1 Department of General Surgery, The Affiliated Changsha Hospital of Xiangya School of Medicine, Central South University, 410005 Changsha, Hunan, China
2 Department of Hepatobiliary Surgery, Hunan Provincial People’s Hospital/The First Affiliated Hospital of Hunan Normal University, 410005 Changsha, Hunan, China
Abstract
Molecular targeted therapies play a crucial role in the management of hepatocellular carcinoma (HCC). Lenvatinib is a standard targeted agent for advanced HCC, while quercetin has emerged as a promising natural compound with anti-HCC potential. This study investigates the combined effect of quercetin and lenvatinib on HCC.
The human HCC cell line Huh7 and a nude mouse subcutaneous xenograft model were utilized. We evaluated cell proliferation, clonogenicity, invasion, migration, and apoptosis through Cell Counting Kit-8 (CCK-8), clonogenic, Transwell, and flow cytometry assays, respectively. Network pharmacology analysis was performed identify common targets. The protein expression levels of B-cell lymphoma 2 (Bcl-2), Bcl-2 associated X-protein (Bax), E-cadherin, N-cadherin, and protein tyrosine kinase 2 (PTK2) were assessed by Western blotting. In vivo experiments were conducted to validate the anti-tumor efficacy of the combination treatment.
The combination of quercetin and lenvatinib significantly enhanced inhibition of Huh7 cell proliferation, colony formation, invasion, and migration (p < 0.01) and promoted apoptosis (p < 0.01) compared to individual treatments. Mechanistically, the combination treatment decreased the expression of Bcl-2 and N-cadherin while upregulating Bax and E-cadherin. PTK2 was identified as a key shared target, and the combination most effectively suppressed its protein expression. In vivo, the combination group demonstrated a higher tumor inhibition rate (p < 0.01) and a Combination Index (CDI) of 0.753.
Quercetin significantly enhances the anti-tumor efficacy of lenvatinib, likely through synergistic inhibition of PTK2 expression.
Keywords
- carcinoma
- hepatocellular
- quercetin
- lenvatinib
- PTK2
Hepatocellular carcinoma (HCC) is one of the most prevalent and lethal malignancies globally. Its high incidence and mortality rates are especially high in regions with endemic hepatitis B virus infection and a high prevalence of cirrhosis, posing a substantial burden to global public health [1, 2, 3].
Lenvatinib, a multi-targeted tyrosine kinase inhibitor, exerts anti-tumor effects by blocking kinases including vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR), and platelet-derived growth factor receptor (PDGFR), thereby disrupting tumor angiogenesis and signaling pathways regulating cell proliferation. It is established as a first-line standard treatment for advanced HCC [4, 5, 6]. However, its effectiveness is limited by the development of resistance. The mechanism behind lenvatinib resistance is complex and multifactorial. Tumor cells can bypass the inhibitory effects of drugs by activating alternative signaling pathways, including c-MET and EGFR. As a result, tumor cells undergo epithelial-mesenchymal transition or increase their stemness, thereby enhancing their invasiveness and survival capabilities. Immunosuppressive cells and fibroblasts in the tumor microenvironment also contribute to the development of drug resistance in tumor cells [7, 8]. Consequently, research is focusing on developing strategies to enhance lenvatinib sensitivity and effective combination therapies for HCC treatment.
Quercetin, a naturally occurring flavonoid found in various vegetables, fruits,
and medicinal herbs, exhibits diverse pharmacological properties such as
anti-inflammatory, antioxidant, and anti-tumor effects [9, 10, 11]. Recent studies
indicate that quercetin inhibits tumor growth through mechanisms including G2/M
phase cell cycle arrest, induction of mitochondrial apoptosis, and suppression of
epithelial-mesenchymal transition (EMT), invasion, and metastasis [12, 13, 14].
Furthermore, it modulates key oncogenic signaling pathways like PI3K/AKT,
NF-
Network pharmacology analysis suggests that quercetin, as a multi-target natural product, influences numerous key factors in HCC pathogenesis, including EGFR, PIK3CA, matrix metalloproteinase-9 (MMP-9), signal transducer and activator of transcription 3 (STAT3), and B-cell lymphoma 2 (Bcl-2), impacting critical biological processes including cell proliferation, apoptosis, angiogenesis, and the tumor microenvironment [21, 22, 23]. A comprehensive exploration of the interactive effects of quercetin and lenvatinib within regulatory networks could illuminate the mechanistic basis for their potential combined antitumor activity.
This study utilized the human HCC cell line Huh7 and a nude mouse xenograft model to investigate the synergistic anti-tumor effects of quercetin combined with lenvatinib. We aimed to elucidate the potential mechanisms involving cell proliferation, apoptosis, invasion, metastasis, and associated molecular pathways, thereby providing novel experimental evidence and strategic options for clinical HCC combination therapy.
The human HCC cell line Huh7 (#CL-0102) was procured from Procell Life Science & Technology Co., Ltd. (Wuhan, China). The Huh7 cells were validated using STR profiling and tested negative for mycoplasma. Cells were maintained in DMEM high-glucose medium supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin at 37 °C in a humidified 5% CO2 incubator. All experiments were completed within the 10th to 25th passage after cell recovery, during the logarithmic growth phase of cells.
Quercetin (HY-18085,
Huh7 cells were seeded in 96-well plates at 5
Huh7 cells (500/well) were plated in 6-well plates and treated with DMSO, 10
µM quercetin, 10 µM lenvatinib, or their combination
for 24 hours. Afterward, the medium was then replaced with drug-free medium, and
cells were cultured for 14 days. Colonies were fixed, stained with 0.1% crystal
violet, and those
Huh7 cells (5
Transwell inserts pre-coated with Matrigel were seeded with treated cells (5
Confluent Huh7 monolayers in 6-well plates were scratched with a sterile 100 µL pipette tip after 24-hour treatments. After washing, fresh medium was added. Wound closure was photographed at 0 and 24 hours, and the healing rate was quantified using ImageJ software (Version 1.54p, National Institutes of Health, Bethesda, MD, USA).
Total protein was extracted, quantified by BCA assay, separated by SDS-PAGE, and transferred to PVDF membranes. Membranes were blocked, incubated with primary antibodies overnight at 4 °C, followed by incubation with HRP-conjugated secondary antibodies. Protein bands were visualized using an ECL kit and analyzed with ImageJ software.
Potential targets of quercetin and lenvatinib were retrieved from the SwissTargetPrediction database. HCC-related targets were obtained from GeneCards. Common targets among quercetin, lenvatinib, and HCC were identified. A Protein-Protein Interaction (PPI) network was constructed using the STRING database (https://cn.string-db.org/), and KEGG pathway enrichment analysis was performed using the DAVID database (https://davidbioinformatics.nih.gov/).
Male BALB/c nude mice (4–6 weeks old) were subcutaneously inoculated with Huh7
cells (5
Tumor tissues from 19 HCC patients, along with their clinical pathological information, were collected for protein tyrosine kinase 2 (PTK2) staining and analysis. Paraffin-embedded tumor sections underwent deparaffinization, antigen retrieval, and blocking of endogenous peroxidase and non-specific sites. Sections were incubated with PTK2 primary antibody overnight at 4 °C, followed by an HRP-labeled secondary antibody. Staining was developed using DAB, counterstained with hematoxylin, and visualized under a microscope.
Data are presented as mean
CCK-8 assays demonstrated that both quercetin and lenvatinib inhibited Huh7 cell
proliferation in a dose-dependent manner (p
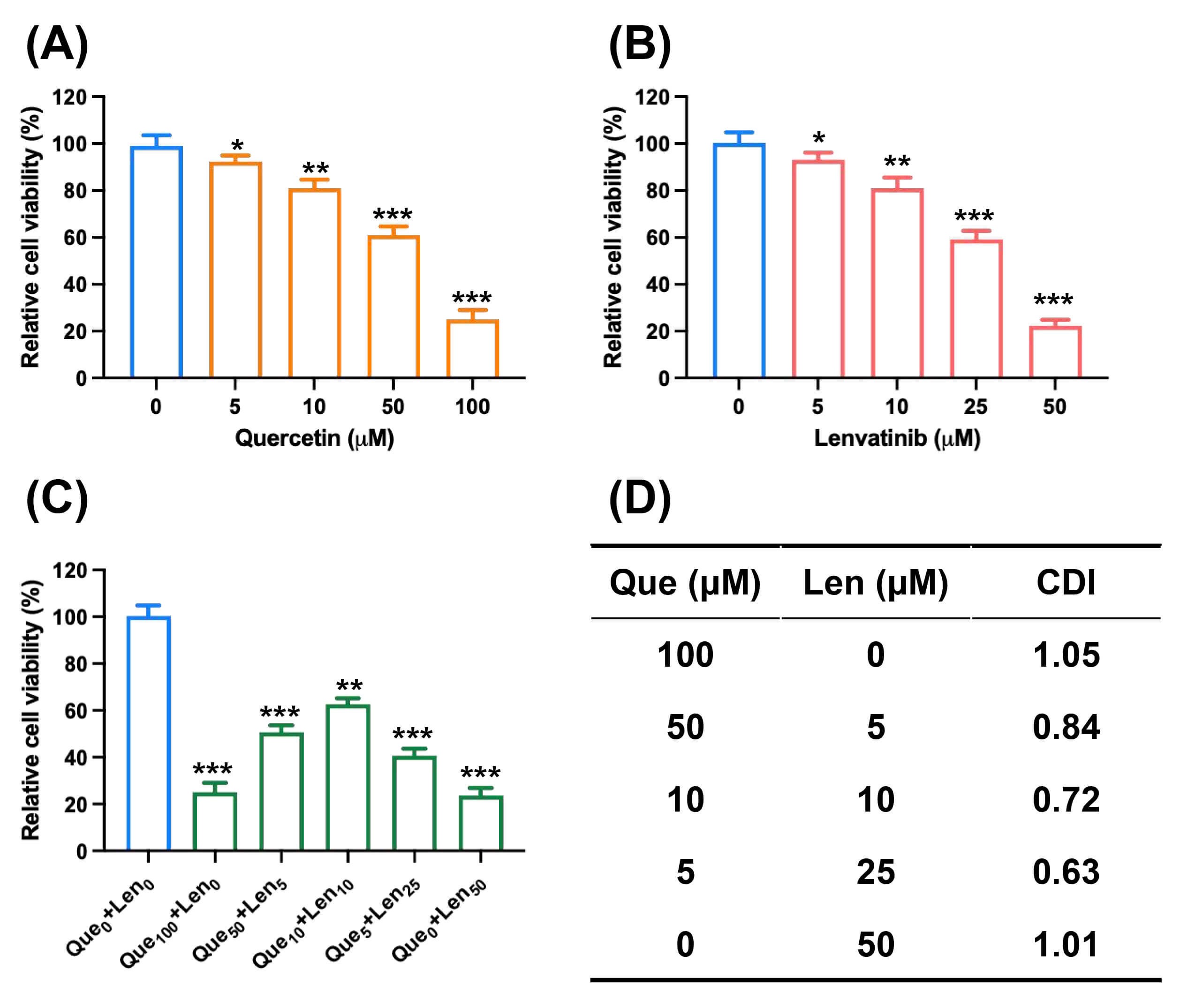 Fig. 1.
Fig. 1.
Quercetin synergizes with lenvatinib to inhibit HCC cell
proliferation. (A) Quercetin at different concentrations inhibited the
proliferation of Huh7 cells (n = 5). (B) Lenvatinib at different concentrations
inhibited the proliferation of Huh7 cells (n = 5). (C) Quercetin and lenvatinib
at different concentrations inhibit the proliferation of Huh7 cells (n = 5). (D)
Combination Index (CDI) according to the results of C indicated that quercetin
and lenvatinib had a combined inhibitory effect on the proliferation of Huh7
cells. Data are mean
Clonogenic assays showed that both quercetin and lenvatinib monotherapies
reduced colony formation (F = 138.9, p
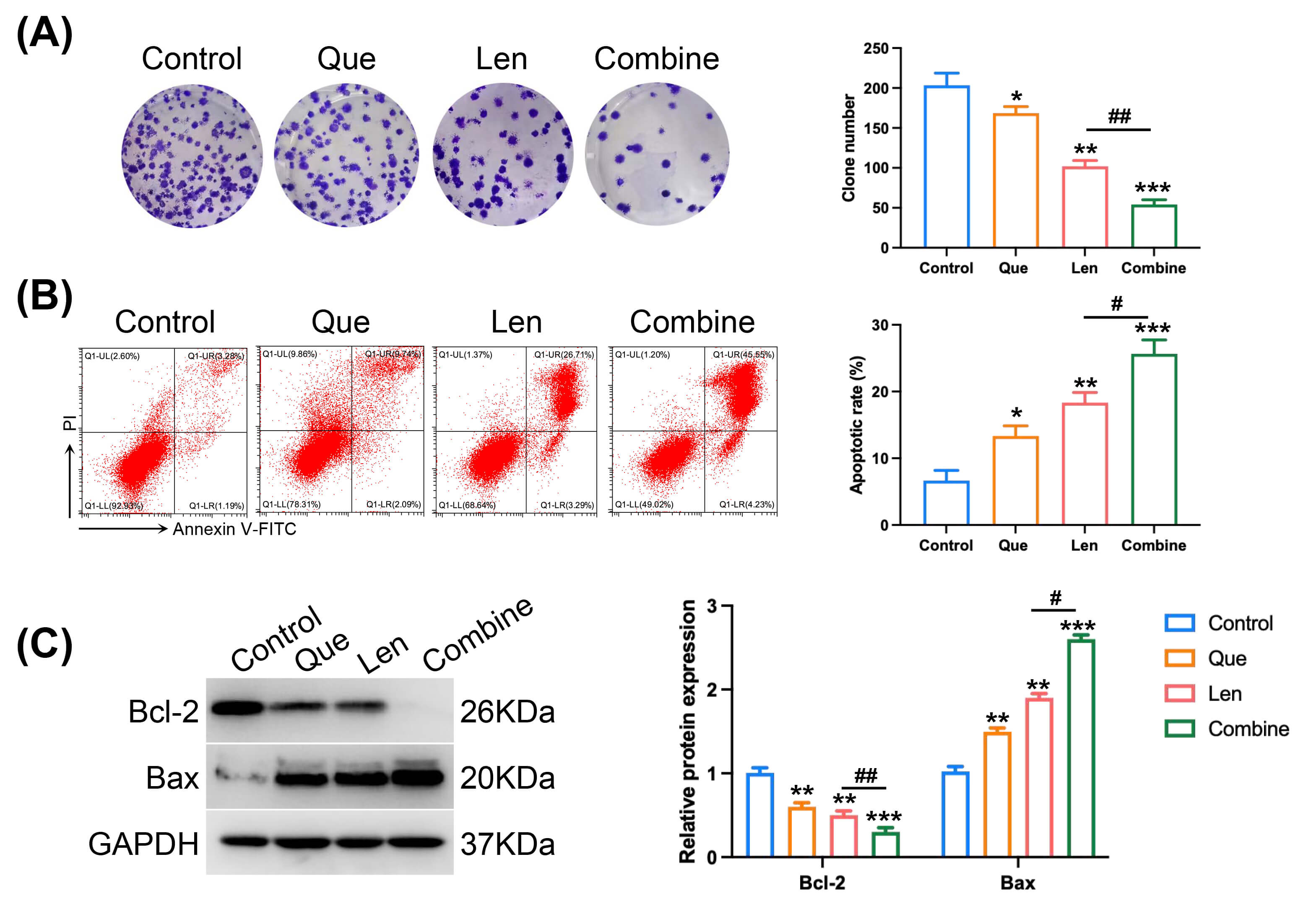 Fig. 2.
Fig. 2.
Effect of quercetin on the pro-apoptotic and anti-clonogenic
activity of lenvatinib in Huh7 cells. (A) Representative images and
quantification of colonies formed by Huh7 cells following treatment (biological
replicates, n = 3). (B) Apoptosis analysis by Annexin V-FITC/PI staining and flow
cytometry after 24-hour treatment (biological replicates, n = 3). (C) Western
blot analysis and densitometric quantification of Bcl-2 and Bax protein levels in
treated Huh7 cells (biological replicates, n = 3), with GAPDH used as a loading
control. The combination of quercetin and lenvatinib significantly enhanced
apoptosis and inhibited colony formation compared to lenvatinib alone. Data are
presented as mean
Transwell invasion assays demonstrated that both drugs inhibited Huh7 cell
invasion when used alone, with the combination yielding a superior effect (F =
107.5, t = 6.86. p
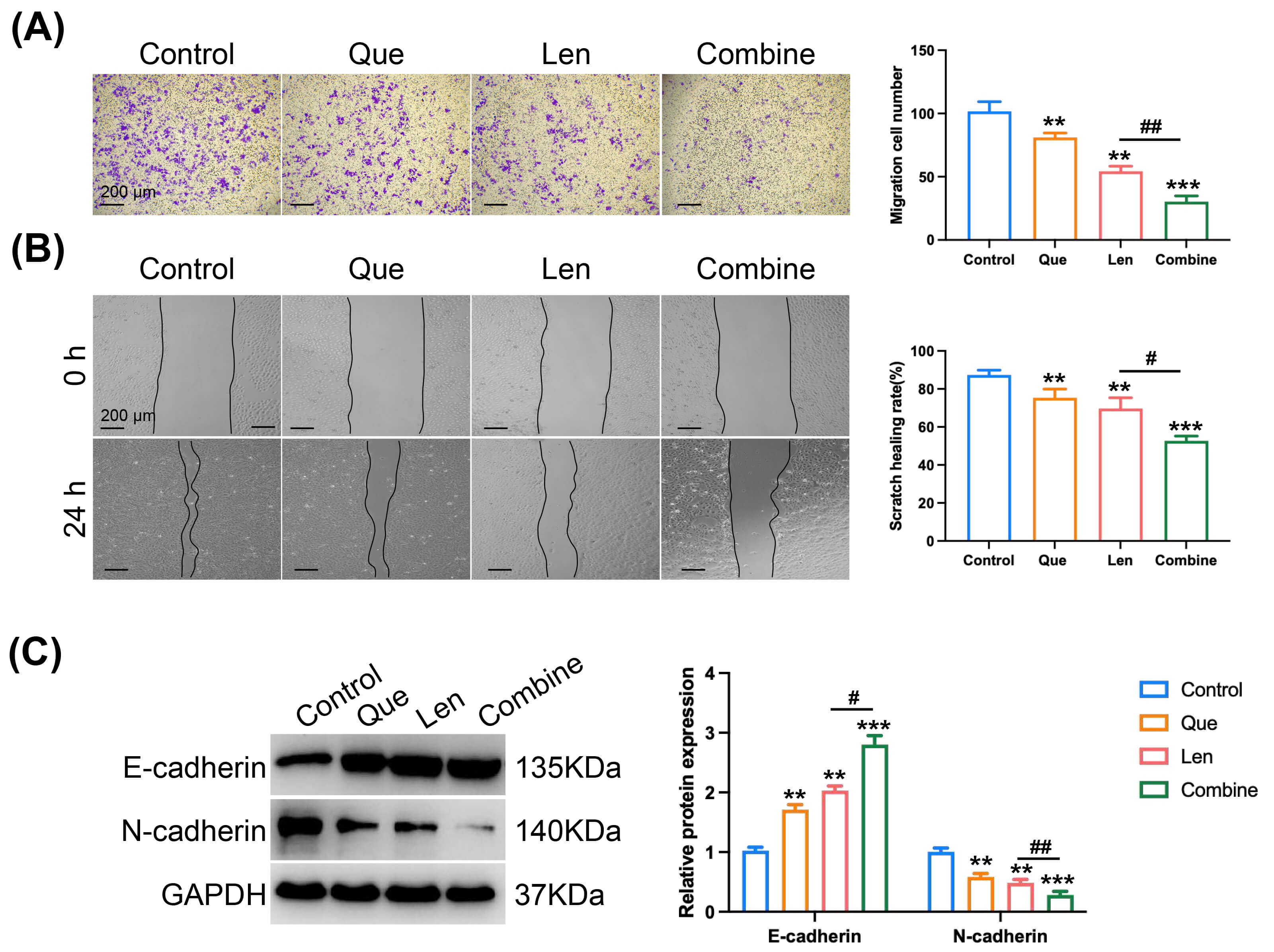 Fig. 3.
Fig. 3.
Effect of quercetin on the anti-invasive and anti-migratory
activity of lenvatinib in Huh7 cells. (A) Representative images and
quantification of invaded Huh7 cells through Matrigel using a Transwell assay
(scale bar: 200 µm, biological replicates, n = 3). (B) Representative images
and quantification of Huh7 cell migration assessed by wound healing assay (scale
bar: 200 µm, biological replicates, n = 3). (C) Western blot analysis and
densitometric quantification of E-cadherin and N-cadherin protein levels in
treated Huh7 cells (biological replicates, n = 3), with GAPDH used as a loading
control. The combination of quercetin and lenvatinib significantly inhibited cell
invasion and migration compared to lenvatinib alone, accompanied by increased
E-cadherin and decreased N-cadherin expression. Cells treated with vehicle
control (DMSO) were used as the reference for response normalization. Data are
presented as mean
The molecular structures of quercetin and lenvatinib are shown in Fig. 4A,B. Network pharmacology initially identified 21 common targets from quercetin, lenvatinib, and HCC-related genes (Supplementary Tables 2–4, Supplementary Fig. 1). Refining the analysis using the top 300 HCC-related targets pinpointed PTK2, KDR, ABCC1, MET, ALK, and PIK3CG as key overlapping targets (Fig. 4C,D). KEGG pathway analysis of the intersecting genes is presented in Fig. 4E.
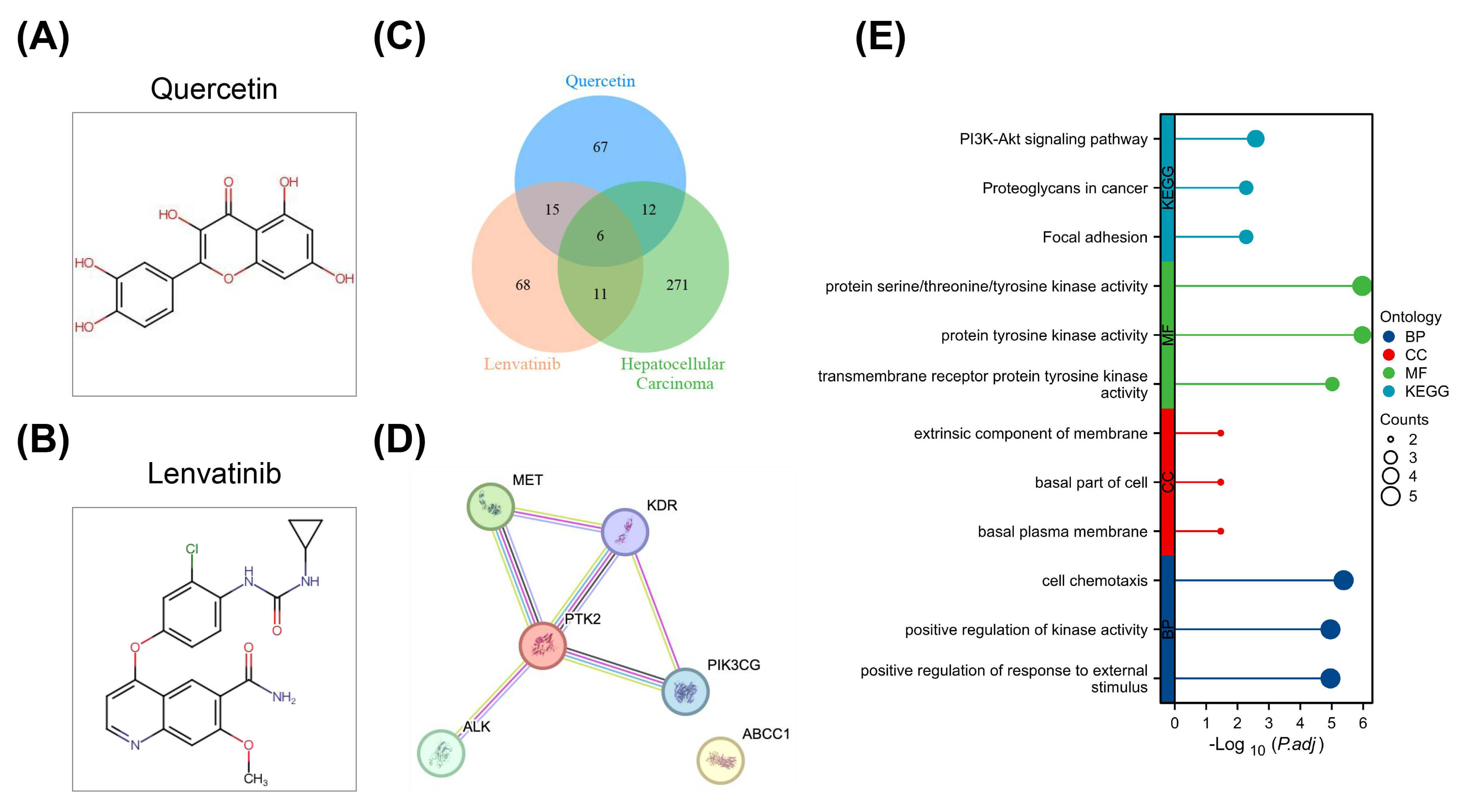 Fig. 4.
Fig. 4.
Network pharmacology analysis of potential targets for quercetin combined with lenvatinib in the treatment of HCC. (A) Molecular structure of quercetin. (B) Molecular structure of lenvatinib. (C) Venn diagram showing the overlap among targets of quercetin, targets of lenvatinib, and HCC-related targets. (D) Protein-protein interaction (PPI) network of overlapping genes. (E) KEGG pathway enrichment analysis for the overlapping genes, indicating that the synergistic effects of the drug combination are likely mediated through regulation of multiple signaling pathways relevant to HCC progression. Analysis was based on established public databases and bioinformatics tools. Data visualization and statistical enrichment were performed to identify key network components and pathways.
An examination of TCGA data revealed significantly elevated mRNA expression of
PTK2, MET, and ABCC1 in HCC tissues compared to normal
liver tissues (p
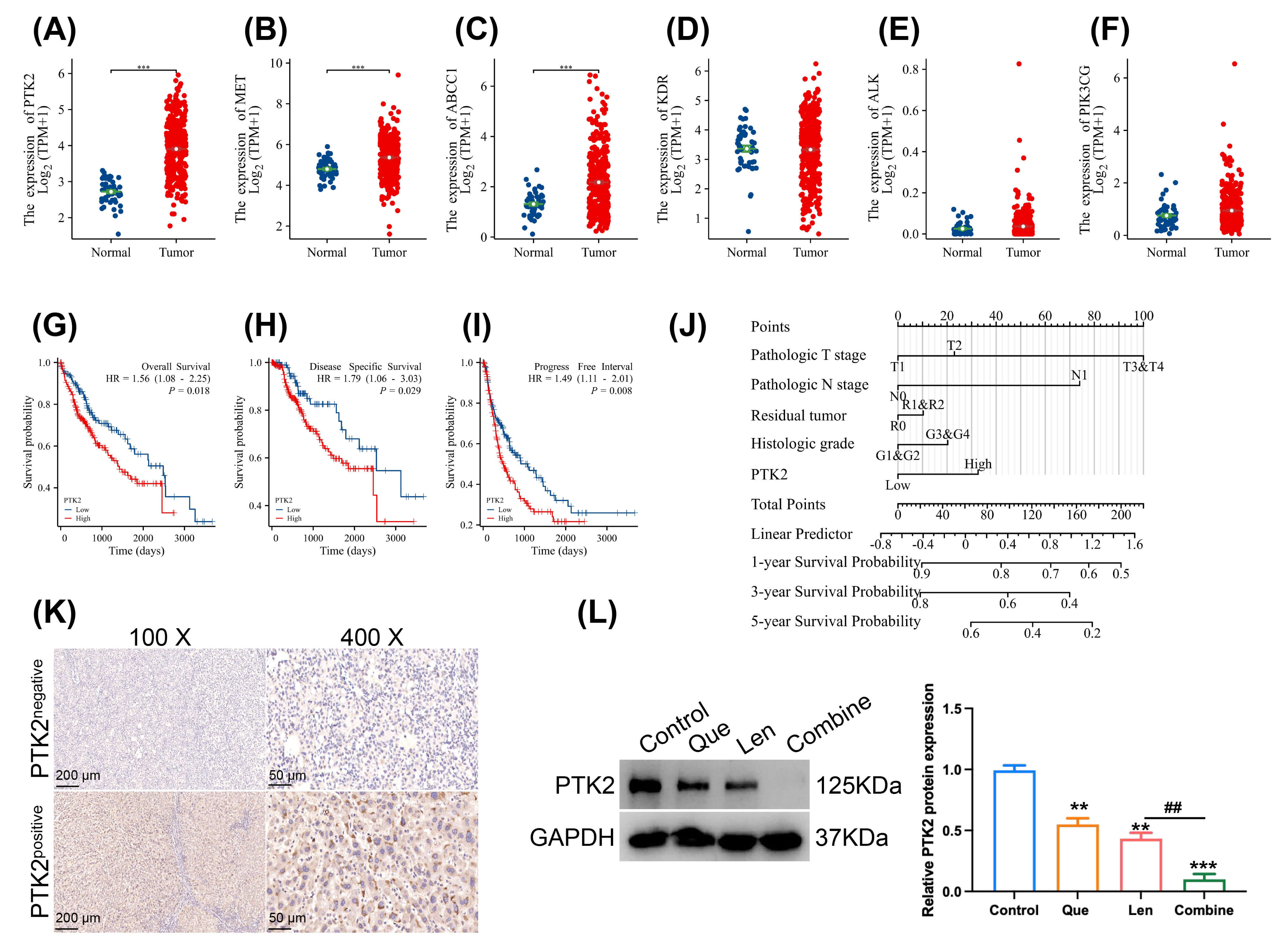 Fig. 5.
Fig. 5.
Identification of PTK2 as a key prognostic and therapeutic
target in HCC. (A–F) mRNA expression levels of PTK2, MET,
ABCC1, KDR, ALK, and PIK3CG in HCC tissues
compared to normal tissues, based on data from The Cancer Genome Atlas (TCGA)
database. (G–I) Kaplan–Meier survival analysis of HCC patients stratified by
PTK2 expression levels: (G) Overall Survival, (H) Disease-Free Survival, (I)
Progression-Free Survival. (J) Forest plot summarizing the prognostic value of
PTK2 mRNA expression in HCC across multiple survival endpoints. (K)
Representative immunohistochemical images showing PTK2-negative and PTK2-positive
staining in human HCC tissue sections (scale bar: 200 µm for 100 ×, scale bar: 50 µm for 400 ×). (L) Western blot
analysis and densitometric quantification of PTK2 protein expression in treated
Huh7 cells (biological replicates, n = 3), with GAPDH used as a loading control.
The combination of quercetin and lenvatinib significantly reduced PTK2 expression
compared to lenvatinib alone. Data are presented as mean
In the Huh7 xenograft model, both monotherapies (Fig. 6A) reduced tumor volume
(F = 94.35, t = 6.45, p
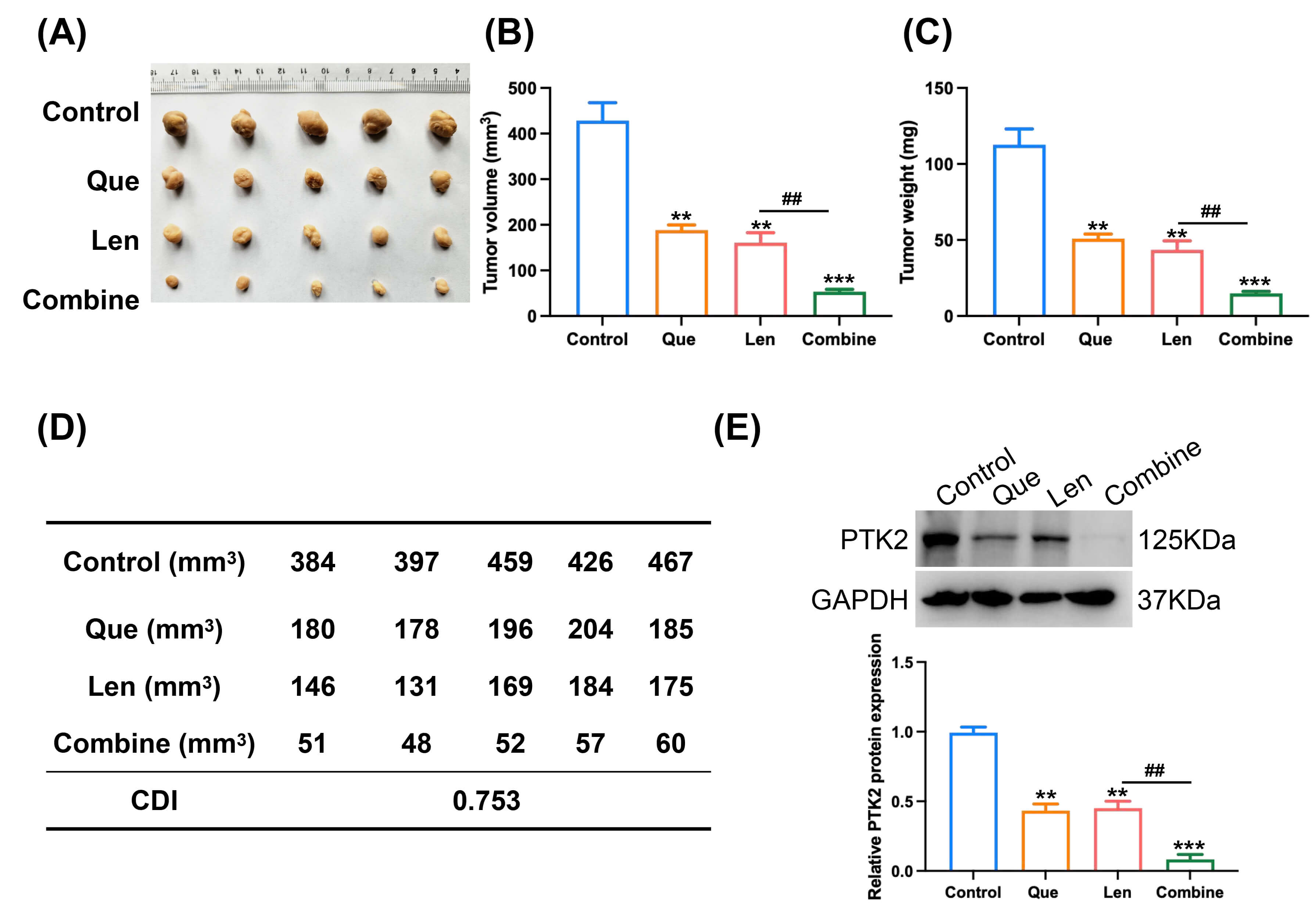 Fig. 6.
Fig. 6.
Quercetin synergizes with lenvatinib to inhibit hepatocellular
carcinoma growth in vivo. (A) Representative photographs of excised
xenograft tumors from each treatment group (n = 5 mice per group). (B) Tumor
growth curves showing volume changes over the treatment period. (C) Final tumor
weights measured at the study endpoint. (D) The in vivo Combination
Index (CI) calculated based on final tumor volumes. The CI value indicated a
synergistic therapeutic effect between quercetin and lenvatinib. (E) Western blot
analysis and densitometric quantification of PTK2 protein expression in xenograft
tumor tissues (n = 3), with GAPDH used as a loading control. The combination
treatment significantly reduced PTK2 expression compared to lenvatinib
monotherapy. Data are presented as mean
This research systematically elucidates the potential of quercetin, a dietary flavonoid, to significantly enhance the anti-tumor activity of lenvatinib in HCC. Our experimental models demonstrate that the combination therapy exhibits a synergistic suppression of key oncogenic traits — proliferation, clonogenicity, invasion, and migration — while simultaneously activating apoptotic pathways. Consistent calculation of Combination Index (CDI) values below one in both cellular and animal studies provided quantitative validation of this pharmacological synergy. Moving beyond phenotypic observation, our integrated strategy, which combines computational network pharmacology with molecular validation, pinpointed PTK2 (Focal Adhesion Kinase) as a critical shared target. This mechanistic insight not only offers a potential clinical strategy to address lenvatinib resistance but also delineates a specific molecular pathway underlying the combined effect.
As a multi-targeted tyrosine kinase inhibitor, Lenvatinib exerts anti-tumor effects by disrupting angiogenic signaling through VEGFR, FGFR, and PDGFR, as well as directly suppressing tumor cell proliferation via RET and KIT inhibition [4, 5, 6, 26]. However, its efficacy in advanced HCC is frequently limited by the development of resistance, mainly due to the pronounced heterogeneity of HCC and its complex stromal microenvironment [27]. While our data confirm the standalone anti-HCC properties of quercetin, its main value appears to lie in its role as a potent sensitizer. The compound significantly lowers the threshold for lenvatinib’s efficacy, transforming a modest cytostatic response into a powerful cytotoxic and anti-metastatic outcome.
The synergy we observed extends beyond a mere additive impact on cell numbers. It represents a coordinated attack on multiple core cancer capabilities. The combination regimen not only more effectively suppressed immediate proliferation but also critically undermined the tumor’s long-term replicative potential, as demonstrated by the drastic reduction in colony-forming ability. Equally important, invasive and migratory behaviors — key drivers of metastasis — were significantly impaired, alongside a marked shift in the cellular population towards apoptosis. The consistent CDI values obtained from independent in vitro and in vivo systems provide robust, multi-level confirmation of a precise synergistic interaction. This pharmacodynamic profile suggests a compelling clinical possibility that quercetin co-therapy could enhance lenvatinib’s therapeutic window, potentially maintaining tumor control at lower, less toxic drug doses [28]. Managing lenvatinib therapy is often complicated by common adverse effects, such as hypertension, proteinuria, and hand-foot syndrome, which can necessitate dose modifications and disrupt treatment continuity, thereby jeopardizing long-term outcomes [29, 30]. In this context, quercetin stands out as a uniquely suitable combination partner. Its status as a ubiquitous dietary component, along with a long history of safe use in humans, positions it as an ideal candidate for mitigating regimen-related toxicity. By synergistically enhancing anti-tumor potency, this combination could theoretically enable effective disease management with reduced lenvatinib exposure, ultimately improving patient tolerability and quality of life, a critical goal in palliative oncology.
The major finding of our work is the identification of PTK2 as a central mediator of this synergy. PTK2 acts as a non-receptor tyrosine kinase and serves as an important signaling hub that integrates inputs affecting cell survival, proliferation, and motility [31]. Its pathogenic role in HCC is well-documented, with numerous reports linking its overexpression to advanced disease stages, vascular invasion, and poor patient survival [32, 33]. Mechanistically, PTK2 sustains tumor cell viability by activating pro-survival cascades like PI3K/Akt/mTOR and Ras/MAPK [34, 35]. Concurrently, it functions as a master regulator of the invasive program, driving metastasis and shaping a permissive tumor microenvironment through effects on angiogenesis and immune evasion [36, 37]. Our results indicate that while both monotherapies can modestly attenuate PTK2 levels, their combination induces a far more substantial downregulation. This cooperative suppression likely disrupts a network of downstream oncogenic signals, ultimately manifesting as the observed potentiation of apoptosis (via Bcl-2/Bax) and reversal of EMT (via E-cadherin/N-cadherin). The congruent modulation of these pivotal pathways strongly supports PTK2’s role as an orchestrator of the synergistic response.
However, this study has some limitations, including the reliance on a single HCC cell line (Huh7) and a subcutaneous xenograft model. In the future study, more HCC cell lines will be applied to study the sensitization effect of quercetin on lenvatinib. Although PTK2 is identified as a key shared target, the precise upstream and downstream molecular pathways through which the combination therapy modulates PTK2 and exerts its synergistic effects require further elucidation. Future work must clarify the precise molecular events by which each agent, and their combination, regulates PTK2. Is control exerted at the transcriptional level, through mRNA stability, or via post-translational modification and protein degradation? From a translational perspective, it is essential to determine if tumors exhibiting high PTK2 expression or activation constitute a biomarker-defined population that derives exceptional benefit from this regimen, paving the way for personalized therapy. Furthermore, evaluating the efficacy of this combination in models with acquired lenvatinib resistance would offer invaluable insights for managing refractory disease.
In conclusion, this study establishes quercetin as a potent enhancer of lenvatinib’s anti-HCC efficacy. The observed synergy, which influences a range of cancer hallmarks from proliferation and survival to invasion and metastasis, is mechanistically rooted in the cooperative targeting of PTK2. These findings provide a strong scientific foundation for clinical exploration of the quercetin-lenvatinib combination, proposing a dual-pronged strategy to enhance therapeutic efficacy and potentially overcome resistance in HCC.
The data sets used or analyzed during the current study are available from the corresponding author upon reasonable request.
YZ designed the research study. JL and QHY performed the research. KL and XC collected and analyzed the data. JL and YZ have been involved in drafting the manuscript and all authors have been involved in revising it critically for important intellectual content. All authors give final approval of the version to be published. All authors have participated sufficiently in the work to take public responsibility for appropriate portions of the content and agreed to be accountable for all aspects of the work in ensuring that questions related to its accuracy or integrity.
The patient samples and information collected in the study were approved by the ethics committee of Hunan Provincial People’s Hospital/The First Affiliated Hospital of Hunan Normal University ([2024] -032). The study was carried out in accordance with the guidelines of the Declaration of Helsinki. All pathological samples obtained were informed to patients or their families/legal guardians and signed an informed consent form. The animal experiments in the study were approved by the ethics committee Hunan Provincial People’s Hospital/The First Affiliated Hospital of Hunan Normal University ([2023] -156). The animal experiments in the study was in compliance with the revised Animals (Scientific Procedures) Act 1986 in the UK and Directive 2010/63/EU in Europe).
Not applicable.
This work was financially supported by the following grant: Research Project of Hunan Provincial Health Commission (20254505).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL47715.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






