1 Department of Cardiology, Affiliated Hospital of Inner Mongolia Minzu University, 028002 Tongliao, Inner Mongolia, China
2 Key Laboratory of Basic Pharmacology of Ministry of Education, Zunyi Medical University, 563000 Zunyi, Guizhou, China
Abstract
Luteolin, a natural flavonoid, is an active ingredient in traditional herbs used to treat cardiovascular diseases. However, little is known about the effects of luteolin on oxidative damage in cardiomyocytes, and the underlying mechanisms remain poorly understood. Therefore, this study aimed to investigate the protective effects of luteolin against hydrogen peroxide (H2O2)-induced mitophagy and apoptosis in cardiomyocytes.
H9c2 cells were exposed to H2O2 for 4 h, which caused severe cellular damage accompanied by apoptosis. The protein expression of β-actin, FK506 binding protein 12, mammalian target of rapamycin (mTOR), acetyl-coenzyme A carboxylase alpha (ACC), sirtuin (silent mating type information regulation 2 homolog) 1 (S. cerevisiae) (SIRT1), peroxisome proliferator-activated receptor gamma, coactivator 1-alpha (PGC-1α), autophagy-related 5 homolog (S. cerevisiae) (ATG5), microtubule-associated protein 1 light chain 3 beta (LC3B), B-cell lymphoma 2 (BCL2)-associated X protein (Bax), B-cell leukemia/lymphoma 2 (Bcl-2), PTEN-induced putative kinase 1 (PINK1), and peroxisome proliferator-activated receptor gamma (PPARγ) was analyzed by Western blotting. Intracellular reactive oxygen species (ROS) levels were assessed using 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) staining (DCFH) oxidation staining. The expressions of phosphorylated AMP-activated protein kinase alpha (p-AMPKα), SIRT1, and caspase 8 were evaluated by immunofluorescence. Mitochondrial membrane potential and mitochondrial permeability transition pore (MPTP) opening were assessed using 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolocarbocyanine iodide (JC-1) staining and an MPTP assay kit, respectively.
H2O2 treatment significantly reduced the viability of H9c2 cardiomyocytes and induced mitochondrial apoptosis. Furthermore, H2O2 upregulated the expression of p-AMPK, SIRT1, mTOR, ACC, and PGC-1α, while downregulating PPARγ expression. Concurrently, H2O2 activated mitophagy, suggesting involvement of the AMPK/mTOR signaling pathway. Notably, pretreatment with luteolin effectively reversed these H2O2-induced alterations by attenuating excessive ROS production, inhibiting MPTP opening, and normalizing the Bcl-2/Bax ratio and caspase 8 expression. Additionally, luteolin suppressed the H2O2-induced upregulation of proteins associated with the AMPK/mTOR signaling axis, mitophagy, and apoptosis.
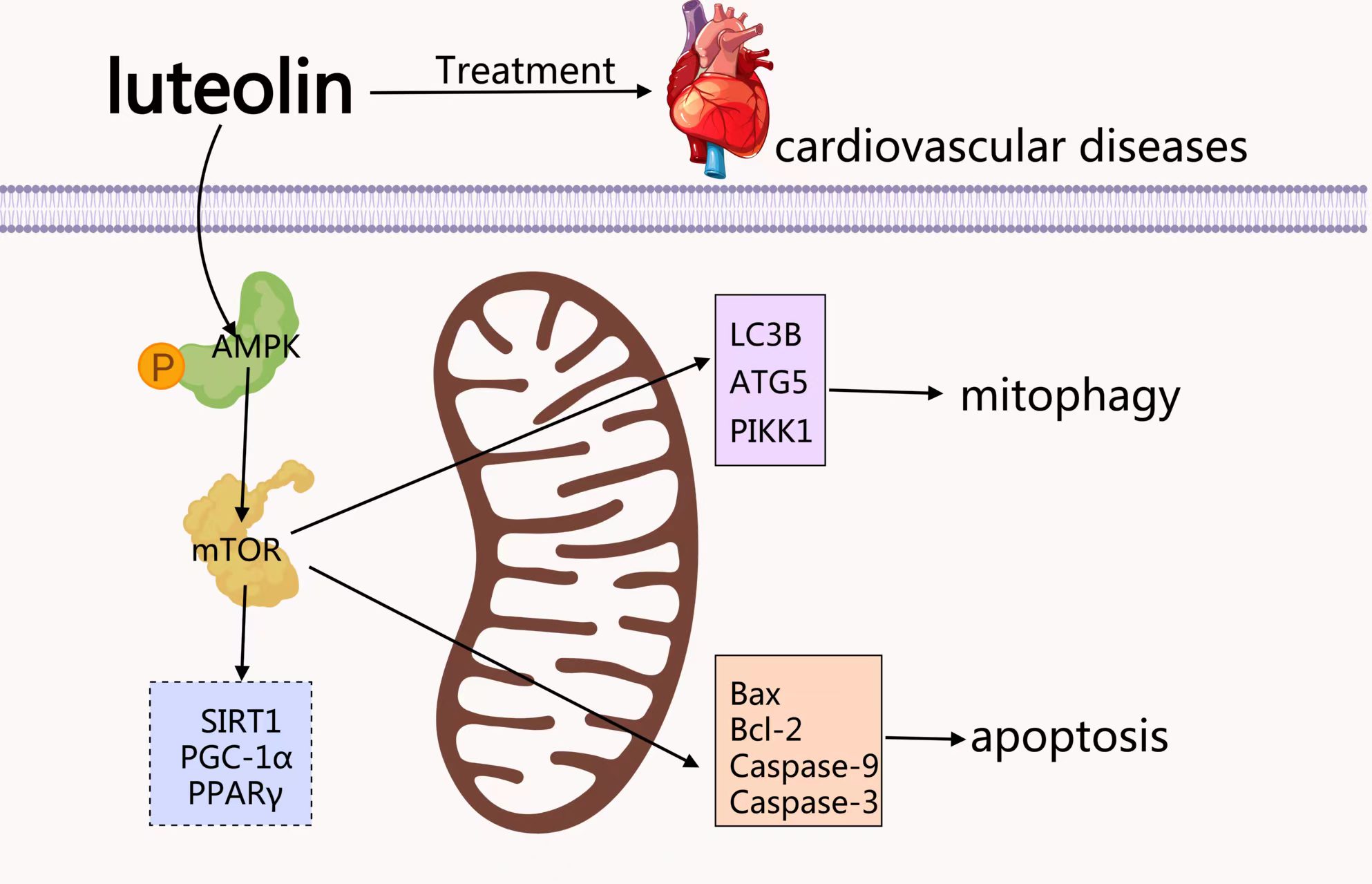
These findings suggest that luteolin protects H9c2 cells from mitochondria-mediated apoptosis by modulating the AMPK/mTOR signaling pathway and inhibiting excessive mitophagy. Moreover, these results suggest that luteolin has potential as a therapeutic agent for preventing and treating cardiovascular diseases.
Graphical Abstract
Keywords
- luteolin
- mitophagy
- reactive oxygen species
- cardiovascular diseases
Cardiovascular disease (CVD) is a group of diseases that entail the heart or blood vessels. CVD contains coronary artery disease, such as angina pectoris and myocardial infarction. At the cellular level, shortly after ischemia or hypoxia, there is a rapid decrease in the amount of the energy molecule adenosine triphosphate (ATP) in cardiomyocytes. If ischemia continues, cytochrome c released from mitochondria triggers apoptosis, ATP depletion following increased mitochondrial permeability, and ultimately necrosis of cardiomyocytes [1, 2]. Oxidative damage to proteins, lipids, and nucleic acids and mitochondrial membrane infiltration is due to excessive accumulation of reactive oxygen species (ROS), which leads to cell death [3]. Excess ROS in turn causes damage to mitochondria and may trigger mitophagy. As an active and highly oxygen-consuming organ, the heart is susceptible to oxidative stress (OS), leading to cell necrosis and apoptosis [4, 5]. O2⁻ is the ROS initially involved in mitophagy induced by glucose, glutamate, pyruvate, or serum loss. In the presence of superoxide dismutase (SOD), superoxide is rapidly dismutated into hydrogen peroxide (H2O2). Compared to O2⁻, H2O2 exhibits greater membrane permeability and acts as a key signaling molecule in redox homeostasis, but its accumulation can also lead to oxidative damage [6]. H2O2 plays an instrumental factor in cellular oxidation-reduction equilibrium and signal transduction due to its better membrane permeability. H2O2 reacts with transition metal ions such as Fe2+ to generate OH, a highly reactive and destructive ROS [7]. Upregulation of OS markers has been shown to predict cardiovascular disease [8]. Therefore, inhibition of OS and cardiomyocyte apoptosis is of great importance for the treatment and prevention of CVD.
Antioxidants have been receiving attention as a new therapeutic strategy for
CVD. Luteolin (Lut) is widely distributed in nature and is a natural flavonoid
compound that can be isolated from a variety of natural herbs, vegetables, and
fruits, with a higher content of whole-leaf cymbidium orchids, chili peppers,
wild chrysanthemums, honeysuckle, and perilla. Lut exhibits various
pharmacological activities, including anti-neoplastic [9, 10], cardiovascular
[11, 12], anti-inflammatory [13], diabetic [14], hepatoprotective [15],
antioxidant [16], and neurological disorders [17]. In addition, Lut inhibits
apoptosis in I/R-injured rat cardiomyocytes by regulating the Toll-like receptor
4 (TLR4)/nuclear factor kappa-B (NF-
Mitophagy is a particular type of autophagy, which is the process of cells breaking down organelles and pathogens that are invading them. Mitophagy is the depolarization of intracellular mitochondria in response to stimuli from internal and external environments, such as ROS, nutrient deficiencies, cellular senescence, etc. Autophagosomes can specifically phagocytose damaged mitochondria and fuse with lysosomes to cause degradation of damaged mitochondria, thus maintaining the stability of the intracellular environment [19]. The mitochondrial marker PTEN induced putative kinase 1 (PINK1) and the microtubule-associated protein 1 light chain 3 beta (LC3B) are usually co-localized in studies to show mitophagy.
It is well known that AMPK and mTOR regulate cellular catabolism and biosynthesis processes through different mechanisms, which together maintain cellular energy balance and metabolic stability [20].
Although attempts have been made to study the pharmacological activity of Lut against CVD, the molecular mechanisms between apoptosis, oxidative stress mitophagy, and Lut remain to be investigated. Elucidating the correlation between these processes may be a mechanism for the treatment of CVD. The paper aimed to demonstrate the molecular mechanism by which Lut affects cardiovascular disease.
Lut (purity
H9c2 cells were cultured in DMEM supplemented with 10% (v/v) FBS and 1% (v/v) penicillin-streptomycin, placed in an incubator at 37 °C with 95% humidity and 5% CO2. Lut (5, 10, and 20 µM) and 5 µM metoprolol treated cells for 24 h. Then cells were divided into control group, H2O2-induced model group, Lut-treated group, and metoprolol-treated group. All groups except the normal group were treated with H2O2 (700 µM) for 4 h. All cell lines were validated by species-specific PCR and tested negative for mycoplasma.
H9c2 cells in the logarithmic growth phase were selected, washed with PBS,
digested with trypsin, and at the end of centrifugation, they were prepared into
cell suspension. Cells were counted under an automatic cell counter (Life
Technologies, USA) using a cell counting plate. They were sequentially inoculated
in 96-well culture plates according to 1.0
H9c2 cells were seeded in 96-well black clear-bottom plates at a density of 1.0
Treat the cells according to the method under 2.3, 1 mL of 1
Treat the cells according to the method under 2.3, add Calcein AM staining solution according to the instructions of the MPTP Assay Kit, and incubate for 30 min at 37 °C in an incubator away from light. At the end of the incubation, replace with fresh pre-warmed culture medium at 37 °C and incubate at 37 °C away from light for 30 min. Then, Cells with the addition of detection buffer were observed under a fluorescence microscope. The fluorescence intensity was quantified using ImageJ software (NIH, USA) and expressed as Mean Fluorescence Intensity.
First, cells were fixed by adding 4% paraformaldehyde for 1 hour at 4 °C; then cells were incubated with 0.3% Triton X-100 (PBS preparation) for 15 minutes and incubated with 5% BSA for 30 minutes. The corresponding primary antibody was added to each well, incubated overnight at 4 °C, and the secondary antibody was added the next day. Finally, incubate with DAPI for 5 minutes and observe under a fluorescence microscope.
Cells were lysed with RIPA cell lysis buffer containing 1% PMSF, placed on ice for 30 min, and centrifuged at 12,000 g and 4 °C for 10 min. Protein samples were prepared based on protein concentration by adding the up-sampling buffer and boiling for 5 min. Samples were separated by 10% SDS-PAGE and transferred to a PVDF membrane, which was blocked with 5% skimmed milk for 1.5 h. The membrane was incubated with primary antibody, overnight for 4 °C, followed by incubation with HRP-conjugated secondary antibody for 1.5 h. Finally, the protein bands were exposed to the ECL ultrasensitive luminescent developer.
Obtain the 2D structure of the small molecule by Pubchem
(https://pubchem.ncbi.nlm.nih.gov/) and convert it to a mol2 file in Chemdraw3D
for backup; query the database in PDB (https://www.rcsb.org/) to obtain the live
3D structure file of the target, such as AMPK
Data were processed and statistically analyzed using GraphPad Prism 8.0 software
(GraphPad Software, San Diego, CA, USA). The normality of data distribution was
assessed using the Shapiro–Wilk test. For comparisons between two groups,
statistical significance was determined using a two-tailed Student’s
t-test. For multiple group comparisons, one-way analysis of variance
(ANOVA) was employed, followed by Tukey’s post hoc test for pairwise comparisons
among all groups, or Dunnett’s post hoc test when comparing multiple treatment
groups against a single control group. All experiments were independently
repeated at least three times, and data are presented as mean
As the concentration of H2O2 increased in Fig. 1A, the cell survival rate gradually decreased. For comprehensive consideration, H2O2 with a moderate degree of cellular damage was chosen to establish a cardiomyocyte injury model by treating with H2O2 at 700 µmol/L for 4 h. As shown in Fig. 1B, the optimal dosing concentration was screened from Lut at concentrations of 5, 10, and 20 µM. As shown in Fig. 1C, Lut/metoprolol (5 µM) pretreatment showed significant protection against H2O2-induced Cytotoxicity.
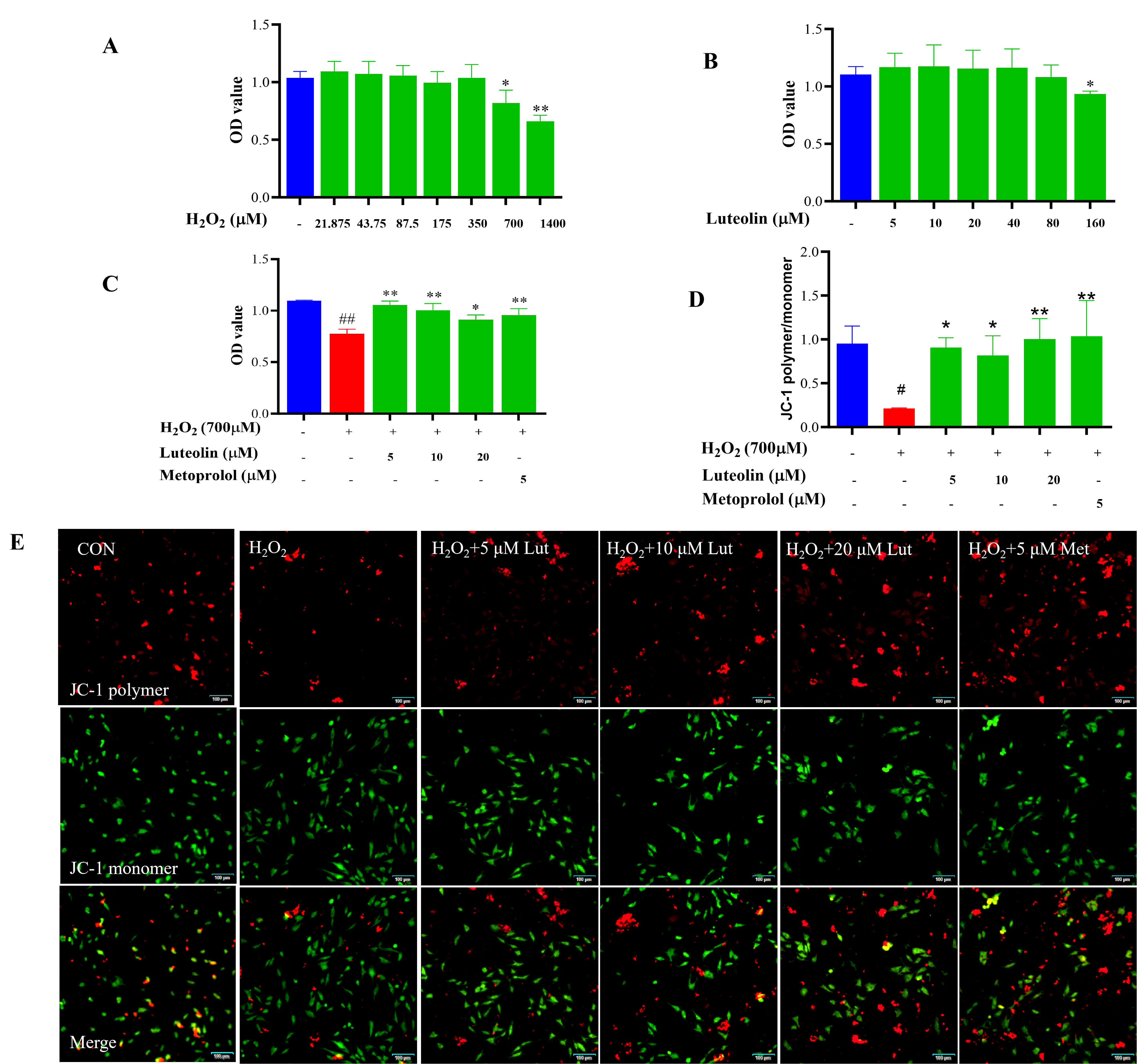 Fig. 1.
Fig. 1.
Effect of Lut on cell survival rate and H2O2-induced
cytotoxicity. H9c2 cells were treated with 700 µM H2O2 for 4 h
and incubated with/without Lut/metoprolol for 24 h. (A) Effect of H2O2
on Cell Survival Rate. (B) Effect of Lut on Cell Survival Rate. (C) Effect of Lut
on H2O2-Induced Cytotoxicity. (D,E) Effect of Lut on
H2O2-Induced cellular MMP by JC-1. Scale bar = 100 µm (applies to
all panels in E). *vs MOD, *p
Decrease in Mitochondrial Membrane Potential (MMP) causes early apoptosis, and in this study, JC-1 fluorescent probe was used to detect MMP in cells. Compared with the control group, the red/green fluorescence ratio of the cells in the model group was decreased, i.e., MMP was decreased; compared with the model group, the red/green fluorescence ratio of the cells in the Lut group was increased, i.e., MMP levels were increased, as shown in Fig. 1D and Fig. 1E.
ROS generated by H2O2 could promote cellular damage. Compared with the
control group, intracellular DCFH-DA probe green fluorescence signal was enhanced
and intracellular ROS levels were elevated in the model group, as shown in Fig. 2A. The intracellular ROS levels were reduced in the Lut (5, 10, 20 µM)
group compared with the model group. Elevated levels of ROS in mitochondria
ultimately lead to the release of cytochrome C and a decrease and loss of MMP. As
shown in Fig. 2B, the fluorescence intensity became weaker in the model group
compared with the control group and increased significantly after treatment with
Lut (p
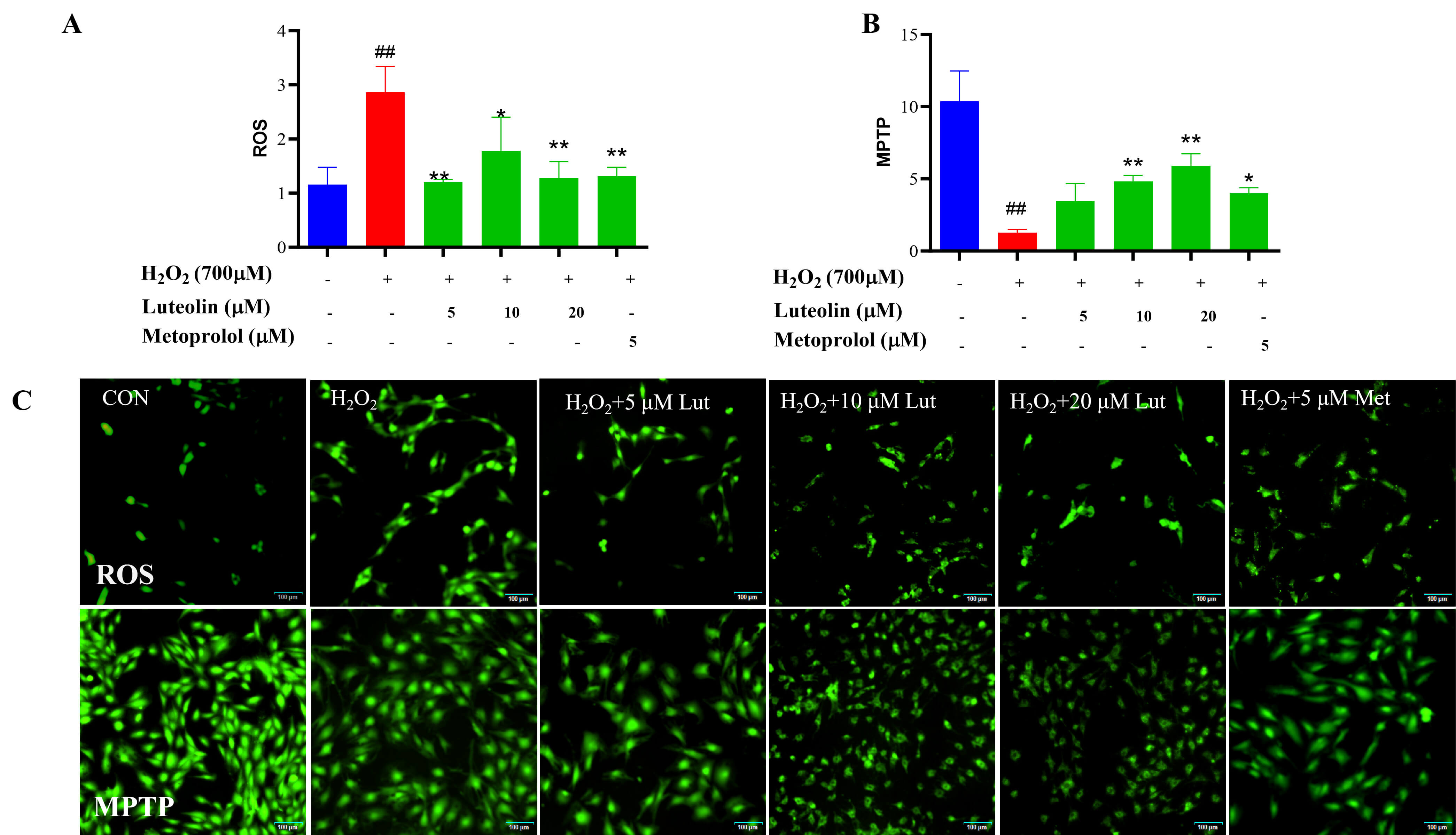 Fig. 2.
Fig. 2.
Effect of Lut on H2O2-induced Apoptosis. H9c2 cells
were treated with 700 µM H2O2 for 4 h and incubated with/without
Lut/metoprolol for 24 h. (A) The effect of Lut on intracellular ROS levels
induced by H2O2. (B) The effect of Lut on H2O2-induced
intracellular MPTP levels. (C) Representative fluorescence images of ROS and MPTP
under different treatment conditions. Scale bar = 100 µm (applies to all
panels in C). * vs MOD, *p
Apoptosis caused activation of the Bcl-2/BAX and Caspase protein families. In this study, the expression of Bcl-2/BAX and Caspase8 proteins was examined. After the cells were treated with H2O2, the ratio of Bcl-2/BAX was decreased and Caspase8 was enhanced, but pretreatment with Lut, the ratio of Bcl-2/BAX was elevated and the expression of Caspase8 was down-regulated (Fig. 3).
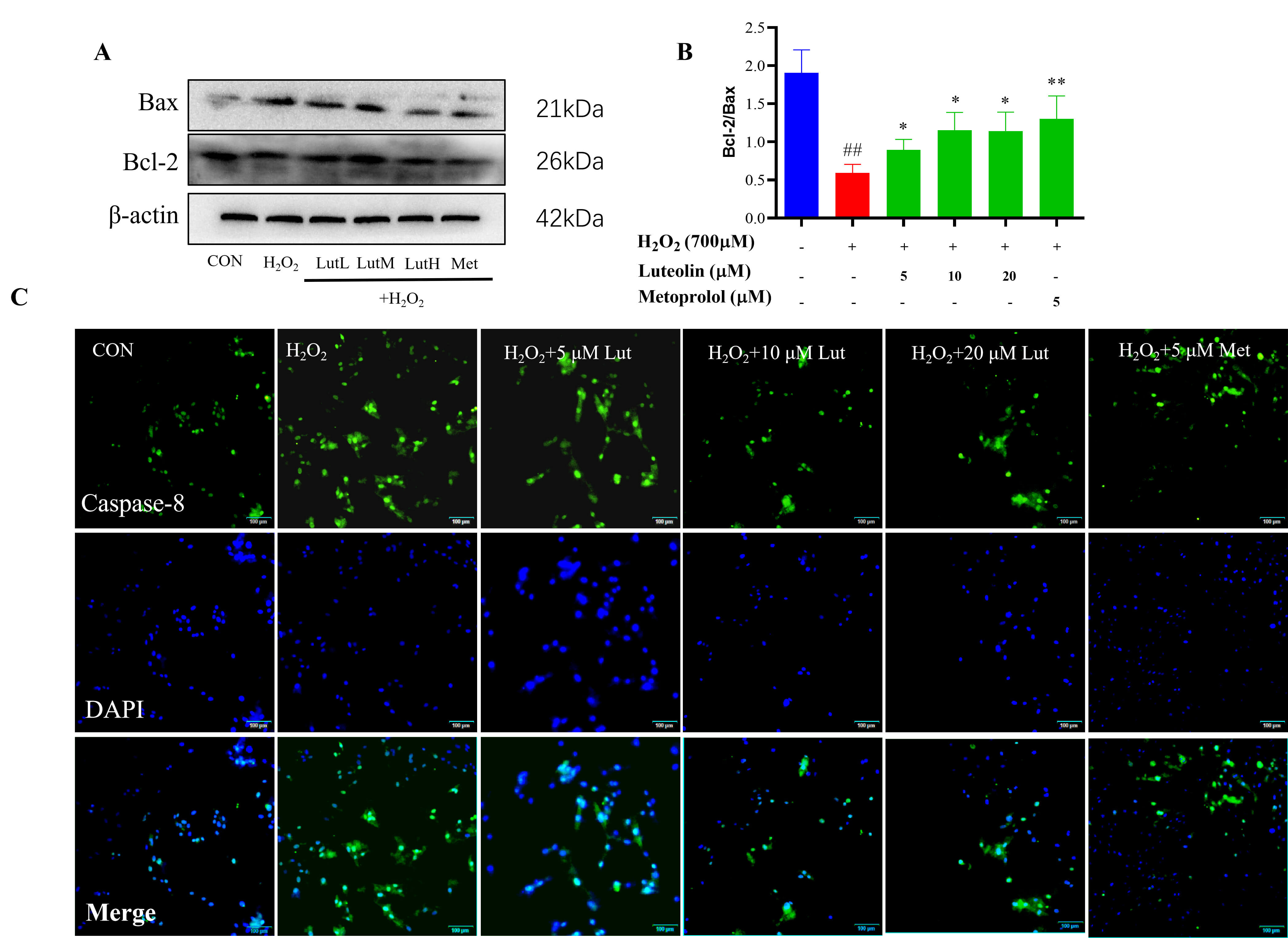 Fig. 3.
Fig. 3.
Effect of Lut on H2O2-induced Apoptosis. H9c2 cells
were treated with 700 µM H2O2 for 4 h and incubated with/without
Lut/metoprolol for 24 h. (A,B) The expression of Bcl-2/BAX was analyzed by
Western blot. (C) The expression of Caspase8 was analyzed by immunofluorescence.
Scale bar = 100 µm (applies to all panels in C. *vs MOD, *p
We further elucidated the mechanism by which Lut attenuates H2O2-induced apoptosis, by examining the mitophagy signaling pathway. Exposure to H2O2 increased the protein level of mTOR and activation of mitophagy related expression of ATG5, PINK1, and LC3B. We interpreted this overactivation as a detrimental, pro-death response that contributes to H2O2-induced cardiomyocyte injury. Treatment with Lut reversed these changes, significantly decreasing the levels of ATG5, PINK1, and LC3B-II. This suggests that the protective effect of Lut is associated with suppressing this pathological overactivation of mitophagy. The involvement of the mTOR pathway was also evident, as H2O2 increased mTOR expression, which was restored by Lut treatment, aligning with the known role of the mTOR in stress-induced mitophagy (Fig. 4).
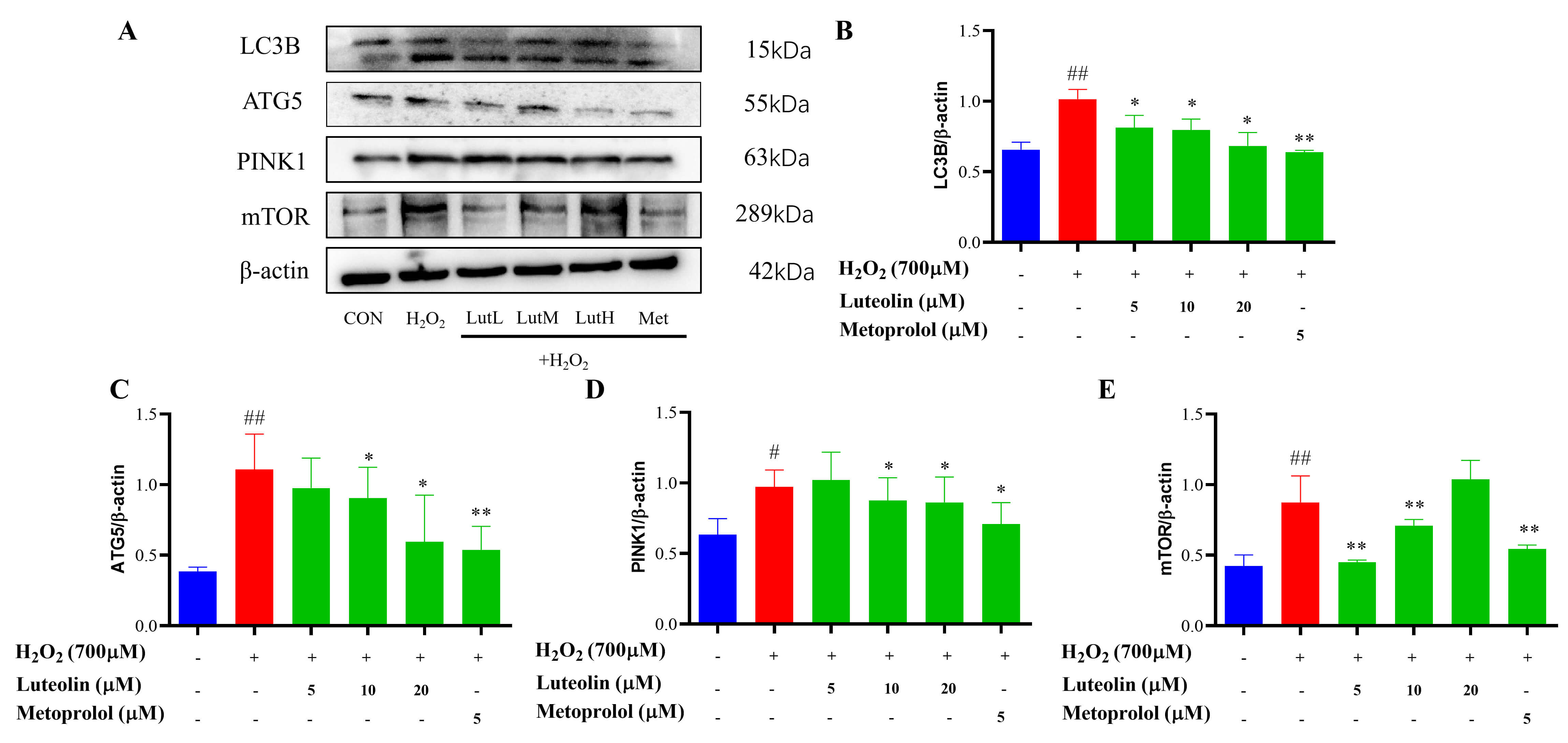 Fig. 4.
Fig. 4.
Effects of Lut on the expression of mTOR and mitophagy. H9c2
cells were treated with 700 µM H2O2 for 4 h and incubated
with/without Lut/metoprolol for 24 h. (A–E) The expression of mTOR, ATG5, PINK1,
and LC3B was analyzed by Western blot. *vs MOD, *p
In this study, Lut decreased the levels of p-AMPK
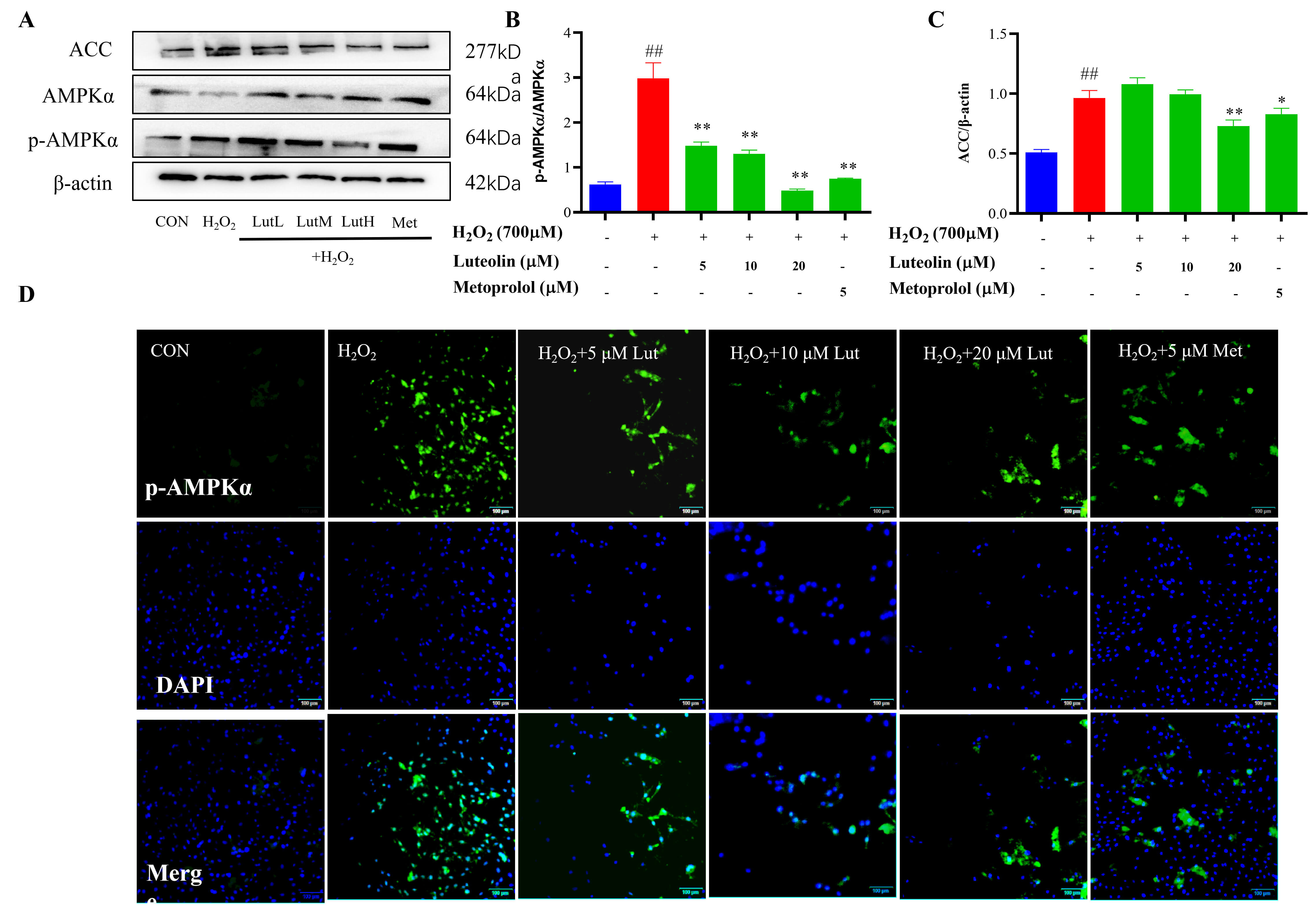 Fig. 5.
Fig. 5.
Effects of Lut on the expression of AMPK/ACC signaling pathway.
H9c2 cells were treated with 700 µM H2O2 for 4 h and incubated
with/without Lut/metoprolol for 24 h. The expression of
The levels of PPAR
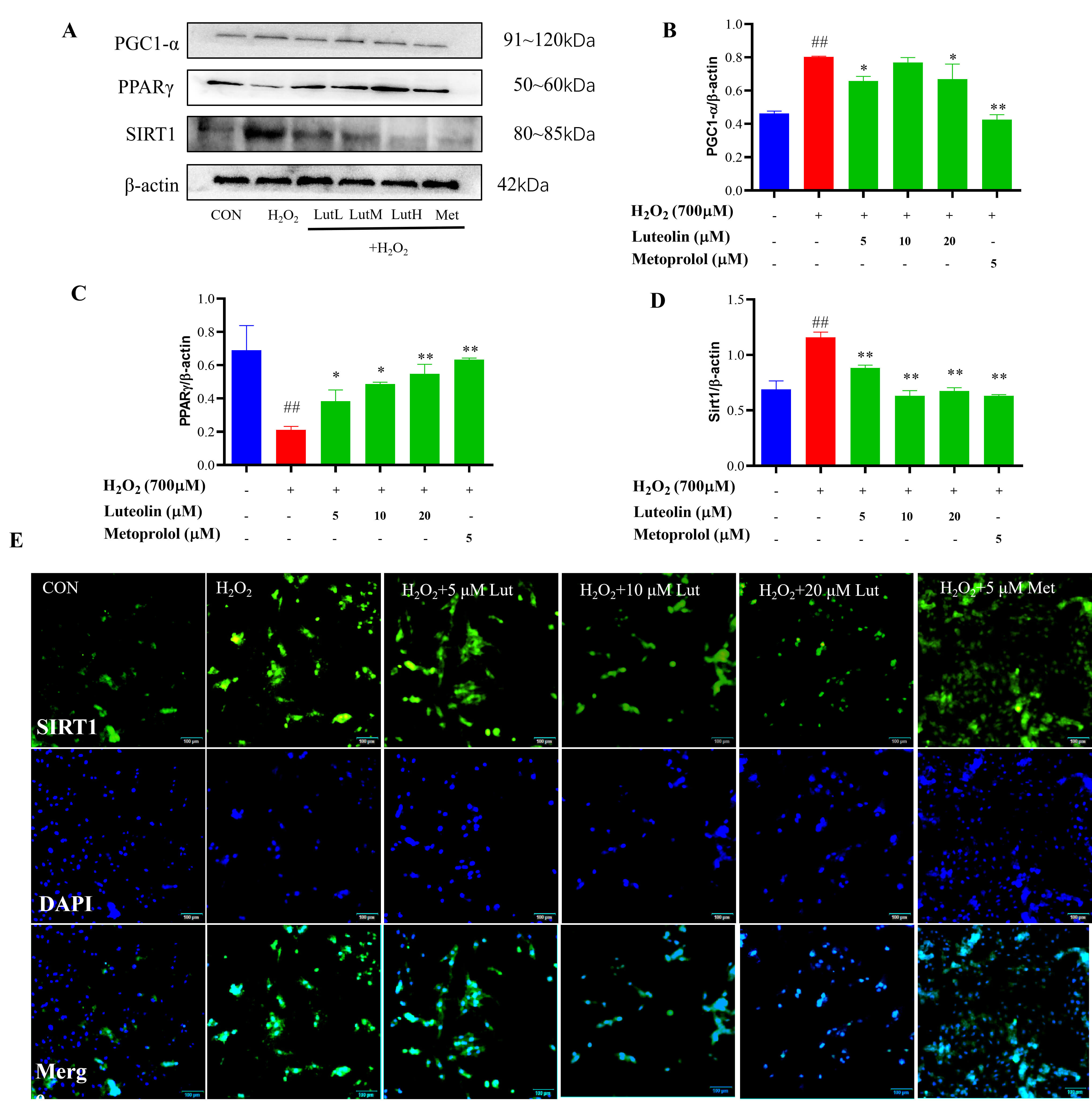 Fig. 6.
Fig. 6.
Effects of Lut on the expression of SIRT1, PGC-1
The results showed that the binding energies of Lut with AMPK
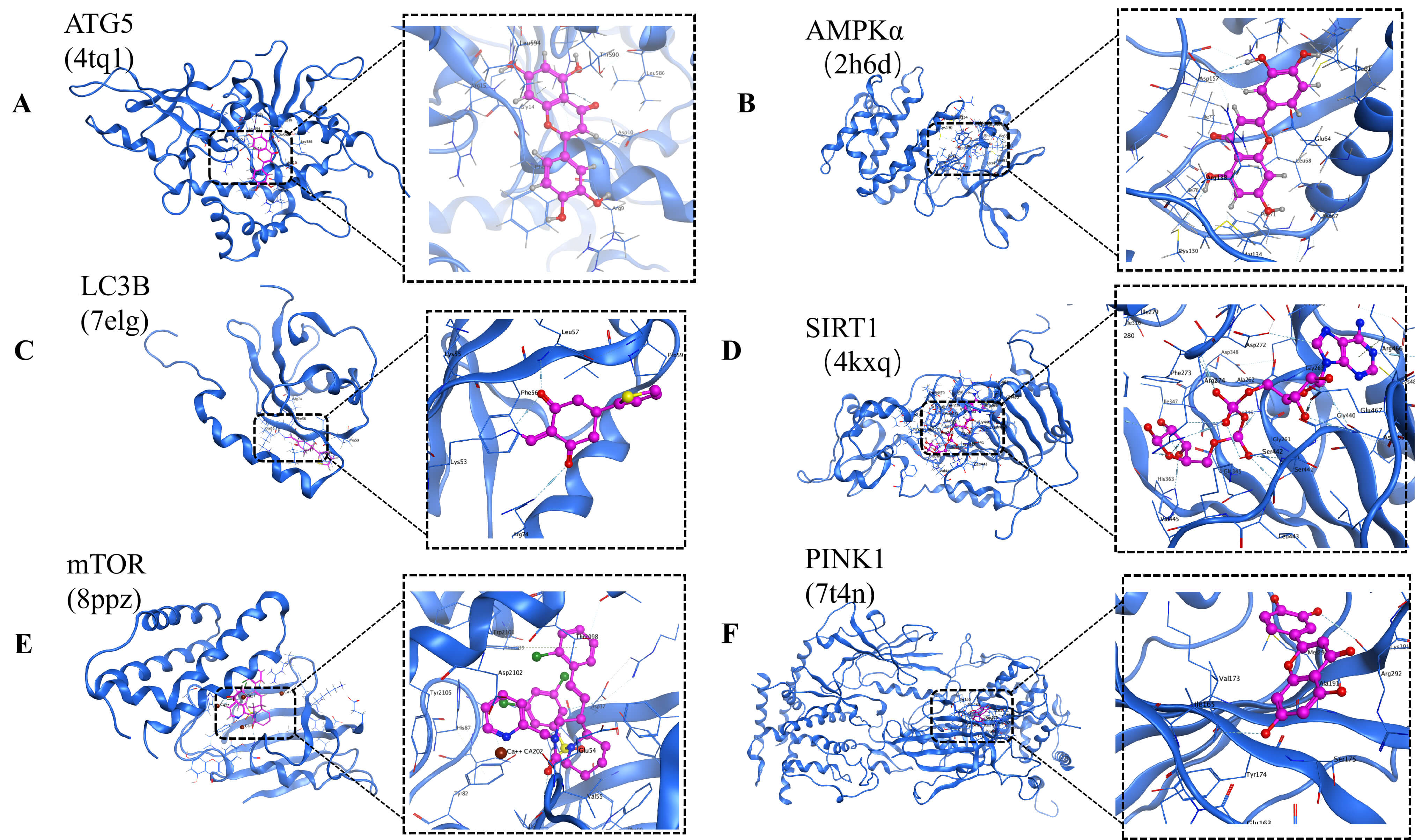 Fig. 7.
Fig. 7.
Docking results of Lut with ATG5 (PDB: 4tq1), AMPK
In recent years, natural compounds have been recognized as promising agents for the treatment of cardiovascular-related diseases. Many Chinese herbal medicines, such as polyphenols and flavonoids, have been shown to have antioxidant and anti-inflammatory effects and to be effective in modulating oxidative stress [27, 28], leading to therapeutic effects. Among them, Lut is widely distributed in nature, having been named because it was initially isolated from the leaves, stems, and branches of the herb Resedaodorata L., and it can be isolated from a variety of natural herbs, vegetables, and fruits. The antioxidant activity of the Lut molecule is mainly attributed to the 3ʹ,4ʹ-dihydroxyl structure (catechol moiety) in the phenolic B-ring, which is essential for the activity of the flavonoid molecule. On the pyran C ring of the flavonoid, a 2,3-double bond is conjugated to a 4-oxo function (i.e., the 1,4-pyranone moiety), which disperses the unpaired electrons on the A, B, and C rings and forms a more stable phenoxyl radical. This may bring better antioxidant activity for flavonoids [29]. Therefore, we utilized its antioxidant activity to protect the mitochondria of cardiomyocytes by effectively reducing the excess ROS produced in H2O2-induced H9c2 apoptosis through mitophagy.
Oxidative stress results from excessive accumulation of intracellular oxygen
free radicals and related metabolites. Among them, mitochondria are the primary
source of ROS in mammals and increased mitochondrial ROS production underlies
cardiovascular and many other diseases [30]. Lut reduced intracellular ROS levels
in H9c2 cells induced by the H2O2. Lut pretreatment significantly
ameliorated oxidative stress in H9c2 cardiomyocytes, suggesting that Lut could be
a candidate for cardiovascular disease. Peroxidative damage to mitochondria can
accelerate the opening of the MPTP, triggering pro-apoptotic factors such as
cytochrome C emission [31]. Peroxidative damage to mitochondrial membranes can
trigger the opening of the mitochondrial permeability transition pore (MPTP),
leading to the release of pro-apoptotic factors such as cytochrome c into the
cytosol [32]. The Bcl-2 protein family serves as a critical checkpoint in the
mitochondrial apoptotic pathway. The anti-apoptotic protein Bcl-2 preserves
mitochondrial integrity, while the pro-apoptotic protein Bax promotes
mitochondrial outer membrane permeabilization [33]. The ratio of Bcl-2 to Bax is
often used as an indicator of apoptotic susceptibility [34]. In our study,
H2O2 treatment significantly decreased the Bcl-2/Bax ratio and
increased Caspase8 expression, indicating that the mitochondrial apoptotic
pathway was engaged. Notably, treatment with luteolin (Lut) reduced intracellular
ROS, restored
Mitochondria are multitasking organelles that are crucial for maintaining a
balance of multiple cellular pathways, including ATP production, redox signaling,
and programmed cell death [35]. Mitochondria play an instrumental regulatory role
during I/R injury [36, 37, 38]. Liu et al. [39] concluded that treatment with
lignocaine could activate the AMPK/mTOR pathway through upregulation of SIRT3,
thereby inhibiting inflammatory response and apoptosis, promoting neurogenesis,
and ameliorating neurological deficits after ischemia in rats after CIRI. In the
present study, p-AMPK
Mitophagy acts as a double-edged sword in the maintenance of cellular homeostasis [47]. Under physiological conditions, it serves as a quality control mechanism to eliminate damaged mitochondria. However, excessive mitophagy can lead to mitochondrial depletion and energy crisis, ultimately triggering apoptosis [48]. The PINK1/Parkin pathway is a central regulator of mitophagy [49]. In healthy mitochondria, PINK1 is continuously cleaved and degraded. Upon mitochondrial damage, the loss of mitochondrial membrane potential stabilizes PINK1 on the outer mitochondrial membrane, where it recruits Parkin and initiates the autophagic clearance of damaged mitochondria [50]. Our results showed that exposure to H2O2 significantly increased the expression of mitophagy-related proteins, including ATG5, PINK1, and LC3B, indicating excessive activation of mitophagy. Notably, treatment with Lut reversed these changes, significantly decreasing the levels of ATG5, PINK1, and LC3B. These findings suggest that the cardioprotective effect of Lut is associated with suppressing this pathologically overactivated mitophagy.
Despite the promising findings, several limitations should be acknowledged. First, this study was conducted exclusively using an in vitro H2O2-induced oxidative stress model in H9c2 cardiomyocytes, which does not fully recapitulate the complex pathophysiology of myocardial I/R injury in vivo. Therefore, the observed protective effects of Lut require further validation in appropriate animal models, and our ongoing work includes such in vivo studies.
Luteolin protects H9c2 cardiomyocytes from H2O2-induced injury by modulating the AMPK/mTOR signaling pathway, inhibiting excessive mitophagy, and thereby reducing mitochondria-mediated apoptosis.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation, to any qualified researcher upon reasonable request.
ZY: Methodology, Resources, Writing – original draft; XL: Data curation, Formal analysis; DL: Methodology, Resources; KM: Data curation, Resources; MZ: Formal analysis, Funding acquisition, Writing – review & editing; YW: Conceptualization, Writing – review & editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to express our gratitude to all those who helped us during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This work has been supported by Natural Science Foundation of Inner Mongolia (NO: 2023LHMS08082), Education Institutions in Inner Mongolia Autonomous Region (NO: NJZY22456). Science and Technology Department of Inner Mongolia Scientific Research Fund Project (2021GG0279).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







