1 Department of Basic Medicine, Gannan Health Vocational College, 341000 Ganzhou, Jiangxi, China
2 College of Pharmacy, Gannan Medical University, 341000 Ganzhou, Jiangxi, China
3 Department of Pharmacy, Gannan Health Vocational College, 341000 Ganzhou, Jiangxi, China
Abstract
Alzheimer’s disease (AD) involves a progressive deterioration of cognitive abilities, memory loss, and persistent brain inflammation. Emerging evidence indicates that pyroptosis mediated by the NOD-like receptor family pyrin domain-containing 3 (NLRP3) inflammasome, contributes significantly to AD development. Docosahexaenoic acid (DHA) has demonstrated neuroprotective properties; however, the precise mechanisms by which it modulates pyroptosis in AD have yet to remained incompletely elucidated.
To explore the role of DHA in modulating microglial pyroptosis via the HOXA9-NLRP3 pathway in an AD model.
Effects of DHA on Aβ25–35-induced pyroptosis were assessed in human microglial clone 3 (HMC3) human microglial cells using CCK-8, western blotting, immunofluorescence, and Enzyme-linked Immunosorbent Assay (ELISA) assays. The role of homeobox A9 (HOXA9) in pyroptosis regulation was evaluated through overexpression and knockdown experiments. Dual-luciferase reporter assays together with chromatin immunoprecipitation (ChIP) were used to verify the interaction of HOXA9 to NLRP3 promoter. Amyloid precursor protein / Presenilin-1 double-transgenic (APP/PS1) transgenic AD mice underwent DHA treatment in vivo, and cognitive performance was assessed using the Morris water maze paradigm. Expression of HOXA9, NLRP3, and pyroptosis-related proteins were analyzed by Quantitative Real-time Reverse Transcription PCR (qRT-PCR), Western blotting, and immunofluorescence.
DHA treatment significantly reduced Aβ25–35-induced microglial pyroptosis, as indicated by decreased levels of p30-Gasdermin D (GSDMD), cleaved-caspase-1, IL-1β, and IL-18. HOXA9 overexpression reversed the protective effects of DHA, whereas NLRP3 inhibition by MCC950 enhanced DHA inhibition of pyroptosis. Dual-luciferase and ChIP assays confirmed that HOXA9 directly regulates NLRP3 transcription. In APP/PS1 mice, DHA administration enhanced cognitive performance while simultaneously decreasing the expression of pyroptosis-related markers and inflammatory mediators in brain. Inhibition of NLRP3 signaling by MCC950 further strengthened the neuroprotective actions of DHA.
DHA ameliorates AD-related cognitive decline and reduces microglial pyroptosis through suppressing the HOXA9-NLRP3 axis. These results offer novel insights into the molecular basis of DHA-mediated neuroprotection and highlight potential therapeutic targets for AD.
Keywords
- Alzheimer’s Disease
- docosahexaenoic acid
- pyroptosis
- homeobox genes
- NLR family pyrin domain-containing 3 protein
- microglia
- neuroinflammation
Alzheimer’s disease (AD) represents a progressive neurodegenerative condition
with a substantial global disease burden, placing a significant strain on
healthcare resources [1, 2]. It is marked by a gradual decline in cognitive
ability, memory loss, and behavioral changes, which arise from multifaceted
pathological mechanisms, including
Pyroptosis is characterized by specific features, including cellular edema, plasma membrane disruption, and the secretion of inflammatory factors [10, 11]. The process amplifies the inflammatory cascades and potentially exacerbates neuronal injury and progressive cognitive impairment in AD [10]. The NOD‑like receptor family pyrin domain-containing 3 (NLRP3) inflammasome is crucial for triggering pyroptosis, as it activates caspase-1 and promotes the processing and activation of inflammatory factors [12].
Increasing studies support the notion that interventions aimed at neuroinflammation and pyroptosis could offer promising therapeutic avenues for AD treatment. In this context, docosahexaenoic acid (DHA), a polyunsaturated omega-3 fatty acid abundant in neural tissues, has gained attention for its potential neuroprotective properties [13, 14]. DHA influences a variety of cellular functions, including the regulation of inflammation, oxidative stress, and synaptic activity [15].
Multiple studies have shown that DHA supplementation can reduce A
Recent findings have revealed notable links between transcription factors and inflammasome activity. Homeobox A9 (HOXA9)has been involved in diverse inflammatory pathways [18]. Although the involvement of HOXA9 in AD has not been thoroughly investigated, its involvement in regulating inflammatory pathways in other conditions suggests a potential link to AD pathogenesis [19]. Notably, bioinformatic analyses predict that HOXA9 may directly bind to the NLRP3 promoter, suggesting a role in the transcriptional regulation of this inflammasome component. This observation indicates the intriguing possibility that HOXA9 may act as a molecular switch in the activation of microglial pyroptosis in AD.
Based on these findings, we hypothesized that DHA exerts neuroprotective effects in AD by modulating the HOXA9-NLRP3 axis, thereby attenuating microglial pyroptosis. Specifically, we propose that DHA suppresses HOXA9 expression, thereby attenuating NLRP3 transcriptional activity and subsequently suppressing inflammasome assembly and pyroptotic cell death. By elucidating this novel mechanism, we aim to provide the potential molecular basis for DHA’s neuroprotective roles and identify potential therapeutic targets for AD.
Human microglial HMC3 cells (ATCC, Manassas, VA, USA) were maintained under
standard conditions, authenticated by STR profiling, and confirmed as
mycoplasma-free. Pyroptosis was induced with 25 µM A
Total RNA was extracted from cultured HMC3 cells and mouse brain tissue using TRIzol reagent (Thermo Fisher Scientific, Waltham, MA, USA). cDNA synthesis was performed using a commercial reverse transcription kit (Takara, Japan). RT-qPCR was conducted with SYBR Green chemistry, and gene expression levels were normalized. Primer sequences, thermal cycling conditions, and reagent details are provided in the Supplementary Methods [22, 23].
Total proteins from cells and brain tissues were isolated using
radioimmunoprecipitation assay buffer containing protease and phosphatase
inhibitors. Equal protein samples were separated by sodium dodecyl
sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) and transferred to
polyvinylidene fluoride membranes. Following blocking, membranes were probed with
primary antibodies targeting HOXA9 (PA5-102516; Invitrogen, Carlsbad, CA, USA),
NLRP3 (MA5-32255, Invitrogen), apoptosis-associated speck-like protein containing
a caspase-recruitment domain (PA5-50915, Invitrogen), p30-gasdermin D (ab215203;
1:1000; Abcam, Cambridge, UK), cleaved-caspase-1 (PA5-38099, Invitrogen),
interleukin (IL)-1
Pro-inflammatory cytokines (TNF-
Cell viability was evaluated using the CCK-8 assay. HMC3 cells were seeded into
96-well plates at 5
ChIP assays were performed using the EZ-ChIP™ Kit (Millipore, Burlington, MA, USA) to investigate HOXA9 binding to the NLRP3 promoter. Cells were cross-linked with formaldehyde, lysed, and sonicated to form shear chromatin. The sheared chromatin was immunoprecipitated using anti-HOXA9 antibodies, followed by DNA purification and qPCR analysis to detect enrichment at the NLRP3 promoter site.
HEK-293T cells were co-transfected with NLRP3 promoter constructs (wild-type or mutant) and either HOXA9 overexpression or control plasmids by Lipofectamine 3000 (Thermo Fisher Scientific). Cells were harvested 48 h post-transfection, and luciferase activities were measured using a Dual-Luciferase Reporter Assay System (Promega, Madison, WI, USA). Relative luciferase activity was calculated by normalizing firefly luciferase signals to Renilla luciferase signals.
Immunofluorescence analysis was performed to detect p30-GSDMD levels in both mouse brain tissue sections and cultured HMC3 microglial cells. Following fixation, permeabilization, and blocking, samples were incubated with primary antibodies and subsequently with fluorescence-conjugated secondary antibodies. Nuclear staining was performed using DAPI, and images were obtained by confocal microscopy (Leica, Wetzlar, Germany).
Male amyloid precursor protein / Presenilin‑1 double-transgenic (APP/PS1) transgenic mice were randomly assigned to control and treatment groups. Mice received daily oral DHA or vehicle for 8 weeks, with some groups additionally receiving intracerebroventricular HOXA9-overexpressing adenovirus or the NLRP3 inhibitor MCC950 [24]. Mice were anesthetized with isoflurane (induction at 4% for 3 minutes, then maintenance at 2% in oxygen for approximately 30 minutes) until loss of pedal reflex, with body temperature maintained on a heating pad. At the experimental endpoint, animals were euthanized by intraperitoneal injection of sodium pentobarbital (200 mg/kg). After deep anesthesia was confirmed, transcardial perfusion with phosphate-buffered saline and 4% paraformaldehyde was performed for histological analysis, or PBS alone for molecular assays. Death was verified by cessation of heartbeat and respiration. Cognitive function was assessed using the Morris water maze test, and brain tissues were analyzed by RT-qPCR, Western blotting, and immunofluorescence.
Data were presented as mean
To assess the influence of DHA on A
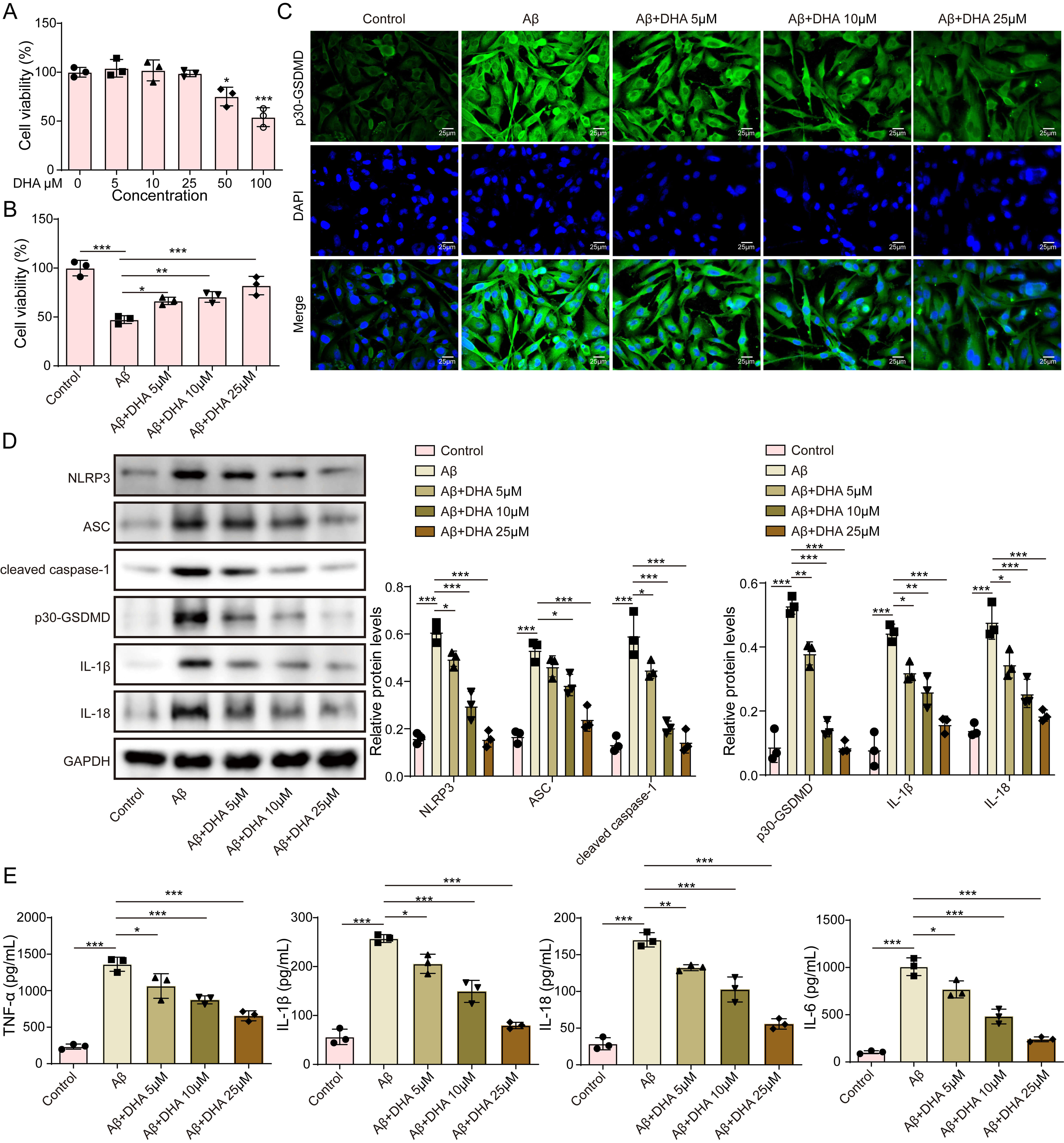 Fig. 1.
Fig. 1.
Docosahexaenoic acid (DHA) inhibits NLRP3 inflammasome
activation and reduces A
We explored the involvement of HOXA9 in the protective effects of DHA against
A
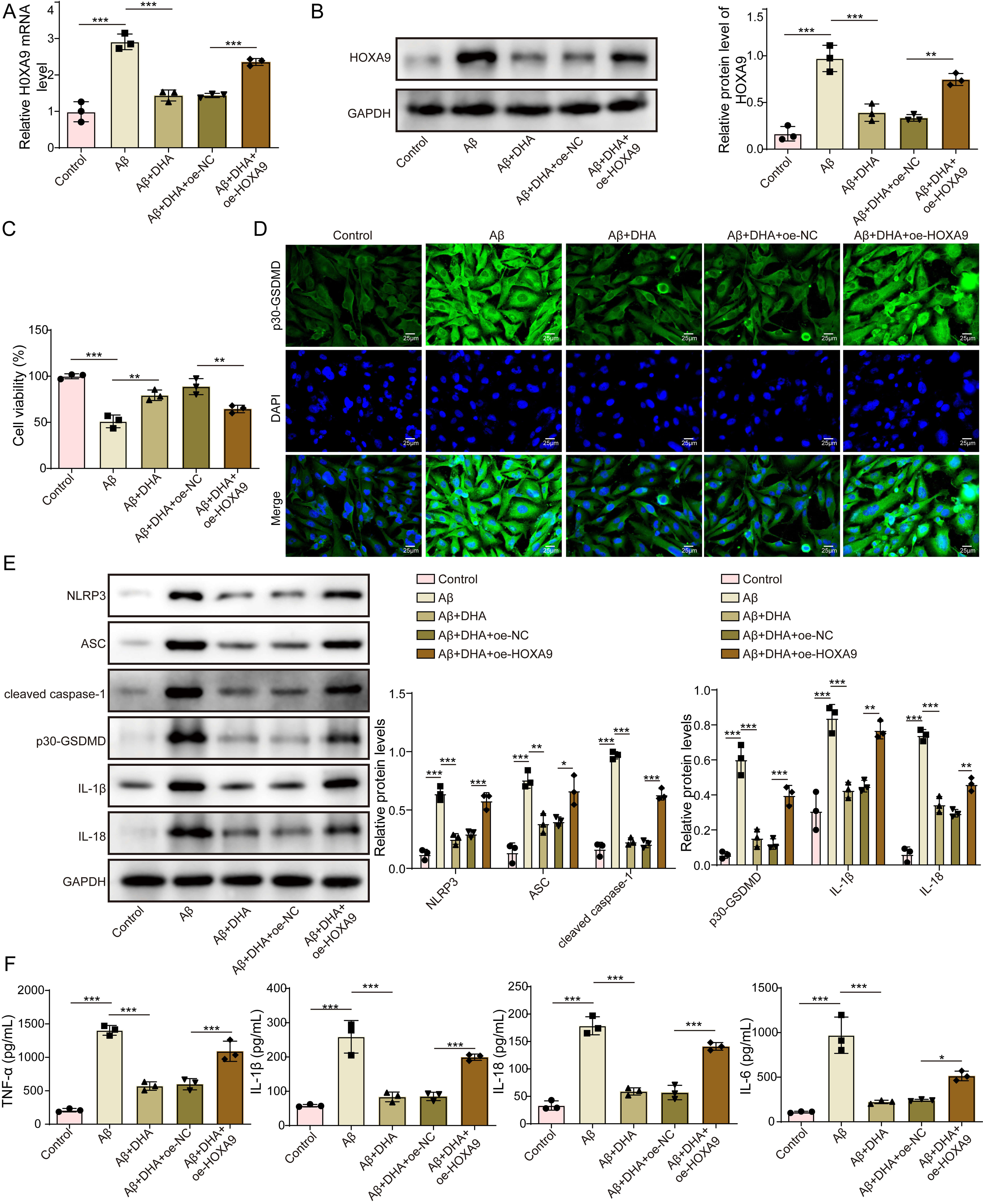 Fig. 2.
Fig. 2.
Docosahexaenoic acid (DHA) inhibits A
To elucidate the cellular pathways regarding DHA’s involvement in NLRP3, we used bioinformatics analysis, which predicted a HOXA9 binding site within the NLRP3 promoter region (Fig. 3A). Dual-luciferase assays indicated that knockdown of HOXA9 (sh-HOXA9) significantly decreased the transcriptional activity of the wild‑type NLRP3 promoter without affecting the mutant construct the mutant promoter (Fig. 3B). This finding suggests that HOXA9 directly regulates NLRP3 transcription via its predicted binding sites. ChIP assays further indicated enrichment of anti-HOXA9 at the NLRP3 promoter sequence in HEK-293T cells (Fig. 3C), providing strong evidence for the direct binding of HOXA9 to the NLRP3 promoter. To evaluate the functional impact of this interaction, we examined NLRP3 expression levels under various conditions. RT-qPCR data revealed that DHA treatment dramatically suppressed NLRP3 mRNA levels, while HOXA9 overexpression promoted NLRP3 mRNA expression (Fig. 3D). Western blot analysis was consistent with these results, demonstrating reduced NLRP3 protein expression following DHA treatment and increased expression with HOXA9 overexpression (Fig. 3E). Taken together, these data demonstrate that DHA suppresses NLRP3 activation by inhibiting HOXA9, which directly regulates NLRP3 transcription.
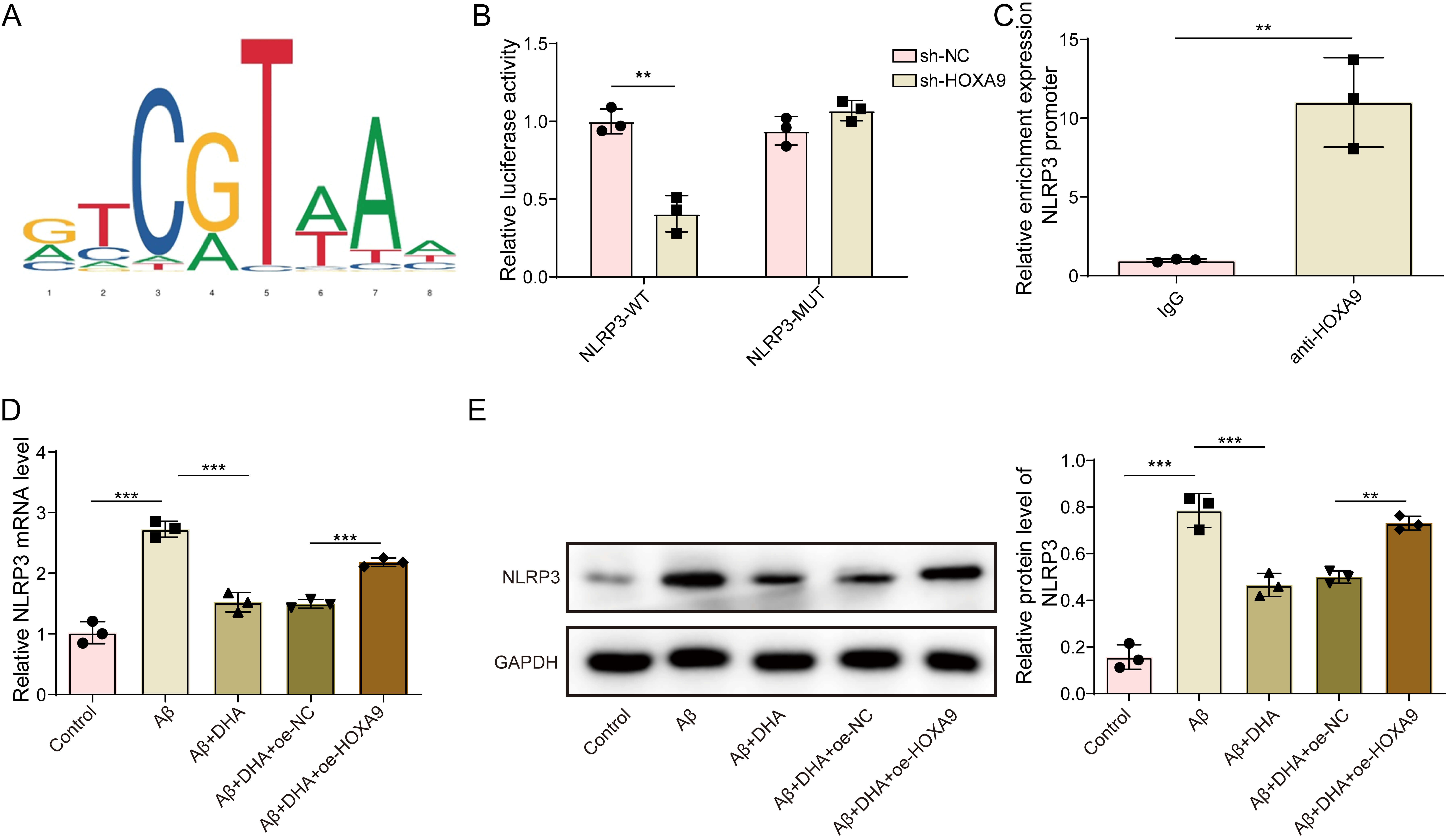 Fig. 3.
Fig. 3.
Docosahexaenoic acid (DHA) suppresses NLRP3 activation through
HOXA9 inhibition. (A) JASPAR prediction of HOXA9 binding site in the NLRP3
promoter region. (B) Dual-luciferase reporter assay results showing the effect of
HOXA9 knockdown on NLRP3 promoter activity in HEK-293T cells. (C) ChIP assay
results demonstrating HOXA9 enrichment at the NLRP3 promoter in HEK-293T cells.
(D) qRT-PCR analysis of NLRP3 mRNA levels under DHA treatment and HOXA9
overexpression conditions. (E) Western blot analysis of NLRP3 protein levels
under DHA treatment and HOXA9 overexpression conditions. **p
Further experiments were conducted to clarify how DHA influences NLRP3
transcriptional activation via HOXA9 in microglial cells. Three experimental
groups were compared: A
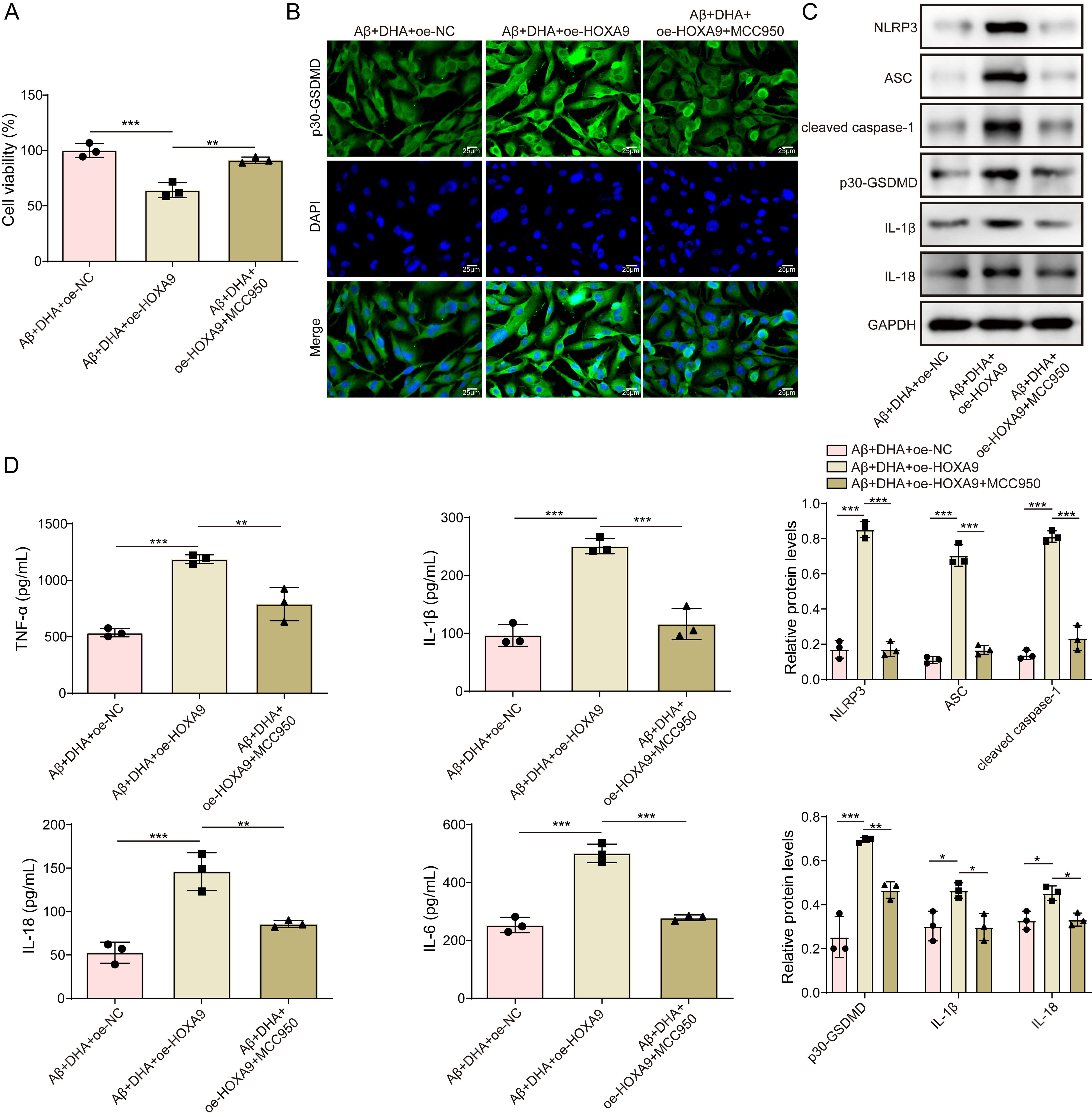 Fig. 4.
Fig. 4.
Docosahexaenoic acid (DHA) inhibits cellular pyroptosis by
regulating NLRP3 transcriptional activation via HOXA9. (A) Cell viability of
HMC3 cells under different treatments, assessed using the CCK-8 assay. (B)
Representative immunofluorescence images and quantification of p30-GSDMD. Scale
bar = 25 µm. (C) Western blot analysis of pyroptosis-related proteins
(p30-GSDMD, cleaved-caspase-1, IL-1
Learning and memory functions of APP/PS1 transgenic mice were assessed using the Morris water maze. The AD model group exhibited a significantly prolonged escape latency, fewer platform crossings, and reduced time spent in the target quadrant. DHA improved these cognitive impairments, as evidenced by a shortened escape latency, increased platform crossings, and prolonged target quadrant time. However, overexpression of HOXA9 (DHA+oe-HOXA9 group) partially reversed the cognitive benefits conferred by DHA, resulting in increased escape latency, reduced platform crossings, and decreased target quadrant time compared with the DHA+oe-NC group. Notably, cotreatment with the NLRP3 inhibitor MCC950 (DHA+oe-HOXA9+MCC950 group) mitigated the adverse effects of HOXA9 overexpression and restored cognitive performance to levels (Fig. 5A).
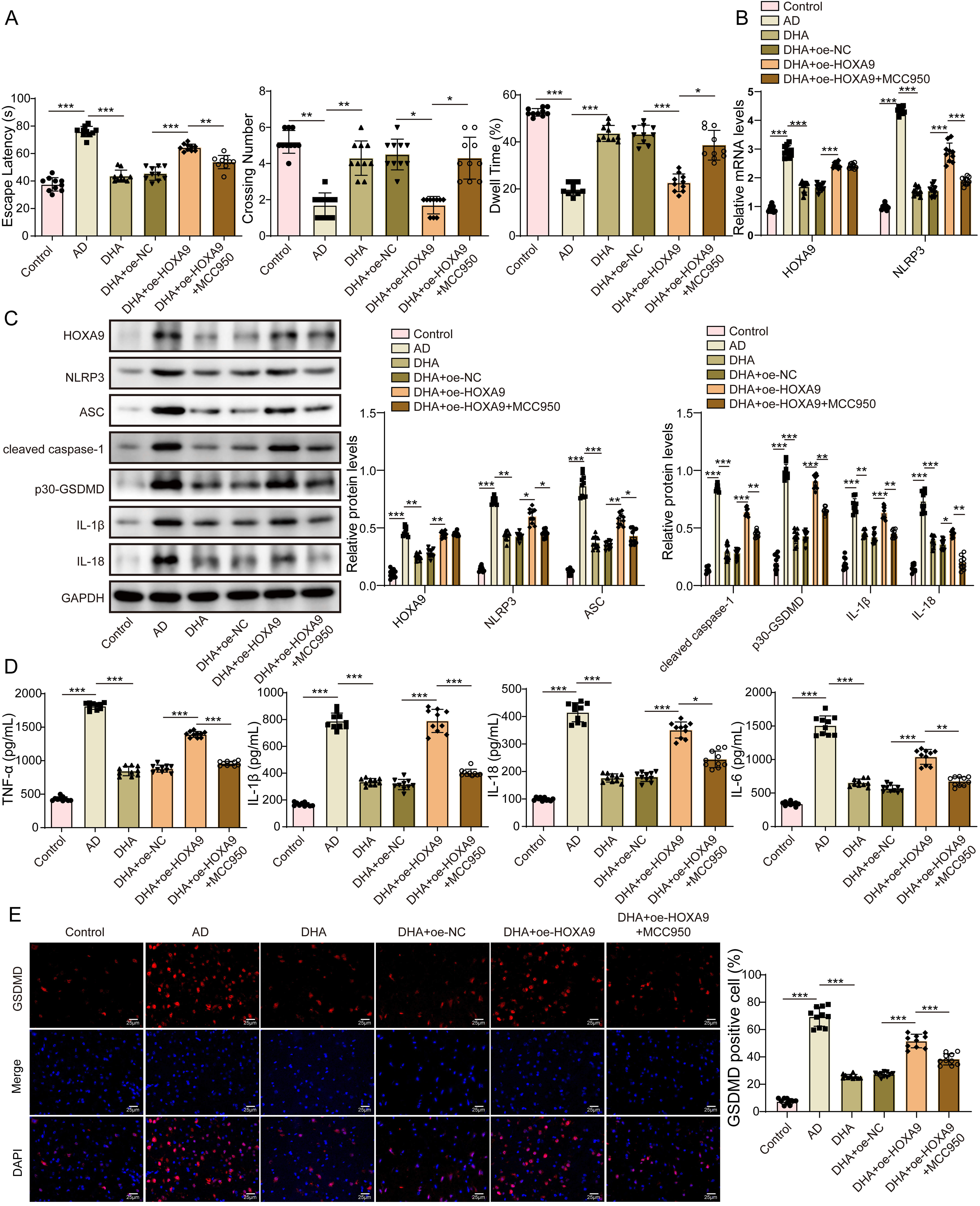 Fig. 5.
Fig. 5.
Docosahexaenoic acid (DHA) improves learning and memory
abilities and reduces pyroptosis levels in Alzheimer’s disease mice via the
HOXA9-NLRP3 Axis (n = 10 mice in each group). (A) Morris water maze performance
of APP/PS1 mice across different treatment groups. Escape latency, platform
crossings, and time spent in the target quadrant were measured. (B,C) qRT-PCR and
Western blot analyses of HOXA9 and NLRP3 expression levels in the brain tissues
of the different groups. (D) ELISA results showing the levels of inflammatory
cytokines (TNF-
The AD model group showed significant upregulation of HOXA9 and NLRP3 expression. Correspondingly, the levels of pyroptosis-related proteins were markedly increased in AD models. DHA treatment dramatically reduced the expression of these proteins, indicating its protective effect against neuroinflammation and pyroptosis. In contrast, overexpression of HOXA9 in DHA-treated mice (DHA+oe-HOXA9 group) resulted in a dramatically increase in HOXA9 and NLRP3 levels, along with higher levels of pyroptosis markers, compared with the DHA+oe-NC group. Co-administration of MCC950 effectively counteracted these effects, resulting in reduced expression of HOXA9, NLRP3, and associated pyroptosis markers in the DHA+oe-HOXA9+MCC950 group (Fig. 5B,C).
The levels of these cytokines were markedly higher in the AD model group,
indicating a pronounced inflammatory response. DHA treatment markedly reduced
TNF-
Immunofluorescence staining of p30-GSDMD was performed to visualize and quantify pyroptosis in brain tissues. The results were consistent with those of the biochemical analyses; the AD model group showed elevated p30-GSDMD levels, indicating increased pyroptosis. DHA treatment reduced p30-GSDMD staining, whereas HOXA9 overexpression in DHA-treated mice (DHA+oe-HOXA9) increased p30-GSDMD levels. Co-treatment with MCC950 in the DHA+oe-HOXA9+MCC950 group significantly diminished p30-GSDMD staining, corroborating the involvement of the HOXA9-NLRP3 axis in pyroptosis regulation in AD (Fig. 5E). These results emphasize the importance of the HOXA9-NLRP3 axis in mediating the neuroprotective effects of DHA against AD-associated cognitive decline and neuroinflammation.
Numerous studies have underscored neuroinflammation as a key contributor to AD
development. particularly regarding the impact of NLRP3 inflammasome activation
on neurodegeneration [25, 26, 27]. Consistent with these observations, our data
demonstrate that DHA administration leads to decreased levels of NLRP3 and
related downstream molecules such as p30-GSDMD, cleaved-caspase-1, IL-1
The innovative aspect of our research lies in identifying HOXA9 plays a central
role in regulating NLRP3 inflammasome signaling. Although much of the existing
literature has focused on NF-
Our in vivo experiments further confirmed that DHA improved cognitive performance in AD mice, as demonstrated by superior outcomes in the Morris water maze. Improved cognitive performance was associated with decreased levels of pyroptosis-associated proteins and inflammatory factors in brain tissues, reinforcing the link between DHA’s anti-pyroptotic effects and its cognitive benefits. This study is consistent with earlier studies suggesting that DHA can alleviate cognitive deficits in AD models, likely through effects on synaptic activity and neuroinflammatory processes [31, 32]. However, our study extends beyond these findings by providing a mechanistic explanation for how DHA influences pyroptosis through the HOXA9-NLRP3 axis.
Notwithstanding these promising findings, this study has certain limitations
that warrant consideration. Although we have demonstrated HOXA9 involvement in
NLRP3-mediated pyroptosis in vitro and in vivo, the exact
molecular interactions between HOXA9 and other transcriptional regulators remain
unclear. Moreover, although the APP/PS1 transgenic mouse is a common model for AD
research, it does not fully replicate the multifaceted nature of human AD. Future
studies should explore the effects of DHA in additional AD models, including
those incorporating tau pathology or other AD-related features. Furthermore, our
study primarily focused on the role of the HOXA9-NLRP3 axis in microglia. Given
the complexity of AD, it is likely that other cells including astrocytes and
neurons also lead to the observed effects of DHA. Future research should
investigate the broader impact of DHA on different cell types within the AD brain
and explore whether similar regulatory mechanisms exist in these cells. Another
limitation was the utilization of relatively young APP/PS1 mice (8 weeks old). At
this age, A
In this study, we demonstrated that DHA dramatically attenuates
A
DHA significantly alleviates AD-related cognitive impairment and neuroinflammation by suppressing microglial pyroptosis. The study demonstrates that DHA inhibits the HOXA9–NLRP3 axis, thereby reducing inflammasome activation and downstream pyroptotic signaling. These findings highlight this regulatory pathway as a potential therapeutic target and support the value of DHA as a promising intervention strategy for AD.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
JLX, XHH, and CLW designed the research study. JLX and QJ performed the experiments. CLW conducted data analysis. JLX drafted the manuscript. JPP participated in the study design, performed experiments, analyzed data, prepared figures and tables, searched references, and was the corresponding author. All authors contributed to editorial revisions, read, and approved the final manuscript. All authors have participated sufficiently in the work to take responsibility for its content and agreed to be accountable for all aspects of the study.
All animal procedures were conducted in accordance with the Guide for the Institutional Animal Care and Use and approved by the Institutional Animal Care and Use Committee of Gannan Health Vocational College (Ethics Number: 20240056). Author confirms all animal experiments were in accordance with the ARRIVE guidelines and the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments, or the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978).
Not Applicable.
This work was supported by the Jiangxi Provincial Natural Science Foundation General Project (No: 20232BAB206049).
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT (developed by OpenAI) to assist with spelling and grammar checking. After employing this tool, the authors carefully reviewed and edited the content to ensure its accuracy and clarity. The authors take full responsibility for the content of this publication and explicitly acknowledge that the scientific interpretation, data analysis, and conclusions were conceived and verified independently by the authors. The use of AI was limited to language refinement and did not influence the scientific results or integrity of the study.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL46572.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





