1 Department of Surgery, University of Kansas Medical Center, Kansas City, KS 66160, USA
2 Department of Cancer Biology, University of Kansas Medical Center, Kansas City, KS 66160, USA
3 Department of Gastroenterology, Hepatology and Nutrition, Children’s Mercy Hospital, Kansas City, MO 64108, USA
Abstract
Inflammatory bowel disease (IBD) is a chronic inflammatory disorder of the gastrointestinal tract characterized by relapsing and remitting disease activity. Despite decades of research, a definitive cure for IBD remains elusive, as cycles of remission and flare-ups mark the disease course and often relies on long-term pharmacologic and supportive strategies. Increasing evidence indicates that diet and nutrition play an important role in modulating the outcomes of IBD. Although a wide range of dietary interventions has been explored, no universally effective approach has been established. Investigations have also proven that different dietary interventions can significantly affect clinical outcomes by altering gut microbial composition. This review explores a novel approach to IBD management by integrating gut microbiome modulation into personalized patient diets. The review examines various dietary interventions commonly used in IBD treatment and their impact on the gut microbiome composition. Furthermore, the study proposes a framework for creating personalized nutrition based on individual microbiome profiles. Finally, the review discusses the potential of personalized nutrition as a long-term therapeutic strategy for IBD management, offering insights into its feasibility and implications for patient care.
Keywords
- inflammatory bowel disease
- gut microbiome
- diet
Inflammatory bowel disease (IBD) has been described numerous times as a chronic condition that causes inflammation of the gastrointestinal tract and clinically includes Crohn’s Disease (CD), which can affect any part of the GI tract, and Ulcerative Colitis (UC), whose inflammation is often limited to the colon [1, 2, 3, 4]. IBD is a painful disease with symptoms including frequent diarrhea, blood in the stool, persistent abdominal pain, and weight loss. Despite decades of research into IBD, there is no permanent cure for IBD as the disease is often characterized by bouts of clinical remission and relapse that heighten the risk for other conditions, particularly colon cancer [5]. Furthermore, there is no singular cause of IBD, but studies have provided evidence that the pathogenesis of IBD is linked to aberrations in host genetics, impaired immune system, dysbiosis of the intestinal microbiota, and other environmental factors such as smoking, diet, and stress [6, 7].
In IBD, disease pathogenesis reflects a complex, bidirectional interplay among host genetics, immune regulation, environmental exposures, and the gut microbiota [8]. Genetic susceptibility and unfavorable environmental factors, such as diets high in saturated fat, can promote immune dysregulation and reshape the baseline microbial ecosystem, thereby favoring dysbiosis toward more pathogenic microbial communities [9, 10, 11]. This dysbiosis state can, in turn, actively contribute to disease initiation and perpetuation by increasing epithelial permeability, enhancing microbial translocation, and skewing mucosal immune responses toward pro-inflammatory pathways [12, 13, 14]. Microbial metabolites and structural components produced during dysbiosis further influence mucosal inflammation by altering immune signaling pathways. Pro-inflammatory products, such as lipopolysaccharides, can activate innate immune responses [15, 16, 17, 18, 19]. In contrast, metabolites including short-chain fatty acids, bile acid derivatives, and tryptophan-derived compounds can either dampen or exacerbate inflammation by shaping cytokine production and T-cell responses at the intestinal mucosa. As a result, many alterations in the microbiota observed in IBD likely represent both contributors to disease activity and consequences of chronic inflammation [20, 21, 22, 23].
Of all the environmental factors contributing to IBD, the relationship between diet, nutrition, and changes in the gut microbiota is critical to understanding and managing disease dynamics. Diet directly affects the composition and function of intestinal bacteria, thereby regulating the gut microbiota. A diet rich in fiber, fermented foods, and prebiotics can promote a healthy and diverse microbiota, which is essential for maintaining gut health and reducing IBD symptoms [24, 25, 26, 27]. Conversely, a diet high in processed foods and sugar may lead to dysbiosis, which can worsen IBD symptoms [28]. Studies continue to show that diet has a profound impact on the gut microbiome and immune response in patients with IBD [29, 30, 31, 32, 33, 34]. In experimental models, dietary interventions have been shown not only to modulate the gut microbiota but also to promote mucosal repair and epithelial regeneration. For instance, supplementation with fermentable fiber (pectin) or the short-chain fatty acid derivative tributyrin ameliorated infectious colitis in mice by restoring microbial diversity, enhancing mucus barrier integrity, and stimulating leucine-rich repeat-containing G-protein coupled receptor 5 (Lgr5+) intestinal stem cell proliferation [29]. These effects highlight how specific dietary components can shape the microbiome’s composition and metabolic output, ultimately influencing epithelial healing, immune balance, and disease outcomes in IBD.
Given the critical role that a healthy microbiome plays in preventing and managing IBD and its complications, many patients seek therapeutic diets as a complement or alternative to conventional medical treatment [27, 35, 36, 37]. Beyond its role in symptom modulation, the gut microbiome is increasingly recognized as a key determinant of therapeutic response in IBD [38].
Emerging evidence indicates that both microbial composition and functional metabolic activity can influence responsiveness to biologic therapies. Distinct microbial signatures have been associated with response to specific agents, including enrichment of butyrate-producing bacteria with vedolizumab and higher Faecalibacterium with reduced Escherichia/Shigella abundance in ustekinumab responders [39]. In addition, bacterial metabolic activity can directly affect drug efficacy, as certain microbial species metabolize 5-aminosalicylic acid (5-ASA), thereby reducing its clinical effectiveness [40]. At the same time, microbial communities are also required for the activation and therapeutic efficacy of sulfasalazine in IBD-associated spondyloarthritis [15, 41].
Understanding these microbiome–drug interactions not only underscores the influence of diet and microbial health on disease trajectory but also points toward future strategies in personalized IBD management, where microbiome profiling could guide both dietary and pharmacologic interventions to optimize treatment response.
IBD patients have explored several dietary patterns while managing the disease [42]. A healthy diet for IBD involves limiting sugar and refined foods while promoting plant-based and whole foods over animal-derived and processed options. This approach is based on the understanding that certain foods can trigger gut inflammation, exacerbating IBD symptoms [43, 44]. Despite extensive research, no single diet can be universally recommended for all IBD patients, as IBD is a highly individualized disease with different triggers and symptoms for each person, leading to differing dietary recommendations from physicians and nutritionists [45]. Generally, diets should ensure an appropriate energy intake and adequate levels of vitamins and minerals such as Vitamin D, iron, calcium, zinc, and folic acid; it should also aim to reduce symptoms and improve the quality of life (QoL) [44, 46, 47]. Some of the well-established dietary interventions that have been utilized in IBD management are highlighted below (Fig. 1).
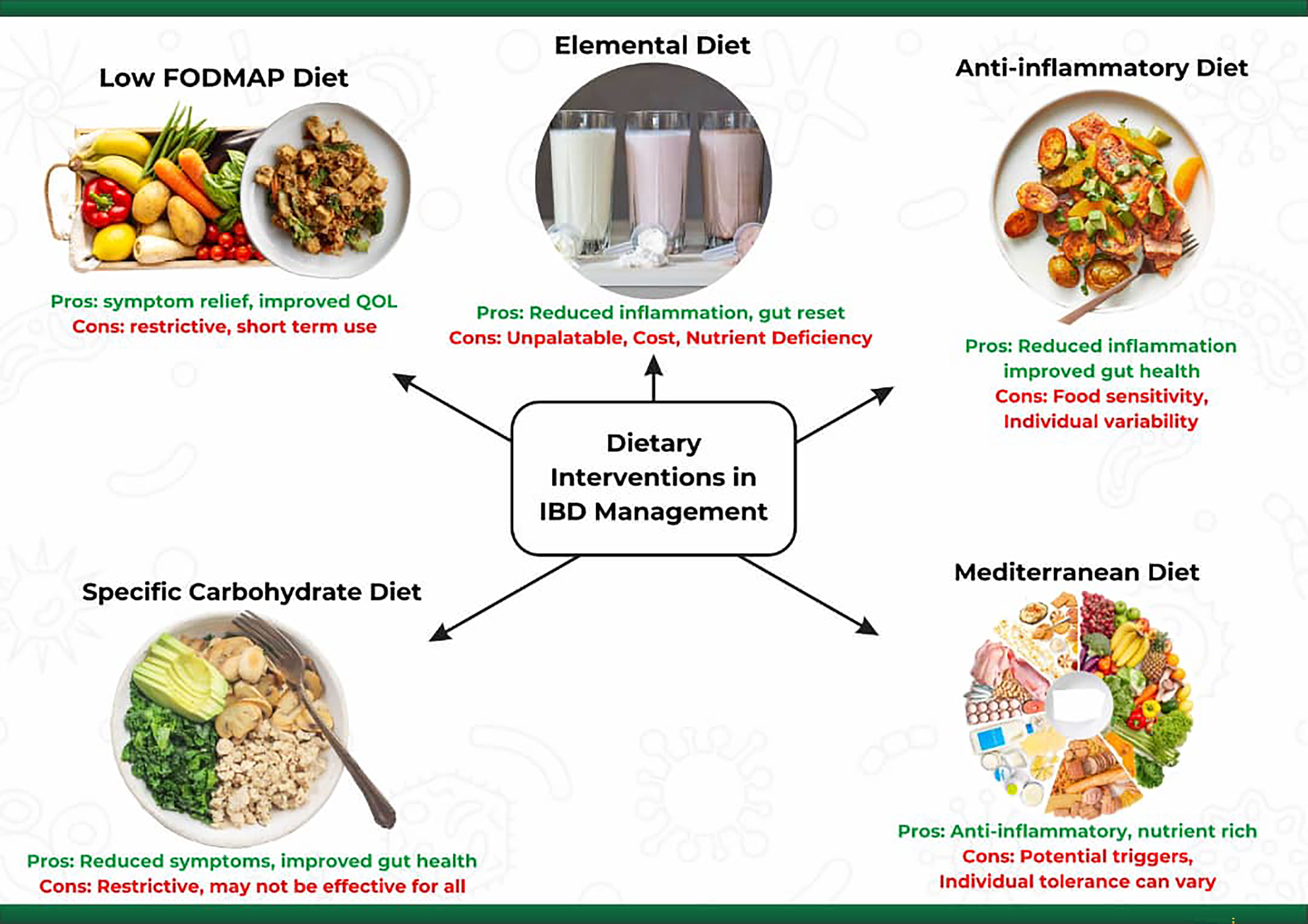 Fig. 1.
Fig. 1.
Summary of major dietary interventions used in IBD management, illustrating potential benefits and limitations of commonly prescribed dietary patterns and emphasizing the variability in patient response. IBD, Inflammatory bowel disease.
The term FODMAP refers to “fermentable oligosaccharides, disaccharides, monosaccharides, and polyols” [48, 49], classified based on the length of their carbohydrate chains and fall into categories of foods poorly digested in the human gut [48, 49]. Instead of being absorbed, FODMAPs are readily fermented by the colonic microbiota and are considered quick fuel for colonic bacteria, producing gas and other byproducts that can cause gastrointestinal discomfort. The symptoms often include diarrhea, bloating, abdominal pain, and constipation. For individuals with IBD, functional gastrointestinal symptoms like diarrhea, abdominal pain, and bloating are quite common [49, 50]. A low-FODMAP diet, which restricts food rich in these fermentable carbohydrates, has gained attention as a potential means to alleviate these symptoms [51, 52]. Some examples of Low-FODMAP foods are proteins (beef, chicken, eggs, fish), grains (gluten-free products, oats, quinoa, rice), vegetables (carrots, cucumbers, spinach, zucchini), fruits (bananas, blueberries, strawberries), and dairy (lactose-free products, hard cheeses). High-FODMAP foods, on the other hand, are vegetables (onions, garlic, cauliflower, asparagus), fruits (apples, pears, mangoes, cherries), dairy (milk, yogurt, soft cheeses), grains (wheat, rye, barley) and legumes (beans, lentils, chickpeas) [53, 54]. For patients whose disease is in remission or only mildly active, adherence to a low-FODMAP diet for approximately 4–6 weeks may improve gastrointestinal symptoms and quality of life; however, its impact on inflammatory biomarkers remains a subject of debate [55].
The Low FODMAP diet restricts the intake of poorly digested carbohydrates, reducing the substrate for bacterial fermentation in the gut. This approach is usually divided into three phases: elimination, reintroduction, and personalization. In the elimination phase, for about 4–6 weeks, high-FODMAP foods are removed from the diet to reduce symptoms. During the reintroduction phase, foods are gradually reintroduced one at a time to determine which FODMAPs cause symptoms. Once trigger foods are identified, a long-term balanced diet is developed in the personalization phase to manage symptoms while incorporating as many foods as possible [53, 56]. Several studies have investigated the effects of a Low FODMAP diet on IBD patients, and the findings have been promising [50, 56, 57, 58, 59]. By minimizing the intake of FODMAPs, individuals with IBD may experience fewer symptoms and better manage their condition.
The low FODMAP diet, initially recognized for managing irritable bowel syndrome (IBS) [51], has extended its application to IBD. By reducing fermentable carbohydrates being consumed, this diet diminishes luminal distension and gas production, easing bloating and discomfort in IBD patients [50, 58, 60]. Adherence to the low FODMAP diet not only reduces symptoms but also significantly improves the quality of life of IBD patients, enhancing their psychological well-being and social functioning [58]. Patients often report increased energy levels, reduced anxiety related to symptoms, and improved social engagement due to decreased gastrointestinal discomfort [61]. In addition, research reveals that certain FODMAPs can increase intestinal permeability in susceptible individuals, and restricting these compounds may help maintain gut barrier integrity and reduce inflammation, offering potential relief for IBD patients [57].
Clinical trials consistently demonstrate significant improvements in abdominal pain, bloating, and overall symptom severity in IBD patients following the low FODMAP diet [57, 58]. A meta-analysis involving 319 IBD patients across six studies revealed that adherence to a low FODMAP diet not only reduced specific gut symptoms but also enhanced functional symptoms while reducing symptom severity (excluding constipation) compared to a regular diet, suggesting its efficacy as a treatment option for IBD [56]. In a randomized 6-week trial, a low FODMAP diet improved fecal biomarkers of intestinal inflammation. It reduced disease activity in patients with CD, even those with mainly quiescent disease, with some improvement in patient quality of life [62]. The benefits of the low FODMAP diet also extend to pediatric IBD patients. In a randomized, single-blinded study involving 84 children aged 5–15 years, those on a low FODMAP diet for six weeks experienced decreased severity of abdominal pain, improved gastrointestinal symptoms, and enhanced health-related quality of life compared to those on a standard diet [54].
The Low FODMAP diet primarily reduces fermentable substrates available to gut bacteria, thereby altering the composition and activity of the gut microbiota [63]. This shift toward less fermentative species is believed to potentially mitigate inflammatory responses in the gut [61]. Despite its efficacy in relieving IBD symptoms, the use of the Low FODMAP diet is recommended for short-term management due to concerns about long-term effects. Prolonged adherence may reduce the diversity and abundance of beneficial gut bacteria and diminish prebiotic fibers essential for maintaining healthy bacterial populations [63, 64]. Also, the diet does not improve colon inflammation and stool consistency in IBD [65, 66]. Studies have suggested that while the diet can alleviate symptoms, it may not significantly impact underlying inflammation or long-term clinical remission. This detail is noteworthy because reducing symptoms without addressing inflammation may mask disease activity rather than control it [67].
However, the diet can be restrictive and may lead to nutritional deficiencies if not properly managed. The diet can decrease key nutrients like short-chain fatty acids such as butyrate, which are crucial for gut epithelial health [68]. There are also concerns about the potential for nutritional deficiencies with prolonged use of the Low FODMAP diet, including fiber, vitamins, and minerals, which could contribute to malnutrition [59]. Additionally, the diet’s restrictive nature involves avoiding a wide range of foods, which can be complex and challenging for patients without comprehensive guidance from a dietitian. This complexity often leads to difficulties in long-term adherence, especially without ongoing professional support. Social and psychological factors further complicate the adoption of a Low FODMAP diet. Patients may experience social isolation, anxiety, and reduced quality of life due to limitations in participating in social events, eating out, and explaining their dietary needs to others [61]. Lastly, the cost and availability of Low FODMAP foods and ingredients present additional challenges, potentially placing financial strain on patients. Effective implementation typically requires the involvement of specialized dietitians, which may not be accessible or affordable for all individuals.
The elemental diet is a nutritionally complete, liquid formula composed of easily digestible, low-residue nutrients in their simplest forms typically amino acids, monosaccharides, minimal fat, and essential vitamins and minerals. This diet involves consuming a formula that contains nutrients in their simplest form, making them easier to digest and absorb. This dietary approach has been explored as a therapeutic strategy in managing IBD, particularly CD [69]. The elemental diet reduces the digestive workload by providing pre-digested nutrients, allowing the intestines to rest and potentially heal. The diet can also alter the composition of gut bacteria, which may reduce inflammatory triggers [70].
Furthermore, an elemental diet eliminates complex proteins and decreases potential antigenic stimulation of the immune system [26]. In addition, the diet aims to provide adequate nutrition when traditional food intake is limited or impossible due to severe symptoms. The elemental diet is stipulated to be free from common allergens and antigens, which might provoke an immune response in the gut. It is usually given orally but can be administered via a nasogastric tube. Treatment typically lasts several weeks, during which patients consume only the elemental formula, followed by continuous monitoring by professionals, which is essential to ensure nutritional adequacy and address potential side effects [71].
Elemental diets are not commonly used as primary therapy but may be beneficial in specific scenarios, such as pre-surgical nutritional optimization in IBD patients [72]. Several studies have shown that elemental diets can induce remission in active Crohn’s disease, with remission rates comparable to corticosteroid therapy in some cases. The diet works by reducing the antigenic load in the gut, minimizing the immune response, and allowing the bowel to heal [73]. The evidence of elemental diet use is present [74] but less robust in ulcerative colitis patients. While it can still reduce symptoms and inflammation in some patients, it is generally not used as a first-line treatment for inducing remission in UC [75].
The elemental diet can significantly reduce gastrointestinal symptoms, leading to an initial improvement in physical well-being. This symptom relief can promote QoL, particularly in patients who have not responded well to other treatments [69]. Another significant benefit of the elemental diet is its steroid-sparing effect. It can be used as an alternative to corticosteroids, particularly in patients who are steroid-dependent or those who experience significant side effects from steroids [72]. Additionally, the elemental diet has effectively lowered inflammatory markers like C-reactive protein (CRP) and fecal calprotectin, indicating decreased disease activity. This reduction in inflammation is associated with clinical improvements and mucosal healing [75]. Additionally, the diet influences the gut microbiota, potentially offering anti-inflammatory benefits. However, the specific changes it induces in the microbiome are complex, and the long-term impact of these alterations remains under investigation [75].
Although this diet is nutritionally complete, its liquid nature and the limited variety of flavors can lead to difficulties with long-term adherence. Patients may experience difficulties related to taste, which they often find unpalatable, and a lack of satisfaction with meals, which can impact their overall well-being [35]. Long-term adherence to the diet is difficult, and reintroducing normal foods can sometimes lead to a relapse of symptoms. Thus, the elemental diet is more commonly used as a short-term management rather than a long-term solution. While the elemental diet is effective for the management of IBD, it is typically not sustainable as a long-term strategy, and patients must shift to a more varied diet. A well-planned modification, often guided by a nutritionist, is essential to avoid causing a setback and to ensure nutritional sufficiency [76].
The Anti-Inflammatory Diet (AID) is increasingly recognized as a promising nutritional approach for managing IBD. This diet focuses on reducing intestinal inflammation, promoting mucosal healing, and supporting overall digestive health [77]. Evidence indicates that the inflammatory potential of dietary patterns significantly influences disease activity, with pro-inflammatory diets correlating with higher rates of active disease even after adjusting for clinical and demographic factors [78]. Conversely, adherence to anti-inflammatory dietary patterns rich in omega-3 fatty acids, antioxidants, fiber, and phytonutrients has been associated with decreased mucosal inflammation and improved clinical outcomes in IBD [79].
This diet emphasizes unprocessed or minimally processed foods such as fresh fruits and vegetables, lean proteins, whole grains, and healthy fats. Processed foods, typically high in additives, preservatives, and unhealthy fats, are discouraged due to their potential to worsen inflammation. Similarly, a wide range of fruits and vegetables, particularly those high in antioxidants, are encouraged, as antioxidants help combat oxidative stress, a key contributor to inflammation [80]. This diet also encourages the consumption of prebiotics and probiotics, which can promote a balanced gut microbiome composition [81].
Examples of anti-inflammatory foods include berries, leafy greens, cruciferous vegetables, and root vegetables. However, during active IBD flare-ups, some raw or high-fiber vegetables may need to be cooked or temporarily avoided to prevent irritation. Omega-3 fatty acids, found in fatty fish such as salmon, mackerel, and sardines, as well as in flaxseeds, chia seeds, and walnuts, are also central to the diet due to their strong anti-inflammatory properties. Balancing omega-3 intake while reducing omega-6 fatty acids (commonly found in processed vegetable oils) is a key strategy in the AID [82]. Common triggers for inflammation or gut irritation in IBD patients such as spicy foods, caffeine, alcohol, and certain high-fiber foods like nuts and seeds are often avoided, particularly during flare-ups [83].
The AID is not one-size-fits-all; it is often personalized based on an individual’s symptoms, triggers, and nutritional needs. A dietitian specializing in IBD can help tailor the diet to meet a patient’s specific needs, making it a valuable complement to medical treatments and improving the quality of life for those with IBD [84].
In recent years, the effects of the anti-inflammatory diet on clinical remission and disease activity in IBD patients have been a subject of increasing interest. Evidence suggests that it can contribute to inducing remission, modulating the immune response, and promoting mucosal healing, particularly when combined with other therapies [85].
Other studies have shown that patients adhering to an AID experience a reduction in IBD symptoms, such as abdominal pain, diarrhea, and fatigue, which are frequently associated with active disease. The diet may not be as immediately potent as more restrictive diets like the elemental diet, but it offers a more balanced and sustainable approach supporting long-term remission. Its effects are often gradual, and its success in inducing remission may depend on the severity of the disease and patient adherence [81].
Furthermore, the anti-inflammatory diet is frequently used alongside conventional medical treatments like biologics, immunosuppressants, and corticosteroids. As a complementary therapy, it can enhance the overall effectiveness of these medications, potentially allowing for lower doses and reducing associated side effects. Patients who follow an anti-inflammatory diet often require lower corticosteroid doses since the diet naturally helps manage inflammation. This is especially advantageous for those who are steroid-dependent or experience significant side effects from steroid use [79].
The anti-inflammatory diet has also reduced inflammatory markers like CRP and fecal calprotectin, often elevated in active IBD and associated with disease activity. By emphasizing fiber-rich, plant-based foods, the diet supports a healthier gut microbiome, increasing beneficial bacteria and reducing harmful species, which helps lower gut inflammation and improve outcomes. This approach promotes mucosal healing, reduces complications like strictures and fistulas in CD, and lessens colitis severity in UC. Unlike restrictive diets, it offers more variety and flexibility, encouraging long-term adherence and consistent disease control. It can be maintained during remission as a preventive strategy, with adjustments made during flare-ups to manage symptoms effectively [79].
The Specific Carbohydrate Diet (SCD) is a dietary approach to manage IBD by eliminating complex carbohydrates (disaccharides and most polysaccharides) that are harder to digest and may contribute to gut inflammation. These complex carbohydrates can ferment in the gut, leading to an overgrowth of harmful bacteria and yeast, exacerbating IBD symptoms [86]. Foods that contain these complex carbohydrates include grains, starchy vegetables, and most legumes [87]. In contrast, the diet permits the consumption of simple carbohydrates (monosaccharides), which are easier to digest and absorb. These include fruits, non-starchy vegetables, nuts, honey, and dairy products like homemade yogurt. Such foods are less likely to contribute to bacterial overgrowth and inflammation [88].
A key aspect of SCD is its emphasis on gut-healing foods such as homemade bone broth, non-starchy vegetables, lean proteins, and fruits, which provide essential nutrients while minimizing inflammation [89]. The diet also prohibits the consumption of all grains, including wheat, corn, rice, oats, and barley. Grains contain complex carbohydrates and gluten, which are thought to irritate the gut and trigger inflammation in susceptible individuals. The SCD instead promotes whole, unprocessed foods, as processed foods often contain additives and preservatives that can further irritate the gut lining [86]. Regarding dairy, SCD allows only lactose-free options, as lactose is a disaccharide excluded in this diet. However, homemade yogurt fermented for 24 hours is permitted because the extended fermentation process breaks down lactose, making it easier to digest [90].
While the SCD and modified SCD diets did not consistently relieve symptoms or inflammation in all IBD patients, some individuals may have benefitted [91]. Research and anecdotal evidence consistently point to the potential of SCD to significantly improve symptoms in IBD patients. Some patients have reported reaching and sustaining clinical remission by following the diet. The SCD may help reduce intestinal inflammation by eliminating specific carbs and sugars, slowing disease activity, and facilitating remission. However, the restricted diet necessitates considerable desire and support for long-term compliance, which might be challenging for certain individuals [91].
SCD is a potential therapy generally used with conventional medical therapies rather than as a stand-alone treatment. Studies have shown that combining SCD with medicines may improve its efficacy in treating IBD and maintaining remission [92]. However, the diet’s stringent nature raises concerns about potential nutritional inadequacies caused by the absence of specific food categories. Proper dietary planning and supplements can reduce these dangers. Despite the encouraging findings of short-term studies and anecdotal accounts, further study is needed to understand the long-term consequences of SCD and maximize its function in IBD therapy [92].
The Mediterranean Diet is renowned for its anti-inflammatory properties and emphasis on whole, nutrient-dense foods, making it a promising approach for managing IBD. This diet emphasizes consuming fruits and vegetables, particularly leafy greens, tomatoes, peppers, berries, and citrus fruits, which provide essential nutrients that minimize oxidative stress and inflammation in IBD management [93]. Consistent with these benefits, evidence-based guidelines now recommend minimizing the intake of ultra-processed foods, especially those containing emulsifiers, and adopting a Mediterranean dietary pattern, which the American Gastroenterological Association endorses for most patients with IBD, unless contraindicated [55].
A central feature of the Mediterranean Diet is the consumption of healthy fats, especially from extra virgin olive oil, rich in monounsaturated fats with anti-inflammatory properties. Whole grains such as oats and quinoa are also encouraged, as they provide essential fiber for gut health. However, during IBD flare-ups, fiber intake may need to be adjusted based on individual tolerance [94]. Legumes, including lentils, chickpeas, beans, and nuts like almonds, walnuts, and hazelnuts, are integral to the Mediterranean Diet. They offer plant-based protein, fiber, and healthy fats, though some individuals with IBD may need to consume these foods in moderation or well-cooked, pureed forms to avoid gut irritation [95].
Likewise, Dairy products, including cheese and yogurt, are included in the diet but should be consumed in moderation, with an emphasis on fermented dairy products that contain probiotics, which support gut health. The diet also incorporates herbs and spices like garlic, oregano, rosemary, and turmeric for flavor. Many of these herbs and spices contain valuable antioxidants in managing IBD. The diet minimizes processed foods, refined sugars, and unhealthy fats, which can worsen inflammation and contribute to poor gut health [96, 97]. Water is the primary beverage; herbal teas are encouraged, while sugary drinks and excessive caffeine are limited.
The Mediterranean Diet also encourages a healthy, diversified, and sustainable approach to eating, with meals frequently consumed in a leisurely and communal setting. This comprehensive approach can improve general well-being and reduce stress. Above all, the diet’s versatility and durability make it a reasonable long-term approach to managing IBD [98].
The Mediterranean Diet is thought to lower systemic and intestinal inflammation, which are key features of IBD etiology. Clinical research and observational studies indicate that this diet can help IBD patients with symptoms such as stomach discomfort, diarrhea, and exhaustion. While the diet may not produce remission in all situations, it can supplement medicinal therapy and improve symptom management [99]. In addition, studies have linked the Mediterranean Diet to decreased recurrence rates, decreased levels of inflammatory markers such as interleukin-6 (IL-6), and improved gut microbiota composition, all of which enhance the QoL for individuals with IBD [99].
Furthermore, the diet is enriched in vital nutrients and vitamins, which help avoid nutritional deficits like anemia and osteoporosis, which are frequent in people with IBD. Its well-rounded strategy promotes general health, including maintaining a healthy weight and preventing weight loss during flare-ups. The diet’s emphasis on savory and varied meals can also make it pleasurable and fulfilling, improving long-term adherence and lowering dietary weariness [98, 100]. Although the Mediterranean Diet is often accessible and affordable, the higher costs of fresh, healthful foods can be a barrier for people with limited resources [101, 102].
The makeup of the gut microbiota is tightly related to the dynamics of IBD [45, 103]. In the early stages of IBD, significant alterations or dysbiosis in the gut microbiota are observed in patients compared to healthy individuals [104]. Research suggests that these changes may be more pronounced in CD than in UC [105, 106]. In IBD patients, beneficial bacteria such as Bifidobacterium longum in UC and Eubacterium rectale, Faecalibacterium prausnitzii, and Roseburia intestinalis in both CD and UC are significantly reduced. At the same time, there is an increased prevalence of harmful bacteria like Bacteroides fragilis [107].
Dysbiosis also disrupts the production of crucial microbial metabolites necessary for gut health. Short-chain fatty acids (SCFAs), particularly butyrate, are known for their beneficial properties and role in maintaining gut mucosal integrity [108]. When dysbiosis occurs, the reduced production of SCFAs can contribute to increased inflammation and compromise the intestinal barrier, thereby worsening IBD symptoms. Also, dysbiosis can impact bile acid metabolism, which is essential for regulating gut microbial populations. Disruptions in bile acid metabolism can create an environment that encourages the growth of pathogenic bacteria, further fueling inflammation and disease progression [109].
Several studies have highlighted that diets high in processed foods, unhealthy fats, and sugars, often referred to as the Western diet, facilitate inflammation and exacerbation of IBD symptoms due to their negative impacts on intestinal permeability and gut microbiome composition [110, 111]. In contrast, a diet rich in fiber, especially fruits and vegetables, promotes the production of SCFAs such as butyrate, which have anti-inflammatory properties and help maintain intestinal integrity [112]. A Western diet is also linked to higher levels of bacteria in the phylum Firmicutes, Proteobacteria, and Enterobacteriaceae, which are associated with dysbiosis and related health issues [113].
While the low-FODMAP diet effectively ameliorates functional IBD symptoms, long-term adherence can adversely affect gut microbiota [114]. Individuals following a low-FODMAP diet often experience a decline in healthy gut bacteria such as Bifidobacteria spp, Akkermansia muciniphila, and species of the Propionibacteriaceae family [115]. A targeted analysis comparing a low FODMAP diet group to a control diet group found that patients following the low FODMAP diet had a significantly reduced abundance of Bifidobacterium adolescentis, Bifidobacterium longum, and Faecalibacterium prausnitzii compared to those on the control diet. However, the two groups had no significant differences in microbiome diversity or inflammatory markers [57]. Nonetheless, more extensive studies are needed to assess whether these microbial changes pose long-term risks, particularly for non-celiac individuals, and determine whether these effects are reversible. To mitigate the potential negative impact on gut microbiota, supplementing the low-FODMAP diet with probiotics has been shown to help restore balance, particularly by increasing levels of Bifidobacterium [116].
Furthermore, the implementation of a Mediterranean diet was associated with a lower Firmicutes/Bacteroidetes ratio, increased Bifidobacterium spp levels, increased short-chain fatty acid (SCFA) production, lower Escherichia coli levels, and increased gut microbiota diversity and richness [117]. The diet’s emphasis on reduced intake of processed foods, saturated fats, and certain amino acids like choline and L-carnitine, combined with a focus on plant-based foods, healthy fats, and fiber, contributes to greater microbial diversity [118]. This includes higher levels of certain bacteria (such as Bacteroides, Lactobacillus, and Bifidobacterium) while reducing the number of pathogenic microorganisms, such as those from the Proteobacteria phylum. The microbial environment fostered by the Mediterranean diet has also been associated with increased SCFA production, which may have a protective effect against the disease [119].
Additionally, a study by Olendzki and associates evaluated the impact of an anti-inflammatory Diet on the gut microbiome and disease outcomes in IBD. The study showed that adherence to the diet improved clinical symptoms and promoted a shift in gut microbiota composition, increasing the abundance of anti-inflammatory bacteria like Faecalibacterium prausnitzii and reducing pathogenic species. Likewise, Yamamoto and colleagues [120] examined patients with Crohn’s disease using an elemental diet. The study found that an elemental diet reduces gut inflammation and modulates the microbiome, decreasing the abundance of pro-inflammatory bacteria such as those in the family Enterobacteriaceae while increasing beneficial species like Bifidobacteria spp [120]. Another group of researchers conducted a pilot study evaluating the effects of the Specific Carbohydrate Diet (SCD) in pediatric CD. The study revealed that SCD significantly altered the gut microbiome by sustaining the growth of anti-inflammatory bacteria such as Bacteroides and reducing pro-inflammatory species. Clinical improvements in disease activity were also observed [121].
Prebiotics, often called fermentable dietary fibers, are non-digestible food components that selectively encourage the growth and activity of beneficial gut bacteria, thereby supporting overall health [122]. Unlike probiotics, which introduce live beneficial microorganisms into the gut, prebiotics nourish these microbes, enhancing their survival and function [123]. Common prebiotics like inulin and fructooligosaccharides (FOS) is naturally found in everyday foods such as onions, garlic, bananas, asparagus, leeks, and chicory root [124]. Through fermentation, these prebiotics are metabolized by gut bacteria, particularly Bifidobacteria and Lactobacilli. This process produces SCFAs like acetate, propionate, and butyrate, which are critical in maintaining gut health [125]. SCFAs lower the pH of the gut environment, which helps inhibit the growth of pathogenic bacteria while fostering the proliferation of beneficial microbes.
While fructan-containing compounds such as inulin are the most recognized prebiotics, other fibers exhibit prebiotic properties and may benefit people with IBD. The potential of prebiotics and dietary fiber for treating IBD has been described [27, 126, 127, 128]. For example, inulin supplementation has been associated with increased levels of Bifidobacterium, which has been linked to improved gut health and reduced inflammation, particularly in patients with UC [129, 130]. However, the effectiveness of prebiotics in IBD still needs to be consistent across studies. This difference can be attributed to the diversity of individual microbiome composition and variations in IBD cases [130, 131].
Probiotics are described as “live microorganisms, primarily beneficial bacteria, which, when utilized in sufficient amounts, provide health benefits to the host.” These beneficial effects often include improving gut health, enhancing the immune system, and supporting intestinal microbiota balance [132]. The widely used probiotic strains belong to the genus Bifidobacterium and Lactobacillus, and their functions include detoxifying harmful substances, altering mycotoxins in food, synthesizing vitamins, and fermenting undigested fibers in the colon [133, 134]. Individuals can ingest probiotics through fermented foods or drinks [125], including dairy and non-dairy options, or as dietary supplements [135]. An effective daily dose of probiotics is typically around 109 colony-forming units (CFU) [136].
Studies have demonstrated that supplementation with probiotic mixtures can significantly alter the gastrointestinal microbiome in patients with IBD, contributing to therapeutic outcomes [137, 138, 139]. For instance, one study reported a notable decrease in the relative abundance of Escherichia spp. during the probiotic therapy [140]. In patients with UC, the intake of a probiotic mixture containing Lactobacillus and Bifidobacterium led to an increased presence of certain Proteobacteria genera and a reduction in Gram-negative rods, indicating favorable shifts in the microbial community [137]. Furthermore, probiotics are well-recognized for modulating host immune responses, an effect observed in several studies on probiotic therapy for IBD [141]. Another study explored using an aerobe-containing probiotic mixture to alleviate Dextran Sodium Sulfate (DSS)-induced chronic colitis in mice, revealing the anti-inflammatory properties of the probiotics in mitigating colitis symptoms [142]. For CD however, the results of the clinical trials are less convincing [143].
Synbiotics are dietary products that combine prebiotics and probiotics to deliver complementary health benefits [144]. By fusing these two ingredients, synbiotics aim to enhance the effects of prebiotics and probiotics through synergy and optimize their combined benefits. There are limited studies about the role of synbiotics in IBD and gut microbiome modulation. As research on synbiotics as an intervention in IBD continues to grow, the few available studies demonstrate positive health effects. For instance, studies have reported that synbiotics can reduce IBS symptoms and improve overall bowel function [145]. Evidence also suggests that synbiotics can modulate the immune response by promoting the action of immunoglobulins and regulating the function of immune cells, thereby helping to reduce inflammation [146, 147].
Extensive research has revealed that diet can shape mucosal immune responses,
indirectly by remodeling microbiome composition and microbial metabolism, and
directly through bioactive nutrients that influence epithelial and immune
signaling [12, 148]. Microbiome-derived metabolites provide a key mechanistic
link between dietary inputs and immune outcomes. Commensal fermentation products
and other microbially modified metabolites, including SCFAs, bile acids, and
tryptophan–pathway derivatives, can influence barrier integrity and immune
regulation. They support regulatory programs such as Treg differentiation and
IL-10 associated anti-inflammatory signaling [12, 149, 150]. In contrast,
dysbiosis and impaired barrier function can increase exposure to microbial
antigens and inflammatory cues. This exposure shifts immune balance toward
pro-inflammatory responses, including Th17 polarization and cytokine programs
involving IL-17, IL-6, and TNF-
Additionally, this regulatory–inflammatory balance is context-dependent. Baseline microbiome structure and host factors influence how dietary changes translate into microbial metabolic outputs and downstream immune effects. This context dependence helps explain why similar dietary interventions may yield variable immunologic and clinical outcomes across individuals with IBD [153].
Microbiome-derived metabolites represent a critical interface through which dietary inputs are translated into host epithelial and immune responses. Among these, SCFAs, including acetate, propionate, and butyrate, are among the most extensively studied mediators of diet–microbiome–host interactions in IBD. SCFAs are primarily generated through microbial fermentation of dietary fibers and exert pleiotropic effects on intestinal homeostasis [108, 119, 127, 154, 155]. Butyrate serves as a significant energy source for colonocytes, enhances epithelial tight junction integrity, maintains the mucus layer, and limits epithelial permeability [112, 156, 157, 158]. Acetate and propionate also support colonic health by strengthening epithelial barrier integrity, regulating immune responses, and promoting microbial cross-feeding to maintain a balanced gut ecosystem [159, 160]. In parallel, SCFAs modulate immune responses through G-protein–coupled receptor signaling and histone deacetylase inhibition, thereby supporting regulatory immune programs and dampening pro-inflammatory cytokine production [161, 162].
While SCFA-mediated effects on epithelial energetics and immune regulation are well established, emerging mechanistic evidence highlights additional diet-sensitive microbial metabolites, including bile-acid and tryptophan-derived pathways, that may further shape barrier integrity and inflammatory tone in IBD. Dietary patterns low in fermentable fiber and enriched in saturated fats can disrupt protective mechanisms of SCFAs by reducing their availability and altering microbial metabolic capacity [163]. Such dietary shifts are also associated with changes in bile-acid metabolism, including increased conversion of primary bile acids into secondary bile acids by gut microbes [164]. While bile acids play essential roles in lipid absorption and metabolic signaling, excessive accumulation of specific secondary bile acids has been linked to epithelial stress, altered barrier function, and inflammatory signaling in genetically or immunologically susceptible hosts [165, 166].
Gut microbes can also convert dietary tryptophan into a range of indole derivatives that activate aryl hydrocarbon receptor signaling in epithelial and immune cells [166]. Activation of this pathway supports epithelial renewal, tight junction stability, and balanced mucosal immune responses [167, 168]. Disruption of microbial tryptophan metabolism or reduced availability of indole derivatives has been associated with impaired barrier integrity and heightened inflammatory activity in IBD, further underscoring the functional consequences of diet-microbiome interactions beyond taxonomic composition alone.
Collectively, these metabolite-mediated pathways illustrate how dietary modulation of microbial function, rather than microbial composition alone, can influence epithelial barrier integrity and inflammatory tone in IBD. This functional perspective provides a mechanistic rationale for focusing on metabolite outputs when considering microbiome-informed dietary strategies.
Biologic therapies, including anti–tumor necrosis factor-
Several observational studies have reported that patients who respond to anti-TNF therapy tend to exhibit greater microbial diversity and enrichment of taxa associated with barrier integrity and anti-inflammatory function, including short-chain fatty acid–producing bacteria, whereas non-response has been linked to dysbiosis and expansion of pro-inflammatory microbial signatures [39, 170]. Beyond taxonomic differences, functional microbial features appear particularly relevant to biological response. Metagenomic and metabolomic analyses indicate that responder-associated microbiomes are enriched for pathways involved in SCFA production, bile acid transformation, and tryptophan metabolism, which may collectively support epithelial repair, immune regulation, and mucosal healing [172].
Diet represents a key modulator of these microbial features and may indirectly influence biologic efficacy by shaping the metabolic and inflammatory capacity of the gut microbiome. Dietary patterns that support SCFA production and microbial diversity may help reinforce a responder-like microbial ecosystem. In contrast, diets that promote proteolytic fermentation or secondary bile acid accumulation could exacerbate inflammatory pathways that undermine therapeutic response [172, 173].
Despite growing interest in dietary modulation of the gut microbiome as a therapeutic strategy in IBD, the current evidence base remains fragmented and methodologically heterogeneous. Many nutritional intervention studies in IBD are small, short-term, and variable in design; they also differ in diet composition, duration, adherence assessment, and patient populations [174, 175]. Microbiome-related endpoints also vary substantially across studies, encompassing differences in sequencing approaches (16S rRNA profiling versus metagenomics), sampling sites (stool versus mucosal), and analytical pipelines [176, 177, 178, 179, 180]. Clinical outcomes are inconsistently defined, ranging from symptom-based indices to objective measures of inflammation, such as fecal calprotectin or endoscopic scoring [181, 182].
Together, these limitations contribute to variable findings and complicate direct translation of microbiome–diet associations into clinical practice. At the same time, host genetics, immune microenvironment, and environmental context impose biological and practical constraints on how uniformly dietary interventions can reshape microbiome composition across individuals. These challenges underscore the need for integrative frameworks that move beyond descriptive associations toward standardized, mechanistically informed approaches capable of accommodating biological constraint and real-world variability. Accordingly, the next section integrates current mechanistic evidence to propose a structured, microbiome-informed framework for personalization of dietary interventions in inflammatory bowel disease.
As mentioned in the preceding sections, diet and gut microbiome modifications are increasingly acknowledged as critical components of IBD management. Hence, personalized nutrition based on gut microbiome modulations represents a novel and underexplored approach to IBD treatment. A study by Zeevi and colleagues [183] revealed that tailoring diets based on gut microbiota profiles could improve postprandial glycemic responses, offering insights into how personalized dietary strategies could be applied in managing chronic conditions like IBD. Additionally, emerging research shows that certain microbial compositions correlate with better responses to anti-inflammatory diets. For example, patients with higher levels of Faecalibacterium prausnitzii tend to experience more significant benefits from fiber-rich diets, which enhance the production of SCFAs and contribute to reduced inflammation [184]. These findings underscore the potential of microbiome-driven dietary interventions as a personalized strategy for IBD management.
The human gut microbiota varies widely between individuals due to factors such as nutrition, antibiotic use, hygiene, and mode of birth. Among these, the influence of diet is substantial, often outweighing the role of genetic factors [185]. Because of these individual differences, the same diet does not provide all patients with the same degree of response [186]. The stages outlined below describe a unique approach for designing personalized diets based on microbiome profiles (Fig. 2).
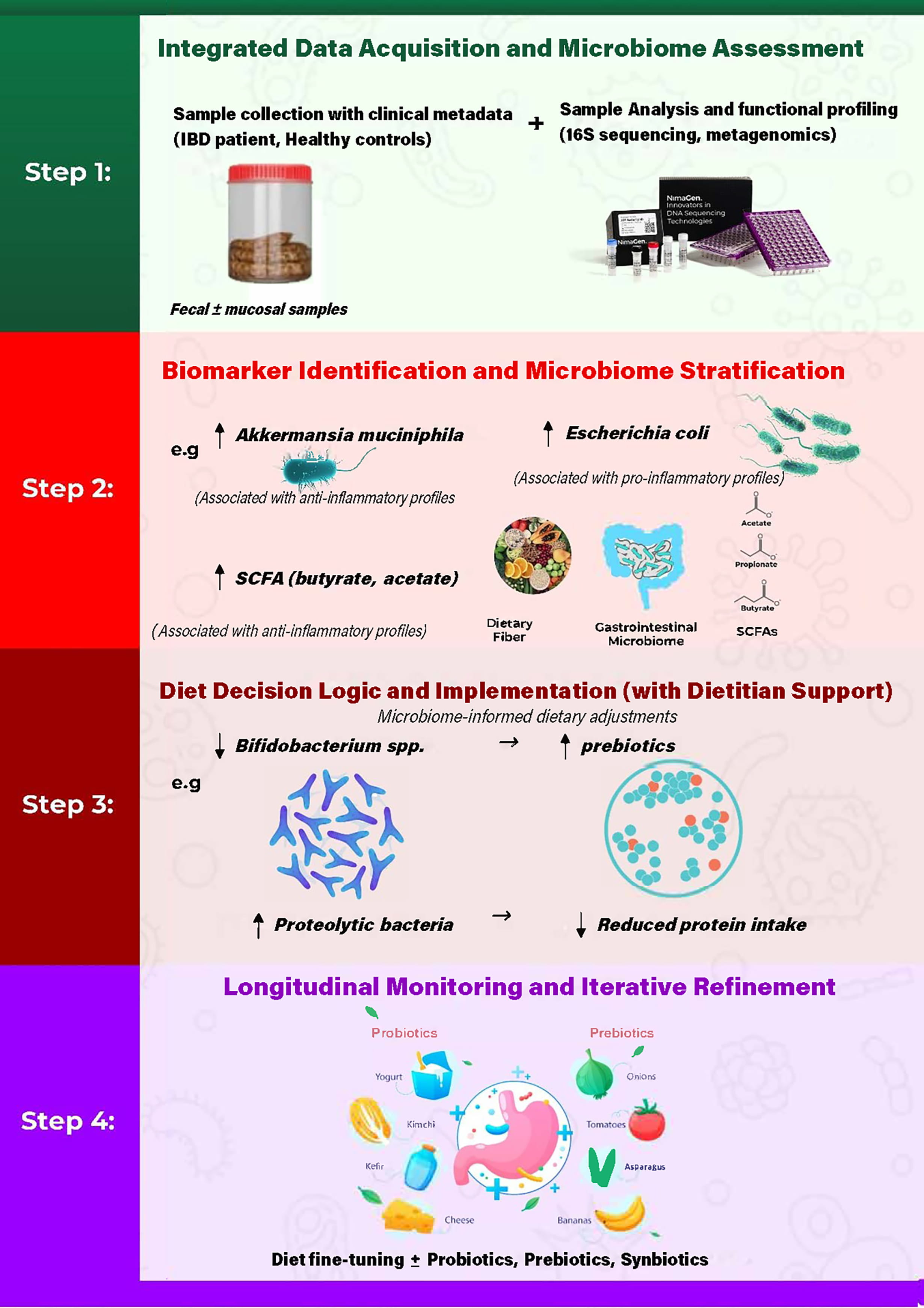 Fig. 2.
Fig. 2.
Proposed workflow for microbiome-informed dietary personalization in inflammatory bowel disease. The framework integrates (i) data acquisition and microbiome assessment with relevant clinical metadata, (ii) biomarker identification and microbiome stratification, (iii) diet decision logic and implementation with dietitian support, and (iv) longitudinal monitoring with iterative refinement to account for temporal variability in microbiome features and disease activity.
The proposed framework does not seek to impose uniform compositional shifts in the gut microbiome across individuals, recognizing that baseline microbial structure is shaped by host genetics, early-life exposures, and long-standing environmental factors that may limit the extent of taxonomic remodeling. Instead, it emphasizes modulation of microbial function and metabolite output, which has been shown to remain responsive to dietary intervention even in the presence of genetic constraints.
To enhance clinical relevance and real-world applicability, the framework recognizes age, cultural dietary practices, regional food availability, and other environmental factors as contextual modifiers when translating microbiome-informed recommendations into practice [187]. Dietary patterns, food accessibility, and cultural norms substantially influence habitual nutrient intake and microbiome configuration, and failure to account for these factors may limit adherence and sustainability of personalized dietary strategies. By embedding microbiome-informed recommendations within culturally and geographically appropriate dietary contexts, this framework aims to support feasible, patient-centered interventions that can be adapted across diverse populations.
The first step in microbiome-informed dietary personalization is comprehensive data acquisition that integrates microbiome profiling with detailed clinical and contextual phenotyping. Microbiome assessment is most commonly performed using fecal samples because they are noninvasive, readily obtainable, and suitable for longitudinal monitoring, making them the primary biospecimen in most gut microbiota studies [188]. While fecal sampling provides a practical overview of luminal microbial communities, mucosal-associated sampling obtained through endoscopy, biopsy, or luminal brushing may offer complementary insights into host–microbe interactions at the epithelial interface in selected clinical or research settings [188].
Collected samples undergo sequencing and functional profiling using next-generation sequencing–based approaches. Among these, 16S rRNA gene sequencing remains the most widely used method for characterizing microbial community composition and diversity. However, higher-resolution approaches, including shotgun metagenomics, meta-transcriptomics, and targeted or untargeted metabolomics, enable functional characterization of microbial metabolic capacity and activity, which is increasingly recognized as more informative for translational applications [179, 189]. Together, these platforms provide complementary insights into microbial structure, functional potential, and metabolite production at the time of sampling.
In addition to microbiome profiling, detailed clinical phenotyping is essential to contextualize microbial data and guide interpretation. Key clinical variables include disease subtype and activity indices, objective inflammatory biomarkers such as C-reactive protein and fecal calprotectin, medication exposure including biologics and immunomodulators, and recent antibiotic use [190]. Dietary intake, age, geographic location, and other environmental covariates should also be captured, as these factors substantially influence both microbiome composition and metabolic output. Integrating microbiome-derived data with clinical and contextual information at this initial stage establishes the foundation for downstream functional analysis and personalized dietary decision-making.
Following integrated microbiome profiling and clinical phenotyping, the next step is to identify biologically and clinically relevant microbial features that can serve as candidate biomarkers for stratification. Depending on the analytical platforms used in Step 1, this process may include evaluating microbial taxa, functional pathways, and metabolite-associated signatures. For example, relative enrichment of commensal taxa such as Faecalibacterium prausnitzii and Akkermansia muciniphila has been associated with epithelial barrier integrity and anti-inflammatory states. In contrast, the expansion of pathobionts, such as Escherichia coli, has been linked to mucosal inflammation and disease activity in IBD [191].
Biomarker identification also encompasses broader patterns of dysbiosis, including reduced microbial diversity and disproportionate representation of pro-inflammatory microbial communities, which are commonly observed in IBD [192]. Functional readouts further refine this stratification by highlighting differences in microbial metabolic capacity, such as altered abundance of short-chain fatty acid–producing bacteria and associated reductions in butyrate, acetate, or propionate production [193]. These functional features may correlate with disease severity, inflammatory burden, and treatment response, providing a more nuanced basis for patient stratification than compositional metrics alone [194].
Importantly, biomarker selection at this stage is not intended to define fixed diagnostic thresholds but rather to identify interpretable microbial and functional features that can inform downstream decision logic. This stratified view of the microbiome bridges descriptive profiling and personalized dietary intervention by linking microbial features to host inflammatory status.
Following biomarker identification and microbiome stratification, dietary recommendations are then developed using structured decision logic that integrates microbial features with clinical context. This step translates microbiome-informed signals into personalized dietary strategies while accounting for disease activity, medication use, nutritional status, and patient-specific constraints.
For instance, reduced abundance of beneficial taxa such as Bifidobacterium spp. or diminished short-chain fatty acid–producing capacity may support dietary strategies emphasizing fermentable fibers or prebiotic-rich foods [129]. Conversely, enrichment of proteolytic or pro-inflammatory microbial signatures, including Escherichia coli or certain Clostridium species, may prompt moderation of specific dietary components, such as excess animal protein or highly processed foods, that can exacerbate inflammatory metabolic pathways [195, 196]. Importantly, these dietary adjustments are not applied as fixed rules, but as flexible, evidence-informed options tailored to individual microbiome profiles and clinical status.
Registered dietitians or trained clinicians would guide the implementation of dietary strategies to ensure nutritional adequacy, cultural relevance, and long-term adherence. Dietitian support allows microbiome-informed recommendations to be translated into practical, patient-centered nutritional plans that can be safely integrated with ongoing pharmacologic therapy and adapted over time as clinical and microbiome parameters evolve.
Personalized dietary strategies informed by microbiome profiling require ongoing evaluation to account for temporal variability in microbial composition, host response, and disease activity. Longitudinal monitoring integrates periodic reassessment of the gut microbiome with clinical follow-up to evaluate how dietary interventions influence microbial function, inflammatory burden, and patient-reported outcomes over time.
Clinical monitoring may include symptom tracking, objective biomarkers of inflammation such as fecal calprotectin or C-reactive protein, and, where appropriate, repeat microbiome profiling to assess functional and compositional shifts. These data provide feedback on the effectiveness and tolerability of dietary interventions within the broader context of pharmacologic therapy and disease course.
Based on longitudinal feedback, dietary strategies can be iteratively refined rather than replaced, allowing adjustments in dietary composition, fiber sources, or food patterns to better align with evolving microbiome features and clinical needs. This adaptive approach acknowledges that microbiome-informed nutrition is dynamic and context-dependent, and it supports sustained personalization through continuous learning rather than static intervention.
Collectively, this framework illustrates how integrating microbiome profiling with clinical phenotyping, biomarker-informed stratification, structured dietary decision-making, and longitudinal monitoring may support more individualized nutritional strategies in inflammatory bowel disease. By emphasizing functional microbial outputs and iterative refinement rather than static dietary prescriptions, this approach aligns with the dynamic nature of both the gut microbiome and IBD disease course. Nevertheless, prospective studies are needed to evaluate the feasibility, durability, and long-term clinical impact of microbiome-informed dietary personalization, including its effects on disease activity, treatment response, and patient-centered outcomes.
Although there is considerable interest in using microbiomes to achieve various health outcomes, significant progress must be made before microbiome-based diagnostics are incorporated into standard clinical care.
One of the primary challenges of utilizing a personalized approach for diet customization based on microbiome profiles is that the gut microbiome is highly personalized and heterogeneous. This heterogeneity makes it difficult to generalize microbiome-based dietary strategies across a broader patient population [197]. Additionally, while research has linked certain microbial groups with IBD and diet, the exact mechanisms by which these microbes influence inflammation and disease progression remain unclear. This knowledge gap limits the precision of personalized dietary interventions [91]. The lack of robust, long-term randomized clinical trials makes it difficult to develop evidence-based guidelines for the use of microbiome-responsive diets in clinical practice. Additionally, trials often have methodological inconsistencies, making it hard to compare results across studies [198, 199]. Ultimately, microbiome sequencing, analysis, and interpretation are costly and time-consuming. Specialized dietary adjustments also demand ongoing patient education, monitoring, and support from dietitians, which might only be feasible in some healthcare settings. Moreover, the cost and accessibility of recommended foods can pose barriers to patients [183, 197].
Looking ahead, the integration of microbiome-based approaches into IBD management will require further exploration of critical microbial signatures associated with disease progression and remission [178]. Artificial intelligence (AI) and machine learning advances could also be crucial in identifying patterns from large datasets and predicting individual responses to dietary interventions. Furthermore, more large-scale longitudinal studies are needed to evaluate the long-term effects of a microbiome-based diet on IBD outcomes [183, 199]. Collaboration between dietitians, gastroenterologists, microbiologists, and bioinformaticians is essential to develop comprehensive, personalized, evidence-based, clinically applicable, and scalable strategies across diverse healthcare settings [198, 199].
Personalized nutrition based on gut microbiome profiles is a novel frontier in IBD management; it offers a tailored intervention that aligns with an individual’s unique microbial composition. While the research is still evolving, early evidence suggests that such approaches could optimize dietary responses, ameliorate symptoms, and improve the quality of life for those with IBD. However, translating these advances into clinical practice requires overcoming challenges related to research, clinical application, and accessibility. By prioritizing integrative research and collaborative models of care, the field can move closer to realizing the full potential of microbiome-driven dietary interventions in IBD.
Several recommendations are suggested for integrating this dietary approach into the future of IBD therapy.
I. Clinicians should consider incorporating microbiome analysis, such as 16S sequencing or metagenomics, as part of the diagnostic and treatment process for IBD. This information can guide more personalized dietary recommendations that align with the patient’s unique microbiome composition.
II. Researchers should also participate in long-term studies to gather more evidence regarding the sustainability and mechanistic pathways involved in personalized diets for IBD.
III. Gastroenterologists, dietitians, and microbiome researchers need to collaborate to design tailored nutritional interventions that address the clinical and microbial aspects of IBD management.
IV. Educating patients on the rationale behind these tailored interventions and providing continuous support, including dietary counseling and monitoring, will improve adherence and optimize outcomes.
V. Personalized diets should be accessible and realistic for patients from diverse socioeconomic backgrounds. Addressing barriers such as cost, food availability, and cultural preferences is vital to ensuring that microbiome-based dietary interventions are effective and sustainable.
Conceptualization, KY and SU; Writing—Original Draft Preparation, KY; Writing—Review and Editing, KY, TMA, MAK, and SU; TMA and MAK, Substantial contributions to the conception or design of the work; Supervision, SU; Funding Acquisition, SU. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


