1 Department of Medicine and Surgery, University of Enna “Kore”, 94100 Enna, Italy
2 Mediterranean Foundation “G.B. Morgagni”, 95125 Catania, Italy
3 Neurovisual Science Technology (NEST) srl, 95123 Catania, Italy
4 Fidia Ophthalmic Research, 95123 Catania, Italy
§Retired July 2022.
Abstract
Therapeutic strategies for ocular diseases are undergoing a transformative shift from symptom management to regenerative and disease-modifying approaches. This review highlights the development of neurotrophin receptor agonists—including recombinant nerve growth factor (NGF) (cenegermin), peptidomimetics (e.g., REC-0559, tavilermide), and synthetic microneurotrophins (BNN27, ENT-A010)—that target tropomyosin receptor kinases (TrkA/TrkB) and the p75 neurotrophin receptor (p75NTR) pathways to promote neuronal survival, synaptic plasticity, and tissue repair in neurotrophic keratitis, dry eye disease, and retinal degenerations. Parallel advances in peptide-based therapies address vascular and inflammatory pathologies: UPARANT and its derivatives modulate urokinase plasminogen activator receptor (uPAR)/formyl peptide receptor (FPR) signaling to inhibit angiogenesis and inflammation in diabetic retinopathy, whereas sphingosine 1 phosphate (S1P)-S1PR3 pepducins and integrin antagonists (risuteganib, THR-687, OTT166) offer multi-targeted strategies to stabilize the blood-retinal barrier and mitigate neovascularization. Innovations in drug delivery, such as dendrimer-peptide conjugates, enhance the stability and bioavailability of these agents. Further, senolytic therapies (e.g., UBX1325, procyanidin C1) are emerging as a promising approach for age-related and diabetic retinal diseases by clearing senescent cells and attenuating senescence-associated secretory phenotype (SASP)-driven inflammation. Together, these approaches exemplify a paradigm of “mimicking nature to modulate vision”, leveraging molecular insights to develop therapies that restore rather than merely preserve ocular function. While clinical validation is ongoing, the convergence of neurotrophic support, vascular modulation, and senescence targeting heralds a new era in precision ophthalmology.
Keywords
- peptidomimetic neurotrophin agonists
- neurotrophic therapy
- ocular regeneration
- neurotrophic keratitis
- retinal neurodegeneration
- senolytic strategies
Vision impairment due to chronic retinal and optic nerve disorders—including age-related macular degeneration (AMD), diabetic retinopathy (DR), and glaucoma—is a leading cause of disability worldwide, particularly affecting older adults and individuals with metabolic diseases such as diabetes and hypertension. The prevalence of these conditions continues to grow, fueled by global population aging, urbanization, and the increasing burden of systemic disorders. The number of individuals with vision-impairing late AMD is projected to rise from approximately 196 million in 2020 to nearly 288 million by 2040 [1], while nearly one in five individuals with diabetes are affected by DR [2]. For glaucoma, the most common cause of irreversible blindness, the estimated global prevalence of primary open-angle glaucoma (POAG) in adults is 2.4%—affecting roughly 68.6 million people in 2020, with projected increase up to 79.9 million by 2025 [3]. The aging of the global population and rising rates of chronic conditions underscore the urgent need for therapeutic advancements across ophthalmic diseases.
Although current treatment strategies have brought considerable benefits, especially in halting or slowing disease progression, they often fail to prevent disease onset or restore damaged retinal structure and function. The growing burden of retinal diseases, such as age-related macular degeneration and diabetic retinopathy, presents significant clinical and socioeconomic challenges, especially given the aging global population and the chronic nature of these conditions [4]. The increasing prevalence of vision-threatening eye diseases, especially in aging populations, highlights a pressing need to expand the therapeutic arsenal beyond currently available approaches.
There is now a broad consensus within the scientific and clinical communities that the future of vision restoration lies in developing interventions that act on the molecular and cellular mechanisms underlying retinal degeneration. Emerging technologies in molecular biology, regenerative medicine, and bioengineering offer promising avenues to intervene earlier in the disease process, protect retinal cells from degeneration, and potentially restore lost visual function [5, 6].
These forward-looking strategies are not limited to treating symptoms or managing end-stage damage. Rather, they aim to reprogram the disease trajectory itself—through approaches that may include modulating cellular stress responses, enhancing tissue regeneration, or replacing dysfunctional cells and tissues. Such innovations, while still in development, signal a paradigm shift from palliative to reparative and even preventive models of care.
While recent reviews have focused on individual therapeutic approaches (e.g., anti-vascular endothelial growth factor (VEGF) therapies or neuroprotective agents alone), this comprehensive analysis provides a unique integration of four emerging paradigms that collectively represent the next frontier in ocular therapeutics: (1) neurotrophin receptor agonism, (2) multitargeted peptide therapies, (3) integrin modulation, and (4) senolytic strategies. Our synthesis reveals how these approaches not only address distinct pathological mechanisms but may synergize to overcome limitations of current treatments. In fact, this review uniquely bridges basic science with translational applications, highlighting both federal drug administration (FDA)-approved breakthroughs (e.g., cenegermin) and investigational agents (e.g., UBX1325) while proposing concrete frameworks for combination therapies.
In this narrative literature review we performed an extensive literature search in multiple electronic databases, including PubMed, Web of Science, and Scopus, covering publications up to August 2025. Key search terms included words related to neurotrophic factors and regenerative therapies (e.g., “nerve growth factor”, “neurotrophin”, “brain-derived neurotrophic factor (BDNF)”, “TrkA agonist”, “peptidomimetic”), ocular diseases and conditions (e.g., “neurotrophic keratitis”, “dry eye”, “age-related macular degeneration”, “diabetic retinopathy”, “glaucoma”), multitargeted peptides and signaling pathways (e.g., “Urokinase Plasminogen Activator Receptor ANTagonist (UPARANT)”, “integrin inhibitor”, “sphingosine-1-phosphate (S1P) pepducin”), and cellular senescence therapies (e.g., “senolytic”, “UBX1325”, “procyanidin C1”). We also included specific drug and compound names (such as cenegermin, REC-0559, Tavilermide, BNN27, ENT-A010, Risuteganib, THR-687, OTT166) to ensure that all relevant studies on these emerging treatments were captured.
During the selection process, we screened titles and abstracts to identify literature that fit the scope of this review. Publications had to focus on therapeutic approaches for ocular diseases (corneal or retinal conditions) that aligned with the four emerging paradigms of interest–neurotrophin receptor agonists, multitargeted peptide therapies, integrin pathway modulators, or senolytic strategies. We included articles discussing the development, mechanism of action, preclinical evaluation, or clinical application of treatments in these categories. We included original research articles (laboratory studies, animal studies, and clinical trials), systematic reviews and meta-analyses, narrative reviews, and select conference proceedings or abstracts if they provided unique data on investigational therapies. The emphasis was on high-quality peer-reviewed journal publications. However, to ensure up-to-date coverage of recent breakthroughs, we also considered authoritative non-journal sources (for example, a regulatory agency press release or updated book chapter) that reported critical information such as drug approvals or clinical trial results relevant to the topic. Given the rapid evolution of this field, we prioritized literature from approximately the last two decades, but earlier pivotal studies were included if they introduced foundational concepts or agents. The final selection spanned publications from 2004 through 2025, reflecting both foundational research and the latest advances.
Applying these criteria yielded a total of 110 references that form the knowledge base of this review. These references encompass a broad range of study designs and publication types, ensuring a well-rounded perspective on the subject matter.
Retinal and optic nerve degeneration—manifesting in conditions such as glaucoma, diabetic retinopathy, and inherited retinal disorders—leads to irreversible vision loss through progressive neuronal death, axonal dysfunction, and synaptic disconnection. Most current treatments target secondary vascular or metabolic drivers, but few directly support neuronal survival or promote repair.
Neurotrophins—a family that includes nerve growth factor (NGF), BDNF, NT-3, and NT-4/5—play critical roles in neuronal development, synaptic plasticity, and survival by activating receptor tyrosine kinases (TrkA–C) and the low-affinity p75NTR receptor (Fig. 1). These signaling pathways are essential for maintaining the viability of retinal ganglion cells, photoreceptors, and interneurons under stress conditions such as elevated intraocular pressure and ischemia-induced injury [7]. This has led to the hypothesis that harnessing neurotrophic signaling may provide an effective means to preserve or restore neuronal integrity in retinal disease [8].
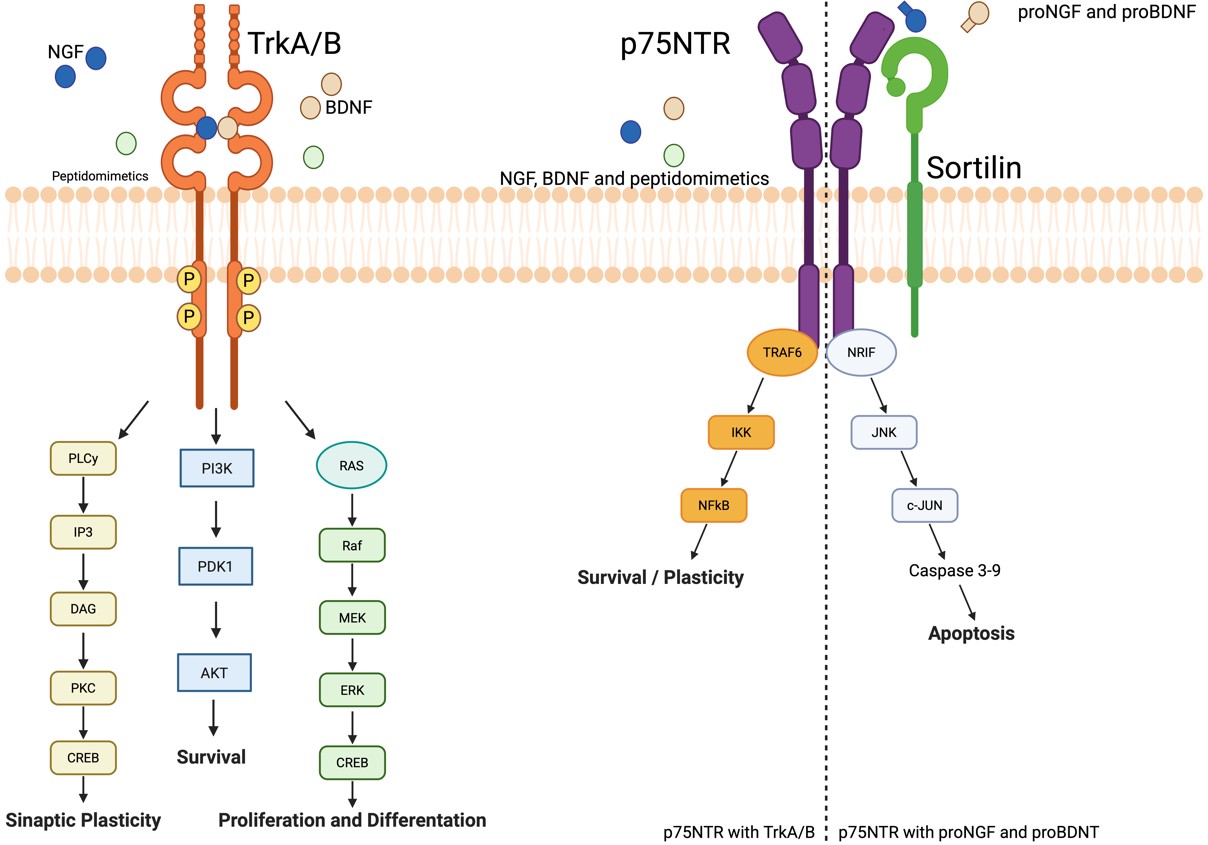 Fig. 1.
Fig. 1.
Neurotrophin receptor signaling and therapeutic modulation.
Schematic representation of the main neurotrophins (NGF, nerve growth factor;
BDNF, brain-derived neurotrophic factor; NT-3, neurotrophin-3; NT-4/5,
neurotrophin-4/5) and their interaction with high-affinity Trk receptors (Trk,
tropomyosin receptor kinases; TrkA–C) and the low-affinity
p75NTR receptor (NTR, neurotrophin receptor p75). Canonical
downstream pathways include PI3K/Akt (PI3K, phosphatidylinositol 3-kinase; AKT, A
serine/threonine kinase; cell survival), MAPK/ERK (MEK, mitogen-activated protein
kinase kinase; ERK, extracellular signal-regulated kinase; differentiation and
plasticity), and PLC
In animal models of retinal degeneration, impaired retrograde transport or reduced neurotrophin expression results in neuronal apoptosis—supporting the “neurotrophin deprivation” hypothesis in optic neuropathies [9, 10]. Preclinical studies have demonstrated that exogenous delivery of neurotrophic factors, or preservation of intrinsic neurotrophin signaling, can mitigate photoreceptor and ganglion cell loss. Notably, growing interest has emerged around neuroprotective and regenerative strategies aimed at halting or even reversing retinal damage [11]. BDNF-based interventions, for instance, have shown the ability to promote neuronal survival, yet their therapeutic potential is hindered by pharmacokinetic limitations and poor tissue penetration [12].
These findings underscore the potential of receptor-specific agonists capable of selectively engaging survival pathways while avoiding detrimental downstream signaling. This chapter will explore the development of molecularly engineered neurotrophin receptor modulators as next-generation tools designed to overcome the limitations of native neurotrophins and pave the way for effective neuroprotection and regeneration in ophthalmic neurodegeneration.
Nerve growth factor (NGF), a key member of the neurotrophin family, has long been recognized for its essential role in neuronal survival, differentiation, and regeneration. In the eye, NGF plays a critical role in maintaining corneal innervation and epithelial integrity, acting through its high-affinity receptor TrkA and low-affinity p75-NTR. These pathways are particularly relevant in the context of neurotrophic keratitis (NK), a degenerative corneal disease marked by decreased or absent corneal sensitivity, leading to impaired wound healing and epithelial breakdown [13].
The clinical application of NGF in ophthalmology reached a milestone with the approval of recombinant human NGF (rhNGF), commercially known as cenegermin, for the treatment of moderate to severe NK. This approval, granted first in the European Union in 2017 [14] and subsequently by the U.S. FDA, followed rigorous clinical trials demonstrating its efficacy and safety [15]. The efficacy of recombinant human NGF (cenegermin) has been confirmed both in individual clinical trials and in aggregate analyses. A systematic review and meta-analysis by Roumeau et al. [16] highlighted significant corneal healing rates and symptom improvement across trials, while Roszkowska et al. [17] provided updated clinical data from a prospective cohort showing consistent therapeutic benefit.
Cenegermin’s mechanism of action includes the promotion of corneal nerve regeneration and enhancement of epithelial healing—effects verified in both animal models and human subjects. Recent studies confirm the effectiveness of topical rhNGF in promoting corneal epithelial regeneration, even in refractory cases, as reported by Dai et al. [18] who observed significant healing in patients with persistent epithelial defects after rhNGF treatment.
Beyond NK, there is growing interest in expanding the therapeutic use of topical NGF to other ocular surface and retinal disorders where neurodegeneration and impaired regeneration play central roles. Experimental studies in animal models of dry eye disease, diabetic keratopathy, and retinal degeneration have demonstrated that topical nerve growth factor (NGF) accelerates epithelial healing, mitigates inflammation, and preserves neuronal structures. In the Ins2-Akita mouse model of diabetic retinopathy, topical NGF preserved retinal nerve fiber layer (RNFL) thickness, improved electrophysiological responses, and prevented retinal ganglion cell loss and microvascular damage [19]. Similarly, topical NGF has been shown to stabilize RNFL/ganglion cell layer (GCL) thickness and maintain retinal function in diabetic mice through neuroprotective mechanisms [20]. For example, in a rat model of retinal ischemia, topical NGF was shown to protect retinal ganglion cells from apoptosis and preserve visual function, likely via retrograde neurotrophic support [21].
In the context of dry eye disease (DED), topical nerve growth factor (NGF) has demonstrated promising effects on tear secretion and ocular surface integrity, although robust clinical trials are still limited. A recent Phase II randomized, vehicle-controlled, dose-ranging study evaluating recombinant human NGF (cenegermin) for moderate-to-severe DED showed significant improvements in both patient symptoms and ocular surface staining after four weeks of treatment [22]. Additionally, in a smaller Phase IIa open-label trial, rhNGF eye drops (administered at 4 µg/mL or 20 µg/mL) were well tolerated and led to reductions in dry eye signs and symptoms; notably, only the higher concentration significantly improved tear function [23]. These early findings support the biological rationale for NGF as a treatment for DED, but confirmatory studies—including larger, controlled Phase III trials—remain necessary before formal clinical adoption is warranted.
Safety data for topical NGF is favorable. The most commonly reported adverse events in clinical trials include transient eye pain, increased lacrimation, and conjunctival hyperemia—symptoms generally considered mild and manageable. Importantly, no systemic toxicity has been reported, a reassuring finding given the molecule’s biological potency and potential off-target effects [13].
Despite these promising outcomes, several challenges remain. NGF is a large protein with limited corneal permeability, necessitating frequent dosing. Stability and storage conditions are also demanding, requiring refrigeration and precise handling. Furthermore, the high cost of production translates into significant financial burden for healthcare systems and patients, raising issues of accessibility and equity [16].
In summary, the successful clinical translation of topical NGF in the treatment of neurotrophic keratitis marks a paradigm shift in ocular surface therapy—introducing a regenerative strategy that directly addresses the pathophysiology of the disease. Ongoing research aims to optimize its delivery, explore alternative indications, and develop next-generation neurotrophin-based agents with improved pharmacological properties. As our understanding of neurotrophic signaling in ocular health deepens, NGF and related molecules are poised to play an increasingly central role in the management of neurodegenerative ocular diseases.
While topical administration of recombinant human NGF (rh-NGF) such as cenegermin has shown clinical efficacy in treating NK, limitations in protein drug delivery, stability, and manufacturing cost have spurred interest in NGF-mimetic compounds—specifically, peptidomimetics designed to replicate the bioactivity of NGF through selective interaction with TrkA receptors.
Peptidomimetics are synthetic molecules that mimic the structure and function of biologically active peptides while offering advantages such as increased stability, improved bioavailability, and lower immunogenicity [24]. Recent advances in generative peptide design, including the incorporation of non-natural amino acids, have further expanded the potential of peptidomimetics as therapeutic agents [25]. In parallel, a growing body of work has focused on the development of low-molecular-weight peptide and non-peptide mimetics of NGF and BDNF that replicate the therapeutic benefits of the full-length proteins while minimizing their pharmacokinetic and immunological limitations [26].
Several rationally designed NGF-mimetic peptidomimetics have demonstrated significant neuroprotective and regenerative effects in preclinical models. A notable example is the synthetic peptide L1L4, derived from loop 1 and loop 4 of the NGF molecule and joined by a flexible linker. L1L4 was found to stimulate TrkA phosphorylation and promote differentiation in PC12 cells and chick dorsal root ganglia, showing selective activity without activating the TrkB receptor. Moreover, in vivo administration in a rat model of neuropathic pain reduced behavioral hypersensitivity and restored neuronal function, indicating potential for broader neuroprotective applications [27]. These findings were further substantiated by follow-up studies on the related NGF-mimetic BB14, which demonstrated the ability to reduce reactive astrogliosis and restore synaptic homeostasis in a rat model of peripheral nerve injury, reinforcing the therapeutic relevance of this peptide family [28].
In a parallel development, a structurally distinct NGF mimetic known as MT2—representing a class of low molecular weight, non-peptidic TrkA agonists—was shown to bind selectively to TrkA with nanomolar affinity. MT2 induced autophosphorylation of key tyrosine residues (Tyr490 strongly, Tyr785 and Tyr674/675 weakly), mirroring the intracellular signaling cascade initiated by native NGF. These effects translated into enhanced ERK1/2 and Akt phosphorylation, improved survival in serum-starved neuronal cultures, and partial neuronal differentiation. In a neuronal model of Alzheimer’s disease, MT2 reversed NGF-deprivation-induced biochemical changes, suggesting a disease-modifying potential [29].
Of particular interest in ophthalmology is the small-molecule NGF mimetic MIM-D3 (Tavilermide), which has been investigated for its ability to promote tear film stability and ocular surface healing in dry eye disease (DED). In preclinical studies, MIM-D3 induced MAPK1/2 phosphorylation and increased conjunctival glycoconjugate secretion both in vitro and in vivo. In a rat model of scopolamine-induced dry eye, topical application of MIM-D3 significantly reduced corneal staining and increased tear film mucin content, supporting its therapeutic potential in ocular surface disorders where NGF signaling is compromised [30]. These encouraging results were further supported in a randomized, placebo-controlled Phase 2 clinical trial, where MIM-D3 ophthalmic solutions showed significant improvements in ocular surface staining and symptom relief in selected patient subgroups, along with a favorable safety profile [31]. More recently, a comprehensive review of approved and investigational therapies for DED has listed MIM-D3 (Tavilermide) among the upcoming potential treatments under assessment, reflecting the continued expansion of the therapeutic pipeline for this multifactorial disease [32].
REC-0559, a TrkA-selective NGF mimetic, has emerged as a clinical-stage candidate for treating NK. Granted orphan drug designation by both the European Medicines Agency in 2014, and the U.S. Food and Drug Administration in 2018, REC-0559 entered phase II clinical trials aimed at evaluating its safety and efficacy in patients with stage 2 and 3 neurotrophic keratitis (https://www.clinicaltrialsregister.eu/ctr-search/trial/2019-002475-34/IT?utm_source=chatgpt.com). While formal trial data remain pending, preclinical findings have demonstrated its ability to activate TrkA selectively and support corneal epithelial regeneration without the drawbacks associated with full-length NGF proteins.
Peptidomimetic NGF agonists represent a next-generation approach to neurotrophic therapy. Their modular design enables the tuning of signaling properties—whether favoring survival, differentiation, or both—by targeting distinct TrkA autophosphorylation patterns and downstream pathways [33]. Such fine control may offer a safer and more effective alternative to rh-NGF, particularly in chronic or refractory conditions where sustained trophic support is essential but continuous protein exposure is impractical.
Therefore, NGF peptidomimetics are promising candidates for future NK therapies and beyond, including broader applications in retinal and optic nerve degenerations. Ongoing trials and optimization of delivery systems will determine their translational success, but the current evidence strongly supports their potential as synthetic surrogates of endogenous neurotrophins.
The emergence of synthetic microneurotrophins (MNTs) represents a pivotal evolution in neurotrophic therapy, addressing key limitations of native neurotrophins while introducing novel pharmacological opportunities. Unlike recombinant proteins such as cenegermin, which face challenges related to stability, delivery, and cost, MNTs are small-molecule compounds designed to selectively activate neurotrophin receptors with improved pharmacokinetic properties [34]. These agents expand the therapeutic potential of neurotrophin-based strategies by enabling targeted modulation of specific receptor pathways critical for neuronal survival, synaptic plasticity, and tissue repair.
A prime example of this approach is BNN27, a synthetic C17-spiroepoxy steroid derivative that mimics the neuroprotective actions of NGF through selective engagement of TrkA and p75NTR receptors [35, 36]. Its ability to cross biological barriers, including the blood-retinal barrier, and resist enzymatic degradation makes it particularly suitable for treating ocular neurodegenerative conditions. In models of diabetic retinopathy, BNN27 has demonstrated remarkable efficacy in preserving retinal function, reducing neuronal apoptosis, and mitigating vascular leakage, effects attributed to its activation of TrkA-mediated survival pathways and modulation of p75NTR signaling [37]. These findings highlight the dual advantage of MNTs: they replicate the beneficial effects of endogenous neurotrophins while overcoming their pharmacological limitations.
The versatility of MNTs is further exemplified by their ability to target different neurotrophin receptors with high specificity. For instance, BNN20, a TrkB agonist, has shown promise in promoting synaptic plasticity and neuronal survival, mirroring the effects of BDNF but with superior bioavailability [38]. In preclinical studies, BNN20 restored synaptic connectivity and reduced neurodegeneration in models of glaucoma and optic nerve injury, underscoring its potential as a therapeutic agent for diseases characterized by synaptic dysfunction and neuronal loss [39]. Similarly, ENT-A010, another TrkB-targeted MNT, has been shown to attenuate microglial activation and oxidative stress in the retina, suggesting a role in mitigating neuroinflammation associated with conditions like age-related macular degeneration [40].
The unique ability of MNTs to modulate p75NTR signaling adds another layer of therapeutic potential. While p75NTR is often associated with pro-apoptotic signaling, certain MNTs, such as ENT-A044, can redirect its activity toward survival pathways in stressed neurons [41]. This context-dependent modulation is particularly relevant for diseases like glaucoma, where retinal ganglion cells are exposed to conflicting trophic and degenerative signals. By fine-tuning p75NTR activity, MNTs offer a level of pharmacological precision that is difficult to achieve with native neurotrophins or their recombinant counterparts [42].
Despite these advances, challenges remain in optimizing the delivery and receptor selectivity of MNTs for ocular applications. While their small size and stability facilitate systemic administration, achieving therapeutic concentrations in specific ocular tissues, particularly the retina, may require localized delivery strategies [34]. Additionally, the pleiotropic effects of some MNTs necessitate careful evaluation of dosing and receptor engagement to minimize off-target effects [43].
Looking forward, the continued development of MNTs is poised to refine their therapeutic profiles and expand their clinical applicability. By leveraging advances in medicinal chemistry and receptor biology, future iterations of these compounds may offer even greater specificity and efficacy, paving the way for their integration into the treatment paradigm for a wide range of ocular neurodegenerative diseases.
In summary, synthetic microneurotrophins represent a significant leap forward in neurotrophic therapy, combining the biological precision of endogenous neurotrophins with the practical advantages of small-molecule drugs. Their ability to selectively target key receptors, modulate neuroinflammation, and promote neuronal survival positions them as a transformative approach for addressing the unmet needs of patients with vision-threatening ocular diseases.
ACD856: Among the emerging neurotrophin-based therapeutic strategies, ACD856, a
positive allosteric modulator (PAM) of tropomyosin receptor kinases (Trk), offers
a compelling rationale for application in retinal neurodegenerative diseases.
While originally developed for Alzheimer’s disease, accumulating evidence
indicates that ACD856 exerts both symptomatic and disease-modifying effects [44, 45], making it a promising candidate for retinal conditions characterized by
neurodegeneration and synaptic dysfunction, such as glaucoma and age-related
macular degeneration (AMD). Retinal ganglion cells (RGCs), which are highly
dependent on neurotrophic support—particularly through BDNF-TrkB
signaling—undergo progressive degeneration under stress conditions. ACD856
enhances BDNF-induced phosphorylation of TrkB and downstream ERK1/2 signaling,
promotes neurite outgrowth, and upregulates SNAP25, a presynaptic protein
critical for synaptic maintenance and function [44]. In addition, ACD856
demonstrates protection against amyloid-
GSB-106: The dipeptide mimetic GSB-106, a low-molecular-weight analog of
brain-derived neurotrophic factor (BDNF) loop 4, has emerged as a promising
neurotrophin agonist with considerable therapeutic potential, particularly in
neurodegenerative and ischemic pathologies affecting the retina. While originally
designed as an antidepressant agent due to its activation of TrkB receptors and
downstream signaling cascades such as PI3K/Akt, MAPK/ERK, and PLC
Cyclic neurotrophin peptides: Recent progress in peptide engineering has enabled the development of neurotrophin-mimetic cyclic peptides that hold significant promise for neuroprotective and regenerative therapies in ophthalmology. One such compound, a synthetic cyclic peptide derived from the N-terminal domain of nerve growth factor (cNGF(1–14)), has demonstrated the capacity to activate TrkA-mediated signaling in a copper-dependent manner, thereby emulating the biological activity of endogenous NGF [55]. This mimic was shown to promote TrkA phosphorylation, downstream ERK1/2 signaling, and CREB activation, leading to increased transcription of brain-derived neurotrophic factor (BDNF) and vascular endothelial growth factor (VEGF)—two mediators intimately involved in neuroprotection, nerve regeneration, and vascular remodeling. Such mechanistic effects are highly relevant to the pathophysiology of the ocular surface and anterior segment, where trophic deprivation contributes to conditions such as neurotrophic keratitis, dry eye disease, and impaired corneal wound healing.
Building on these findings, the same research group extended the strategy to design cyclic mimetics of BDNF and NT-3 (cBDNF(1–12) and cNT-3(1–13)), which showed analogous bioactivity in neuronal models, again dependent on copper ion modulation and Ctr1/CCS intracellular transport [56]. The dual role of these peptides as neurotrophic agonists and copper ionophores offers additional therapeutic potential in ocular settings where oxidative stress and metal dyshomeostasis exacerbate neuroinflammation and tissue damage, such as in glaucomatous optic neuropathy or ischemic retinopathies. Moreover, the VEGF-inducing effects of these agents, when precisely controlled, may serve to support reparative angiogenesis in the retina or optic nerve without incurring pathological neovascularization. Their small size and cyclic backbone confer notable resistance to proteolytic degradation and enhanced molecular stability, characteristics that make them suitable candidates for topical ocular administration, a route often limited by enzymatic breakdown and low retention time on the ocular surface. In sum, the integration of neurotrophic signaling, metal transport modulation, and formulation stability in these cyclic peptides positions them as innovative molecular tools for the treatment of neurodegenerative and inflammatory eye diseases [55, 56].
H105A: The search for effective therapeutic interventions for retinal degenerative diseases, such as retinitis pigmentosa (RP), has increasingly turned toward biologically active peptides due to their specificity, favorable safety profiles, and ease of delivery. Among these, the H105A variant of the 17-mer peptide derived from pigment epithelium-derived factor (PEDF) has emerged as a particularly promising candidate, demonstrating robust neuroprotective and retinoprotective activity in preclinical models. Recent studies have shown that topical application of H105A eye drops leads to significant preservation of photoreceptor cells and improved retinal function in RP mouse models carrying rd10 and RhoP23H mutations, underscoring its efficacy across diverse genetic backgrounds [57]. These protective effects are not limited to animal models; the peptide has also been shown to mitigate oxidative stress-induced photoreceptor death in human retinal organoids, reinforcing its translational potential [57]. The therapeutic mechanism appears to involve specific interaction with PEDF-R, as evidenced by diminished activity in the 17-mer[R99A] variant lacking receptor-binding ability and enhanced efficacy in the H105A variant with increased receptor affinity [58]. Furthermore, the antioxidant and cytoprotective capacity of PEDF[H105A] has been validated in a rabbit model of paraquat-induced oxidative retinal damage, a system that more closely approximates human ocular anatomy and pathophysiology [59]. Importantly, PEDF and its fragments have also demonstrated anti-inflammatory properties, such as downregulation of IL-6 in retinal pigment epithelial cells [60], supporting a dual role in both neuroprotection and inflammation modulation. The capacity of PEDF-derived peptides to preserve cone photoreceptors in models of focal phototoxicity further extends their relevance to conditions where cone survival is critical for maintaining central vision [61]. Collectively, these findings suggest that PEDF-derived peptides, particularly H105A, represent a viable and minimally invasive strategy for slowing or preventing photoreceptor degeneration. Their successful translation into clinical practice could signify a pivotal advance in peptide-based therapies for chronic retinal diseases such as RP, AMD, and diabetic retinopathy.
Retinal diseases such as diabetic retinopathy (DR), diabetic macular edema (DME), and age-related macular degeneration (AMD) result from a complex interplay of vascular dysfunction, chronic inflammation, and neurodegeneration, which together compromise retinal integrity and visual function. Building upon the neurotrophic and regenerative principles discussed in Section 2.2, new therapeutic paradigms now aim to restore tissue homeostasis by addressing the vascular and inflammatory dimensions of retinal disease alongside neuronal protection. These innovative approaches—spanning peptide-based and small-molecule agents—share convergent mechanistic goals despite acting on distinct molecular targets. UPARANT and its derivatives modulate uPAR/FPR signaling, counteracting both angiogenesis and inflammation. GPCR-directed pepducins, such as S1P–S1PR3 antagonists, further refine this concept by targeting intracellular receptor loops to regulate endothelial activation and neovascular remodeling. Integrin inhibitors (Risuteganib, THR-687, OTT166) intervene downstream, stabilizing cell–matrix adhesion and restoring vascular barrier function. Collectively, these therapies form a pathophysiological continuum, addressing complementary facets of the same disease network—angiogenic, inflammatory, and degenerative. The following subsections (2.3.1–2.3.5) explore these agents in detail, emphasizing their shared capacity to modulate pathological angiogenesis, preserve vascular integrity, and attenuate chronic inflammation. When viewed together, they exemplify the shift toward multitargeted, systems-level interventions designed to re-establish retinal equilibrium rather than suppress isolated disease symptoms.
Among these integrated approaches, the development of UPARANT exemplifies how targeted peptide design can simultaneously address angiogenic and inflammatory pathways that drive retinal pathology. The foundational work by Carriero et al. [62] established UPARANT as a stabilized urokinase receptor-derived peptide with enhanced pharmacokinetic properties and potent antiangiogenic activity, specifically designed to inhibit VEGF-driven pathological neovascularization. Further mechanistic analysis in primary human retinal endothelial cells confirmed that UPARANT dose-dependently suppresses VEGF-A-induced migration, invasion, and tube formation, while preserving endothelial barrier integrity by inhibiting VEGFR-2 and STAT3 phosphorylation as well as downstream ERK, JNK, p38, and AKT signaling [63]. However, subsequent research has revealed that UPARANT’s mechanism of action extends beyond simple uPAR antagonism, involving complex interactions with the formyl peptide receptor (FPR) system that significantly contribute to its therapeutic effects.
Early preclinical studies demonstrated UPARANT’s efficacy in ocular angiogenesis models, including oxygen-induced retinopathy and laser-induced choroidal neovascularization, where it effectively reduced pathological vessel growth [64, 65]. These antiangiogenic effects were initially attributed primarily to its interference with the uPAR system. However, further work provided crucial insight into UPARANT’s additional mechanism of action through FPR modulation. This research showed that human vitreous humor from diabetic retinopathy patients contains N-formyl peptides that activate FPRs to promote angiogenesis, and that UPARANT can counteract this effect [66]. This discovery positioned UPARANT as a dual modulator of both uPAR and FPR pathways in ocular diseases.
The anti-inflammatory properties of UPARANT, demonstrated in systemic inflammation models [67], likely stem in part from its FPR interactions, as these receptors play key roles in regulating inflammatory responses. This dual mechanism becomes particularly relevant in diabetic ocular complications, where UPARANT has shown remarkable efficacy in preserving retinal function and blood-retinal barrier integrity [68, 69]. The peptide’s ability to simultaneously target both uPAR-mediated angiogenic pathways and FPR-driven inflammatory processes may explain its superior performance in these models compared to single-pathway inhibitors.
Further supporting this dual mechanism, UPARANT has demonstrated therapeutic benefits in diverse ocular conditions including rubeosis iridis [70] and diabetic nephropathy [71], where both angiogenesis and inflammation contribute to pathology. Recent work using human iris tissue has confirmed that UPARANT’s effects involve modulation of receptor interactions beyond just uPAR, likely including FPR family members [72].
The pharmacokinetic profile of UPARANT, characterized by good stability and bioavailability [73], combined with its dual mechanism of action, makes it particularly promising for treating complex ocular diseases where multiple pathological pathways are activated. Even in neurodegenerative conditions like retinitis pigmentosa, where UPARANT showed unexpected protective effects [74], FPR modulation may contribute to its efficacy, as these receptors are involved in neuroinflammation and cellular stress responses.
This evolving understanding of UPARANT’s polypharmacology, particularly its interactions with both uPAR and FPR systems, suggests it may offer advantages over more targeted therapies in treating multifactorial ocular diseases. The peptide’s ability to simultaneously address angiogenic, inflammatory, and possibly neurodegenerative processes through these receptor systems positions it as a uniquely versatile candidate for ocular therapeutics. Future research should further elucidate the relative contributions of uPAR versus FPR modulation to UPARANT’s various biological effects, potentially leading to even more optimized therapeutic peptides for ocular diseases.
Building upon the dual uPAR/FPR modulation of UPARANT, recent efforts have refined this strategy toward more selective FPR1 antagonism, De Fenza et al. [75] have developed N-19004, a novel turn-adopting peptidomimetic designed as a specific FPR1 antagonist. This rationally designed compound, inspired by UPARANT’s structural features but optimized for FPR1 specificity, incorporates a 2,6-diamino-thiophenyl scaffold that mimics the critical Arg residues in UPARANT’s bioactive conformation. In a mouse model of choroidal neovascularization, systemic administration of N-19004 significantly reduced lesion size and promoted edema resolution, demonstrating that selective FPR1 antagonism alone can achieve therapeutic effects comparable to UPARANT’s broader mechanism [75]. This work not only validates FPR1 as a viable therapeutic target in ocular neovascular diseases but also suggests that UPARANT’s efficacy may be partially attributable to its FPR1 antagonistic properties. The development of N-19004 represents an important step toward more targeted therapies while simultaneously providing new insights into UPARANT’s polypharmacology. These findings open exciting possibilities for both improving upon UPARANT’s design and developing combination therapies that simultaneously target uPAR and FPR pathways with optimized specificity. As our understanding of these receptor systems in ocular pathophysiology deepens, such structure-guided drug design approaches may yield next-generation therapeutics with enhanced potency and selectivity for treating complex retinal diseases.
The growing understanding of G protein-coupled receptor (GPCR) signaling in pathological angiogenesis has opened new therapeutic avenues for ocular diseases characterized by aberrant neovascularization. Recent work by Avnet et al. [76] has highlighted the sphingosine-1-phosphate (S1P)-S1PR3 axis as a particularly promising target, demonstrating that specific S1PR3 antagonists can inhibit endothelial cell tubulogenesis by up to 75% in vitro, while also reducing vascular sprouting in advanced 3D microfluidic models. These findings are especially relevant for ocular pathologies like diabetic retinopathy and neovascular age-related macular degeneration (AMD), in which dysregulated angiogenesis drives disease progression.
The study’s use of KRX-725-II and its derivatives as allosteric modulators of
S1PR3 activity suggests that pepducin-based strategies—lipidated peptides
designed to target intracellular GPCR domains—could be adapted for ocular
applications. Specifically, KRX-725-II (Myristoyl-GRPYDAN-NH2) is a
nine-amino-acid pepducin modeled on the second intracellular loop of S1PR3. After
membrane insertion, it binds the inner face of the receptor—acting as a biased
allosteric antagonist that blocks Gi-dependent activation of the MEK–ERK cascade
and prevents
Pepducins were first discovered in 2002 by Covic et al. [78] as
cell-penetrating modulators of GPCR signaling, derived from the receptor’s own
transmembrane sequences. Unlike traditional orthosteric drugs, pepducins act as
biased allosteric regulators, offering precise control over specific downstream
pathways (e.g., G-protein vs.
The CXCL8-CXCR1/2 axis represents another GPCR-mediated pathway with significant relevance to ocular angiogenesis. Campbell et al. [83] detailed how this pro-inflammatory signaling system promotes endothelial proliferation and vascular remodeling, suggesting that targeted inhibition could benefit conditions like proliferative diabetic retinopathy. This is supported by earlier work in ovarian cancer models, where a CXCR1/2-targeting pepducin (X1/2pal-i3) potently inhibited IL-8-driven angiogenesis and tumor growth [84]. The ability of such pepducins to disrupt paracrine signaling between tumor cells and the vasculature suggests they could similarly interfere with pathological crosstalk between retinal cells and choroidal or retinal vasculature in ocular diseases.
The translational potential of these approaches is further evidenced by studies showing that pepducins targeting protease-activated receptors (PARs) can effectively inhibit metalloprotease-driven angiogenesis and ascites formation in ovarian cancer [85]. Given the established role of matrix metalloproteases in breakdown of the blood-retinal barrier and choroidal neovascularization, such PAR-targeting strategies could be adapted for ocular use. The demonstrated biodistribution and pharmacokinetic properties of pepducins, including their ability to act locally while avoiding systemic effects [81], make them particularly suitable for ocular administration, whether through intravitreal injection or sustained-release formulations.
Collectively, these studies paint a compelling picture of GPCR-targeting strategies-particularly pepducin-based approaches-as versatile tools for combating pathological angiogenesis in the eye. From S1PR3 antagonism to CXCR1/2 and PAR1 modulation, the growing arsenal of GPCR-targeting agents offers multiple entry points for disrupting the complex signaling networks that drive ocular neovascularization. Future research should focus on optimizing these compounds for ocular delivery and testing their efficacy in models of diabetic retinopathy, AMD, and other vision-threatening conditions where current anti-VEGF therapies show limitations. The ability to simultaneously target angiogenic and inflammatory pathways through GPCR modulation may provide much-needed alternatives for patients with resistant or recurrent disease.
The pathogenesis of diabetic retinopathy (DR) and its complications—diabetic
macular edema (DME) and proliferative diabetic retinopathy (PDR)—involves a
multifaceted disruption of vascular integrity, inflammation, and aberrant
angiogenesis. Central to these processes is the dysregulation of integrin
signaling, particularly
Risuteganib (Luminate®), a first-in-class multitargeted integrin
antagonist, disrupts the same
THR-687, a small-molecule pan-RGD integrin antagonist, extends this strategy by
potently blocking the same
Preclinical studies have provided important insights into the pharmacokinetic profile of THR-687. Using rabbit and minipig models, researchers developed a novel integrated pharmacokinetic model to characterize its intravitreal distribution and systemic exposure following administration [95]. The findings demonstrated sustained vitreous retention and minimal systemic bioavailability, a crucial feature that may reduce the risk of off-target effects compared to therapies with significant systemic spillover. This localized action is particularly advantageous in chronic retinal conditions requiring repeated dosing, as it may mitigate cumulative systemic toxicity.
Clinical translation of THR-687 has shown encouraging results. A Phase 1 open-label, dose-escalation study evaluated its safety and preliminary efficacy in patients with center-involved DME [96]. The trial, which tested doses ranging from 0.4 mg to 2.5 mg, reported no dose-limiting toxicities or serious adverse events, supporting a favorable safety profile. Notably, treatment-related ocular events were mild and transient, resolving without intervention. Beyond safety, the study documented rapid functional improvements, with patients gaining an average of 7.2 early treatment diabetic retinopathy study chart (ETDRS) letters by day 7 (95% CI, 4.1 to 10.3 letters) —an effect that persisted through the three-month follow-up period. Anatomical benefits, including reductions in central subfield thickness, further underscored its therapeutic potential. These early clinical outcomes align with preclinical data, reinforcing THR-687’s mechanism of action and durability.
The promise of THR-687 extends beyond its efficacy in DME. Given its pan-integrin antagonism, it may also hold value in other retinal vascular disorders, including neovascular AMD, where integrin signaling contributes to choroidal neovascularization and fibrosis [94]. Unlike anti-VEGF monotherapy, which primarily addresses vascular permeability, THR-687’s broader mechanism could simultaneously target inflammation and aberrant tissue remodeling, key drivers of disease progression in advanced retinopathies. This multifaceted activity positions it as a compelling candidate for combination regimens, potentially enhancing outcomes in patients with suboptimal responses to existing treatments.
Further clinical trials are needed to fully define THR-687’s therapeutic potential. Phase 2 studies should assess efficacy in larger cohorts and explore combination strategies with anti-VEGF therapies, while long-term safety and durability evaluations remain crucial for chronic retinal conditions. If confirmed in later-phase trials, THR-687 may offer a next-generation alternative for patients with limited responses to current treatments. In summary, THR-687 represents a significant advance in retinal therapeutics, with preclinical and early clinical data supporting its role as a well-tolerated, effective, and durable integrin-targeted therapy.
The development of dendrimer-peptide conjugates represents a significant advancement in targeted drug delivery for retinal diseases. Dendrimers are highly branched, nanoscale polymers with precise architectural control, enabling them to serve as versatile carriers for therapeutic peptides. Their multivalent surface allows covalent attachment of peptide payloads while protecting them from rapid enzymatic degradation–a critical limitation of free peptide therapies [86]. This protective capacity was demonstrated in vitro, where dendrimer conjugation preserved ~90% of integrin-binding peptide ALG-1001 after 1.5 hours of proteinase exposure, compared to 90% degradation of the free peptide. Beyond stabilization, dendrimers enhance tissue penetration and prolong therapeutic effects, as evidenced by sustained anti-angiogenic activity in choroidal neovascularization models [86, 97]. These properties make dendrimer-peptide systems particularly valuable for ocular conditions, where anatomical barriers and chronic disease nature demand durable, localized drug action.
Against this technological backdrop, the integrin inhibitor OTT166 emerges as a compelling case study in translating molecular targeting to clinical benefit. As a topical selective integrin antagonist, OTT166 was evaluated in a Phase 1b randomized trial for diabetic retinopathy and macular edema [98]. The study design—twice-daily eyedrops for 28 days with a 28-day follow-up—directly addressed the unmet need for non-invasive therapies in chronic retinal conditions. Safety outcomes were notably favorable, with no drug-related serious adverse events and only two mild treatment-emergent adverse events among 44 participants. While best-corrected visual acuity remained stable, optical coherence tomography revealed meaningful anatomical improvements: 37% of participants were classified as “responders”, defined as those showing a measurable reduction in central retinal thickness (CRT) from baseline, who exhibited a median CRT decrease of 46.6 µm by day 28, progressing to 67.4 µm by day 56. This delayed peak effect suggests that OTT166 may initiate biological changes that continue beyond treatment cessation, potentially linked to integrin-mediated modulation of extracellular matrix remodeling.
The differential response between treatment-naïve and previously treated subjects further illuminates OTT166’s mechanism. Naïve patients showed superior responses (median –41.5 vs. –16.5 microns in pretreated groups at 2.5% dose), implying that early intervention with integrin inhibition may yield optimal outcomes [98]. This aligns with preclinical data showing integrins regulate both vascular permeability and inflammatory cascades in diabetic retinopathy pathogenesis [99]. The topical delivery achievement is particularly significant when contrasted with intravitreal dendrimer-peptide approaches; while systemic D-ALG required nanocarrier conjugation for retinal bioavailability [86], OTT166’s apparent corneal penetration as a free molecule underscores the diversity of viable delivery strategies for integrin modulators.
Together, these developments highlight two parallel frontiers in retinal therapy: the engineering of sophisticated delivery platforms like dendrimer-peptides to overcome pharmacological barriers, and the clinical validation of targeted agents like OTT166 that address disease biology beyond VEGF. As both fields progress, their convergence may yield even more effective solutions–perhaps dendrimer-formulated integrin inhibitors combining optimized delivery with multi-pathway therapeutic effects.
The pathogenesis of diabetic retinopathy and its complications involves a
multifaceted disruption of vascular integrity, inflammation, and aberrant
angiogenesis, with integrin signaling playing a central regulatory role. The
development of integrin antagonists like Risuteganib, THR-687, and OTT166
represents a paradigm shift in retinal therapeutics, offering mechanisms that
extend beyond VEGF inhibition to address the complex pathophysiology of diabetic
eye disease. Preclinical and clinical studies demonstrate these agents’ ability
to simultaneously target vascular permeability, pathological neovascularization,
and oxidative stress through selective modulation of
The accumulation of senescent cells—dysfunctional cells resistant to apoptosis that secrete pro-inflammatory and pro-angiogenic factors—has emerged as a key contributor to the pathogenesis of age-related and metabolic ocular diseases, including diabetic retinopathy (DR), diabetic macular edema (DME), and age-related macular degeneration (AMD). Cellular senescence disrupts tissue homeostasis by promoting chronic inflammation (via the senescence-associated secretory phenotype, SASP), vascular dysfunction, and extracellular matrix degradation. In the retina, senescent retinal pigment epithelial (RPE) cells and endothelial cells drive disease progression by exacerbating oxidative stress, blood-retinal barrier breakdown, and pathological angiogenesis [101, 102].
Senolytic agents, which selectively eliminate senescent cells, and senomorphics, which suppress SASP activity, represent a promising therapeutic strategy. Preclinical studies have demonstrated that senolytics such as ABT-263 (navitoclax), a Bcl-2/Bcl-xL inhibitor, reduce senescent RPE burden and attenuate retinal degeneration in AMD models by restoring phagocytic function and decreasing inflammatory cytokine release [103]. Similarly, procyanidin C1, a natural flavonoid with dual senolytic and senomorphic properties, improved retinal structure and function in aged mice by clearing senescent cells and reducing SASP-mediated damage [104]. Notably, UBX1325 (foselutoclax), a Bcl-xL inhibitor optimized for intravitreal delivery, has advanced to clinical trials for DME. In a phase 1 study, a single injection of UBX1325 in patients with anti-VEGF-resistant DME led to sustained reductions in central retinal thickness and improvements in visual acuity, with effects persisting for up to 48 weeks [105, 106]. These findings suggest that senolysis may offer durable benefits compared to conventional therapies, which require frequent dosing.
Innovative delivery approaches are also being explored. Supramolecular senolytic peptides, designed to oligomerize intracellularly in response to elevated reactive oxygen species (ROS) in senescent cells, have shown efficacy in AMD models by selectively eliminating dysfunctional RPE without harming healthy tissue [107]. Additionally, HSP90 inhibitors (e.g., IPI-504) exhibit senomorphic effects by downregulating SASP factors like IL-6 and MCP-1 in RPE cells, offering an alternative strategy for AMD [108].
The translational potential of senotherapeutics is further highlighted in diabetic retinopathy, where senescent endothelial cells contribute to microvascular dysfunction. Preclinical studies indicate that senolytics can ameliorate retinal vascular leakage and inflammation, suggesting utility in both non-proliferative DR and DME [109]. Clinical trials of UBX1325 now aim to validate these mechanisms, with early data showing promise in patients with refractory DME [106].
Challenges remain, including optimizing tissue-specific delivery and minimizing off-target effects. However, the convergence of senolytic research with advancements in nanotechnology (e.g., ROS-responsive peptides) and biomarker identification (e.g., p16INK4a expression) is accelerating the development of precision therapies. As evidence grows for senescence as a unifying pathway in retinal diseases, senolytics may soon complement or even replace current standards of care, particularly for patients with suboptimal responses to anti-VEGF therapy. Future directions include combination strategies—pairing senolytics with anti-angiogenics or neuroprotectants—to address the multifactorial nature of diseases like AMD and DME [110].
In summary, senolytic therapy represents another paradigm shift in ocular disease treatment, moving beyond symptom management to target root causes of cellular dysfunction. With ongoing clinical validation, these agents could redefine therapeutic approaches for degenerative retinal conditions, offering hope for long-term disease modification (Fig. 2).
 Fig. 2.
Fig. 2.
Mechanistic and therapeutic convergence in retinal disease. Integration of four major therapeutic axes in ocular pharmacology: (1) neuroprotection through neurotrophins and receptor agonists; (2) vascular stabilization via integrin inhibitors and peptide modulators (e.g., risuteganib, UPARANT, Urokinase Plasminogen Activator Receptor ANTagonist); (3) clearance of senescent cells using senolytic or senomorphic agents (e.g., UBX1325, procyanidin C1); and (4) advanced drug delivery systems enabling targeted and sustained release. The figure illustrates how these pillars converge on shared pathophysiological processes—oxidative stress, chronic inflammation, and vascular dysfunction—across diseases such as AMD, diabetic retinopathy, glaucoma, and neurotrophic keratitis. Together, they represent a multidimensional therapeutic paradigm aimed at restoring rather than merely preserving vision. Created in BioRender. Avitabile, A. (2025) https://BioRender.com/uw6fw2q.
Despite encouraging progress, several challenges temper the translational outlook of these innovative strategies. Peptide-based therapeutics, including UPARANT and microneurotrophins, are inherently susceptible to enzymatic degradation, short systemic half-life, and potential immunogenicity—issues that necessitate advanced formulation approaches such as sustained-release matrices, intravitreal depots, or conjugation with biodegradable carriers [111, 112]. Moreover, their target selectivity can vary depending on receptor polymorphisms and tissue microenvironment, complicating the predictability of in vivo outcomes.
Similarly, integrin antagonists and GPCR-targeting compounds face hurdles related to dose optimization and receptor desensitization. While topical or intravitreal delivery has shown promise, maintaining consistent therapeutic concentrations in posterior tissues remains a technical challenge. Long-term studies are also required to establish ocular and systemic safety profiles, particularly regarding potential interference with physiological angiogenesis or wound healing [113].
Finally, the application of senolytic drugs to ocular diseases—though mechanistically compelling—raises concerns about off-target cytotoxicity and chronic-use safety. The long-term effects of clearing senescent cells within the delicate neurovascular retina are not yet fully understood, and refined dosing schedules or chronotherapeutic regimens may be needed to minimize collateral tissue stress [114, 115].
A critical next step in the clinical development of these therapies lies in balancing potency with safety, improving targeted delivery, and integrating temporal and molecular precision to fully realize the regenerative potential of these approaches in human retinal disease.
The current landscape of ophthalmic pharmacology is characterized by a shift from isolated interventions to a multidimensional therapeutic matrix. This framework strategically integrates four complementary pillars: neuroprotection, vascular stabilization, senescence clearance, and advanced drug delivery. Neurotrophins and peptidomimetics, such as the FDA-approved topical cenegermin which supports neuronal survival through receptor activation [14], provide the foundation for halting and reversing neuronal degeneration. Simultaneously, integrin antagonists and peptide-based agents like UPARANT—which demonstrates antiangiogenic and anti-inflammatory effects by inhibiting the urokinase receptor and modulating formyl peptide receptor (FPR) signaling [70]—directly address vascular leakage and pathological angiogenesis. The third pillar is revolutionized by senolytic compounds such as UBX1325 (foselutoclax), which targets Bcl-xL to selectively eliminate senescent cells and has shown encouraging efficacy in retinal diseases like diabetic macular edema, particularly for patients unresponsive to anti-VEGF therapy [106]. Critically, these mechanistic advances are enabled by sophisticated delivery platforms—such as dendrimer-peptide conjugates and optimized topical formulations—that overcome traditional pharmacological barriers to ensure effective and sustained drug action at the target site.
The synergy between these pillars creates powerful potential for combination strategies. In conditions like age-related macular degeneration, the concurrent application of a senolytic to remove dysfunctional RPE cells and a neuroprotective microneurotrophin could yield greater benefits than either approach alone. Similarly, in diabetic retinopathy, an integrin modulator that stabilizes the vasculature may work synergistically with an agent like UPARANT that suppresses inflammatory and angiogenic pathways. This integrated, multi-target strategy represents the core of next-generation ocular therapeutics, moving beyond single-pathway inhibition to address the complex, interconnected pathophysiology of degenerative eye diseases.
The principal classes of peptide-based therapeutics currently under development for ocular diseases—including recombinant neurotrophins, NGF peptidomimetics, synthetic microneurotrophins, and multitargeted vascular/inflammatory modulators—are summarized in Table 1.
| Therapeutic class | Examples | Indications | Mechanism of action | Stage of development |
| Recombinant proteins | Cenegermin | NK | Recombinant human nerve growth factor (rhNGF); activates TrkA/p75NTR to promote corneal nerve regeneration and epithelial healing | Approved (EMA & FDA) |
| NGF peptidomimetics | REC-0559, Tavilermide, MIM-D3 | NK, Dry Eye Disease | Small-molecule or peptide NGF mimetics; selectively activate TrkA or modulate NGF receptor signaling to support trophic repair without full NGF liability | Preclinical–Phase II |
| Synthetic microneurotrophins | BNN27, ENT-A010, BNN20 | DR, AMD, Glaucoma | Steroid-derived TrkA/TrkB/p75NTR ligands; neuroprotective and anti-inflammatory effects in retinal and optic nerve models | Preclinical |
| Other peptide agents | UPARANT, Pepducins, Integrin inhibitors (Risuteganib, THR-687, OTT166) | DR, CNV, AMD | Peptide modulators of uPAR/FPR, formyl peptide receptor or GPCR signaling (UPARANT, pepducins) and RGD-integrin antagonists ( |
Phase I–III (depending on compound) |
AMD, Age-related macular degeneration; CNV, Choroidal neovascularization; DR, Diabetic retinopathy; NK, Neurotrophic keratitis; NGF, Nerve growth factor; RGD, Arg-Gly-Asp motif; Trk, Tropomyosin receptor kinase; FPR, formyl peptide receptor; GPCR, G protein-coupled receptor; NTR, neurotrophin receptor; EMA, European Medicine Agency; FDA, Federal Drug Administration; UPARANT, Urokinase Plasminogen Activator Receptor ANTagonist.
An emerging dimension in ocular regenerative pharmacology concerns the temporal (circadian) dimension of tissue responsiveness and drug-target oscillation—in other words, the promise of chronotherapy. Several of the molecular pathways discussed in this review show evidence of circadian regulation, suggesting that aligning treatment timing with endogenous rhythms may enhance therapeutic efficacy and reduce side-effects. For example, the low-affinity neurotrophin receptor p75NTR is itself a bona fide clock-controlled gene, with expression driven by the CLOCK:BMAL1 complex and shown to influence rhythmicity of clock genes in central and peripheral tissues [116]. Moreover, its cognate high-affinity receptor TrkB (for BDNF) exhibits a circadian pattern of activation in retina and hippocampus—a finding directly relevant to ocular neurotrophic interventions [117, 118]. This linkage raises the possibility that neurotrophin-based treatments (for example RGC/neuroprotection in glaucoma or optic neuropathy) might show improved outcomes if dosed at times when the receptor/ligand system is at peak responsiveness.
Turning to the senescence and regenerative side, cellular senescence itself appears to modulate circadian clocks: replicative senescent human fibroblasts show prolonged circadian periods and delayed phases [119, 120]. More broadly, clock regulators such as CLOCK and BMAL1 are now recognized as upstream modulators of senescence and the senescence-associated secretory phenotype (SASP) [121]. Recent evidence indicates that circadian disruption amplifies SASP activity and inflammatory signaling, whereas timed senolytic administration aligned with peak SASP oscillations may enhance clearance efficiency and reduce collateral stress responses [121, 122]. In the ocular context—where senolytic agents are gaining traction for age-related pathologies (macular degeneration, glaucoma, etc.)—it follows that timing senolytic administration to when SASP expression or inflammatory surges are maximal may yield better clearance of senescent cells and less collateral damage to non-senescent tissue.
Finally, the retina itself is endowed with strong intrinsic circadian oscillators, modulating visual processing and light-entrainment pathways [123, 124]. Disruption of retinal circadian machinery (e.g., via RGC loss in glaucoma) can therefore impair not only vision but systemic circadian entrainment. Accordingly, therapies that restore RGC function (e.g., neurotrophins) may have the added benefit of re-establishing circadian integrity. Conversely, chronobiological factors (light exposure timing, sleep–wake schedules) may influence therapeutic efficacy and should be considered in clinical trial design and patient management.
In sum, incorporating the temporal axis into precision ophthalmology—i.e., what time of day, in addition to what therapy and what dose—offers a complementary layer of optimization. We suggest that future pre-clinical and clinical studies in regenerative ophthalmology formally record and report timing of drug/biologic administration, include chronotype and sleep-wake background of subjects, and when feasible test diurnal-versus-noon-versus-night dosing arms. This chrono-pharmacological dimension adds minimal complexity but could yield outsized benefit in maximizing the regenerative potential of the modalities discussed in this review.
The paradigm for treating ocular disease is undergoing a profound transformation, shifting from palliative management to active restoration of visual function. This review has highlighted the convergence of innovative strategies—neurotrophin receptor agonism, multitargeted peptide therapies, integrin modulation, and senolytic clearance—that collectively target the multifaceted nature of conditions like diabetic retinopathy, age-related macular degeneration, and neurotrophic keratitis. The future of ocular therapeutics lies not in isolated magic bullets, but in the rational combination of these approaches, enabled by advanced delivery systems. By simultaneously supporting neuronal survival, stabilizing the vascular environment, eliminating senescent cells, and ensuring precise drug delivery, this integrated framework heralds a new era of precision medicine capable of fundamentally altering disease trajectories and restoring vision.
DR, AA, and CG designed the research. DR and CG developed the overall concept. DR wrote the initial draft of the paper. DR and CG integrated and refined the key highlights. AA conducted the literature search and provided help and advice on figures and table design. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We gratefully acknowledge the logistic support of NEST (Neurovisual Science Technology, Catania, Italy) given to Alessandro Avitabile.
This research received no external funding.
All authors declare no conflicts of interest. Despite Dr. Alessandro Avitabile is affiliated with Neurovisual Science Technology (NEST) srl; Dr. Dario Rusciano Fidia is affiliated with Ophthalmic Research; the judgments in data interpretation and writing were not influenced by this relationship. Given his role as the Editorial Board member, Dario Rusciano had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Graham Pawelec.
During the preparation of this work the authors used ChatGpt-3.5 and DeepSeek in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


