1 Department of Radiobiology, Cancer Research Institute, Biomedical Research Center, Slovak Academy of Sciences, 845 05 Bratislava, Slovakia
2 Department of Laboratory Medicine, Faculty of Health Sciences, Catholic University in Ružomberok, 034 01 Ružomberok, Slovakia
†These authors contributed equally.
Abstract
Accurate biodosimetry is essential for effective radiological triage, precise clinical monitoring, and assessment of risks from diagnostic exposures. Immunofluorescent detection of phosphorylated histone H2AX (γH2AX) and p53-binding protein 1 (53BP1) DNA double-strand break repair foci provides high sensitivity for radiation dose assessment within the first hours after exposure. However, inter-laboratory reproducibility of γH2AX/53BP1-based biodosimetry remains limited, and the contribution of pharmacological modifiers is unresolved.
Here, we conducted an inter-laboratory comparison of in vitro radiation dose–response relationships of foci yields measured in cryopreserved umbilical cord blood lymphocytes (UCBLs) and freshly isolated peripheral blood lymphocytes (PBLs) from healthy donors using fluorescent microscopy with emphasis on workflow harmonization and reproducibility across laboratories.
Under low-dose γ irradiation, both UCBLs and PBLs exhibited a strong linear, dose-dependent induction of γH2AX, 53BP1, and co-localized foci, with co-localization emerging as the most sensitive endpoint. γH2AX pan-nuclear staining was observed exclusively in UCBLs and functioned as a distinct endpoint under the examined conditions. Under harmonized low-dose conditions, calyculin A at a non-toxic concentration of 1 nM did not provide measurable stabilization or enhancement of ionizing radiation-induced foci (IRIF) yields. Although IRIF yields differed between the two laboratories, dose–response slopes were highly concordant, demonstrating reproducibility under harmonized experimental conditions.
These findings demonstrate that inter-laboratory reproducibility of γH2AX/53BP1-based biodosimetry is achieved primarily through disciplined workflow harmonization rather than through pharmacological enhancement. By reinforcing assay reproducibility and biological consistency, this work supports the translational applicability of IRIF-based biodosimetry for broader application in radiation exposure assessment.
Keywords
- DNA double-strand breaks
- DNA damage response
- H2AX histone
- 53BP1 protein
- calyculin A
- biodosimetry
- lymphocytes
- phosphatase inhibitors
- ionizing radiation
- immunofluorescence
Accidental or deliberate exposure to ionizing radiation (IR) remains a serious public health concern, particularly in the context of nuclear accidents, radiological emergencies, and increasing geopolitical instability. Consequently, rapid, sensitive, and reliable biodosimetric tools are indispensable for population triage following radiological events [1]. Beyond emergency response, biodosimetry is increasingly applied in clinical practice, for example, for individualization of radiotherapy and monitoring of patient responses to IR [2, 3]. In addition, sensitive biodosimetric approaches are required for the detection and evaluation of low-dose radiation exposures originating from diagnostic imaging procedures, including conventional X-rays [4, 5], computed tomography [6, 7], nuclear medicine [8, 9, 10], and other radiological examinations [11, 12, 13, 14, 15, 16].
Among currently available assays, immunofluorescent detection of DNA
double-strand break (DSB) repair foci, particularly those marked by
phosphorylated histone H2AX (
In an effort to overcome these limitations, pharmacological modulation of DNA
damage signaling pathways has been explored [28]. Among several phosphatase
inhibitors, including fostriecin and okadaic acid, calyculin A (cal A) has
attracted particular attention. Cal A is a potent inhibitor of serine/threonine
protein phosphatases 1 and 2A (PP1/PP2A) [29, 30] and has been reported to
stabilize
Beyond practical biodosimetric implications, cal A interferes with fundamental
DNA damage response mechanisms. Through inhibition of serine/threonine
phosphatases, cal A can alter chromatin-associated signaling pathways,
complicating the interpretation of radiation-induced foci (IRIF)-based readouts.
In particular, apoptosis-associated
Our previous work directly examined the effects of cal A on
All experimental procedures were designed to ensure maximal reproducibility between laboratories. Wherever possible, identical reagents, suppliers, incubation times, irradiation conditions, and image acquisition parameters were used. Methodological details are provided to explicitly document harmonization steps critical for inter-laboratory comparison rather than to serve as a procedural manual.
Reagent grade chemicals were obtained from Sigma-Aldrich (St. Louis, MO, USA), Merck KGaA (Darmstadt, Germany) and Life Technologies (Carlsbad, CA, USA). Cal A was purchased from Abcam Biochemicals (Cambridge, UK) and dissolved in dimethyl sulfoxide (DMSO; SERVA Electrophoresis GmbH, Heidelberg, Germany).
UCB mononuclear cells (MNC) were extracted as previously described [27] from 3 healthy newborns after full-term pregnancies and provided by Eurocord-Slovakia, Bratislava, Slovakia. In Laboratory 1, cryopreserved UCB MNC from three donors were thawed using standard protocols and cultured in RPMI 1640-based basal medium supplemented with fetal bovine serum and antibiotics. Monocytes were removed by short-term adherence.
In Laboratory 2, UCB MNC from three independent donors obtained under identical inclusion criteria were analyzed as part of a previously published dataset [36] and used here exclusively for inter-laboratory comparison at matched irradiation doses and time points. Except for fixation timing, identical cell handling and processing steps were applied in both laboratories.
Peripheral blood (PB) MNC were isolated from six healthy adult donors by density-gradient centrifugation as previously described [39]. Monocytes were removed by adherence, yielding predominantly lymphocyte populations. Cell viability exceeded 95% in all experiments as assessed by Trypan blue exclusion.
All experiments were performed using primary human mononuclear cells isolated directly from umbilical cord blood or peripheral blood of healthy donors. These cells were not cultured or passaged and were used immediately after isolation (PBL) or after standardized cryopreservation and thawing (UCBL). Cell identity was ensured by established lymphocyte isolation procedures, donor documentation, and ethical approval. Cell morphology was examined by light microscopy, and cell viability exceeded 95% as assessed by trypan blue exclusion. As the study did not involve immortalized cell lines or long-term cell culture, STR profiling and routine mycoplasma testing were not applicable.
Following equilibration, lymphocytes were divided into three treatment groups: cal A (1 nM), DMSO vehicle control (0.1%), and untreated controls. Cells were incubated on ice for approximately 40 min prior to irradiation to synchronize handling between laboratories. To avoid prolonged exposure-related toxicity, cal A and DMSO were added immediately after irradiation in both laboratories, consistent with our previous observations [36].
Cells were irradiated on ice using acute
At defined time points post-irradiation (0.5, 1, and 2 h for UCBL; 2, 4, and 6 h
for PBL), cells were cytocentrifuged onto poly-L-lysine-coated slides, fixed with
paraformaldehyde, permeabilized, and blocked according to standard protocols.
Image acquisition was performed in both laboratories using Metafer Slide
Scanning Systems V 3.9 (MetaSystems GmbH, Altlussheim, Germany) coupled to Zeiss
Axio Imager fluorescence microscopes (Carl Zeiss Microscopy GmbH, Gottingen,
Germany) under identical acquisition settings. For each condition, at least 600
cells were analyzed. Automated scoring of
The yields of foci (Y) were expressed per cell. At each experimental dose point
in cells of each blood donor the foci-per-cell distribution was checked for
compliance with the Poisson model by
Dose-response relationships for the foci yield were fitted by the iteratively reweighted least squares method to the linear model:
where Y - foci yield per cell; c - the background foci level;
For each UCBL donor, the foci-per-cell distributions in unirradiated and irradiated samples were consistent with Poisson statistics, as expected for homogeneous exposure to low-LET radiation (data not shown). Donor-specific regression coefficients are summarized in Supplementary Table 1, and pooled regressions for each laboratory are shown in Table 1 and Fig. 1.
| End-point | Laboratory 1 | Laboratory 2 | ||||
| c |
r | c |
r | |||
| 0.4199 |
7.2150 |
0.9976 | 0.7105 |
7.4690 |
0.9987 | |
| 53BP1 | 0.2436 |
7.6600 |
0.9491 | 1.0829 |
5.1370 |
0.9951 |
| 0.2021 |
6.0960 |
0.9778 | 0.3081 |
4.2980 |
0.9958 | |
Values represent coefficients of the linear dose-response model Y =
c (
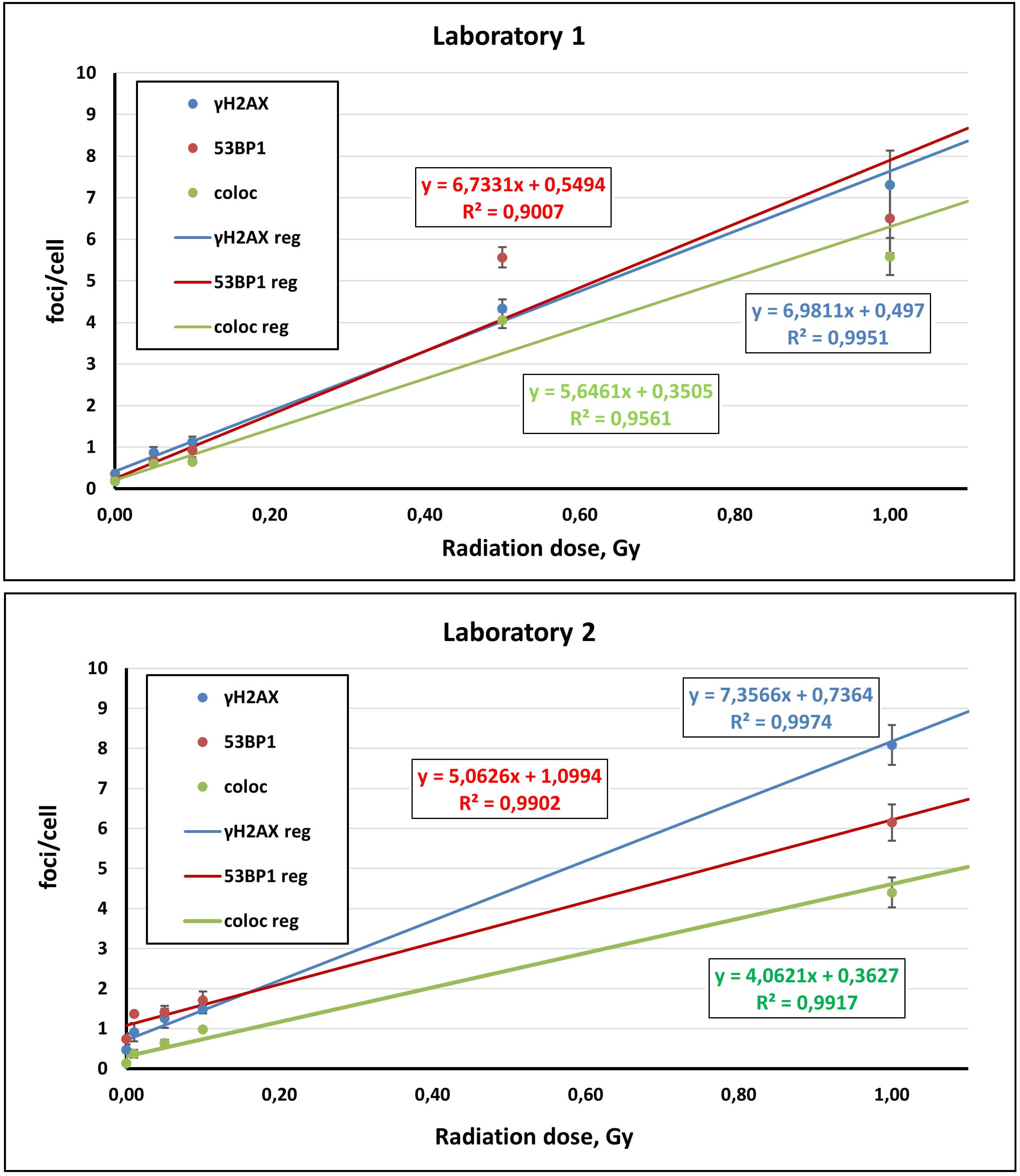 Fig. 1.
Fig. 1.
Linear dose-response relationships for
Across both laboratories,
Importantly, because the two laboratories quantified foci at different
post-irradiation times (0.5 h vs 2 h), absolute foci yields between laboratories
are not interpreted as direct head-to-head “inter-laboratory disagreement”.
Instead, the inter-laboratory comparison is focused on (i) the reproducibility of
the dose-response form (linearity), (ii) the similarity of regression slopes
(
Together, these results establish that the
Laboratory 1 assessed the effects of irradiation dose (0, 10, 50, 100 mGy),
treatment condition (cal A, DMSO vehicle, untreated), and post-irradiation time
(0.5, 1, 2 h) on
| Variables | Factorial ANOVA | Categorical factors | p-values adjusted by FDR | ||||||||
| 53BP1 * | Multivariate | Univariate | Dose | Treatment | Post-irradiation Time | Dose | Treatment | Post-irradiation Time | |||
| + | + | + | + | + | – | + | + | + | 0.00000 | 0.38884 | 0.20207 |
| + | – | – | – | – | + | + | + | + | 0.00000 | 0.72719 | 0.65372 |
| – | + | – | – | – | + | + | + | + | 0.00000 | 0.29701 | 0.13051 |
| – | – | + | – | – | + | + | + | + | 0.00000 | 0.84937 | 0.43122 |
| – | – | – | + | – | + | + | + | + | 0.59673 | 0.40271 | 0.33894 |
Categorical factors: radiation dose (Dose), cal A/DMSO treatments (Treatment), and post-irradiation time (0.5, 1, and 2 h). Where applicable, multivariate (across foci endpoints) and univariate tests are shown. Reported p-values are FDR-adjusted. * mean, foci per cell; + the data were included in analysis; – data were not included in analysis. Statistically significant values are marked in red. UCBL, umbilical cord blood lymphocytes; FDR, false discovery rate.
Univariate testing confirmed a highly significant dose dependence for
Untreated UCBL were compared with matched DMSO-treated samples at identical doses and time points. No significant differences were detected, validating DMSO as an appropriate vehicle control. For clarity, untreated data were omitted from Fig. 2, which focuses on direct cal A vs DMSO comparisons. As shown in Fig. 2, foci yields are presented across multiple doses, time points, and treatments to illustrate overall assay behavior under the experimental design; no direct quantitative comparison between post-irradiation time points is intended.
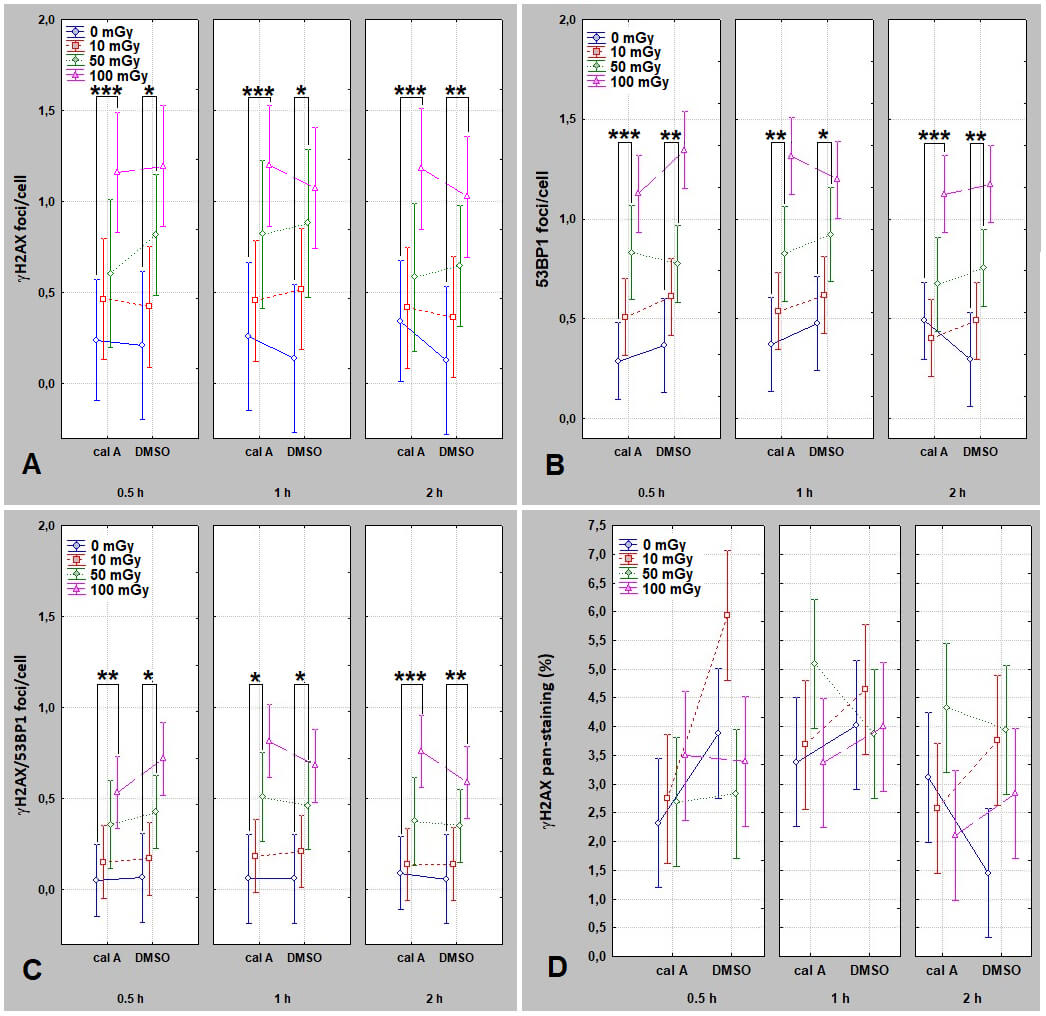 Fig. 2.
Fig. 2.
Dose- and time-dependent induction of DNA repair foci in UCBL
treated with DMSO or cal A - Laboratory 1. Panels show mean
As shown in Fig. 2,
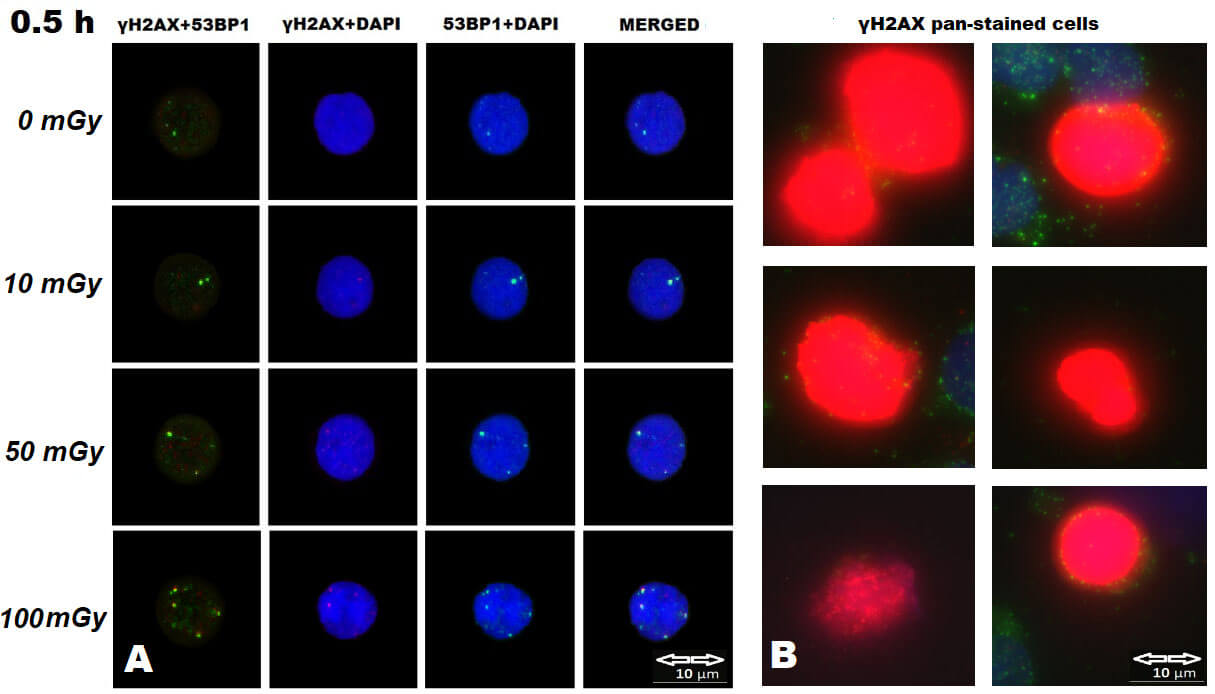 Fig. 3.
Fig. 3.
Representative immunofluorescence images of UCBL nuclei analyzed
for
To enable a time-matched inter-laboratory comparison, we evaluated UCBL data acquired at 2 h post-irradiation (Laboratory 1:2 h time point from the present study; Laboratory 2:2 h dataset reported previously and included here specifically for comparative evaluation). A three-way factorial ANOVA (Laboratory, Treatment, Dose) was performed (Table 3).
| Variables | Factorial ANOVA | Categorical factors | p-values adjusted by FDR | ||||||||
| 53BP1 * | Multivariate | Univariate | Laboratory | Treatment | Dose | Laboratory | Treatment | Dose | |||
| + | + | + | + | + | – | + | + | + | 0.00000 | 0.32265 | 0.00000 |
| + | – | – | – | – | + | + | + | + | 0.00000 | 0.38422 | 0.00000 |
| – | + | – | – | – | + | + | + | + | 0.00000 | 0.93173 | 0.00000 |
| – | – | + | – | – | + | + | + | + | 0.00001 | 0.29985 | 0.00000 |
| – | – | – | + | – | + | + | + | + | 0.23427 | 0.07822 | 0.54648 |
Categorical factors: laboratory relevant (Laboratory), radiation dose (Dose), and cal A/DMSO treatments (Treatment). Where applicable, multivariate (across foci endpoints) and univariate tests are shown. Reported p-values are FDR-adjusted. * mean, foci per cell; + the data were included in analysis; – data were not included in analysis. Statistically significant values are marked in red. UCBL, umbilical cord blood lymphocytes; FDR, false discovery rate.
A significant laboratory factor was detected for
Fig. 4 visualizes the 2 h comparison and confirms three core outcomes: (i)
robust dose-dependent induction of
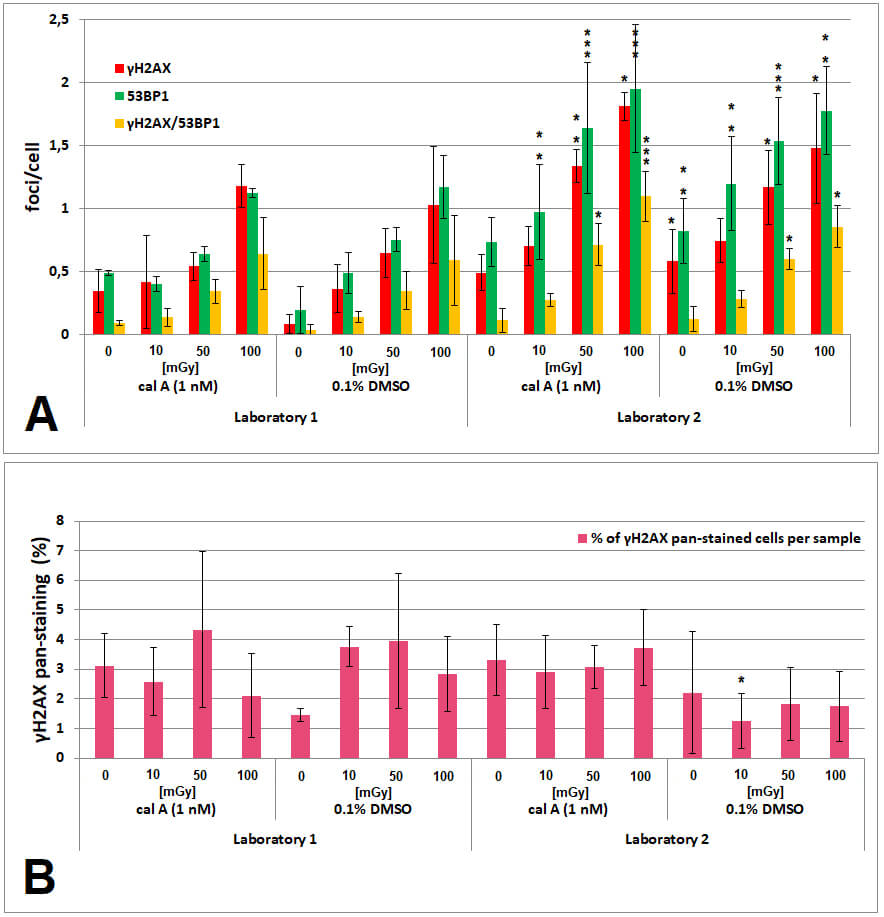 Fig. 4.
Fig. 4.
Inter-laboratory comparison of
Taken together, the time-matched comparison shows that while absolute yields can differ between laboratories, dose-response relationships and the qualitative treatment conclusion (no cal A stabilization at 1 nM) are reproducible under harmonized workflows.
To assess whether findings in cryopreserved UCBL generalize to fresh adult
lymphocytes and to reduce potential freeze-thaw confounding, peripheral blood
lymphocytes (PBL) were analyzed in Laboratory 2 across doses and time points
(Table 4; Fig. 5). Factorial ANOVA demonstrated highly significant dose effects
for all foci endpoints at 2 h (FDR-adjusted p
| Variables | Factorial ANOVA | Categorical Factors | p-values adjusted by FDR | |||||||
| 53BP1 * | Multivariate | Univariate | Dose | Treatment | Post-irradiation Time | Dose | Treatment | Post-irradiation Time | ||
| + | + | + | + | – | + | + | – | 0.00000 | 0.12145 | – |
| + | – | – | – | + | + | + | – | 0.00000 | 0.64753 | – |
| – | + | – | – | + | + | + | – | 0.00000 | 0.26433 | – |
| – | – | + | – | + | + | + | – | 0.00000 | 0.08321 | – |
| + | + | + | + | – | + | + | + | 0.00040 | 0.40890 | 0.86156 |
| + | – | – | – | + | + | + | + | 0.00018 | 0.37226 | 0.87154 |
| – | + | – | – | + | + | + | + | 0.00461 | 0.97609 | 0.77227 |
| – | – | + | – | + | + | + | + | 0.00006 | 0.38582 | 0.47825 |
Factors included in the first 4 row-based cases - 0, 10, and 1000 mGy radiation dose (Dose) and cal A-, DMSO-, and non-treatments (Treatment) measured at 2 h post-irradiation/treatment; factors included in the last 4 row-based cases - 0 and 100 mGy radiation dose, cal A-, DMSO-, and non-treatments, and Post-irradiation Time (2, 4, and 6 h). Where applicable, multivariate (across foci endpoints) and univariate tests are shown. Reported p-values are FDR-adjusted. * mean, foci per cell; + the data were included in analysis; – data were not included in analysis. Statistically significant values are marked in red. PBL, peripheral blood lymphocytes; FDR, false discovery rate.
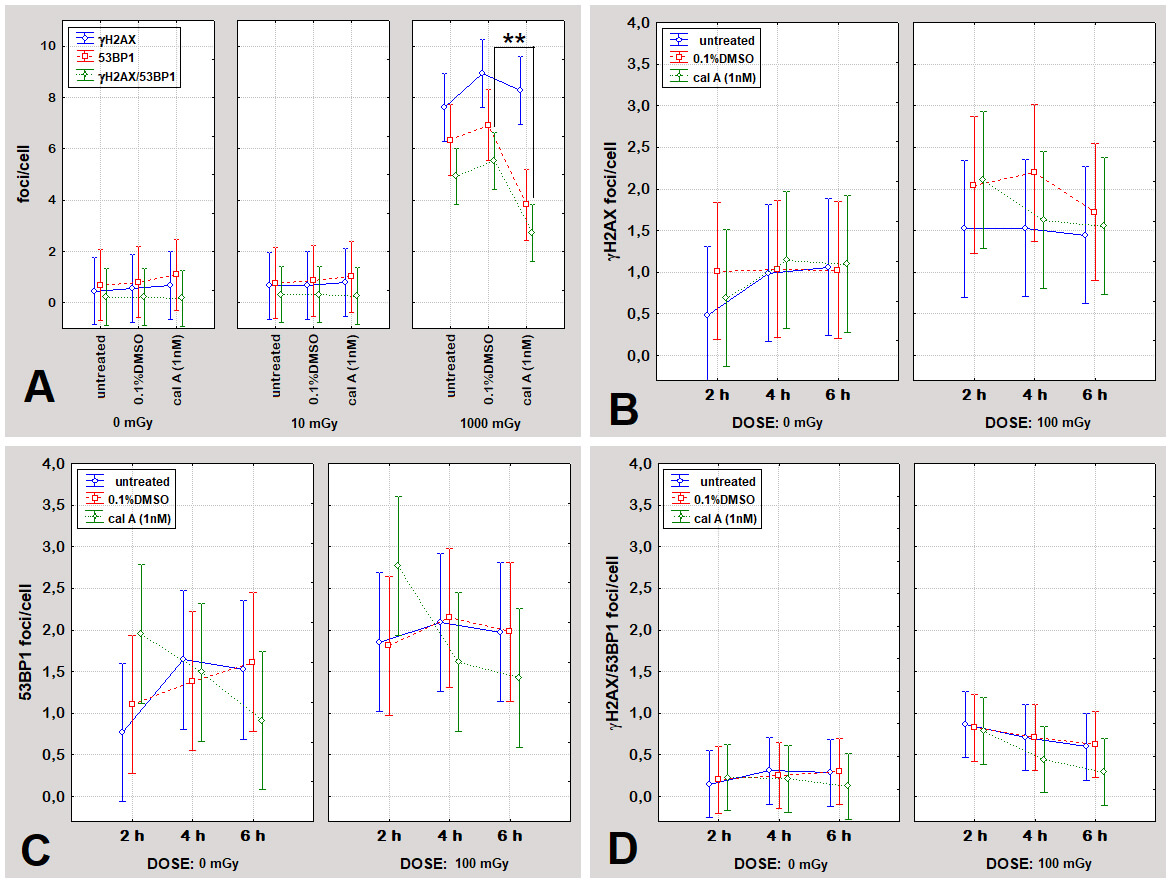 Fig. 5.
Fig. 5.
Induction of DNA repair foci in PBL across treatments and
irradiation conditions - Laboratory 2. (A) Mean numbers of
Notably,
Inclusion of untreated irradiated PBL alongside DMSO- and cal A-treated samples confirmed that DMSO did not alter foci induction compared with untreated controls, supporting its use as vehicle control (Fig. 5). At low doses (0, 10, 100 mGy) and across time points (2, 4, 6 h), cal A did not significantly modify foci induction relative to DMSO or untreated controls.
A significant treatment-dependent effect emerged only at the highest dose
tested: at 1000 mGy, co-localized
Under comparable experimental conditions, both UCBL (in both laboratories) and
PBL exhibited dose-dependent induction of
Overall, these results support high robustness of IRIF dose-response relationships under harmonized workflows while indicating that the impact of cal A is strongly context-dependent and not reliably stabilizing under the tested conditions.
Across two independent laboratories,
Although absolute IRIF yields differed between laboratories, the form and
linearity of the dose-response relationships were preserved across endpoints.
Crucially, the qualitative conclusion regarding cal A was consistent across
laboratories. Although these findings confirm the expected low-dose sensitivity
of
Analysis of freshly isolated PBL served as a within-laboratory control to assess
whether conclusions derived from cryopreserved UCBL were workflow-dependent. PBL
showed consistent dose-dependent induction of all IRIF endpoints, with foci
yields at 100 mGy exceeding those observed in UCBL from Laboratory 1 and closely
matching UCBL data from Laboratory 2. Importantly,
Phosphatase inhibition by cal A has been reported to affect
The PBL dataset was included to test the internal consistency of conclusions
under conditions free of cryopreservation-related stress. While UCBL showed no
cal A effect across the examined dose range, PBL displayed a reduction in
The present findings directly extend and reinforce our earlier observations in
UCBL [36]. In that study,
Inclusion of freshly isolated PBL adds an important dimension, indicating that donor variability, fixation timing, and cryopreservation stress contribute to absolute yield differences [43, 44, 45].
Across both studies, DMSO was confirmed as a neutral vehicle control, and cal A at 1 nM consistently failed to stabilize IRIF. Higher concentrations previously shown to induce cytotoxicity and apoptosis [36] were deliberately excluded from the present comparison. Together, these datasets converge on a coherent interpretation in which low, non-toxic concentrations of cal A do not measurably enhance IRIF stability, whereas higher concentrations trigger apoptosis-related chromatin alterations that confound biodosimetric readouts.
Systematic offsets in absolute IRIF yields between laboratories are a well-recognized feature of multicenter biodosimetry studies and do not, by themselves, indicate methodological failure [15, 17, 46]. In the present study, such offsets did not alter dose-response linearity or qualitative treatment conclusions. These findings are directly relevant to ongoing biodosimetry standardization efforts within international networks such as RENEB and EURADOS, which increasingly prioritize workflow harmonization over absolute inter-laboratory yield matching. Accordingly, the objective of inter-laboratory comparison in this study was not to eliminate absolute yield differences, but to determine whether harmonized workflows preserve interpretable and reproducible dose-response behavior. Recent methodological analyses converge on the same conclusion: reproducibility in IRIF-based biodosimetry is achieved through disciplined workflow alignment rather than through incremental assay sensitivity gains [47, 48].
Consistent correlations among
Standardized IRIF assays without pharmacological modifiers provide reliable and interpretable dose-response information in the low-dose range relevant for biodosimetry [2, 6, 49, 50]. While cal A remains useful in specific radiobiological applications, such as premature chromosome condensation assays [51, 52, 53, 54], its routine use as an enhancer of IRIF-based biodosimetry is not supported under harmonized low-dose workflows.
Future work should focus on validating IRIF-based biodosimetry under realistic operational constraints, including delayed processing and inter-operator variability, rather than on pharmacological signal amplification [55]. From a translational perspective, these findings argue for abandoning cal A as a default ‘fix’ for inter-laboratory variability and instead prioritizing disciplined workflow harmonization.
This study provides an inter-laboratory evaluation of
Our results demonstrate that
Collectively, these findings indicate that cal A should not be regarded as a
default enhancer of
By demonstrating reproducibility across laboratories and biological systems, this work strengthens confidence in IRIF-based biodosimetry as a standardized and translatable tool. These results further support integration of IRIF-based biodosimetry into automated and predictive scoring platforms for radiation preparedness and clinical triage. Recent integrative analyses further underscore that such progress toward automation and predictive biodosimetry critically depends on rigorously validated, reproducible experimental foundations rather than on incremental assay sensitivity gains alone. Instead, reproducibility of IRIF-based biodosimetry is best achieved through disciplined workflow harmonization rather than through pharmacological intervention.
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Conceptualization, LZ, LJ; methodology, LZ, LJ; software, LZ; validation, JK, PP; formal analysis, LZ, JK, PP and LJ; investigation, LZ, LJ and JK; resources, LZ; data curation, LZ, JK and LJ; writing-original draft preparation, LZ and LJ; writing-review and editing, LZ; visualization, LZ and LJ; supervision, LZ; project administration, LZ; funding acquisition, LZ. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. UCB samples were obtained from Eurocord-Slovakia, a non-profit public umbilical cord blood bank. According to Slovak national regulations, the use of fully anonymized biological material provided by a public tissue bank for research purposes does not require additional ethical committee approval. Donor recruitment and primary UCB sample collection were conducted in accordance with national regulations and institutional ethical guidelines. PBL donor recruitment and participation were supervised by the local Ethics Committee of the Biomedical Research Center, Slovak Academy of Sciences, Bratislava, Slovakia (approval No. EK/BmV-01/2023). The local ethical protocols required that all PBL donors or their legal guardians provided informed consent prior to blood sample collection.
The authors would like to thank M. Kubes, Eurocord-Slovakia, Bratislava, Slovakia, for providing UCB MNC; V. Vinnikov, PhD. and I. Belyaev, Assoc. Prof., D.Sc., Department of Radiobiology, Cancer Research Institute, Biomedical Research Center, Slovak Academy of Sciences, Bratislava, Slovakia, for consulting on statistical issues of the study; E. Hajtmanova, M.D. and Z. Zaborska, Oncology Centre of Jessenius Faculty of Medicine in Martin, Comenius University in Bratislava and Martin University Hospital, Martin, Slovakia, and G. Kralik and K. Kontrisova, St. Elisabeth Cancer Institute, Bratislava, Slovakia for assistance and in vitro irradiations of the cells.
This research was funded by the Recovery and Resilience Plan of the Slovakia under the project [CU-ESG-2024-05-PD] - administered by the Research Agency under the Grant Agreement No. 09I03-03-V05-00019 and by the Grant Agency of the Slovakia (VEGA) under Grant [2/0140/23].
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL47070.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





