1 Department of Cardiology, Zhejiang Province Rongjun Hospital, 314000 Jiaxing, Zhejiang, China
2 Department of Cardiology, Affiliated Central Hospital of Dalian University of Technology, 116033 Dalian, Liaoning, China
3 Department of Heart Intensive Care Unit, The First Affiliated Hospital of Dalian Medical University, 116011 Dalian, Liaoning, China
Abstract
Hyperlipidemia is highly prevalent worldwide and can affect cardiac pathophysiology. This study aimed to compare the effects of high-intensity interval training (HIIT) and moderate-intensity continuous training (MICT) on the molecular mechanisms of myocardial stress and pathological remodeling in non-obese apolipoprotein E knockout (ApoE⁻/⁻) mice with hypercholesterolemia.
Thirty-five 8-week-old male ApoE⁻/⁻ mice were randomly assigned to four groups as follows: control (normal diet); HFD (high-fat diet); HFD+MICT (60% maximal running speed); and HFD+HIIT (85% maximal running speed). After a 12-week intervention, serum levels of blood lipids and B-type natriuretic peptide (BNP) as well as pathological changes in the myocardial tissue (hematoxylin and eosin staining and Masson’s trichrome staining) were detected. Protein expression analyses of lipid metabolism markers (CD36, CD68 (Cluster of Differentiation 36/68), lectin-type oxidized low-density lipoprotein receptor 1 and peroxisome proliferator-activated receptor-gamma), antioxidant regulators (sirtuin 1/3 [SIRT1/3], nuclear factor erythroid 2-related factor 2 [NRF2], and superoxide dismutase 2 [SOD2]), inflammatory cytokines (interleukin [IL]-6 and IL-18), and fibrosis-related proteins (transforming growth factor-beta 1 [TGF-β1], collagen I/III) was performed using immunohistochemistry and western blotting.
The HFD condition increased serum total cholesterol (TC) and triglyceride (TG) levels, but did not increase body weight, consistent with a lean hyperlipidemia model. Compared with the MICT condition, the HIIT condition demonstrated superior efficacy in reducing HFD-induced TC, TG and BNP levels (p < 0.05). Histologically, HIIT reduced myocardial fibrosis and inflammation. HIIT downregulated lipid transporters CD36/CD68, upregulate the antioxidant SIRT1/3-NRF2-SOD2 axis, inhibit pro-inflammatory factors IL-1β, IL-6, and IL-18, and reduce the deposition of fibrotic TGF-β1 and collagen I and III (p < 0.05).
In a non-obese, hypercholesterolemic ApoE⁻/⁻ model, HIIT elicited more favorable molecular signatures than MICT for ameliorating myocardial stress and pathological remodeling in terms of lipid deposition, oxidative stress, inflammation and fibrosis pathways.
Keywords
- hyperlipidemia
- ApoE⁻/⁻ mice
- myocardial stress
- high-intensity interval training
- moderate-intensity continuous training
- lipid metabolism
- oxidative stress
Hyperlipidemia is characterized by abnormally high levels of lipids or
lipoproteins in the blood due to abnormal fat metabolism or function, caused by
obesity and hereditary disorders such as familial hypercholesterolemia (FH) or
metabolic syndrome [1, 2]. Patients with hyperlipidemia are approximately twice as
likely to develop cardiovascular disease (CVD) than healthy individuals [3].
Studies have demonstrated that hyperlipidemia can lead to vascular
atherosclerosis, and cardiac fat accumulation and affect cardiac function and
electrophysiological activity [3, 4, 5]. Elevated total cholesterol (TC) and free
fatty acids levels trigger systemic oxidative stress and proinflammatory milieu
[6, 7]. Cardiomyocytes reportedly take up oxidized low-density lipoprotein (LDL)
through CD36 and lectin-type oxidized low-density lipoprotein receptor 1 (LOX-1),
further activating the nuclear factor-kappa B (NF-
Exercise training is a critical non-pharmacological intervention for metabolic
and CVD. Moderate-intensity continuous training (MICT) improves insulin
sensitivity, while high-intensity interval training (HIIT) preferentially
enhances mitochondrial biogenesis and oxidative capacity. Structured aerobic
exercise reduces the incidence of metabolic and CVD by 30–40% [13]. Exercise
exerts cardioprotective effects via antiapoptotic, antifibrotic, and antioxidant
mechanisms, mediated by SIRT1/3-NRF2 regulation and mitochondrial quality control
[9, 13, 14, 15, 16]. Exercise reportedly modulates inflammation and lipid metabolism
through intensity-dependent regulation of CD36-mediated fatty acid uptake and
peroxisome proliferator-activated receptor-gamma (PPAR-
This study compared the molecular adaptations between HIIT and MICT in a hypercholesterolemic apolipoprotein E knockout (ApoE⁻/⁻) model by focusing on lipid transport, redox homeostasis, and inflammatory-fibrotic signaling.
Thirty-five 8-week-old male ApoE⁻/⁻ mice were procured from the Beijing
Weitong Lihua Experimental Animal Company Beijing Biotechnology Co., Ltd.
(Beijing, China) and randomly allocated into four groups. The groups were defined
as follows: Control (n = 7), which received a standard diet (10% calories from
fat); HFD (n = 9), which received a high-fat diet (35% fat calories, 1.5%
cholesterol); HFD+MICT (n = 10), which received a high-fat diet along with
moderate-intensity continuous exercise (60% maximal speed); HFD+HIIT (n = 9),
which received a high-fat diet along with high-intensity intermittent exercise (9
To determine the maximum running speed, ApoE⁻/⁻ mice in the exercise group underwent a running test on an XR-PT-10B treadmill (Shanghai XinRuan Information Technology Co., Ltd., Shanghai, China). The test commenced at a speed of 10 m/min with an incline of 0° for 20 min, and the speed was gradually increased by 4 m/min until exhaustion was reached. The mice were considered exhausted if they remained stationary on the grid for 3 s or exhibited no movement in response to gentle nudges with a soft brush. The maximum velocity achieved during exercise was defined as maximum running speed (Vmax). All exercise groups underwent a 5-minute warm-up at 40% Vmax before formal training. For the HFD+HIIT group, the exercise regimen involved nine sets of higher-intensity treadmill exercises at 85% Vmax, each lasting 1.5 min, with a 1-min recovery interval at 40% Vmax between each sprint set, totaling 21.5 min per session. No adverse events (injury or death) occurred during the training, confirming the safety of the intensity protocol for this genotype. Conversely, the HFD+MICT group participated in continuous endurance training, maintaining a speed equivalent to 60% Vmax until their running distance matched that of the HFD+HIIT group. This design ensured that any observed differences were attributable to the intensity modality rather than the total work volume. Following formal training, both groups underwent a 5-min recovery period at 40% Vmax. This exercise routine was repeated five times per week for 12 weeks.
Serum lipid (TC and triglyceride [TG]) and B-type natriuretic peptide (BNP) levels were quantified using a commercial assay kit, following the protocols outlined by the manufacturer (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
The procured hearts were fixed in 10% neutral-buffered formalin for 24 h, then
dehydrated in graded ethanol (75%, 85%, 90%, and 100%, each for 5 min each),
and embedded in paraffin. Sections (4 µm) were cut and mounted on glass
slides coated with poly-L-lysine. To prepare the sections for analysis,
deparaffinization was achieved by immersion in xylene, with three changes of
xylene (5 min each). Rehydration was then performed using reduced concentrations
of alcohol (100 to 75% for 5 min each). Hematoxylin and eosin (HE) staining was
performed according to standard protocols. Masson’s trichrome staining was used
to visualize the collagen deposition (blue) and myocardial architecture (red) to
assess interstitial fibrosis. Imaging was conducted using an upright light
microscope (Murzider, Beijing, China) at 200
Heart tissue sections were fixed in 10% formalin, dehydrated using an ethanol
series, and embedded in paraffin for a histological evaluation. After
deparaffinization and antigen retrieval (citrate buffer, pH 6.0, 95 °C,
20 min), the sections were blocked with 3% H2O2 (15 min) and 5%
bovine serum albumin (BSA), then incubated overnight with primary antibodies at 4
°C (all from Proteintech, Wuhan, China). The dilutions were as follows:
rabbit anti-CD36 (1:600, Cat# 32371-1-AP), rabbit anti-CD68 (1:500, Cat#
30929-1-AP), rabbit anti-PPAR-
Total protein was extracted using Radio Immunoprecipitation Assay (RIPA) buffer
(P0013B; Beyotime Biotechnology, Shanghai, China) supplemented with
protease/phosphatase inhibitors. The protein concentration was determined using a
bicinchoninic assay (Beyotime Biotechnology, Shanghai, China) with bovine serum
albumin (BSA) standards and transferred to polyvinylidene fluoride membranes
(Immobilon, MilliporeSigma, Billerica, MA, USA). After being blocked with 5%
milk/Tris-buffered saline containing 0.1% Tween-20 at room temperature for 1
hour, the membranes were incubated overnight at 4 °C with primary
antibodies (all from Proteintech, Wuhan, China) at the following dilutions:
rabbit anti-CD36 (Cat# 32371-1-AP), anti-CD68 (Cat# 30329-1-AP),
anti-PPAR-
All data are presented as the
mean
To investigate the effects of different exercise intensities on hyperlipidemia,
we established a hyperlipidemia model using a high-cholesterol diet that
exhibited a lean hyperlipidemic phenotype. Serological findings indicated notably
higher TC and TG in the HFD group, but no significant increase in total body
weight or heart weight (Fig. 1), confirming the successful establishment of the
familial hypercholesterolemic mouse model in which cardiac damage precedes
systemic obesity. This allowed us to isolate direct myocardial lipotoxicity from
the confounding hyperlipidemic effects. In contrast, the HIIT+HFD group exhibited
significantly reduced TC and TG levels (p
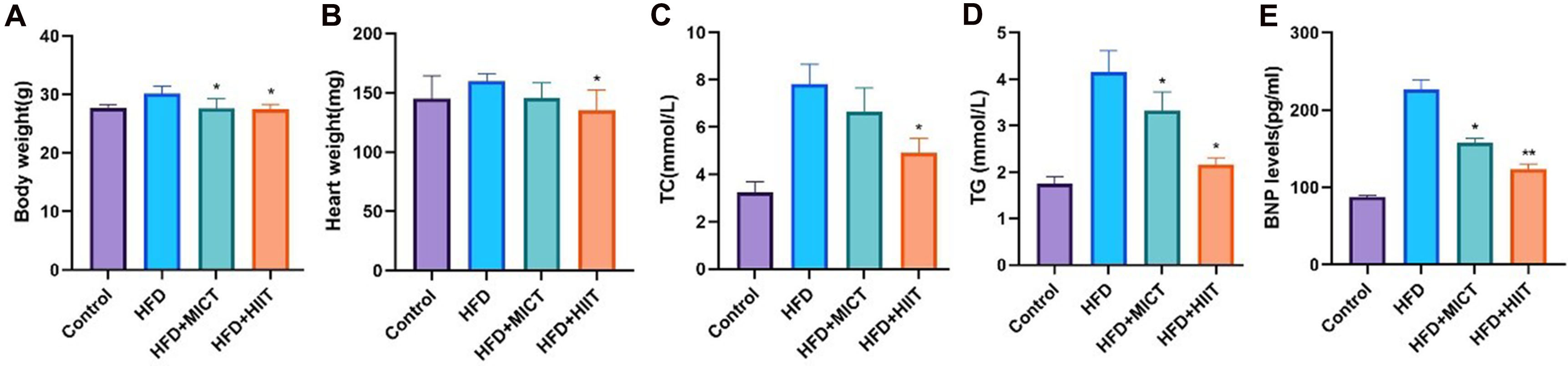 Fig. 1.
Fig. 1.
Effects of high-intensity interval training (HIIT) on body
weight, blood lipids levels and B-type natriuretic peptide (BNP) levels in mice.
(A) Body weight. (B) Heart weight. (C) Total cholesterol (TC). (D) Triglyceride
(TG). (E) BNP. *p
The HE results confirmed HFD-induced inflammatory infiltration and microvesicular steatosis in the myocardial tissue. MICT improved these lesions moderately, whereas HIIT significantly reduced them (Fig. 2). Masson’s trichrome staining revealed no histological abnormalities in the myocardial structures in the control group. However, the mice in the HFD group showed significant myocardial interstitial fibrosis (blue). The exercise interventions reduced fibrosis, with a modest effect of MICT and a more dramatic effect of HIIT.
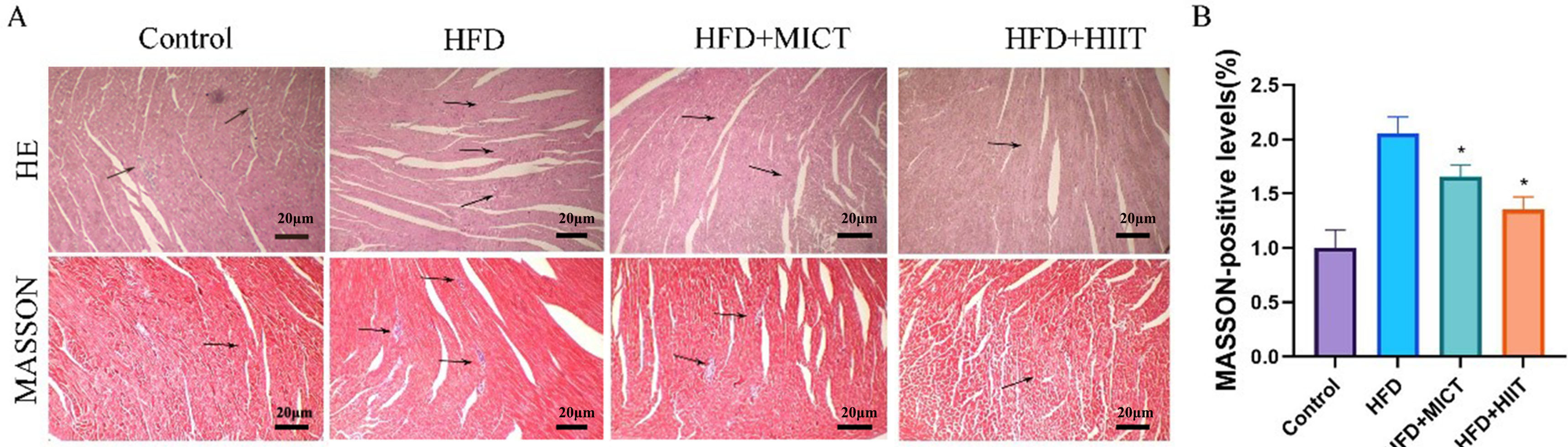 Fig. 2.
Fig. 2.
Histological analysis (hematoxylin and eosin [HE] and Masson’s
trichrome staining) of the hearts of HFD-fed apolipoprotein E knockout
(ApoE⁻/⁻) mice subjected to HIIT. (A) HE-stained tissue showing
inflammatory infiltration and microvesicular steatosis in the HFD group.
Masson-stained tissue reflecting collagen deposition (blue) and myocardial
architecture. 200
These lipid deposition markers collectively capture the full spectrum of lipid
handling ranging, from cellular uptake (CD36) and oxidized LDL recognition
(LOX-1) to storage regulation (PPAR-
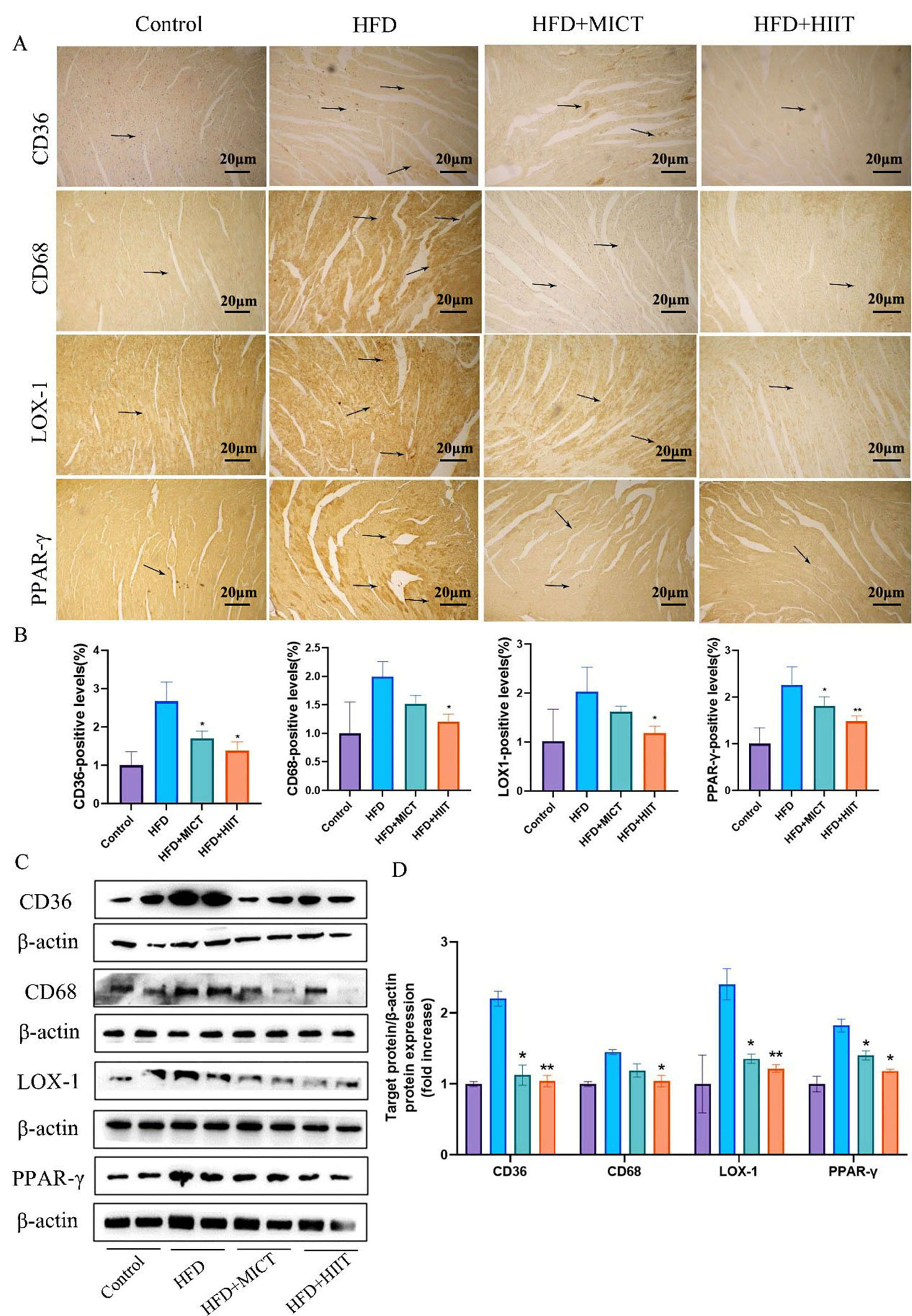 Fig. 3.
Fig. 3.
HIIT reduced lipid metabolism-related protein expression in
HFD-fed ApoE⁻/⁻ mice. (A) Immunohistochemical staining of CD36, CD68
(Cluster of Differentiation 36/38), lectin-type oxidized low-density lipoprotein
receptor 1 (LOX-1), and peroxisome proliferator-activated receptor-gamma
(PPAR-
Hyperlipidemia is associated with increased reactive oxygen species (ROS) production; therefore, here we examined the effects of different exercise on oxidative stress markers and antioxidants in mice. These markers collectively reflect the mitochondrial antioxidant defense axis, spanning upstream regulators (SIRT1/3), transcriptional activators (NRF2), and the key effector enzyme (SOD2). Our findings revealed a notable reduction in SIRT1, SIRT3, NRF2, and SOD2 levels in HFD mice versus control mice. However, HIIT attenuated the reduction in SIRT1, SIRT3, NRF2, and SOD2 levels observed in the HFD group, as shown in the HIIT+HFD group (Fig. 4).
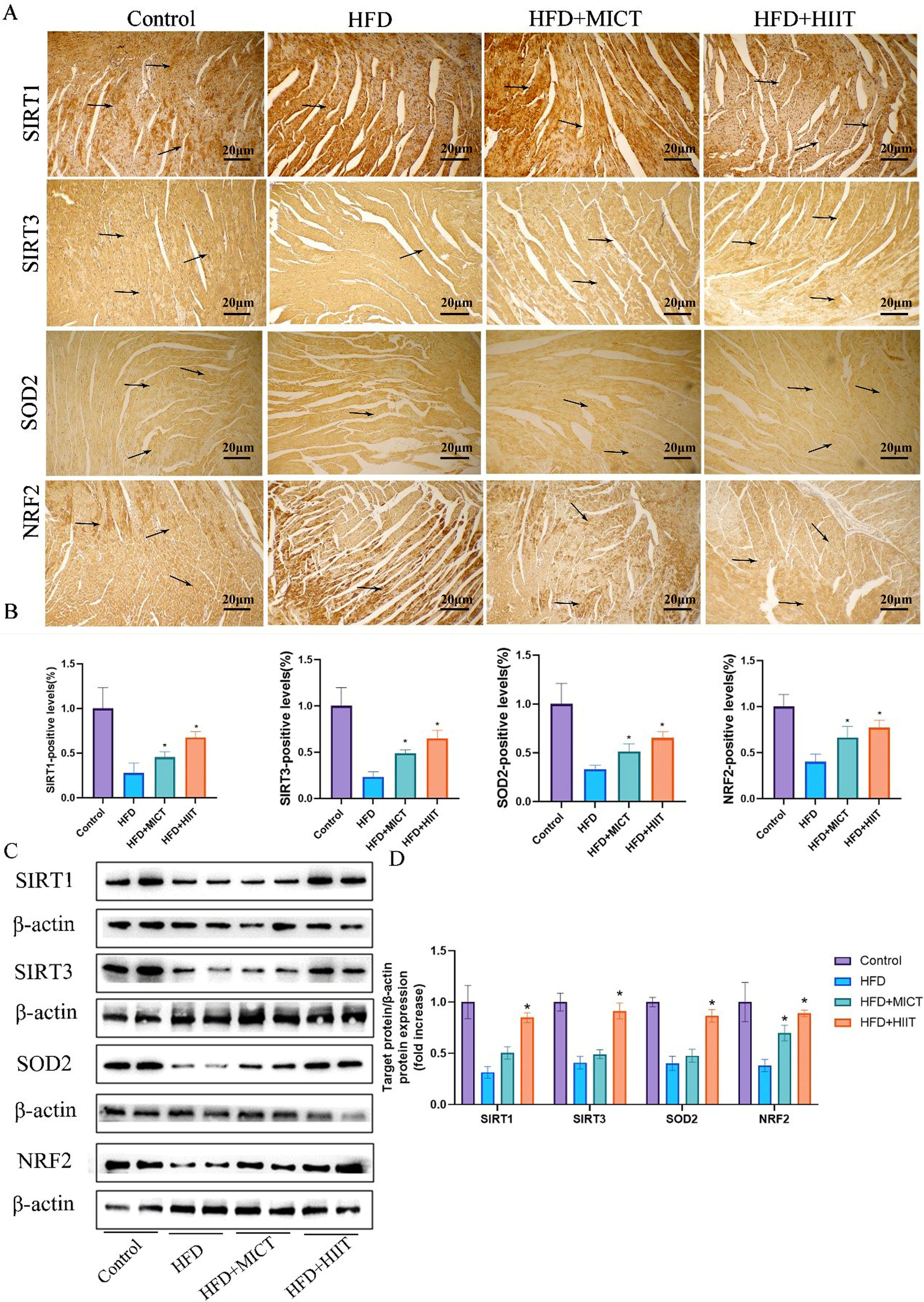 Fig. 4.
Fig. 4.
HIIT reduces antioxidant protein expression in HFD-fed
ApoE⁻/⁻ mice. (A) Immunohistochemical staining of sirtuin 1 (SIRT1),
sirtuin 3 (SIRT3), superoxide dismutase 2 (SOD2), and nuclear factor erythroid
2-related factor 2 (NRF2). The arrows indicate positively stained cells. Scale
bar = 20 µm at 200
IHC was performed to detect the expressions of the HFD-induced
inflammation-related factors. We observed increases in IL-1
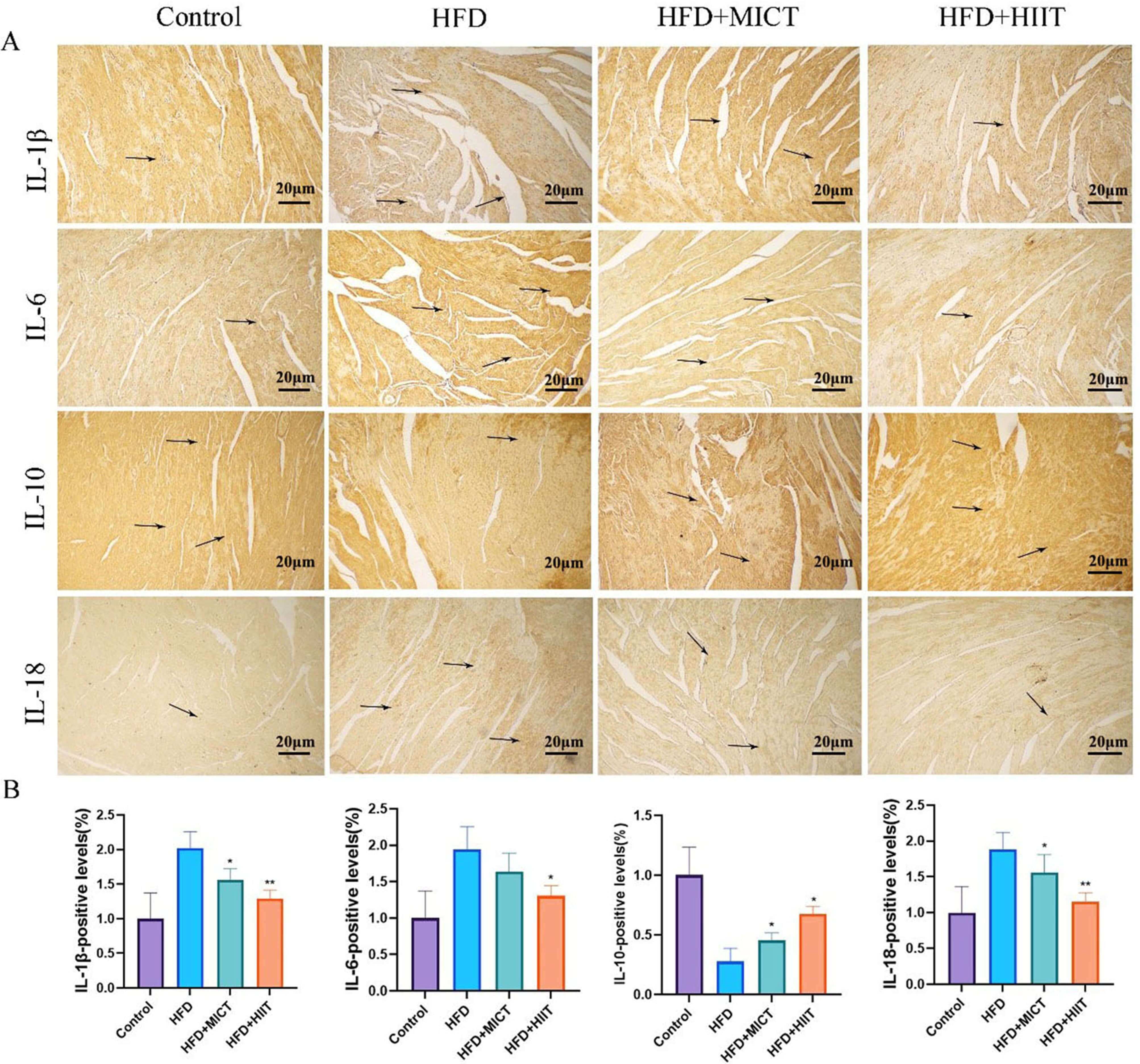 Fig. 5.
Fig. 5.
HIIT reduces inflammatory marker expression in HFD-fed
ApoE⁻/⁻ mice. (A) Immunohistochemical staining for interleukin
(IL)-1
Markers such as TGF-
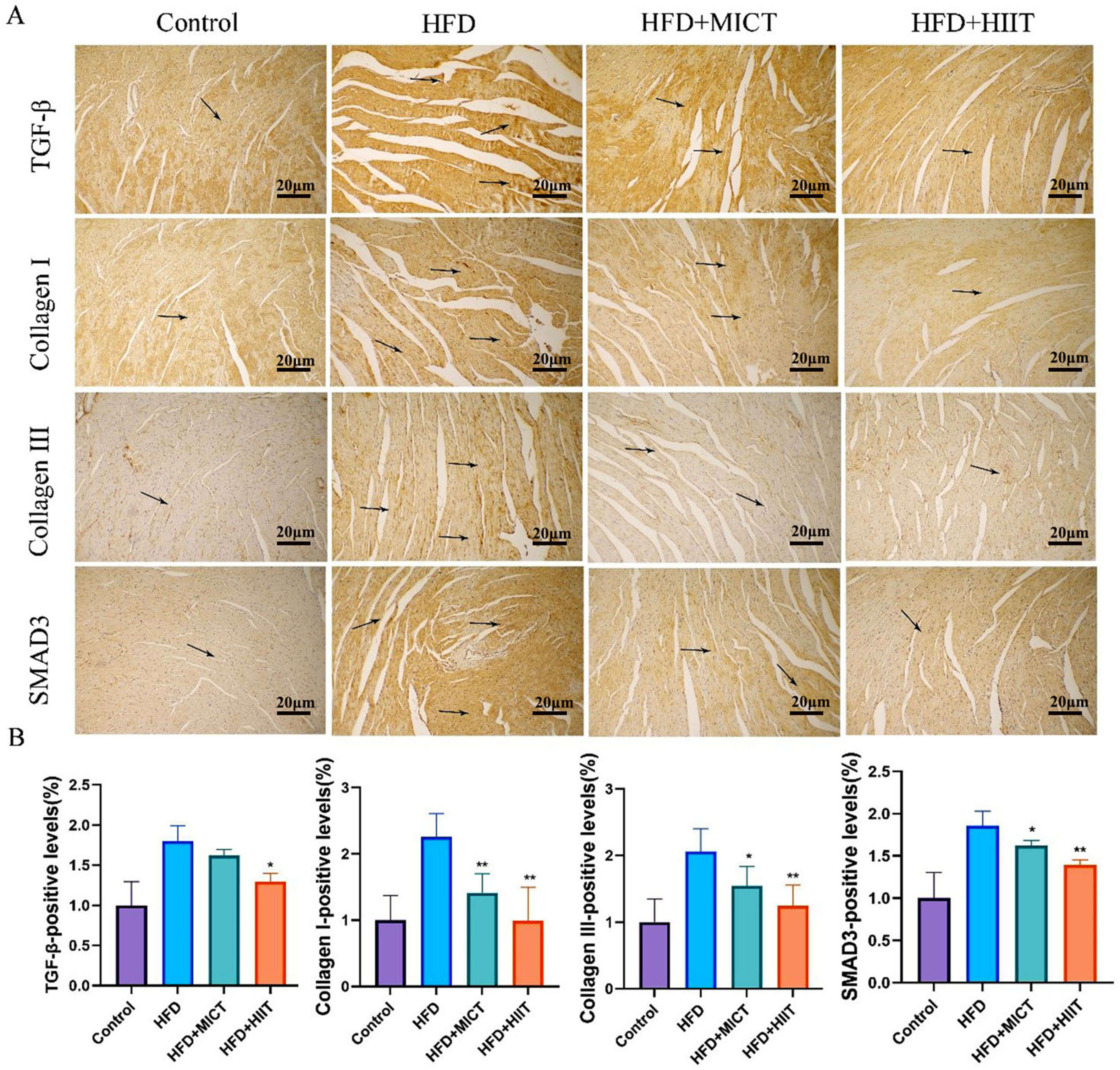 Fig. 6.
Fig. 6.
HIIT reduces the expression of fibrotic markers in HFD-fed
ApoE⁻/⁻ mice. (A) Immunohistochemical staining of collagen I, collagen
III, mothers against decapentaplegic homolog 3 (SMAD3), and transforming growth
factor-beta (TGF-
This study used a hypercholesterolemic ApoE⁻/⁻ model without obesity, in which cardiovascular risk is independent of obesity-related factors. Our systematic comparison of HIIT and MICT revealing that HIIT elicits superior signatures, including the downregulation of lipid transporters, upregulation of the SIRT3-NRF2 antioxidant axis, reduction expression of pro-inflammatory cytokines and fibrotic signaling. These intensity-dependent effects are correlated and the requirement for functional validation and causal mechanistic experiments are major limitation (Fig. 7).
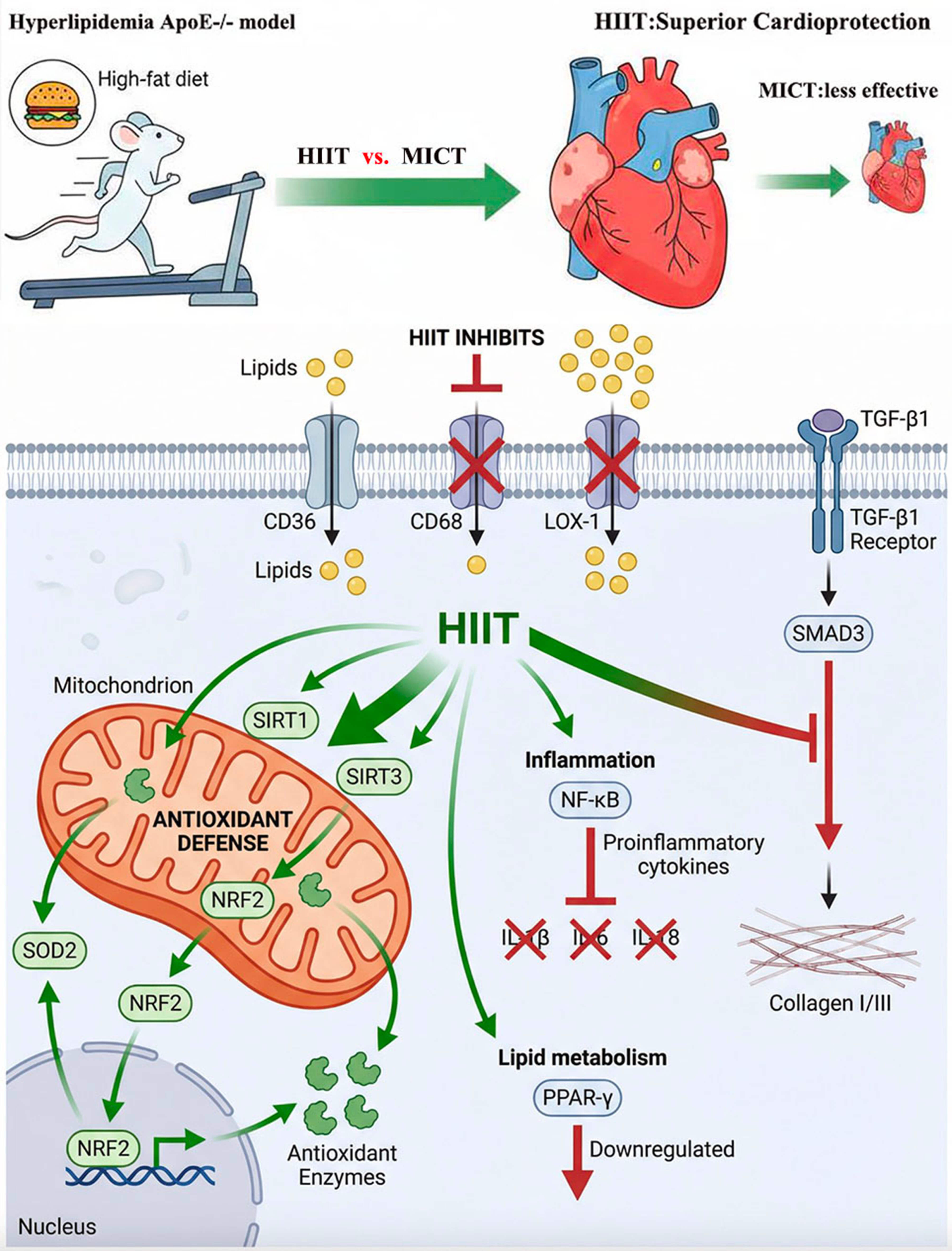 Fig. 7.
Fig. 7.
Proposed molecular mechanism illustrating the superior cardioprotective effects of HIIT versus MICT in hyperlipidemic ApoE⁻/⁻ mice. Created using Gemini, Nanobanana, and Adobe Photoshop (https://gmn.hopeai.cc; https://www.nanoai.cn).
Interestingly, Geng et al. [20] proposed that excessive exercise paradoxically exacerbates cardiac lipotoxicity via lipid redistribution under high-fat, high-calorie dietary conditions. However, Li et al. [21] proposed that compared with MICT, HIIT can comprehensively improve cardiac function and has greater potential to prevent cardiac aging in aged mice. Our research group previously demonstrated that swimming exercise ameliorated hyperlipidemia-induced cardiac injury through anti-inflammatory and antioxidant mechanisms. However, the study did not distinguish between exercise intensities [9]. Hyperlipidemia, a primary CVD risk factor, shows undefined exercise intensity responses in hypercholesterolemic ApoE⁻/⁻ models [9, 15]. In this study, a hypercholesterolemic ApoE⁻/⁻ model was successfully established using a dietary formula with 35% fat and 1.5% cholesterol. This model can reproduce myocardial stress caused by hypercholesterolemia without inducing obesity, similar to the clinicopathological features of FH [5, 9]. This preliminary study compared HIIT and MICT to identify the optimal exercise protocol for subsequent mechanistic work [15]. In this study, we compared the abilities of HIIT and MICT to reduce hypercholesterolemia-induced myocardial stress within an ApoE⁻/⁻ model without obesity or advanced age. HIIT showed superior protective effects against various aspects of lipid metabolism, oxidative stress, inflammation, and fibrosis cascades, thereby extending previous observations that did not directly compare these exercise modalities. The advantages of HIIT for metabolic diseases were previously reported. Our contribution is the parallel assessment of multiple molecular pathways in an obesity-free mouse model that provided insight into the protective effects of different exercise intensities against hyperlipidemic myocardial stress.
CD36 is a membrane fatty acid transporter whose myocardial upregulation
contributes to lipotoxicity [7, 17]. CD68 is a marker of macrophage infiltration
that reflects inflammatory cell recruitment [8, 22]. LOX-1 mediates the uptake of
atherogenic lipoproteins, thereby triggering oxidative stress [7, 22, 23].
PPAR-
Oxidative stress is the major manifestation of peripheral atherosclerosis. SIRT1/3 is a Nicotinamide Adenine Dinucleotide (NAD+)-dependent deacetylases that governs mitochondrial function and antioxidant responses [15, 25]. SIRT1 orchestrates lipid and glucose metabolism by deacetylating target proteins, thus assuming a crucial regulatory function in cellular stress resistance, energy metabolism, and tumorigenesis [25, 26]. NRF2 is a master transcriptional activator of cellular antioxidant defense because it regulates over 200 cytoprotective genes in response to oxidative stress [27, 28]. SOD2 is a mitochondrial antioxidant enzyme that mediates myocardial protection via NRF2 [28]. SIRT3, a NAD+-dependent deacetylase primarily situated within the mitochondria, governs numerous mitochondrial proteins through post-translational modifications [15, 26]. SIRT3 activates SOD2 via deacetylation, thereby enhancing intracellular ROS scavenging activity [29]. Exercise significantly reduces oxidative stress caused by hyperlipidemia. In addition, HIIT was associated with greater attenuation of hyperlipidemia-induced cardiac oxidative stress damage compared to MICT group.
The chemokine system plays a key role in the pathophysiology of cardiometabolic
diseases. Myocardial IL-1
Hyperlipidemia drives myocardial fibrosis through TGF-
Although BNP can be used as a valid surrogate for cardiac load [34], the lack of echocardiographic assessments is a major limitation. Therefore, our conclusions are limited to molecular and histological correlates of myocardial stress rather than functional outcomes. Due to limited sample resources, comprehensive transcriptional validation was not performed. All mechanistic conclusions in this study are hypothesis-generating rather than definitive. This study only compared different exercise intensities at the molecular level, and in-depth mechanistic studies are required in the future. Our study cannot explain the general superiority of HIIT in myocardial damage in patients with hypercholesterolemia, especially in those with obesity and metabolic syndrome or in the elderly population.
In the non-obese hypercholesterolemic ApoE⁻/⁻ model used in this study, HIIT elicited superior molecular adaptations compared to MICT by upregulating antioxidant defenses, downregulating of lipid deposition, suppressing pro-inflammatory cytokines, and reducing fibrotic signaling. However, these conclusions are limited by the absence of functional cardiac assessment and direct quantification of myocardial lipid content; thus further functional validation of our findings is required (Fig. 7).
All data generated and analyzed during this study are included in this published article and its supplementary information files. The datasets analyzed during the current study are available from the corresponding author upon reasonable request.
Conceptualization, supervision and project administration, HL and YS. Writing-original draft and data analysis, CQ; Animal experiments and sample collection, HL and ZP; Investigation and data curation, ZY and MS; Formal analysis and Methodology, MS and YZ; Funding acquisition, HL and YS. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal procedures adhered to the guidelines stipulated in the “Guide for the Care and Use of Laboratory Animals” and were approved by the Animal Ethics Committee of Affiliated Central Hospital of Dalian University of Technology (approval No. [2023-057-55]).
Not applicable.
This research was funded by Jiaxing Key Laboratory of Cardiac Rehabilitation, grant number 2022002.
The authors declare no conflict of interest. Given his role as the Guest Editor, Zuowei Pei had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Ioanna-Katerina Aggeli.
During the preparation of this work, the author used Gemini and Nanobanana to generate figures and visualizations. The author reviewed and edited the content as needed and takes full responsibility for its accuracy and integrity.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







