1 Department of Orthopaedic Surgery, Shanghai Jiao Tong University Affiliated Sixth People’s Hospital, 200233 Shanghai, China
†These authors contributed equally.
Abstract
Rotator cuff injuries are common musculoskeletal disorders and are frequently complicated by impaired tendon–bone healing and high re-tear rates after surgical repair. Exosomes derived from adipose-derived stem cells (ADSCs) have shown regenerative potential through paracrine mechanisms; however, the role of exosomal insulin-like growth factor 1 (IGF1) in tendon–bone healing remains unclear.
Exosomes were isolated from rat ADSCs with or without lentiviral knockdown of IGF1. A rat supraspinatus tendon tear and repair model was established, and 200 μg of exosomes was administered systemically post-surgery. Tendon–bone healing was evaluated at 8 weeks post-operation using histological, immunohistochemical, and micro-computed tomography analyses. Early molecular responses were assessed at 1-week post-surgery by Western blot and RT-qPCR. Angiogenic markers (vascular endothelial growth factor (VEGF), CD31, α-SMA), inflammatory cytokines (interleukin (IL)-1β, IL-18), pyroptosis-related proteins (gasdermin D N-terminal fragment (GSDMD-N)), and NLRP3 inflammasome components were examined.
ADSC-derived exosomes significantly enhanced bone mineral density, fibrocartilage formation, vascularization, and biomechanical strength at the tendon–bone interface. These effects were accompanied by reduced inflammatory cytokine expression, inhibition of pyroptosis, and suppression of NLRP3 inflammasome activation. In contrast, exosomes derived from IGF1-deficient ADSCs exhibited markedly reduced therapeutic efficacy, with attenuated angiogenic, anti-inflammatory, and anti-pyroptotic effects.
Exosomal IGF1 plays a critical role in promoting angiogenesis, suppressing inflammation and pyroptosis, and improving structural and biomechanical outcomes during tendon–bone healing. IGF1-enriched ADSC-derived exosomes represent a promising therapeutic strategy for enhancing rotator cuff repair.
Keywords
- rotator cuff injuries
- exosomes
- adipose-derived stem cells
- insulin-like growth factor 1
- tendon-bone healing
The rotator cuff is a complex anatomical structure composed of four tendons that stabilize the glenohumeral joint and facilitate normal shoulder motion [1]. Rotator cuff injuries are among the most common musculoskeletal disorders, particularly in individuals engaged in repetitive overhead activities and in aging populations [2]. These injuries range from tendinopathy and partial-thickness tears to full-thickness tendon ruptures, leading to pain, weakness, restricted range of motion, and functional impairment [3]. Despite advancements in surgical and non-surgical treatment modalities, rotator cuff tears present a significant clinical challenge due to persistently high rates of re-tear and suboptimal tendon-to-bone healing [4]. Consequently, current research is increasingly focused on understanding the biological environment of the tendon, enhancing regenerative approaches, and developing novel biomaterials to improve clinical outcomes [5].
Adipose-derived stem cells (ADSCs) have emerged as a promising source of mesenchymal stem cells (MSCs) with significant potential in regenerative medicine and tissue engineering [6]. Isolated from the stromal vascular fraction of adipose tissue, ADSCs exhibit multilineage differentiation capacity, immunomodulatory properties, and a high proliferative profile [7]. Compared with other MSC sources, such as bone marrow, ADSCs offer advantages including minimal invasiveness during harvest, higher yield, and reduced donor site morbidity [8]. ADSCs exert therapeutic effects not only through their differentiation capacity but also, increasingly, via paracrine mechanisms-especially by releasing extracellular vesicles (EVs), such as exosomes [9]. Exosomes are nanoscale vesicles (30–150 nm in diameter) derived from the endosomal pathway, which serve as carriers of bioactive components, including proteins, lipids, mRNAs, and microRNAs [10]. These vesicles mediate intercellular communication and play a critical role in regulating inflammation, stimulating angiogenesis, preventing apoptosis, and supporting tissue regeneration [11].
ADSC-derived exosomes (ADSC-exo) are gaining attention as a cell-free therapeutic modality that recapitulates many of the regenerative effects of stem cell transplantation without the associated risks of tumorigenicity, immune rejection, or vascular occlusion [12, 13]. Accordingly, in this study, our objective is to investigate the effects of ADSC-exo on angiogenesis, inflammation, and pyroptosis at the tendon-bone interface during rotator cuff repair, and to evaluate their potential to enhance rotator cuff healing after surgical repair. Furthermore, we aim to elucidate the role of the key protein insulin-like growth factor 1 (IGF1) contained within ADSC-exo.
ADSCs were isolated from the inguinal adipose tissue of 4-week-old Sprague-Dawley rats according to the protocol described in previous research [14], and the rats were euthanized in the same manner as in Section 2.6. Briefly, adipose tissue was harvested under sterile conditions, minced into approximately 1-mm3 fragments, and enzymatically digested with collagenase. The digested suspension was filtered, centrifuged, and the resulting cell pellet was resuspended and cultured in complete culture medium. Cells were maintained at 37 °C in a humidified atmosphere containing 5% CO2, and medium was refreshed every 2–3 days. ADSCs at passage 3 were used for all subsequent experiments. All primary ADSCs were validated for cell identity by surface marker analysis using flow cytometry and were confirmed to be mycoplasma-negative, ensuring the reliability and reproducibility of the experimental results.
To suppress IGF1 expression, ADSCs were transduced with lentiviral vectors encoding short hairpin RNA (shRNA) targeting rat Igf1 (GenePharma, Shanghai, China). The shRNA sequence targeting Igf1 was GGTGGATGCTCTTCAGTTC, while a non-targeting sequence (ACTTACGCTGAGTACTTCG) served as the negative control. Lentiviral transduction was performed according to the manufacturer’s instructions. Knockdown efficiency was confirmed at both mRNA and protein levels.
Exosomes were isolated from ADSC-conditioned medium using a standard
differential ultracentrifugation protocol. Briefly, culture supernatants were
sequentially centrifuged at 1000
Exosome purity was evaluated by calculating the particle-to-protein ratio (particles/µg protein).
To evaluate whether IGF1 knockdown affects ADSC viability, a Cell Counting Kit-8
(CCK-8) assay was performed. Control ADSCs and IGF1-knockdown ADSCs (shIGF1) in
the logarithmic growth phase were seeded into 96-well plates at a density of 5
To evaluate whether systemically administered exosomes can reach the
tendon–bone injury site, exosome biodistribution was assessed using DiI
fluorescent labeling. Purified ADSC-derived exosomes were incubated with the
lipophilic fluorescent dye DiI at a final concentration of 5 µM for 30 min
at 4 °C in the dark. To remove unbound dye, the labeled exosomes were
ultracentrifuged at 100,000
DiI-labeled exosomes were administered to rats with rotator cuff tear via tail-vein injection. Three days after injection, rats were euthanized in the same manner as in Section 2.6, and tendon tissues from the repair site were harvested, embedded, sectioned, and examined under a fluorescence microscope to detect DiI fluorescence signals.
The rat rotator cuff tear and repair model were established following the surgical procedure described in previous research [15, 16]. A total of 42 adult female Sprague–Dawley rats (4–5 months old, 258–552 g) were used. Animals were randomly assigned to experimental groups using a computer-generated random number table. Investigators performing outcome assessments were blinded to group allocation. General anesthesia was induced with 2–3% isoflurane in 100% oxygen. A transverse incision was made lateral to the deltoid muscle to expose the rotator cuff complex. The supraspinatus tendon was sharply detached from the greater tuberosity, and the footprint was gently debrided to simulate clinical tendon avulsion prior to repair.
Postoperatively, 200 µg of ADSC-derived exosomes suspended in 200 µL PBS were administered via tail vein injection immediately after surgery (day 0), on days 3 and 7, and once weekly thereafter until the designated endpoints. Animals were euthanized at 1 or 8 weeks post-surgery under deep anesthesia by intraperitoneal injection of sodium pentobarbital (150 mg/kg). Death was confirmed by the absence of respiration, corneal reflex, and heartbeat.
At 1 week post-surgery, tendon tissues from the repair site were harvested and
homogenized in RIPA lysis buffer. Protein samples were separated by SDS–PAGE and
transferred to PVDF membranes. Membranes were incubated with primary antibodies
against IGF1 (1:2000, #ab322659, Abcam, Cambridge, MA, USA), VEGFA (1:1000,
#ab214424, Abcam), CD31 (1:1500, #ab222783, Abcam), interleukin (IL)-1
qRT-PCR was performed using SYBR Green Master Mix (#4344463, Thermo Fisher Scientific, Waltham, MA, USA) on a CFX96 Real-Time PCR System (Bio-Rad, Hercules, CA, USA). GAPDH was used as the internal reference gene. The primer sequences used were as follows:
GAPDH:
F: 5′-TCAAGAAGGTGGTGAAGCAG-3′;
R: 5′-GGTGGAAGAGTGGGAGTTGC-3′.
Igf1:
F: 5′-CCTGCTTGCTCACCTTTACC-3′;
R: 5′-GGTAGCTCAGGCATGTCCAG-3′.
Vegfa:
F: 5′-GAGAGGTACAGTGCTGCCCT-3′;
R: 5′-CACACAGGACGGCTTGAAGA-3′.
Acta2:
F: 5′-GACCTTGAGAAGAGTTACGAGTTG-3′;
R: 5′-TAGAGAGACAGCACGATGGG-3′.
Cd31:
F: 5′-GCTGGTGCTGTTCTTCCTGT-3′;
R: 5′-AGGTGCCATCCAGGTACTTG-3′.
Il18:
F: 5′-TGCCATGTCAGAAGACTCTGC-3′;
R: 5′-TGGGTCACAGCCAGTTCTTC-3′.
Il1b:
F: 5′-TGCAGCTGGAGAGTGTGGAT-3′;
R: 5′-TGTCGTTGCTTGGTTCTCCT-3′.
At 8 weeks post-surgery, shoulder joint specimens were fixed in 4% paraformaldehyde, decalcified, embedded in paraffin, and sectioned at 5-µm thickness. Hematoxylin and eosin (H&E) staining was used to assess overall tissue morphology, while Safranin O–Fast Green staining was performed to evaluate fibrocartilage formation at the tendon–bone interface. For immunohistochemistry, sections were incubated with primary antibodies against VEGF, followed by secondary antibodies and DAB visualization to assess neovascularization.
Micro-computed tomography (micro-CT) analysis was conducted to assess bone structure and mineralization at the greater tuberosity [17]. Following scanning, 3D reconstruction was used to evaluate bone volume parameters and trabecular architecture. For biomechanical testing, the humerus-tendon-scapula complex was harvested and subjected to uniaxial tensile loading to determine maximum load to failure and stiffness, thereby assessing the mechanical integrity of the tendon-to-bone repair.
All data are presented as mean
To verify whether IGF1 knockdown affected ADSC viability, a CCK-8 assay was performed to assess cell viability. From day 1 to day 4 of culture, the relative cell viability of control ADSCs and shIGF1-transduced ADSCs increased in parallel, and no significant differences were observed at any time point (day 4: t = 0.87, df = 10, p = 0.483, Cohen’s d = 0.30; Supplementary Fig. 1). These results demonstrate that IGF1 knockdown did not impair ADSC viability.
To determine whether suppression of Igf1 in ADSCs altered IGF1 expression and
its incorporation into secreted exosomes, ADSCs were transduced with lentiviral
shRNA targeting Igf1. Quantitative RT-PCR and Western blot analyses
confirmed a significant reduction in Igf1 mRNA and IGF1 protein levels
in shIGF1-transduced ADSCs compared with controls (Fig. 1A–C; p
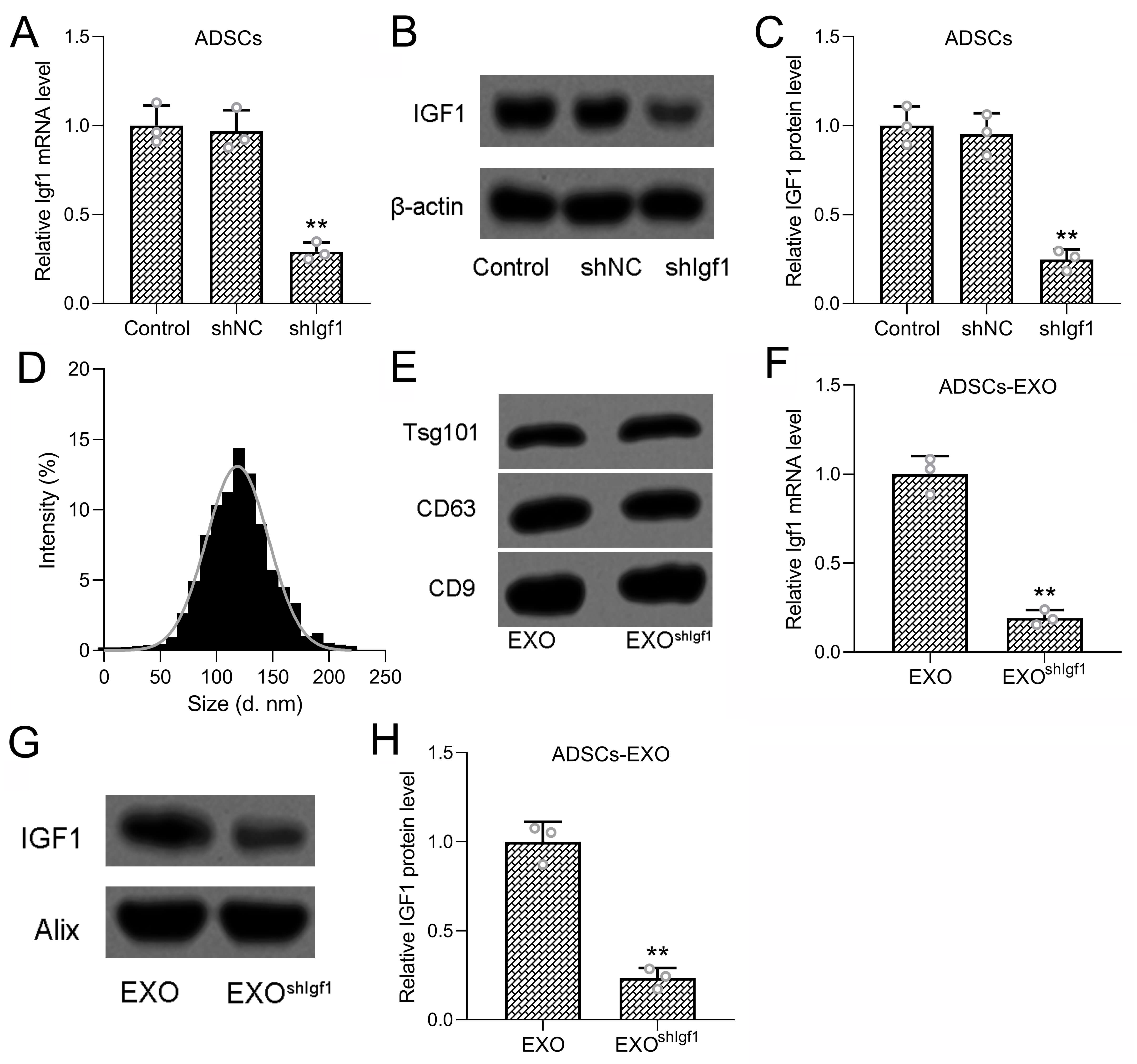 Fig. 1.
Fig. 1.
Inhibition of Igf1 in ADSCs attenuated the content of
IGF1 in ADSCs derived exosomes. (A) ADSCs were transfected with shIgf1 for 48
hours, the mRNA and protein expressions of IGF1 were measured (B,C). 3 repeats
were conducted. (D) Size distribution of the exosomes derived from ADSCs with
Igf1 inhibition. (E) Detection of Tsg101, CD63, CD9 expressions by Western
blotting analysis from exosomes derived from ADSCs and ADSCs with Igf1
inhibition. (F) Detection of Igf1 mRNA expressions by qRT-PCR from
exosomes derived from ADSCs and ADSCs with Igf1 inhibition. (G,H) Detection of
IGF1 and Alix protein expressions by Western blotting analysis from exosomes
derived from ADSCs and ADSCs with Igf1 inhibition. 3 repeats were conducted. Data
was shown with mean
Exosomes isolated from control and shIGF1 ADSCs were subsequently characterized. Western blotting further confirmed the presence of canonical exosomal markers TSG101, CD63, and CD9, with no detectable differences between control and shIGF1-derived exosomes (Fig. 1D,E), indicating that Igf1 knockdown did not affect exosome biogenesis or structural integrity. To further evaluate whether Igf1 knockdown affects exosome yield and purity, particle concentration and total protein content of isolated exosomes were quantified, and the particle-to-protein ratio was calculated. No significant differences were observed between control ADSC-derived exosomes (EXO) and Igf1-knockdown ADSC-derived exosomes (EXOshIGF1) in particle concentration, protein concentration, or particle-to-protein ratio (Supplementary Fig. 2A–C). These results further indicate that Igf1 knockdown does not affect basic exosome biogenesis, yield, or purity, but specifically reduces IGF1 cargo within the exosomes.
Importantly, analysis of exosomal cargo revealed a substantial reduction in IGF1
content following Igf1 knockdown. qRT-PCR demonstrated significantly decreased
Igf1 mRNA levels in exosomes derived from shIGF1-transduced ADSCs
compared with controls (Fig. 1F; p
Given that exosomes were administered systemically in this study, it was first necessary to determine whether intravenously delivered exosomes could reach the tendon–bone injury site. Accordingly, DiI-labeled ADSC-derived exosomes were injected via the tail vein. Distinct DiI fluorescence signals were detected in tendon tissues three days after injection, indicating that systemically delivered exosomes are able to successfully reach and persist at the injury site (Supplementary Fig. 3).
To assess the structural and functional outcomes of rotator cuff repair,
micro-CT and biomechanical analyses were performed at 8 weeks post-surgery.
Micro-CT analysis demonstrated that treatment with exosomes derived from control
ADSCs (EXO group) significantly improved bone microarchitecture at the
tendon–bone interface compared with the untreated model group
(Supplementary Fig. 4). Specifically, bone mineral density (BMD), bone
volume fraction (BV/TV), and trabecular spacing (Tb.Sp) were all significantly
improved in the EXO group (Fig. 2A–C; Brown–Forsythe ANOVA: F = 20.37, df =
2.15, partial
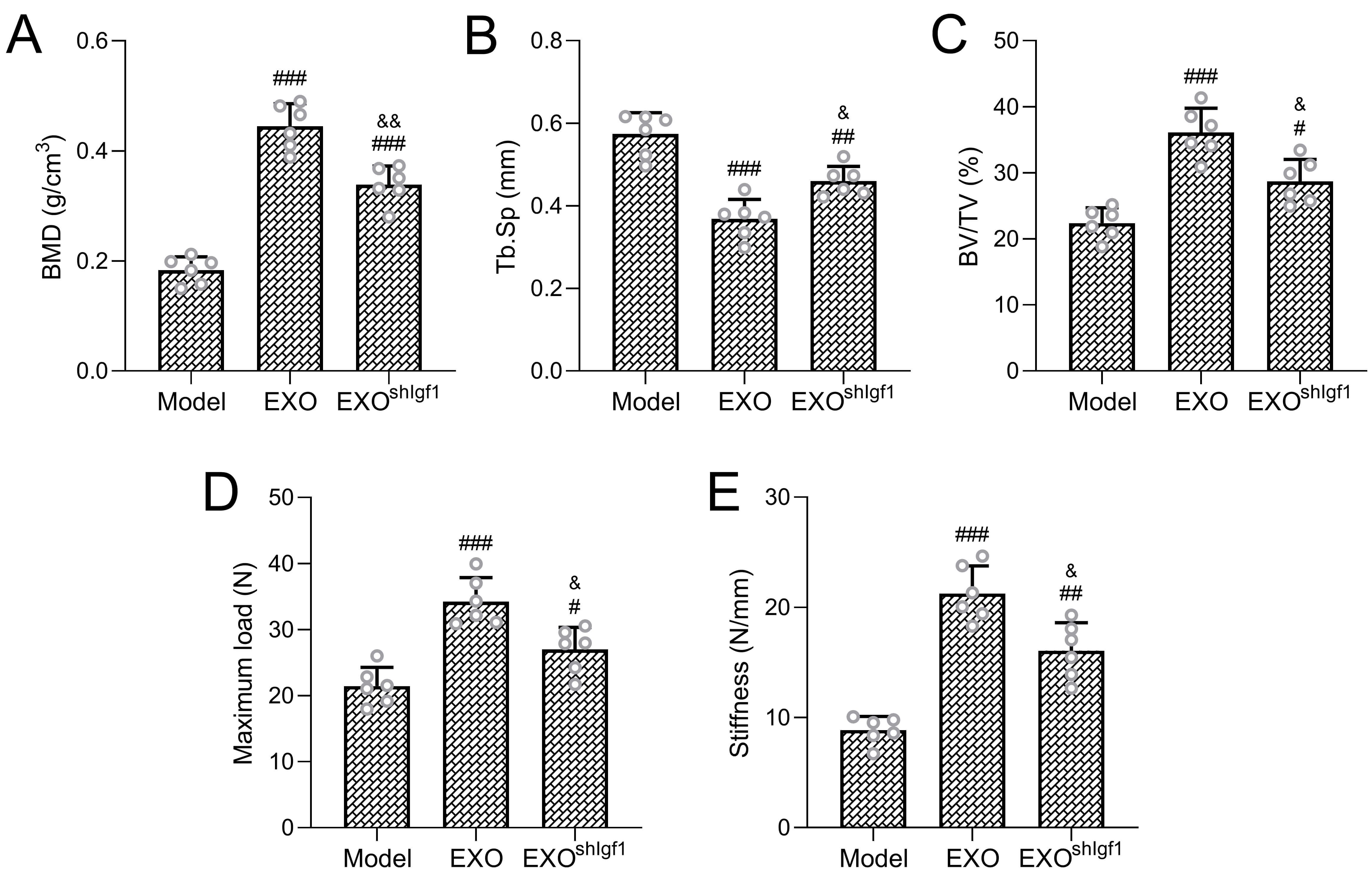 Fig. 2.
Fig. 2.
Therapeutic efficacy in rotator cuff repair. Micro-computed
tomography was used to evaluate bone remodeling at the tendon–bone interface,
including bone mineral density (BMD; A), trabecular separation (Tb.Sp; B), and
bone volume fraction (BV/TV; C). Mechanical properties of the repaired rotator
cuff were assessed by measuring ultimate failure load (D) and stiffness (E). Six
rats were included in each experimental group. Data are shown as mean
Consistent with the micro-CT findings, biomechanical testing revealed that the
maximum load to failure and interfacial stiffness were significantly higher in
the EXO group than in the untreated model group, confirming enhanced mechanical
integration of the repaired tendon (Fig. 2D,E; Brown–Forsythe ANOVA: F = 22.68,
df = 2.15, partial
To further evaluate histological remodeling at the tendon–bone interface
following rotator cuff repair, H&E staining and SO–FG staining were performed
on specimens harvested 8 weeks post-surgery. Significantly lower scores were
revealed in the EXO group compared with the model group, indicating reduced
inflammatory features and more orderly fibrocartilage structure (Fig. 3A,B;
Brown–Forsythe ANOVA: F = 16.82, df = 2.15, partial
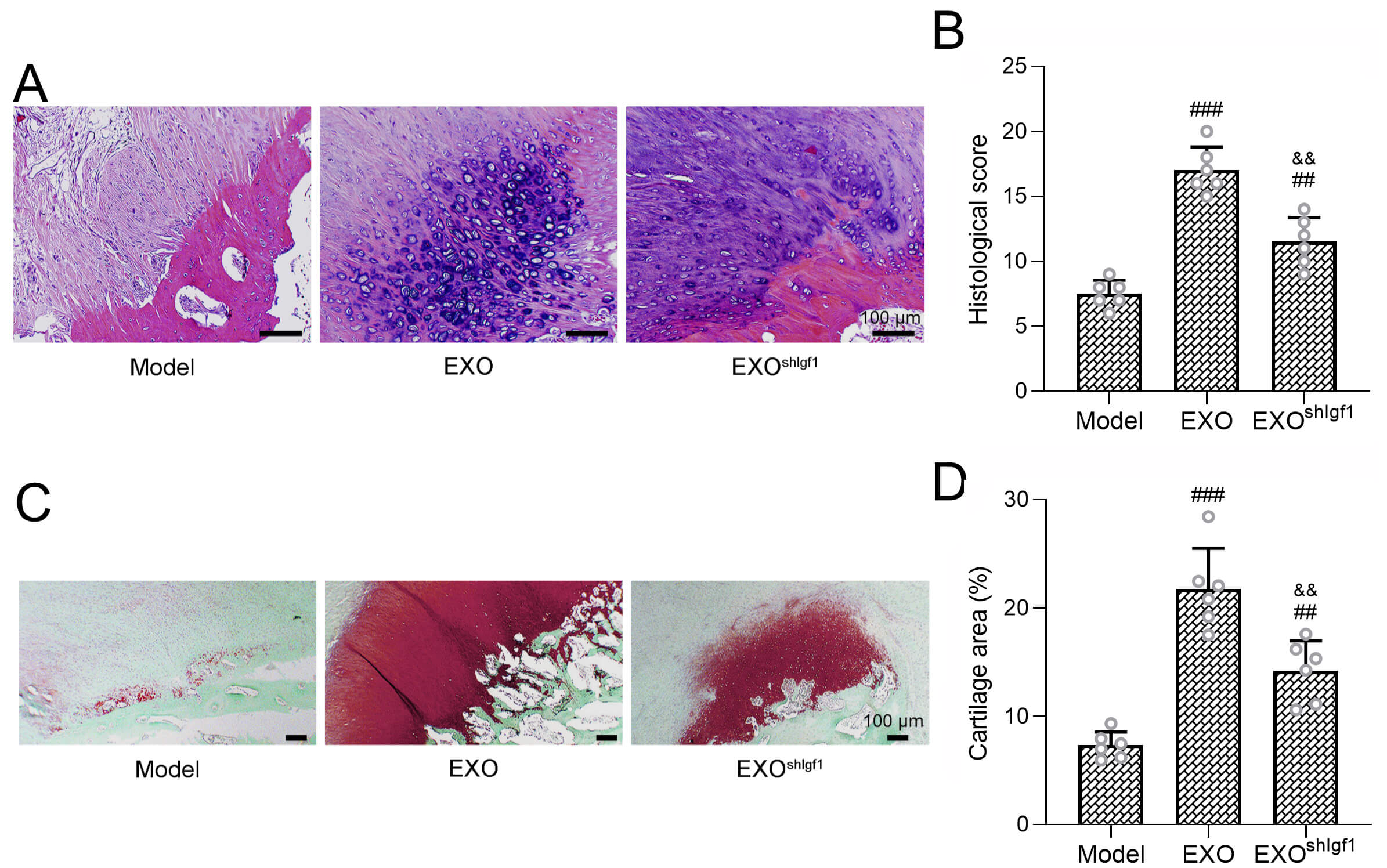 Fig. 3.
Fig. 3.
Tissue organization and fibrocartilage formation at the
repair site. (A) Representative histological sections stained with hematoxylin
and eosin (H&E), and (B) quantitative evaluation of overall histological scores.
Scale bar = 100 µm. (C) Representative Safranin O–Fast green (SO–FG), and
(D) Safranin O–positive cartilage area. Scale bar = 100 µm. Six rats were
included in each group. Results are expressed as mean
To investigate the contribution of ADSC-derived exosomes to angiogenesis during
tendon–bone healing, immunohistochemical staining and gene expression analyses
were performed on tendon tissues harvested 8 weeks after surgery. A significant
increase in VEGF-positive staining was demonstrated in the EXO group (Fig. 4A,B,
Brown–Forsythe ANOVA, F = 14.92, df = 2.15, partial
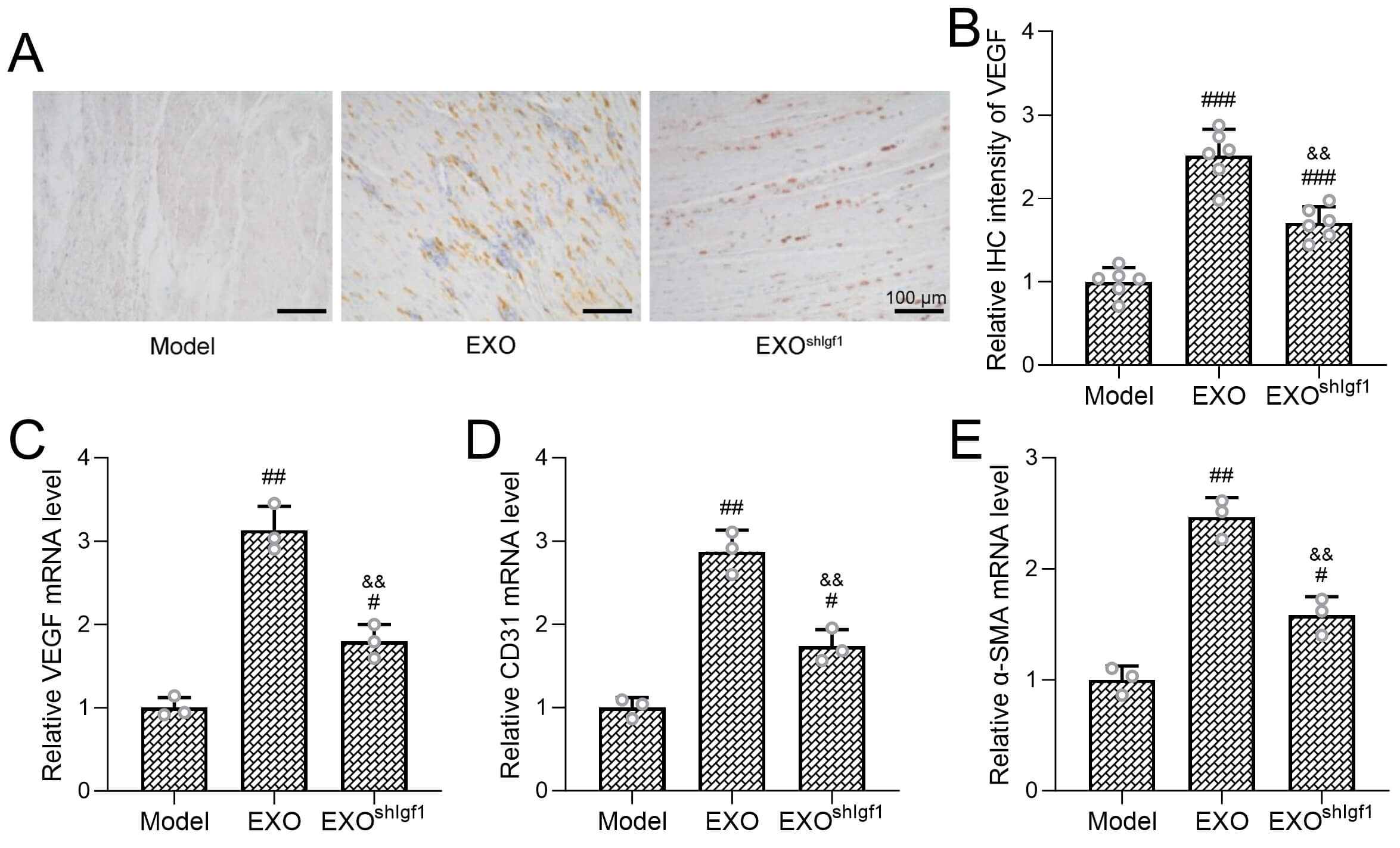 Fig. 4.
Fig. 4.
Igf1 Deficiency decreases exosome’ pro-angiogenic effects in
rotator cuff repair. (A) Representative images of immunohistochemical staining
of vascular endothelial growth factor (VEGF), and (B) intensity of VEGF
in tendon tissues. Scale bar = 100 µm. Transcriptional levels of
angiogenesis-related genes, including Vegfa (C), Cd31 (D), and
In contrast, the pro-angiogenic responses were substantially attenuated when exosomes were derived from Igf1-deficient ADSCs. Compared with the EXO group, the shIGF1-EXO group exhibited significantly reduced VEGF staining intensity as well as lower transcript levels of Vegfa, Cd31, and Acta2. These findings indicate that IGF1 plays a critical role in mediating the angiogenic activity of ADSC-derived exosomes in the context of rotator cuff repair.
To evaluate the effects of ADSC-derived exosomes on tendon cell pyroptosis
following rotator cuff injury, tendon tissues were harvested 1 week after surgery
for propidium iodide (PI) staining and Western blot analysis. A sham-operated
group was included as a baseline control. Compared with the sham group, the
untreated model group exhibited a marked increase in PI-positive cells,
indicating enhanced pyroptotic activity in the injured tendon tissue (Fig. 5A,B).
Treatment with exosomes derived from control ADSCs (EXO group) significantly
reduced the number of PI-positive cells relative to the model group, suggesting
an inhibitory effect of exosomes on tendon cell pyroptosis during the early
healing phase (Fig. 5B; Brown–Forsythe ANOVA: F = 17.25, df = 3.20, partial
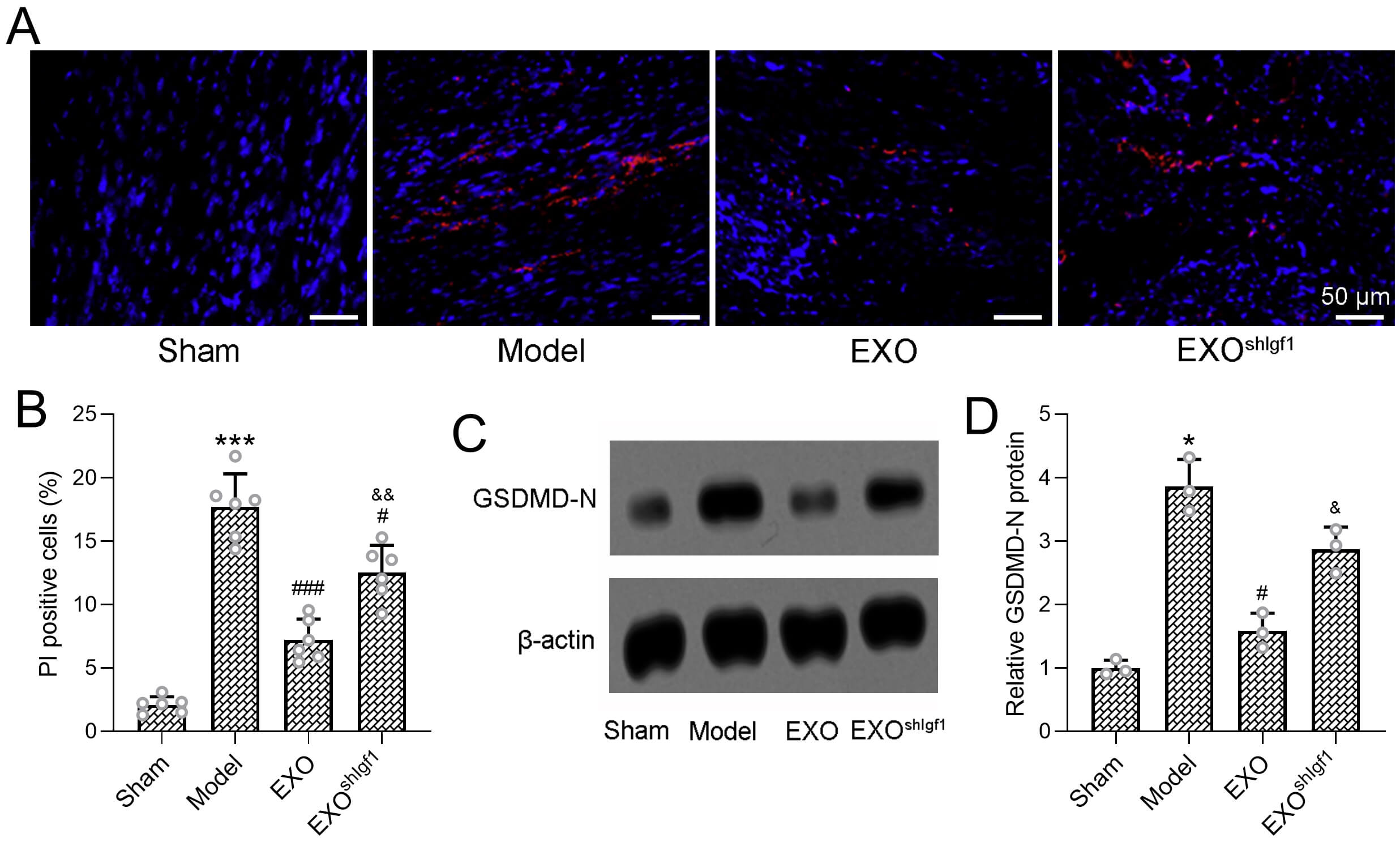 Fig. 5.
Fig. 5.
Igf1-deficient exosomes exhibit reduced inhibitory effects on
pyroptosis. (A) Representative propidium iodide (PI) fluorescence
staining of tendon sections, and (B) quantitative analysis revealed PI-positive
cells in membrane-compromised cells. Scale bar = 50 µm. Protein expression
of the pyroptosis executor gasdermin D N-terminal fragment (GSDMD-N) in tendon
tissues was assessed by Western blotting (C).
Consistent with these findings, Western blot analysis demonstrated a pronounced
upregulation of gasdermin D N-terminal fragment (GSDMD-N), a key executor of
pyroptosis, in the model group, whereas exosome treatment markedly suppressed
GSDMD-N expression toward baseline levels (Fig. 5C). Quantitative analysis of
GSDMD-N normalized to
Notably, the anti-pyroptotic effects of exosome treatment were significantly attenuated when exosomes were derived from Igf1-knockdown ADSCs. Compared with the EXO group, the shIGF1-EXO group exhibited a higher proportion of PI-positive cells and significantly elevated GSDMD-N protein levels. These results indicate that IGF1 is required for the exosome-mediated suppression of tendon cell pyroptosis during the early stage of rotator cuff healing.
To further examine the regulatory effects of ADSC-derived exosomes on the
inflammatory response associated with NLRP3 inflammasome activation and
pyroptosis, the protein and mRNA expression levels of the pro-inflammatory
cytokines IL-1
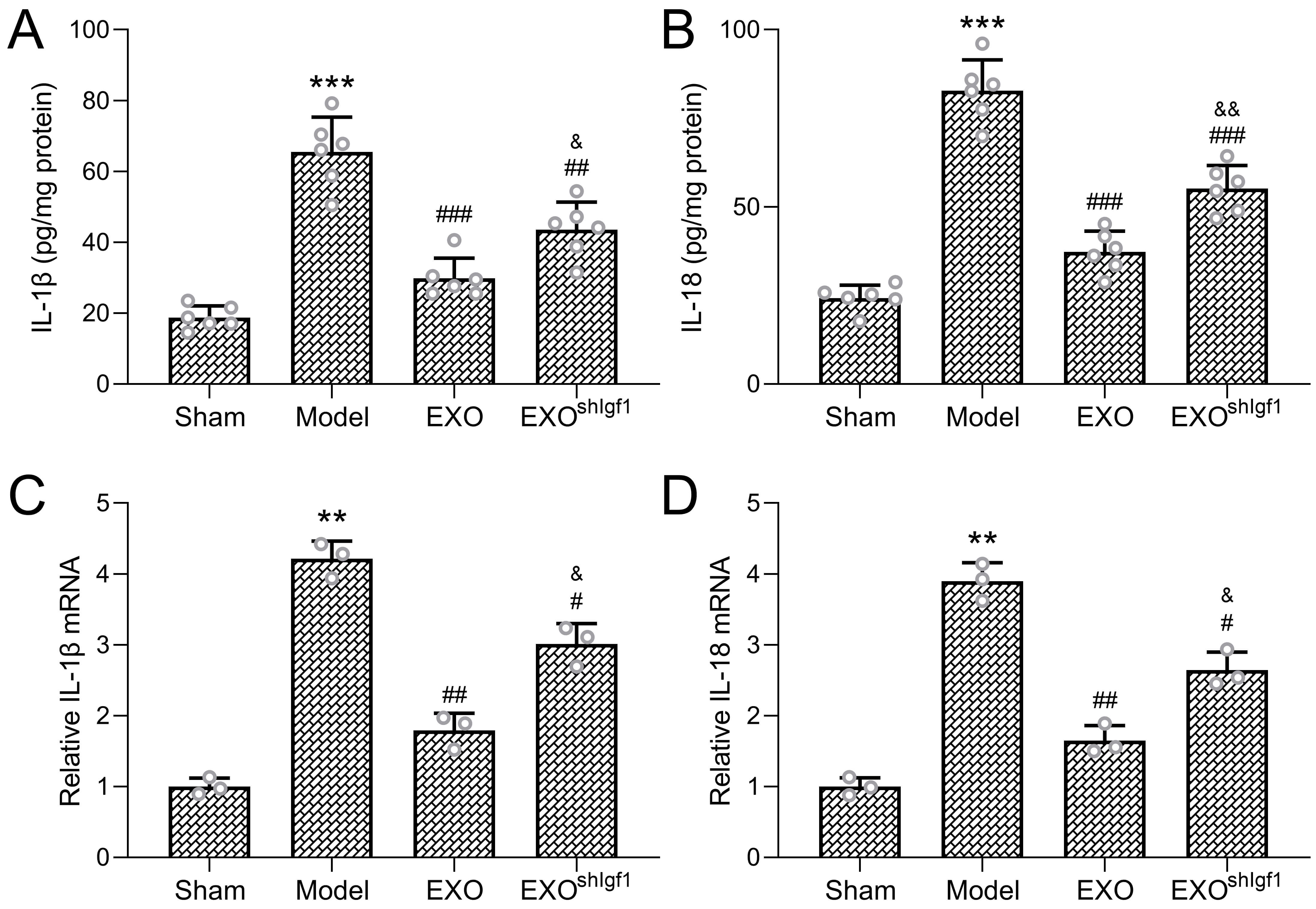 Fig. 6.
Fig. 6.
Igf1 Knockdown attenuates the anti-inflammatory
effects. Protein concentrations of the pro-inflammatory cytokines
interleukin-1
Notably, these anti-inflammatory effects were substantially attenuated when
exosomes were derived from Igf1-inhibited ADSCs. Compared with the EXO group, the
shIGF1-EXO group exhibited significantly higher levels of IL-1
To investigate the influence of ADSC-derived exosomes on NLRP3 inflammasome
activation in the early phase of tendon healing, Western blot analysis was
performed on tendon tissues collected one-week post-surgery. The NLRP3
inflammasome is a critical innate immune signaling complex that detects
pathogenic stimuli or cellular stress. Once activated, NLRP3 recruits the adaptor
protein ASC through PYD–PYD interactions. ASC then facilitates the conversion of
pro-caspase-1 into its active form, triggering the release of pro-inflammatory
cytokines and initiating pyroptosis [18, 19, 20]. Compared to the sham group, the
model group showed marked upregulation of NLRP3 (Fig. 7A,B, Brown–Forsythe ANOVA,
F = 13.25, df = 3.8, partial
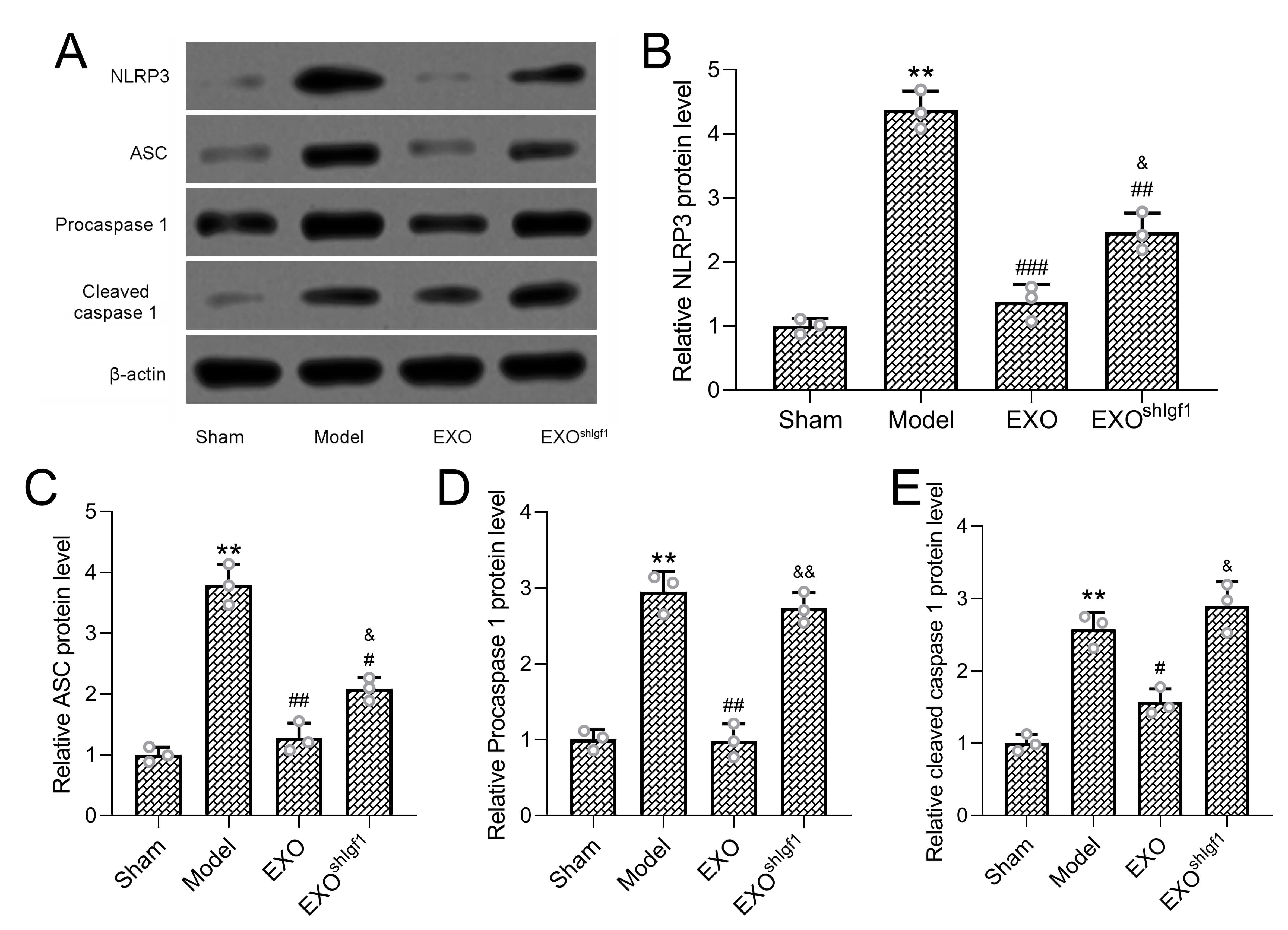 Fig. 7.
Fig. 7.
Effect of ADSC-Derived exosomes on NLRP3 inflammasome
activation. Protein levels of NLRP3 inflammasome–related components, including
NLRP3, ASC, pro-caspase-1, and cleaved caspase-1, were examined in tendon tissues
by Western blot analysis (A).
Treatment with exosomes derived from control ADSCs (EXO group) significantly suppressed the expression of these inflammasome-related proteins compared with the model group, indicating effective inhibition of NLRP3 inflammasome activation. In contrast, this suppressive effect was markedly attenuated when exosomes were derived from Igf1-knockdown ADSCs. Compared with the EXO group, the shIGF1-EXO group exhibited significantly higher levels of NLRP3, ASC, and cleaved caspase-1, demonstrating incomplete inhibition of inflammasome signaling.
Collectively, these findings indicate that IGF1 is essential for the ability of ADSC-derived exosomes to suppress NLRP3 inflammasome activation during the early inflammatory phase of rotator cuff tendon healing, thereby linking exosomal IGF1 to coordinated regulation of inflammation, pyroptosis, and tissue repair.
In the present study, we demonstrate that exosomes derived from rat ADSCs significantly promote tendon–bone healing in a rat model of rotator cuff injury. Importantly, our findings indicate that these therapeutic effects are mediated, at least in part, by IGF1 contained within the exosomes. ADSC-derived exosomes promoted structural regeneration, and angiogenesis, while concurrently suppressing inflammation and pyroptosis. Notably, exosomes derived from IGF1-silenced ADSCs exhibited markedly reduced therapeutic efficacy, supporting a critical functional role for IGF1 in exosome-mediated tendon–bone repair.
Rotator cuff injuries often result in incomplete healing due to insufficient biological reattachment between tendon and bone, leading to high rates of re-tear [21]. Conventional surgical repair methods restore anatomical continuity but do not adequately address the biological deficiencies at the repair interface [22]. Our results support the use of ADSC-derived exosomes as a promising cell-free regenerative strategy capable of enhancing the biological healing response. Exosome treatment significantly improved bone microarchitectural parameters, including BMD and BV/TV, increased biomechanical strength at the tendon–bone junction, and promoted fibrocartilage formation with improved tissue organization. These findings are consistent with accumulating evidence that mesenchymal stem cell–derived exosomes exert potent regenerative effects in musculoskeletal tissues.
IGF1 has been shown to play a key role in tendon biology by promoting matrix
synthesis, cell proliferation, and neovascularization [23, 24]. In the present
study, exosome treatment significantly enhanced neovascularization at the repair
site, as evidenced by increased VEGF immunoreactivity and upregulated expression
of angiogenic markers such as VEGF, CD31, and
Beyond structural and vascular regeneration, we found that ADSC-exosomes attenuates pyroptosis, a form of inflammatory cell death implicated in chronic tissue damage. The expression of GSDMD-N, a marker of pyroptosis [25], was elevated in injured tendons but significantly decreased with exosome treatment. Additionally, markers of NLRP3 inflammasome activation—including NLRP3, ASC, and cleaved caspase-1-were suppressed by exosome therapy, indicating that ADSC-derived exosomes modulate early inflammatory signaling pathways. IGF1 knockdown largely diminished these effects, further highlighting its central role in the anti-pyroptotic and anti-inflammatory action of exosomes.
Collectively, our findings suggest that IGF1 within ADSC-derived exosomes is not merely a positive biomarker, but rather a functionally active cargo that contributes substantially to therapeutic efficacy. Rather than acting through a single pathway, exosomal IGF1 appears to participate in a coordinated, multifaceted regenerative process that includes enhancement of angiogenesis, suppression of inflammasome-driven inflammation and pyroptosis, and support of extracellular matrix remodeling. Such integrative regulation is particularly advantageous in complex tendon–bone interfaces, where successful healing requires tightly coordinated cellular and molecular responses.
Several limitations of this study should be acknowledged. First, the experiments were conducted in a rat model, and although this model is well established, healing dynamics differ between rodents and humans. Validation in large animal models will be necessary before clinical translation. Second, the present study focused on short- to mid-term outcomes (1–8 weeks), and long-term evaluation of tendon integrity, mechanical durability, and re-tear rates remains to be performed. Third, while we identified IGF1 as a key functional component of ADSC-derived exosomes, the downstream signaling pathways in recipient cells—such as PI3K–Akt or MAPK signaling—were not directly investigated. Future studies should explore the cell-type–specific mechanisms by which IGF1-containing exosomes influence tenocytes, endothelial cells, macrophages, and resident stem cells. Finally, although systemic (tail vein) administration was selected for its clinical feasibility and was shown to allow exosomes to reach the tendon tissue, alternative local or biomaterial-assisted delivery strategies may further enhance therapeutic efficiency and warrant future investigation.
In conclusion, this study identifies IGF1 as a key bioactive component of ADSC-derived exosomes and demonstrates their therapeutic potential as a cell-free strategy for promoting tendon–bone healing after rotator cuff injury. By enhancing angiogenesis, suppressing inflammation and pyroptosis, and improving structural and mechanical integration at the tendon–bone interface, IGF1-containing exosomes significantly improve repair outcomes in a preclinical model. These findings provide a strong rationale for the further translational development of engineered exosome-based therapies for musculoskeletal regeneration.
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
FW designed the research study and wrote the initial draft. WQ, YJ, HS, YL and FW conducted experiments. WQ and YJ analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was approved by Shanghai Jiao Tong University Affiliated Sixth People’s Hospital (KFJ200887), this study was performed in strict accordance with the NIH guidelines for the care and use of laboratory animals (NIH Publication No. 85-23 Rev. 1985).
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL47168.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







