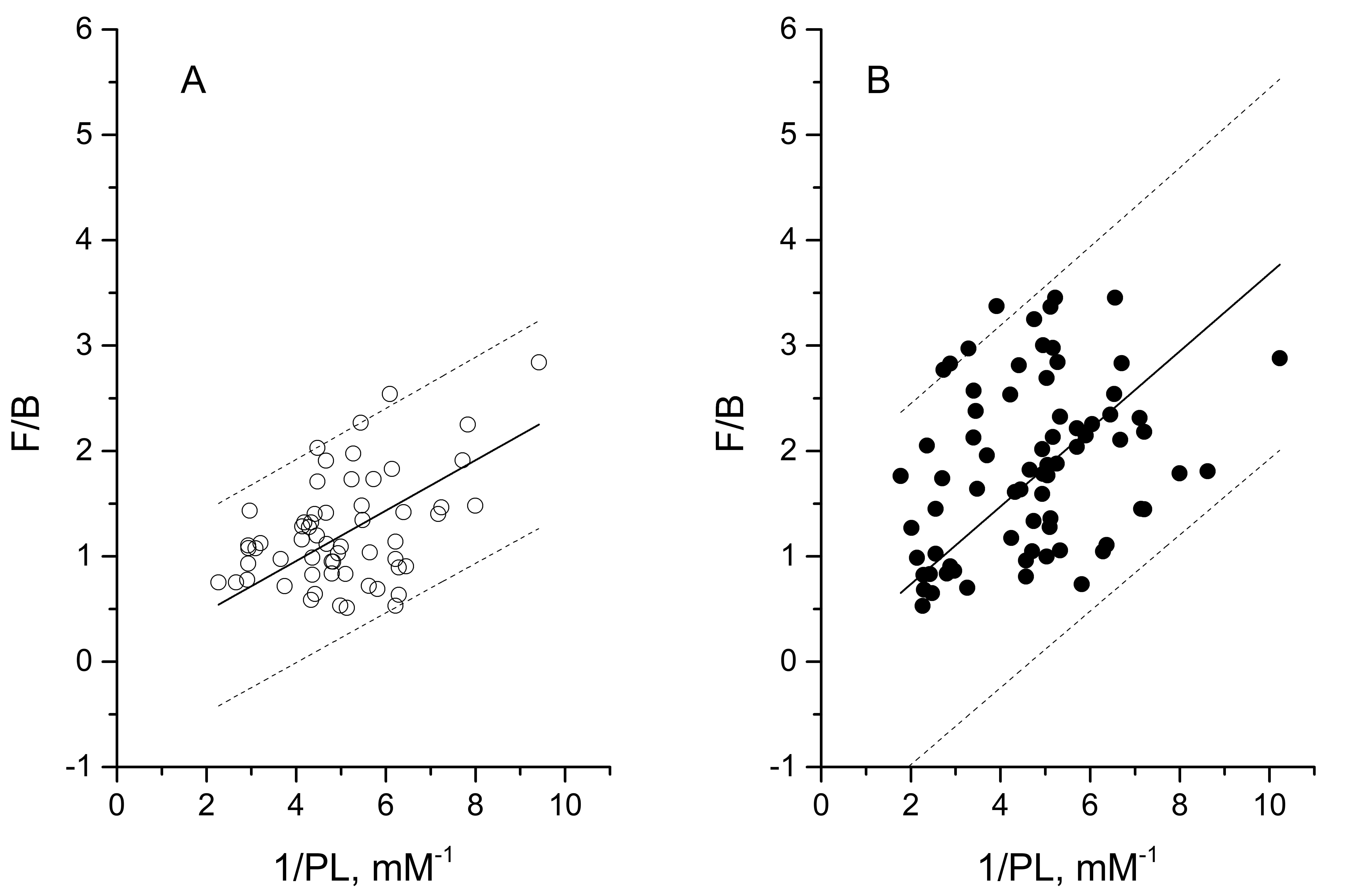 Fig. 1.
Fig. 1.
ApoA-I distribution between water and lipid phases of pHDL
predenatured by 4.25 M urea. (A) pHDL from control patients (n = 59). (B) pHDL
from CAD patients (n = 73). The 95% prediction bands are given as thin dashed
lines. The Y-intercepts passed through zero according to the distribution model.
Slopes were 0.239
The similar values for free energy change have been obtained for the exchange of apoA-I variants between discoidal HDL-bound and lipid-free pools (1–3 kJ/mol) [25] and the lipid transfer for nanodisks consisting of apoA-I and DMPC (1.7 kJ/mol) [26]. ApoA-I in HDL from CAD patients possesses higher free energy and, thus, exhibits a higher dissociation. Of note, the increased PL:apoA-I and Chol:apoA-I ratios for pHDL from CAD patients relative to controls were measured throughout the whole range of HDL-C concentration (data not shown). To reveal the associations between apoA-I dissociation and lipoprotein metabolism, the expression of 65 genes in peripheral blood mononuclear cells, sensitive to HDL metabolism and atherogenesis [20, 24], was analyzed. The significant correlations between D and gene expression are included in Table 1. For control patients, the positive correlations of the D parameter with the ABCA1 and TLR8 expression levels were revealed. For patients with CAD, the positive (for PRKACB, HMGCR, PLTP, CD36, SLPI, and OLR1) and inverse (for CUBN, ALB, and IL1R1) associations of gene expression in peripheral blood with the degree of apoA-I dissociation were revealed.
First, the efflux measurements were done with intact pHDL as cholesterol acceptors. Both for control and CAD patients, there were significant inverse correlations between apoA-I dissociation and ABCA1-mediated cholesterol efflux (Table 1).
Second, due to the involvement of lipid-free and lipid-bound apoA-I in
cholesterol transport by the ABCA1 transporter, the decrease of efflux efficiency
in CAD may be associated with the less efficient efflux to each or both
acceptors. To separate individual contributions, the functionality of lipid-free
apoA-I was studied (Fig. 2). To induce apoA-I dissociation from the HDL lipid
phase to the water phase, pHDL from control and CAD patients were denatured for 6
h at 25 °C by 4.25 M urea at the middle of the denaturation transition.
The ABCA1-mediated efflux for 2 h to pHDL, at fixed or serial dilutions, was then
measured, and the data were fitted to Michaelis-Menten kinetics. The kinetic
parameters are given in Table 2. The concentration of lipid-free
pre
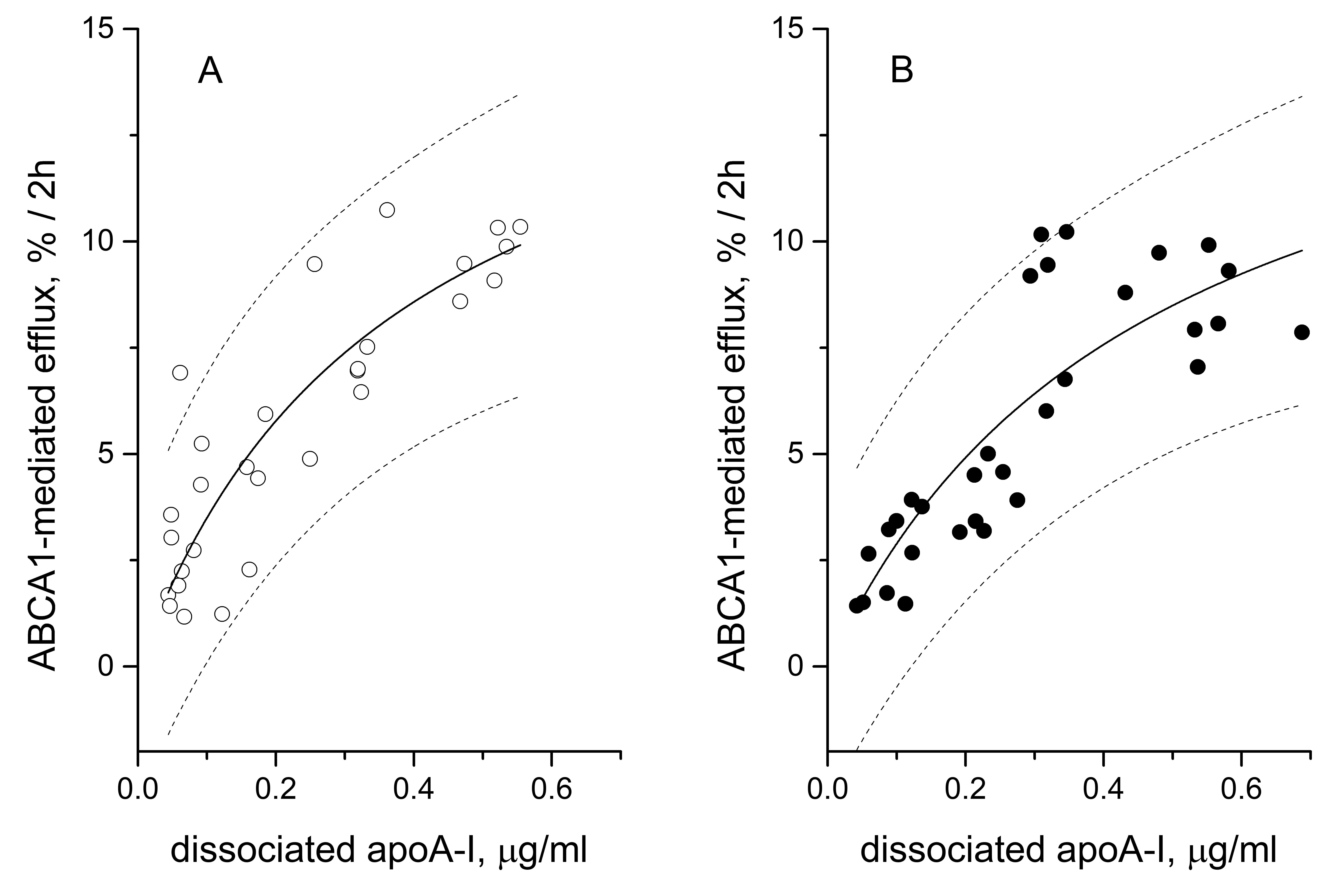 Fig. 2.
Fig. 2.
The kinetics of ABCA1-mediated efflux to pHDL predenatured by 4.25 M urea. (A) pHDL from control patients (n = 29). (B) pHDL from CAD patients (n = 31). The 95% prediction bands are given as thin dashed lines.
| Group | Urea, M | n | Vm, %/2 h | Km, µg/mL | Vm/Km |
| Control | 4.25 | 29 | 16.6 |
0.38 |
44.2 |
| 4.25 | 5 | 16.8 |
0.69 |
24.4 | |
| 7.50 | 5 | 15.0 |
0.74 |
20.3 | |
| CAD | 4.25 | 31 | 16.5 |
0.47 |
35.0 |
| 4.25 | 4 | 18.9 |
0.61 |
30.8 | |
| 7.50 | 4 | 17.7 |
0.62 |
28.3 |
Mean
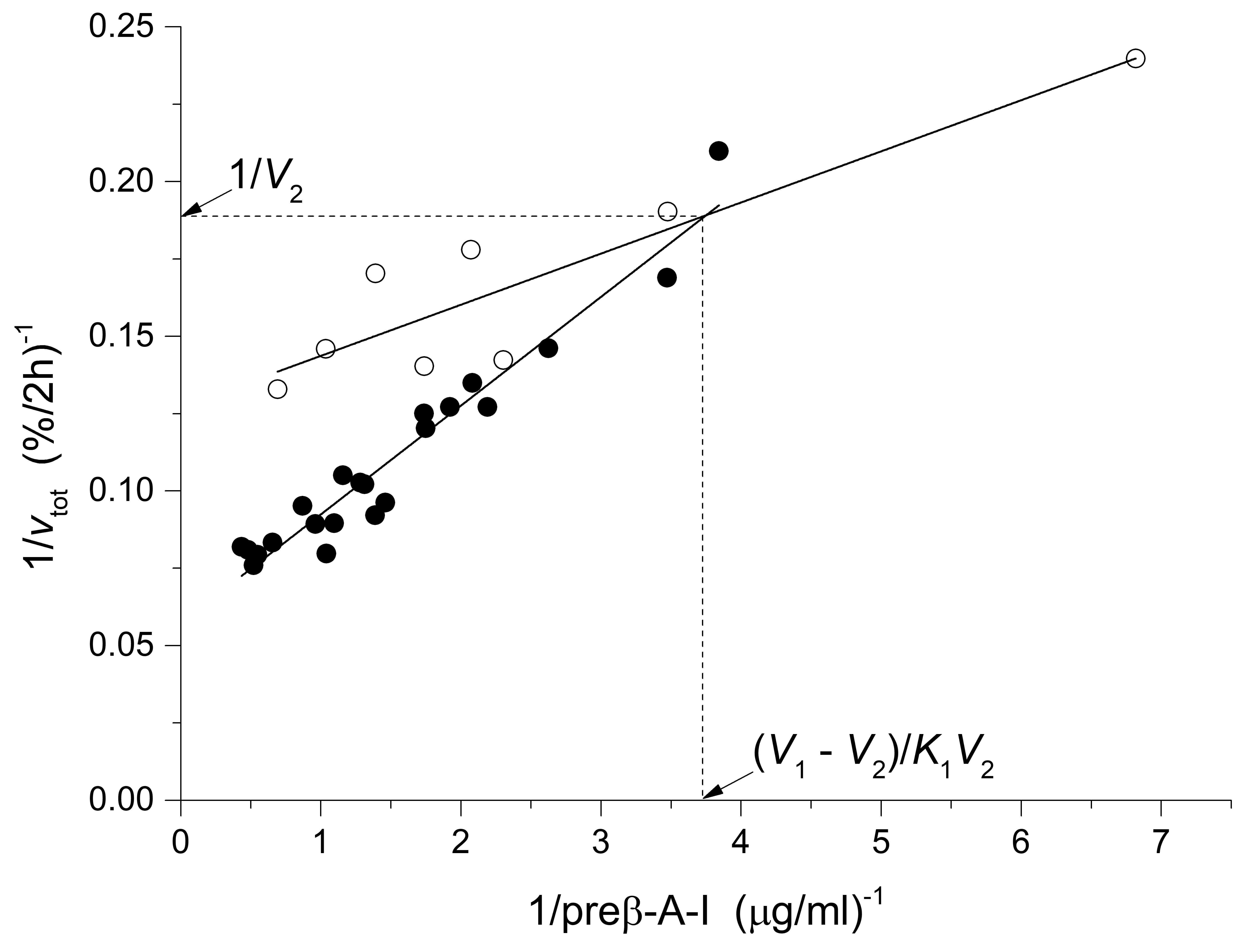 Fig. 3.
Fig. 3.
The dependency of the total initial rates of ABCA1-mediated
cholesterol efflux vtot on S1 concentration in double
reciprocal plots for intact and denatured pHDL from the CAD group with low plasma
HDL-C levels. The data were fitted to a linear function 1/vtot =
intercept + slope
We used PEG 7000–9000 to prepare crude HDL for subsequent cholesterol efflux
measurements. ApoB depletion by PEG is the most frequent approach in such studies
[15, 22, 27] due to quick and efficient plasma/serum fractionation with
preservation of HDL integrity. To avoid possible contamination by apoB-containing
aggregated lipoproteins, we discarded the thin surface layer after the
centrifugation step, if present. Importantly, the procedure, if performed at room
temperature, neither involves apoA-I dissociation [28] nor an overall efflux
change [29]. These properties are crucial in the preservation of individual
efflux steps with lipid-free apoA-I and
There are three major findings in the present study revealed by the comparison of compositional and functional properties of HDL from CAD and control patients.
First, for HDL from CAD patients, apoA-I partitioned more into the water phase during urea-induced HDL denaturation. ApoA-I dissociation is determined by the equilibrium [7, 31] and/or kinetic [32] nature of HDL stability. The first condition implies apoA-I distribution between lipid and water phases by mass law action, while the second condition includes apolipoprotein shedding from the HDL surface induced by metabolic transformation. Metabolic remodeling that includes the transitions between different lipoprotein structures is accompanied by the apolipoprotein A-I dissociation from the HDL surface and particle fusion; lipolytic and transfer activities are the most important. CETP- and HL-induced dissociation of lipid-free apoA-I from HDL with the subsequent formation of discoidal HDL occurs in the presence of VLDL or LDL [33, 34]. This apoA-I dissociation also occurs at HDL thermal or chemical denaturation [6, 21, 32, 35]. The chaotropic agent-induced apoA-I dissociation is irreversible [21], and HDL denaturation induces apoA-I unfolding and partitioning from HDL to water (Fig. 1). Partitioning also occurred in the absence of denaturants [7] that may result in the appearance of lipid-free apoA-I as a principal ligand for the ABCA1 transporter. The spontaneous exchange of apoA-I between lipid-free and lipid-bound forms [8, 36] differs from dissociation, as no net transfer occurs in the exchange. Irrespective of the nature of dissociation, this process results in the existence of up to 10% of total plasma apoA-I in lipid-free form.
Distribution parameter D was inversely associated with the absolute and apoA-I-normalized levels of choline-containing phospholipids and cholesterol in pHDL, opposite to the lack of any association for pHDL from control patients. The increased denaturation of HDL from patients with acute coronary syndrome has been described [35]. Importantly, HDL particles from CAD patients are more enriched with PL and cholesterol normalized by apoA-I level throughout the whole range of HDL-C. The enrichment of HDL with cholesterol, with the concomitant increase of competition between apoA-I and cholesterol for the binding to phospholipid molecules adjacent to apoA-I, is suggested to be involved in the increased apolipoprotein dissociation from HDL in CAD. Cholesterol molecules have been shown to be partially excluded from boundary lipids in reconstituted HDL [37]. The free cholesterol molecules may increasingly accumulate in the vicinity of apoA-I molecules in cholesterol-overloaded pHDL from CAD patients with the additional perturbation of PL structural order and dynamics. What could be a driving force for additional cholesterol accumulation in HDL from CAD patients compared to control patients? Earlier, we measured the upregulated expression of CETP, LPL, and PLTP genes and the downregulated expression of the LCAT gene among 65 genes selected to follow atherogenesis-prone changes in CAD [20, 24]. The differential expression of these genes is suggested to be involved in lipid enrichment of HDL in CAD. Indeed, the enrichment of HDL with phospholipid and cholesterol molecules in CAD could occur due to a decreased phospholipid hydrolysis by LCAT, which possesses phospholipase activity with a concomitant lowering of cholesteryl ester generation. PLTP is capable of transforming small into large HDL particles with a concomitant release of apoA-I [5]; the effect of upregulated expression of the pLTP gene lies in accordance with the increase of apoA-I-normalized cholesterol and phospholipid levels in CAD. The entropically favorable processes of both apoA-I exchange between discoidal HDL-bound and lipid-free pools [25] and the lipid transfer for nanodisks consisting of apoA-I and DMPC [26] with the increased activation entropy and the decrease in the standard entropy have been suggested. The exact contribution of entropy and enthalpy to apoA-I increased dissociation from mature spherical HDL in CAD remains to be determined. The different exposure of the hydrophobic amino acid residues of apoA-I to the aqueous phase in the activated state at apolipoprotein denaturation [21] may be involved. Interestingly, cholesterol in discoidal HDL increased the number of apoA-I tryptophan residues accessible to the aqueous phase but decreased their mean degree of hydration [38].
Second, we revealed significant associations between the apoA-I dissociation parameter and expression of several genes (Table 1). These associations may reveal proatherogenic or atheroprotective effects, and any causal links remain to be separately studied. For HDL from control patients, the D parameter was positively correlated with the ABCA1 and TLR8 expressions. The decrease of lipid-free apoA-I with the increase of ABCA1 activity is suggested to result in the increase of dissociation of lipid-bound apolipoprotein. Of note, the increased apoA-I dissociation seems to possess a proatherogenic effect in control patients due to the positive associations between the D parameter and TLR8 gene expression, which is involved in inflammation [39].
For HDL from CAD patients, the D parameter was associated with the expression of
a number of genes involved in lipoprotein metabolism (PRKACB, HMGCR,
PLTP, OLR1, CUBN, and ALB) and systemic inflammation (CD36,
SLPI, and IL1R1). Interestingly, the expression of apoptosis-related
genes IL1R1 and PRKACB with the inverse associations with the D
parameter has also been regulated inversely in acute myocardial infarction [40].
The observed inverse associations of the D parameter with CUBN
and ALB gene expression may be associated with the increased catabolism
of lipid-free apoA-I and the accompanying decrease of the pre
Third, lipid-free/lipid-poor apoA-I that may originate from the HDL-bound state,
both for CAD and control HDL, seems to be equally efficient in ABCA1-mediated
efflux. The two dependencies in the double reciprocal plot intersected in the
upper right corner (Fig. 3), which is definite evidence of competition of two
substrates for a single enzyme [23] with the relation V1 (pre
The decreased ABCA1-mediated efflux efficiency of intact HDL as a mixture of
lipid-free and lipid-bound apoA-I in CAD has been described [17, 18]. Despite the
attempts to normalize efflux to pre
Limitations include modest sample size, use of only RAW 264.7 murine macrophages, restriction of urea-induced denaturation as an in vitro model, and causal contribution of apoA-I dissociation to CAD development.
For CAD patients, apoA-I partitions more into the water phase at urea-induced
HDL denaturation due to the higher free energy of apolipoprotein in the lipid
phase with the negative correlations of the dissociation parameter with absolute
and normalized per apoA-I cholesterol and phospholipid levels in HDL particles.
For control patients, the dissociation of apoA-I in the water phase is associated
with ABCA1 and TLR8 gene expression. The upregulated expression
of CETP, LPL, and PLTP genes and the downregulated
expression of the LCAT gene in CAD are suggested to result in the lipid
enrichment of HDL. PLTP gene expression may underlie the increased
apoA-I dissociation in CAD. The total initial rates of ABCA1-mediated cholesterol
efflux from RAW 264.7 macrophages, when pre
All raw data reported in this paper will also be shared by the lead contact upon request.
ADD – concept of the study; writing text of the paper; supervision of the study; MAP – patient selection, coronary angiography, laboratory analysis, and search references; VBB – conducting experiments; preparation of figures and tables. ADD and VBB – analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The project was approved by ethics committee of the M.F. Vladimirsky Moscow Regional Research and Clinical Institute MONIKI (protocol no. 12479/2019, February 17, 2019). All involved patients provided voluntary informed consent to participate in the study.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



