1 Department of Biosciences, Biotechnology and Environment, University of Bari, 70121 Bari, Italy
2 Department of Health Science, University of Florence, 50139 Florence, Italy
3 Pancreas Unit, Careggi University Hospital, 50134 Florence, Italy
4 Department of Experimental and Clinical Medicine, University of Florence, 50134 Florence, Italy
5 Department of Experimental and Clinical Biomedical Sciences “Mario Serio”, University of Florence, 50134 Florence, Italy
†These authors contributed equally.
Abstract
Extracellular vesicles (EVs) are small nanometric particles surrounded by a lipid bilayer and actively secreted by different cell types. EVs play a key role in cell-to-cell communication, and the vast array of biomolecules that EVs transport reflects the molecular profile of the originating cells. In cancer, EVs are key components of the tumor microenvironment (TME); meanwhile once released into the peripheral circulation, EVs can travel systemically and transmit signals beyond the primary tumor site, making EVs ideal candidates for liquid biopsy. Notably, EVs can be isolated from a blood sample and analyzed to obtain real-time information on tumor biology, enabling early diagnosis, monitoring of treatment response, and evaluation of disease evolution, with high sensitivity and specificity. This review examines the biological significance and clinical utility of EVs expressing the stemness-associated glycoproteins CD44 and CD133 in gastrointestinal (GI) malignancies. Cancer stem cells (CSCs) expressing these surface markers are known to exhibit enhanced tumorigenic potential, metastatic capacity, and therapy resistance. In particular, we focus on the increasing evidence that EVs enriched in CD44+ and CD133+ populations play critical roles in key aspects of tumor progression in cholangiocarcinoma, pancreatic, colorectal, and gastric cancers. Following internalization by recipient cells, CD44+ and CD133+ EVs drive phenotypic reprogramming, foster more aggressive cellular states, and promote chemoresistance by delivering specific molecular cargo. Mechanistically, CD44 isoforms, particularly CD44v6 and CD44v9, activate key oncogenic signaling pathways, including Wnt/β-catenin and phosphoinositide 3-kinase (PI3K)/serine/threonine kinase AKT (AKT). In parallel, CD133-enriched EVs help maintain stemness and contribute to TME reorganization, thereby facilitating tumor progression. Despite ongoing challenges in EV isolation and standardization, EVs positive for stemness markers show great potential as liquid biopsy analytes for noninvasive disease monitoring, prognostic evaluation, and patient stratification. This review summarizes the expanding body of knowledge on cancer stem cell (CSC)-derived EVs in GI tumors, underscoring the potential of these particles for early diagnosis, prognosis, and the development of targeted therapies to overcome treatment resistance.
Keywords
- liquid biopsy
- extracellular vesicles
- cancer stem cells
- CD44
- CD133
- biomarkers
Extracellular vesicles (EVs) are nano-sized, membrane-bound structures released into the extracellular space by various cell types either constitutively or in response to stimuli. They are crucial mediators of cell-cell communication, capable of altering the behavior of target cells both nearby and at distant sites [1, 2]. EVs encompass distinct subtypes [2, 3, 4] categorized by size and biogenesis:
Apoptotic bodies (Ø = 1000–5000 nm), ectosomes (Ø = 200–1000 nm),
exosomes (Ø = 30–150 nm) and exomeres (Ø
Exosomes begin with the formation of a small endocytic vesicle (early endosome) by the budding of the inner cell membrane, a process that is controlled by intracellular calcium levels. Then the enlargement of the endocytic vesicle forms a late endosome that generates, by invagination of its membrane inward, nanometer intraluminal vesicles (ILVs) collectively called multivesicular bodies (MVB). Ultimately, ILVs are redirected either toward lysosomal degradation through MVB-lysosome fusion or toward secretion into the extracellular space through MVB-plasma membrane fusion [6, 7]. Exosome production is regulated via two main pathways: the endosomal sorting complex for transport (ESCRT)-dependent and ESCRT-independent mechanisms. The highly conserved ESCRT machinery, comprising four multiprotein complexes (0-II-II-III), orchestrates endosomal membrane deformation and the sorting of ubiquitinated cargo into ILVs, thereby preventing recycling and retrograde transport [8]. In contrast, the ESCRT-independent pathway relies predominantly on lipid rafts and tetraspanins, involving proteins such as caveolin-1, flotillins, and Rab31 to mediate ILV formation [9]. Annexins (in the ESCRT-dependent route) and Rab GTPases (in the ESCRT-independent pathway) regulate late endosome budding from early endosomes and influence MVB trafficking and docking at the plasma membrane [1, 10, 11]. Various factors at the endo-lysosomal interface modulate EV production and release [12], and perturbations in these processes have been implicated in several diseases, including cancer [13, 14]. Notably, alterations in endosomal pH within early and recycling endosomes have been associated with tumor development and therapy resistance [15]. Additionally, dysregulation of the lysosomal system, such as overactivation of the c-MYC/HDAC2-MiT/TFE axis, can suppress lysosome biogenesis and autophagy, contributing to cancer progression. Monitoring EV release and cargo in response to these alterations is therefore essential for understanding disease mechanisms [13].
At each stage, from the endocytic vesicle to the exosome, EVs carry diverse biological cargos such as DNA, RNAs, proteins, and lipids, which may have metabolic significance and signaling potential since they reflect their cell of origin [16]. Exosomes play a crucial role in tumor progression by regulating processes such as angiogenesis, metastasis and immune suppression [17]. Importantly, cancer cells secrete significantly higher amounts of EVs compared to normal cells [18], suggesting the modulation of exosome release as a potential therapeutic strategy.
As main mediators of cell-cell communication, EVs play a pivotal role in shaping the TME and forming the pre-metastatic niche [19, 20] mainly through the integrin-driven organotropism, which directs EVs to specific target organs [21].
Within this context, tumor-derived exosomes (TDEs), which can be readily isolated from biological fluids, have emerged as promising biomarkers for liquid biopsy—a non-invasive tool for diagnosis, patient monitoring, and stratification in the era of precision oncology [3, 22, 23, 24]. Of particular interest are stemness-related molecules as putative biomarkers for various cancers, including gastrointestinal (GI) tumors [25]. Stemness is a crucial process in cancer development [26] and the dissemination of CSCs with the support of TME is believed to be responsible for the metastatic progression [27]. Epithelial cell adhesion molecule (EpCAM), Leucine-rich repeat-containing G-protein coupled receptor 5 (Lgr5), aldehyde dehydrogenase (ALDH) and particularly CD44 and CD133 are the most well-known and described CSC markers and their role in the promotion of tumorigenic potential has been deeply explored [26]. EpCAM is highly expressed in epithelial-derived tumors, such as hepatocellular carcinoma (HCC), and has been identified as a negative prognostic marker for recurrence and survival following surgical resection [28, 29, 30]. Regarding Lrg5, although many studies have reported a pivotal role in supporting cancer progression, inconsistent results were obtained when evaluating its expression in relation to patient survival [31]. ALDH is another stemness marker that has been described as involved in tumor relapse and development of chemoresistance and the possibility to target stem cells expressing ALDH has been proposed as a strategy to improve patient response to therapy in many cancers [32].
CD44 and CD133 are probably the most studied CSC markers, they have been widely used either alone or in combination to sort and characterize this specific cell population to better understand mechanisms driving tumor development of chemoresistance [33, 34]. Along with the idea of taking advantage of CD44 and CD133 expression to detect CSCs, the investigation of CSC-derived EVs appears as a novel, promising solution to provide an accessible representation of the tumor of origin and monitoring tumor progression [35]. The study of EVs derived from CSCs gained even more attention considering that CD63, one of the main EV markers, has been recently described as a novel liver stem cell marker [36].
Overall, the application of CD44+ and CD133+ EVs in liquid biopsy is still a field to be explored and an evident gap in literature was observed despite multiple studies conducted on the role of CD44 and CD133 in different tumors, including GI cancers.
In this work, we review current literature on this topic trying to shed light on possible therapeutic applications to identify potential stemness-related targets (Fig. 1) and opening a new perspective in the use of EVs as informative biomarkers.
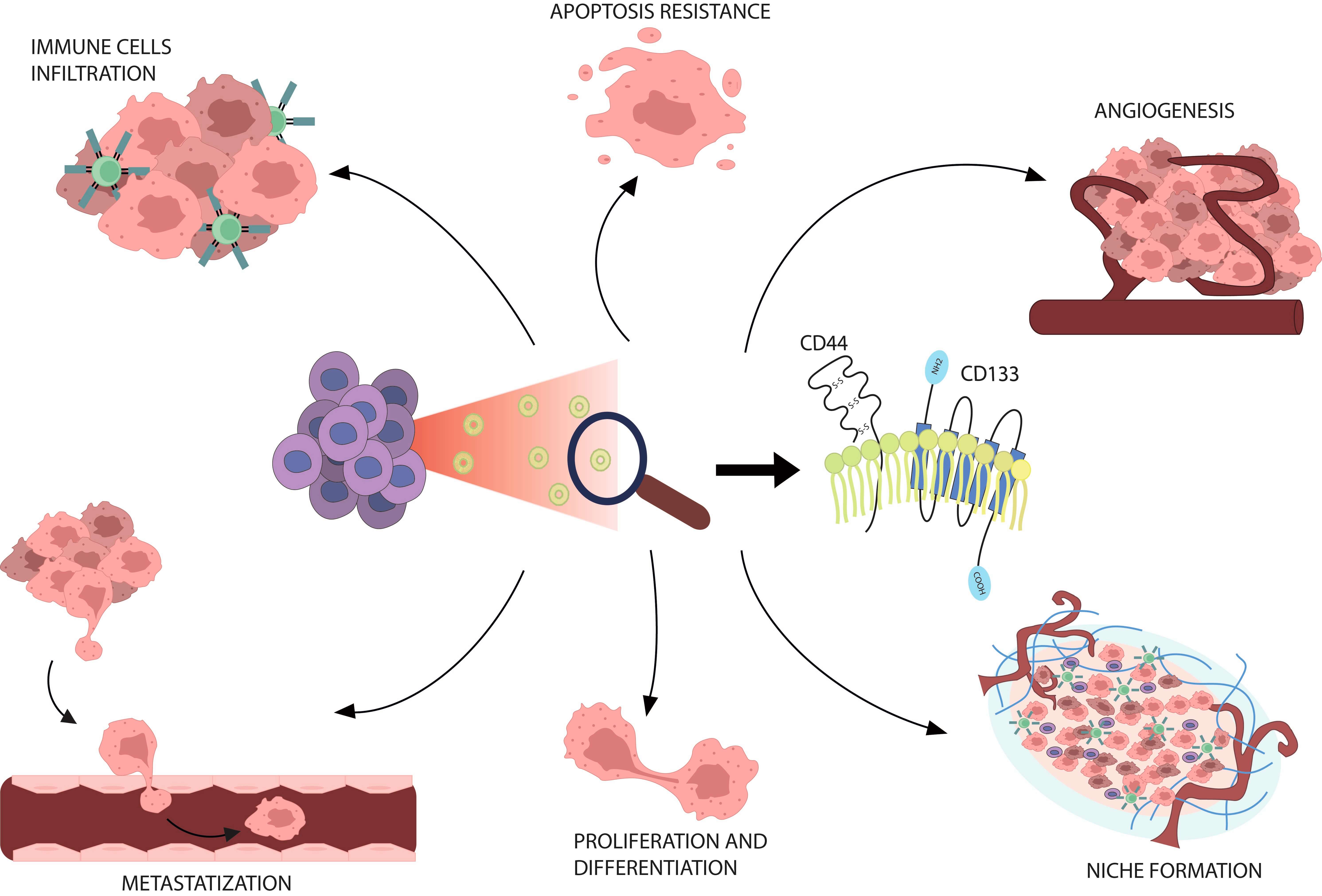 Fig. 1.
Fig. 1.
CSC-derived EVs and cancer. CD44+ and CD133+ EVs released in circulation by CSCs are involved in a variety of tumor progression mechanisms in GI cancers. CSCs, cancer stem cells; EVs, Extracellular vesicles; GI, gastrointestinal.
Localized on chromosome 11, CD44 is a transmembrane glycoprotein with
constitutive expression. Due to alternative splicing, it exists in different
isoforms, mainly, CD44 standard (CD44s) and CD44 variant (CD44v) [37] that can
interact with various ligands, including hyaluronic acid (HA), osteopontin (OPN)
and matrix metalloproteinases (MMPs) [38]. In cancer cells, CD44 expression has
been observed mostly in filopodia implying a role in tumor spreading [39, 40]. As
mentioned, the upregulation of its expression, together with CD133, ICAM1, and
LGR5 is distinctive in CSCs [41, 42]. The interaction of CD44 with its ligand of
choice, HA, triggers tumor proliferation and suggests a possible targeting of
this process to manage progression of cancer [37, 43]. Signaling pathways
activated by HA-CD44 binding involve Ras, mitogen-activated protein kinases
(MAPK), phosphoinositide 3-kinase (PI3K) and
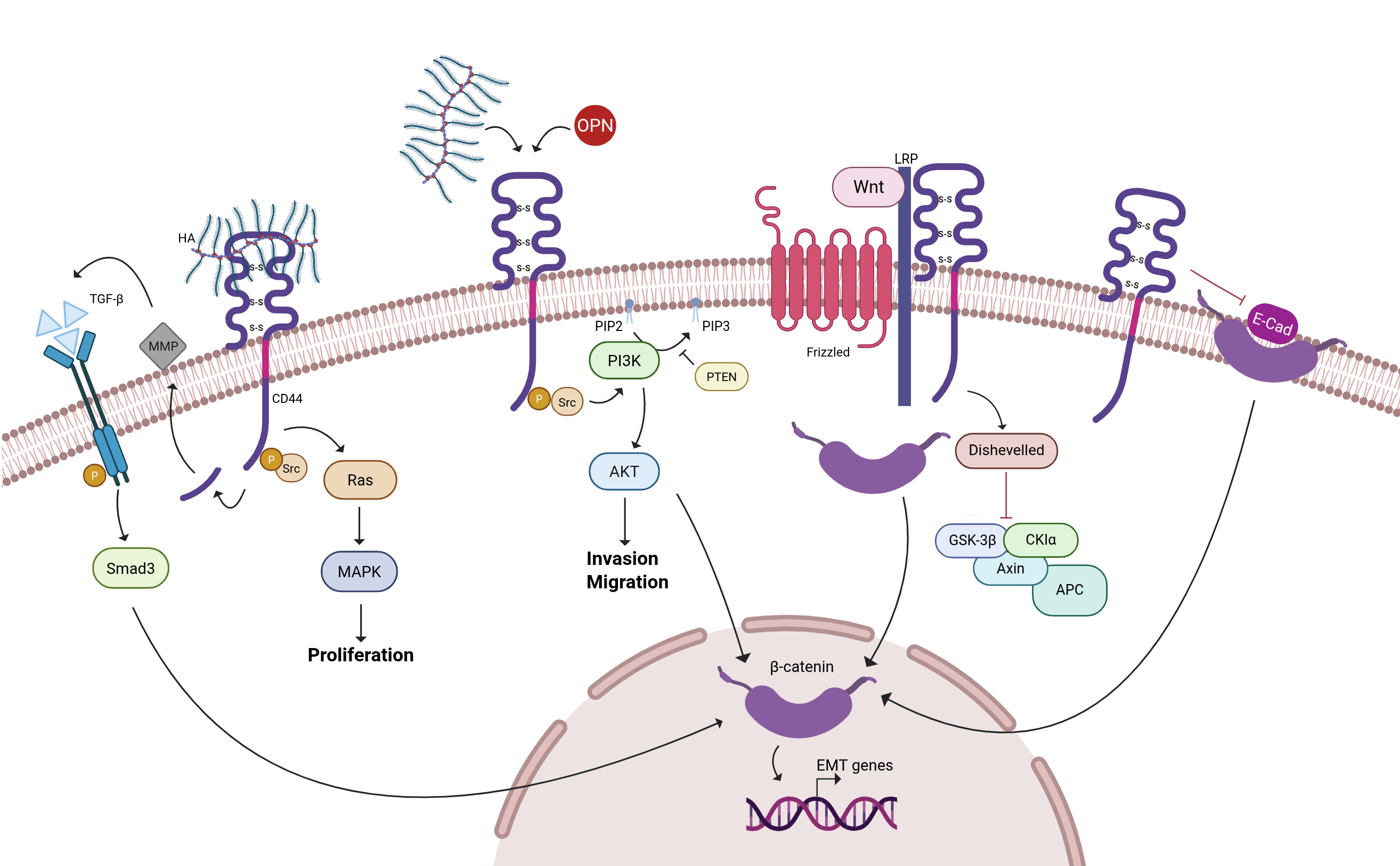 Fig. 2.
Fig. 2.
Signaling pathways regulated by CD44. CD44 and CD44v can
interact with various ligands, including hyaluronic acid (HA), osteopontin (OPN)
and matrix metalloproteinases (MMPs). These interactions contribute to the
regulation of numerous key cellular signaling pathways, including PI3K-Akt,
Ras-MAPK and Wnt/
The interaction with OPN has also been widely studied for the implication in tumor progression. OPN binds predominantly to CD44v6 and CD44v and activates many pathways including PI3K, Akt pathway and c-Jun-NH (2)-kinase (JNK) to promote the metastatic process [45]. Interestingly, in glioma, the interaction has been reported to facilitate resistance to radiation through the promotion of a stemness phenotype [46]. Similar results were obtained in nasopharyngeal carcinoma, where the inhibition of the CD44-Stat3 axis and consequent reversion of epithelial-mesenchymal transition (EMT) by afatinib promotes sensitivity to radiation [47].
In general, the targeting of CD44 is the strategy of choice over the targeting of ligands [45], although limited success was obtained in several clinical trials that have investigated the efficacy of treatments that aim at CD44+ CSCs [37]. Mostly, anti-CD44 monoclonal antibodies have been screened, of interest the humanized anti-CD44v6 monoclonal antibody (mAb) BIWA4 (bivatuzumab)-mertansine drug conjugate was tested in a clinical phase I trial, on patients with head and neck squamous cell carcinoma. However, severe skin toxicities were observed and determined a suspension of the study [48].
More recently, the humanized anti-pan-CD44 mAb, RG7356 has shown a decent safety profile in patients with acute myeloid leukemia and advanced CD44-positive solid tumors in phase I trials. Indeed, no clinical and/or pharmacodynamic dose–response followed the treatment, implying a deeper study of anti-CD44 antibody optimization [49]. Another strategy adopted was the development of CD44v6-specific chimeric antigen receptor-modified T (CAR T) cells that have been tested in various clinical trials (clinicaltrials.gov ID: NCT04427449, clinicaltrials.gov ID: NCT04430595), but so far, the results of these studies have not been published [50].
Promising preclinical studies paved the way to the use of novel approaches involving HA-modified liposomes [43] or HA-curcumin nanocarriers engineered to be internalized through the interaction with CD44 to maximize the effect of therapy in MCF-7 and 4T1 cells [51], but more studies are needed to support this approach and, eventually, extend it to the use of EVs. The possibility of blocking the loading of CD44 in TDE or directly CD44+ EV-release [39] seems promising; indeed the targeting of EVs could overcome several limitations observed in CD44 clinical trials, although more research is necessary.
CD133 is a glycoprotein formed by five transmembrane domains and encoded by PROM1 gene; various alternative splicing of the main transcripts have been described with splice variants that differ in the N- and C-terminal domain sequences. Although evidence of CD133 as a membrane receptor is lacking, probably because ligands are still unknown, its role in the regulation of several signaling pathways has been deeply described [52].
In particular, the subcellular localization of CD133 in plasma membrane protrusions, such as microvilli, depends at least in part on its interaction with cholesterol within membrane microdomains known as lipid rafts, rich in specific lipids such as cholesterol and sphingolipids [53].
The involvement of CD133 in the organization of plasma membrane protrusions, as
well as in the regulation of oncogenic signaling pathways such as PI3K/Akt,
Src-FAK, and Wnt/
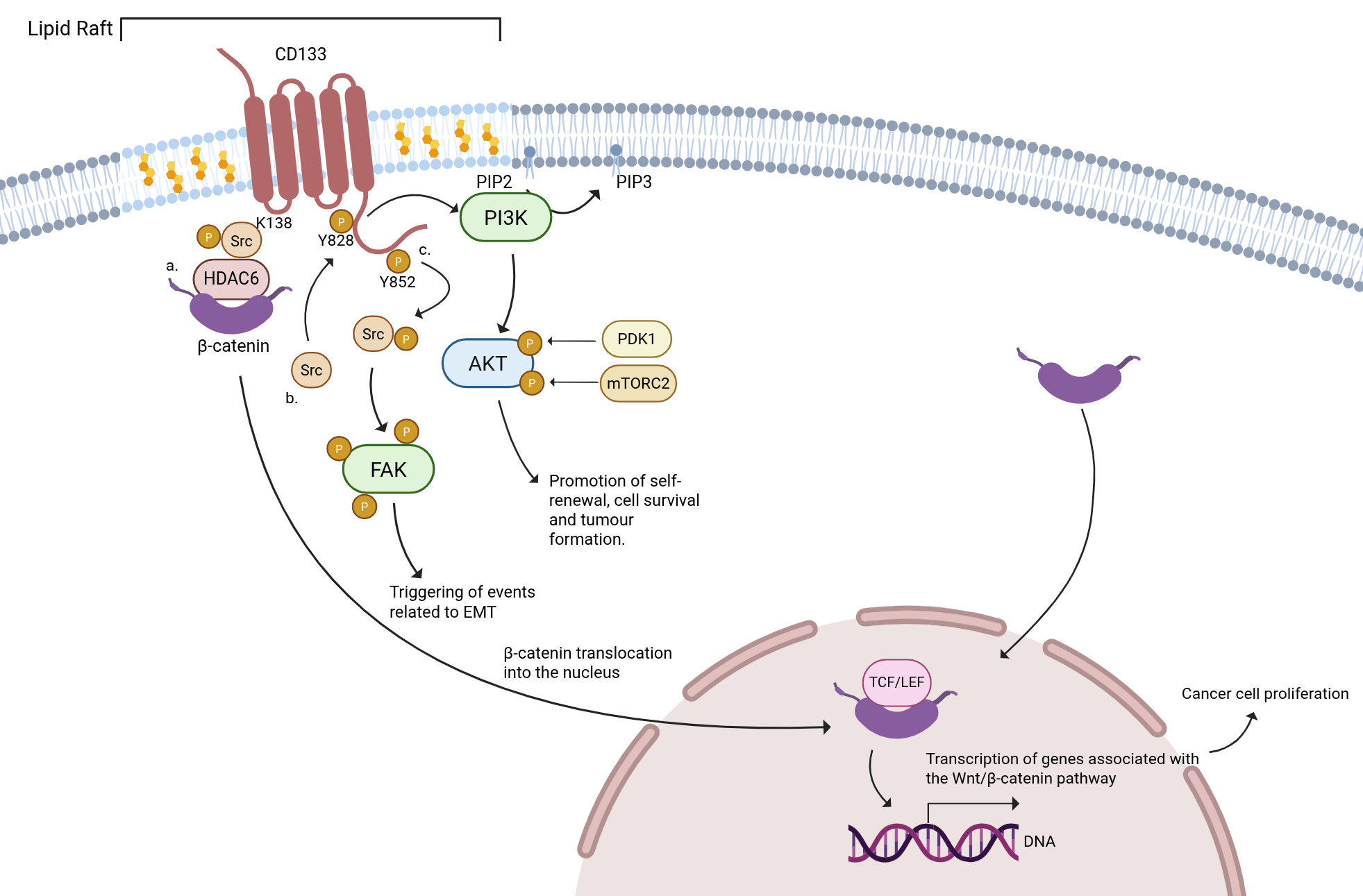 Fig. 3.
Fig. 3.
Signaling pathways regulated by CD133. The interaction of CD133
and HDAC6 mediated by cytoplasmic K138 in the first cytoplasmic loop (IC1) has an
impact on the proliferation and differentiation of cancer cells. CD133 forms a
ternary complex with HDAC6 and
Along with this, CD133 is also considered a marker of stem cells in different
types of cancer, originally described as a marker for hematopoietic stem cells;
recently, its role in the context of CSCs has been investigated [55]. Mainly, it
has been observed that its expression promotes the proliferation of CSCs together
with the induction of their spreading to develop metastasis. In glioma, when
hypoxic conditions are present, the glycosylation of CD133 has been linked to the
improvement in survival of CSCs [56], suggesting a possible correlation between
oxygen level, CD133 expression and glycosylation status. Also, CD133 can manage
the triggering of nuclear factor-kappa B (NF-
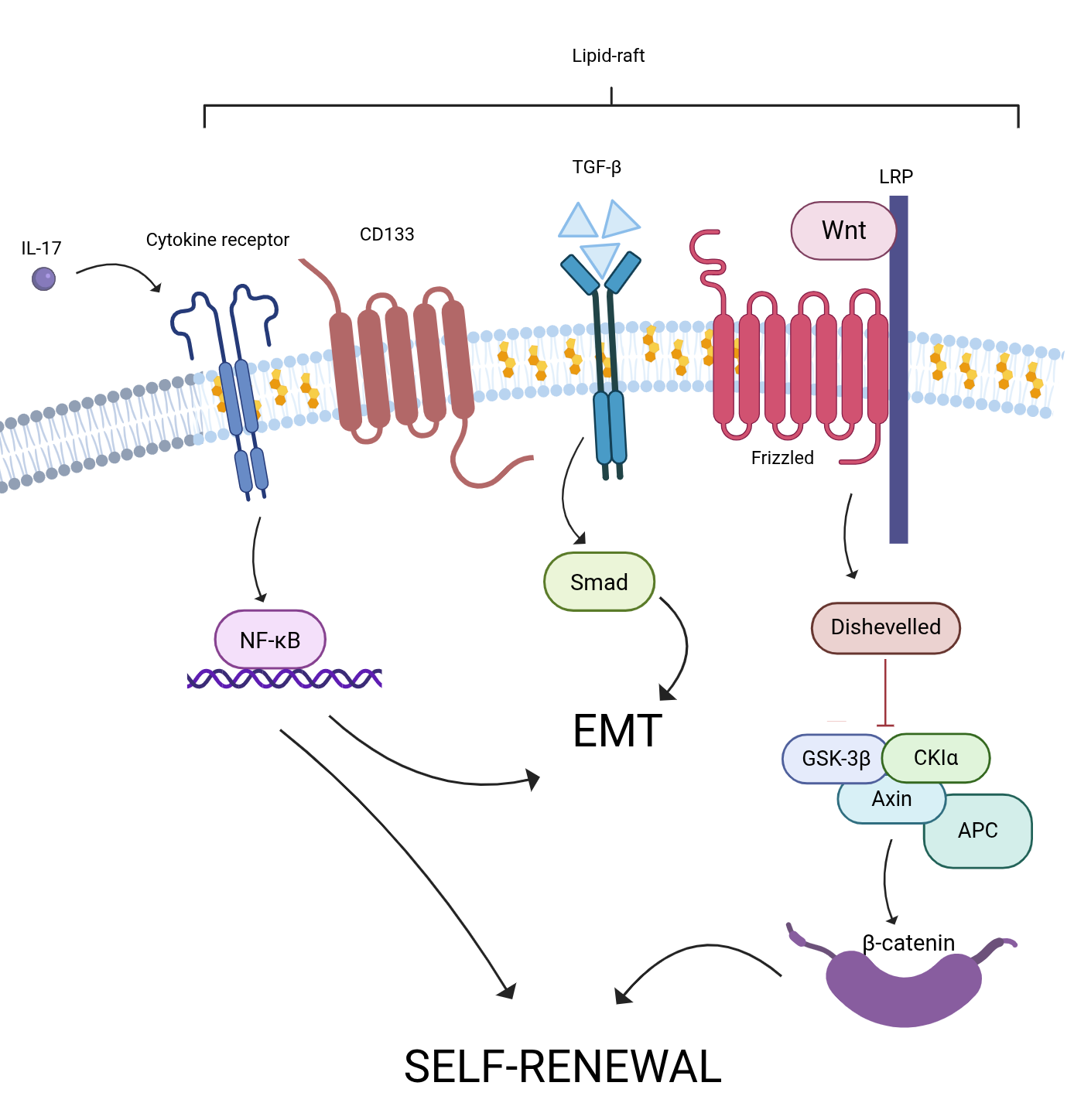 Fig. 4.
Fig. 4.
CD133 and lipid rafts: key players in CSC signaling. Lipid
rafts of CSCs CD133+ promote self-renewal through: (1) The Wnt/
The pivotal role of CD133 in tumorigenesis and maintenance of stemness properties in CSCs suggests a possible use as a novel target for therapy, although the different splice variants and post-translational modifications limit its targeting. Indeed, glycosylation is often responsible for hiding specific epitopes with a subsequent lack of antibody efficacy; this was also true for commercially used Miltenyi antibodies such as CD133/1 (AC133 or W6B3C1) or CD133/2 (AC141 or 293C3) that, despite the targeting of glycosylated epitopes, can fail due to modifications in the glycosylation pattern. Few antibodies have been developed for non-glycosylated regions, with even more conflicting results achieved [55, 59].
Many in vitro studies investigated the reduction of tumor angiogenic or proliferation properties after CD133 silencing [58, 60] and the managing of chemoresistance through the targeting of CSCs [57, 61, 62]. A paper by Fan et al. [63] tested NOTCH signaling pathway inhibition as a strategy to deplete CD133+ CSCs in glioblastoma; in HCC similar results were obtained by Hemati et al. [64]. The use of an anti-CD133 mAb conjugated with a cytolethal distending toxin gave positive results in head and neck squamous cell carcinoma cells [65], but no clinical trials followed this study.
Interestingly, the promising approach of using CAR T cells CD133-directed therapy has been explored in a phase II clinical trial (NCT02541370) on hepatocellular carcinoma patients, achieving encouraging findings in terms of safety and managing tumor recurrence. Specifically, 13% of patients enrolled in the study reached partial remission, 60% stable disease and the 3-month disease control rate was 65.2% [66, 67].
Regarding the targeting of CD133+ EVs a lot still needs to be done, but the possibility of selectively acting on the so-called “prominosomes” [68] is fascinating and deserves further investigation. Of interest, in a recent paper, the presence of CD133 has been observed in a new defined class of mRNA-enriched vesicles negative for the expression of the main EV markers called intercellsomes capable of migrating between cells [69], unraveling a novel role of CD133 in intra- and inter-cellular signals.
CCA is a heterogeneous group of malignant neoplasms arising from the biliary epithelium. Characterized by asymptomatic progression that may persist until advanced stages, CCA is associated with high malignancy, frequent recurrence, and significant diagnostic and therapeutic challenges. Despite advances in the molecular understanding of the disease, the management of CCA remains complex and often ineffective, highlighting the need for novel targeted therapeutic strategies [70]. Due to the lack of reliable risk factors and specific biomarkers, early diagnosis remains difficult. Currently, diagnostic approaches are primarily based on imaging techniques, such as ultrasonography, computed tomography (CT) and magnetic resonance imaging, along with histological examination of tumor biopsies and nonspecific serum biomarkers, such as carbohydrate antigen 19-9 (CA19-9) and carcinoembryonic antigen (CEA) [71]. Among the CSC markers in CCA, CD44 and CD133 have been widely studied. In particular, the CD44v9 variant has been linked to tumor growth and inflammation; in fact, when silenced, stem-like features such as drug resistance and invasion are reduced, showing its important role in keeping the cancer aggressive [72, 73]. Other variants like CD44v6 and CD44v8-10 have also been studied as possible markers of tumor progression, worse outcomes for patients and a shorter recurrence-free survival as shown in a retrospective study by Padthaisong et al. [74].
As for CD133, Mizukami et al. [75] analyzed 82 patients with extrahepatic bile duct cancer and gallbladder cancer and showed that its cytoplasmic expression is linked to poor prognosis and worse tissue differentiation, making it an independent prognostic factor. However, the role of CD133 in intrahepatic CCA is still debated. For example, Shimada et al. [76] found that cytoplasmic CD133 is related to poor prognosis, but Leelawat et al. [77] found no link between CD133 and patient survival. On the other hand, Fan et al. [78] found that CD133 expression was associated with longer survival. These different results may be due to several factors, such as differences in lab methods, how CD133 positivity is defined (e.g., cytoplasmic vs membrane expression), and variation in patient samples (disease stage, treatments, surgery).
Despite multiple findings on the role of CD44 and CD133 in CCA, little is known on CD44+ and CD133+ EVs. Few studies opened up to the possibility of using these two markers alone or in combination to support the diagnosis and monitoring of CCA. The expression of CD44 in EVs has been linked to tumor growth and progression in patients with inoperable CCA that received selective internal radiotherapy. Interestingly, CD44v6 was screened together with other molecules to be used as an EV-surface marker of CCA, even though the combination of MUC1, EGFR, and EPCAM was shown to outperform CD44 in detecting tumor-derived EVs in bile samples in a prospective study by Jeong et al. [79].
A paper by Urban et al. [80] evaluated the specificity of the combination of surface antigens AnnV, CD133, CD44v6 and CD326 (EpCAM) to discriminate biliary cancers from HCC, obtaining positive results in both cell line-derived EVs and patient serum-derived EVs. Promising data on the diagnostic potential of CD133 in patient serum were already obtained by Julich-Haertel et al. [81] in tumor-associated microparticles, even though few advancements followed these findings.
Given the limited number of studies, further investigations seem to be required to better shape how CSC-derived EVs take part in tumor progression in the context of CCA, especially in light of recent advancements in the use of EVs as a source of biomarkers [82].
Pancreatic ductal adenocarcinoma (PDAC) is the most common pancreatic cancer (PC), accounting for about 90% of all pancreatic tumors. It is associated with a poor prognosis since it does not show specific symptoms and often is diagnosed at an advanced stage when metastases are present and patients are inoperable [83]. For this reason, the identification of novel specific molecules for an earlier diagnosis seems necessary. Both the standard and the variant forms of CD44 have been studied and linked to tumor progression processes in PDAC. In particular, in vitro studies corroborated the idea of CD44s as a driver in the acquisition of a mesenchymal phenotype [84] through the Snail-MMP axis [85] and CD44s high cells have been related to the development of gemcitabine resistance, although more recent studies have described the variant CD44v9 as the major player in the promotion of resistance to therapy [86].
CD44v6 variant seems to be involved mainly in promoting a metastatic phenotype through the interaction with different pathways, including HGF-induced MET phosphorylation and VEGFR2 [87], and, lately, through the regulation of metastasis-promoting tetraspanin Tspan8 and promotion of drug efflux by MDR1 (multidrug resistance 1) modulation [88].
Accordingly, CD133+ human PC cells have been reported as predisposed to metastasis formation and EMT. Interestingly, the first studies to be conducted showed the induction of EMT only when a hypoxic environment was present [89] while others demonstrated the direct interaction of CD133 with the ERK pathway and the modulation of Slug and N-Cadherin gene expression [90]. This was also confirmed in another paper by Nomura et al. [91], where the upregulation of stemness-related genes such as KITLG, LIN28B, c-MYC, KLF4, GLI1, SOX2, NANOG, SIRT1, POU5F1, and CXCR4 was observed in cells where CD133 was overexpressed.
The co-expression of CD44 and CD133 has been used in several studies to detect and characterize CSCs in PDAC. In a study by Chen et al. [92] formalin-fixed paraffin-embedded samples of human pancreatic carcinoma were tested and CD44+/CD133+/TF+ (tissue factor) CSC subpopulations were found more invasive and capable of forming metastases. In PANC-1 and MiaPaCa-2 cell lines, Mortoglou et al. [93] showed that the upregulation of CD44 and CD133, along with several EMT markers, seems to be triggered by miR-210 which has a pivotal role in the maintenance of a hypoxic microenvironment which is in line with first in vitro findings on CD133 that correlated its high expression with low oxygen conditions [89].
Surprisingly, CD44 and CD133 co-expression has been connected with the dysregulation of presenilin enhancer 2 (Pen2), a gamma-secretase regulator involved in key pathways like Notch signaling. In particular, it was demonstrated that CD44+, CD133+ and EpCAM+ PANC-1 cells showed higher expression of Pen2 than cells that are not expressing these markers. Moreover, Pen2+ cells were more resistant to gemcitabine, enhanced tumor spheres formation and led to increased tumor growth when injected into nude mice compared with Pen2- cells [94].
Interestingly, exosomes released by pancreatic CD44+ and CD133+ CSCs seem to play a crucial role in communicating with non-stem cancer cells (NSCCs); in a paper by Ruivo et al. [95], the establishment of this specific network by EVs promotes the adaptation of cancer cells in the tumor environment, ensuring disease progression. Furthermore, less malignant cancer cells can uptake EVs derived from malignant cells and increase their metastatic capabilities [95].
Supporting the involvement of EVs in the initiation of pre-metastatic niche, Mu
et al. [96] showed the increased expression of CD44 and integrin
These findings confirmed what was demonstrated back in 2009 by Jung et al. [98] on the involvement of CD44v in promoting the assembly of a soluble matrix in the rat pancreatic adenocarcinoma model BSp73ASML. This specific matrix supports exosomes, which can modulate target tissue cells and facilitate the formation of the pre-metastatic niche. The knockdown of CD44v4-v7 in this model (ASMLkd) confirmed that the cooperation between CD44v and exosomes is necessary to generate the pre-metastatic niche [98].
The effects of exosome-delivered CD44v6 isoform were also studied in regard to PDAC liver metastasis by Xie et al. [99]. Specifically, exosomes isolated from mouse PDAC cells expressing CD44v6 were injected in C57BL/6 mice via intrasplenic injection. In this study, they showed that the interaction between CD44v6 and C1QBP (complement C1q binding protein), forming the CD44v6/C1QBP complex, activated insulin-growth factor (IGF-1) signaling in hepatic satellite cells (HSCs) to modulate the microenvironment and promote metastasis. Supporting these findings, patients with high levels of exosomes-delivered CD44v6/C1QBP complex were shown to have a worse prognosis and a higher risk of developing liver metastasis [99].
Of note, the presence of CD133+ exosomes has been recently identified in malignant ascites from 133 PC patients, here CD133 expression seems more represented compared to other stemness markers and the glycosylation profile has been linked to patient prognosis [100]. Indeed, it has been observed that there is a correlation between the glycosylation grade of CD133 and enhanced survival in patients with advanced PC. Taken together, these findings open up a new possibility for the use of the glycosylation profile of CD133 in exosomes derived from malignant ascites as a novel biomarker to select patients with a better prognosis [101].
Colorectal cancer (CRC) is one of the diseases responsible for the most cancer-related deaths in the world, thus representing a major health problem [102]. Ongoing research for new molecular biomarkers and specifically, blood-based markers, needs implementation for the possibility of an earlier diagnosis and decrease in mortality rate [103], also considering that the translation of preclinical findings into clinics is still limited [104]. In the context of CRC and CSCs, CD44 overexpression in patients has been correlated with unfavorable prognosis and immune cell infiltration [105], which is not surprising given previous in vitro studies that demonstrated its role in several tumorigenic mechanisms such as the promotion of metastasization [106] and chemosensitivity [107].
Regarding CD133, it is considered a validated marker of CSCs in CRC [108], and its expression has been described as potentially prognostic in terms of survival rate of patients, especially when evaluated in circulating tumor cells (CTCs) [109, 110]. Importantly, concomitant expression of both CD44 and CD133 has been shown to have a significant impact on the prediction of metastatic disease [111] and the development of therapy resistance [112].
Despite numerous studies on the applicability of CD44 and CD133 in the context of the identification of new biomarkers for CRC, the putative role of EVs positive for these stem cell markers is still under investigation. As highlighted in other tumors, CRC-derived CD44+ exosomes are involved in niche formation, and their cargo contributes to invasion and motility [113, 114]. Regarding cell motility, a study by Kelemen et al. [115] compared cells with low or high expression of CD44, showing a higher proliferative capacity and increased EV release in CD44 high cells compared to cells with a lower expression of CD44. A different expression of CD44 was able to distinguish EV-cargo, in particular miR-100, miR-95 and miR-365 were specific for CD44high EVs and miR-345 for the CD44low EVs. Furthermore, EVs derived from CD44high subpopulation were able to induce the activation of fibroblasts and stimulate their proliferation [115]. Promotion of metastasis by CD44+ EVs was confirmed in Suwakulsiri et al. [116], where the enrichment of CD44 in exosomes and shed microvesicles (MVs) was observed in SW620, a CRC metastatic cell line compared to SW480 CRC cell line. The observed enrichment of CD44 and other integrins (ANXA1, NOTCH1/2 and CLDN7), sustained the idea of exosome modulation of pre-metastatic niche formation [116].
In a paper by Gao et al. [117], CD44 expression on exosomes was correlated with the disruption of core 1-mediated O-glycosylation in LS174T and LSC cell lines. Considering the main function of exosomes in promoting the network between cancer cells and other cell types, the transfer of truncated CD44, and therefore the activation of tumor-related pathways, could identify a novel function of exosomes in CRC as a source for a potential biomarker of abnormal O-glycosylation [117].
On CD133+ EVs in CRC, even more promising results have been obtained [118], considering that in vitro experiments have demonstrated that one of the mechanisms by which CD133+ cells could sustain tumor development is through the upregulation of the autophagic process, probably to avoid apoptosis [119]. This lays the basis for a meticulous study of CD133+ EVs in CRC. It is known that the production of EVs and the autophagy process are strictly interconnected [120], and a modulation of the endosomal system could reflect an alteration in EV production that could be exploited when searching for new biomarkers.
Indeed, the increase of CD133+ EVs concentration in the bloodstream of CRC patients, compared with healthy controls, was associated with a shorter overall survival. Higher blood levels of CD133+ EVs CRC patients were also correlated with a weaker response to a first-line fluoropyrimidine-based therapy [121], which was in line with a previous study showing the use of CD133 siRNA to promote the sensitivity to Oxaliplatin while reducing stemness and migration properties in HT-29 cells [122].
Related to the study of CD133 in EVs, in several papers, the expression of CD133 in CRC was evaluated in MVs, whose size ranges from 50 nm to 1 µm in diameter.
Interestingly, the epidermal growth factor (EGF) was found to promote CD133 expression in CRC, which in turn regulates MVs release through RhoA and Rac1 regulation. Also, CD133+ MVs derived from KRAS mutant CRC cell line (HCT116) were shown to be able to carry the oncoprotein KRAS mutant, effectively mediating the development of chemoresistance [123, 124]. In another paper by Kim et al. [125], MVs containing CD133 were reported to play an important role in the crosstalk between tumor associated macrophages (TAMs) and CRC cells in TME. Specifically, CD133-containing MVs merged with M0 macrophages, supporting the acquisition of an M2-like TAM phenotype, and promoting IL-6 secretion [125].
Exosomes from CD133+/CD44+ CRC cells also seem to have a crucial role in the induction of stemness in recipient cells through long intergenic noncoding RNA 01315 (LINC01315) [126]. Long non-coding RNAs together with circRNAs have recently emerged as key regulators of CRC progression [127], although their possible use as biomarkers is still at infancy. Long non-coding RNAs (lncRNAs) and circular RNAs (circRNAs) carried by tumor-derived exosomes are emerging as stable, non-invasive biomarkers for cancer detection. Their resistance to degradation, due to exosomal encapsulation or circular structure and tissue-specific expression profiles, supports their diagnostic potential. However, the lack of standardized protocols and the difficulty in isolating tumor-specific exosomes limit clinical applicability. Large-scale studies and functional characterization are needed to fully exploit their biomarker and therapeutic potential. Nevertheless, these studies support a deeper investigation of exosome cargo in the context of the identification of new biomarkers, fueling the research of mechanisms that drive a selective enrichment in vesicles of specific molecules that could be informative for the pathology.
Gastric cancer (GC) is the second leading cause of cancer-related deaths globally. Despite treatment advancements, the prognosis for patients with advanced disease remains poor due to metastasis and recurrence. These factors significantly contribute to mortality, highlighting the need for efficient and accurate diagnostic methods for early metastasis detection [128]. CD44, is a generally recognized key marker for CSCs in GC, in multiple studies, it has been linked to worse prognosis and reduced survival rates in patients with primary adenocarcinoma of the stomach [129, 130, 131], although sometimes conflicting results have been obtained. Nosrati et al. [132] analyzed 95 samples and a correlation between CD44 expression and the intestinal subtype of the tumor, tumor size (4–8 cm), invasion depth, and moderate differentiation was identified, these findings were not confirmed in a retrospective study by Ahadi et al. [133] where no correlation between CD44 expression and the histology of the tumor was found. Probably, differences in the number of patients included in the studies as well as methodologies used for the detection, impacted the overall interpretation of the studies which implies the need for a deeper characterization of this marker in GC.
Along with the standard CD44 isoform, variants CD44v6 and CD44v9 have been linked to unfavorable prognosis. Interestingly, the latter variant has been proposed in combination with Ki-67 as a prognostic marker to detect early GC in a retrospective study by Go et al. [134].
The potential of CD44v9 as a biomarker of GC progression has also been described in association with the development of 5-fluorouracil resistance, driven, among other mechanisms, by its interaction with glutamate–cystine transporter xCT. In particular, the stabilization of xCT consequent to CD44v9 interaction drives the accumulation of GSH and glutathione peroxidase 2 (GPx2) induction and improves cell survival in stress conditions through the suppression of ROS [135, 136]. In a retrospective study by Jogo et al. [137], the expression of CD44v9 and GPx2 was evaluated in GC patients that received or not preoperative chemotherapy before surgery. In line with previous findings, CD44v9 was found to be associated with tumor progression and GPx2 induction regardless of preoperative treatment [135, 136]. The diagnostic and prognostic relevance of CD44v9 was recently further investigated in relation to T cell immunoglobulin and mucin domain‑containing protein 3 (TIM3) expression with the aim of improving the accuracy of CD44 as a biomarker.
Contradictory findings have been reported on the expression of CD133: in a recent paper, a reduction in mortality rate was observed in patients receiving neoadjuvant chemotherapy, suggesting a protective function of CD133 [138], but previous in vitro and in vivo findings reported its detrimental effect in the progression of the tumor [139]. The inconsistent results were probably due to different cohorts of patients examined, with neoadjuvant chemotherapy playing a role in the evaluation of CD133 expression in immunohistochemical analysis.
Notably, CD133 seems to be involved in drug resistance mechanisms, indeed CD133+ bone marrow-mesenchymal stem cells (BM-MSCs) have been shown to enhance chemoresistance by regulating apoptosis-related genes such as Bcl-2 and BAX [140, 141]. In this context, a study by Li et al. [142] evaluated oxaliplatin resistance in GC in relation to CD133 and PARP1, which is an important factor in maintaining the function of CD133+ CSCs. It was found that sequencing data from CD133+ and CD133- cells after tumourigenesis in human GC organoids showed higher PARP1 expression in CD133+ cells compared to CD133- cells. Overall, CD133+ CSCs mediated a strong DNA damage repair capacity through the enhanced stability of PARP1 mediated by N6-methyladenosine (m6A) Methyltransferase METTL3 [142]. The role of the binding protein of m6 heterogeneous nuclear ribonucleoprotein A2B1 (hnRNPA2B1) was explored in another paper where its presence was correlated with poor prognosis and limited response to chemotherapy in patients carrying high levels of CD44 and CD133 [143].
The study of CSC-EVs in GC has mainly focused on CD44+ vesicles that, interestingly, have been investigated also in the context of liquid biopsy. The expression of CD44 has been analyzed together with other potential markers (FASN and PTEN) in EVs isolated from the plasma of GC patients. Results obtained showed that CD44 expression is related to patients clinical treatment course, in particular, mRNA levels in EVs were found to decrease after neoadjuvant chemotherapy and gastrectomy, opening up to the possibility of using CD44 as a marker useful for patients stratification based on therapy response [144].
The role of exosomal CD44 was investigated as a marker of prognosis in GC, also in another paper, where its expression has been described to be responsible for the regulation of RhoA/YAP/Prox1/CPT1A signaling axis and promotion of lymph node metastasis (LNM) capacity [145]. The direct involvement of CD44+ EVs in the metastatic process has been further reported in a paper by Härkönen et al. [146] using CD44-negative human GC cell line, MKN74, manipulated to stably express CD44s. It was observed that the expression of CD44s does not significantly affect the overall secretion of EVs by tumor cells, yet CD44s is efficiently incorporated into EVs. Due to its strong binding affinity for HA, CD44 present on EVs may play a crucial role in modulating the interactions between EVs and the extracellular matrix (ECM) [146].
Despite a growing interest in EVs from CSCs in GC and their potential as biomarkers, to date, there is a paucity of studies investigating CD133+ EVs, which is disappointing given the relevance of CD133 in the pathology, as seen in other tumors, the potential of CSC-vesicles is paramount and should be exploited also in GC.
CD44 and CD133 are widely recognized markers of CSCs in several cancers. Stem cells are crucial players in tumor spreading and the initiation of pre-metastatic niche; in GI tumors, they have been implicated in the pathogenesis, supporting a deeper study of their specific functions in light of the identification of new targets. Despite promising results obtained in clinical trials on therapies targeting both CD44 and CD133, especially using CAR T cells, few advancements have been made, probably due to limited safety profiles and little efficacy. The optimization of strategies targeting CSCs together with the need for new biomarkers in the context of GI tumors is, in fact, an open challenge considering the limited prognostic values of markers that have been used in clinical settings. In this regard, liquid biopsy represents a powerful tool since it can help monitor the disease, stratify patients and determine the therapy of choice. Circulating exosomes isolated from liquid biopsy samples are an important source of molecules that can be informative of cells of origin and that can be used as a window to unravel mechanisms involved in tumor progression. The presence of CD44 and CD133 on EV surface has been broadly described and novel high-throughput technologies [147] could help in the application of this specific EV subpopulation as new biomarkers. Prior to EVs translation into clinics, many limitations, mostly related to the standardization of isolation methods need to be faced, indeed, based on the protocol used for EV purification and the EV source, several contaminants have been shown to impact downstream analyses, undermining their potential as well as data reproducibility [148].
Among several EV isolation methods, ultracentrifugation (UC) is the technique of choice in terms of purity, but the yield and the fact that it is a time-consuming technique sometimes undermine its use. To overcome these limitations, size exclusion chromatography (SEC) can be used since it showed great potential in terms of yield and EV integrity, also when samples are isolated from biological fluids. If the isolation of a specific EV population is required, immunoaffinity capture can be coupled to either UC or SEC even though the output and costs related to these techniques are often limiting. Other methods include ultrafiltration, precipitation (using commercially available kits) and microfluidic-based techniques, despite their use being restricted to rapid isolation procedures [149]. With the aim of establishing a rigorous isolation method for the therapeutic application of plasma-EVs, a recent paper compared nine different techniques, with the result of identifying immunoaffinity-capture and UC as optimal methods in terms of purity and yield, respectively [150]. This statement is in contrast with previous findings where precipitation was found to be the method of choice for high recovery [151, 152], further reinforcing the lack of consensus in the field.
In order to escalate the use of vesicles as a tool of liquid biopsy capable of giving a better representation of the tumor in terms of diagnosis or prognosis, a lot still needs to be done, especially when considering EV-based clinical trials. In the latest years, an increasing number of clinical trials have shown a growing interest in EV-focused studies [153, 154, 155] mostly for diagnostic purposes compared to therapeutics. Among others, an ongoing phase I trial on PC, tested mesenchymal stromal cells-derived exosomes with KrasG12D siRNA on metastatic patients harboring KrasG12D mutation, sustaining the use of engineered vesicles to improve delivery cargo [156]. To the best of our knowledge, no clinical trials involving CD44+ and/or CD133+ EVs have been established despite promising results obtained in preclinical models. In GC, it has been demonstrated that targeting of CD133+ and CD44+ gastric CSCs through CD44/CD133antibody-conjugated all-trans retinoic acid-loaded poly (lactide-co-glycolide)-lecithin-PEG nanoparticles is functional in selective delivery to CSCs and in improving the efficacy of the treatment [157]. This is an example of how new technologies represent a powerful resource to enhance therapeutic efficacy and lays the basis for the use of this approach in other types of tumors also using EVs.
The presence of CD44 and CD133 on EV surface has been broadly described and recently optimized antibody-based assays could help in the application of this specific EV subpopulation as new biomarkers. Also, the possibility of engineering EVs to express antibodies that can specifically bind to tumor cells or selectively deliver their cargo opens a new, promising scenario in the management of cancer and other diseases [158], although many challenges still need to be considered. In particular, their high immunogenicity limits the stability due to clearance by the immune system and strategies to improve this aspect are still under investigation [159].
In conclusion, CD44+ and CD133+ EVs hold promise as novel biomarkers in the context of GI tumors; evidence of the involvement of this specific subpopulation of vesicles in mechanisms driving tumor progression or development of therapy resistance has been shown in many papers reported in this review (Table 1, Ref. [79, 95, 96, 97, 98, 99, 113, 114, 115, 116, 119, 122, 123, 124, 145, 146]).
| Cholangiocarcinoma | Pancreatic cancer | Colorectal cancer | Gastric cancer | |
| Cell proliferation | [79] | [115] | ||
| Invasion | [95, 97, 98] | [113, 114, 115, 119, 122] | [146] | |
| Metastasis | [95, 96, 97, 98, 99] | [116] | [145] | |
| Chemoresistance | [97] | [122, 123, 124] |
While numerous findings have supported the fine regulation of CD44+ and CD133+ EVs in the establishment of a network with non-tumor cells and in the initiation of a pre-metastatic niche in pancreatic and CRC cancers, more research is needed for CCA and gastric cancers. Nevertheless, despite few studies on molecular tumor dynamics, the expression of exosomal CD44 has been considered informative of therapy response and diagnosis in GC and CCA patients, respectively. As discussed, investing resources in the standardization of EV isolation and detection could overcome several current limitations and push towards the reshaping of standards in liquid biopsy. Moreover, a multiomics approach directed towards exosome characterization could serve as a tool to implement the identification of signatures for patient stratification. Overall, the EV field is evolving rapidly with the aim to manage their heterogeneity and evaluating its biological relevance. Putting together the research that has been conducted up to now, it seems that the correlation between CSC-EVs and clinical outcomes should be further investigated to open up a new scenario in their applicability in liquid biopsy.
EV, extracellular vesicle; TME, tumor microenvironment; GI, gastrointestinal; CSC, cancer stem cell; CCA, cholangiocarcinoma; ILV, intraluminal vesicle; MVB, multivesicular body; ESCRT, endosomal sorting complex for transport; TDE, tumor derived exosomes; HCC, hepatocellular carcinoma; CD44s, standard isoform of CD44; CD44v, variant isoform of CD44; HA, hyaluronic acid; OPN, osteopontin; MMP, matrix metalloproteinase; MAPK, mitogen-activated protein kinases; PI3K, phosphoinositide 3-kinase; EMT, epithelial-mesenchymal transition; mAb, monoclonal antibody; CART, chimeric antigen receptor-modified T; NF-
CP conceptualization, writing-original draft, final manuscript, BM conceptualization, writing-original draft, final manuscript, MP figures and writing-original manuscript, ET conceptualization, writing-original manuscript, CB conceptualization, writing-original manuscript, AC conceptualization, writing-original draft, GP conceptualization, writing original draft, SP conceptualization, writing-original draft, final manuscript, AG conceptualization, writing-original draft, final manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
Bando Grandi Attrezzature 2024 (B13C25001000007) awarded to AG and SP by Cassa di Risparmio Firenze.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




