1 Emergency Center, Dingxi People’s Hospital, 743000 Dingxi, Gansu, China
2 Heart Center, The First Hospital of Lanzhou University, 730000 Lanzhou, Gansu, China
3 Gansu Province Clinical Research Center for Cardiovascular Diseases, 730000 Lanzhou, Gansu, China
4 The First Clinical Medical College of Lanzhou University, 730000 Lanzhou, Gansu, China
5 Department of Cardiology, Dingxi People’s Hospital, 743000 Dingxi, Gansu, China
†These authors contributed equally.
Abstract
The gut microbiome is increasingly recognized as a modifiable contributor to coronary artery disease (CAD). This narrative review integrates mechanistic and clinical evidence regarding short-chain fatty acids (SCFAs), trimethylamine-N-oxide (TMAO), and bile acids, and appraises therapeutic modulation via diet; probiotics, prebiotics, and synbiotics; fecal microbiota transplantation (FMT); and drug–microbiome interactions. SCFAs generally confer anti-inflammatory and lipid-regulatory effects, whereas bile acid signaling exhibits context-dependent metabolic actions. Findings regarding TMAO are inconsistent; in several cohorts, associations with cardiovascular risk become null or attenuated after adjustment for renal function (estimated glomerular filtration rate [eGFR]) and dietary patterns. Most interventional studies are small, use surrogate endpoints, and vary in strains and dosing, limiting certainty. Microbiome profiles differ across geographic regions, racial and ethnic groups, and dietary patterns, underscoring the need for stratified approaches. Routine FMT in CAD remains constrained by safety, feasibility, and ethical and logistical considerations. Overall, the microbiome represents a promising yet unproven therapeutic target in CAD. Future trials should standardize interventions, rigorously control for confounders, evaluate drug–microbiome interactions, and be adequately powered to detect clinical events to enable precision medicine.
Keywords
- coronary artery disease
- gastrointestinal microbiome
- precision medicine
Coronary artery disease (CAD), the most common manifestation of atherosclerotic cardiovascular disease (ASCVD), is the focus of this review. Its pathogenesis is complex, involving lipid metabolism disorders, immune inflammatory response and other aspects [1, 2]. CAD dominates the world’s major medical burden. Worldwide, CAD causes more than 8 million deaths each year [3]. Risk factors for CAD include age, gender and genetic background, smoking habits, hyperlipidemia, hypertension, diabetes, etc. These risk factors significantly increase the risk of CAD by promoting atherosclerosis or enhancing the inflammatory response. In recent years, diet and metabolic factors are closely related to gut microbes, gradually becoming a new perspective for understanding the etiology of CAD [4, 5, 6].
Gut microbiota refers to the microbial community present in the human intestine, including bacteria, viruses, fungi, archaea and other microorganisms, with a number of 100 trillion, and the number of its genes is 150 times more than the human body’s own genes. Under physiological conditions, healthy intestinal microbial communities usually show high diversity and have certain genus dominance, including two major phyla: Bacteroidetes and Firmicutes [7, 8, 9]. The gut microbiome mainly affects the health of the host through the metabolites it produces. Short-chain fatty acids (SCFAs) are one of the key metabolites produced by the fermentation of dietary fiber under the action of gut microbiome. They play an important role in maintaining the integrity of the intestinal barrier and regulating systemic inflammation and metabolism [10, 11]. Another important metabolite is trimethylamine-N-oxide (TMAO), which is formed by the metabolism of choline and carnitine by intestinal bacteria and is then further oxidized in the liver. Studies have shown that elevated TMAO levels are closely related to the progression of atherosclerosis and coronary artery disease [12, 13]. The dynamic balance of the gut microbiome structure is highly dependent on factors such as the host’s diet, lifestyle, and immune system. Fiber and polyphenols in dietary ingredients are believed to increase the relative abundance of probiotics, while high-fat and high-sugar diets may induce gut dysbiosis, thereby affecting overall health. This complex microecological system plays a core role in cardiovascular health by regulating inflammatory responses, immune tolerance, and metabolic homeostasis. For this reason, gut microbiome provides a new research direction for the prevention and treatment of CAD [14, 15]. This work is a narrative review. It does not report a protocolized database search or meta-analysis; studies were selected to illustrate mechanisms and translational relevance, and uncertainties are explicitly discussed.
In recent years, the microbiome has emerged as one of the focal points of research. Over the past two decades, numerous studies have untangled the impact of microbial communities on the physiology and metabolism of multicellular organisms, further influencing health and disease states. Bacteria are present in various parts of the human body, including the oral cavity, gastrointestinal tract, and skin. Within the digestive tract, the colon stands as the primary contributor to the total bacterial count, while the stomach and small intestine contribute to a lesser extent. Hence, the colon is a key site for estimating the bacterial load in the body [16]. The gut microbiota refers to the microbial community residing in the human intestine, comprising a diverse array of microorganisms such as bacteria, viruses, fungi, and archaea. With a population of up to 100 trillion microorganisms and a gene count exceeding 150 times that of the human genome, it is often described as the “second genome” of the human body [17]. Under physiological conditions, a healthy gut microbial community typically exhibits high diversity and possesses certain dominant taxa, including Bacteroidetes, Firmicutes, Proteobacteria, and Actinobacteria [18]. These microorganisms are predominantly anaerobic bacteria that typically colonize at birth. As the organism develops, its microbiota gradually diversifies, reaching an adult-like configuration by around 3 years of age and remaining relatively stable until old age [19, 20, 21, 22]. The richness and diversity of the gut microbiota are determined by various factors such as environment, genetics, and age, but are primarily influenced by the dietary habits of the host [23, 24]. The gut microbiota can degrade nutrients, produce vitamin B and vitamin K, promoting the growth, metabolism, and developmental processes of the host. Simultaneously, the gut microbiota protects the host in various ways through the production of short-chain fatty acids via the fermentation of dietary fiber, providing energy for intestinal cells and regulating the immune system. Furthermore, microorganisms maintain intestinal development and integrity by secreting epithelial renewal signals and inducing intestinal angiogenesis. In summary, accumulating evidence indicates that the gut microbiota and their metabolites contribute to host physiology relevant to cardiovascular health; however, the magnitude, direction, and generalizability of these effects vary across populations and disease states [25, 26]. Patients with CAD exhibit reduced gut microbiota diversity compared to healthy individuals, a pattern observed across multiple cohorts [27, 28]. In CAD patients, there is a significant increase in proinflammatory bacteria such as Escherichia-Shigella, while the population with anti-inflammatory properties, such as Faecalibacterium and Roseburia, decreases significantly. This suggests that the ecological structure of intestinal bacteria may play a key role in the pathophysiology of CAD [29, 30, 31]. The findings of Cui et al. [32] also indicate an increase in the Firmicutes phylum and a decrease in the Bacteroidetes phylum among CAD patients. Jie et al. [33] reported an increase in the Enterobacteriaceae family and Streptococcus species, accompanied by a reduction in Roseburia intestinalis and Faecalibacterium prausnitzii. Meanwhile, studies have shown that the microbiota of CAD patients exhibits lower fermentation capacity and enhanced inflammatory characteristics. A meta-analysis and systematic review involving 21 studies have indicated differences in the composition of gut microbiota between patients with CAD and healthy controls. One of the most significant disparities is the reduction of beneficial Bacteroidetes and Helicobacter species in CAD patients, coupled with an increase in Enterobacteriaceae, Actinobacteria, and Verrucomicrobia populations. Available studies report that CAD-related shifts in gut microbiota composition are associated with quantitative changes in putative atherogenic microbial products—such as lipopolysaccharides, trimethylamine-N-oxide, and selected uremic toxins—and with intermediate phenotypes (e.g., systemic inflammation and endothelial dysfunction). These observations are largely derived from observational cohorts and small interventional studies, and results—particularly for TMAO—remain heterogeneous; therefore, causality and effect sizes should be interpreted with caution [34].
However, multiple factors can influence the gut microbiome. Across populations, gut microbiome composition separates by country and lifestyle, with U.S. adults showing lower diversity and distinct community structure compared with Malawian and Amerindian cohorts; age explains some variation, but geography remains a major axis throughout life [24]. People who consume animal protein/fat for a long time are prone to the development of Bacteroides-rich flora, while a carbohydrate/fiber-rich diet is conducive to the growth of Prevotella; compared with their European peers, children with a high-fiber diet in rural areas (such as Burkina Faso) have rich fiber-degrading bacteria and higher levels of SCFAs [35]. Even within the same national environment, subtle but consistent microbiome differences are detectable across self-identified ethnic groups in the U.S., involving sets of taxa that replicate across cohorts and may intersect with health disparities biology [36].
ASCVD—which encompasses coronary disease, cerebrovascular disease, and peripheral artery disease—this review focuses on CAD. Atherosclerosis is the predominant pathological substrate of CAD, although plaque burden and clinical presentation vary widely across individuals. Beyond inherited risk, environmental contributors—including the gut microbiota—modulate CAD susceptibility and disease expression, providing a rationale to examine microbially derived metabolites in CAD pathophysiology.
TMAO is a product of co-metabolism between the host and gut microbiota. Its generation begins with the breakdown of precursors such as choline and carnitine from the diet into trimethylamine by gut microbiota. This trimethylamine is then oxidized into TMAO in the liver by the action of flavin-containing monooxygenase (FMO). Research has indicated that TMAO plays a pathogenic role in the occurrence and development of CAD [37, 38]. TMAO promotes atherosclerosis through multiple pathological mechanisms. It enhances cholesterol absorption, inhibits bile acid synthesis, disrupts reverse cholesterol transport, and accelerates the production of foam cells. Simultaneously, TMAO activates inflammatory responses, which also contribute to cholesterol overload in foam cells and exacerbate endothelial damage. Furthermore, TMAO can exacerbate the progression of CAD by promoting platelet activation and increasing the risk of thrombosis [39, 40, 41]. It has been reported that impaired intracellular calcium transport in platelets leads to enhanced platelet reactivity and increased platelet adhesion to collagen fibers. TMAO also affects liver lipid metabolism and systemic metabolic homeostasis. Specifically, it regulates genes related to liver lipid metabolism, leading to fatty liver disease, insulin resistance, altered cholesterol metabolism, and accelerated plaque formation [37, 42]. Koeth et al. [43] confirmed in a mouse model study that trimethylamine oxide inhibits the expression of Cyp7a1 enzyme and bile acid transporter. Cyp7a1 is responsible for bile acid synthesis and suppression of cholesterol catabolism. The absence of Cyp7a1 leads to reduced bile acid synthesis and secretion, resulting in the progression of atherosclerosis. Simultaneously, decreased expression of bile acid transporters in the liver negatively impacts the body’s primary pathway for cholesterol elimination. Clinical studies have validated the positive correlation between elevated levels of TMAO and the risk of CAD. For instance, a study involving 199 patients with acute coronary syndrome (ACS) revealed a significant increase in blood TMAO concentrations compared to healthy controls, and these concentrations were directly proportional to the severity of coronary artery stenosis [44]. Furthermore, according to Senthong and colleagues [45], elevated plasma TMAO levels predict a higher long-term risk of death among patients with stable coronary artery disease who are receiving optimal medical therapy. These findings underscore the value of TMAO as a metabolic marker in CAD and provide a target basis for interventional therapies. Fig. 1 shows the multi-faceted mechanism of TMAO-induced CAD. Notably, not all cohorts show a positive association between circulating TMAO and cardiovascular risk. In two large prospective studies—one comprising 4132 patients with suspected CAD and another community cohort of 6393 adults—TMAO did not predict 10-year all-cause or cardiovascular mortality after multivariable adjustment including renal function [46]. Similarly, in a population-based study of early–middle-aged adults, TMAO was not associated with subclinical atherosclerosis (incident coronary artery calcium, CAC progression, or carotid intima-media thickness) [47]. While TMAO has been associated with atherosclerosis and adverse cardiovascular outcomes in multiple cohorts, results are not uniform. Associations can attenuate after accounting for renal function, diet, and medications; study assays and sampling (fasting vs. non-fasting) also vary. Experimental data support endothelial and hemostatic effects, yet effect sizes and generalizability remain uncertain, and causality has not been definitively established. Accordingly, we use cautious phrasing and avoid overreliance on any single line of evidence. For TMAO, confounding by renal function is a major consideration because TMAO is largely cleared by the kidney. In several cohorts, the association between circulating TMAO and CAD or atherosclerotic phenotypes attenuates after adjustment for eGFR or when individuals with chronic kidney disease are analyzed separately [48, 49, 50]. Additional sources of heterogeneity include dietary patterns (e.g., fish and carnitine intake), gut microbial composition, antibiotic or proton-pump inhibitor use, and liver and bile acid metabolism. Prospective and Mendelian-randomization evidence is mixed, with some studies reporting null associations with incident clinical events. These inconsistencies underscore the need for standardized phenotyping, repeated measures, and careful modeling of kidney function and diet.
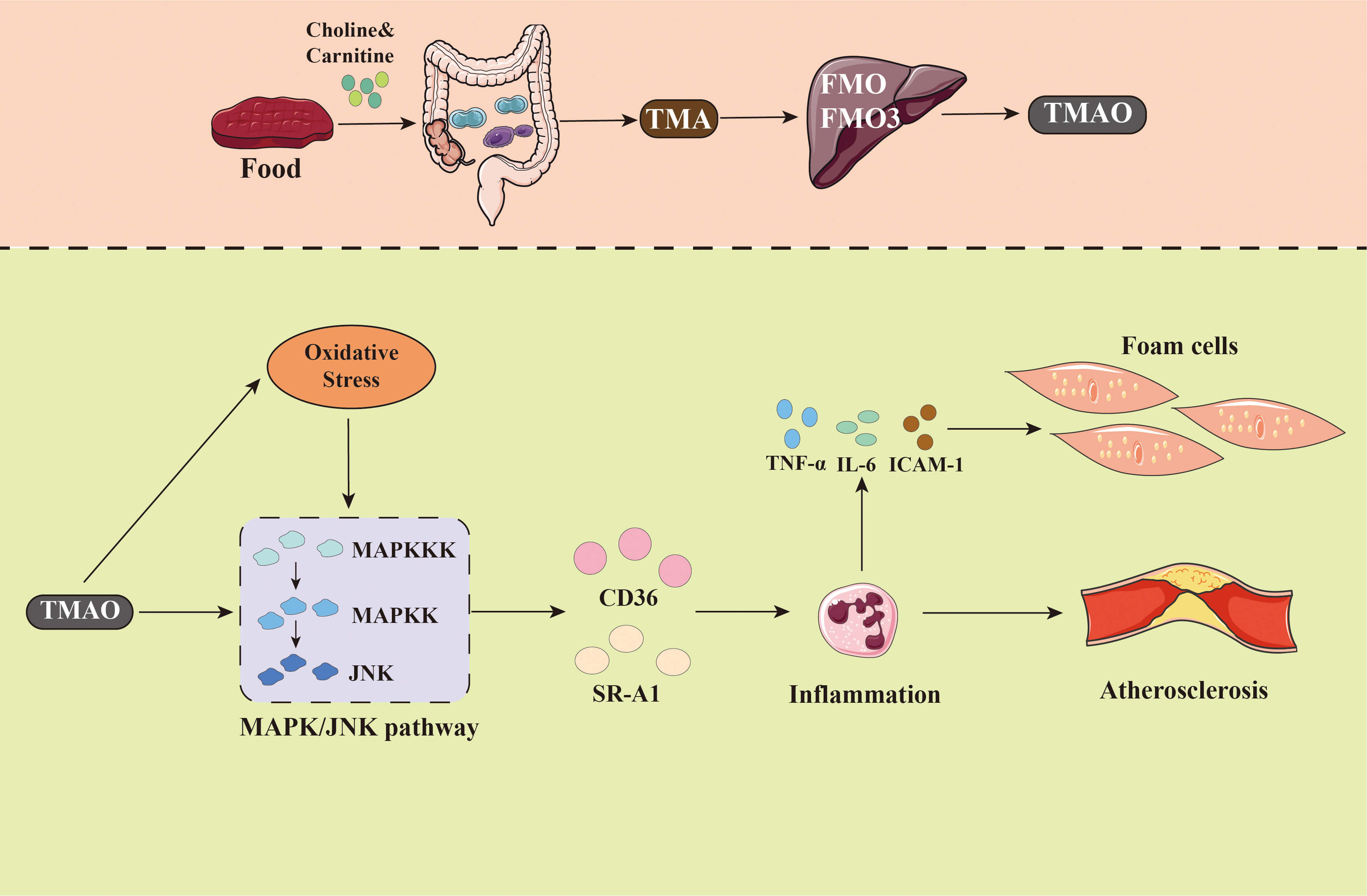 Fig. 1.
Fig. 1.
The gut microbiota metabolite TMAO is closely related
to the occurrence and development of CAD. TMAO is a metabolite produced from
TMA, which is generated by the gut microbiota metabolism of dietary components
such as choline, phosphatidylcholine, and carnitine. TMA is then oxidized in the
liver by FMO3 to form TMAO. The mechanism by which TMAO contributes to
atherosclerosis and coronary heart disease involves multiple levels. CAD,
coronary artery disease; TMAO, Trimethylamine N-oxide; MAPKKK, Mitogen-Activated
Protein Kinase Kinase Kinase; MAPKK, Mitogen-Activated Protein Kinase Kinase;
JNK, c-Jun N-terminal Kinase; MAPK, Mitogen-Activated Protein Kinase; CD36,
Cluster of Differentiation 36; SR-A1, Scavenger Receptor Class A Member 1;
TNF-
SCFAs, such as acetic acid, propionic acid, and butyric acid, are crucial
metabolites produced by gut microbiota through the fermentation of dietary fiber.
These SCFAs exert multifaceted protective effects on cardiovascular health. By
binding to G protein-coupled receptors (e.g., GPR41 and GPR43) and activating
histone deacetylases (HDACs), SCFAs modulate the host immune system, thereby
ameliorating low-grade inflammation and enhancing intestinal epithelial barrier
function [2, 29]. Previous studies have indicated that acetic acid, butyric acid,
and propionic acid play significant roles in atherosclerosis by regulating the
production of Regulatory T cells and inhibiting histone deacetylase [51, 52].
This influence permeates multiple aspects of atherosclerosis. Tian et
al.’s [53] research on butyrate has shown that suppressing the expression of
endothelial cell nicotinamide adenine dinucleotide phosphate oxidase 2 and
reactive oxygen species through the PPAR
| Metabolites | Source | Function | Effects on CAD | Evidence source |
| TMAO | Choline, L-Carnitine | Promote atherosclerosis | Promote | Clinical |
| Thrombosis | ||||
| Inflammation | ||||
| SCFAs | Dietary fiber | Anti-inflammatory | Protect | Animal |
| Improve vascular function | ||||
| BCAAs | Protein | Insulin resistance | Promote | Clinical (with animal support) |
| Lipid metabolism disorders | ||||
| LPS | Gram-negative bacteria | Pro-inflammatory | Promote | Clinical |
| Vascular damage | ||||
| BA | BA metabolism | Cholesterol metabolism | Promote | Clinical (with animal support) |
CAD, coronary artery disease; TMAO, Trimethylamine N-oxide; SCFAs, Short-chain fatty acids; BCAAs, Branched-Chain Amino Acids; LPS, Lipopolysaccharide; BA, Bile Acid.
The intestinal barrier plays a crucial role in maintaining homeostasis and shielding the body from pathogenic invasion. Its functional integrity relies on the synergistic action of tight junctions in intestinal epithelial cells, mucosal secretions, and the immune system. When the intestinal barrier is damaged, such as by dietary factors high in fat and sugar, persistent stress, or improper use of antibiotics, the barrier permeability can significantly increase, inducing chronic low-level inflammation [57]. Increasing evidence suggests an elevated intestinal permeability in patients with CAD and atherosclerosis. Intriguingly, the presence of gut-derived bacteria has been reported in human atherosclerotic plaques, indicating a role of bacterial translocation in the pathogenesis of atherosclerosis [58, 59]. In the pathological process of CAD, this low-level chronic inflammation exhibits a significant impact. Inflammation leads to intestinal dysfunction, and the damaged intestinal barrier allows bacteria and their metabolites (such as lipopolysaccharide, LPS) from the intestinal lumen to enter the bloodstream, activating a systemic immune response. This process accelerates the formation of atherosclerosis and plaque instability by increasing the production of pro-inflammatory cytokines and affecting the recruitment of white blood cells (Fig. 2). These are the main characteristics of CAD development [60, 61, 62].
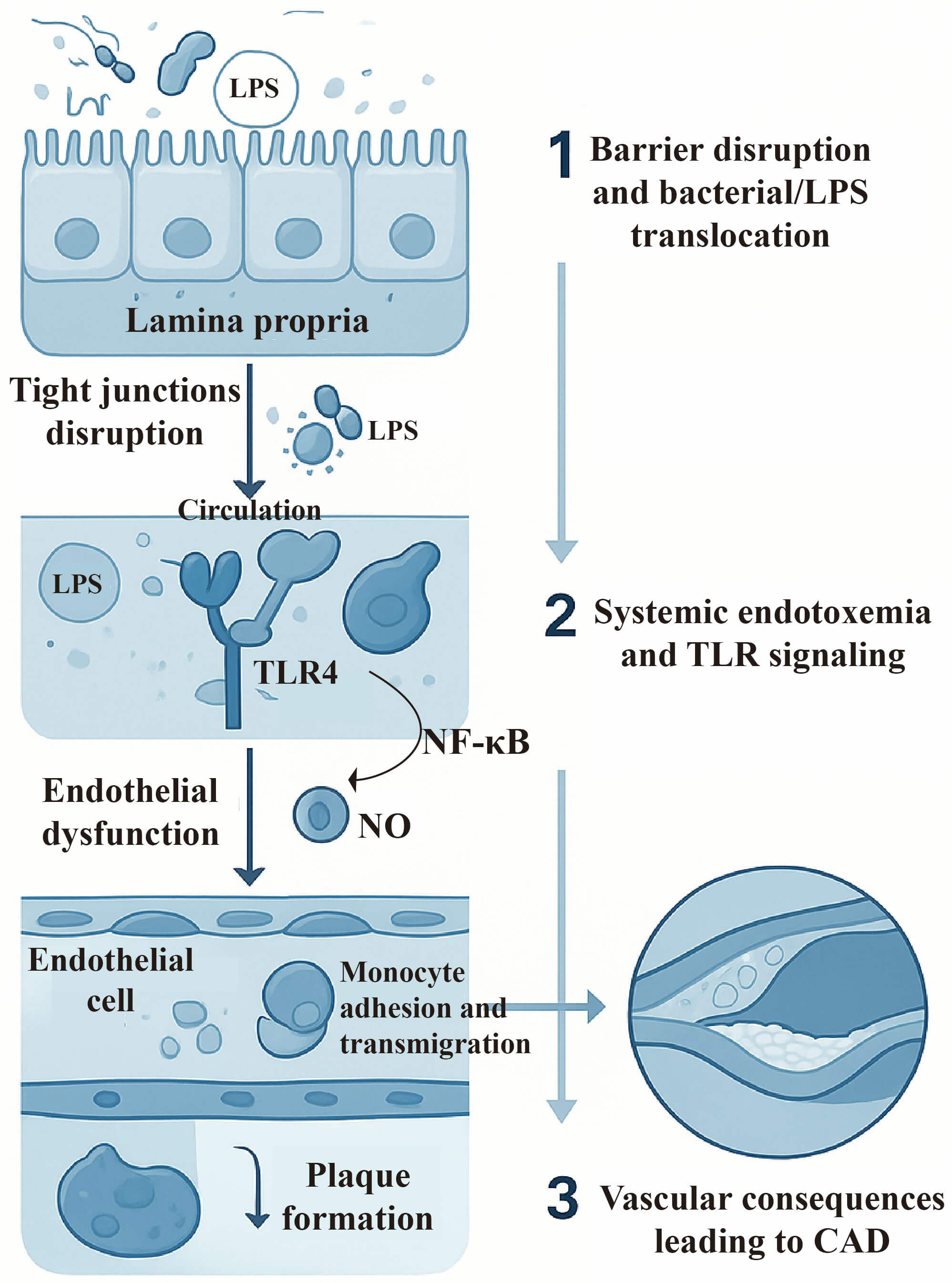 Fig. 2.
Fig. 2.
Gut barrier dysfunction contributing to the development
of coronary artery disease. The upper panel depicts a disrupted intestinal
epithelial barrier, allowing translocation of gut-derived bacteria and LPS into
the circulation. The middle section illustrates systemic endotoxemia,
characterized by the presence of LPS and microbial components in the bloodstream.
These microbial products activate inflammatory signaling pathways via receptors
such as Toll-like receptor (TLR) on vascular cells. The lower panel shows the
downstream vascular consequences, including endothelial dysfunction,
inflammation, and the formation of atherosclerotic plaques, ultimately leading to
coronary artery disease. LPS, lipopolysaccharide; TLR, toll-like receptor; NO,
nitric oxide; NF-
Lipopolysaccharide (LPS), a primary structural component of intestinal
Gram-negative bacteria, serves as a crucial mediator of gut-derived
proinflammatory signals. When intestinal barrier permeability increases, LPS
tends to disseminate through the bloodstream and activate immune cells
systemically. The proinflammatory effects of LPS are primarily achieved through
the TLR4 signaling pathway, leading to the activation of nuclear factor kappa B
(NF-
The application of probiotics and prebiotics in the treatment of CAD has become a research hotspot in recent years. Probiotics refer to live microorganisms that are beneficial to the health of the host, while prebiotics are substances that promote the growth of probiotics. Common probiotics include Lactobacillus, Bifidobacterium, Lactococcus, Streptococcus, and Enterococcus. Most prebiotics are carbohydrates found in natural products such as fruits, vegetables, and grains [69]. Their potential for CAD treatment is based on the theoretical foundation of the gut-heart axis, and is related to inflammation and lipid metabolism [70] (Fig. 3). A recent cross-sectional study has demonstrated that the consumption of probiotics is beneficial to cardiovascular health among patients with existing atherosclerotic disease [71]. Furthermore, a recent meta-analysis has also indicated that the addition of probiotics or synthetic bacteria to conventional medications for CAD can enhance patient outcomes [72]. In a randomized controlled trial, the combined supplementation of probiotics and inulin for 8 weeks among CAD subjects was found to exert beneficial effects on depression, anxiety, and inflammatory biomarkers. The addition of inulin to probiotic supplements proved more effective in improving psychological outcomes and inflammatory biomarkers compared to the separate administration of the two supplements [73]. In addition, several clinical studies have demonstrated the significant efficacy of specific probiotics as adjunctive therapy for CAD. According to Malik and colleagues, Lactobacillus plantarum 299v can enhance endothelial function and reduce systemic inflammation in CAD patients. Circulating gut-derived metabolites may underlie these improvements, warranting further investigation [74]. According to Sun et al.’s study [75], combining Bifidobacterium lactis Probio-M8 with conventional treatment provides additional benefits to CAD patients, compared to conventional treatment alone. Accumulating evidence suggests that combination therapies, such as probiotics and prebiotics, can modulate gut microbiota, thereby influencing the brain-gut axis or gut-heart axis. Despite limitations in the selection and validation of functional strains, numerous probiotic or prebiotic preparations are commercially available and exhibit good patient compliance, making them promising candidates for novel interventions in CAD. However, the evidence for probiotic or synbiotic supplementation for the treatment of CAD remains limited and highly heterogeneous. Most trials were single-center, short-term, and primarily focused on surrogate endpoints (e.g., lipid profiles, inflammatory markers, endothelial function, mood symptoms, or circulating TMAO) rather than clinical events. Reported benefits often varied by strain (e.g., Lactobacillus plantarum 299v; Bifidobacterium lactis Probio-M8), with small sample sizes, varying doses and durations, and inconsistent co-interventions [72, 74, 76]. Some studies demonstrated neutral or mixed effects of probiotic or synbiotic supplementation on TMAO or surrogate cardiometabolic markers, with substantial inter-study heterogeneity [77, 78]. External validity was further limited by differences in baseline microbiome, background diet, renal function (which has a significant impact on TMAO), and concomitant medications (e.g., statins, PPIs, antibiotics). Risk of bias considerations included unclear allocation concealment, incomplete blinding, per-protocol analysis, and selective reporting. In summary, current data suggest that specific strains may improve specific surrogate outcomes in specific populations, but the quality of evidence is low to very low (GRADE), and no conclusions about reductions in cardiovascular events can be drawn. Well-designed, adequately powered, preregistered, multicenter randomized controlled trials with standardized strain characteristics, dose, duration of treatment, dietary control, and adjudicated outcomes are needed before routine use can be recommended.
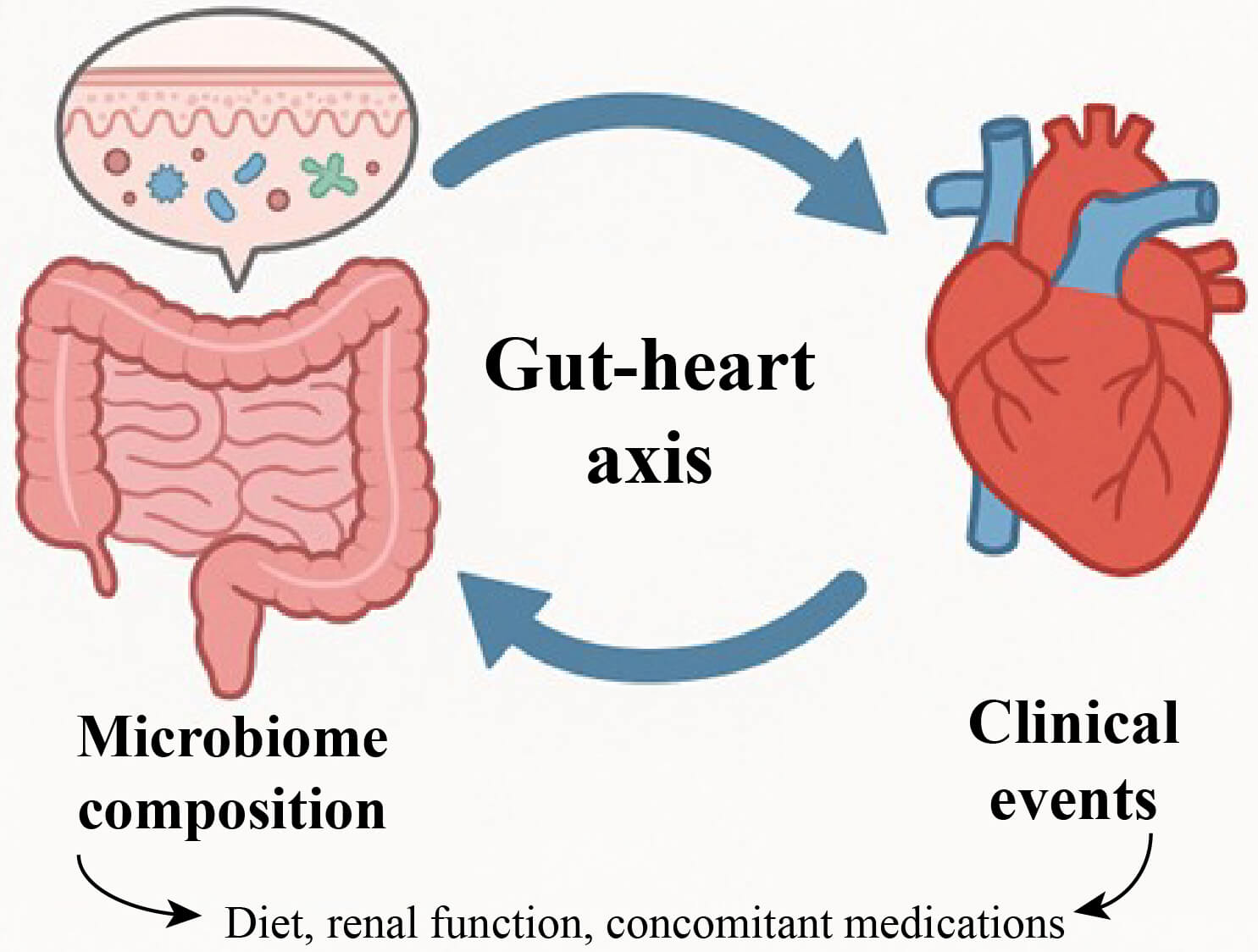 Fig. 3.
Fig. 3.
The gut-heart axis links gut microbes to cardiovascular disease.
Healthy dietary patterns have demonstrated a significant role in rebalancing gut
microbiota and reducing the risk of CAD. Some successful intervention strategies,
including the Mediterranean diet or cessation of red meat consumption, represent
more achievable methods to lower TMAO levels and potentially decrease
cardiovascular risk. These dietary approaches, characterized by rich dietary
fiber,
Fecal microbiota transplantation (FMT) currently stands as the most radical intervention targeting the gut microbiota. FMT represents an innovative therapy that restores intestinal microecological balance by transferring fecal microbiota from healthy donors to patients. Its application in the treatment of CAD reflects a cutting-edge approach in modulating the gut-heart axis [81]. The theoretical basis of FMT primarily derives from the close association between gut microbiota dysbiosis and imbalances in metabolism and inflammatory states. The goal is to restore a healthy microbial ecosystem, which aids in the reconstruction of the host’s intestinal mucosal barrier function, thereby reducing oxidative stress and vascular damage caused by microorganisms and their metabolites [82, 83]. ALW-II-41-27 is a specific inhibitor of the Ephrin type-A receptor 2 (EphA2) receptor, exhibiting anti-inflammatory properties. Lu et al. [84] employed fecal microbiota transplantation in their study. The results indicated that the feces of mice subjected to ALW-II-41-27 treatment showed a reduction in atherosclerotic plaques [84]. In the current study, FMT demonstrates a significant role in improving metabolic syndrome, regulating TMAO production, and modulating SCFAs levels. While this therapy has shown potential superiority in certain cardiovascular diseases, its specific mechanism of action and clinical application in the field of CAD are still in the exploratory phase. The main technical challenges include the complexity of donor selection (related to donor microbiota diversity and health status consistency), safety issues during feces processing and preservation, and the evaluation of long-term effectiveness after transplantation [85]. Furthermore, ethical controversies reflect variations in patient acceptability of FMT, particularly in aspects such as donor sources and privacy security, which require strict management. Hence, further standardizing FMT-related procedures, optimizing transplantation techniques, and conducting long-term tracking of treatment outcomes will constitute significant breakthroughs in this field in the future.
The introduction of gene editing technologies, such as CRISPR-Cas9, has provided revolutionary insights for the treatment of CAD by directly modifying microbial genomes or host genes. For instance, by targeting and controlling the expression of specific bacterial genes related to TMA production, it is possible to effectively inhibit the generation of TMAO and indirectly reduce the risk of atherosclerosis [4, 86]. Despite ethical and long-term effectiveness concerns, these technologies’ multi-level intervention models have pointed the way forward for precision medicine in CAD. The interdisciplinary integration of bioinformatics, synthetic biology, and metabolomics techniques will provide novel tools for optimizing disease intervention strategies.
Microbiome-targeted interventions face non-trivial practical and ethical barriers. For fecal microbiota transplantation (FMT), recent FDA safety communications highlight rare but serious donor-to-recipient transmission of multidrug-resistant organisms and the need for stringent donor screening (including SARS-CoV-2 testing), which has tightened the regulatory environment and limited the former policy of enforcement discretion to narrow circumstances [87]. Manufacturing, traceability, and quality control also shift FMT and next-generation consortia into the “live biotherapeutic product” framework, with associated chemistry, manufacturing and controls requirements that complicate scaling and global trials [88]. For in situ microbiome gene editing (e.g., phage- or particle-delivered CRISPR/base editors), off-target effects, payload containment, horizontal gene transfer, ecological disturbance, and long-term reversibility remain unresolved; current evidence is preclinical (mouse) or early translational [89]. Ethically, informed consent must address uncertain long-term risks, potential community-level externalities, and data governance around metagenomes; operationally, centers require biosafety level-appropriate handling, donor/strain registries, and post-marketing safety surveillance [90]. Table 2 (Ref. [89, 91, 92, 93, 94, 95, 96, 97, 98]) summarizes microbiome-directed interventions relevant to cardiometabolic risk, including fecal microbiota transplantation (FMT), probiotics, prebiotics, next-generation microbes, enzyme inhibition of TMA lyase pathways, and early in situ microbiome gene-editing approaches.
| Modality | Primary endpoint | Result | Notes/Limitations | Reference |
| FMT (lean |
Peripheral insulin sensitivity at 6 weeks | Positive ( |
Small, short follow-up; allogenic vs. autologous; no CV outcomes. | [91] |
| FMT responder-dependence | Insulin sensitivity; bile acid/metabolic readouts | Heterogeneous; benefit depends on baseline microbiome | Generalizability limited; durability uncertain. | [92] |
| Probiotic (L. reuteri NCIMB 30242) | LDL-C change | Positive (LDL-C |
Surrogate outcome; strain-specific; not hard CV events. | [93] |
| Probiotic (various) on TMAO | Circulating TMAO | Negative/Neutral overall | Small samples; heterogeneity high. | [94] |
| Probiotic (L. plantarum GLP3) | Plasma TMAO | Positive (TMAO |
Single study; journal tier modest; replication needed. | [95] |
| Prebiotic (inulin) | Fasting & postprandial TMAO | Negative (no reduction) | Short duration; specific fiber type; no CV endpoints. | [96] |
| Next-gen microbe (Akkermansia) | Safety; insulin resistance; metabolic markers | Positive on metabolic markers | Pilot; no CV endpoints; effects may depend on baseline abundance. | [97] |
| Enzyme inhibition (TMA lyase pathway) | Weight gain/metabolic disturbance | Positive mechanistic signal | No human CV data; translation pending. | [98] |
| In situ microbiome gene editing | On-target editing; strain control | Feasibility shown | Early-stage; safety/containment/regulation unresolved in humans. | [89] |
At the pharmacokinetic–pharmacodynamic interface, the gut microbiome can transform many cardiovascular agents, altering exposure and response. Large systematic screens show that dozens of commonly used oral drugs are chemically modified by gut bacteria, which helps explain inter-individual variability [99]. Specific Eggerthella lenta strains harbor a cardiac-glycoside reductase (cgr) operon that inactivates digoxin to dihydrodigoxin, reducing efficacy; this phenotype depends on the presence of the cgr genes and is inhibited by arginine [100]. Dietary nitrate relies on oral nitrate-reducing bacteria to generate bioactive nitrite/NO; antiseptic mouthwashes that suppress these bacteria blunt nitrite bioavailability and can raise blood pressure in controlled studies—an effect relevant to patients on BP-lowering regimens [101]. The gut-derived metabolite TMAO enhances platelet reactivity and thrombosis potential; mechanistically, choline/TMAO can also impair clopidogrel bioactivation via NOX-ROS/Nrf2-CES1 signaling, offering one explanation for variable antiplatelet response [102]. Antibiotic perturbation of gut flora reduces bacterial vitamin-K synthesis and is a recognized cause of INR elevation; patient-level data further link gut taxa (e.g., prevotella, Eubacterium) and vitamin-K status with warfarin dose/response variability [103]. A growing body of work shows that gut and oral microbes can directly biotransform cardiovascular agents or indirectly modulate host drug-metabolizing pathways, thereby shaping inter-individual variability in exposure, efficacy, and toxicity.
The concept of precision medicine emphasizes personalized diagnosis and treatment for patients, and this approach holds significant potential in the study of gut microbiota and CAD. The microbiota of each patient is unique, and this diversity directly determines metabolic characteristics and treatment outcomes in pathological processes. Therefore, the introduction of high-throughput sequencing technology for the microbiota is crucial to precisely analyze the composition of microbial species and functional genes in patients’ intestines. By integrating metabolomic data, key metabolic pathways leading to disease progression can be identified, enabling targeted intervention strategies. For instance, patients with significantly elevated TMAO levels can benefit from supplementation with specific probiotics that inhibit the production of trimethylamine, a precursor substance, thereby indirectly reducing cardiovascular risk. Furthermore, the design of clinical trials is particularly important at this stage. A preferred approach is to adopt a multicenter randomized controlled design to ensure the broad applicability of the results. In terms of intervention measures, combinations of probiotics, prebiotics, and other functional foods can be adjusted based on patients’ microbial profiles. Therapeutic efficacy indicators should be specific and clear, utilizing measurements such as inflammatory biomarker levels, dynamic changes in metabolites, and cardiovascular function assessments to comprehensively evaluate the effects of intervention [104].
The rapid advancement of artificial intelligence (AI) technology is reshaping the field of medical research, particularly in the broad application prospects for CAD risk assessment. By integrating AI algorithms, such as deep learning and random forests, data from various research sources can be effectively combined to generate high-precision predictive models. The initial step in model construction involves data preparation, encompassing high-quality microbiome sequencing data, metabolomic indicators, and relevant clinical features. During the data processing phase, algorithmic optimization and feature selection are employed to reduce redundant data and enhance predictive performance. Through model training and validation, patterns of correlation between various microbial characteristics and CAD risk are untangled. Additionally, AI technology enables dynamic model updates to accommodate the incorporation of multiple new detection indicators or risk factors. In practical applications, these models could potentially serve as preclinical screening tools for identifying high-risk populations. Furthermore, the precise risk assessment outputs can guide the development of interventional treatment plans, facilitating a seamless transition from disease prediction to intervention [105].
Conducting high-quality gut microbiome research relies on the standardization of sampling procedures and the consistency of data analysis workflows. Currently, the diversity in sampling methods, storage conditions, and data analysis tools directly poses challenges to the reproducibility of research findings. The promotion of standardized operational procedures has become one of the core issues urgently needing to be addressed in this field. Researchers should drive the establishment of internationally unified sampling guidelines, including sample collection time, methods, and preservation conditions. In terms of data analysis, establishing unified quality control standards and adopting analysis tools with strong platform compatibility will further enhance the comparability between different studies and the convenience of data sharing. The study of the gut microbiome and CAD is an interdisciplinary field involving microbiology, immunology, metabolomics, and cardiovascular disease. Future progress requires strengthening interdisciplinary collaboration to integrate research capabilities in these areas.
The gut microbiome has emerged as a modifiable layer of CAD pathobiology and treatment response. Converging evidence links microbially derived metabolites (e.g., TMAO, bile acids, SCFAs) with atherosclerosis, while interventional strategies—dietary pattern shifts, pre/pro/postbiotics, targeted enzyme inhibition, bile-acid modulation, and selected bacteriotherapies—show promise but remain variably supported across endpoints and populations. Heterogeneity is the rule: microbiome profiles and metabolite baselines differ by geography, ethnicity, and diet, and common cardiovascular drugs exhibit bidirectional interactions with the microbiome (e.g., digoxin inactivation, nitrate–nitrite–NO dependence on oral taxa, TMAO–clopidogrel dynamics, vitamin-K/warfarin, statin response variability). These realities underscore the need to report and stratify by region, diet, and ancestry, and to account for concomitant medications in both trials and practice. A near-term research agenda should prioritize multicenter randomized studies with harmonized phenotyping, robust dietary assessment, and standardized metabolomic readouts; integration of metagenomics with host genomics, immunometabolism, and plaque imaging; and prospective validation of microbiome-informed risk scores and “treat-to-metabolite” targets. Safety, durability, manufacturing quality (for live biotherapeutics), and regulatory and ethical considerations require equal attention. If executed with careful population context and medication review, microbiome-guided strategies can move CAD care from association to action—advancing precision prevention and therapy rather than offering one-size-fits-all solutions.
Each author made significant contributions to the manuscript. PPW, LQD, and YY made substantial contributions to the conception and framework of the review, conducted targeted literature acquisition and screening, and participated in the critical interpretation and synthesis of the evidence that informed the manuscript’s key sections and conclusions. PPW and LQD drafted the initial version. YZ and ZKL provided substantial revisions and additional content. ZKL and YZ were responsible for reference collection and the design of figures and tables. YY critically reviewed the manuscript and offered valuable feedback. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



