1 Second Clinical Medical College, Guizhou University of Traditional Chinese Medicine, 550025 Guiyang, Guizhou, China
2 Key Laboratory on the Property & Effect of Chinese Medicine (Ethnic Medicine), 550025 Guiyang, Guizhou, China
†These authors contributed equally.
Abstract
This review examines the applications of natural coumarins in the context of bone and joint disorders. It provides comprehensive coverage of their fundamental pharmacology, the epidemiology and pathological mechanisms of osteoarticular diseases, as well as the pharmacological actions, clinical applications, future prospects, and ongoing challenges associated with these compounds. Through systematic analysis of relevant studies, we summarize the basic characteristics of natural coumarins, including their chemical structures, metabolic pathways, and pharmacological activities. We also examine their mechanisms of action in conditions such as osteoarthritis, rheumatoid arthritis, and osteoporosis, with a particular emphasis on their anti-inflammatory and cartilage-repairing properties. Furthermore, we summarize current clinical trials, formulation development, delivery strategies, and safety evaluations. We also explore potential new indications, combination therapies, and personalized treatment approaches. Several controversies and ongoing challenges are discussed, including variability in dose-dependent efficacy, divergent therapeutic outcomes across diseases, and concerns regarding long-term safety. This review aims to provide a foundational reference for further research and clinical application of natural coumarins in the treatment of bone and joint disorders.
Graphical Abstract
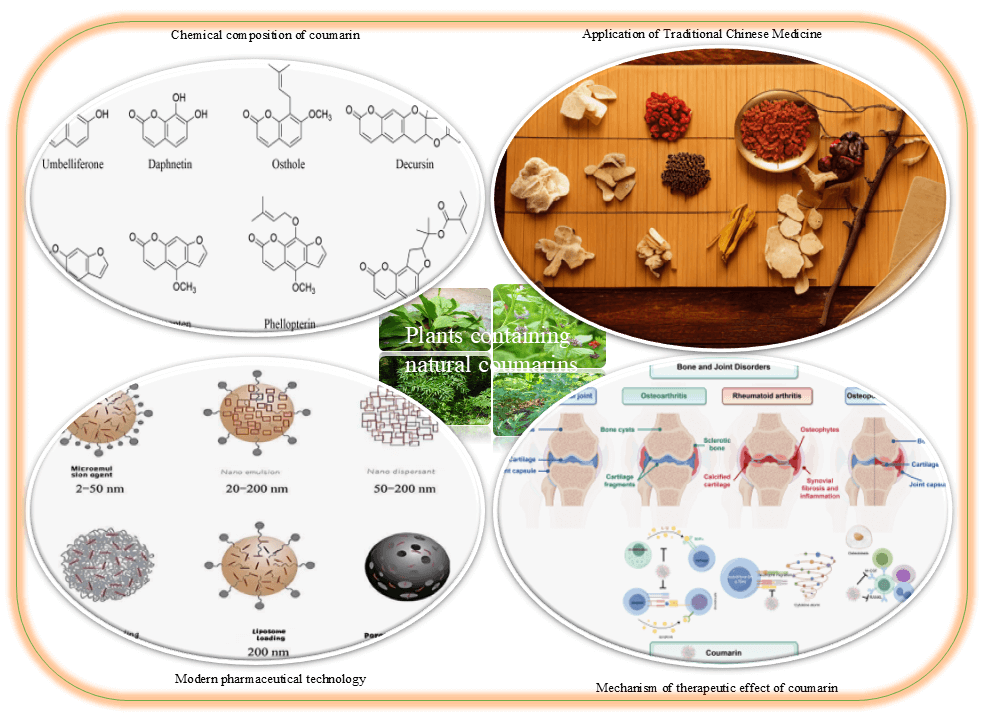
Keywords
- natural coumarins
- bone and joint disorders
- pharmacological mechanism
- clinical application
- potential for medicinal use
Coumarins are a class of lactones characterized by an
Osteoarticular disorders encompass a spectrum of pathological conditions of bone and joint arising from degeneration, inflammation, trauma, metabolic disturbances, infection or neoplasia. These conditions include rheumatoid arthritis (RA), osteoarthritis (OA), bursitis, synovitis, cervical and lumbar spondylopathies, adhesive capsulitis, osteophyte formation, avascular necrosis of the femoral head, and degenerative joint disease [10]. They are highly prevalent in middle-aged and older adults, and are characterized primarily by chronic pain and loss of function [11]. Progressive disease often leads to joint stiffness and deformity, resulting in substantial functional impairment and representing a major cause of disability in the elderly. The demographic shift towards an aging population has led to a rising incidence of osteoarticular disease, with associated increases in social and healthcare burdens [12].
Consequently, the identification of effective strategies for the prevention and treatment of osteoarticular disease remains a key priority in geriatric medicine. Traditional Chinese medicine (TCM) has a long history of treating joint disorders and has yielded numerous empirically effective formulas [13]. Contemporary pharmacological investigations indicate that natural coumarins in medicinal herbs contribute substantially to therapeutic effects by mediating anti-inflammatory actions, inhibition of ferroptosis, suppression of pyroptosis, as well as other protective mechanisms [14]. Coumarins therefore hold considerable promise for the management of osteoarticular disease. Our research group has also identified several coumarin derivatives from traditional medicines that have drug development potential [15, 16].
For this review, we performed a literature search using the keywords “coumarin”, “osteoarthritis”, “rheumatoid arthritis”, and “osteoporosis” across the Web of Science (https://webofscience.clarivate.cn) and China National Knowledge Infrastructure (https://www.cnki.net). Literature from the SCIE and Chinese Core Journal Directory within the past five years was included. Irrelevant topics (such as non-disease research), low-quality research, conference abstracts and preprints were excluded. Records retrieved from these databases were manually screened to remove duplicates and irrelevant works. Based on classical TCM texts and the primary literature, all studies meeting our inclusion criteria were analyzed and synthesized. This review summarizes current advances in the study of natural coumarins for the treatment of osteoarticular disease, with the aim of informing future efforts to exploit their therapeutic potential.
The basic structural framework of coumarin compounds consists of a benzopyrone nucleus [17]. These compounds occur predominantly in dicotyledonous plants either as aglycones or glycosides [18], and are found in traditional Chinese medicinal herbs such as Psoralea corylifolia, Peucedanum praeruptorum, Angelica pubescens, Angelica dahurica, Fraxinus rhynchophylla, Cnidium monnieri, and Artemisia capillaris [19]. Chemical modifications to the core structure can significantly influence their pharmacological properties [20]. Based on their chemical structures, coumarins derived from medicinal plants can be categorized into four classes: simple coumarins, furanocoumarins, pyranocoumarins, and other structural variants.
Simple coumarins are defined by the presence of substituents solely on the benzene ring, without the formation of additional furan or pyran rings at the 6- or 8-position relative to the 7-hydroxy group. Oxygen-containing functional groups—such as hydroxyl or methoxy groups—are frequently observed at the C-7 position, although substitutions may also occur at other sites. Representative examples include umbelliferone from Peucedanum praeruptorum, and daphnetin from Daphne odora.
Complex coumarins arise through the annulation of heterocyclic rings onto the benzene moiety of simple coumarins, leading to the formation of furanocoumarins and pyranocoumarins. Furanocoumarins typically result from cyclization of an isopentenyl group with an adjacent phenolic hydroxyl on the coumarin backbone. These are further subdivided into 6,7-furanocoumarins (linear type, e.g., psoralen from Psoralea corylifolia), and 7,8-furanocoumarins (angular type, e.g., angelicin).
Pyranocoumarins are characterized by a 2,2-dimethylpyran ring formed via cyclization between an isopentenyl group at C-6 or C-8 and a neighboring phenolic hydroxyl. These are also classified into linear (6,7-) and angular (7,8-) types, exemplified by xanthyletin from Zanthoxylum simulans and seselin from Seseli indicum, as well as decursin and decursinol from Peucedanum praeruptorum.
Beyond these three primary categories, natural coumarins also exhibit additional
structural diversity. For example, dicoumarols represent dimers of coumarin
units, which may involve linkages between simple coumarins or between
pyranocoumarins in linear–linear or linear–angular configurations. Isocoumarins
are isomers of coumarins and occur predominantly in plants as dihydroisocoumarin
derivatives. Furthermore, certain coumarins carry substituents at the C-3 or C-4
position of the
| No. | Type | English name | Molecular formula | Source (Botanical origin) | Ref. |
| 1 | Simple Coumarin | Umbelliferone | C9H9O3 | Peucedanum praeruptorum | [21] |
| 2 | Daphnetin | C9H6O4 | Daphne genkwa | [22, 23] | |
| 3 | Scoparone | C11H10O4 | Artemisia scoparia | [24] | |
| 4 | Osthole | C15H16O3 | Cnidium monnieri | [25, 26, 27] | |
| 5 | Fraxetin | C10H8O5 | Fraxinus rhynchophylla | [28] | |
| 6 | Esculetin | C9H6O4 | Fraxinus rhynchophylla | [29] | |
| 7 | Scopoletin | C10H8O4 | Angelica dahurica | [30] | |
| 8 | Pyranocoumarin | Xanthoxyletin | C15H14O4 | Zanthoxylum americanum | [31] |
| 9 | Xanthyletin | C14H12O3 | Zanthoxylum bungeanum | [32] | |
| 10 | Luvangetin | C15H14O4 | Toddalia asiatica | [33] | |
| 11 | Decursin | C19H20O5 | Angelica decursiva | [34] | |
| 12 | Decursinol | C14H14O4 | Angelica decursiva | [35] | |
| 13 | Seselin | C14H12O3 | Seseli indicum | [36] | |
| 14 | 5-Hydroxyseselin | C14H12O4 | Seseli indicum | [37] | |
| 15 | 5-Methoxyseselin | C15H14O4 | Seseli indicum | [38] | |
| 16 | Praeruptorin A | C21H22O7 | Peucedanum praeruptorum | [39] | |
| 17 | Praeruptorin B | C24H26O7 | Peucedanum praeruptorum | [40] | |
| 18 | Praeruptorin C | C24H28O7 | Peucedanum praeruptorum | [41] | |
| 19 | Furanocoumarin | Psoralen | C11H6O3 | Psoralea corylifolia | [42, 43, 44] |
| 20 | Imperatorin | C16H14O4 | Angelica dahurica | [45] | |
| 21 | Isoimperatorin | C16H14O4 | Angelica dahurica | [46] | |
| 22 | Xanthotoxin | C12H8O4 | Angelica pubescens | [47] | |
| 23 | Bergapten | C12H8O4 | Citrus medica | [48, 49] | |
| 24 | Phellopterin | C17H16O5 | Angelica dahurica | [50] | |
| 25 | Byakangelicol | C17H16O6 | Angelica dahurica | [51] | |
| 26 | Cnidicin | C21H22O5 | Angelica sinensis | [52] | |
| 27 | Oxypeucedanin | C16H14O5 | Peucedanum praeruptorum | [53] | |
| 28 | Isopsoralen | C11H6O3 | Psoralea corylifolia | [54] | |
| 29 | Isobergapten | C12H8O4 | Notopterygium incisum | [55] | |
| 30 | Pimpinellin | C13H10O5 | Heracleum hemsleyanum | [56] | |
| 31 | Angenomalin | C14H12O3 | Angelica dahurica cv. | [57] | |
| 32 | Columbianadin | C19H20O5 | Angelica sinensis | [58] | |
| 33 | Other Coumarins | Euphorbetin | C18H10O8 | Euphorbia lathyris | [59] |
| 34 | Daphnoretin | C19H12O7 | Stellera chamaejasme | [60] | |
| 35 | Dicoumarol | C19H12O6 | Medicago sativa | [61] | |
| 36 | Capillarin | C13H10O2 | Artemisia capillaris | [62] | |
| 37 | Agrimonolide | C18H18O5 | Agrimonia pilosa | [63] | |
| 38 | Plumbapin | C11H8O3 | Plumbago zeylanica | [64] | |
| 39 | Pachyrrhizin | C19H12O6 | Pachyrhizus erosus | [65] |
Natural coumarins exhibit a broad spectrum of pharmacological activities, including anti-inflammatory, antioxidant, antitumor, antibacterial, and antiviral effects [66]. In terms of anti-inflammatory activity, numerous coumarins act by suppressing key inflammatory signaling pathways. For instance, umbelliferone and methyl gallate have been shown to significantly inhibit the secretion of nitric oxide (NO) and prostaglandin E2 (PGE2) in lipopolysaccharide (LPS)-stimulated macrophages, concurrently downregulating the expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), thereby exerting anti-inflammatory effects [67]. Common mechanisms underlying these anti-inflammatory actions are summarized in Table 2 (Ref. [21, 27, 44, 49, 50, 66, 68, 69]) and Fig. 1.
| Chemical compound | Test subject | Mechanism of action | Reference |
| Decursin | Mouse chondrocytes | Inhibits IL-1 |
[66] |
| Phellopterin | Human immortalized keratinocytes (HaCaT) | Upregulates SIRT1 and downregulates ICAM-1 | [50] |
| Osthole | Human keratinocyte cell line (HaCaT) | Modulates secretion of IL-1, TNF- |
[27] |
| Umbelliferone | Arthritic rat model | Mediates MAPK signaling via the NF-κB pathway | [21] |
| Daphnetin | Inflammatory pain mouse model | Negatively regulates the spinal NF-κB pathway and preferentially activates the Nrf2/HO-1 signaling pathway | [68] |
| Bergapten | Mouse monocyte-macrophage leukemia cells (RAW264.7) | Inhibits JAK/STAT activation and ROS production | [49] |
| Columbianadin | Human peripheral blood mononuclear cells (hPBMCs) | Suppresses LPS-induced production of inflammatory cytokines and downregulates the NOD1/NF-κB pathway | [69] |
| Psoralen | Mouse monocyte-macrophage leukemia cells (RAW264.7) | Inhibits NF-κB activation and MAPKs phosphorylation | [44] |
ROS, reactive oxygen species.
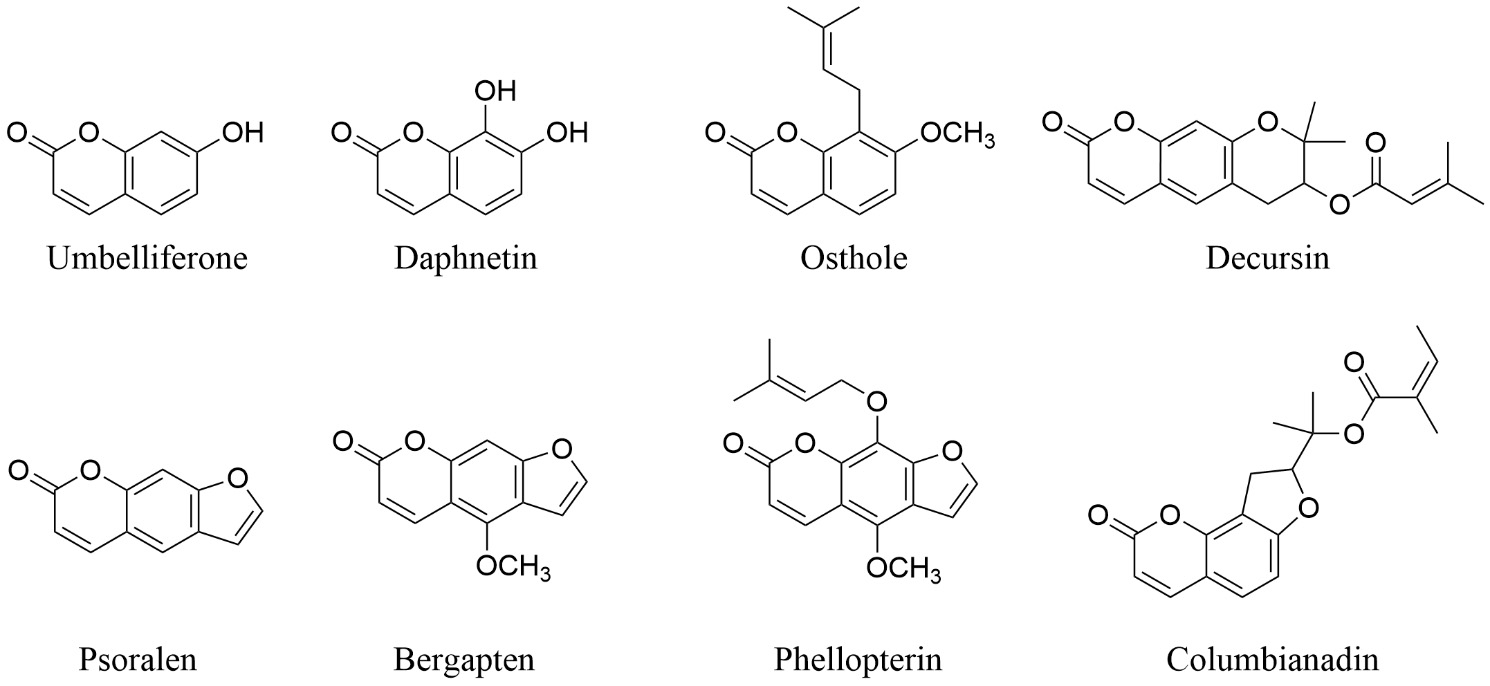 Fig. 1.
Fig. 1.
Representative coumarins with anti-inflammatory effects.
The antioxidant properties of coumarins are primarily mediated through free radical scavenging and modulation of the antioxidant enzyme system. Notably, 7-hydroxycoumarin demonstrates considerable antioxidant capacity by effectively neutralizing DPPH (1,1-diphenyl2-picrylhydrazyl) radicals. This activity is closely associated with the presence of hydroxyl groups in its structure [70]. Furthermore, coumarins have diverse biological activities, such as antioxidant and anticoagulant properties. For example, coumarin and several of its derivatives (e.g., esculetin, scoparone, and 4-methylumbelliferone) were evaluated for their impact on carbon tetrachloride-induced hyperlipidemia in rats. Structural variations among these compounds were found to confer distinct modulatory effects on the serum lipid profile, underscoring the importance of chemical structure in determining biological activity [71].
With regard to antitumor applications, certain coumarin derivatives can induce apoptosis, suppress proliferation, and inhibit the metastasis of tumor cells. Specifically, some natural coumarins isolated from plants have been reported to modulate the expression of cyclins and apoptosis-related proteins, thereby promoting cancer cell apoptosis and impairing migration and invasive capacities [72]. A series of furo [3,2-c] coumarin derivatives were synthesized and assessed for their anti-proliferative activity against MCF-7 breast cancer and HCT-15 colon cancer cell lines. Among these, compounds 1b and 1d exhibited potent activity, and UV-Vis spectroscopic studies demonstrated binding affinity toward DNA and bovine serum albumin (BSA) [73].
Natural coumarins also demonstrate notable antibacterial and antiviral
activities through mechanisms such as the disruption of bacterial cell membranes
and inhibition of viral replication, highlighting potential applications in
anti-infective therapeutics [74]. Several coumarin derivatives that inhibit a
range of microorganisms have been identified. For example, novel Cu(II) complexes
synthesized with 6-acetyl-7-hydroxy-4-methylcoumarin (HL1) and
8-acetyl-7-hydroxy-4-methylcoumarin (HL2) show enhanced antibacterial and
cytotoxic activities compared to their parent ligands. Notably, the complex
Cu(HL2)2
OA is a prevalent chronic and degenerative joint disorder [10]. Medical expenses
related to OA account for 1–2.5% of gross domestic product (GDP) in high-income
countries of Europe and North America [76, 77], with joint replacement procedures
constituting a major proportion of these costs. As global demographic aging
continues to increase, OA is projected to become one of the most common diseases
worldwide [78]. The pathophysiological process of OA involves alterations in
multiple joint tissues, including articular cartilage, synovium, and subchondral
bone. Inflammation plays a central role in the initiation and progression of OA.
The release of various cytokines such as interleukin-1
Isofraxidin has been found to inhibit IL-1
RA is a chronic and progressive systemic autoimmune disorder characterized
primarily by inflammation of the synovial membrane within joints. The global
prevalence of RA ranges from 0.18% to 1.07%, while the reported prevalence in
mainland China is 0.42%, corresponding to an affected total population of
approximately 5 million individuals [86]. The pathogenesis of RA involves complex
interactions among genetic, environmental, and immunological factors [87]. Genetic
susceptibility plays a critical role, particularly alleles of the human leukocyte
antigen (HLA) genes, such as specific variants of HLA-DRB1
(e.g., shared epitope). These alleles influence antigen presentation and T-cell
activation, thereby increasing disease risk. RA shows familial aggregation, with
first-degree relatives of RA patients having a three-fold higher risk of
developing the disease. Moreover, the concordance rate among monozygotic twins
ranges from 12–15%, underscoring the contribution of genetic factors.
Immunological dysregulation is central to RA pathology. Abnormal activation of
CD4+ T helper cells leads to the secretion of proinflammatory cytokines,
including TNF-
Coumarin derivatives exert multifaceted effects in the pathological process of
RA. For instance, 8-methoxychromen-2-one (MCO), isolated from Ruta graveolens,
significantly reduces the arthritis index and clinical scores in a rat model of
collagen-induced arthritis (CIA), while also mitigating joint damage and reducing
plasma levels of TNF-
With regard to apoptosis, the combination of daphnetin with Bcl2-targeted small
interfering RNA (si-Bcl2) was shown to modulate the expression of anti-apoptotic
genes in fibroblast-like synoviocytes (FLS) derived from CIA rats. Both si-Bcl2
and daphnetin alone downregulated the mRNA and protein expression of Bcl2, and
reduced the mRNA level of signal transducer and activator of transcription 3
(STAT3). Their combined application resulted in a stronger pro-apoptotic effect
on RA FLS, providing a theoretical and experimental foundation for novel RA
treatments based on daphnetin-based siRNA combination therapies [95].
Furthermore, 7-hydroxycoumarin (7-HC) was shown to inhibit proliferation and
induce apoptosis of FLS in the CIA rat model by suppressing the
Wnt/
Coumarins have also been shown to modulate osteoclast differentiation and function. For instance, cinnamoyloxy-mammeisin (CNM) inhibited macrophage colony-stimulating factor (M-CSF)- and receptor activator of nuclear factor kappa-B ligand (RANKL)-induced osteoclast differentiation, as well as the expression of osteoclast-specific markers, thereby reducing bone resorption and alleviating bone destruction in RA [97].
Osteoporosis (OP) is a common bone disorder, particularly prevalent among
postmenopausal women and the elderly [98]. The maintenance of bone homeostasis
relies on a delicate balance between bone formation mediated by osteoblasts, and
bone resorption facilitated by osteoclasts. The pathophysiology of OP is
characterized by an imbalance between these processes, wherein bone resorption
exceeds bone formation, leading to reduced bone mass and the deterioration of
bone microarchitecture [99]. Osteoblasts promote the differentiation of
osteoclast precursors into mature osteoclasts by secreting M-CSF and RANKL,
thereby accelerating bone resorption [100]. Concurrently, osteoblasts also produce
osteoprotegerin (OPG), which competitively binds to RANKL and inhibits osteoclast
differentiation and maturation [101]. Consequently, the RANKL/OPG ratio serves as
a critical indicator of bone resorption activity [102]. Aging as well as
declining estrogen levels also contribute to a state of chronic, low-grade
inflammation. Proinflammatory cytokines such as TNF-
Natural coumarin compounds demonstrate considerable potential for the prevention
and treatment of OP. For example, the XLGB-B fraction, isolated from the TCM
formulation Xianlinggubao (XLGB) and known to contain multiple compounds
including coumarins, effectively prevents bone loss in ovariectomized mice.
XLGB-B improves bone quality, microstructure, and strength, while also modulating
bone turnover markers [106]. Furthermore, in vitro studies have shown
that osthole and imperatorin stimulate the proliferation of MCF-7 cells and
upregulate the mRNA levels of estrogen receptor-regulated genes, including
estrogen receptor-
Bone and joint diseases including OA, RA and OP represent major global public health challenges [110]. There is an urgent need to develop safe and effective therapeutics for these conditions. Herbal formulas containing multiple ingredients are commonly prescribed to patients in TCM practice. These formulas often include herbs rich in coumarin.
Coumarin-containing TCM formulas have long been employed in clinical practice,
and contemporary investigations have provided experimental and clinical support
for the therapeutic contributions of coumarins. For example, Duhuo Jisheng Tang
(DJT), with Duhuo as the monarch herb, contains multiple coumarin constituents
including dihydrooxypeucedanin, angeloylcolumbianetin, and osthol derivatives.
DJT is used clinically to dispel wind-damp, relieve arthralgia, and tonify the
liver and kidneys—actions that correspond conceptually to the TCM pattern of
“deficiency underlying excess” in osteoarticular disorders. A randomized
controlled trial allocated patients with knee OA to hyaluronate, DJT, or
combination therapy [111]. After 8 weeks, the combination group had significantly
lower scores on the Western Ontario and McMaster Universities Osteoarthritis
Index (WOMAC), lower TCM syndrome scores, and lower post-treatment serum
TNF-
Jitongning Tablet (JTNT), a TCM formula for ankylosing spondylitis (AS), has progressed to a phase II clinical trial (NCT03932019). AS is an inflammatory spondyloarthropathy related to RA. UPLC-Q-TOF-MS profiling of JTNT metabolites in rat biofluids (plasma, urine and brain tissue), together with network pharmacology and molecular docking analyses, have implicated the regulation of inflammatory and immunerelated genes as key mechanisms of JTNT action. Moreover, coumarins were predicted to be among the principal active constituents. Such “constituent–metabolite–target” multidimensional investigations provide a mechanistic foundation for elucidating the role of coumarins within complex formulas. They also guide subsequent targeted isolation and development of active compounds [113].
Several proprietary Chinese medicines derived from traditional formulas owe part of their efficacy to coumarin-bearing herbs. Xianling Gubao capsule, developed by Professor Shi Guangda from an ethnomedicinal Miao prescription and produced using modern pharmaceutical techniques [114], contains Epimedium, Psoralea and Dipsacus among its principal ingredients. Psoralea is rich in psoralen and isopsoralen, while Dipsacus also contains coumarin derivatives. The Clinical Practice Guideline for Chinese Patent Medicines in the Treatment of Osteoporosis [115] indicates that Xianling Gubao combined with conventional anti-OP therapy (bisphosphonates) significantly improves lumbar and femoral neck bone mineral density. Additionally, it has clinical relevance for preventing and treating osteoporotic vertebral compression fractures and femoral neck fractures. In a comparative clinical study, Fu and Shi [116] evaluated oral calcium carbonate with vitamin D3 plus intravenous zoledronic acid versus the same regimen combined with Xianling Gubao. The combined treatment group demonstrated a superior overall response rate of 92.68% vs. 75.61%.
Collectively, these findings indicate that coumarins are not only active ingredients of traditional TCM, but also bioactive substances that can undergo modern scientific evaluation and inform precision clinical use and drug development. This body of evidence forms an integrated “clinic–mechanism–practice” loop and exemplifies a pragmatic pathway for the modernization of TCM.
Combination therapy is an important strategy to increase efficacy while reducing singleagent doses and adverse effects. Coumarins demonstrate substantial potential for synergistic interactions with conventional therapeutics.
Synergy with immunosuppressive approaches: studies have shown that daphnetin combined with Bcl-2-targeting small interfering RNA (siRNA) markedly enhanced the apoptosis of pathological synovial cells in CIA rats. This result suggests a novel “drug–gene” combinatorial paradigm for RA therapy, whereby coumarins paired with nucleicacidbased agents can eliminate pathogenic cell populations more effectively [117].
Synergy with natural products: a synergistic effect of imperatorin and
The therapeutic efficacy of natural coumarins varies across different bone and joint disorders. As previously discussed, multiple natural coumarins demonstrate beneficial effects in OA by inhibiting inflammatory responses and modulating chondrocyte metabolism. In contrast, although certain coumarin compounds can promote bone formation and suppress bone resorption in OP, their mechanisms and effectiveness may differ significantly depending on structural variations. Furthermore, research on natural coumarins remains relatively limited for other bone and joint diseases, such as RA and AS, and their efficacy in these conditions is not yet well-established. These variations in treatment outcomes may be attributed to the complex and diverse pathophysiological mechanisms underlying different disorders. Each disease involves distinct cell types, signaling pathways, and tissue injury mechanisms, leading to differences in the targets and effects of natural coumarins. Therefore, a deeper understanding of the mechanisms of action of natural coumarins in various bone and joint diseases, along with the optimization of treatment strategies for specific conditions, is essential for maximizing their therapeutic potential.
Personalized medicine is the future goal of clinical practice, with the application of natural coumarins in the management of bone and joint disorders holding considerable promise. Given the variations in genetic background, disease status, and lifestyle among individuals, patient responses to natural coumarin-based therapies may differ significantly. For example, genetic polymorphisms influence the drug metabolism and efficacy of coumarin-derived oral anticoagulants. Polymorphisms in Cytochrome P450 proteins 2C9 (CYP2C9) and vitamin K epoxide reductase complex subunit 1 (VKORC1) are strongly associated with warfarin dosing requirements, such that patients carrying certain mutant alleles often require lower doses to avoid adverse effects such as bleeding [119]. Therefore, genetic testing and biomarker assessments can help to characterize a patient’s genetic profile in clinical practice, enabling the design of individualized treatment regimens involving natural coumarins. This includes the selection of appropriate drug formulations, determination of optimal dosages, and adjustment of dosing frequency. Additionally, comorbidities and concomitant medications must also be considered in order to comprehensively evaluate the risk-benefit profile of treatment, thereby achieving precision therapy and improving both therapeutic outcomes and quality of life. Personalized treatment is increasingly recognized as essential in the context of RA, and natural coumarins may play a valuable role. Due to patient heterogeneity in RA pathogenesis, disease progression, and treatment response, personalized strategies aim to tailor therapies based on individual characteristics.
At the genetic level, polymorphisms in genes encoding drug-metabolizing enzymes, such as cytochrome P450 isoforms, may influence the efficacy and safety of coumarins by altering their pharmacokinetics. Genetic profiling can provide guidance for dose individualization and rational combination therapies. From a clinical perspective, markers of disease activity such as CRP and erythrocyte sedimentation rate (ESR), together with joint damage assessments, can inform coumarin dosing and formulation selection. Patients with mild disease may benefit from low-dose coumarins combined with gentle therapeutic regimens, whereas those with severe inflammation may require higher doses, or combination with more potent agents. Furthermore, the presence of comorbidities such as cardiovascular disease or diabetes should guide the choice of coumarin compounds or delivery systems to minimize adverse impacts on concurrent conditions. Through this integrated approach, personalized treatment with natural coumarins can be optimized to enhance therapeutic efficacy and improve patient quality of life. The dose-response relationship of natural coumarins remains a subject of ongoing debate. Various studies have demonstrated that different doses of coumarins can lead to distinct pharmacological outcomes. For instance, coumarin and its derivatives exerted differential effects on the serum lipid profile in experimental rat models. At a specific dosage, 4-methylumbelliferone significantly reduced triglyceride and very-low-density lipoprotein cholesterol levels in rats with carbon tetrachloride-induced hyperlipidemia, but failed to restore total cholesterol levels. In contrast, umbelliferone and scoparone were effective in preventing the reduction of high-density lipoprotein cholesterol in the same model of lipid dysregulation [71].
In the context of bone and joint disorders, the optimal therapeutic dosage for many coumarins has yet to be conclusively established. Some studies suggest that increasing the dose may enhance efficacy, but this could also increase the risk of adverse reactions. For example, excessively high doses of coumarin-based anticoagulants increase the likelihood of serious adverse events such as bleeding, while insufficient dosing may fail to achieve the desired anticoagulant effect. Therefore, defining the optimal dosage range for natural coumarins in various bone and joint diseases, where efficacy is balanced with safety, represents a significant challenge in current research. Further clinical studies and robust data are essential to resolve these controversies and establish evidence-based dosing guidelines.
The application of natural coumarins in RA faces several controversies and challenges. From a clinical perspective, only a limited number of high-quality trials have investigated coumarins in RA patients. Large-scale, multi-center validations of their efficacy and safety are still lacking. Although coumarin derivatives have shown promising therapeutic effects in animal models, interspecies differences complicate the extrapolation of these results to humans. Additionally, potential drug–drug interactions between coumarins and conventional RA treatments—such as disease-modifying antirheumatic drugs (DMARDs) and NSAIDs—are not yet fully understood, which may impact the safety and efficacy of combination therapies. Moreover, while some mechanistic studies have identified molecular targets and signaling pathways through which coumarins may act, the complex pathophysiology of RA raises questions about their precise mechanisms of action in vivo and the possibility of undiscovered side effects. Challenges also remain in the extraction, purification, and formulation of natural coumarins, including how to ensure batch-to-batch consistency, improve bioavailability, and achieve stable and reproducible product quality.
Future research on coumarins in RA should prioritize several key areas. First, well-designed clinical studies are essential. Large-scale, randomized, multi-center controlled trials are needed to rigorously evaluate the efficacy and safety of coumarins in RA patients and to define optimal treatment protocols across different patient subpopulations. Second, further investigation into the mechanisms of action of coumarins is warranted. Despite current insights into the targets and pathways of coumarins, the complexity of RA pathogenesis necessitates deeper exploration of how they interact within biological networks and with endogenous molecules. This should provide a more robust theoretical foundation for drug development. In terms of pharmaceutical innovation, continued efforts should be made to optimize coumarin structures through computer-aided drug design and synthetic modification, leading to the development of derivatives with enhanced potency and reduced toxicity. The development of novel delivery systems, such as smart nanocarriers, should also be pursued to improve targeting and bioavailability. Finally, the integration of multi-omics technologies (e.g., genomics, proteomics, metabolomics) will help to elucidate the systemic effects and influencing factors of coumarin therapy in vivo, supporting the advancement of personalized treatment strategies.
The application of coumarins in RA research has a long history, and several coumarin-containing plants have been used in the treatment of RA and related conditions for many years. For instance, Saussurea involucrata has been used in traditional Uyghur, Mongolian, Kazakh, and Chinese medicine for its purported effects in promoting blood circulation, dispelling cold-dampness, and reducing inflammation. With advances in modern science and technology, research on coumarins has progressively expanded. Initial studies were primarily focused on the phytochemical analysis of coumarin-rich plants, leading to the identification of coumarin structures and their natural occurrence. Subsequent investigations delved into their biological activities, revealing that coumarins possess anti-inflammatory, antioxidant, and immunomodulatory properties, all of which are highly relevant to the pathological mechanisms of RA. This prompted further exploration of their therapeutic potential in RA.
The use of coumarins for the treatment of osteoarticular diseases is similar to
that of other compounds. Although a variety of components play a role in the
treatment of these diseases, their mechanisms and focus differ. For example, the
flavonoids Epimedium and daidzein exert estrogen-like effects, promote
osteogenesis, inhibit osteoclastogenesis, have antioxidant and anti-inflammatory
properties, promote chondrocyte proliferation, and inhibit matrix degradation
[120, 121]. Alkaloid Sinomenin and Matrine strongly inhibit the inflammatory
response and suppress chondrocyte apoptosis and osteoclast formation [122, 123].
Polyphenolics such as Resveratrol and Curcumin have strong antioxidant and
anti-inflammatory properties, regulate chondrocyte metabolism, and exert
multi-target actions and cardiovascular protection [124, 125]. Coumarin compounds
such as Daphnetin and Bergapten can promote osteoblast differentiation and
inhibit osteoclast activity, while also being anti-inflammatory via the
regulation of NF-
Beyond their applications in OA and OP, natural coumarins exhibit potential for a range of new therapeutic indications. Due to their broad pharmacological spectrum including anti-inflammatory and antioxidant properties, they may offer therapeutic benefits for other bone and joint disorders associated with inflammation and oxidative stress. For example, in RA, where inflammatory responses are central to disease progression, natural coumarins may alleviate symptoms by inhibiting key inflammatory signaling pathways, similar to their mechanism in OA [128].
Structural modification of natural coumarins is an important strategy for
enhancing their therapeutic efficacy in the treatment of bone and joint
disorders. Interactions with biological targets can be optimized by altering the
coumarin scaffold, thereby improving pharmacological activity. For example, a
series of 3-(4-aminophenyl)coumarin derivatives were synthesized and subjected to
activity screening. Among these, compound 5e demonstrated the strongest
inhibitory activity against the proliferation of fibroblast-like synoviocytes
(FLS), along with suppression of RA-related cytokines including IL-1, IL-6, and
TNF-
A coumarin compound isolated from Glycine tabacina, dolichosin A, was identified
as a potential therapeutic agent for RA by integrating network pharmacology and
experimental validation. Network pharmacology predictions suggest its involvement
in the PI3K/AKT and MAPK signaling pathways. Experimental results confirmed that
dolichosin A significantly inhibits IL-1
Another active research area is the development of novel coumarin-based therapeutics, including structural modification of natural coumarins to enhance efficacy and reduce toxicity and side effects. Additionally, combination therapies involving coumarins and other drugs are being explored to leverage synergistic effects and improve treatment outcomes. The design of advanced coumarin delivery systems is also a focus, with efforts directed toward enhancing target specificity and bioavailability while minimizing systemic adverse effects. Examples include smart drug delivery platforms that respond to pathological features of RA, such as the inflammatory microenvironment or metabolic abnormalities, enabling precise and controlled drug release.
The development of advanced formulations of natural coumarins is critical for enhancing their bioavailability and therapeutic efficacy. Several innovative delivery systems have been investigated, including nanoparticle-based approaches. A nanoparticle formulation effectively increased the Bax/Bcl-2 expression ratio and induced apoptosis, suggesting it can substantially improve the therapeutic potential of natural coumarins [131].
Similarly, a micellar system based on poly(ethylene
glycol)-block-poly(
A metabolically bioresponsive hydrogel was also designed for the controlled release of psoralen in the treatment of RA. This enables on-demand release of psoralen and oxygen in response to inflammatory stimuli, thereby modulating homeostasis and metabolic disorders in the hypoxic arthritic microenvironment. Such pathology-responsive delivery systems represent a novel strategy for coumarin administration, with the potential to enhance treatment efficacy while minimizing systemic side effects [133].
The administration route significantly influences the absorption and distribution of natural coumarins. For example, some orally administered coumarin compounds may be affected by gastric acid, digestive enzymes, and other gastrointestinal factors, resulting in reduced bioavailability. Further investigation of appropriate delivery routes and formulations—such as lipid-based carriers (e.g., liposomes), microspheres, or transdermal delivery systems—holds promise for improving their pharmacokinetic profile and therapeutic efficacy. However, factors such as drug-carrier interactions and formulation stability must be carefully evaluated to ensure safety and effectiveness.
After entering the systemic circulation, natural coumarins are primarily metabolized in the liver through the action of various enzymes, with the cytochrome P450 (CYP) superfamily playing a central role. For example, coumarin is metabolized in both rat and human liver microsomes, with CYP1A2 catalyzing its conversion to o-hydroxyphenylacetic acid, and CYP2A6 mediating 7-hydroxylation to form 7-hydroxycoumarin. Notably, human hepatocytes have an approximately 10-fold higher metabolic capacity for the 7-hydroxylation of coumarin compared to rat hepatocytes [134].
The bioavailability of coumarin compounds is influenced by multiple factors, with their chemical structure playing a critical role. Specific substituents can affect gastrointestinal absorption, plasma protein binding, and metabolic conversion. Physiological factors such as age, sex, genetic background and disease status also contribute to interindividual variability. For example, genetic polymorphisms in CYP enzymes are associated with differences in the rate of coumarin metabolism, thereby altering bioavailability. Drug–drug interactions must also be considered, as the co-administration of coumarins with agents that induce or inhibit CYP activity can modify metabolic pathways and rates, ultimately affecting the bioavailability of coumarin [135].
The safety profile of natural coumarins is a critical consideration for their clinical application. Several studies have conducted preliminary safety assessments. For example, toxicogenomic and toxicological analyses of Soulatrolide and Mammea (A/BA + A/BB) coumarins extracted from Haploclathra paniculata (Brazilian plant) found that short-term oral administration (100 mg/kg/day for one week) induced changes in hepatic gene expression, primarily involving drug-metabolizing enzymes. However, no histopathological damage was observed in the liver, kidneys, or spleen, suggesting a favorable short-term safety profile [136].
Saussurea involucrata, a traditional herbal medicine containing coumarins among other constituents, is commonly used in the treatment of RA and other conditions. Although it demonstrates various beneficial biological activities, precise quality control and comprehensive toxicological studies are essential to ensure consistency and safety in clinical applications [137].
Studies on Angelicae pubescentis radix (AP) have revealed that its coumarin components exhibit a wide range of pharmacological effects. However, due to the complexity and diversity of its chemical composition and mechanisms, standardized criteria for evaluating its quality and efficacy are still lacking. Further research is needed to elucidate the structure–activity relationships among its constituents, as well as potential synergistic or antagonistic effects. Pharmacokinetic and toxicity studies in humans are also needed to ensure safe application [138].
Ardisia gigantifolia Stapf is used in traditional medicine for the management of RA, with clinical observations showing no significant toxic side effects. This provides supportive evidence, albeit preliminary, for the safety of coumarin-containing preparations in RA treatment. Nonetheless, detailed mechanistic toxicological studies are required to confirm the safety of individual active compounds [139].
Several studies have reported potential adverse effects associated with coumarin derivatives. Long-term use of oral coumarin-based anticoagulants, such as warfarin, has been linked to an increased risk of OP and fractures. Although the evidence remains controversial, this potential risk may be clinically significant for chronic users [140].
Long-term follow-up of patients using coumarin drugs has revealed serious adverse events, including gastrointestinal bleeding and intracranial hemorrhage. Furthermore, the impact of prolonged natural coumarin consumption on hepatic and renal functions requires further evaluation. While some studies indicate a favorable safety profile for certain natural coumarins within specific dosage ranges and treatment durations, the potential risks associated with extended use necessitate additional clinical research and vigilant monitoring to ensure patient safety.
Additionally, some coumarin compounds may exert embryotoxic effects at high doses. For example, several 4-phenyldihydrocoumarin derivatives demonstrated higher embryotoxicity in a zebrafish model than coumarin itself [141]. Thorough safety assessment and close monitoring of potential adverse reactions are therefore imperative when using natural coumarins in therapeutic applications.
In summary, natural coumarins represent a promising class of bioactive compounds with multi-target therapeutic potential in bone and joint disorders. Their broad anti-inflammatory, chondroprotective, and bone-modulating properties are underpinned by diverse molecular mechanisms, supporting their use in conditions such as OA, RA, and OP (Fig. 2). However, challenges remain in optimizing bioavailability, ensuring long-term safety, and validating efficacy through rigorous clinical trials.
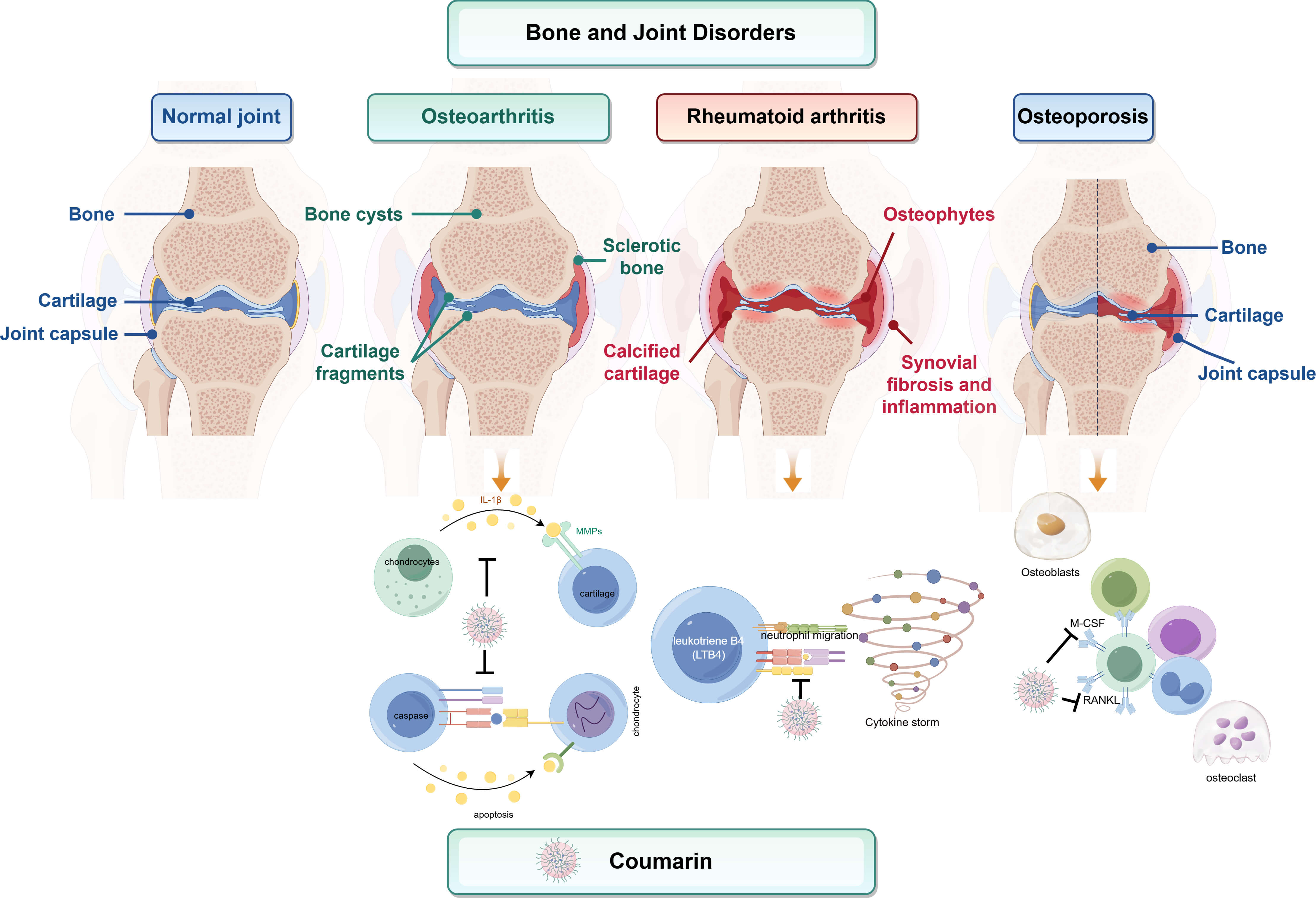 Fig. 2.
Fig. 2.
Mechanism diagram of bone and joint diseases. This schematic
compares normal joint architecture with pathological features of three major bone
and joint disorders: osteoarthritis (OA), rheumatoid arthritis (RA), and
osteoporosis (OP). It further outlines how natural coumarins intervene in
disease-specific mechanisms. In OA, coumarins inhibit IL-1
This review analyzed and summarized the literature and research data on the chemical composition and pharmacological effects of natural coumarins, their relevance to clinical practice, efficacy in traditional medicine, and metabolism of circulating components. Future research should focus on achieving structural optimization and developing smart delivery systems and personalized treatment strategies. This should enable the translation of these natural agents into clinically viable therapies, while providing guidance for further discoveries of the potential of natural coumarins as drugs and treatments.
AAA, abdominal aortic aneurysm; ALP, alkaline phosphatase; anti-CCP, anti-cyclic citrullinated peptide; ALT, alanine transaminase; AST, aspartate transaminase; AS, ankylosing spondylitis; AVF, arteriovenous fistula; CCL2, motif chemokine ligand 2; CCL5, motif chemokine ligand 5; CNM, cinnamoyloxy-mammeisin; COX-2, cyclooxygenase-2; CRP, C-reactive protein; CRRT, continuous renal replacement therapy; CTSK, Recombinant Human Cathepsin K; CT, computed tomography; CYP2C9, Cytochrome P450 proteins 2C9; CIA, collagen-induced arthritis; DBIL, direct bilirubin; 7-HC, 7-hydroxycoumarin; OA, osteoarthritis; DPPH, 1,1-diphenyl2-picrylhydrazyl; Dex, dexamethasone; DMARDs, disease-modifying antirheumatic drugs; ESR, erythrocyte sedimentation rate; FLS, fibroblast-like synoviocytes; GDP, gross domestic product; HLA, human leukocyte antigen; ICAM-1, intercellular cell adhesion molecule-1; ICU, intensive care unit; IL-1 interleukin-1; IL-1
Conceptualization, CH and ZQL; methodology, WJW and JHM; software, LW; data curation, LW and XDZ; writing—original draft preparation, JHM; writing—review and editing, JHM; visualization, WJW; supervision, CH; project administration, CH; funding acquisition, CH. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All data reported in this paper will also be shared by the lead contact upon request.All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research was funded by National Natural Science Foundation of China (No.82160805) and Guizhou University of Traditional Chinese Medicine (TD NO.[2022]004).
The authors declare no conflict of interest.
During the preparation of this work the authors used DeepSeek-V3 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


