1 Department of Ophthalmology, Aier Eye Hospital, Jinan University, 510071 Guangzhou, Guangdong, China
2 Department of Fundus Diseases, Nanning Aier Eye Hospital, 530000 Nanning, Guangxi, China
3 School of Life Science, Northeast Agricultural University, 150030 Harbin, Heilongjiang, China
†These authors contributed equally.
Abstract
While the systemic metabolic role of fibroblast growth factor 21 (FGF21) is well-established, its function in retinal homeostasis and its link to retinal diseases like age-related macular degeneration (AMD) and diabetic retinopathy (DR) remains poorly understood. This study investigated the impact of endogenous FGF21 deficiency on the retinal immune microenvironment.
Retinal structure was assessed in FGF21 KO and wild-type mice using spectral-domain optical coherence tomography. Transcriptomic profiles of the retina/choroid were analyzed by RNA-seq. Differentially expressed genes (DEGs) were identified (DESeq2, FDR <0.05), clustered, and interrogated by Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment. Immune-cell composition was inferred with ImmuCellAI.
FGF21 KO mice showed no overt retinal structural defects under baseline conditions. Nevertheless, 449 DEGs were identified (293 up, 156 down in knockout). Pathway analysis revealed significant enrichment of cytokine–cytokine receptor interaction, chemokine signaling, and Jak-STAT cascades. Immune deconvolution indicated a significant increase in M2-polarised macrophages (p < 0.01) without change in total macrophage number. Expression of key inflammatory mediators including Il1b was concordantly altered.
This work establishes endogenous FGF21 as a crucial local immunomodulator and defines a novel mechanistic link to retinal disease susceptibility, supporting its further exploration as a therapeutic target.
Graphical Abstract
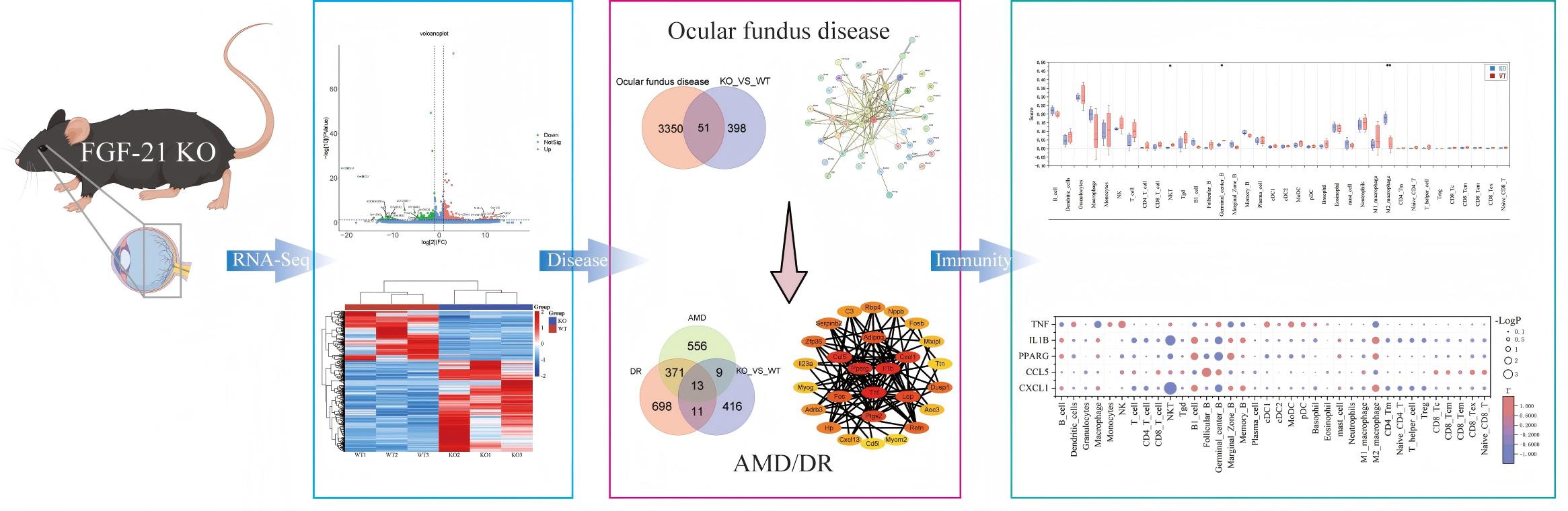
Keywords
- FGF21
- retina
- RNA-Seq
- macrophages
- immunomodulation
The retina is essential for vision, and its dysfunction is a major cause of irreversible blindness worldwide. With aging populations, the prevalence of sight-threatening retinal diseases such as diabetic retinopathy (DR) and age-related macular degeneration (AMD) has risen markedly, representing a critical public health challenge [1]. Understanding the pathogenesis of these conditions and developing effective interventions are therefore central goals in ophthalmology research.
Fibroblast Growth Factor 21 (FGF21) is a hepatokine with systemic metabolic regulatory functions [2]. Previous studies have demonstrated its pivotal role in modulating glycolipid metabolism [3], exerting antioxidant [4] and anti-inflammatory [5], and attenuating the aging process [6]. Intriguingly, recent studies suggest that exogenous FGF21 administration may protect against various retinal pathologies, including DR and dry AMD [7, 8, 9, 10, 11], implying a potential role in maintaining retinal health. However, the mechanisms underlying these protective effects, and particularly the physiological function of endogenous FGF21 in the retina, remain poorly understood.
To elucidate the functional mechanism underlying FGF21’s protective role in retinal homeostasis, the FGF21 knockout mice were used to investigate whether endogenous FGF21 influences retinal homeostasis. We first assessed retinal structure in vivo and then performed transcriptomic analysis of retinal/choroidal tissues to identify molecular changes caused by FGF21 deficiency. Our results reveal that loss of endogenous FGF21 disrupts retinal immune homeostasis, notably by shifting macrophage polarization toward an M2-dominant phenotype, thereby rendering the retina more susceptible to injury. These findings establish endogenous FGF21 as a key local immunomodulator and provide a mechanistic foundation for exploring its therapeutic potential in retinal diseases.
All animal experiments conducted in this study were approved in advance by
Northeast Agricultural University. FGF21 knockout mice (FGF21 KO mice, KO group)
were purchased from Cyagen Biosciences Inc. (Suzhou, Contract No.
KOAIP180401WZ1+KOAIP180401WZ2). C57BL/6 mice of equivalent age (WT group) were
obtained from Liaoning Chang Sheng Biotechnology Co., Ltd. (Liaoning, China). All
animals were housed in a controlled environment with a 12-hour light/12-hour dark
cycle, provided with ad libitum access to food and water, and shielded from
external stimuli. The ambient temperature for housing was maintained at 22
To determine whether endogenous FGF21 deficiency affects retinal resilience
under pathological stress, mice were allocated into two independent
disease-modeling experiments (n = 6 per genotype per model). For diabetic
retinopathy (DR) Model, because of the hypersensitivity of FGF21 KO mice to
streptozotocin (STZ) (HY-13753, MedChemExpress, Shanghai, China), diabetes was
induced using genotype-specific protocols. WT mice received intraperitoneal
injections of STZ (30 mg/kg/day for 5 consecutive days) while maintained on a
high-fat diet (HFD, 1135DM). FGF21 KO mice were fed the same HFD without STZ
injection until their fasting blood glucose levels exceeded 11.1 mmol/L. Diabetic
mice (fasting glucose
To investigate the direct effect of endogenous FGF21 deletion on retinal homeostasis in the absence of external stressors, a separate cohort of mice (n = 6 per genotype, WT and FGF21 KO) was examined at 6 weeks of age under normal physiological conditions. After baseline OCT imaging, mice were euthanized, and neural retina/choroid complexes were promptly harvested. Tissues from each group were pooled for bulk RNA sequencing (n = 3 mice), and other mice per group were used for subsequent qPCR validation (n = 3 retina/choroid complexes) and immunohistochemistry (n = 3 retina/choroid complexes).
Eyeballs were fixed with 4% paraformaldehyde, dehydrated, cleared, and embedded in paraffin. Sections (4–5 µm) were dewaxed and subjected to heat-induced epitope retrieval (HIER). After blocking endogenous peroxidase activity with 3% H2O2 and nonspecific sites with 10% horse serum, sections were incubated with primary antibodies against CD163 (bs2527R, 1:100, Bioss, Beijing, China). Following PBS washes, sections were incubated with a Goat Anti-Rabbit IgG H&L antibody (bs-0295G-HRP, 1:200, Bioss, Beijing, China), developed with 3,3-diaminobenzidine (DAB) (HY-W014212, MedChemExpress, Shanghai, China), and counterstained with hematoxylin (C0107, Beyotime, Shanghai, China).
For semi-quantification of CD163 staining, whole-section images were analyzed using ImageJ software (NIH, Bethesda, USA) with the “IHC Toolbox” plugin. The integrated optical density (IOD) of the DAB (brown) signal, which reflects the amount of CD163 protein within the tissue, was measured in a defined region of the entire retinal section.
Following a 12-h fast, mice were weighed and baseline blood glucose was measured prior to oral gavage of glucose (2 g/kg). Blood glucose levels were then monitored at 30-minute intervals for 120 minutes. Glucose tolerance was determined by calculating the area under the curve (AUC) for blood glucose over time using Origin 2021 (OriginLab Corporation, Northampton, MA, USA) software.
Mice were anesthetized via intraperitoneal Avertin (1.25% w/v solution, 30 µL/g, MA0478, Dalian Meilun Biotechnology Co., Ltd, Dalian, Liaoning, China). Pupillary dilation was achieved using tropicamide-phenylephrine eye drops (1–2 drops per eye, Santen Pharmaceutical Co., Ltd, Suzhou, Jiangsu, China). Throughout the procedure, ophthalmic hydroxypropyl methylcellulose (UV-Gel) (Shanghai Haohai Biological Technology Co., Ltd., Shanghai, China) was intermittently applied to the eyes of the mice to safeguard the cornea. OCT images were captured using the VG200D imaging camera system (SVision imaging, Ltd, San Jose, CA, USA).
RNA-Seq testing was commissioned to the Biotechnology Company (Shanghai, China).
Total RNA was extracted from tissues using the TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA). Then, the first-strand cDNA was synthesized using a HiScript II 1st Strand cDNA Synthesis Kit (R212-01, Vazyme, Nanjing, Jiangsu, China). The gene expression was detected with a Taq Pro Universal SYBR qPCR Master Mix Kit (Q712-02, Vazyme, Nanjing, Jiangsu, China), according to the manufacturer’s protocol. The expression for each gene was calculated using the expression 2-△△Ct method. The Real-time PCR primers used in this study were synthesized by Sangon Biotech (Shanghai) Co., Ltd. The primer sequences are shown in Table 1.
| Gene | Forward primer | Reverse primer |
| Tnf | GCCACCACGCTCTTCTGTCTACT | TGGTTTGTGAGTGTGAGGGTCTGG |
| Il1b | ATCTCGCAGCAGCACATCAACAAG | GGTCCACGGGAAAGACACAGGTAG |
| Pparg | TCGCCAAGGTGCTCCAGAAGATG | GGTGAAGGCTCATGTCTGTCTCTGT |
| Ccl5 | ACTCCCTGCTGCTTTGCCTACC | TTGGCGGTTCCTTCGAGTGACAA |
| Cxcl1 | ATGGCTGGGATTCACCTCAAGAACA | GAGTGTGGCTATGACTTCGGTTTGG |
| Ptgs2 | AACACCTGAGCGGTTACCACTTCAA | AGGCAATGCGGTTCTGATACTGGAA |
| Lep | GGTTCCTGTGGCTTTGGTCCTATCT | GGATACCGACTGCGTGTGTGAAATG |
| Adipoq | GCCTGGAGAAGCCGCTTATGTGTA | ACTTGCCAGTGCTGCCGTCAT |
| Fos | GCCAGTCAAGAGCATCAGCAACG | AGGAACCGGACAGGTCCACATCT |
| Retn | TCTTCCTTGTCCCTGAACTGCTGG | GCTCAAGACTGCTGTGCCTTCTG |
| CD163 | GGTTCTTCTTGGAGGTGCTGGATCT | CCGCCAGTCTCAGTTCCTTCTTCA |
| CD206/MRC | ACCTGGCAAGTATCCACAGCATTGA | GCAGTCCTCCTGTCTGTTGTTCTCA |
DESeq was employed for the analysis, with the screening criteria set as
p value
The relevant genes obtained from the screening were imported into the STRING website (http://string-db.org) to construct the protein-protein interaction (PPI) network [12]. The results of the PPI network were then visualized and enhanced using Cytoscape (version 3.10.0, https://cytoscape.org, National Resource for Network Biology, USA). To comprehensively analyze the PPI network and identify key components, the cytoHubba [13] plugin was utilized. Specifically, the Maximal Clique Centrality (MCC) algorithm was employed to screen for key genes, facilitating the construction of sub-networks and identification of pivotal components within the PPI network.
The analysis was carried out by using R software (v.4.2.2) package clusterProfiler (v.4.5.0) [14] through Hiplot Pro (https://hiplot.com.cn/), a comprehensive web service for biomedical data analysis and visualization.
The correlation coefficients between the variables were computed through Pearson correlation analysis. A grid plot was generated to display the genes in the group with an absolute correlation coefficient greater than 0.8 and a p-value less than 0.05. This facilitated the visualization of interactions between significantly correlated feature nodes, aiding in the identification of relationships between the variables. The process of calculation and plotting was conducted using CNSknowall (https://cnsknowall.com/).
Immune Cell Abundance Identifier (ImmuCellAI) is a tool to accurately estimate immune cell abundance from gene expression datasets, including RNA-Seq [15]. In the present study, the Immune Cell Abundance Identifier for mouse (ImmuCellAI-mouse) [16] is used to estimate the abundance of 36 immune cell (sub) types in mouse retina/choroid RNA-Seq data.
Data are presented as the mean
To assess the intrinsic role of endogenous FGF21, we first examined retinal
structure and systemic metabolism in unchallenged, 6-week-old mice. In
vivo spectral-domain optical coherence tomography (OCT) revealed comparable
retinal thickness, reflectivity, and overall architecture between wild-type (WT)
and FGF21 knockout (KO) mice (Fig. 1A,B). Furthermore, FGF21 KO mice showed no
significant differences in body weight, fasting blood glucose, or glucose
tolerance compared to WT littermates (all p
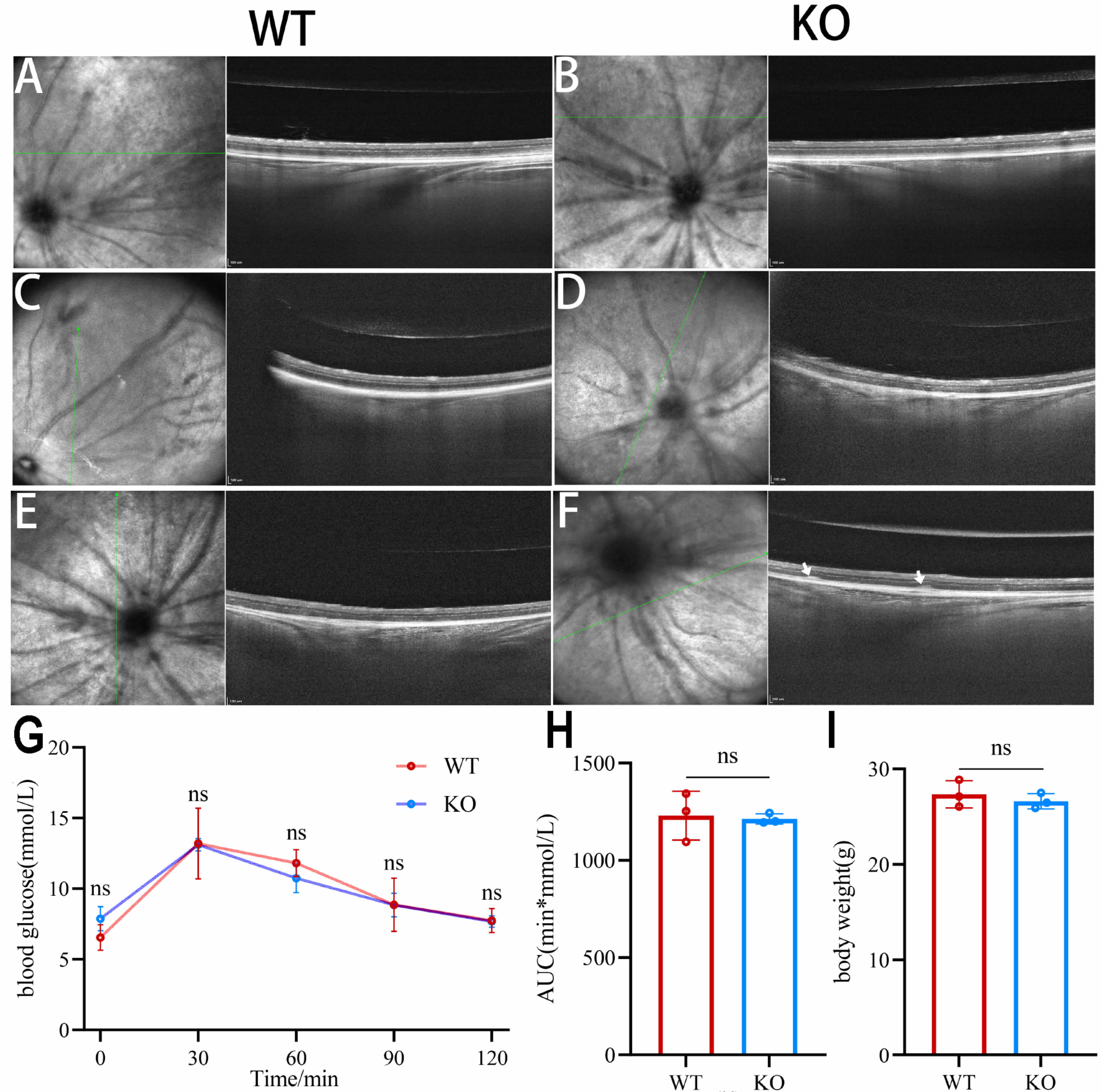 Fig. 1.
Fig. 1.
Fibroblast Growth Factor 21 (FGF21) knockout exacerbates retinal
structural damage induced by hyperglycemia and oxidative stress in mouse models.
(A–F) OCT detection results of the fundus of mice. (A) 6-week-old WT mice. (B)
6-week-old FGF21 KO mice. (C) WT diabetic mice. (D) FGF21 KO diabetic mice. (E)
WT AMD mice. (F) FGF21 KO AMD mice. Green lines indicate the position of the OCT
scan line. White arrows indicate drusen, the typical lesions of dry age-related
macular degeneration. (G) Oral glucose tolerance test (OGTT) performed in
6-week-old FGF21 KO and WT mice. (H) Area under the curve (AUC) of blood glucose
during the OGTT. (I) Body weight measurements of 6-week-old FGF21 KO and WT mice. KO: FGF21 knockout; WT: normal control; ns: p
We next investigated whether the absence of FGF21 affects retinal resilience to pathological insults. Quantitative analysis of retinal layer thickness by OCT revealed that FGF21 KO mice exhibited significantly exacerbated structural damage under both pathological conditions.
When subjected to diabetic conditions or hydroquinone-induced oxidative stress (a model for dry age-related macular degeneration), FGF21 KO mice exhibited significantly more severe retinal structural damage than their WT counterparts, as quantified by OCT (Fig. 1C–F). Retinal thickness measurements were performed on the averaged image from three consecutive scans. Using the segmented line tool, three perpendicular measurements were taken from the internal limiting membrane (ILM) to the retinal pigment epithelium (RPE) to determine the total retinal thickness. The values were then averaged to yield a single thickness value per eye.
Under diabetic conditions, the total retinal thickness in FGF21 KO mice was
thinner than in WT controls (p
Transcriptomic analysis of retinal/choroidal complexes from 6-week-old FGF21 KO
and WT mice revealed 449 differentially expressed genes (DEGs) (p
 Fig. 2.
Fig. 2.
Transcriptomic profiling reveals differential gene expression patterns in the retina/choroid of FGF21 KO mice compared to WT controls. (A) Volcano plot illustrating differentially expressed genes (DEGs) between KO and WT groups. Significantly up-regulated genes are highlighted in red, while significantly down-regulated genes are shown in green. Blue dots represent non-significantly expressed genes. (B) Principal component analysis (PCA) plot demonstrating the clustering pattern of samples based on the global transcriptome profiles, with PC1 and PC2 indicating the first and second principal components, respectively. (C) Hierarchical clustering heatmap of differentially expressed mRNA. Rows represent individual genes, and columns represent individual samples. Color scale indicates normalized expression levels, with red denoting high expression and blue denoting low expression. (D) Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis of DEGs. KO: FGF21 knockout; WT: normal control.
Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis of these DEGs highlighted significant involvement in immune and inflammatory pathways. The most enriched terms included Cytokine-cytokine receptor interaction signaling pathway, Motor proteins, Alcoholic liver disease, C-type lectin receptor signaling pathway, and TNF signaling pathway (Fig. 2D).
To explore the disease relevance of the transcriptomic changes, we intersected
the 449 DEGs with known fundus disease-associated genes from the GeneCards
database (relevance score
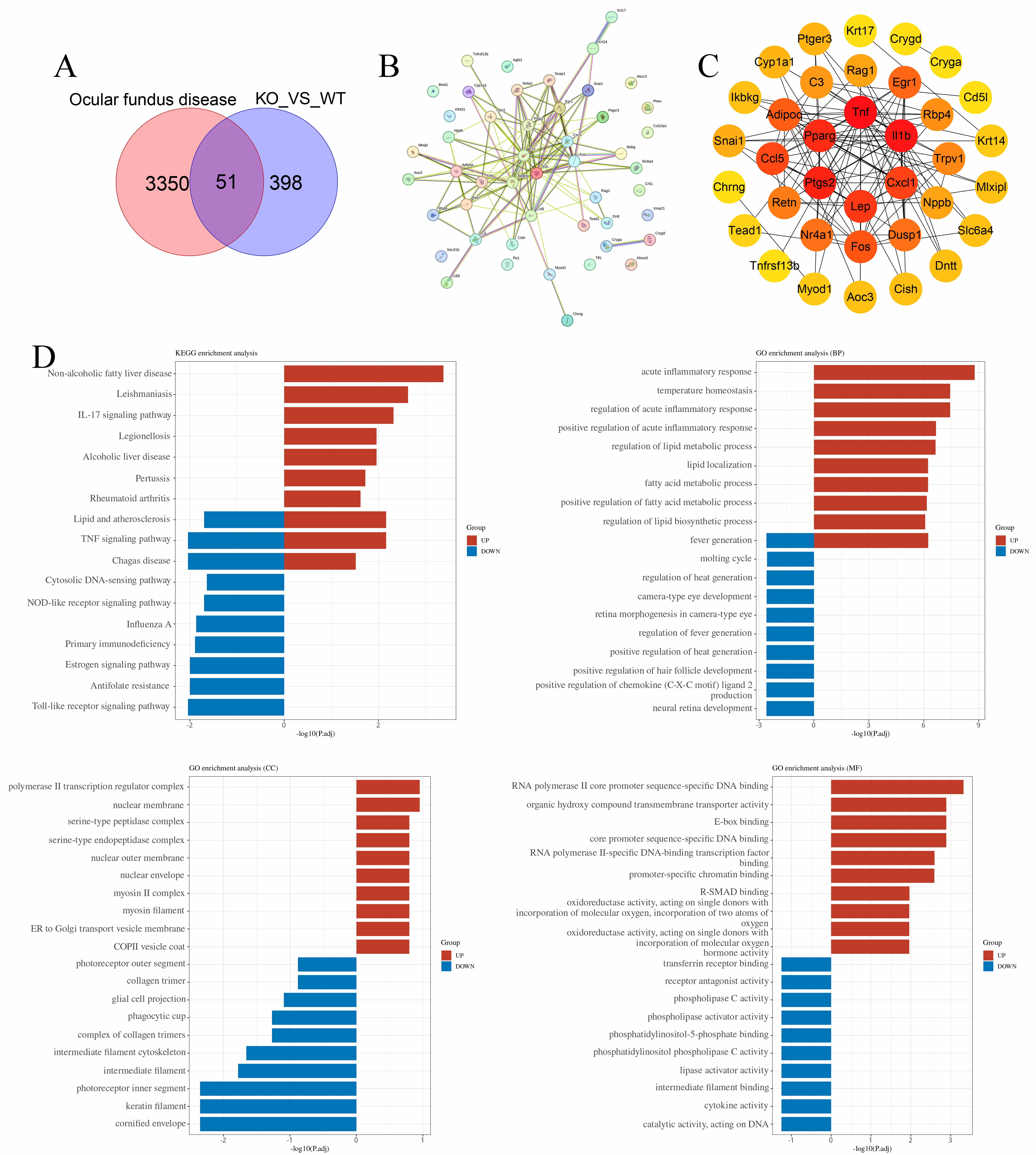 Fig. 3.
Fig. 3.
Integration analysis identifies FGF21 deficiency-associated retinal/choroidal differentially expressed genes (DEGs) overlapping with ocular fundus disease signature genes and their functional interaction networks. (A) Venn diagram depicting the intersection between retinal/choroidal DEGs from FGF21 KO mice and ocular fundus disease-associated genes curated from the GeneCards database. (B) PPI network diagram. Nodes represent individual proteins, and edges indicate predicted functional interactions. (C) Identification of hub genes from the PPI network using the Maximal Clique Centrality (MCC) algorithm implemented in CytoHubba plugin of Cytoscape. The top-ranked core genes are displayed in descending order of MCC scores, with color intensity proportional to the centrality ranking. (D) KEGG versus GO enrichment analysis. KO: FGF21 knockout; WT: normal control.
Functional enrichment analysis of these hub genes highlighted their involvement in key pathways and biological processes. KEGG pathway analysis showed enrichment in the TNF signaling pathway, Non-alcoholic fatty liver disease, and Leishmaniasis. Gene Ontology (GO) analysis indicated associations with the Acute inflammatory response, Temperature homeostasis, Photoreceptor inner segment, Keratin filament, Cornified envelope, RNA polymerase II core promoter sequence-specific DNA binding (Fig. 3D).
To delineate the specific relevance of FGF21 deficiency to age-related macular
degeneration (AMD) and diabetic retinopathy (DR), we intersected our DEG list
with disease-associated genes from GeneCards, DisGeNET, and Therapeutic Target
Database. After applying a relevance score filter (
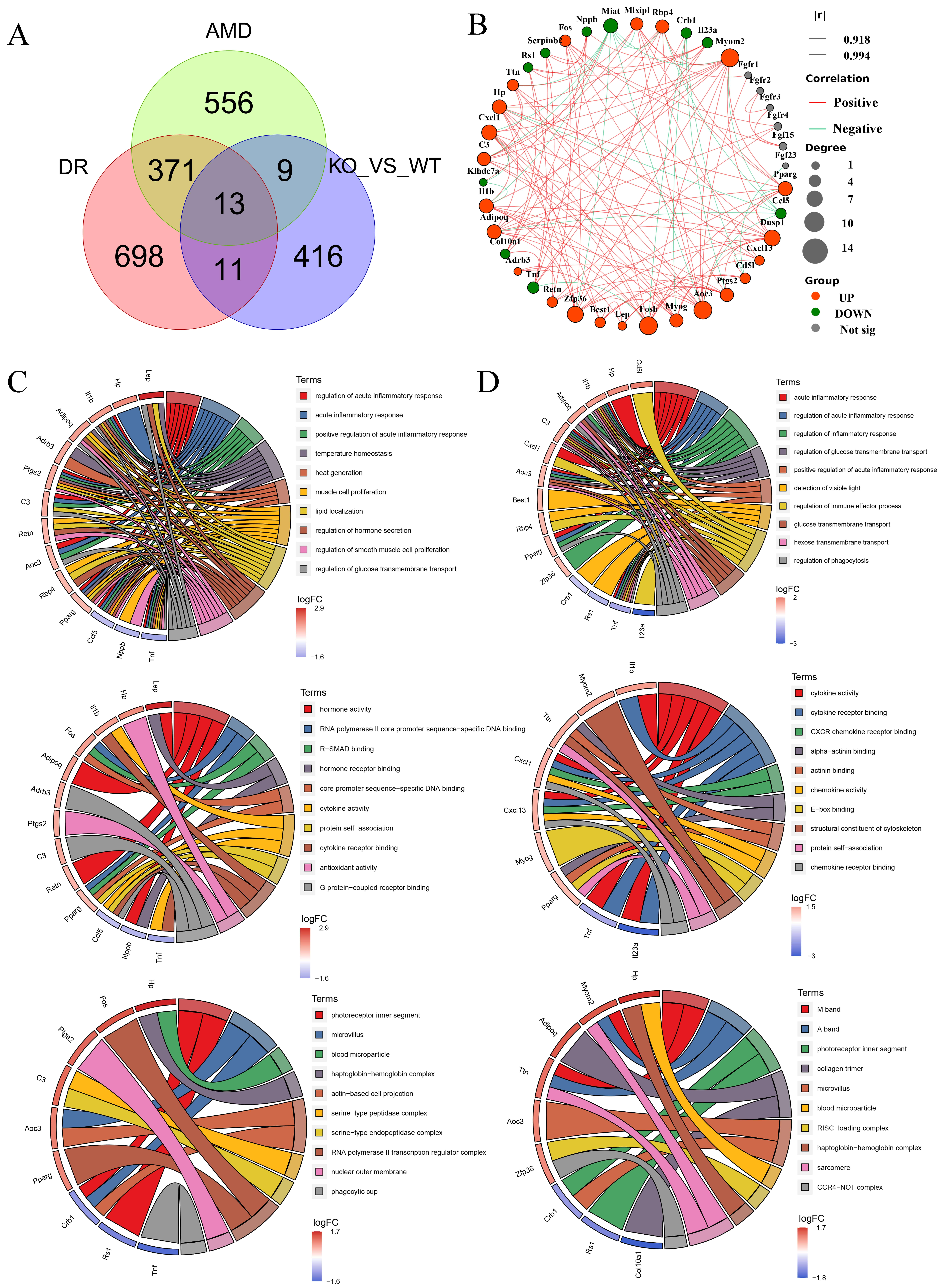 Fig. 4.
Fig. 4.
Gene enrichment analysis of age-related macular degeneration (AMD)/diabetic retinopathy (DR) associated DEGs between KO and WT groups. (A) Venn diagrams depicting DR and AMD-related genes with significantly different genes from three databases: GeneCards, DisGeNET, and Therapeutic Target Database. (B) Correlation between disease (AMD and DR) related genes, FGF receptors, and FGF19 subfamily members. (C) Chord plot illustrating GO enrichment analysis of DR-related differential genes. (D) Chord plot illustrating GO enrichment analysis of AMD-associated differential genes. Differential genes are presented on the left, with color changes indicating alterations in log2FC from small to large, with red indicating up-regulated and blue indicating down-regulated genes. Enrichment term results are displayed on the right, and the two are linked to indicate gene enrichment into that term. KO: FGF21 knockout; WT: normal control.
Pearson correlation analysis between these 33 candidate genes and members of the FGF19 subfamily (including FGF21) and their receptors (FGFRs) revealed several significant associations: Mlxipl and Aoc3 showed strong positive correlations with FGFR1, Fos, Retn, and Adipoq correlated positively with FGFR4, and Fos also correlated with FGF15. In contrast, Cd5l exhibited a strong negative correlation with Fgf23 (Fig. 4B–D). Functional enrichment analysis of these 33 overlapping genes highlighted acute inflammatory response as a key biological process linking the FGF21-deficient retinal phenotype to both AMD and DR pathogenesis (Fig. 5A,B).
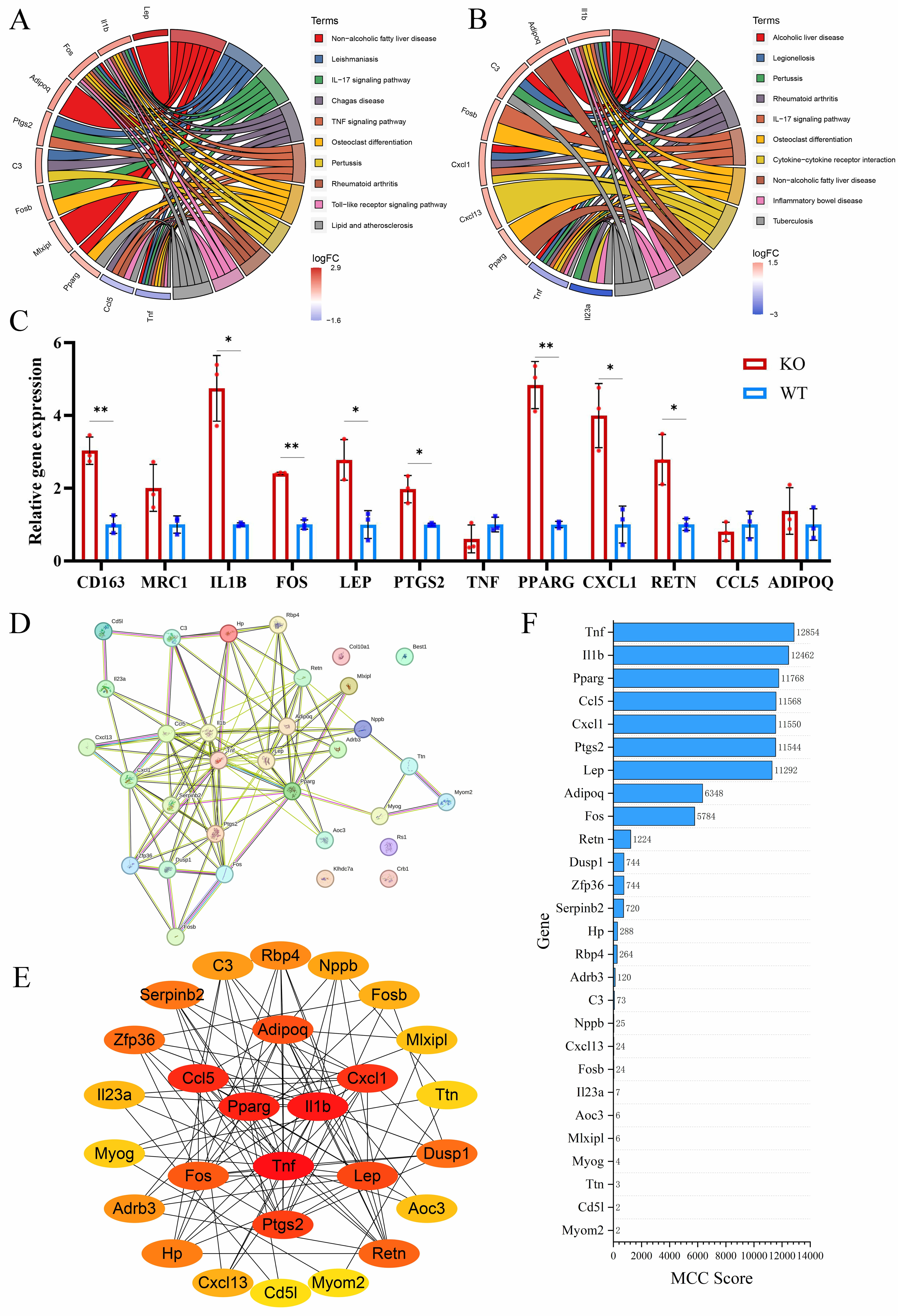 Fig. 5.
Fig. 5.
Screening of core DEGs related to AMD/DR in the retina/chorioid
of FGF21 KO mice. (A) STRING diagram illustrating KEGG enrichment analysis of
DR-related differential genes. (B) STRING diagram illustrating KEGG enrichment
analysis of AMD-related differential genes. On the left side are the differential
genes, with color changes indicating the change in log2FC from small to large,
where red represents up-regulation and blue represents down-regulation. On the
right side are the enrichment term results, and the two are connected to indicate
gene enrichment into that term. (C) Real-time PCR analysis for differential genes
in indicated groups. (D) PPI network diagram. (E) Core differential genes
identified by the MCC algorithm. (F) MCC scores and rankings of related genes.
KO: FGF21 knockout; WT: normal control. Statistical significance is indicated as
* p
In addition, we constructed a PPI network from these genes using STRING and
Cytoscape, which comprised 27 nodes and 109 interactionsFunctional enrichment
analysis of these 33 overlapping genes (Fig. 5D). Application of the MCC
algorithm identified five central hub genes: tumor necrosis factor (Tnf),
interleukin 1
To investigate the effect of FGF21 deficiency on the immune microenvironment, we estimated immune cell abundances from the RNA-seq data using the ImmuCellAI-mouse. The results revealed a significant shift in immune cell composition in FGF21 KO retinal/choroidal tissues. Specifically, the abundance of M2 macrophages was markedly increased, whereas NKT cells and germinal center B cells were decreased compared to WT controls (Fig. 6A,B).
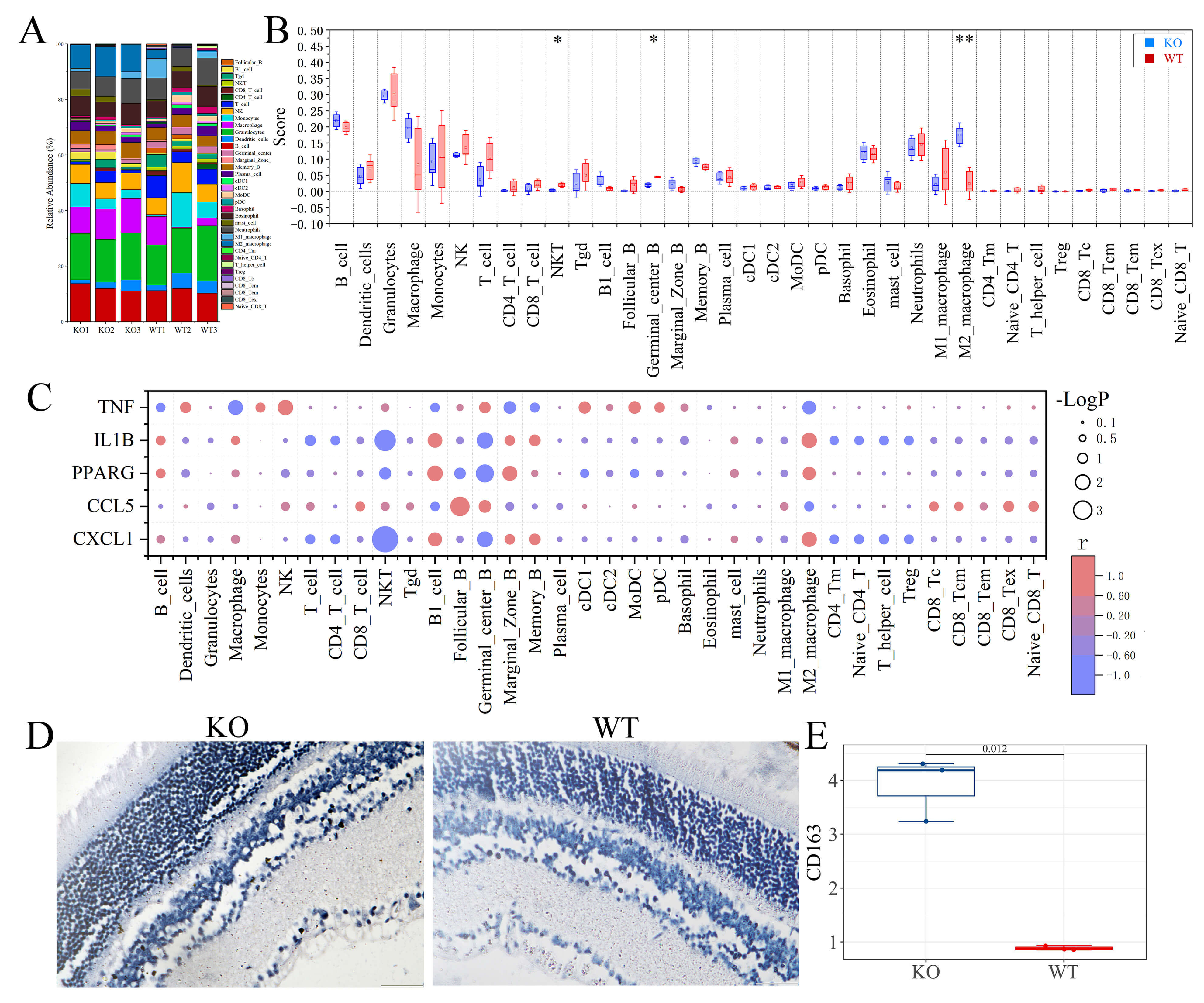 Fig. 6.
Fig. 6.
Immune infiltration analysis. (A) Relative percentages of 36
infiltrating immune cells in the KO group versus the WT group. (B) Box plot of 36
infiltrating immune cells in the KO group versus the WT group. (C) Bubble plots
showing the correlation between differential genes and immune cells. Bubble
colors represent the magnitude of correlation coefficients, indicating positive
and negative correlations. The size of the bubbles represents the magnitude of
the p-value, signifying the significance level of this sample. (D) Representative images of IHC staining for CD163. (E) Quantification of CD163 staining. Scale bar =
50 µm. *, p
Subsequently, Pearson correlation analysis between the abundance of these altered immune cells and key DEGs (Tnf, Il1b, Pparg, Ccl5, Cxcl1) showed a clear dichotomy. The increased M2 macrophage signature positively correlated with Il1b, Pparg, and Cxcl1, but negatively correlated with Tnf and Ccl5. The opposite correlation pattern was observed for the diminished NKT and Germinal center B cell populations (Fig. 6C). Together, these data indicate that FGF21 deficiency dramatically alters the retinal immune milieu, primarily by disrupting macrophage homeostasis.
Exogenous administration of fibroblast growth factor 21 (FGF21) or its analogs has shown therapeutic promise in animal models of various retinal disorders, including diabetic retinopathy, retinitis pigmentosa and retinal neovascularization [7, 8, 9, 10]. Our prior work further demonstrated its protective effect against the progression of dry age-related macular degeneration (dAMD) [11]. These consistent findings across models underscore FGF21 signaling as a potential therapeutic target for retinal diseases.
However, a critical gap exists between the well-characterized pharmacological
effects of FGF21 and the understanding of its endogenous physiological functions.
Notably, whereas administration of recombinant FGF21 improves systemic metabolism
(reducing hyperglycemia and body weight) [17, 18, 19, 20], FGF21 KO or its coreceptor
Importantly, while exogenous FGF21 alleviates retinal pathology, overt structural lesions are rarely reported in unchallenged FGF21 KO mice. In line with this, our study also found no detectable fundus abnormalities in 6-week-old KO mice. The results indicate that endogenous FGF21 is not required for maintaining gross retinal structure under physiological conditions.
However, transcriptomic analysis revealed a contrasting picture at the molecular level. Despite the absence of structural or systemic metabolic defects, FGF21 deficiency significantly altered multiple inflammatory and metabolic pathways within the retinal/choroidal tissue. The lack of concurrent systemic metabolic dysregulation in our mice allows us to attribute these transcriptional changes primarily to the local absence of FGF21 signaling in the retina/choroid, rather than secondary effects of whole-body metabolic disturbance.
The disease relevance of these transcriptomic alterations was underscored by the significant overlap between our DEGs and genes associated with major retinal diseases, particularly AMD and DR. Among the intersecting genes, key regulators of inflammation and immunity emerged, including Il1b, Cxcl1, and Pparg (upregulated), as well as Tnf and Ccl5 (downregulated). This signature strongly indicates that FGF21 deficiency fundamentally reshapes the retinal inflammatory microenvironment.
Retinal inflammation is a pivotal driver in the pathogenesis of AMD and DR
[24, 25, 26]. This study reveals that endogenous FGF21 deficiency creates a complex
immune milieu distinct from the anti-inflammatory effects of its pharmacological
supplementation. This complexity is exemplified by two observations: first,
the downregulation of Tnf in KO mice, contrasting with reports that exogenous
FGF21 can reduce TNF-
The first paradox likely originates from the fundamental difference between
chronic genetic deficiency and acute pharmacological intervention. The former
permits long-term adaptations, such as the marked compensatory upregulation of
Pparg we observed. Given that PPAR
The second observation can be understood by looking beyond the classical M1/M2
dichotomy. Recent studies emphasize that macrophage activation exists on a
functional continuum, where specific subsets can produce pro-inflammatory factors
including IL-1
Thus, the immune microenvironment in the retina of FGF21 KO mice appears to be
reconfigured rather than simply attenuated. The concurrent rise in Il1b suggests
a shift towards a specific inflammatory axis, while the PPAR
Additionally, the observed shift toward an M2-like macrophages underscores a disruption in macrophage polarization homeostasis, a critical aspect of retinal immune balance. Given the high plasticity of macrophages, this M2-skewed state in young FGF21 KO mice may represent a specific adaptation to the early, FGF21-deficient microenvironment. It is plausible that this balance could shift further under prolonged stress or with aging. Regardless of the specific phenotype, the overall increase in macrophage presence and the imbalance itself are recognized as key drivers of retinal pathology. Both M1 and M2 macrophages can contribute to pathogenic processes such as abnormal neovascularization through distinct mechanisms, thereby promoting the progression of diseases like AMD and DR [33, 34, 35]. Thus, the dysregulated macrophage landscape we identified provides a plausible cellular mechanism linking endogenous FGF21 deficiency to increased retinal disease susceptibility.
While our study in FGF21 KO mice establishes a principled link between
endogenous FGF21 and retinal immune homeostasis, several considerations must be
addressed when translating these findings to human disease. First,
inherent species differences in retinal anatomy and immune cell repertoire may
modulate the phenotype. Second, human age-related macular degeneration (AMD) and
diabetic retinopathy (DR) are multifactorial, chronic disorders influenced by
genetics, environment, and aging. In contrast, this study uses a single-gene
knockout model raised in a controlled, simplified laboratory environment, which
cannot fully replicate the multifaceted nature of the human conditions.
Nonetheless, our discovery provides a testable hypothesis for human pathology.
Future studies should prioritize: (1) validating this immune signature in human
retinal sections or vitreous samples from AMD/DR patients, correlating FGF21
pathway activity with macrophage polarization markers; (2) employing human
retinal organoids or induced pluripotent stem cell (iPSC)-derived
microglia/macrophage co-cultures to dissect the cell-autonomous effects of FGF21
signaling in a human genetic background; and (3) exploring targeted therapeutic
strategies, such as developing eye-localized delivery systems for FGF21 analogs
or modulators of its downstream targets (PPAR
This study also has several limitations. First, as an exploratory investigation, the sample size for the transcriptomic analysis, though aligned with similar discovery-phase studies, may limit the detection of genes with subtle expression changes. Nevertheless, the robust pathways and cell population shifts we identified provide strong preliminary evidence and clear targets for future hypothesis-driven studies with appropriate power analysis.
Second, while we have provided protein-level validation for the central immunological phenotype (M2 macrophage increase via CD163 IHC, Fig. 6D,E) and transcriptional validation for key cytokines, we acknowledge that we did not perform comprehensive protein quantification for all identified differentially expressed genes (DEGs). This was primarily due to the limited protein yield from mouse retinal/choroidal tissues. Our validation strategy was therefore intentionally focused on confirming the most robust and functionally central finding. Finally, while our data reveal a strong association between the M2-skewed microenvironment and increased disease susceptibility. Direct functional assays, such as macrophage-specific depletion or adoptive transfer experiments, are required to establish causality. Future work should prioritize such functional validation alongside the proteomic and spatial transcriptomic approaches noted above.
The raw data supporting the conclusions of this article will be made available by the authors on request.
Conceptualization, TZ and BS; Methodology, MC and HG; Software, MC; Investigation, TZ, MC, WW, HG; Formal analysis, BS, MC; Validation, YH; Data curation, TZ, WW, MC; Writing—original draft preparation, TZ and MC; Writing—review and editing, WW; Supervision, BS, WW. All authors have read and agreed to the final version of the manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the ARRIVE guidelines (https://arriveguidelines.org) and was approved by the Animal Ethics Committee of Northeast Agricultural University (NEAUEC20240135).
We give our sincere thanks to Grace Liu, Oliver Kang and Lismarie Sun from Intalight company for their help in this study.
This research was funded by grants from the Guangxi Natural Science Foundation Project, China (2025GXNSFAA069182); Natural Science Foundation of Hunan Province, China (2023JJ70050); Guangxi Zhuang Autonomous Region Health Commission Self-funded Scientific Research Project (Z-A20251080).
The authors declare no conflicts of interest. No funding or employment was received from Intalight company.
In this study, to enhance the linguistic quality of the paper, we employed an artificial intelligence language polishing tool, ChatGPT, to optimize certain paragraphs. We chose to use this tool due to its outstanding performance in terms of language fluency and grammatical accuracy. We ensured that all polishing work was carried out without altering the original research data and analysis results. The specific polishing process involved a section-by-section assessment of the initial draft, followed by modifications based on the tool’s suggestions. We believe that this method of polishing has improved the readability and professionalism of the paper while maintaining the integrity and accuracy of the research content. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL47702.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






