1 Department of Analytical Chemistry, Medical University of Bialystok, 15-222 Bialystok, Poland
2 Ruđer Bošković Institute, Laboratory for Oxidative Stress (LabOS), HR-10000 Zagreb, Croatia
3 Instrumental Analysis Open Access Centre, Faculty of Natural Sciences, Vytautas Magnus University, LT-44404 Kaunas, Lithuania
Abstract
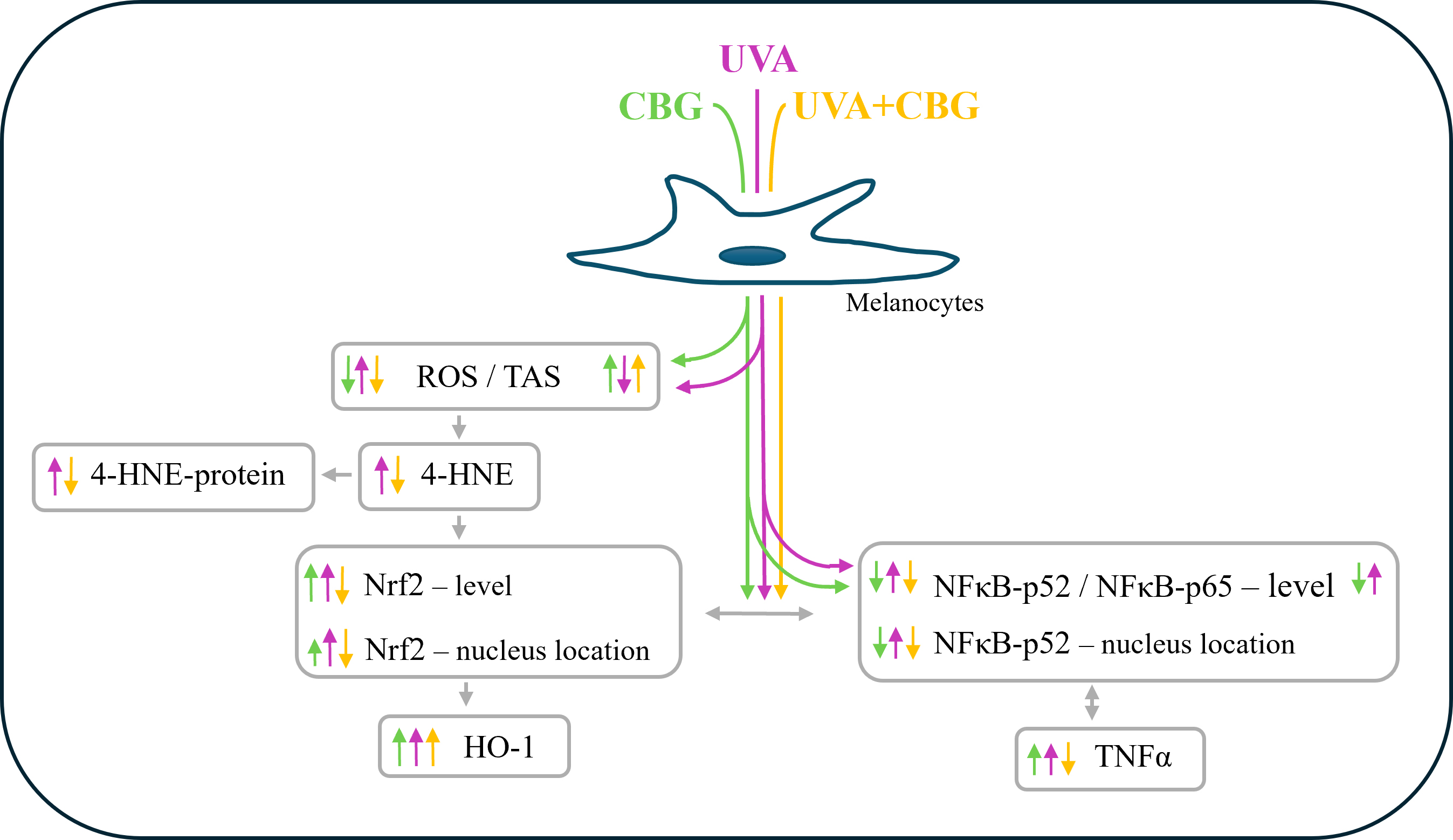
Ultraviolet A (UVA) radiation is a major environmental factor contributing to melanoma development. Melanocytes synthesize melanin, which provides partial protection against UVA-induced oxidative damage; however, these cells remain highly susceptible to oxidative and pro-inflammatory effects of UVA exposure.
In melanocytes, the following parameters were assessed: total antioxidant status (TAS-photometrically), reactive oxygen species (ROS-ESR), lipid peroxidation (4-HNE-GC-MS/MS), 4-HNE-protein adducts, and the expression/localization of key signaling proteins including phosphorylated nuclear factor erythroid 2-related factor 2 (pNrf2) and nuclear factor kappa B (NFκB) subunits [ELISA/fluorescence microscopy].
Cannabigerol (CBG) is a cytoprotective phytocannabinoid. In vitro studies showed that CBG attenuated UVA-induced oxidative stress in human melanocytes exposed to UVA radiation and significantly reduces lipid peroxidation, as measured by the levels of 4-hydroxynonenal (4-HNE) and its protein adducts. The biosynthesis of antioxidants was also regulated by CBG, even when administered post-irradiation. CBG attenuated the effects of UVA radiation by downregulating Nrf2, Kelch-like ECH-associated protein 1 (Keap1), BTB domain and CNC homolog 1 (Bach1), potent cyclin-dependent kinase inhibitor (p21), KRAB-associated protein 1 (KAP1), and multifunctional adaptor protein (p62). CBG also partially inhibited the pro-inflammatory NFκB signaling pathway by reducing the level of the activator (pIκB) and increasing the levels of the inhibitors (IKKα/β).
These results suggest that CBG may protect melanocytes from UVA-induced oxidative changes and lipid peroxidation by activating the Nrf2-dependent antioxidant system and inhibiting NFκB-based pro-inflammatory signaling. CBG can therefore create favorable conditions for the physiological functioning of melanocytes after UVA exposure, ultimately reducing the risk of inflammatory skin responses and neoplastic transformation.
Graphical Abstract
Keywords
- cannabigerol (CBG)
- melanocytes
- UVA
- inflammation
- protein modification
- 4-hydroxynonenal (4-HNE)
Due to its protective role over the entire body, human skin is constantly exposed to physical and chemical environmental factors. Physical factors that affect the integrity and proper functioning of the skin include ionizing radiation, such as cosmic and environmental radiation (e.g., radionuclides), as well as non-ionizing radiation, such as solar radiation [1]. Ionizing radiation is capable of directly damaging DNA [2]. Non-ionizing radiation mainly affects the skin by inducing oxidative stress, which leads to photoaging, disorders of the epidermal barrier, and consequently photocarcinogenesis [3]. The skin is constantly exposed to radiation, and its cumulative effects constitute a major risk factor for metabolic modifications of skin cells [1]. Moreover, the skin has a number of functions, such as receiving various stimuli, maintaining water balance, thermoregulation, and, to some extent, vitamin D biosynthesis [4]. To meet these challenges under harsh environmental conditions, skin cells, including melanin-producing melanocytes, protect other skin cells from the harmful effects of UV radiation from the sun [5] and from artificial sources [6].
While UVB radiation is absorbed primarily by epidermal keratinocytes, up to 50%
of UVA radiation penetrates the basal layer of the epidermis, directly affecting
melanocytes and stimulating melanin synthesis [7]. However, UVA exposure also
promotes the oxidation of endogenous photosensitizers, leading to excessive
production of reactive oxygen species (ROS) in melanocytes, often exceeding the
levels observed in keratinocytes and fibroblasts [8]. This overproduction of
pro-oxidant factors induces oxidative stress, leading to DNA damage as well as
activation of proinflammatory signaling pathways [9, 10]. Oxidative stress further
modifies lipid metabolism, with enhanced non-enzymatic ROS-dependent peroxidation
increasing the formation of reactive aldehydes such as 4-hydroxynonenal (4-HNE)
[11, 12]. In addition, enhanced enzymatic lipid metabolism leading to cyclization
products formation, including 15-deoxy-
There is currently great interest in bioactive substances, especially those of
natural origin, that do not have adverse effects and could prevent the
undesirable effects of UV radiation. These include antioxidants that reduce the
level of oxidative damage and protect the associated metabolic pathways [18].
Among such compounds are non-psychoactive phytocannabinoids derived from
Cannabis sativa L. These exhibit a range of therapeutic benefits for
various conditions, including those affecting the skin [19]. The most widely
studied phytocannabinoid is cannabidiol (CBD). CBD can protect melanocytes from
the effects of UVB radiation, even increasing their survival, due to its
antioxidant, anti-inflammatory, and cytoprotective properties [20]. The most
recent in vitro data and pilot clinical studies have reported a
reduction in oxidative stress-induced DNA damage and inflammation following
topical application of CBD [21, 22]. Moreover, CBD indirectly induces
melanogenesis via MAPK-based stimulation of MITF gene expression, which plays a
key role in the development of melanocytes [23]. Another increasingly studied
phytocannabinoid is cannabigerol (CBG). Similar to CBD, CBG easily penetrates
cell membranes and also exhibits antioxidant and anti-inflammatory properties.
This is evidenced by its direct bioactive properties, and indirectly through
interactions with G protein-coupled receptors (GPCRs) [24, 25]. Compared to CBD,
CBG acts more selectively on pathways related to inflammation, lipid metabolism,
and mitochondrial function [26]. When applied topically to the skin, CBG enhanced
the expression of key genes, including collagen (type I, III, and IV), elastin,
fibronectin, hyaluronan synthase 2, and IL‑10. In addition, CBG inhibited
pro-inflammatory cytokines (e.g., IL‑1
Despite the documented bioactive properties of CBG, its modulation of
UVA-induced oxidative stress-inflammation interactions in human melanocytes has
not been investigated. Therefore, in the present study, we examined the effect of
CBG on UVA-induced metabolic changes in human melanocytes, with a particular
emphasis on antioxidant and proinflammatory signaling pathways mediated by the
Nrf2 and NF
Standard human melanocytes isolated from the foreskin of a newborn and purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA; Cat. PCS-200-012) were used in this study. The cell line was validated by STR profiling and tested negative for mycoplasma. In accordance with ATCC guidelines, cells were grown in specific melanocyte medium (Dermal Cell Basal Medium [DCBM], ATCC) supplemented with Melanocyte Growth Kit (ATCC). Penicillin (50 U/mL) and streptomycin (50 µg/mL) were added to the medium to avoid bacterial contamination. During incubation, melanocyte cell cultures were kept at 37 °C in a humidified atmosphere containing 5% CO2.
Once the melanocytes (passage 3) reached 90% confluence, the cells were placed in plastic dishes surrounded by ice and 15 cm from UV 5 lamps, each with 8 W power. They were then exposed for 70 min to UVA radiation at 365 nm (Bio-Link Crosslinker BLX /365; Vilber Lourmat, Germany) and an intensity of 4.08 mW/cm2. The radiation dose was selected to achieve approximately 75% cell viability, as measured by MTT assay (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; Merck Millipore, Burlington, MA, USA) [33]. In parallel, melanocytes not exposed to UVA radiation (control group) were kept under the same conditions but in the dark and protected from any light, including UVA radiation.
To assess the effect of CBG (Sigma-Aldrich, St. Louis, MO, USA) on melanocytes
following exposure to UVA radiation, the cells were incubated for 24 h in medium
supplemented with a final concentration of 1 µM CBG, obtained after
dilution of a 32 mM stock solution dissolved in ethanol. Because the CBG solution
was prepared in ethanol, the respective control cultures were also treated with
the same final concentration of ethanol (0.3%). The specific concentration of
CBG was used because it does not alter cell morphology, proliferation, and
viability as assessed by the MTT assay and as describe previously [34]. IC50
(inhibitory concentration at 50%) of CBG was
The melanocyte cultures were divided into four experimental groups (each consisting of 5 independent replicates) according to the treatment applied:
I. Control: melanocytes cultured in standard medium only;
II. CBG: melanocytes cultured in medium containing CBG (1 µM) for 24 h;
III. UVA: melanocytes exposed to UVA radiation (18 J/cm2) and cultured in standard growth medium for 24 h;
IV. UVA+CBG: melanocytes exposed to UVA radiation (18 J/cm2) and cultured in medium containing CBG (1 µM) for 24 h.
At the end of the respective treatments, cells were washed with PBS, sonicated on ice using a Sonic Ruptor 400 (Omni International, Kennesaw, GA, USA), and centrifuged for 15 min at 12,000 g to obtain lysates for quantitative determination. All parameters were adjusted according to the protein content, as determined by Bradford assay [35].
Total antioxidant status (TAS) in melanocytes was determined using 2,2′-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS; Merck Millipore, Burlington, MA, USA) based on the method of Zhang et al. [36]. A microplate reader was used to measure absorbance at a wavelength of 734 nm. Results were calculated and presented as the percentage change in TAS in treated melanocytes compared to the control group.
The generation of total ROS in melanocytes was determined using an electron spin resonance (ESR) spectrometer (Noxygen GmbH/Bruker Biospin GmbH, Germany). This measures the oxidation products of cyclic hydroxylamine spin trap (CMH) by ROS in cell lysates, as described in a previous study [37]. Results were calculated and presented as the percentage change of ROS in melanocytes compared to the control group.
Melanocytes were cultured in 96-well culture plates for 24 h, rinsed with PBS,
and fixed with ice-cold methanol. Cell membranes were permeabilized with 0.1%
Triton X-100, and the samples were incubated for 1 h with a 1:10,000 dilution of
primary antibodies against phospho-Nrf2 (phospho-Ser40; Abcam, Cambridge, UK) or
phospho-NF
The expression levels of pNrf2 and NF
The levels of 4-hydroxy-2-nonenal (4-HNE) were determined by the GC-MS/MS method [39]. 4-HNE was converted into its O-PFB-oxime-TMS derivative via derivatization with O-(2,3,4,5,6-pentafluorobenzyl) hydroxyamine hydrochloride (0.05 mol/L) (Sigma-Aldrich, St. Louis, MO, USA). Samples were then extracted with hexane and, after evaporation under argon, suspended in N,O-bis(trimethylsilyl)trifluoroacetamide (Sigma-Aldrich, St. Louis, MO, USA). The samples were subsequently analyzed using a 7890A GC–7000 quadruple MS/MS (Agilent Technologies, Palo Alto, CA, USA) in a selected ion monitoring (SIM) mode: m/z; 352.0, 271.2 (4-HHE-PFB-TMS) and m/z; 307.0 (internal standard derivatives). Results were presented as percentage changes of 4-HNE in comparison to the melanocyte control group.
The levels of 4-HNE-protein adducts in melanocytes were determined using an ELISA method [38]. Briefly, samples containing cell lysates were incubated overnight with a mouse monoclonal antibody against 4-HNE-His (Invitrogen, Burlington, ON, Canada). The primary antibody was then removed, and the samples were washed with PBS containing 0.1% Tween 20. After incubating with secondary antibody (goat anti-mouse antibody EnVision+ Dual Link/HRP; Agilent Technologies, Santa Clara, CA, USA), the samples were subsequently incubated with chromogen substrate solution (TMB; 0.1 mg/mL). Absorbance was then recorded at 450 nm, with 4-HNE-protein adduct levels expressed as a percentage relative to untreated control cells.
Statistical comparisons were performed using one-way analysis of variance
(ANOVA), followed by Tukey’s multiple comparisons test (GraphPad Prism 9, Dotmatics, Boston, MA, USA). Data
are presented as the mean value with standard deviation (SD) for N = 5.
Differences were considered statistically significant at p
Incubation of melanocytes with CBG for 24 h altered their endogenous antioxidant capacity. The total antioxidant potential (TAS) increased by approximately 30%, while ROS levels decreased (Fig. 1). Exposure of melanocytes to UVA radiation reduced TAS by approximately 50% compared to the control group. In contrast, treatment with CBG after UVA irradiation increased TAS, restoring it to the level of the control group.
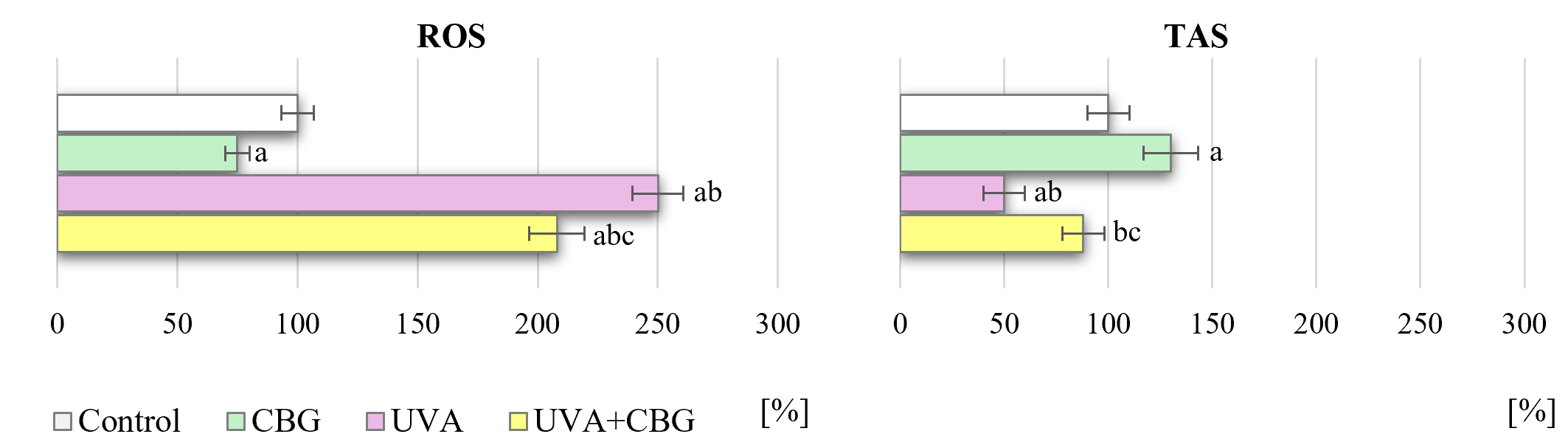 Fig. 1.
Fig. 1.
Effect of CBG (1 µM) on the level of reactive
oxygen species (ROS) and total antioxidant status (TAS) in melanocytes (presented
as a percentage of control cells). The four experimental groups were: controls;
treatment with CBG for 24 h; exposure to UVA (18 J/cm2) followed by culture
for 24 h; and exposure to UVA (18 J/cm2) followed by treatment with CBG for
24 h. Values represent the mean
The changes in melanocyte TAS may be related to the expression (Fig. 2) and cytosolic-nuclear localization (Fig. 3 and Supplementary material) of the transcription factor Nrf2, which is responsible for the biosynthesis of antioxidant proteins. CBG was found to increase the level of pNrf2 and the expression of heme oxygenase (HO-1), which is an indicator of Nrf2 transcriptional efficiency (Fig. 2). Following cell exposure to UVA radiation, the levels of pNrf2 and HO-1 increased significantly by approximately 120% and 100%, respectively. Furthermore, microscopy results showed that exposure of melanocytes to UVA radiation induced strong increases in Nrf2 expression in both the nucleus and cytoplasm. However, treatment with CBG following UVA irradiation caused a significant decrease in pNrf2 levels, accompanied by an increase in HO-1 levels. CBG also modified the intracellular localization of Nrf2, partially preventing UVA-induced Nrf2 translocation to the nucleus (Fig. 3).
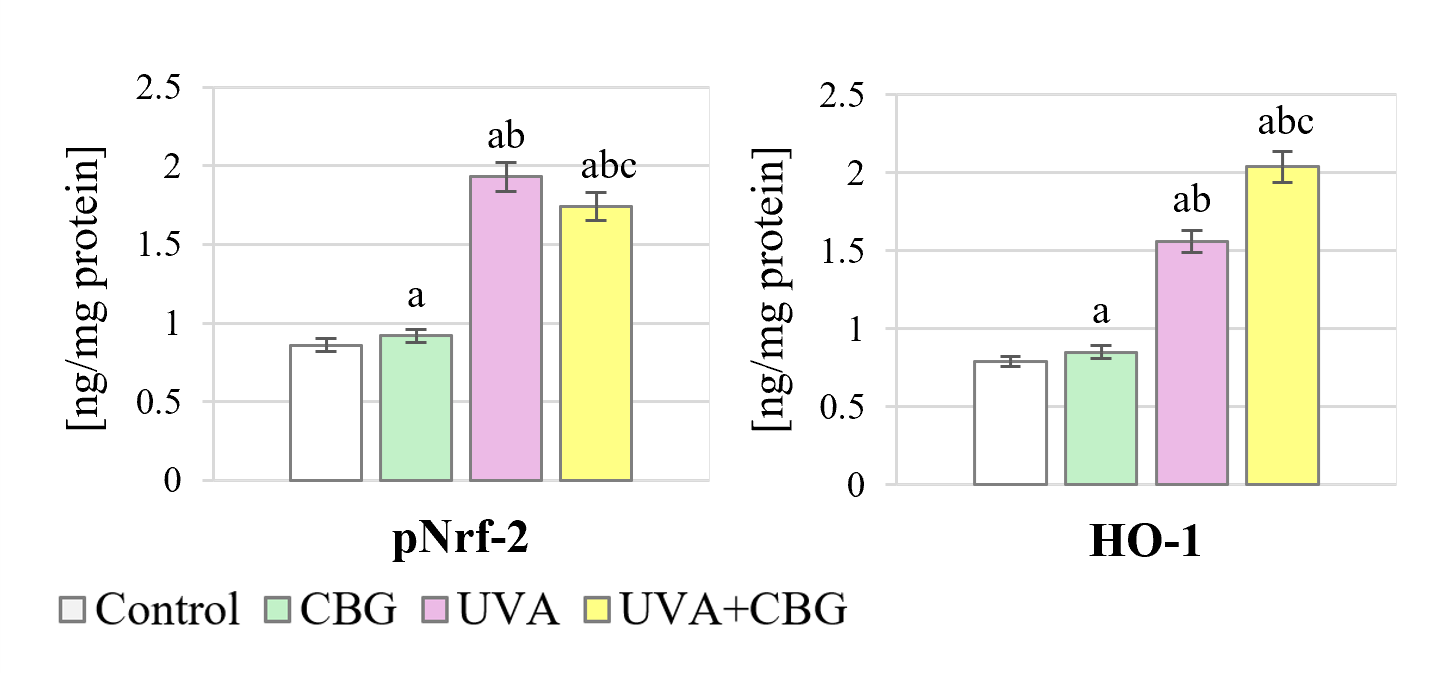 Fig. 2.
Fig. 2.
Effect of CBG (1 µM) on the level of phospho-Nrf2
(Ser40) (pNrf2) and heme oxygenase (HO-1) in melanocytes. The four experimental
groups were: controls; treatment with CBG for 24 h; exposure to UVA (18
J/cm2) followed by culture for 24 h; and exposure to UVA (18 J/cm2)
followed by treatment with CBG for 24 h. Values represent the mean
 Fig. 3.
Fig. 3.
Effect of CBG (1 µM) on phospho-Nrf2 (Ser40) (pNrf2) intracellular distribution in melanocytes. The four experimental groups were: controls; treatment with CBG for 24 h; exposure to UVA (18 J/cm2) followed by culture for 24 h; and exposure to UVA (18 J/cm2) followed by treatment with CBG for 24 h. Localization of Nrf2 is shown in green. Scale bar = 50 µm.
The levels of pNrf2 activators in melanocytes exposed to UVA and CBG are presented in Fig. 4. CBG was found to cause significant changes in the levels of activators (except p62), corresponding to the increased pNrf2 level (Fig. 2). Significant increases in activator levels were observed after UVA irradiation of cells, particularly for pMAPK (approximately 550%) and pERK (approximately 210%). However, addition of CBG to the medium after UVA exposure caused a decrease in the levels of KAP1 and pERK proteins, accompanied by a slight increase in p62 level.
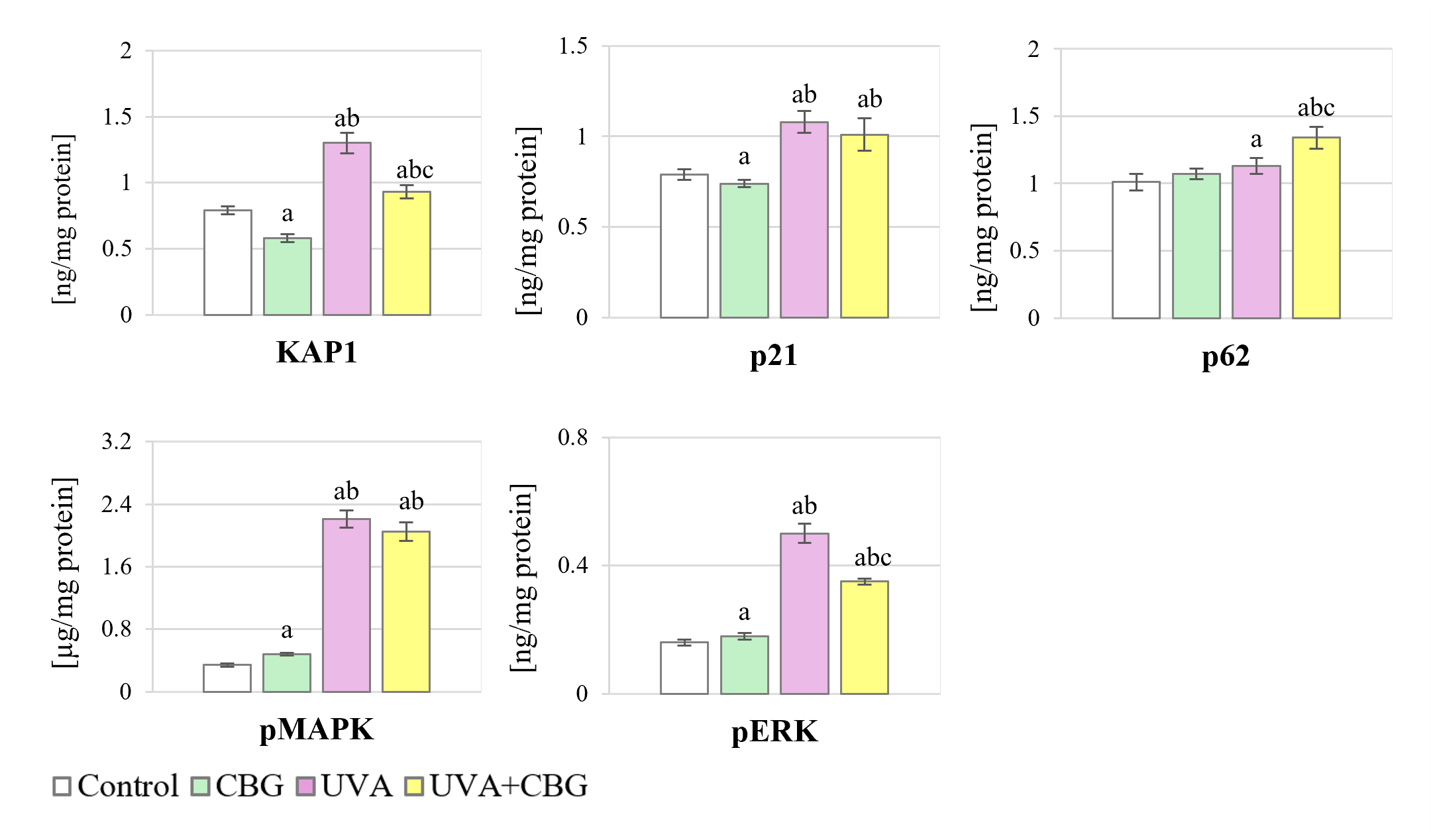 Fig. 4.
Fig. 4.
Effect of CBG (1 µM) on the level of Nrf2
activators in melanocytes. The four experimental groups were: controls;
treatment with CBG for 24 h; exposure to UVA (18 J/cm2) followed by culture
for 24 h; and exposure to UVA (18 J/cm2) followed by treatment with CBG for
24 h. Values represent the mean
CBG treatment also caused changes in the levels of pNrf2 inhibitors, including increases in Keap1 and PGAM5, and a decrease in the nuclear inhibitor Bach1 (Fig. 5 and Supplementary material). The response of these inhibitors to UVA radiation was also variable, with a decrease in Keap1 and an increase in Bach1 and PGAM5 levels. However, the addition of CBG to cells previously exposed to UVA radiation modified the response of Nrf2 inhibitors. Adding CBG to control cells increased Keap1 and PGAM5 levels and decreased Bach1 levels. Conversely, adding CBG to UVA-exposed melanocytes reversed the decrease in Keap1 level, and reversed the increase in Bach1 and PGAM5 levels. However, following CBG treatment, the levels of all Nrf2 inhibitors were different from those of the control group, with Keap1 and Bach1 being lower, and PGAM5 being higher.
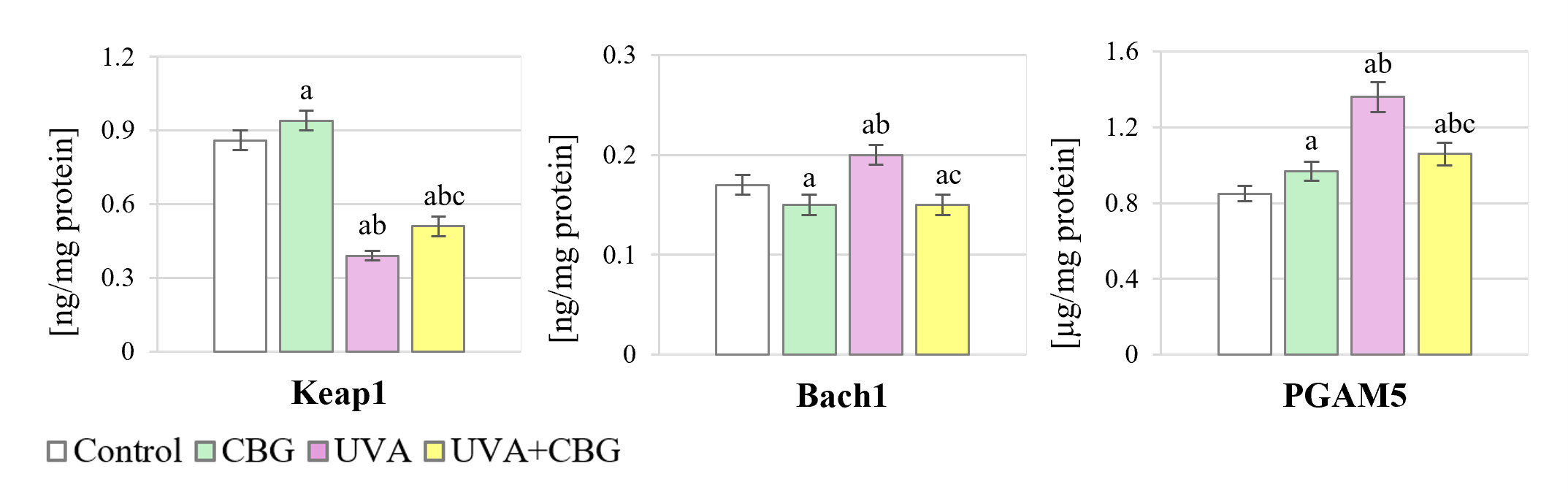 Fig. 5.
Fig. 5.
Effect of CBG (1 µM) on the level of Nrf2
inhibitors in melanocytes. The four experimental groups were: controls;
treatment with CBG for 24 h; exposure to UVA (18 J/cm2) followed by culture
for 24 h; and exposure to UVA (18 J/cm2) followed by treatment with CBG for
24 h. Values represent the mean
Changes in pNrf2 levels are also influenced by the WTX and DDP3 proteins, which are activators of Keap1 (Fig. 6 and Supplementary material). CBG alone caused significant decreases in the levels of WTX and DPP3. In contrast, UVA radiation increased WTX by 60% and DPP3 by 270%. Moreover, CBG applied after UVA reversed the UVA-induced increase of both activators.
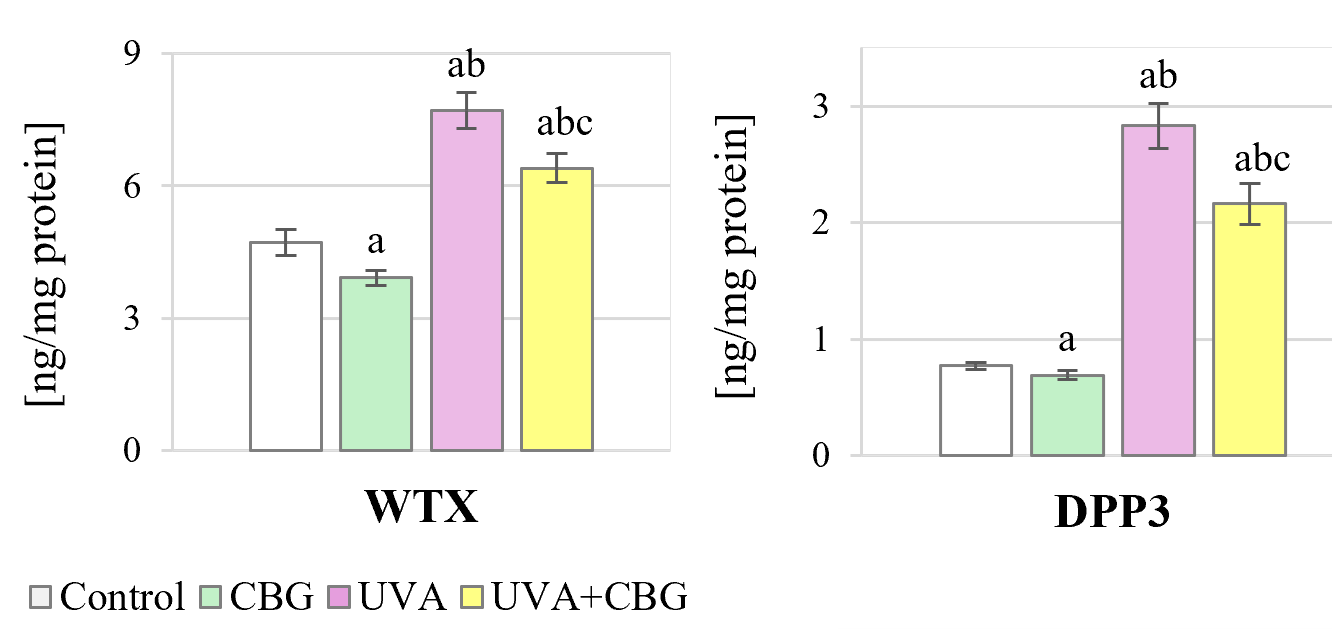 Fig. 6.
Fig. 6.
Effect of CBG (1 µM) on the level of Keap1
activators in melanocytes. The four experimental groups were: controls;
treatment with CBG for 24 h; exposure to UVA (18 J/cm2) followed by culture
for 24 h; and exposure to UVA (18 J/cm2) followed by treatment with CBG for
24 h. Values represent the mean
The shift in redox balance towards oxidation following UVA irradiation of melanocytes was reduced by the application of CBG (Fig. 1). This was reflected by the increase in ROS-dependent lipid peroxidation, which consequently manifested as increased levels of both 4-HNE and its protein adducts (approximately 117%) (Fig. 7). These UVA effects were attenuated by treatment with CBG, which caused a significant reduction in lipid peroxidation. In melanocytes treated with CBG after UVA irradiation, the 4-HNE level decreased by approximately 40%, and 4-HNE-adducts with protein formation by approximately 24%.
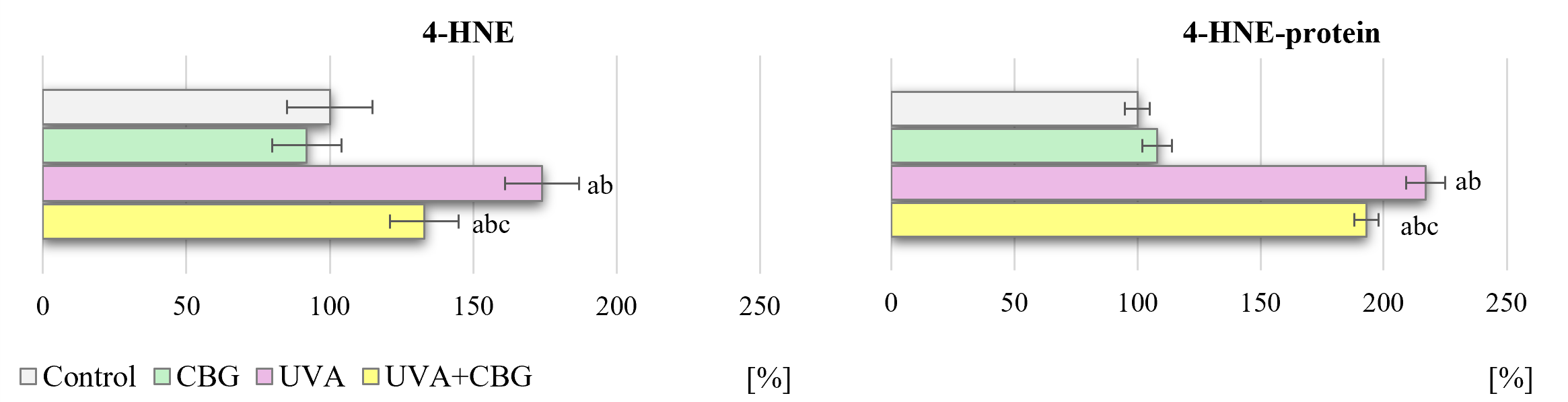 Fig. 7.
Fig. 7.
Effect of CBG (1 µM) on the level of
4-hydroxynonenal (4-HNE) and 4-HNE-protein adducts in melanocytes (presented as a
percentage of control cells). The four experimental groups were: controls;
treatment with CBG for 24 h; exposure to UVA (18 J/cm2) followed by culture
for 24 h; and exposure to UVA (18 J/cm2) followed by treatment with CBG for
24 h. Values represent the mean
The antioxidant transcription factor Nrf2 is known to interact with the
proinflammatory transcription factor NF
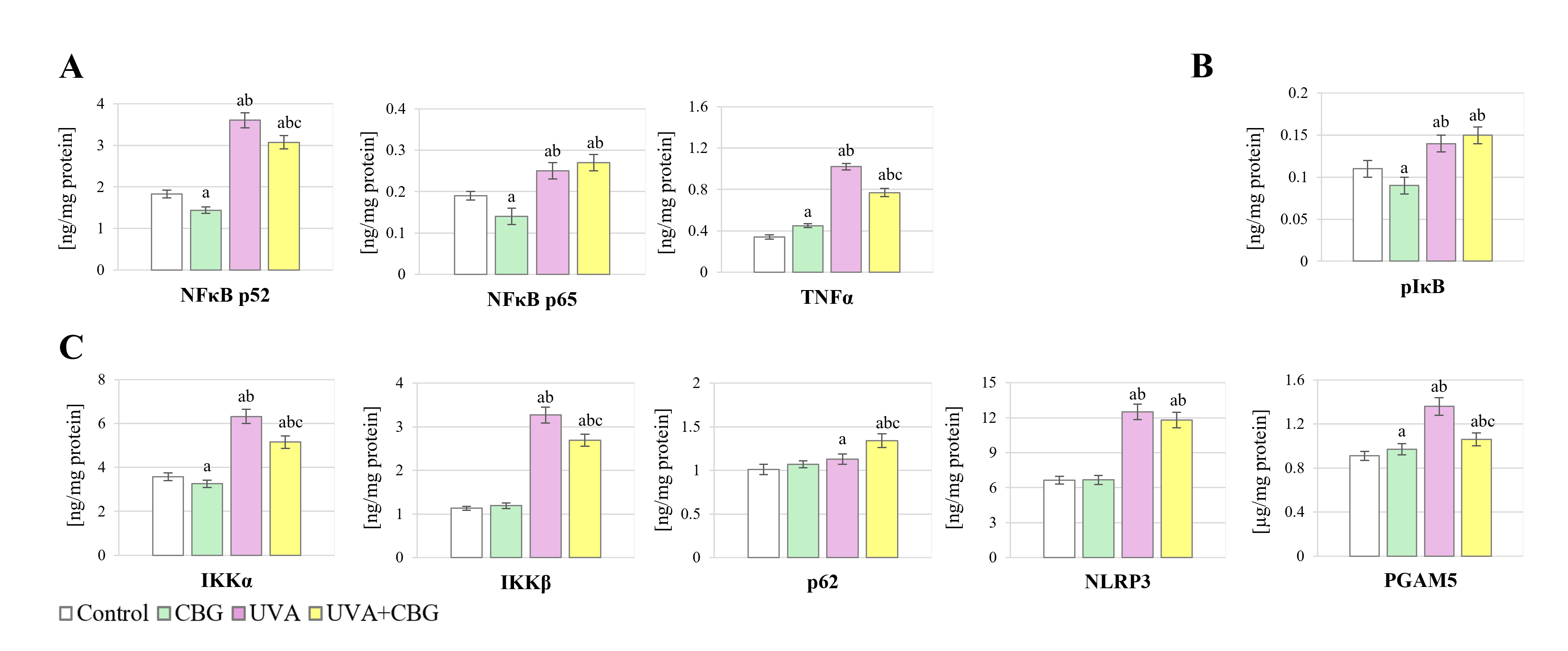 Fig. 8.
Fig. 8.
Effect of CBG (1 µM) on the level of (A)
NF
 Fig. 9.
Fig. 9.
Effect of CBG (1 µM) on phosphorylated form of
subunit p52 NF
The exposure of melanocytes to UVA radiation led to increased expression of both
NF
Changes in the levels of the NF
Epidemiological data indicate that UVA radiation induces oxidative stress in skin cells, including melanocytes, thereby altering the structure and function of macromolecules. This causes excessive cell proliferation and/or mutations [40] that subsequently exert cytotoxic, proinflammatory, and carcinogenic effects [41], leading to the development of melanoma, a dangerous skin cancer type [42, 43]. Hence, there is a need to protect skin from UVA radiation, preferably through the use of natural compounds such as phytocannabinoids that have a low probability of generating harmful effects. CBD and CBG have been shown to exhibit antioxidant and anti-inflammatory effects on keratinocytes and dermal fibroblasts [25, 44]. Furthermore, CBG has greater metabolic efficiency than CBD [28]. The safety of long-term topical CBG use has already been demonstrated [28], suggesting it may be a promising bioactive ingredient in sunscreens and skin care products. Therefore, this study investigated the effect of CBG on melanocyte metabolism.
Melanocytes are located in the basal layer of the epidermis and produce
endogenous chromophores under the influence of UVA radiation, in particular the
cytoprotective agent melanin [17, 42]. Melanin synthesis is regulated by numerous
modulators, including cytokines such as TNF
Previous studies have shown that sunlight (containing a UVA component) not only increases melanogenesis, but due to its reaction with dopaquinone, a pigment precursor [49], also decreases the antioxidant capacity [50, 51]. It does this by reducing the levels of cysteine and consequently glutathione (GSH), which may influence the effectiveness of other cellular antioxidants. In the current study, CBG demonstrated cytoprotective effects by reducing ROS levels in both control and UVA-irradiated melanocytes, while simultaneously increasing the total antioxidant status (TAS), which is known to promote cell viability [34]. The metabolic bioactivity of CBG is believed to result from its terpene structure. CBG is estimated to have approximately twice the ROS-reducing capacity of CBD, making it a common ingredient in skin preparations [52].
The synthesis of a wide range of antioxidant proteins is regulated by the activity of the transcription factor Nrf2. The level and efficiency of Nrf2 is in turn regulated by cytoplasmic and nuclear activators and inhibitors [25]. The results of the current study showed relatively low levels of pNrf2 in the cytoplasm of control melanocytes. This indicates that it may form adducts with the cytosolic inhibitor Keap1, causing Nrf2 to be directed towards proteasomal degradation [53]. Similar to CBD, CBG can also modify Cys15 of Keap1, which is involved in the formation of the Cul3–Keap1 complex necessary for interaction of this inhibitor with Nrf2 [54]. However, under the oxidative stress conditions of UVA-exposed melanocytes, Nrf2 dissociates from Keap1 and translocates to the cell nucleus, where it heterodimerizes with one of the small Maf proteins. Heterodimers recognize ARE sequences present in the regulatory regions of Nrf2 target genes, which are essential for the recruitment of key factors [55]. Nuclear Nrf2 induces the expression of a broad spectrum of genes involved in cellular antioxidant defense, detoxification, and redox homeostasis. In addition to the canonical redox-dependent mechanism, Nrf2 can also be activated via non-canonical pathways. In non-canonical pathways, proteins such as p62, p21, DPP3, and WTX interact directly with Keap1, impair Keap1-mediated ubiquitination of Nrf2, and promote its stabilization, nuclear accumulation, and transcriptional activity by disrupting the Nrf2–Keap1 complex [56].
A previous finding relevant to the present results is that UVA irradiation of melanocytes may increase pNrf2 levels and induce HO-1 expression, thereby inhibiting Bach1 degradation [57]. Furthermore, Bach1-mediated repression of Maf-regulated genes has been shown to dominate over Nrf2-mediated activation of these genes [58]. These results suggest that Nrf2 activation is able to offset the increase in Bach1 levels and activate the transcription of antioxidant genes, even in the presence of high Bach1 levels. Under reduced Keap1 levels following UVA exposure, Bach1 protein stabilization might be induced by Nrf2-dependent upregulation of HO-1, promoting a decrease in free heme levels.
CBG increased the level of free Keap1 in control and UVA-irradiated melanocytes. This may indicate less involvement of the cytosolic Nrf2 inhibitor in the formation of Nrf2–Keap1 interactions, as the biological activity of Keap1 depends on the availability of functional cysteine thiol groups. However, these are highly susceptible to modification by ROS and electrophilic aldehydes, including 4-HNE generated during lipid peroxidation [59]. Increased levels of 4-HNE and its protein adducts in the cytosol were observed after UVA irradiation of melanocytes, but were reduced after CBG treatment, thereby maintaining the normal phospholipid structure of the cell membrane [34]. However, even after treatment with CBG, the level of 4-HNE-protein adducts did not reach the control values. This may indicate the persistence of oxidative stress after CBG treatment, which is also supported by the increased level of ROS and 4-HNE. Modification of Keap1 by 4-HNE is known to increase the level of free Nrf2, thus enhancing its translocation to the cell nucleus, as confirmed by the microscopy results of our study. However, the levels of PGAM5, another cytosolic Nrf2 inhibitor [60], and Bach1, a nuclear Nrf2 inhibitor [61, 62], were also observed to increase after UVA irradiation of melanocytes. In control melanocyte cultures and those treated with CBG, Bach1 was present primarily in the cytoplasm. In contrast, Bach1 shifted to the nucleus following UVA irradiation, where it is more effective at inhibiting Nrf2 activity. Furthermore, CBG treatment alone increased the expression of PGAM5 phosphatase, which can interact with both Keap1 and Nrf2 [63], thereby confirming the regulatory role of this phytocannabinoid. Due to the reduced expression and likely structural modifications of Keap1 in UVA-irradiated and CBG-treated melanocytes, Nrf2 can more easily relocate to the nucleus compared to control cells. Thus, by increasing ROS generation, UVA irradiation simultaneously increases the expression of Nrf2 in the cytosol, inducing the transcription of cytoprotective proteins essential for maintaining redox balance under conditions of elevated ROS [59]. The level of the primary target of this transcriptional activity, HO-1 [64], was shown to increase after UVA exposure, and even more after CBG treatment.
Regardless of the Nrf2-Keap1 interaction, metabolic links are known to exist between the Nrf2-activating protein p62 and the Keap-1 inhibitor. These links connect the Nrf2 and autophagy signaling pathways [65], which are essential for maintaining cellular antioxidant homeostasis [66] and increasing the biosynthesis of cytoprotective proteins [67]. Autophagy also acts as a cell survival mechanism under various physicochemical conditions, allowing p62 to form a positive feedback loop with Nrf2 [68], thus inhibiting p62 accumulation. UVA irradiation of melanocytes and their exposure to CBG significantly increased the level of p62, which may be important because autophagy plays a role in regulating melanin production and skin pigmentation [69]. Autophagy is also involved in melanin degradation via the PI3K/AKT/mTOR pathway [70]. Moreover, increased autophagy in melanocytes provides more effective protection against the accumulation of oxidatively modified macromolecules. Autophagy disorders can therefore increase the sensitivity of melanocytes to oxidative stress [71].
It is well established that overproduction of ROS leads to oxidative stress,
which plays a major role in inflammatory processes [16]. At the same time, the
NF
CBD is structurally similar to CBG and can also modify the structure of Keap1
[77]. It can therefore be assumed that CBG influences the regulation of both Nrf2
and NF
CBG confers beneficial metabolic effects on melanocytes, especially those exposed to UVA radiation. However, it is important to note the limitations of this study. The experiments were conducted exclusively in vitro and using only monocultures of melanocytes, without considering possible interactions with other skin cells, such as fibroblasts. Furthermore, only a single dose of CBG and UVA radiation was used in the experiments, which especially in the case of UVA, does not reflect real-world conditions. Nevertheless, our study represents the first attempt to assess the protective effects of CBG on melanocytes, which may constitute a barrier to the neoplastic transformation of these cells. Further studies are planned to analyze the metabolic consequences of increased doses of CBG and the length of exposure to melanocytes. Furthermore, studies using co-cultures of fibroblasts and melanocytes are planned. Although there is no clear evidence of any harmful effects of CBG, an in vivo analysis of its effect on the metabolism of skin cells, including melanocytes, in experimental animals should also be conducted.
Furthermore, the results of this study only describe the mechanism of action of CBG, without comparing its effects with well-described and commonly used cytoprotective compounds such as ascorbic acid or CBD. These are characterized by both similar and diverse physicochemical properties, and consequently diverse biological effects that could significantly increase the interpretability of our results. Further analysis of the metabolic potential of CBG should include comparative studies of this phytocannabinoid with other protective compounds that have similar and diverse physicochemical and biological properties. Such research could then lead to the formulation of more rational conclusions.
CBG has regulatory effects on the metabolism of melanocytes, especially those exposed to UVA radiation. This phytocannabinoid generates favorable conditions for the physiological or near-physiological functioning of melanocytes, even after exposure to harmful environmental conditions such as UVA radiation. By increasing the antioxidant capacity of melanocytes and reducing inflammatory processes, CBG may reduce the risk of neoplastic transformation of these cells.
Skin exposure to solar radiation is not only a risk factor for premature skin
aging, but also a trigger for metabolic changes leading to the development of
skin cancer. Our results suggest that CBG protects melanocytes against
UVA-induced oxidative changes by activating the Nrf2-dependent antioxidant
system, and/or inhibiting pro-inflammatory signaling based on NF
The datasets obtained during this study are available from the corresponding author upon reasonable request.
IJ-K and ES designed the study. NZ and AM conducted the formal analysis. IJ-K and AG developed the methodology, conducted the research, and were responsible for data collection. ES managed the project administration and provided funding and supervision for the study. IJ-K, AG and AM prepared the visualizations and wrote the original manuscript, while NZ and ES analyzed and revised the final version. All authors contributed to editorial changes to the manuscript after peer review. All authors read and approved the final version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
The article has been supported by the Polish National Agency for Academic Exchange under the “Strategic Partnerships” programme (project no. BPI/PST/2024/1/00008).
The authors declare no conflict of interest. Given his role as the Guest Editor and Editorial Board member, Neven Žarković had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Alexandros G. Georgakilas.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL47793.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









