1 Department of Hematology, The First Affiliated Hospital of Guangxi Medical University, 530021 Nanning, Guangxi, China
2 Key Laboratory of Hemotology, Guangxi Medical University, Education Department of Guangxi Zhuang Autonomous Region, 530021 Nanning, Guangxi, China
†These authors contributed equally.
Abstract
Acute myeloid leukemia (AML) is a hematological malignancy of the myeloid lineage with poor clinical outcomes due to limited targeted therapies. This study elucidates the role of inositol 1,4,5-trisphosphate receptor type 2 (ITPR2) in regulating cell apoptosis by modulating mitochondrial calcium (Ca2+) levels and underscores the clinical significance of ITPR2.
ITPR2 expression in patients with AML compared with that of healthy controls using the quantitative real-time PCR (RT-qPCR) method. The role of ITPR2 in AML and its association with immune infiltration levels were investigated through bioinformatics analyses. ITPR2 knockdown in Tohoku Hospital Pediatrics-1 (THP-1) cells were achieved using small interfering RNA (siRNA) and 2-aminoethyl diphenylborinate (2-APB), followed by comprehensive molecular characterization employing RT-qPCR, western blotting (WB), cell counting kit-8 (CCK-8) assays, and flow cytometry.
ITPR2 was validated as being highly expressed in patients with AML, and this expression correlated with risk stratification and poor prognosis. Functional enrichment analysis revealed that ITPR2 is involved in Ca2+ signaling pathways and mitochondrial-related biological processes, and its expression level was negatively correlated with immune infiltration levels. Knockdown of ITPR2 or inhibition of its activity with 2-APB reduced Ca2+ ion concentrations in both the cytoplasm and mitochondria, leading to mitochondrial dysfunction (characterized by elevated intracellular reactive oxygen species (ROS) levels, reduced mitochondrial membrane potential (MMP) levels, and reduced mitochondrial DNA copy number) and eventually AML cell apoptosis.
ITPR2 facilitates AML progression via the Ca2+-mitochondrial axis and may serve as a prognostic factor and potential therapeutic target.
Graphical Abstract
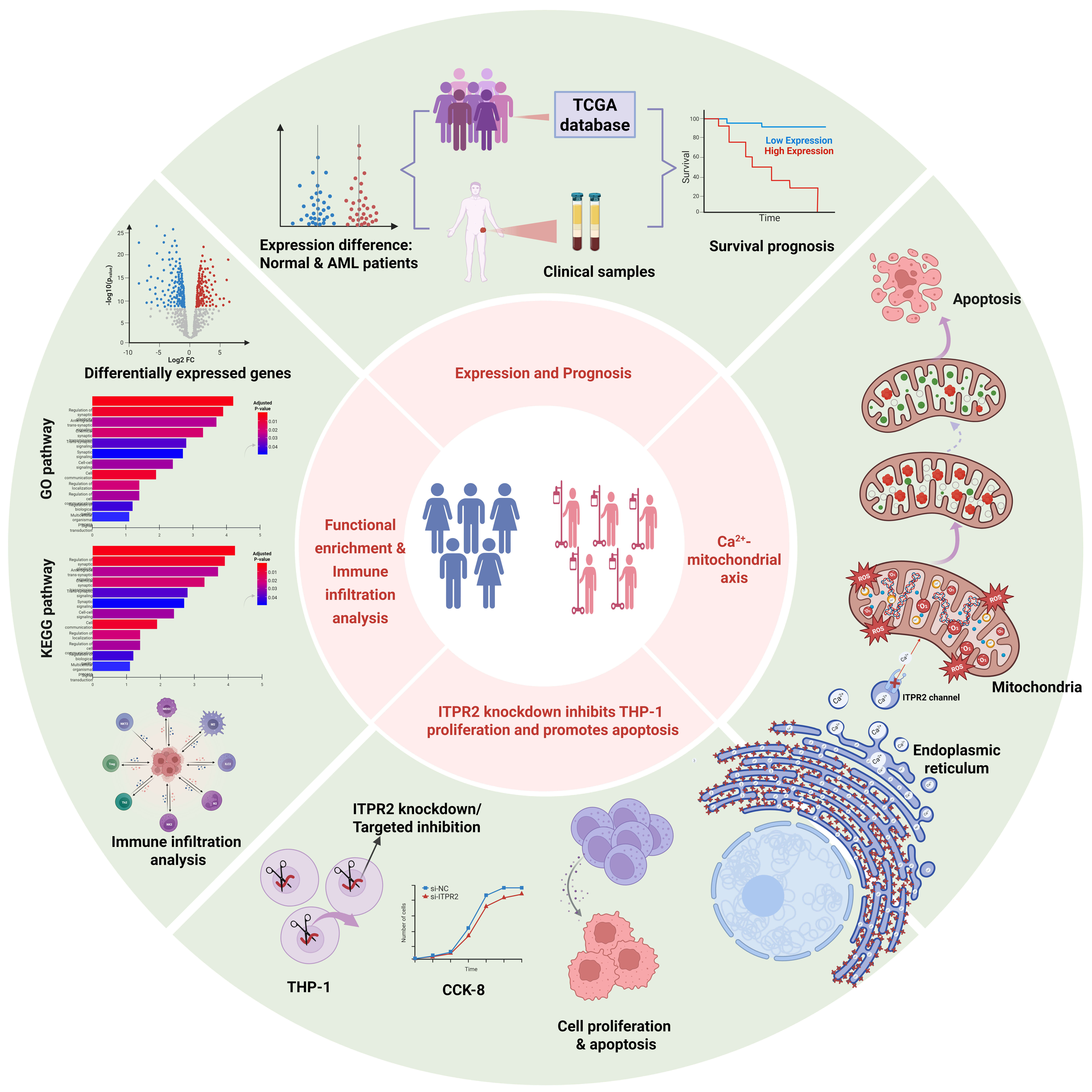
Keywords
- acute myeloid leukemia
- mitochondrial
- calcium
- prognosis
The acute myeloid leukemia (AML) is a highly heterogeneous clonal malignancy of the haematopoietic system and the most prevalent form of acute leukaemia in adults [1]. Abnormal gene expression and mutations are central mechanisms that regulate the initiation and progression of AML. Although the clinical application of targeted therapeutic agents, such as FMS-like tyrosine kinase 3 (FLT3) inhibitors, isocitrate dehydrogenase 1/2 (IDH1/2), B-cell lymphoma-2 (Bcl-2) inhibitors, has broadened treatment options for AML [2], the overall survival (OS) of patients has not significantly prolonged. Therefore, there is an increasing clinical need to systematically clarify the core molecular mechanisms involving AML pathogenesis and to explore potential therapeutic targets with clinical translational value. This endeavor is of great scientific significance to explore new and effective treatment methods aimed at improving patient prognosis.
Inositol 1,4,5-trisphosphate receptors (ITPRs), crucial intracellular calcium (Ca2+) ion channel proteins, are widely distributed in mammalian tissue cells, mediating Ca2+ ion release and signal transduction. The Ca2+ ion signaling pathway regulates fundamental physiological processes like cell proliferation, apoptosis, and differentiation [3, 4]. Due to their central regulatory role, ITPRs are vital targets for understanding disease pathogenesis. Some investigators found that aberrant expression of ITPRs in solid tumors like breast cancer [5], cholangiocarcinoma [6], and renal cell carcinoma [7] correlates with advanced tumor stage and poor prognosis, suggesting their potential as diagnostic markers and therapeutic targets. Research on ITPRs in hematological diseases is limited. Our single-cell sequencing data revealed upregulation of inositol 1,4,5-trisphosphate receptor type 2 (ITPR2) in AML [8], consistent with prior reports [9], indicating a key oncogenic role of ITPR2 in AML. Nevertheless, the biological functions and mechanisms of ITPR2 in AML remain largely unknown.
This study reveals that ITPR2 is significantly upregulated in AML, with its elevated expression associated with poor prognosis in AML patients. Mechanistically, ITPR2 knockdown disrupts mitochondrial Ca2+ homeostasis, caused oxidative stress and mitochondrial dysfunction, which in turn promotes apoptosis in AML cells. These results clarify the fundamental role of ITPR2 in AML, and provide essential experimental evidence for understanding the molecular mechanisms that underlie AML pathogenesis and for developing treatment methods targeting ITPR2.
This study downloaded the uniformly standardized pan-cancer dataset from the University of California, Santa Cruz Genomic (UCSC) database and extracted ITPR2 expression data for each sample. Acute myeloid leukemia-M3 (AML-M3) samples were excluded, and all expression data were transformed to log2(x + 1) form. Differences in ITPR2 expression between samples were analyzed using R software (version 4.2.1, R Foundation for Statistical Computing, Vienna, Austria) [10]. The association between ITPR2 level and OS was investigated using data from the Gene Expression Omnibus (GEO) database (accession No: GSE12417; n = 242), with the median ITPR2 level used as the threshold for grouping.
Bone marrow (BM) samples were obtained from 82 newly diagnosed AML patients and 30 normal donors at the First Affiliated Hospital of Guangxi Medical University, China, from 2013 to 2021. The diagnosis of AML was confirmed according to the World Health Organization (WHO) classification system. Ethical approval for this research was granted by the Ethical Review Commission of the First Affiliated Hospital of Guangxi Medical University, Nanning, China (Approval code: 2025-E0668).
Bone marrow mononuclear cells (BM-MNCs) were isolated by centrifugation, from
which total RNA was extracted. RNA purity was assessed via spectrophotometer,
with a 260/280 nm ratio greater than 1.8 indicating adequate purity. The total
RNA was then converted into complementary DNA by reverse transcription. Human
ITPR2 expression data were extracted from The Cancer Genome
Atlas–Acute Myeloid Leukemia (TCGA-LAML) (non-M3) database. The samples were
divided into high and low expression groups based on the median ITPR2
level. Differential expression analysis between the two groups was performed
using the limma package, with differentially expressed genes (DEGs)
judgement criteria of
Tohoku Hospital Pediatrics-1 (THP-1) cells were acquired from the Shanghai Cell Bank (https://www.cellbank.org.cn). THP-1 cells were cultured in RPMI-1640 (Gibco, NY, USA) with 10% fetal bovine serum (FBS) (Biological Industries, Cromwell, CT, USA) and 1% penicillin-streptomycin at 37 °C. All cell lines were validated by STR profiling and tested negative for mycoplasma.
siRNA was used to silence the ITPR2 gene. The plasmid construct was synthesized and generated by Guangzhou Ribobio Co., Ltd. (a commercial biotechnology company specializing in nucleic acid synthesis and vector construction). The interfering sequences targeting the ITPR2 gene were as follows: siRNA-ITPR2-1: 5′-CAACGAAATTAGCGAGAGA-3′, siRNA-ITPR2-2: 5′-CCCTTAGCCTACCACATCA-3′, siRNA-ITPR2-3: 5′-GTGGCGCTTTCATGTCGAA-3′. THP-1 cells were seeded in 6-well plates and cultured until 50% confluency. Subsequently, transfection of 20 nM negative control siRNA (si-NC) with a scrambled sequence that does not target any human genes or ITPR2-specific siRNA into THP-1 cells was performed using the CALNP™ RNAi in vitro (Beijing D-Nano Therapeutics, Beijing, China) reagent at 37 °C for 24 h. Subsequently, at other indicated time points, the cells were harvested and stored.
Western Blot (WB) cellular proteins were lysed using radio-immunoprecipitation assay (RIPA) buffer
and centrifuged for 5 minutes at 4 °C. Equal amounts of protein (40
µg) were loaded into each lane and separated by 10% SDS-polyacrylamide gel
electrophoresis (SDS/PAGE). The proteins were then electrotransferred to a
polyvinylidene fluoride (PVDF) membrane. The membranes were blocked via 8%
nonfat milk for 1 h and overnight incubated with primary antibodies at 4
°C. The specific primary antibodies used in this study were: anti-ITPR2 (Affinity, CAT#DF13336, dilution ratio 1:1000), anti-Cyclin D1 (Proteintech, CAT#26939-1-AP, dilution ratio 1:2000), anti-Cyclin E1 (Proteintech, CAT#11554-1-AP, dilution ratio 1:2000), anti-CDK6 (Proteintech, CAT#14052-1-AP, dilution ratio 1:1000), anti-Bcl-2 (Proteintech, CAT#12789-1-AP, dilution ratio 1:4000), anti-Bax (Proteintech, CAT#50599-2-Ig, dilution ratio 1:5000), and anti-actin (Abcam, CAT#ab8226, dilution ratio 1:2000). After washing via phosphate-buffered saline (PBS), the membranes
were incubated for 1 h with corresponding secondary antibodies (Anti-Rabbit IgG (Zenbio, CAT#511203, dilution ratio 1:5000)
Anti-Mouse IgG (Zenbio, CAT#511103, dilution ratio 1:4000)) in a blocking
solution. Chemiluminescence was detected using a kit from chemiluminescence
detection kit (Bio-Rad) [16] with
CCK-8 (NCM Biotech, Suzhou, Jiangsu, China) was utilized to evaluate cell viability. Ten microliters of CCK-8 were given each well and cultured for 2 h at 37 °C. The cells absorbance were detected with a spectrophotometer (Molecular Devices, San Jose, CA, USA) at 0, 24, 48, 72 h post-seeding.
Cell Cycle Analysis Kit (Beyotime, Shanghai, China) was applied to conduct the cell cycle assessment. Cells were subjected to two rounds of washing with PBS. After that fixed them in ethanol at 4 °C for 12 hours. Subsequently, centrifuged at 800 rpm for 5 minutes, then stained with propidium iodide (PI) and RNase A, and incubated at 37 °C for 30 minutes without light. The stained cells were detected via the CytoFLEX flow cytometer (Beckman Coulter, Brea, CA, USA). G0/G1, S, and G2/M phase distribution were measured using FlowJo software (v10.8.1, FlowJo LLC, Ashland, OR, USA).
The Annexin V-Fluorescein Isothiocyanate (FITC) Apoptosis Detection Kit (Beyotime) was utilized to measure cellular apoptosis following the relevant interpretion. Cells from various groups were plated in 6-well plates, then collected and washed with PBS. Next, 195 µL of Annexin V-FITC binding solution was used to resuspend them. For each tube, 5 µL of Annexin V-FITC and 10 µL of PI were added, then cultured for 20–25 minutes without light. Apoptosis rates of cells from each group were evaluated by flow cytometry. Additionally, late apoptosis was evaluated via labeling DNA fragments of apoptotic cells with a Terminal Deoxynucleotidyl Transferase dUTP Nick-End Labeling (TUNEL) Assay Kit (Elabscience, Wuhan, China). Following fixation, permeabilization, equilibration, labeling, and washing per the instructions, detected with flow cytometer. TUNEL-positive cells were quantified with FlowJo software (v10.8.1).
Fluorescent indicators Fluo-4 AM (Beyotime) and Rhod-2 AM (Yesen, Shanghai, China) were employed to determine the levels of cytoplasmic Ca2+ and mitochondrial Ca2+, respectively. THP-1 cells were prepared as single-cell suspensions and loaded with 5 µM Fluo-4 AM and 5 µM Rhod-2 AM by incubation at 37 °C for 30 minutes. Then, the cells were washed with PBS to eliminate any extracellular Fluo-4 AM and Rhod-2 AM. The cytoplasmic and mitochondrial Ca2+ concentrations were then simultaneously measured by CytoFLEX flow cytometry (Beckman Coulter).
The MMP was assessed via flow cytometry with JC-1 staining, following the manufacturer’s instructions (C2006, Beyotime) [17]. Cells were collected and resuspended in 0.5 mL of cell culture medium and then incubated with 0.5 mL of a JC-1 staining solution in the dark at 37 °C for 20 minutes. Then, the cells were harvested, washed three times with JC-1 staining buffer, and resuspended in JC-1 staining buffer. The red/green fluorescence of JC-1-labelled cells was detected using a CytoFLEX flow cytometry (Beckman Coulter). The difference in MMP were determined by the color transformation of the fluorescence.
Intracellular ROS levels were detected with a DCFH-DA probe (Beyotime). The cells were treated with serum-free media containing 10 µM DCFH-DA probe and incubated at 37 °C in the dark for 30 minutes, with gentle agitation every 5 minutes. After incubation, the cell pellets were collected, washed with PBS, and resuspended in PBS for CytoFLEX flow cytometry (Beckman Coulter) detection. The resulting green fluorescence was measured and the mean fluorescence intensity (MFI) serves as an indicator of the relative intracellular ROS levels.
A Genomic DNA Mini Preparation Kit (Beyotime) was used to isolate total DNA from
treated cells. Reactions for RT-qPCR were performed with Real Star Fast SYBR qPCR
(Genstar, Shenzhen, China). Human
Cells were seeded in the medium containing varying concentrations of
2-Aminoethyl diphenylborinate (2-APB) or dimethyl sulfoxide (DMSO) control. After
a specified incubation interval, 10 µL of CCK-8 reagent was added, and the
plates were cultured for viability of the cells was determined according to this
formula: cell viability rate = [(As–Ab)/(Ac–Ab)]
Statistical analysis were performed on SPSS 25.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism 8.0 software. Each
in vitro experiment was replicated a minimum of three times for every
sample, repeat, group, and condition. The t-test was used to judge
statistical variances in quantitative datasets. For nonparametric data, the
Mann-Whitney U test was implemented. Correlations between two parameters were
determined via the Pearson’s analysis. The criteria of p
Given the potential critical role of ITPR2 in AML, we first investigated the expression profiles of ITPR2 across malignancies. This research collected a harmonized pan-cancer dataset from the UCSC database, which revealed that ITPR2 expression exhibits significant variations among multiple tumor types (Fig. 1A). To explore the clinical significance of ITPR2 in AML prognosis, the gene expression and clinical data of 242 non-M3 AML patients from the GSE12417 dataset were downloaded. Kaplan–Meier analysis revealed that high ITPR2 level was associated to significantly shorter OS (Fig. 1B)
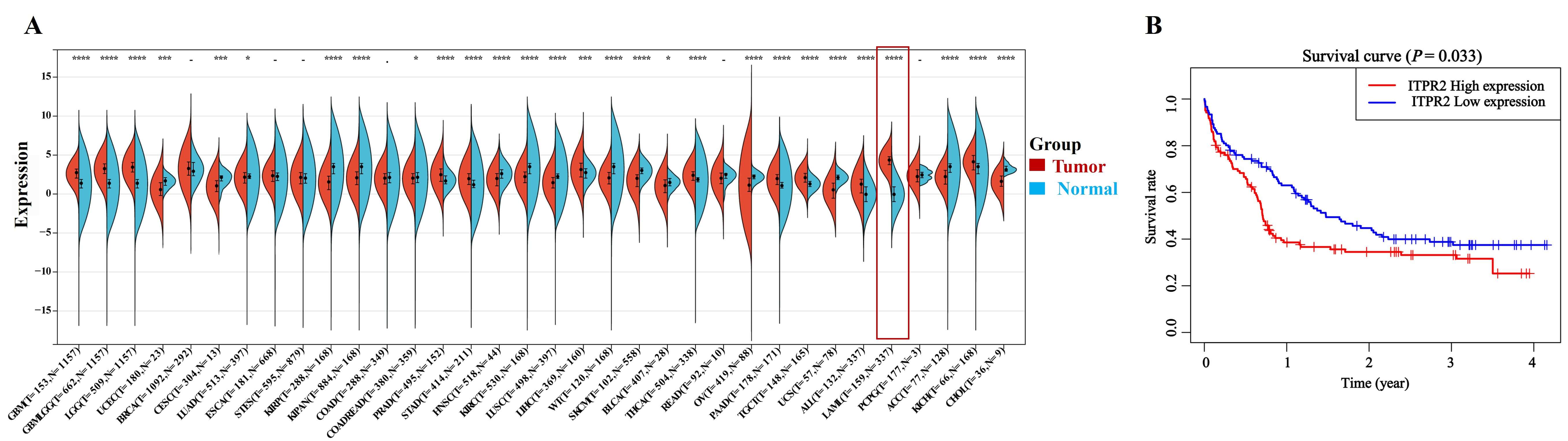 Fig. 1.
Fig. 1.
ITPR2 expression was increased in public databases vs.
normal group and associated with prognosis. (A) Expression of ITPR2 in
pan-cancer. (B) OS of GSE12417 patients (n = 242) stratified by high vs. low
ITPR2 expression. * p
To validate the results, BM samples and clinical data from 82 non-M3 AML
patients and 30 healthy donors were collected. RT-qPCR analysis revealed
significantly higher ITPR2 expression in AML patients than in controls
(Fig. 2A). Receiver operating characteristic (ROC) curve analysis revealed an
area under the curve (AUC) of 0.906 (95% CI: 0.851–0.961, p
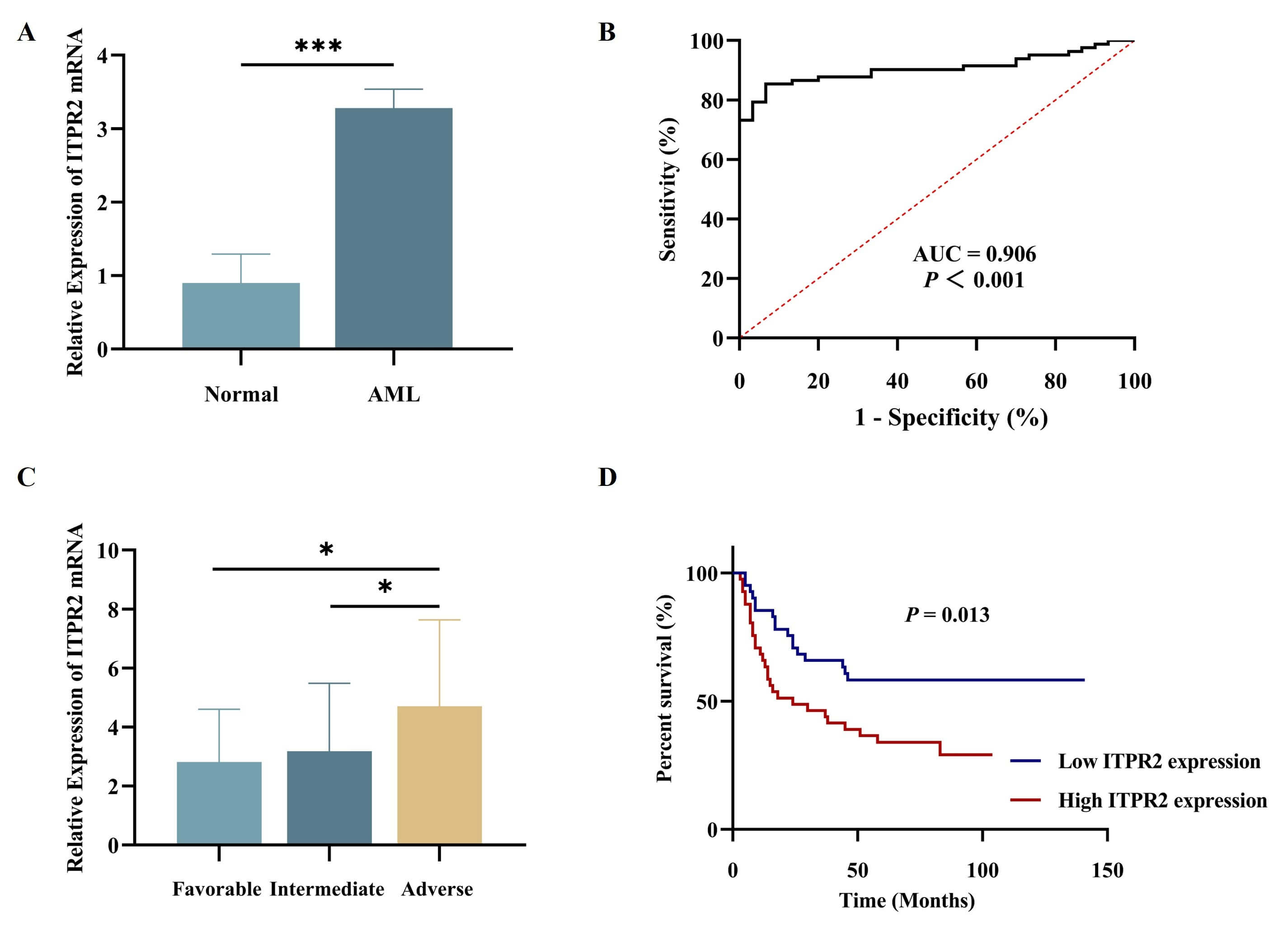 Fig. 2.
Fig. 2.
ITPR2 expression was increased in clinical samples vs.
normal group and associated with clinical features. (A) The expression of
ITPR2 in AML patients (n = 82) and normal group (n = 30). (B) ROC
analysis of ITPR2 for diagnosing AML. (C) ITPR2 expression
associated with increasing risk stratification. (D) Kaplan-Meier analysis of AML
patients showed significantly lower survival in the ITPR2 high
expression group compared with the low expression group. * p
To further analysis the relationship between ITPR2 expression and OS in
AML patients, subjects were divided into low and high ITPR2 expression
groups with the median relative ITPR2 expression as the cutoff. The
Kaplan-Meier analysis indicated a significantly shorter OS in AML cases with high
ITPR2 level versus those with low ITPR2 level (Fig. 2D). Table 1 compares the clinical and biological characteristics of the two groups.
Notably, high ITPR2 expression was significantly associated with a
greater number of BM blasts (p = 0.01). The Cox regression analyses of
the AML subgroups indicated that high ITPR2 level was a significant
independent factor for short OS (p
| Clinical characteristics | ITPR2 level | p value | |||
| Low (n = 41) | High (n = 41) | ||||
| Age (years), median (range) | 37 (22–62) | 32 (19–67) | 0.388 | ||
| Sex | 0.656 | ||||
| Male | 18 | 16 | |||
| Female | 23 | 25 | |||
| Leukocyte, ×109/L, median (range) | 15.90 (0.49–182.50) | 29.48 (0.09–242.10) | 0.310 | ||
| Hemoglobin, g/L, median (range) | 77.80 (46.00–126.20) | 72.60 (40.10–115.6) | 0.926 | ||
| Platelet, ×109/L, median (range) | 33.30 (8.00–168.60) | 44.40 (7.00–237.10) | 0.568 | ||
| Bone marrow (BM) blasts, %, median (range) | 51.50 (20.00–94.20) | 69.00 (21.20–92.20) | 0.010 | ||
| FAB type, n (%) | 0.679 | ||||
| M1 | 1 (2.44) | 4 (9.76) | |||
| M2 | 14 (34.15) | 11 (26.83) | |||
| M4 | 14 (34.15) | 16 (39.02) | |||
| M5 | 12 (29.26) | 9 (21.95) | |||
| M6 | 0 (0.00) | 1 (2.44) | |||
| Cytogenetic risk n (%) | 0.061 | ||||
| Favorable | 15 (36.58) | 12 (29.27) | |||
| Intermediate | 23 (56.10) | 20 (48.78) | |||
| Adverse | 3 (7.32) | 9 (21.95) | |||
| Mutation status, n (%) | |||||
| Nucleophosmin 1 (NPM1) | 0.243 | ||||
| Mutation | 9 (21.95) | 5 (12.20) | |||
| Wild type | 32 (78.05) | 36 (87.80) | |||
| Fms-like Tyrosine Kinase 3 (FLT3) | 0.801 | ||||
| Mutation | 10 (24.39) | 11 (26.83) | |||
| Wild type | 31 (75.61) | 30 (73.17) | |||
| DNA Methyltransferase 3 (DNMT3) | 0.535 | ||||
| Mutation | 7 (17.07) | 5 (12.20) | |||
| Wild type | 34 (82.93) | 36 (87.80) | |||
| CCAAT/Enhancer Binding Protein Alpha (CEBPA) | 0.193 | ||||
| Mutation | 7 (17.07) | 12 (29.27) | |||
| Wild type | 34 (82.93) | 29 (70.73) | |||
| Wilms Tumor 1 (WT1) | 0.148 | ||||
| Mutation | 15 (36.59) | 9 (21.95) | |||
| Wild type | 26 (63.41) | 32 (78.05) | |||
| Hematopoietic stem cell transplantation (HSCT), n (%) | 0.263 | ||||
| Yes | 14 (34.15) | 19 (46.34) | |||
| No | 27 (65.85) | 22 (53.66) | |||
| Complete remission, n (%) | 0.355 | ||||
| Yes | 29 (70.73) | 25 (60.98) | |||
| No | 12 (29.27) | 16 (39.02) | |||
FAB, French-American-British Classification System.
| Factor | Univariate | Multivariate | ||
| Hazard ratio (95% CI) | p value | Hazard ratio (95% CI) | p value | |
| ITPR2 (high vs. low) | 2.113 (1.155–3.866) | 0.015 | 1.972 (1.073–3.622) | 0.029 |
| Age ( |
1.494 (0.803–2.779) | 0.205 | ||
| Sex (male vs. female) | 0.994 (0.969–1.019) | 0.614 | ||
| Leukocyte ( |
0.835 (0.464–1.501) | 0.547 | ||
| Hemoglobin, ( |
0.758 (0.422–1.362) | 0.355 | ||
| Platelet ( |
1.346 (0.743–2.438) | 0.327 | ||
| BM blasts ( |
1.180 (0.655–2.126) | 0.581 | ||
| NPM1 (mutated vs. wild) | 0.700 (0.296–1.654) | 0.416 | ||
| FLT3 (mutated vs. wild) | 1.379 (0.711–2.675) | 0.341 | ||
| DNMT3 (mutated vs. wild) | 0.536 (0.192–1.497) | 0.234 | ||
| CEBPA (mutated vs. wild) | 1.131 (0.573–2.235) | 0.722 | ||
| WT1 (mutated vs. wild) | 0.479 (0.222–1.032) | 0.060 | ||
| Cytogenetic risk (Favorable vs. Intermediate & adverse) | 1.834 (0.945–3.559) | 0.073 | ||
| Complete remission (yes vs. no) | 6.149 (3.203–11.805) | 4.968 (2.464–10.018) | ||
| HSCT (yes vs. no) | 0.400 (0.206–0.775) | 0.007 | 0.627 (0.306–1.285) | 0.203 |
To analysis the functional significance of ITPR2 in AML, TCGA samples
were divided into two different expression groups based on the median
ITPR2 level. Differential expression analysis was performed using the
limma package. DEGs were screened with
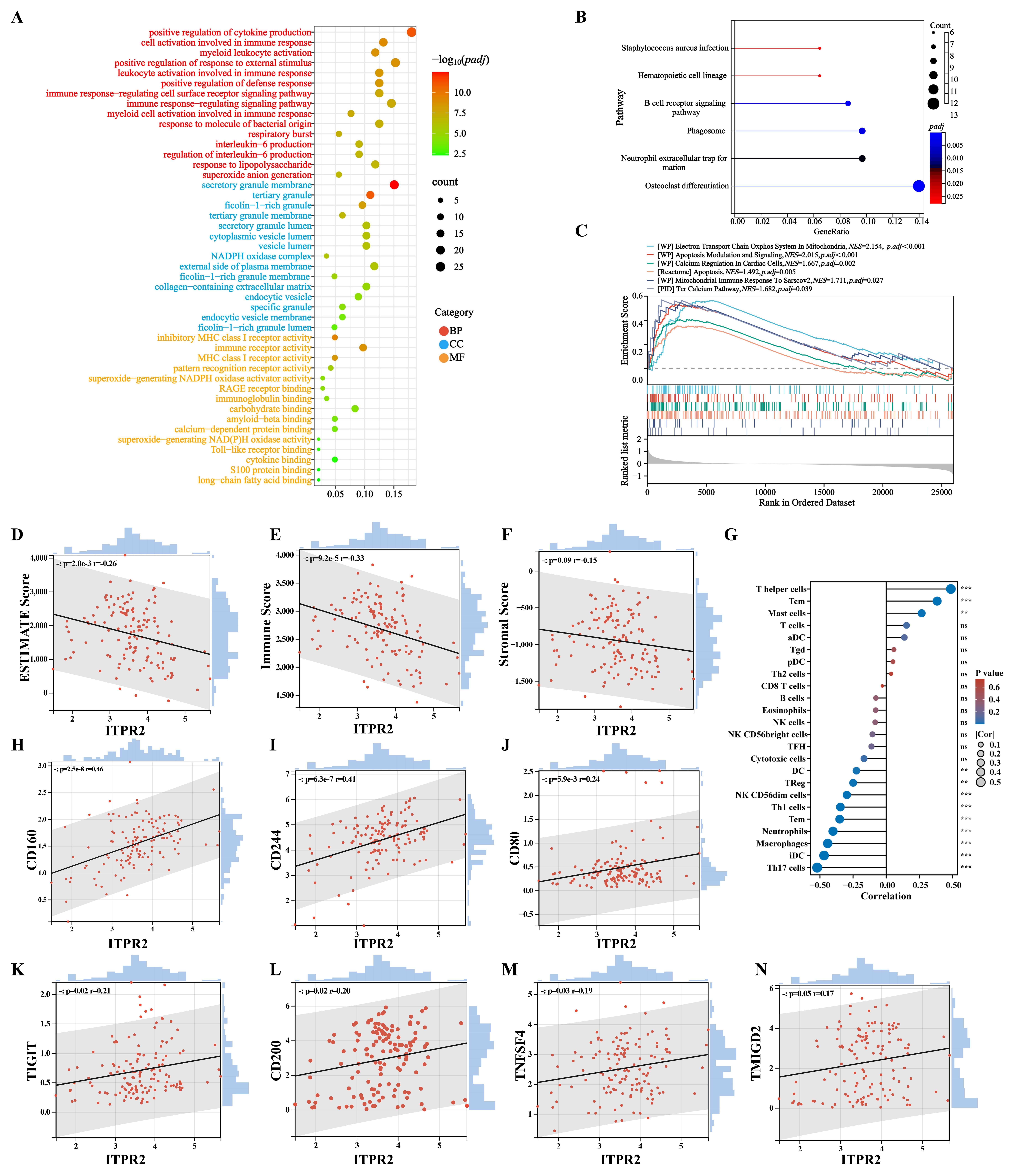 Fig. 3.
Fig. 3.
Functional enrichment and immune infiltration analysis of
ITPR2 expression in TCGA-LAML patients. (A,B) GO and KEGG enrichment
analysis of DEGs. (C) GSEA analysis of DEGs. (D–F) Correlation analysis of
ESTIMATE score (D), immune score (E) and stromal score (F), respectively. (G)
Correlation analysis of ITPR2 with different immune cells. (H–N)
Correlation between ITPR2 and the immune checkpoint CD160 (H), CD244
(I), CD80 (J), TIGIT (K), CD200 (L), TNFSF4 (M) and
TMIGD2 (N), respectively. ** p
For the purpose of exploring ITPR2’s role in AML-related immune regulation, the ESTIMATE algorithm was utilized. Outcomes revealed an inverse association between ITPR2 level and both the ESTIMATE and immune score (Fig. 3D–F). Using the ssGSEA algorithm, the correlation of ITPR2 with the infiltration score was analysed (Fig. 3G). ITPR2 level was positively related to the T helper and T-cell infiltration but negatively related to the Tregs, Th17 cells, macrophages, and NK CD56dim cells. Furthermore, the associations between ITPR2 and immune checkpoint genes were evaluated through pearson correlation analysis. ITPR2 was positively correlated with the expression of CD160, CD244, CD80, etc. (Fig. 3H–N).
To clarify the role of ITPR2 in AML, siRNAs were used to knockdown
ITPR2 in the THP-1 cell line. The knockdown efficiency was verified by
RT-qPCR and WB (Fig. 4A,B). In functional validation experiments, CCK-8 assay
data confirmed that ITPR2 knockdown obviously blocked the proliferation
of THP-1 cells (Fig. 4C). According to the result of flow cytometry, it was found
that the loss of ITPR2 caused an increase in the ratio of cells in the
G0/G1 stage (Fig. 4D) and suggested a potential induction of apoptosis (Fig. 4E,F). WB analysis further verified that ITPR2 knockdown not only reduced
the expression of G1/S transition regulatory factors (cyclin D1, cyclin E1, and
cyclin-dependent kinase 6 (CDK6)) but also downregulated the levels of Bcl-2 and
Bcl-2-associated X protein (Bax) (Fig. 4G). While both Bcl-2 and Bax decreased,
the resulting Bcl-2/Bax ratio
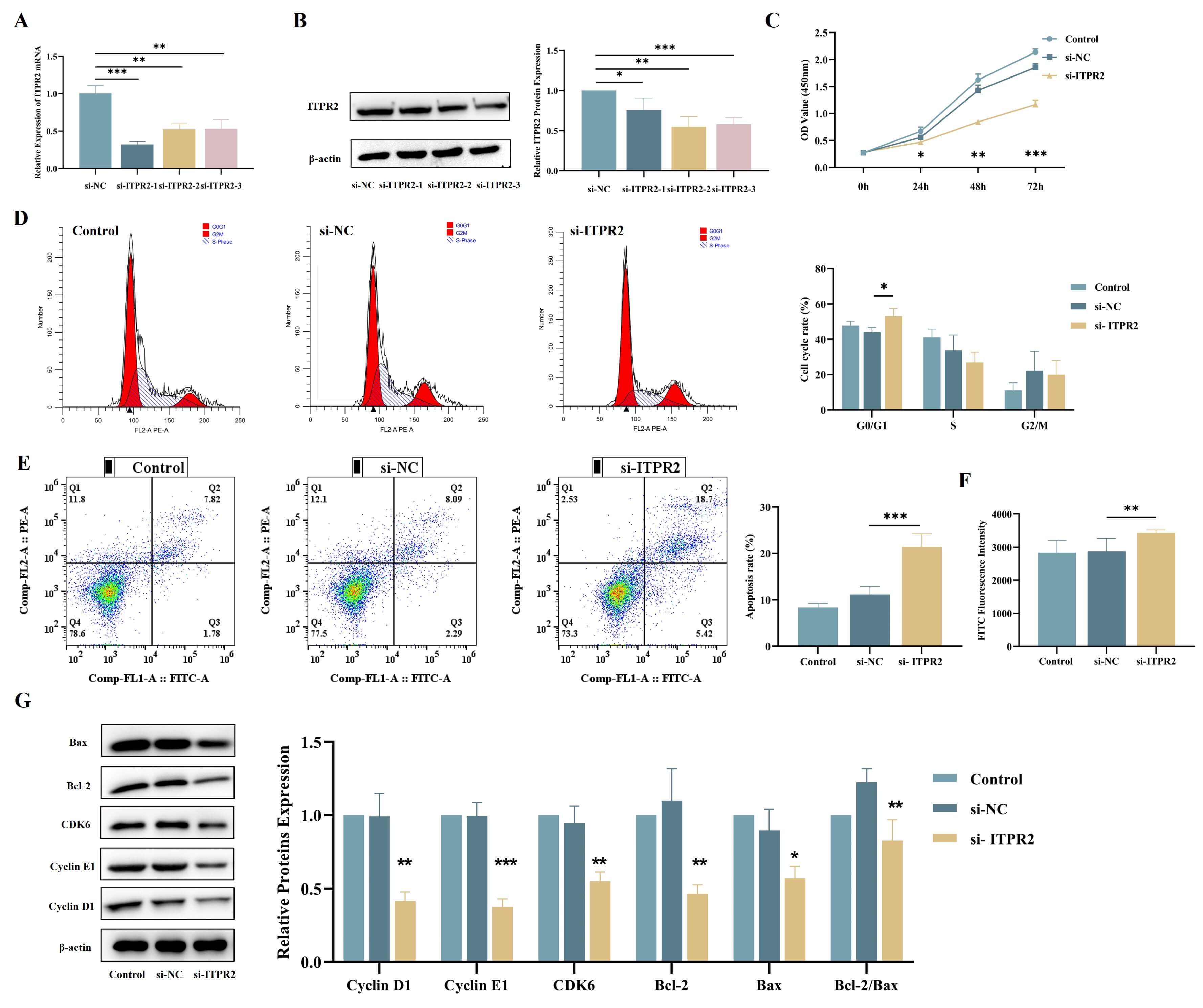 Fig. 4.
Fig. 4.
siRNA-mediated ITPR2 knockdown suppresses THP-1 cell
proliferation and induces apoptosis. (A,B) RT-qPCR and WB analysis were
employed to examine the efficiency of ITPR2 knockdown in THP-1 cells (n
= 3). (C) CCK-8 assay demonstrated that ITPR2 knockdown inhibited THP-1
proliferation (n = 3). (D) Cell cycle distribution of ITPR2-knockdown
THP-1 cells analyzed by flow cytometry (n = 3). (E,F) The apoptotic rate of
ITPR2 knockdown THP-1 cells was increased by using the Annexin V-FITC
Apoptosis Detection Kit (E) and the One-step TUNEL Apoptosis Kit (n = 3) (F) for
cell staining separately through flow cytometry. (G) WB analysisdemonstrated the expression levels of cell cycle and apoptosis-related proteins in ITPR2 knockdown cells. Grayscale values were quantified using ImageJ. The quantitative results of the levels of cycle and apoptosis-related proteins were adjusted using the level of
To explore the treatment potential of targeting ITPR2, an in vitro drug sensitivity assay was performed. 2-APB is a specific inhibitor of ITPRs [19]. RT-qPCR and WB confirmed that 2-APB effectively inhibited the expression of ITPR2 (Fig. 5A,B). The viability of THP-1 cells exposed to 2-APB was assessed with CCK-8 assays. The results revealed that prolonged drug exposure significantly decreased cell viability, with an IC50 value of 202 µM after 48 h (Fig. 5C). Therefore, in the subsequent experiments, this concentration and treatment time were selected for the subsequent tests. Additionally, the results of the cell proliferation assay indicated that compared with the control group, 2-APB led to more pronounced inhibition of cell growth (Fig. 5D). Flow cytometry analysis revealed that 2-APB increased the proportion of cells arrested in the G0/G1 phase (Fig. 5E) and suggested a potential induction of apoptosis (Fig. 5F,G). WB analysis further demonstrated that 2-APB treatment reduced the levels of cyclin D1, cyclin E1, Bcl-2, and Bax (Fig. 5H).
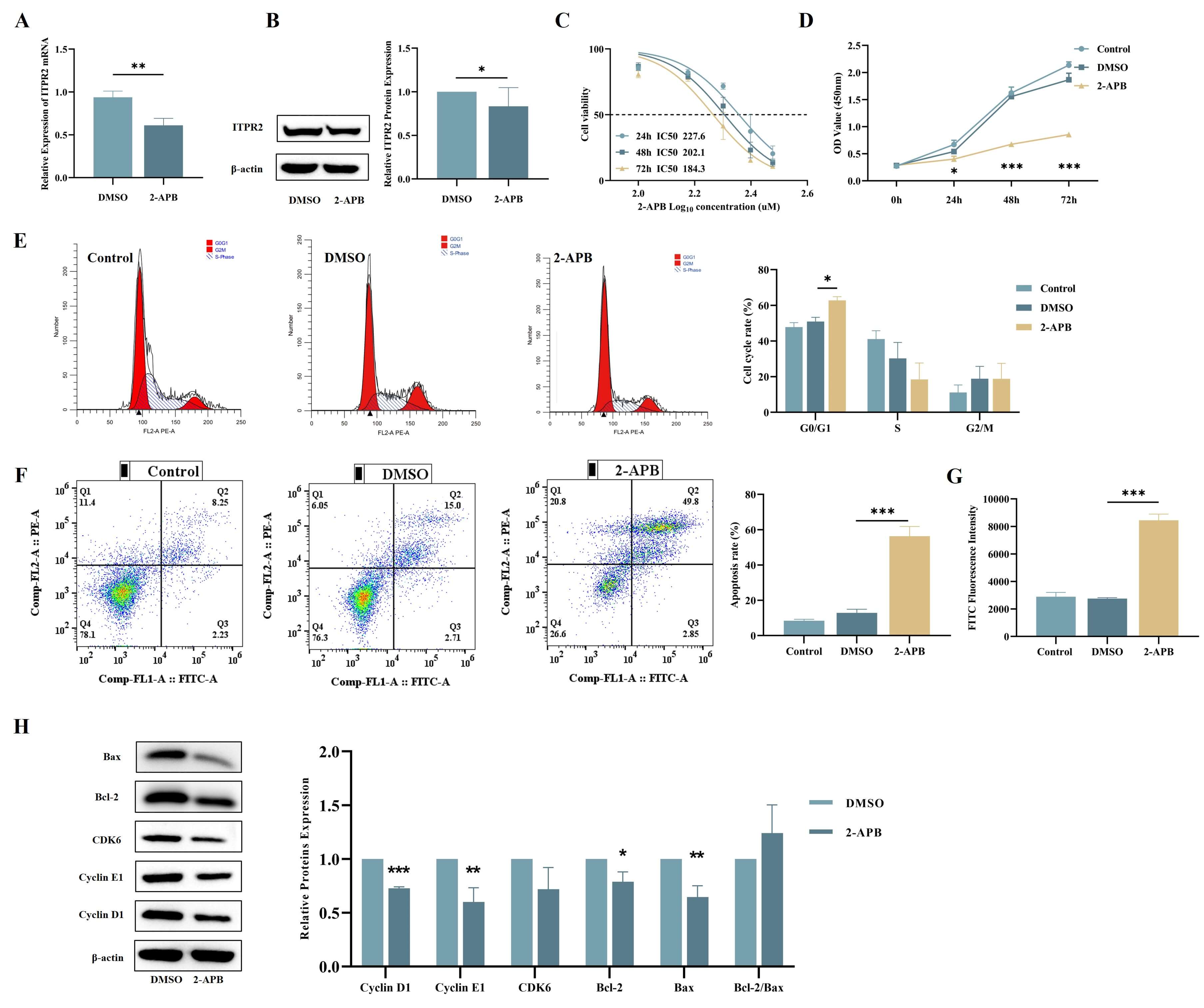 Fig. 5.
Fig. 5.
Exposure of THP-1 cells to 200 µM 2-APB for 48 h resulted
in impaired proliferation and enhanced apoptosis. (A,B) RT-qPCR and WB analysis
of THP-1 cells treated with 2-APB revealed decreased ITPR2 expression (n
= 3). (C) Effect of different concentrations of 2-APB on the cell viability of
THP-1 cell line (n = 6). (D) CCK-8 assay demonstrated 2-APB treatment effectively
suppressed THP-1 growth (n = 3). (E) Cell cycle distribution of 2-APB-treated
THP-1 cells analyzed by flow cytometry (n = 3). (F,G) The apoptotic rate of
2-APB-treated THP-1 cells was increased by using the Annexin V-FITC Apoptosis
Detection Kit (F) and the One-step TUNEL Apoptosis Kit (G) for cell staining
separately through flow cytometry (n = 3). (H) WB examined cell cycle and apoptosisrelated protein expression in 2-APB-treated THP-1 cells. Grayscale values were quantified using ImageJ. The quantitative results of the levels of cycle and apoptosis-related proteins were adjusted using the level of
ITPR2 mediates Ca2+ transport. To investigate whether the aberrant upregulation of ITPR2 in AML cells disrupts intracellular Ca2+ homeostasis, this research detected the cytosolic and mitochondrial Ca2+ levels via the Fluo-4 and Rhod-2 probes, respectively. Our results showed that compared with si-NC, si-ITPR2 significantly reduced Ca2+ concentrations in both compartments (Fig. 6A,B). Mitochondrial Ca2+ imbalance will result in elevated oxidative stress and mitochondrial dysfunction. With the MMP-sensitive fluorescent dye JC-1, changes in the MMP were evaluated by analyzing the red/green fluorescence ratio. Knockdown of ITPR2 significantly depolarized the MMP (Fig. 6C), a hallmark of mitochondrial dysfunction associated with early apoptosis. The intracellular ROS levels were determined using DCFH-DA. We found that ITPR2 knockdown increased the level of intracellular ROS (Fig. 6D). To evaluate the effect of ITPR2 on mtDNA-encoded respiratory chain gene expression, NADH dehydrogenase 1 (ND1) mRNA was quantified in THP-1 cells. Knockdown of ITPR2 significantly downregulated ND1 mRNA (Fig. 6E). These results are consistent with those in 2-APB-treated THP-1 cells (Fig. 6A–E).
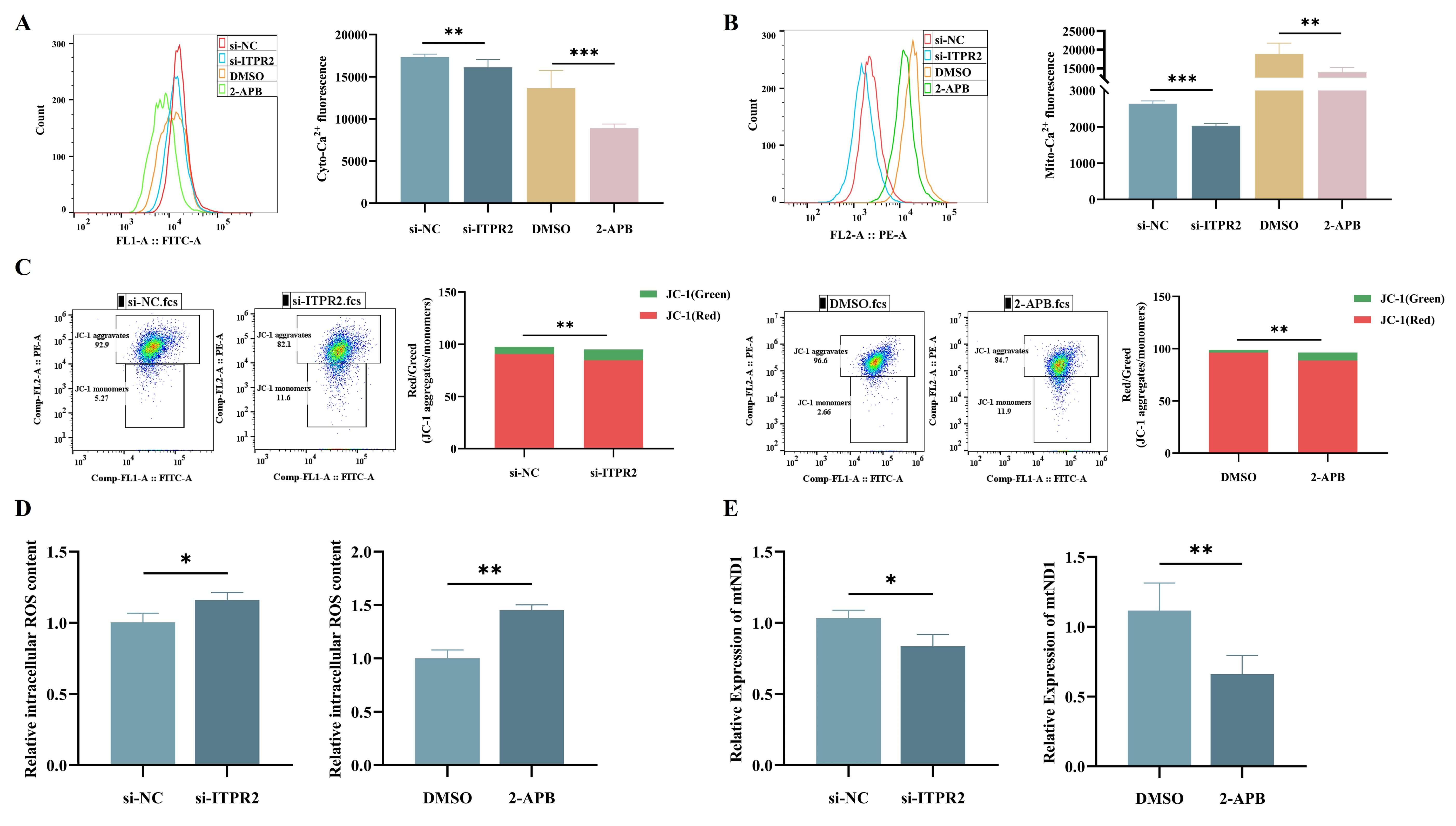 Fig. 6.
Fig. 6.
ITPR2 Knockdown and 2-APB treatment reduce
intracellular Ca2+ and induce mitochondrial dysfunction in THP-1 Cells. (A,B) Flow cytometric analysis revealed reduced intracellular Ca2+ (A) and
mitochondrial Ca2+ (B) levels in THP-1 cells after ITPR2 knockdown
or 2-APB treatment. (C) Flow cytometry showed reduced MMP in THP-1 cells
following ITPR2 knockdown or 2-APB treatment. (D) Flow cytometry showed
increased ROS levels in THP-1 cells following ITPR2 knockdown or 2-APB
treatment. (E) RT-qPCR showed reduced mtND1 expression in THP-1 cells
with ITPR2 knockdown or 2-APB treatment. The image represents one of the
3 independent parallel experiments. si‑, small interfering; NC, negative control.
Compared with si-NC or DMSO * p
Recent studies have highlighted the oncogenic potential of ITPR2 across various malignancies. The overexpression of ITPR2 has been implicated in a poor prognosis for lung adenocarcinoma cases [20], renal cell carcinoma [21], and chronic lymphocytic leukaemia [22]. In lung cancer A549 cells, the overexpression of endoplasmic reticulum protein 44 (ERP44) reduces intracellular Ca2+ release via ITPR2, alters cell morphology, and significantly inhibits cell migration [23]. Zhang et al. [24] reported that Aflatoxin B1 (AFB1) induced pyroptosis in mouse livers by activating the caspase-12/caspase-3 pathway through ITPR2 activation. The above findings indicate that ITPR2 could serve as a novel target for the therapy of AFB1-induced liver injury. In breast cancer patients, ITPR2 expression levels are specifically elevated in tumour tissues compared with adjacent nontumoral tissues. ITPR inhibition or siRNA-mediated silencing results in compromised bioenergetics and in increased ROS production and autophagy, which ultimately leads to cell death [4, 23].
This study suggests that ITPR2 may serve as a novel and critical therapeutic target for the therapy of AML. RT-qPCR results indicated that ITPR2 expression was notably elevated in BM-MNCs from AML patients, with higher levels correlating with a worse prognosis and shorter OS. ITPR2 supports proliferation in AML cells. This finding not only suggests that ITPR2 may serve as a key driver molecule in AML progression but also supports its potential as a prognostic biomarker, thereby further expanding the clinical implications of ITPR2 in hematological malignancies.
Immune dysregulation is one of the key mechanisms underlying the progression and therapeutic resistance of AML. To investigate the function of ITPR2 in AML, we first focused on its association with the AML immune microenvironment to uncover the potential mechanisms by which it regulates disease progression. Results from immune scoring and ESTIMATE analysis indicated that ITPR2 level was obvious negatively related to these scores, suggesting that ITPR2 may be involved in the formation of an immunosuppressive tumor microenvironment (TME) by regulating immune cell infiltration. Further ssGSEA revealed that ITPR2 was positively correlated with T helper cells and central memory T cells, while negatively correlated with innate immune cells (e.g., dendritic cells (DCs)/immature DCs (iDCs), etc.), effector memory T cells, Th1/Th17 cells, regulatory T cells. Notably, as immune memory cells, the enrichment of Tcm may increase the risk of AML recurrence, whereas the reduction of Th1/Th17 cells impairs antitumor immune responses. This bidirectional regulatory pattern is relatively rare among other tumor-related molecules, indicating that ITPR2 exerts a unique role in shaping the immune landscape of the AML TME.
Immune checkpoint molecules such as T cell immunoreceptor with Ig and ITIM domains (TIGIT), CD160, and CD244 are predominantly expressed on the T cells and NK cells. These molecules transmit inhibitory signals through binding to their ligands, which induces immune cell exhaustion, impairing their ability to recognize and kill tumour cells, and thus represents a key mechanism underlying tumour immune evasion. A preclinical research indicated that tumour-infiltrating TIGIT+ NK cells were exhausted in transplant mouse tumour models and that blockade of TIGIT can unleash the antitumour function of NK cells and promote the CD8+ T-cell antitumour response. The blockade of TIGIT alone or combined with anti-programmed cell death protein 1 (anti-PD-1) or anti-programmed death-ligand 1 (anti-PD-L1) blockade is being tested in many clinical trials [25, 26], highlighting its value of treatment target. In AML, high level of CD244 may associate with the construction of an immunosuppressive TME by inducing T-cell depletion, which impairs the antileukaemia function of these cells [27]. A similar mechanism has also been observed in chronic lymphocytic leukemia (CLL). It has been reported that higher level of CD244, PD-1, CD160 on the T cells of untreated CLL cases, is often accompanied by abnormal of cytotoxicity [28]. Here, we found that the abovementioned immune checkpoint molecules were significantly positively correlated with ITPR2, suggesting that ITPR2 may directly inhibit the immune activity of T and NK cells and indirectly promote the immune escape of tumour cells through the upregulation of the expression of these molecules. On the basis of this correlation, ITPR2 can be seemed as a predictor for the efficacy of relevant immunotherapies.
Beyond its immunomodulatory function, the regulation of malignant phenotypes in
AML cells by ITPR2 also relies on its core regulation of mitochondrial
function. Previous studies have demonstrated that upregulated ITPR2
promotes massive Ca2+ influx into mitochondria, leading to abnormal
elevation of Ca2+ concentration in the mitochondrial matrix. This alteration
activates Ca2+-dependent enzymes in mitochondria, thereby impairing
mitochondrial respiratory chain function. Inhibition of ITPR and Ca2+
transfer to mitochondria causes cell cycle arrest and cell death in T-cell acute
lymphoblastic leukaemia cells [29]. In AML cell lines, elevated mitochondrial
Ca2+ levels are observed in cells that can evade treatment with cytarabine
and venetoclax [30]. Zhong et al. [31] reported that abnormally
increased expression of oxysterol-binding protein-related protein 4L (ORP4L) can
activate ITPRs, leading to ER Ca2+ release to increase
mitochondrial respiration and promote the survival of leukaemia stem cells [32].
Our experimental data demonstrated that targeted knockdown of ITPR2
significantly reduced mitochondrial Ca2+ uptake and induced depolarization
of MMP (
Modulation of ion channels (both inhibition and activation) in cells and their organelles, as well as the modulation of cell redox states, has been used to treat various diseases. 2-APB, one of the most extensively studied boron-containing compounds, is capable of modulating the activities of a variety of ion channels and is used as an inhibitor of ITPRs [33]. 2-APB has been demonstrated to inhibit Ca2+ uptake by isolated liver mitochondria in a dose-dependent manner and to reduce cellular Ca2+ accumulation in hepatocellular carcinoma G2 cells [34]. In the present study, 2-APB, by inhibiting ITPR2 function, also induces reduced mitochondrial Ca2+ uptake, mitochondrial dysfunction, and ultimately AML cell apoptosis in AML cells. Given that ITPR2 is highly expressed in AML patients and associated with poor prognosis, these findings collectively confirm the potential feasibility of ITPR2 as a therapeutic target for AML, providing critical experimental evidence for the development of ITPR2-targeted therapeutic strategies. However, its clinical translational value requires further validation through in-depth follow-up studies.
Severals limitations of this study should be acknowledged. Firstly, confining the study to the THP-1 cell line, which lacks cell type diversity, and assessing only a restricted range of apoptotic markers (e.g., excluding the evaluation of cleaved caspase-3 and other essential apoptotic proteins) may constrain the generalizability of our findings. Therefore, forthcoming research will expand the variety of cell types and apoptotic markers for assessment, explore further the signaling pathways accountable for the observed effects. Secondly, we could not validate the in vitro results in AML primary cells. In the future, we will isolate primary cells from AML patients following standardized clinical processing and validation protocols, and corroborate the outcomes from THP-1 cell line experiments in the patient samples encompassed in the study. Thirdly, due to constraints in experimental duration and resources, in vivo validation remains incomplete. Complex in vivo microenvironment, such as immune regulation and intertissue crosstalk, may alter ITPR2-mediated molecular mechanisms and drug responses, necessitating further verification of in vitro findings using in vivo models. Additionally, the pharmacokinetic and pharmacodynamic properties of 2-APB in vivo—such as its bioavailability, tissue distribution, metabolism, and off-target effects—may differ substantially from its performance in cell culture systems, which could impact its actual therapeutic potential and safety profile. Therefore, future studies should prioritize validating these observations using relevant preclinical models, including AML xenograft mice, patient-derived xenograft models, or conditional ITPR2 knockout models, to confirm the physiological significance of ITPR2 in AML progression and rigorously evaluate the in vivo efficacy and safety of ITPR2-targeted therapies.
This study systematically clarifies the core role and clinical value of ITPR2 in AML, offering an innovative perspective. Mechanistically, we first demonstrate that ITPR2 drives AML progression via two pathways: (1) maintaining mitochondrial integrity through regulating Ca2+ transport via the “Ca2+ signaling-mitochondrial homeostasis” axis to promote proliferation and inhibit apoptosis; (2) establishing an immunosuppressive microenvironment by modulating immune cell infiltration and checkpoint molecules (e.g., TIGIT) to facilitate immune escape. The small-molecule inhibitor 2-APB targets ITPR2 to induce AML cell mitochondrial dysfunction and apoptosis, supporting ITPR2 as a potential therapeutic target. While lacking in vivo validation, this work provides a new basis for AML prognostic stratification and ITPR2-targeted strategies, with subsequent preclinical studies to solidify its clinical translation.
The data generated in the present study may be requested from the corresponding author.
XH and NL conducted most of the experiments and wrote the manuscripts. SZ collected and confirmed the authenticity of all raw data. XL analyzed the data. YH drew the pictures. JL completed the tables. ZL designed the study and critically revised the manuscript. All authors have read and approved the final version of the manuscript. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This research has been reviewed and approved by the Ethical Review Commission of the First Affiliated Hospital of Guangxi Medical University, Nanning, China (Approval code: 2025-E0668). The Ethical Review Commission of the First Affiliated Hospital of Guangxi Medical University approved this study protocol and waived the obligation for informed consent because of the retrospective nature of the study. This study was conducted in accordance with the ethical standards of the Declaration of Helsinki.
The authors sincerely thank the patients whose valuable data were vital to this research.
This work was supported by the National Natural Science Foundation of China (No. 82160039 and No. 82560035), the Natural Science Foundation of Guangxi Province (No. 2025GXNSFAA069052), the Open Research Project from Key Laboratory of Clinical Laboratory Medicine of Guangxi Department of Education (No. GXGXLCJYZDX2025007), and the Middle-aged and Young Teachers’ Basic Ability Promotion Project of Guangxi (No. 2024KY0112).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






