1 Department of Gastroenterology, West China Hospital, Sichuan University, 610041 Chengdu, Sichuan, China
2 Department of Pharmacology, West China School of Basic Medical Sciences & Forensic Medicine, Sichuan University, 610041 Chengdu, Sichuan, China
†These authors contributed equally.
Abstract
A gastric ulcer is a common gastrointestinal disorder, particularly associated with alcohol abuse, leading to acute ulcers. Proton pump inhibitors, such as esomeprazole, are effective for treating acute alcoholic ulcers. However, their therapeutic effects vary, and the underlying reasons for these differences remain unclear. In this study, we investigated gender differences in the effects of esomeprazole on ethanol-induced acute gastric injury in mice. We evaluated the potential different gastroprotective effects of esomeprazole by modulating the receptor-interacting protein kinase1 (RIPK1) and nuclear factor kappa-B (NF-κB) pathways.
The effects of esomeprazole on ethanol-induced acute gastric injury in vivo were analyzed. Macroscopic observation and pH measurement, histological analysis, and pepsin activity were used to assess the gastroprotective effects of esomeprazole. Serum levels of interleukin (IL-6) and tumor necrosis factor alpha (TNF-α) in female mice were measured by enzyme-linked immunosorbent assay (ELISA). Immunohistochemistry (IHC) assay and western blotting analysis were used to evaluate the anti-inflammatory effects and underlying mechanisms of esomeprazole.
Ethanol-induced acute gastric injury in mice, including ulcer area, ulcer index score, ulcer depth, bleeding and inflammation, pH, and pepsin, was reversed by esomeprazole pretreatment in vivo. Esomeprazole exhibited significant gastroprotective effects on alcohol-induced ulcers in mice, whether administered by injection or gavage, at both high and low doses. Notably, there were gender differences in the treatment effect. The improvement in these indicators was more pronounced in female mice, especially in response to the esomeprazole enteric capsule 3.03 mg/kg. We found that esomeprazole can reverse the elevation of serum inflammatory factors such as IL-6 caused by alcoholic gastric ulcer. Esomeprazole notably reduced the inflammatory injury score in gastric tissue, with decreased RIPK1 and NF-κB protein levels, accompanied by increased Schiff’s periodic acid staining and enhanced caspase 8 expression. The anti-necrosis, anti-inflammation effect of esomeprazole in ethanol-induced acute gastric injury is partly via RIPK1 and NF-κB-dependent.
Esomeprazole has excellent gastroprotective and anti-inflammatory effects in ethanol-induced acute gastric injury by modulating RIPK1and NF-κB in a dependent manner. The improved response demonstrated in female mice may be attributed to the protective effects of estrogen on the gastrointestinal tract.
Keywords
- gender differences
- esomeprazole
- ethanol-induced acute gastric injury
Gastric ulcer is a common gastrointestinal disorder characterized by mucosal damage and a complex pathogenesis involving multiple factors, such as Helicobacter pylori infection, acid secretion dysregulation, NSAID use, alcohol abuse, and genetic susceptibility. Epidemiological studies highlight significant gender disparities in gastric ulcer (gastrointestinal disorders) incidence, clinical manifestations, and treatment outcomes [1, 2, 3, 4]. In terms of incidence, males exhibit a higher prevalence, whereas females may display distinct pathological features linked to hormonal fluctuations. Higher male prevalence may correlate with smoking and alcohol consumption, while female susceptibility could involve psychosocial factors, necessitating gender-stratified prevention approaches, etc. Furthermore, clinical observations suggest gender-specific differences in therapeutic responses include adverse reactions (e.g., to proton pump inhibitors (PPI) or antibiotics) and ulcer recurrence rates [5]. Proton pump inhibitors such as esomeprazole are effective drugs for treating acute ulcers. In terms of treatment, omeprazole may be more effective for female patients with gastric ulcers [5, 6, 7]. Different studies have found that the efficacy of PPI in treating ulcer bleeding varies, and long-term use may also cause varying degrees of lung damage [6, 7]. The differences in the effectiveness of PPIs in alcoholic ulcers remain unclear. These differences prompt us to consider whether the therapeutic effect of PPI is gender related, and what may be the underlying mechanisms of these differences?
Therefore, the study of gender differences in ulcers has important clinical significance and scientific value; however, systematic investigations into these disparities remain limited, and the underlying mechanisms are poorly understood. The mechanism in gastric mucosal protection and repair requires further exploration, particularly their interactions with inflammation, oxidative stress, and H. pylori infection. Clarifying gender differences and inflammation mechanisms could inform personalized treatment strategies, such as some special drug therapy for females or adjusted drug dosages for males [8, 9]. The sexual dimorphism in necroptotic signaling has been evidenced in kidney ischemia-reperfusion injury. Female mice, which sustain milder kidney injuries, exhibit delayed and attenuated necroptotic signaling, involving molecules such as receptor-interacting protein kinase1 (RIPK1), receptor-interacting protein kinase 3 (RIPK3), and mixed lineage kinase domain-like protein (MLKL) [10]. By comparing therapeutic outcomes between genders in mice, this study aims to provide a theoretical basis for gender-specific clinical treatment of proton pump inhibitors. Therefore, further investigation into the treatment differences and mechanism of gastric ulcers related to gender is of great significance for optimizing clinical treatment plans and improving proton pump inhibitor therapeutic efficacy.
In accordance with the Committee on the Ethics of Animal Experiments of Sichuan
University (Permit Number: 20240611004), a total of 60 Kunming mice (18–22 g,
6–8 week-old, 30 male and 30 female) were purchased from Da-Shuo Biological
Technology Co., Ltd. (Chengdu, China) and housed under standard conditions (room
temperature: 23 °C
All treatment groups were pre-treated with the corresponding dose of esomeprazole, once a day for 5 days. The control group and the ethanol group were given the same volume of saline. Two hours after the last pre-treatment, 100% ethanol 0.2 mL was administered orally to induce acute gastric injury, according to our preliminary experiments [12]. The control group received saline 0.2 mL orally. After 1 h from induction, all mice were euthanized with pentobarbital sodium (20 mg/mL, 150 mg/kg, intraperitoneally).
The gastric tissues of all mice were removed and opened along the greater
curvature. Gastric ulcerative lesions were assessed by two blinded observers and
photographed. On the mucosal side of the stomach, according to the diameter of
gastric ulcer, the ulcer index (UI) was scored as follows: 0: no lesion; 1: ulcer
area with the longest diameter
The gastric juice pH value was determined with pH test paper.
Then, the gastric tissues were cut into 2 halves. One was kept in 10% Neutral formalin solution for further histological studies, and one-half was kept at –80 °C.
After fixation in 10% Neutral formalin solution for 24 h, gastric tissues were embedded in paraffin and cut into 4 µm sections. These sections were stained with hematoxylin and eosin (HE) and Schiff’s periodic acid (PAS) for histological evaluation. The morphology of the gastric tissue was observed blindly using a light microscope (DM250, LEICA, Germany), and gastric injury was graded from 0 (normal) to 4 (severe). Ulcer depth, bleeding, and inflammation were scored, respectively. Finally, the gastric injury score was obtained by adding the individual scores for each type.
PAS stain was used to assess the mucus production in the gastric tissue.
For the IHC assay, the 4-µm sections were incubated with primary receptor-interacting protein kinase1 (RIPK1) antibodies (dilution 1:50, Proteintech, USA) overnight at 4 °C, washed three times with phosphate-buffered saline (PBS), and then stained with secondary antibody (goat anti-rabbit IgG labeled by horseradish peroxidase (HRP)) for 4 hours. 3,3’-diaminobenzidine (DAB) was used for IHC signal detection after washing three times with PBS, then hematoxylin counterstaining for 2 min. Observe the images using an optical microscope (DM250, LEICA, Germany) and perform semi-quantitative analysis blindly through ImageJ software.
Gastric tissues were homogenized with the extracting solution and centrifuged at
4 °C, 10,000 rpm for 10 min, and the
protein concentrations in supernatant (Cpr) were measured by BCA at 562 nm.
Pepsin activity from gastric tissues was determined by ultraviolet spectroscopy.
The opticle density (OD) values were detected at 275 nm using a
spectrophotometer. Pepsin activity (U•mg prot-1) = 1.31
Blood samples of mice were centrifuged at 3000 g for
15 min at 4 °C, then serum was collected, and the concentrations of
IL-6 and TNF-
Total proteins were extracted using radioimmunoprecipitation assay (RIPA) Lysis
Buffer (Beyotime Institute of Biotechnology, China), and the protein
concentrations were measured with a BCA protein assay kit (Beyotime Institute of
Biotechnology, China). Equal amounts of total protein (40 µg/10 µL)
were loaded onto 8% SDS-PAGE gels at 120 v for 60 min, electro-transferred to
polyvinylidene difluoride (PVDF) membranes, blocked in 5% non-fat milk at room
temperature for 2 h, and incubated with the diluted primary antibodies, including
Caspase8 (Cell Signalling Technology, 4790S, 1:1000), RIPK1 (Proteintech,
17519-1-AP, 1:1000), and NF-
All values were expressed as mean
To investigate the gender difference of the gastroprotective effect of
esomeprazole, the mouse acute ethanol (EtOH)-induced model of gastric injury was
established by intragastric administration of 100% EtOH. First, the gastric
mucosal injury was observed in gross tissues, and the ulcer index (UI) was used
to evaluate the extent of gastric injury. As shown in Fig. 1A, no visible lesions
developed in the control group, but 100% EtOH markedly resulted in gastric
mucosa ulcer and bleeding in female and male mice, manifested by an increased UI
compared with the control group (p
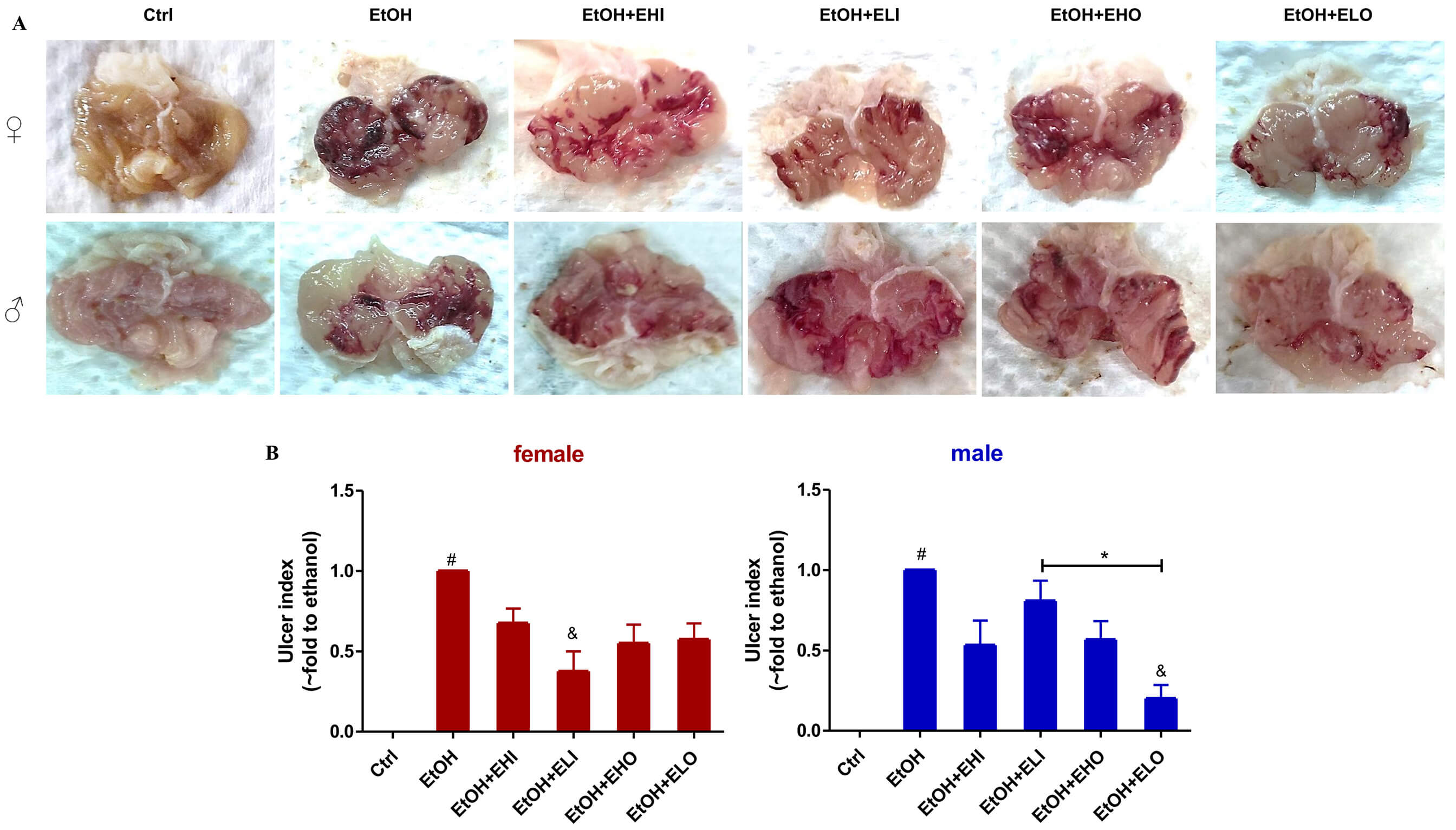 Fig. 1.
Fig. 1.
Effects of esomeprazole on the EtOH-induced gastric injury in
mice. Representative images of the mouse stomach from (A) control, EtOH, EtOH +
esomeprazole 6.06 mg/kg i.p (EtOH + EHI), EtOH + esomeprazole 3.03 mg/kg i.p (EtOH
+ ELI), and EtOH + esomeprazole 6.06 mg/kg gavage (EtOH + EHO), EtOH + esomeprazole
3.03 mg/kg gavage (EtOH + ELO). (B) The ulcer index was compared between mice of
different genders. Significant differences were observed after administration of
esomeprazole at the dose of 3.03 mg/kg i.p in female mice and 3.03 mg/kg p.o in
male mice. # p
Next, histopathological evaluation of the damage in the gastric mucosa was
performed using HE. In the control group, the gastric mucosa of mice was intact,
and there was no inflammatory cell infiltration and mucosal hemorrhage (Fig. 2A).
100% EtOH induced an extensive disruption of the gastric mucosal layer with a
marked presence of ulcerative plaque, gland atrophy, and hemorrhage, inflammatory
cell infiltration in the mucosal and submucosal layer, with an increased gastric
injury score compared with the control group (Fig. 2B). As compared to the EtOH
group, two dose forms of esomeprazole significantly reduced EtOH induced gastric
mucosal injury with a significant inhibition in ulcer/hemorrhage score and total
score, except for the ELI group (left and right panel, Fig. 2B). However, there
was obviously inhibition in inflammatory cell infiltration only in female mice
(middle panel, Fig. 2B). Therefore, as shown in the right panel of Fig. 2B, we
found pre-treatment with ELO at 3.03 mg/kg (clinical equivalent
dose 20 mg) significantly prevented the presence of ulcerative plaque,
hemorrhage, and inflammatory cell infiltration, especially in female mice
(p
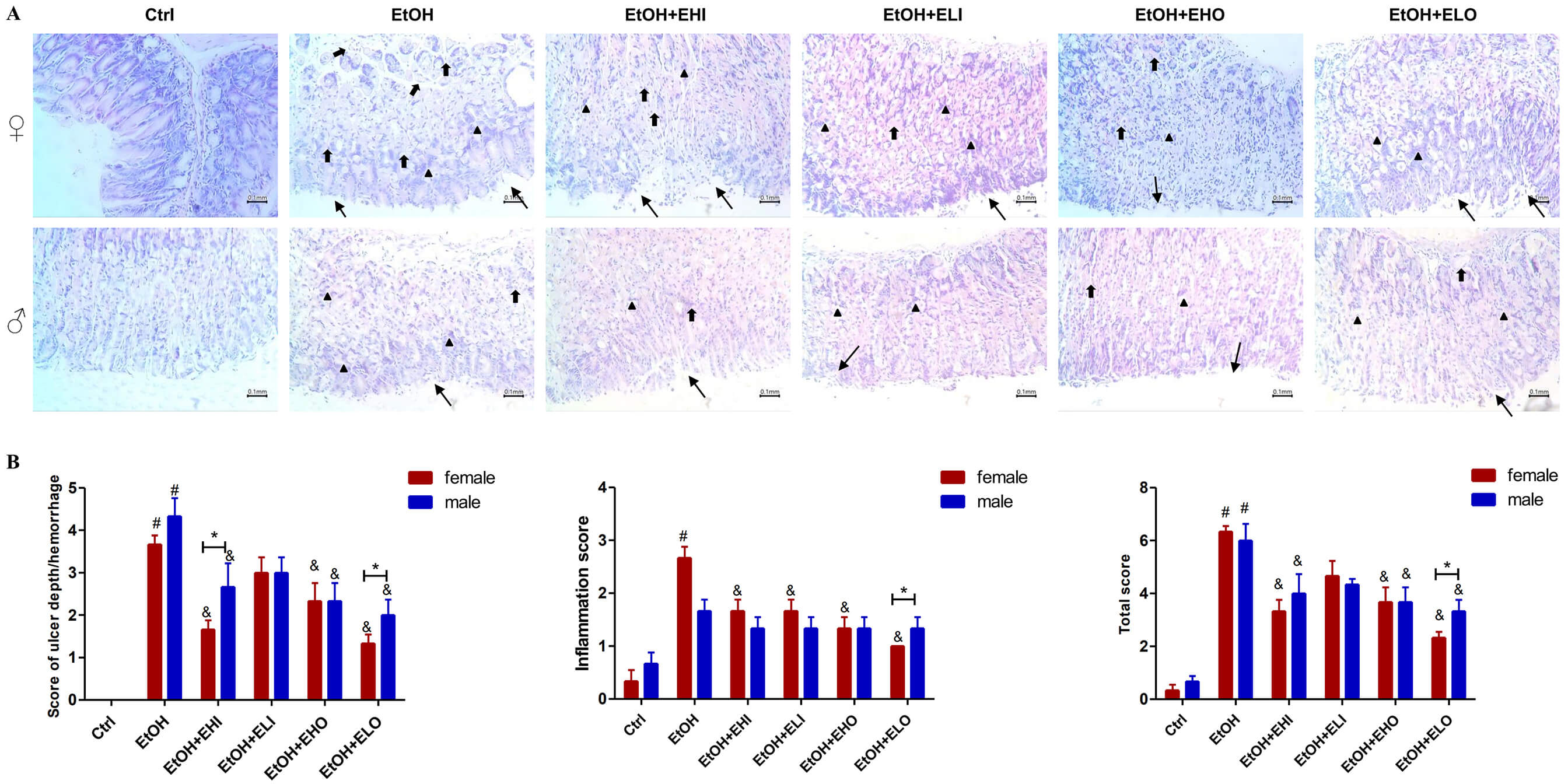 Fig. 2.
Fig. 2.
Gastroprotective effect of esomeprazole on the EtOH-induced
gastric injury in mice by histopathological evaluation. (A) Representative
images of histopathological evaluation of the damage in the gastric mucosa
stained with hematoxylin and eosin (HE) stains (
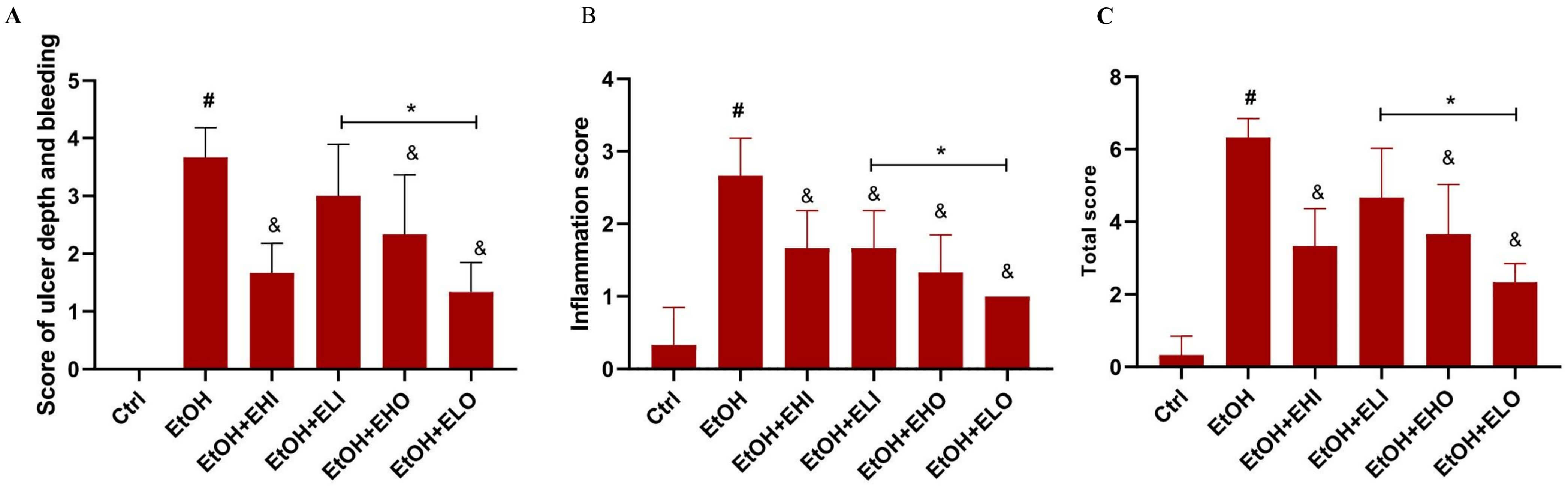 Fig. 3.
Fig. 3.
Gastroprotective and anti-inflammatory effects of esomeprazole
in the EtOH-induced gastric injury in female mice. (A) Gastric injury scores
include ulcer depth, bleeding, (B) inflammation score and (C) total score were
compared from control, EtOH, EtOH + esomeprazole 6.06 mg/kg i.p (EtOH + EHI), EtOH
+ esomeprazole 3.03 mg/kg i.p (EtOH + ELI), and EtOH + esomeprazole 6.06 mg/kg
gavage (EtOH + EHO), EtOH + esomeprazole 3.03 mg/kg gavage (EtOH + ELO). * p
To examine the gender different effect of esomeprazole on acid secretion and
pepsin activity, we measured the pH value with pH test paper first and found the
pH values of the EtOH group in both female and male mice were little difference
compared with that of the control group (p
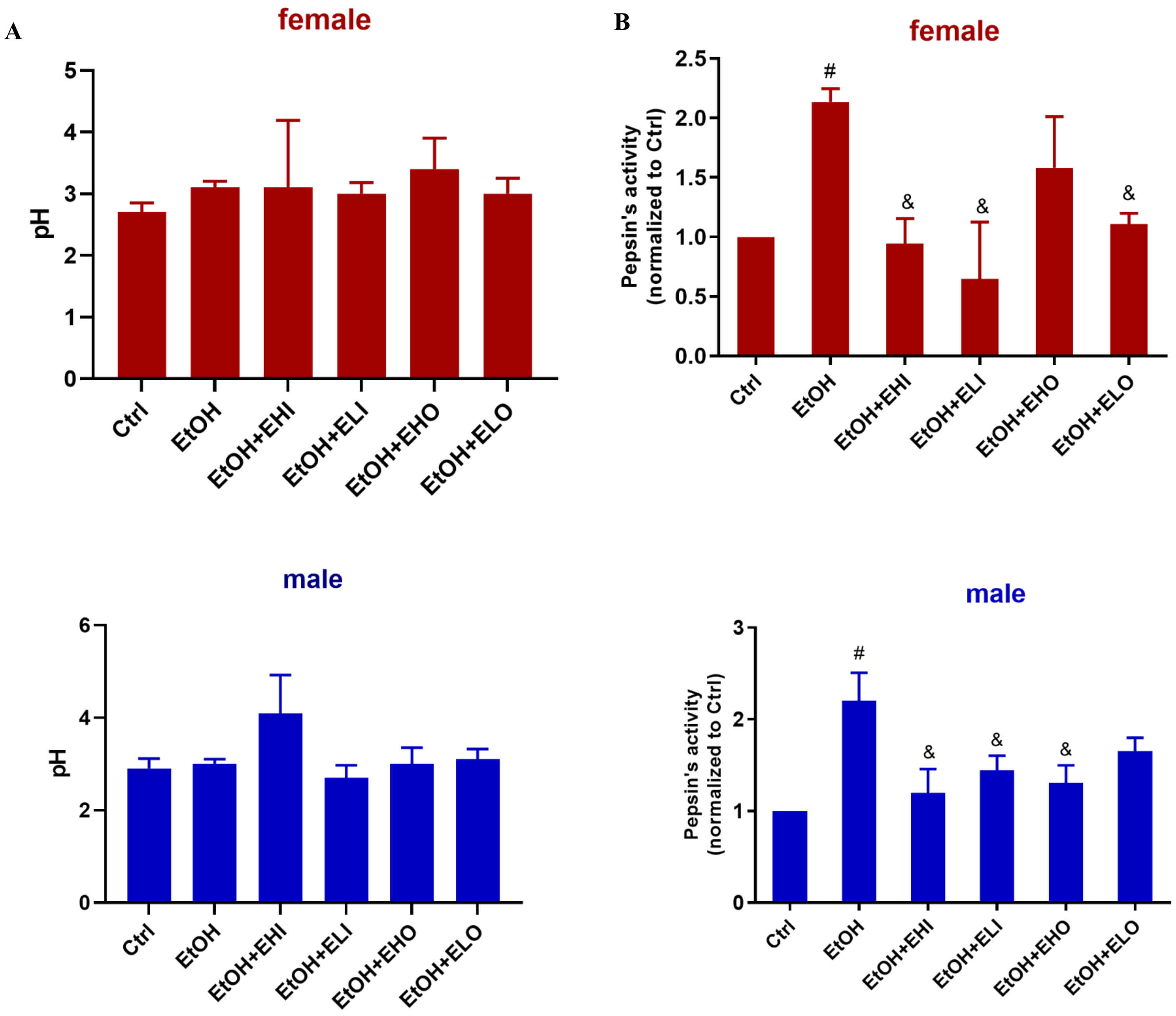 Fig. 4.
Fig. 4.
Effects of esomeprazole on gastric acid secretion in the
EtOH-induced gastric injury in mice. (A) pH value with pH test paper from
control, EtOH, EtOH + esomeprazole 6.06 mg/kg i.p (EtOH + EHI), EtOH +
esomeprazole 3.03 mg/kg i.p (EtOH + ELI), and EtOH + esomeprazole 6.06 mg/kg
gavage (EtOH + EHO), EtOH + esomeprazole 3.03 mg/kg gavage (EtOH + ELO). (B) Pepsin
activity was compared. Significant differences were observed after administration
of esomeprazole EtOH + EHI, EtOH + ELI, and EtOH + ELO group in female mice, EtOH
+ EHI, EtOH + ELI, and EtOH + EHO in male mice. # p
Considering the gender difference in esomeprazole efficacy, we chose female mice as the animal model in the subsequent experiment. PAS staining was used to evaluate the content of gastric mucosal glycoprotein. As we expected, the PAS staining of the gastric mucus was weaker in the EtOH group than that in the control group, which was significantly increased in all esomeprazole pretreatment groups (Fig. 5A).
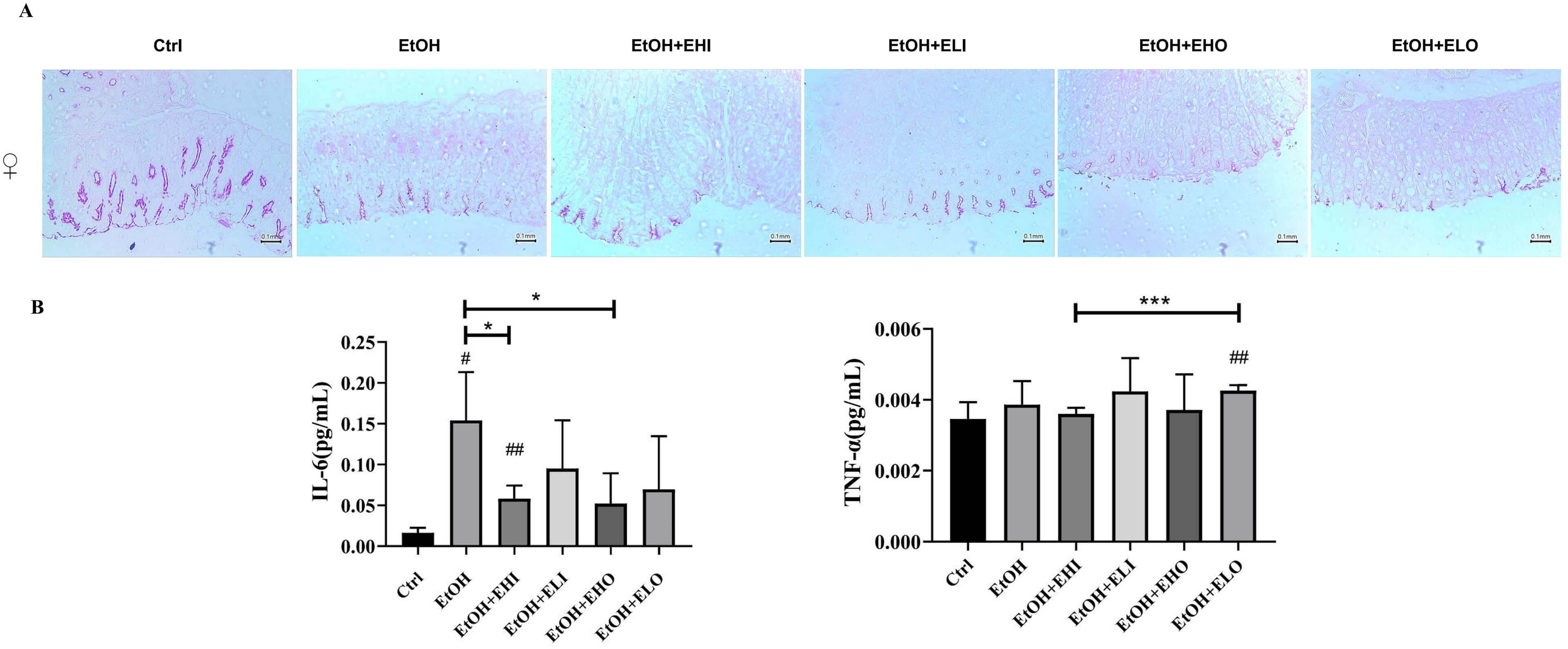 Fig. 5.
Fig. 5.
Effects of esomeprazole on serum inflammatory factors and
gastric mucosal glycoprotein expression in the EtOH-induced gastric injury in
female mice. (A) Representative PAS staining images of the mouse stomach from
control, EtOH, EtOH + esomeprazole 6.06 mg/kg i.p (EtOH + EHI), EtOH +
esomeprazole 3.03 mg/kg i.p (EtOH + ELI), and EtOH + esomeprazole 6.06 mg/kg
gavage (EtOH + EHO), EtOH + esomeprazole 3.03 mg/kg gavage (EtOH + ELO). PAS
staining of the gastric mucus was weaker in the EtOH group than in the control
group; mucosal glycoprotein expression increased in all esomeprazole groups.
Scale bar: 0.1 mm. (B) Serum concentrations of inflammatory factors IL-6 and
TNF-
Our studies demonstrated that the score of the EtOH-induced gastric injury
tissues in female mice was significantly attenuated following esomeprazole
pretreatment (p
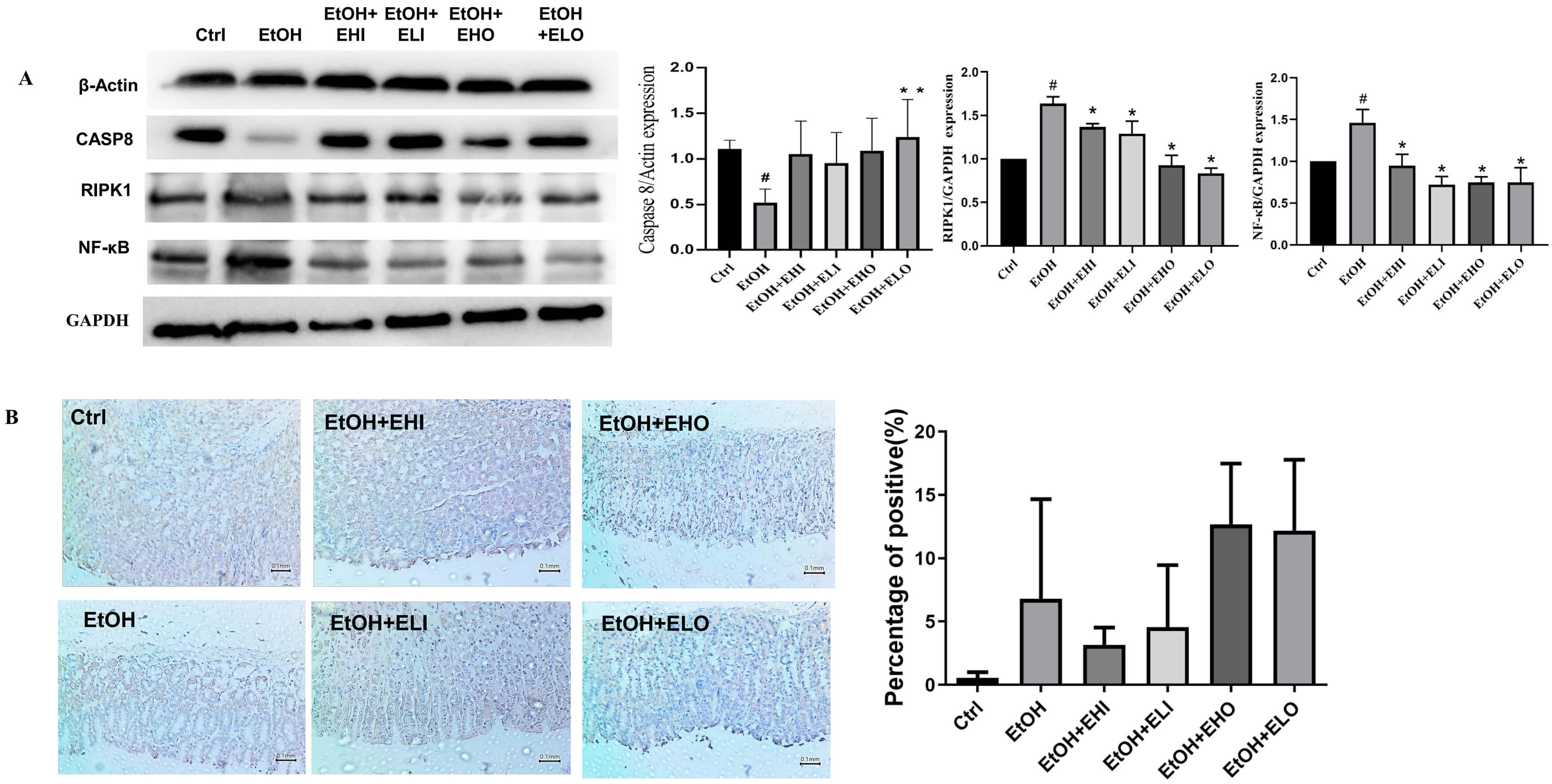 Fig. 6.
Fig. 6.
Anti-inflammatory effects of esomeprazole expression via
receptor-interacting protein kinase1 (RIPK1)/NF-
In the present study, we demonstrated that esomeprazole, a proton pump
inhibitor, exerts significant gastroprotective effects in ethanol-induced gastric
injury. The result of our study indicated that it has a good therapeutic effect
on ethanol-induced gastric ulcers in mice, with a significant gender difference,
and the therapeutic effect is better in female mice. Moreover, our study has
shown that the gastroprotective mechanism of esomeprazole is related to its
anti-inflammatory, anti-necrosis action, which is partly associated with the RIPK1
and NF-
Esomeprazole, a widely used proton pump inhibitor, is well-known for its ability to reduce gastric acid secretion by inhibiting the H+/K+-ATPase enzyme in parietal cells. This mechanism has made it a cornerstone in the treatment of acid-related disorders such as gastroesophageal reflux disease and peptic ulcers. However, the findings from the mentioned research open new avenues for understanding its potential therapeutic applications beyond acid suppression.
As a result, the EtOH + esomeprazole group had less severe inflammation. This
observation that esomeprazole can effectively treat ethanol-induced gastric
ulcers in mice suggests that it may have additional protective mechanisms in the
gastrointestinal tract. Ethanol is a well-known irritant that can cause severe
gastric mucosal damage, leading to ulcer formation. Our research found that the
effect of pH on alcoholic ulcers was not significant, but esomeprazole still had
a good anti-ulcer effect, which was not significantly related to changes in pH;
and its effect had significant gender differences, so we analyzed whether it is
related to other mechanisms to play a protective role against ulcers. The fact
that esomeprazole shows a better therapeutic effect in female mice indicates that
there might be gender-specific differences in the pathophysiology of
ethanol-induced ulcers or in the pharmacodynamics of esomeprazole. Hormonal
differences between male and female mice could potentially influence the
susceptibility to gastric injury and the response to treatment. For example,
estrogen has been demonstrated to exert protective effects on the
gastrointestinal tract through estrogen-related receptor alpha, which potentially
plays a crucial role in regulating immune homeostasis by maintaining a balanced
pro-inflammatory and anti-inflammatory response, thus contributing to the
preservation of epithelial integrity [14]. This might contribute to the
enhanced therapeutic effect observed in female mice. Histological observations
have found that esomeprazole can significantly reduce tissue inflammation, ulcer
necrosis area, and depth; and esomeprazole can reverse the elevation of serum
inflammatory factors such as IL-6 caused by alcoholic gastric ulcer in female
mice. Although our research found that there was no change in TNF-
Moreover, the association of esomeprazole’s gastroprotective effect with its
anti-inflammatory action is intriguing. Inflammation and necroptosis play a
crucial role in the development and progression of gastric ulcers [16]. Members
of the receptor-interacting protein kinase (RIPK) family are important regulators
of inflammation and cell death [17, 18, 19]. RIPK1 complex regulates necroptosis,
which is a highly controlled programmed cell death and works against
pathogen-mediated infections, and morphologically features cellular organelle
swelling followed by plasma membrane rupture [20, 21, 22]. The classical necroptosis
is initiated with TNF-
In conclusion, the research findings highlight the potential of esomeprazole as
a therapeutic agent for ethanol-induced gastric ulcers, particularly in female
mice. Further studies are needed to elucidate the exact mechanisms underlying its
anti-inflammatory effects and its interaction with the RIPK1and NF-
However, there are many limitations in the present study. Firstly, the use of pH test paper is relatively imprecise. Secondly, this study did not evaluate sex hormones, estrous cycles, or sex-dependent pharmacokinetics, and the intentional focus on females in subsequent experiments introduces systematic bias, thereby weakening the claim of truly gender-dependent mechanisms. In future studies, we will conduct a thorough assessment of sex hormone levels and investigate gender-dependent mechanisms in male mice.
Esomeprazole has excellent gastroprotective and anti-inflammatory effects in
ethanol-induced acute gastric injury, especially in female mice, by modulating
the RIPK1 and NF-
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
LZ, ZHY, and LHW conceived the idea, and designed the study; LZ wrote and revised the manuscript; YW, YY and FL contributed to most of the animal and molecular biology experiments; TZ, YZ, and LLL assisted in completing animal experiments and participated in data analysis. All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work. All authors read and approved the final manuscript.
All animal experiments should comply with the ARRIVE guidelines. All methods were carried out in accordance with relevant guidelines and regulations. The study was approved by the Animal Ethics Committee of Sichuan University (NO. 20240611004). The authors have read the ARRIVE guidelines, and the manuscript was prepared and revised according to the ARRIVE guidelines.
We would like to express our gratitude to all those who helped us during the writing of this manuscript, and to the public experimental platform of West China School of Basic Medical Sciences & Forensic Medicine for providing some testing instruments. Thanks to all the peer reviewers for their opinions and suggestions.
This work was supported by the Key Project of the Science and Technology Department in Sichuan Province (No. 23ZDYF1182). The authors received no specific funding for this work.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






