1 Department of Nephrology, Shanghai Changzheng Hospital (Second Affiliated Hospital of Naval Medical University), 200003 Shanghai, China
2 Department of Nephrology, Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, 201203 Shanghai, China
3 TCM Institute of Kidney Disease of Shanghai University of Traditional Chinese Medicine, 201203 Shanghai, China
4 Key Laboratory of Liver and Kidney Diseases, Ministry of Education, Shanghai Key Laboratory of Traditional Chinese Clinical Medicine, 201203 Shanghai, China
5 Department of Nephrology, The 960th Hospital of the PLA Joint Logistics Support Force, 250031 Jinan, Shandong, China
6 Department of Nephrology, Shuguang Hospital Affiliated Baoshan Branch, Shanghai University of Traditional Chinese Medicine, 201901 Shanghai, China
7 Department of Nephrology, Affiliated Nantong Hospital of Shanghai University (The Sixth People’s Hospital of Nantong), 226000 Nantong, Jiangsu, China
†These authors contributed equally.
Abstract
Recent studies have identified impaired renal gluconeogenesis as a hallmark of chronic kidney disease. Triptolide is a natural compound widely used in China for the treatment of renal diseases. This study investigated whether triptolide mitigates renal fibrosis by promoting renal gluconeogenesis.
Renal fibrosis was induced in vivo by unilateral ureteral obstruction (UUO) surgery in mice. Transforming growth factor-β (TGF-β)-stimulated human kidney-2 (HK-2) cells were used as an in vitro model to investigate renal fibrosis. Metabolomics, western blotting, immunohistochemistry (IHC), and metabolic assays were performed to investigate the underlying mechanisms.
Triptolide reduced the expression of several fibrotic markers in the kidneys of UUO mice. Metabolomic analysis revealed enhanced renal gluconeogenesis following treatment with triptolide, which was confirmed by analyzing gluconeogenic enzyme expression and lactate concentration in UUO kidneys. The pro-gluconeogenic effect of triptolide was further confirmed in TGF-β-stimulated HK2 cells. Inhibition of phosphoenolpyruvate carboxykinase 1 (PCK1) reversed the anti-fibrotic and pro-gluconeogenic effects of triptolide in TGF-β-stimulated HK2 cells. We further demonstrated that peroxisome proliferator-activated receptor-gamma co-activator 1 alpha (PGC1α) expression was downregulated in TGF-β-stimulated HK2 cells and UUO kidneys, and that triptolide reversed this downregulation. Moreover, the PGC1α inhibitor reversed the effect of triptolide on PCK1 expression and glucose metabolism. Finally, IHC analysis revealed that triptolide inhibited histone lactylation in UUO kidneys, which was associated with a decreased production of inflammatory factors and reduced macrophage infiltration.
Triptolide may inhibit renal fibrosis by increasing the PGC1α/PCK1 axis, thereby promoting renal gluconeogenesis. This cascade may reduce histone lactylation and renal inflammation, providing a mechanistic pathway for its anti-fibrotic effect.
Keywords
- triptolide
- gluconeogenesis
- fibrosis
- lactic acid
- kidney
Chronic kidney disease (CKD) is a significant public health concern, affecting
more than 10% of the global population [1]. Renal fibrosis is a common
pathological pathway and key manifestation of CKD progression [2]. It is
characterized by the excessive deposition of extracellular matrix (ECM) in the
kidney, such as fibronectin and collagen-I in renal interstitial regions [3].
Furthermore, the upregulation of epithelial-mesenchymal transition (EMT) markers
and activation of the transforming growth factor-beta (TGF-
Impaired renal gluconeogenesis has recently been identified as a key feature of acute and chronic kidney diseases [5, 6, 7]. Clinically, this impairment is associated with poor renal prognosis and increased mortality in patients with acute or chronic kidney dysfunction [5, 7]. Overexpression of tubular phosphoenolpyruvate carboxykinase 1 (PCK1), an important enzyme involved in renal gluconeogenesis, inhibits disease progression in an animal model of CKD [8]. These findings suggest that improving renal gluconeogenesis is a novel therapeutic strategy to halt CKD progression.
Given the critical role of gluconeogenesis in various disease states, the
regulatory mechanisms governing this process have been the focus of extensive
research [9, 10]. Peroxisome proliferator-activated receptor-gamma co-activator 1
alpha (PGC1
Triptolide, a diterpenoid trioxide isolated from the medicinal plant Tripterygium wilfordii Hook F [13, 14], has been demonstrated to confer renoprotective effects through various mechanisms, including anti-fibrotic, anti-inflammatory, and immunosuppressive actions [15]. Clinically, triptolide-containing formulations have been used to treat human renal diseases such as nephritis, minimal change disease, and membranous nephropathy [16, 17]. Preclinical studies in animal models further substantiate its advantages. Triptolide inhibits renal fibroblast activation, attenuates renal tubular EMT, and reduces collagen deposition in the renal interstitium [16]. However, the complete spectrum of mechanisms underlying triptolide-mediated renoprotection remains unclear. Notably, recent studies have reported that triptolide improves glucose metabolism in intrahepatic cholangiocarcinoma and rheumatoid arthritis models [18, 19], prompting the question of whether it exerts similar effects on glucose metabolism in CKD, a prospect that remains unexplored.
In this study, we aimed to evaluate the hypothesis that triptolide inhibits renal fibrosis by improving renal gluconeogenesis.
Wild-type male C57BL/6 mice (body weight 20–25 g) were obtained from Shanghai SLAC Laboratory Animal Co., Ltd., and housed in a specific pathogen-free-grade animal facility in the Shanghai University of Traditional Chinese Medicine under the local regulations. Shanghai University of Traditional Chinese Medicine has approved the animal experiments (PZSHUTCM18111601).
Mice were anesthetized with pentobarbital sodium (P3761; Sigma-Aldrich Co., St. Louis, MO, USA) at a dose of 100 mg/kg via intraperitoneal (i.p.) injection before surgery. The UUO surgery was performed by ligating the left ureter twice using 4-0 nylon sutures. Mice were randomly divided into four groups: (1) Sham/vehicle (n = 6), (2) Sham/triptolide (n = 7), (3) UUO/vehicle (n = 7), and (4) UUO/triptolide (n = 7). Triptolide (Topscience, T2179, Shanghai, China) was dissolved in dimethylsulfoxide (DMSO) and diluted with normal saline to prepare a working solution containing 1% DMSO. Sham or UUO mice were treated with vehicle or 0.25 mg/kg triptolide daily by i.p. injection for 10 days starting from day 0. The triptolide dosage (0.25 mg/kg/day) was selected based on a previous study that demonstrated its protective effect against renal disease in mice [20]. Mice were sacrificed on day 10. Mice were euthanized by cervical dislocation under anesthesia with 100 mg/kg pentobarbital sodium at the experimental endpoint for kidney collection.
Renal proximal tubular epithelial (HK2) cells were obtained from the Cell Bank
of Shanghai Institute of Biological Sciences (Chinese Academy of Science, Cat.
BFN60700259). The cell line used in this study was identified by short tandem
repeat profiling and tested negative for mycoplasma. HK2 cells were seeded in
6-well plates to 40%–50% confluence and starved overnight with DMEM/F12 medium
(GNM12400, GENOM, China) containing 0.5% fetal bovine serum. The following day,
the medium was replaced with fresh medium containing 0.5% fetal bovine serum
(04-001-1ACS, Biological Industries, Israel), and the cells were exposed to 2.5
ng/mL TGF-
Mouse kidneys were sliced, fixed, and embedded in paraffin, and cut into 4 µm-thick sections. Paraffin-embedded kidney sections were stained with hematoxylin, followed by ponceau red liquid dye acid complex, and then incubated in phosphomolybdic acid solution. Finally, the tissues were stained with aniline blue liquid and acetic acid. Images were acquired using a microscope (Nikon 80i, Tokyo, Japan).
Cell or kidney proteins were extracted using lysis buffer (P0013; Beyotime
Biotech, Nantong, Jiangsu, China). The BCA Protein Assay Kit (P0012S, Beyotime Biotech,
Nantong, Jiangsu, China) was used to quantify the protein concentration. Protein samples
were dissolved in 5
The renal cortex was collected for protein extraction. Protein samples were
isolated using 10
The supernatant was collected from HK2 cells and centrifuged to remove cell debris. Glucose and lactate concentrations were determined using commercial kits, as described previously.
Paraffin-embedded tissue sections were dewaxed and immersed in a retrieval
vessel containing citrate-based antigen retrieval buffer (pH 6.0, RC03; Shanghai
Huilan Biotech) for high-pressure antigen retrieval. Endogenous peroxidase
activity was blocked with 3% hydrogen peroxide. The sections were incubated
overnight at 4 °C with primary antibodies: Histone H4 (Lys12)
(PTM-1411RM, PTM bio, Hangzhou, Zhejiang, China, 1:100), F4/80 (HA721745, HUABIO,
Hangzhou, Zhejiang, China, 1:100), or interleukin-1 beta (IL-1
Non-targeted metabolomics analysis was performed by Beijing Biomarker
Technologies Co., Ltd. (Beijing, China) on six mouse kidney samples using
LC-QTOF. Metabolites were extracted and analyzed, and the data were processed
using Progenesis QI for peak alignment and identification against METLIN, HMDB,
LIPID MAPS, and in-house databases. The quality control sample reproducibility
was high (correlation
The results are presented as mean
The effect of triptolide on renal tubulointerstitial fibrosis was assessed using a UUO mouse model. Masson staining revealed extensive interstitial collagen deposition in the kidneys of UUO mice at 10 days postoperation. Notably, triptolide therapy significantly reduced collagen deposition (Fig. 1A,B). Western blotting analysis further demonstrated that ECM markers, including collagen-I and fibronectin, were upregulated in UUO kidneys, whereas triptolide treatment significantly decreased their levels (Fig. 1C,D). Importantly, triptolide treatment did not significantly affect the expression of these ECM markers in Sham-operated kidneys (Fig. 1C,D).
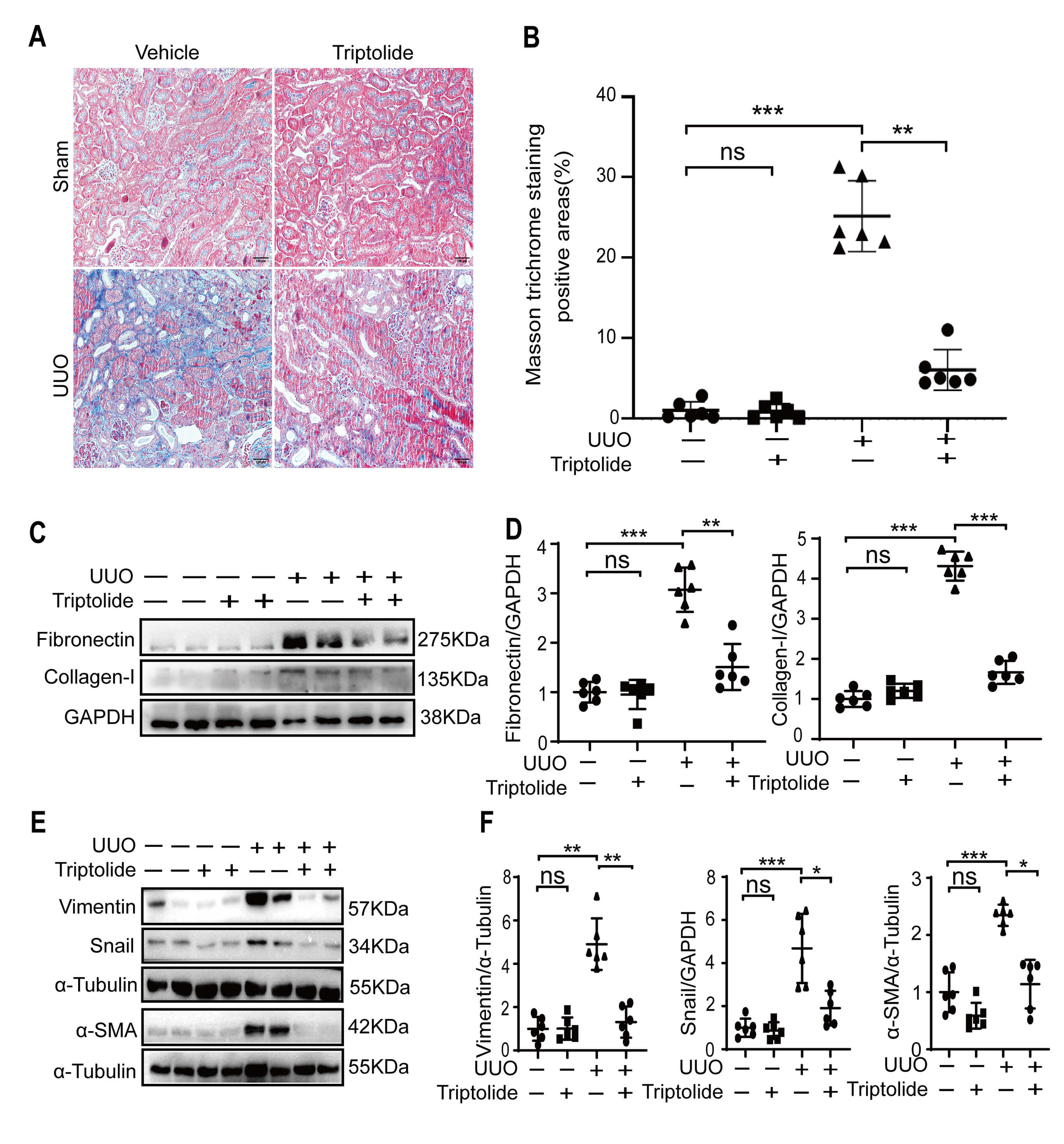 Fig. 1.
Fig. 1.
Triptolide inhibits tubulointerstitial fibrosis in obstructive
mouse kidneys and renal cells. Sham (-) or UUO (+) operation was performed on
wild-type C57BL/6 mice, followed by 10 days of treatment with DMSO or triptolide.
(A,B) Renal fibrosis was assessed using Masson’s trichrome staining and
quantified. Scale bar = 100 µm. (C,D) Expression
levels of fibronectin and collagen-I were analyzed using Western blotting and
quantified. Data are presented as mean
Furthermore, we evaluated the effect of triptolide on EMT in the fibrotic
kidneys. The expression levels of three key EMT markers, vimentin,
Collectively, these findings demonstrate that triptolide exhibits anti-fibrotic effects in mice with obstructive kidney injury.
To explore the mechanism underlying the renoprotective effects of triptolide in the fibrotic kidneys, we performed untargeted metabolomics analysis. A total of 260 differentially expressed metabolites (comprising 85 upregulated and 175 downregulated) were identified between the kidneys of UUO mice treated with vehicle and those treated with triptolide (Fig. 2A). KEGG enrichment analysis revealed that glucose metabolism, including gluconeogenesis and glycolysis, was among the most significantly dysregulated pathways affected by triptolide (Fig. 2B).
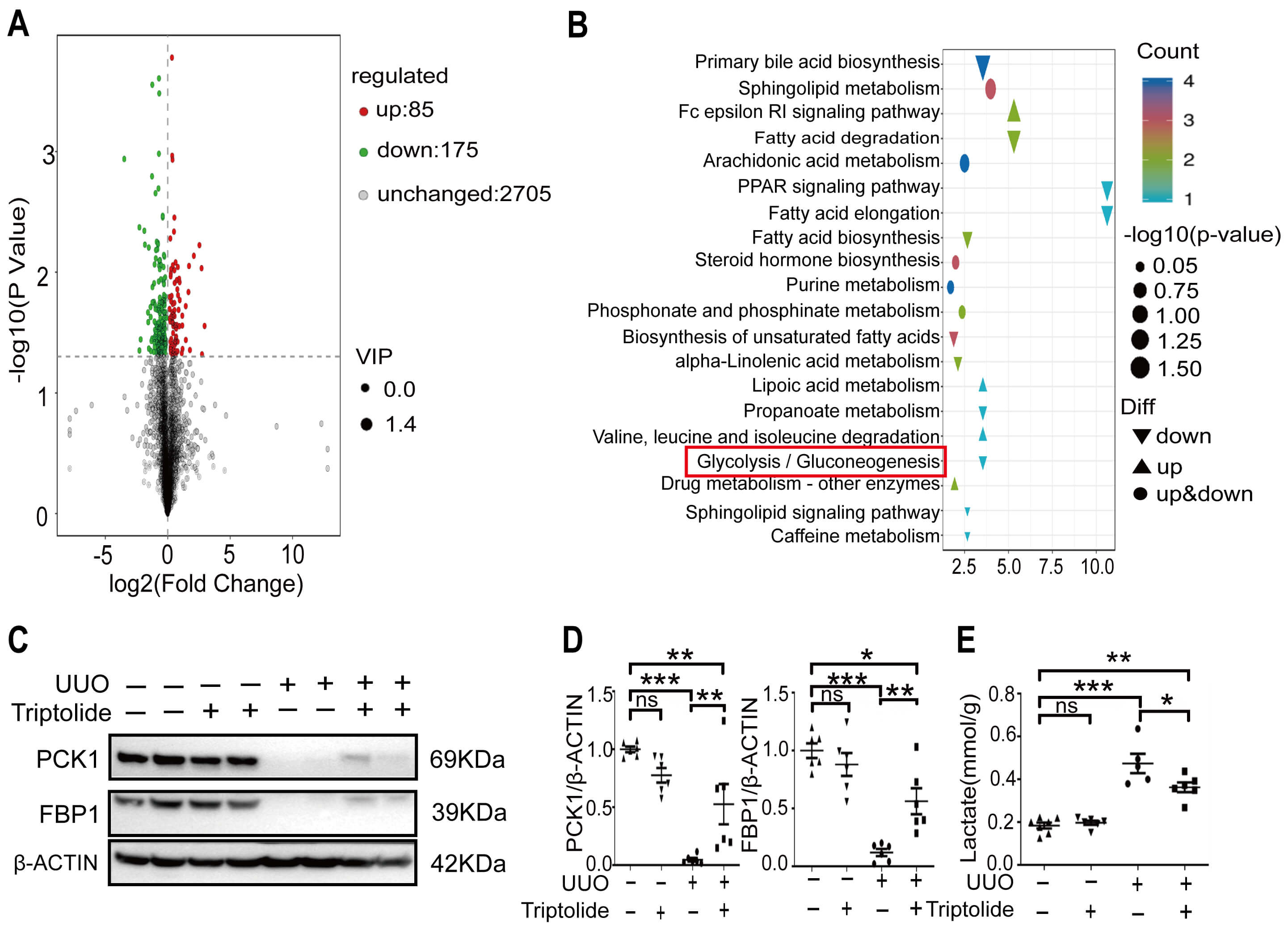 Fig. 2.
Fig. 2.
Renal gluconeogenesis is promoted by triptolide in
fibrotic kidneys. (A) Volcano plots of differentially expressed metabolites in
mouse kidneys between UUO/DMSO and UUO/triptolide groups based on untargeted
metabolomics analysis. (B) KEGG pathway enrichment analysis of
triptolide-targeted metabolites by untargeted metabolomics. (C,D) Expression levels of PCK1 and FBP1 in Sham (-) or UUO (+) kidneys were analyzed
using Western blotting and quantified. (E) Lactate levels in whole kidney samples
were determined. Data from at least three independent experiments are presented
as one sample. Data are presented as
mean
Subsequently, we validated the effects of triptolide on renal gluconeogenesis in fibrotic kidneys. The expression levels of PCK1 and FBP1, two rate-limiting enzymes involved in renal gluconeogenesis, were significantly downregulated in UUO kidneys compared to those in Sham-operated kidneys (Fig. 2C,D). Notably, triptolide treatment significantly increased the expression of these two enzymes in UUO kidneys; however, it failed to fully restore PCK1 and FBP1 expression in UUO kidneys to the levels observed in Sham-operated kidneys (Fig. 2C,D). Importantly, triptolide exhibited no significant effect on the expression of these two enzymes in Sham-operated kidneys (Fig. 2C,D).
Furthermore, renal lactate concentration was higher in UUO kidneys than in Sham kidneys (Fig. 2E), and triptolide treatment reduced lactate levels in UUO kidneys (Fig. 2E). However, triptolide did not completely reverse the UUO-induced increase in renal lactate to the baseline levels of Sham-operated kidneys (Fig. 2E). No significant difference in renal glucose concentration was observed between Sham and UUO kidneys (data not presented).
Collectively, these findings demonstrate that triptolide promotes renal gluconeogenesis in the fibrotic kidneys.
The anti-fibrotic effect of triptolide was validated in vitro using
TGF-
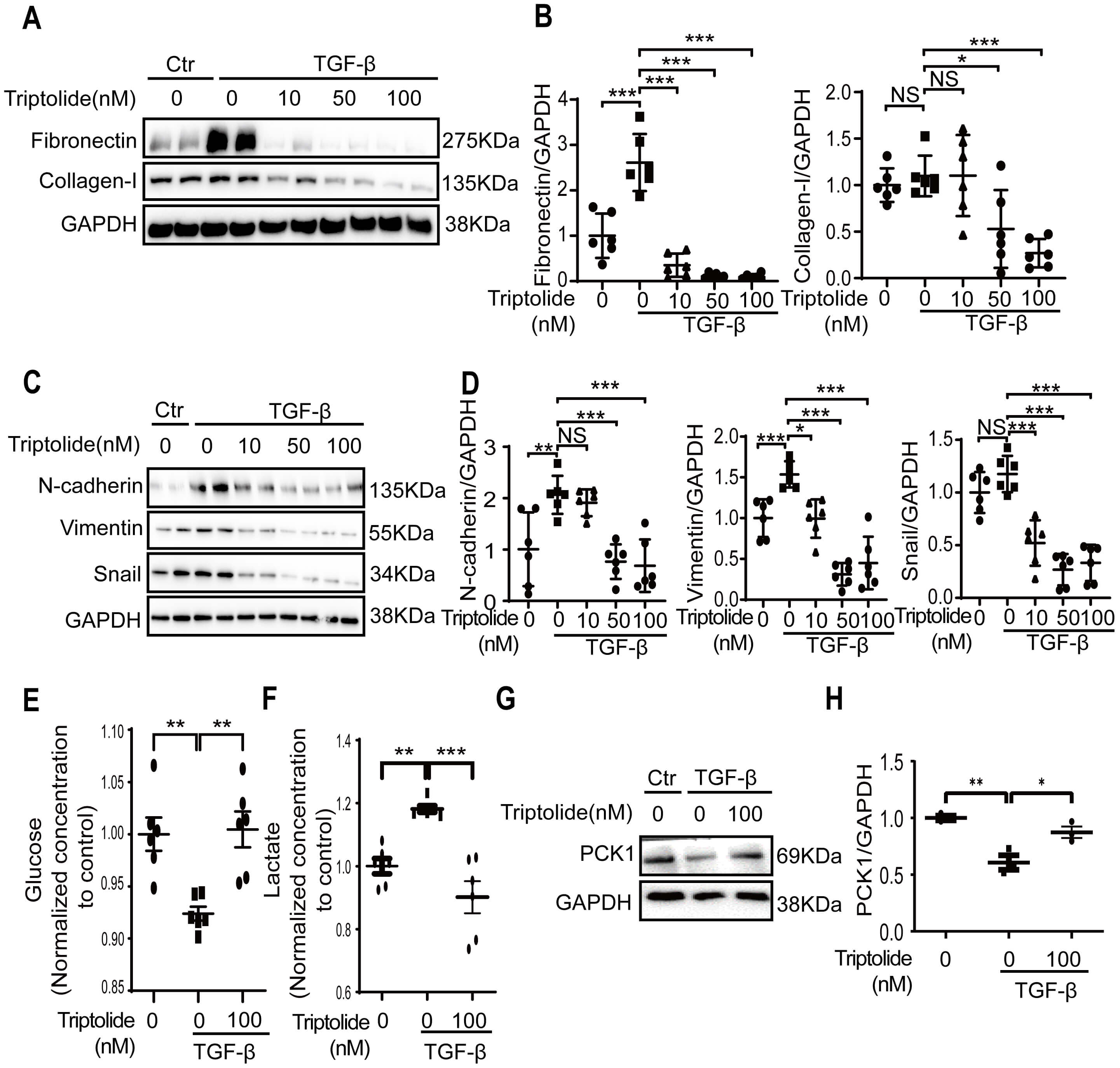 Fig. 3.
Fig. 3.
Triptolide prevents fibrotic changes and promotes glucose
metabolism in TGF-
We further observed that TGF-
To determine whether the anti-fibrotic effects of triptolide depend on
gluconeogenesis, we treated TGF-
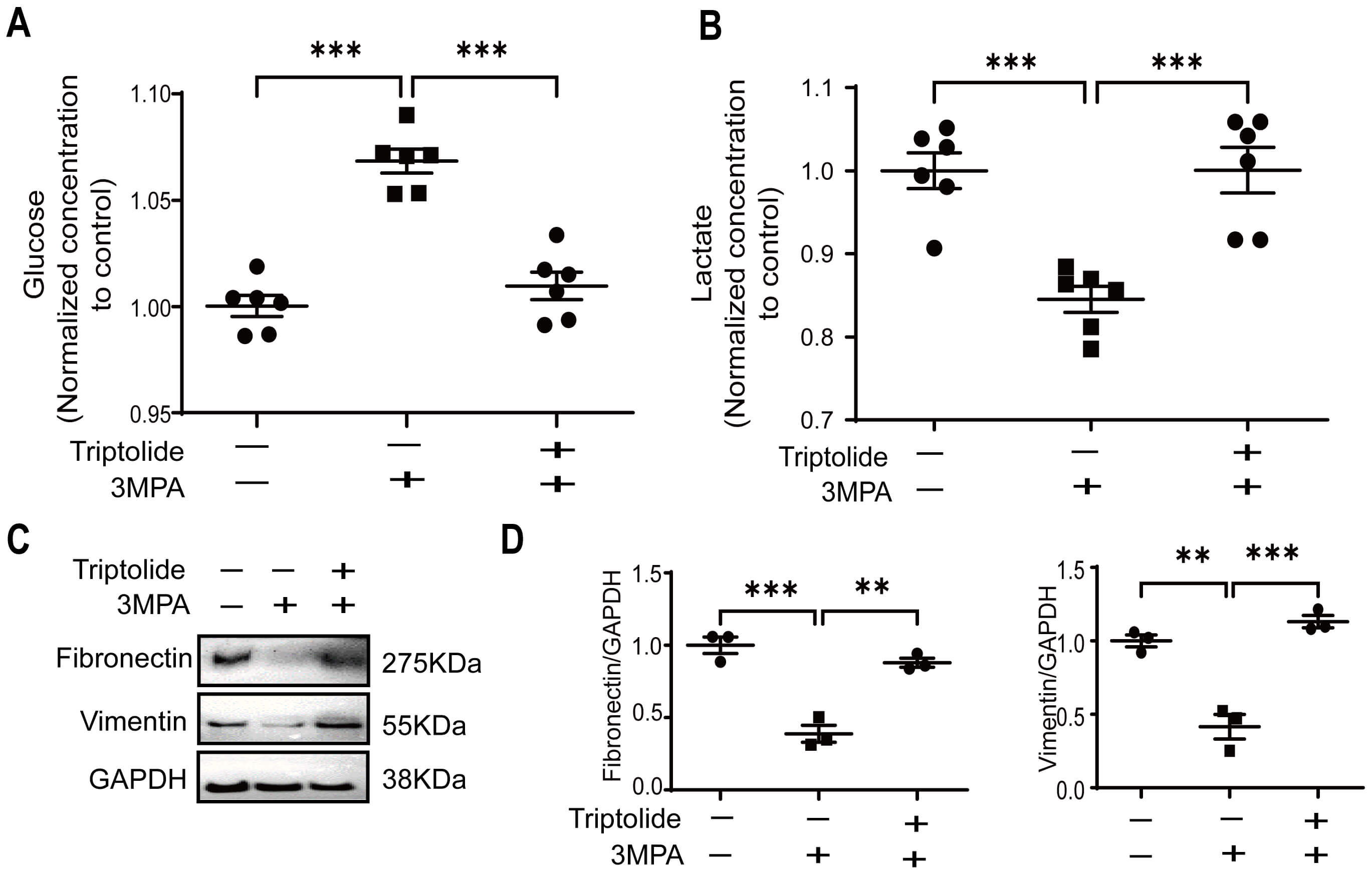 Fig. 4.
Fig. 4.
Triptolide inhibits renal fibrosis by promoting
gluconeogenesis. (A,B) Glucose and lactate levels in HK2 cells treated with (+)
or without (-) 1 mM PCK1 inhibitor 3MPA were measured. (C,D) Western blotting was
performed to evaluate the expression of fibronectin and vimentin in HK2 cells
treated with (+) or without (-) the PCK1 inhibitor 3MPA and triptolide. The
results were quantified. Data are presented as mean
We subsequently investigated the expression levels of PGC1
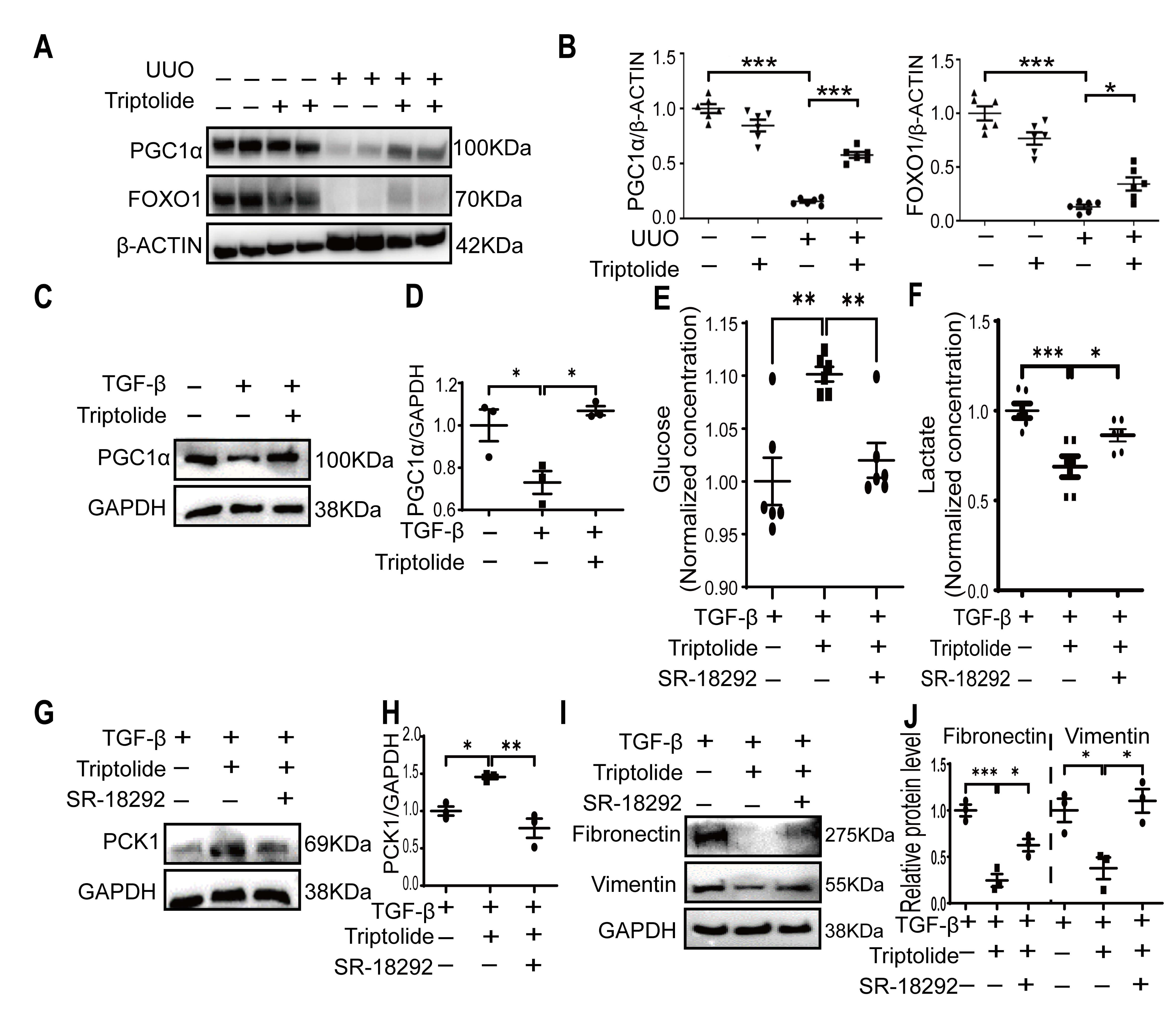 Fig. 5.
Fig. 5.
Triptolide promotes glucose metabolism through PGC1
Collectively, these findings suggest that PGC1
A recent study has demonstrated that lactate, previously considered only a metabolic waste product, promotes renal fibrosis by inducing H4K12 histone lactylation-mediated inflammatory responses in tubular cells [22]. Therefore, we investigated whether triptolide inhibits renal fibrosis by suppressing histone lactylation-mediated inflammation, a mechanism that lies further downstream of gluconeogenesis.
IHC staining revealed elevated H4K12 lactylation in the renal tubular cells of
fibrotic kidneys (Fig. 6A,B). This elevation was associated with increased
macrophage infiltration, as indicated by F4/80 staining (Fig. 6C,D), and
upregulated the expression of inflammatory factors such as IL-1
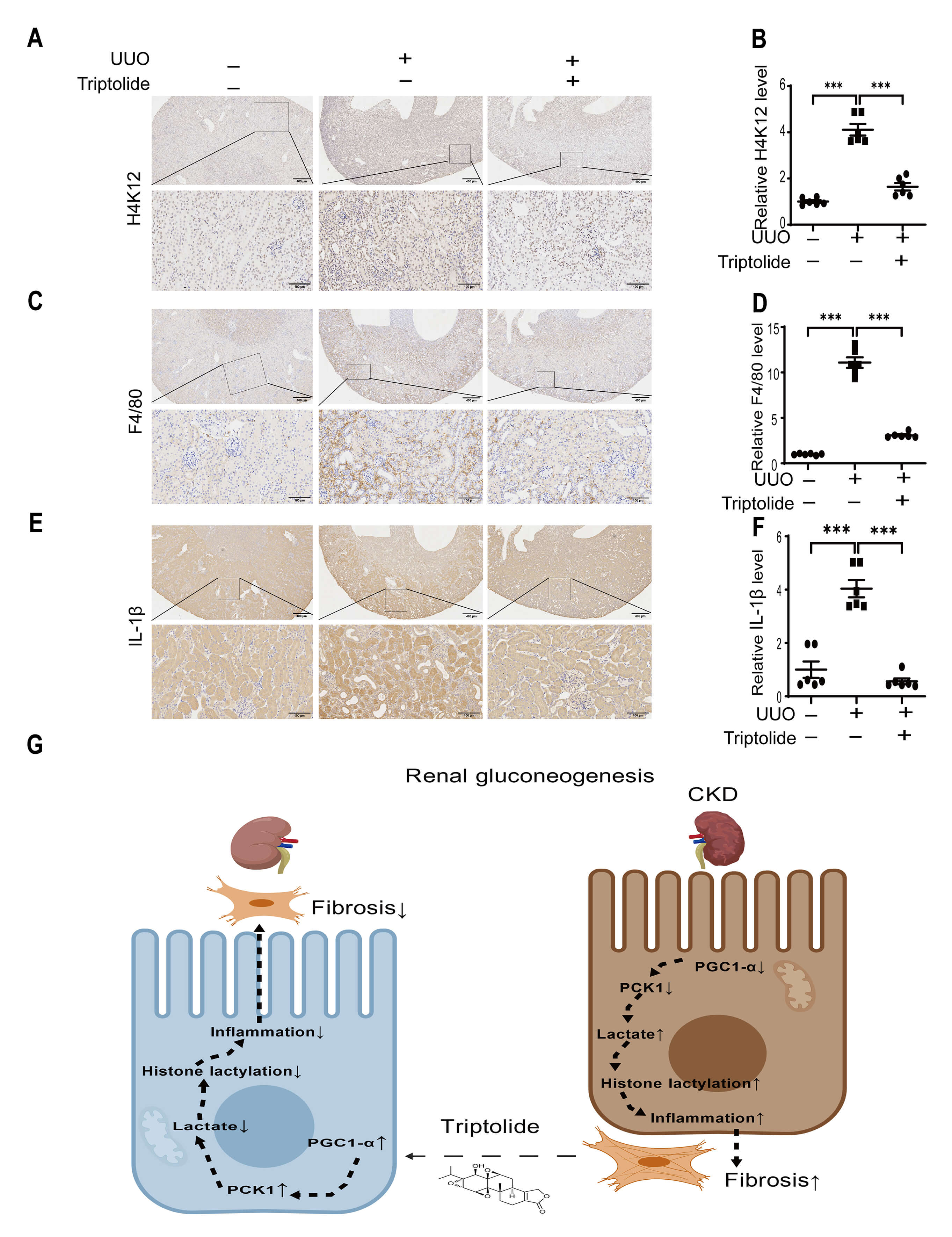 Fig. 6.
Fig. 6.
Triptolide inhibits histone lactylation and inflammatory
responses in UUO kidneys. (A,B) ICH analysis and quantification of
H4K12 in sham (-) or UUO (+) mouse kidneys with (+) or without (-) triptolide
treatment. Scale bar = 400 µm or 100 µm. (C,D) ICH analysis and quantification of F4/80 in the kidneys of triptolide-treated
UUO mice. Scale bar = 400 µm or 100 µm. (E,F) ICH
analysis and quantification of IL-1
These findings imply that the anti-fibrotic effect of triptolide involves the suppression of histone lactylation, a pathway that contributes to inflammation in fibrotic kidneys.
Formulations containing triptolide are widely used in China to treat patients with CKD [23]. In this study, we elucidated a novel mechanism underlying the renoprotective effect of triptolide, which inhibits renal fibrosis by promoting renal gluconeogenesis. First, we confirmed the anti-fibrotic effect of triptolide in UUO mice. Metabolomics analysis revealed that glucose metabolism is a downstream pathway mediating the anti-fibrotic effects of triptolide in UUO kidneys. This finding was further corroborated by analyses of gluconeogenic enzyme expression and metabolite levels. Furthermore, triptolide enhanced the expression of gluconeogenic enzymes and improved glucose metabolism in a cellular model of renal fibrosis. Moreover, in vitro studies demonstrated that the inhibition of gluconeogenesis by a PCK1 inhibitor reversed the anti-fibrotic effect of triptolide.
This study has several significant advancements in the current understanding of the mechanism of action of triptolide and the pathogenesis of renal fibrosis. First, it reveals a previously unrecognized role of triptolide in regulating renal glucose metabolism, particularly promoting gluconeogenesis, which differs fundamentally from its previously reported metabolic effects in other diseases [18, 19]. Previous studies have demonstrated that triptolide improves glucose metabolism by inhibiting glycolysis, as observed in intrahepatic cholangiocarcinoma via the Akt/mammalian target of rapamycin pathway signaling pathway to suppress cancer cell proliferation [18], and rheumatoid arthritis by reducing glycolytic enzyme expression and lactate concentrations to alleviate joint inflammation [19]. Notably, these previously documented beneficial effects of triptolide on glucose metabolism are all associated with its inhibition of glycolysis. This study is the first to demonstrate that triptolide improves glucose metabolism by promoting gluconeogenesis.
Second, we demonstrated that triptolide upregulated PGC1
Third, we identified a novel link between metabolic regulation (gluconeogenesis
promotion) and epigenetic/inflammatory modulation (histone lactylation
inhibition) in the anti-fibrotic mechanism of triptolide. Recent study has
demonstrated that lactate (a byproduct of impaired gluconeogenesis) promotes
renal fibrosis via H4K12 histone lactylation-mediated inflammatory responses
[22]. Notably, histone lactylation, including at the H4K12 site, can exhibit
context-dependent roles in inflammation. While our data and a recent study
support a pro-inflammatory role of H4K12 lactylation in renal tubular cells [22],
emerging evidence also suggests that lactate and lactylation may promote
macrophage polarization toward an M2 phenotype [26]. In this study, we observed a
positive correlation between IL‑1
Furthermore, our findings have implications for translating preclinical research
into clinical practice. Clinically approved agents targeting renal
gluconeogenesis are scarce, and triptolide-containing formulations are already
used in China to treat renal diseases such as nephritis and membranous
nephropathy [16, 17]. By identifying the
PGC1
This study has several limitations. First, although we observed changes in the
protein expression of PCK1 and FBP1, and alterations in the levels of downstream
metabolites (such as glucose and lactate) that suggested changes in enzymatic
activity, we were unable to directly measure the enzymatic activities of PCK1 and
FBP1. Similarly, while treatment with inhibitors (3-MPA and SR-18292) produced
the expected metabolic and molecular phenotypic changes, we did not provide
direct evidence for the inhibition of PCK1 enzymatic activity or PGC1
In conclusion, our findings suggest that triptolide may attenuate renal
tubulointerstitial fibrosis by modulating the PGC1
The paper is listed as, “Triptolide ameliorates renal tubulointerstitial fibrosis through EZH2” as a preprint on bioRxiv at: https://www.biorxiv.org/content/10.1101/2023.01.29.526092v1.
All relevant data were included in the study. The datasets generated during this study are available from the corresponding author upon reasonable request.
LL, DC, LY, and MW funded the project. LL, GW, MW and DC conceived this project. MW coordinated the study and wrote the paper. YW, YJ and GW conducted the in vitro experiments. MW, YW, LY, and DC performed the animal experiments. YW, LL and LY performed and analyzed the Western blotting. All authors reviewed the results and approved the final version of the manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experiments involving animals were performed in accordance with the National Institutes of Health guidelines and approved by the Animal Experimentation Ethics Committee of Shanghai University of Traditional Chinese Medicine (PZSHUTCM18111601).
Not applicable.
This work was supported by Key Disciplines Group Construction Project of Pudong Health Bureau of Shanghai (PWZxq2017-07); The Three Year Action Plan Project of Shanghai Accelerating Development of Traditional Chinese Medicine (ZY(2018-2020)-CCCX-2003-08); Shanghai Key Laboratory of Traditional Chinese Clinical Medicine (20DZ2272200), Shanghai, PR China; Scientific Research Foundation of Shanghai Municipal Commission of Health and Family Planning (201740193) to MW; National Natural Science Foundation of China (81470914) to LL; National Natural Science Foundation of China (81400687) and the Western Medicine Guiding Project of Shanghai Science and Technology Commission (18411961000) to DC; Baoshan District Health Commission Excellent Youth (Yucai) Program BSWSYC-2025-03 to LY; National Natural Science Foundation of China (82205018) to LY.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






