1 School of Acupuncture-Moxibustion and Tuina, Nanjing University of Chinese Medicine, 210023 Nanjing, Jiangsu, China
2 Yizheng Clinical School, Jiangsu Health Vocational College, 211400 Yangzhou, Jiangsu, China
3 Department of Ophthalmology, Nanjing Drum Tower Hospital, The Affiliated Hospital of Nanjing University Medical School, 210000 Nanjing, Jiangsu, China
4 Now with Rehabilitation Department, Xuzhou Tongshan District Hospital of Traditional Chinese Medicine, 221100 Xuzhou, Jiangsu, China
5 Department of Ophthalmology, Affiliated Hospital of Nanjing University of Chinese Medicine, 210029 Nanjing, Jiangsu, China
6 Traditional Chinese Medicine Rehabilitation Center, The Second Affiliated Hospital of Nanjing University of Chinese Medicine, 210017 Nanjing, Jiangsu, China
†These authors contributed equally.
Abstract
Altered homeostasis of the ocular surface microenvironment is a hallmark of dry eye disease (DED). The alpha-7 nicotinic acetylcholine receptor (α7nAChR) plays a key role in DED pathophysiology. In this study, we established a rabbit model of DED using scopolamine hydrobromide (Scop) to determine the effect of electroacupuncture (EA) on ocular surface damage in DED and to explore its underlying mechanisms.
New Zealand White rabbits (1.5–2.0 kg) were subcutaneously administered Scop for 21 days prior to EA treatment. After 35 days, the homeostasis of the ocular surface microenvironment was evaluated using the Schirmer I test (SIT), tear break-up time (BUT), corneal fluorescein (FL) staining, and measurement of tear osmolarity. The expression levels of ACh, α7nAChR, and high mobility group box 1 (HMGB1) were detected via histopathological examination of the cornea, lacrimal glands, and conjunctiva, combined with immunohistochemistry and western blotting. Additionally, protein chip technology was used to determine the expression levels of downstream factors.
EA stimulation significantly improved the homeostasis of the ocular surface microenvironment, as evidenced by increased SIT values and BUT, reduced corneal FL intensity, and decreased tear osmolarity. It also alleviated pathological damage to the cornea, conjunctiva, and lacrimal glands; upregulated the expression of ACh and α7nAChR; and downregulated the expression of HMGB1 and related inflammatory factors. However, these changes were reversed following administration of α-Bungarotoxin.
EA stimulation improves ocular surface homeostasis and reduces inflammation in DED, potentially via activation of the α7nAChR signaling pathway, which in turn inhibits the expression of HMGB1 and inflammatory factors.
Keywords
- electroacupuncture
- dry eye
- inflammation
- alpha-7 nicotinic acetylcholine receptor
- high mobility group box 1
Dry eye disease (DED) is a prevalent multifactorial ocular surface disorder triggered by factors such as pharmaceutical use, hormonal imbalance, corneal refractive surgery, systemic diseases, and allergic reactions [1]. These etiologies disrupt tear film stability and homeostasis, initiating a self-perpetuating cycle of ocular surface inflammation and damage to the ocular surface microenvironment [2]. Clinical manifestations include dryness, foreign body sensation, burning, and blurred vision, which significantly impair patients’ quality of life [3, 4]. Current therapeutic strategies for DED only provide transient symptomatic relief. Long-term use of corticosteroids may cause ocular surface damage, while cyclosporine-A ophthalmic emulsion requires several weeks to months to take effect and exhibits limitations in inhibiting innate pro-inflammatory mediators [5]. Thus, there is an urgent need to develop safer and more effective therapeutic approaches for DED.
Electroacupuncture (EA), a key therapeutic modality in traditional Chinese medicine, involves delivering electrical stimulation to specific acupoints with adjustable frequency and intensity to modulate neurological, immunological, and other physiological functions [6, 7]. As a safe and cost-effective intervention, EA exerts anti-inflammatory effects and is recommended for restoring homeostatic balance [7, 8, 9, 10]. Studies have confirmed the efficacy, safety, and feasibility of EA in improving DED symptoms and signs, with significant enhancements in the Ocular Surface Disease Index (OSDI), Schirmer I test (SIT) scores, and tear film break-up time (BUT) [11, 12]. Our previous research verified that EA can suppress inflammation in the cornea and lacrimal glands, potentially through vagus nerve-mediated regulation of neuroimmune responses to inhibit inflammatory reactions [13, 14]. As previously reported, EA can activate the cholinergic anti-inflammatory pathway [15].
This pathway is initiated by the binding of acetylcholine (ACh)—released from
the vagus nerve—to the alpha-7 nicotinic ACh receptor (
HMGB1, a non-histone nuclear protein belonging to the damage-associated molecular pattern family, has been detected in the tear fluid of patients with ocular surface inflammatory conditions such as conjunctivitis, blepharitis, and DED [19]. Under quiescent conditions, HMGB1 is localized intracellularly as an inflammatory mediator; however, its extracellular release can activate the innate immune response, triggering a pro-inflammatory vicious cycle that sustains immune activation and contributes to tissue damage.
To date, the mechanism underlying the role of the
Adult New Zealand white rabbits (1.5–2.0 kg) were purchased from the Qinglong
Mountain Experimental Animal Center (No. SCXK Jiangsu 2024-0001; Nanjing, China)
and housed in the Pharmacology Animal Laboratory of Jiangsu Province Hospital of
Traditional Chinese Medicine (Jiangsu, China). Housing conditions were maintained
at a constant temperature of 22 °C
The DED model was established by subcutaneous injection of Scop at the nape of the neck (Fig. 1A) at a dose of 2.0 mg/kg per injection, administered four times daily (8:00, 11:00, 14:00, and 18:00) for 21 consecutive days. Successful model establishment was confirmed by strong positive corneal fluorescein (FL) staining (Fig. 1B).
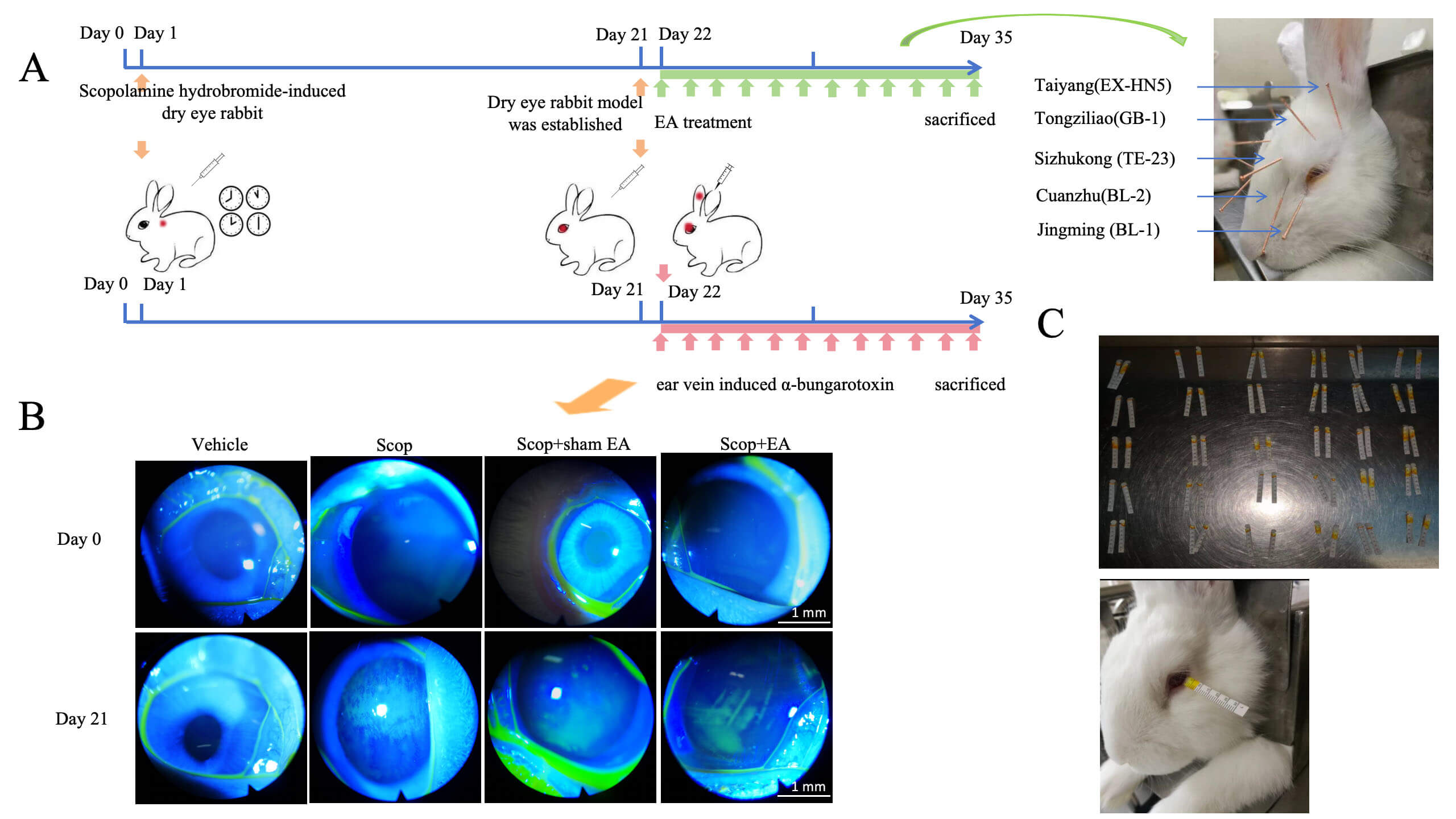 Fig. 1.
Fig. 1.
Schematic diagram of the experimental principle. (A) Adult New
Zealand white rabbits received four daily subcutaneous injections of Scop for 21
consecutive days. From day 22, EA stimulation was applied to bilateral ocular
acupoints Jingming (BL-1), Cuanzhu (BL-2), Sizhukong (TE-23), Tongziliao (GB-1),
Taiyang (EX-HN5). And
Experiment 1: Male adult New Zealand white rabbits were randomly divided into
four groups (n = 6 per group): (1) Vehicle control group; (2) Scop group; (3)
Scop + sham group; and (4) Scop + EA group. Experiment 2: Adult New
Zealand white rabbits of both sexes were randomly divided into five groups (n = 6
per group): (1) Vehicle control group; (2) Scop group; (3) Scop + EA group; (4)
Scop +
EA treatment was initiated on day 22 and administered once daily for 14
consecutive days. For the Scop + sham EA group, blunt needle puncture at
acupoints was performed daily from day 22 for 14 days. A selective
EA stimulation was performed using the Hwato Electronic Acupuncture Stimulator SDZ-IIB Health Care Device (Nanjing Jisheng Medical Technology Co., Ltd., Nanjing, China). Rabbits were restrained in a specialized rabbit fixation box. Bilateral ocular acupoints, including Jingming (BL-1), Cuanzhu (BL-2), Sizhukong (TE-23), Tongziliao (GB-1), and Taiyang (EX-HN5), were selected for EA stimulation. Acupuncture needles (0.3 mm diameter, 15 mm length; Hwato) were inserted subcutaneously to a 5 mm depth. EA was delivered with a sparse-dense wave (2 Hz/20 Hz), 0.2 ms pulse width, and 1 mA intensity (adjusted to elicit slight muscle twitching at the insertion site). The positive electrode of the EA device was connected to Jingming (BL-1), and the negative electrode to the ipsilateral Taiyang (EX-HN5). EA stimulation was applied for 20 min per day.
A corneal staining filter paper strip was placed into the lower eyelid fornix of
the rabbit and moistened. FL was evenly distributed over the cornea through
blinking. Corneal epithelial damage was graded using a cobalt blue filter. The
cornea was divided into four quadrants, with scoring criteria as follows: 0 (no
staining), 1 (
After moistening the FL sodium staining paper, it was gently touched to the conjunctival fornix of the lower eyelid 2–3 times. The rabbit was encouraged to blink several times to facilitate complete diffusion and distribution of the dye across the ocular surface. Under a slit lamp (Chongqing Shang Bang Medical Equipment Co., Ltd., Chongqing, China), the timer recorded the time of the first corneal dry spot, repeatedly measured three times, and the average time was used.
One end of the Schirmer strip was folded and inserted into the space between the middle and outer one-third of the lower eyelid and left in place for 5 min. The wet length of the strip was recorded after the strip was taken out (Fig. 1C).
Tear samples (20–50 µL) were collected from the lateral palpebral fissure (lacrimal lake) using a glass microcapillary pipette. Tear osmolarity was measured using the Vapro 5600 Osmometer (Wescor Inc., South Logan, UT, USA).
After the experimental protocol, animals were euthanized for the collection of corneas, lacrimal glands, and conjunctivae. These excised tissues were subsequently fixed in 4% paraformaldehyde, embedded in paraffin, sectioned at 5 µm, dried, and stained with hematoxylin and eosin. Histological changes were assessed under light microscopy.
A portion of the conjunctival tissue was excised, rinsed thoroughly with
physiological saline, and weighed. Then the tissue was homogenized under ice bath
conditions. Subsequently, the homogenate was diluted with 300 µL
physiological saline and centrifuged, after which the supernatant was collected.
A cholinesterase inhibitor (neostigmine, 10 µM) was added during
tissue collection/homogenization. Samples were kept on ice, and assays were
completed within 2 h of homogenization. The concentrations of ACh (JEB-14586;
Nanjing Jin Yibai, China) and
Conjunctival tissues were processed for embedding and sectioning, followed by antigen retrieval and blocking. Sections were incubated with a primary anti-HMGB1 antibody (1:500, GB11103-100; Servicebio, Wuhan, China) at 4 °C overnight, followed by incubation with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG secondary antibody (1:1000, RS0002; ImmunoWay, San Jose, CA, USA) and staining with 3,3′-diaminobenzidine. Nuclei were counterstained with hematoxylin. Using Image-Pro Plus 6.0 software (National Institutes of Health, Bethesda, MD, USA), the integrated optical density (IOD) of positive staining and pixel area were measured, and the areal density was calculated as IOD/area.
Conjunctival paraffin sections were dewaxed through a standard xylene-ethanol
gradient and rehydrated. After antigen retrieval (citrate buffer, pH 6.0, 95 °C
for 20 min) and blocking in 5% goat serum (room temperature, 30 min), sections
were divided into two groups for single-labeling detection. Then the sections
were incubated overnight at 4 °C with primary antibodies against HMGB1 (1:300,
GB11103-100; Servicebio) and
Conjunctival paraffin sections were dewaxed and stained with Periodic-acid Schiff reagent to highlight intracellular stored mucus and the secretory products of goblet cells. Positively stained goblet cells in the conjunctiva were counted, and the distance between the first and last goblet cells was measured to calculate the average number of goblet cells per millimeter.
Initial fixation was conducted in 0.1 mol/L phosphate buffer (pH 7.4) with 2.5% glutaraldehyde, followed by post-fixation in 1% osmium tetroxide. After dehydration through an ascending series of alcohols, the samples were embedded in epoxy resin. Then, small blocks (1 mm3) were excised from the central region of the conjunctiva. These blocks were subjected to double staining with lead acetate and uranyl acetate, after which they were observed under a transmission electron microscope.
Equal amounts of cell lysates were extracted from the conjunctiva, and the
protein concentration was determined using the BCA protein assay. Protein was
resolved by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis,
electrotransferred to a PVDF membrane (Millipore, Burlington, MA, USA), blocked
in 5% non-fat dry milk, and incubated overnight at 4 °C with the
following primary antibodies: anti-HMGB1 rabbit polyclonal antibody (1:1000, GB11103, Servicebio) ,
anti-
Total protein was extracted from the conjunctiva, and the protein concentration
was measured using the BCA protein assay. A rabbit cytokine quantification array
(QAL-CYT-1 kit; RayBiotech, Inc., Norcross, GA, USA) was employed to detect the
expression of 10 cytokines (interleukin 1
All experimental procedures were performed in at least three independent
replicates. All data are presented as the mean
To determine whether EA stimulation can alleviate ocular surface functional abnormalities resulting from DED, a series of assessments was conducted following 3 and 5 weeks of continuous subcutaneous injection of Scop. At 3 and 5 weeks, the FL was elevated in the Scop and Scop + Sham EA groups compared to the Vehicle control group, indicating severe corneal epithelial damage in these Scop-treated groups. Concurrently, the BUT was significantly reduced in all three groups relative to the vehicle control group (Fig. 1B and Fig. 2B,D). After 5 weeks of Scop injection, the tear osmolarity was markedly increased, while the SIT was significantly decreased in the Scop and Scop + Sham EA groups (Fig. 2A,B,D,F,G). EA stimulation for 2 weeks counteracted these adverse effects. These results suggest that EA exerts protective effects against corneal epithelial damage, consistent with the improved epithelial layer integrity observed in the histological sections (Fig. 2C). Additionally, EA alleviated ocular surface damage and mitigated ocular functional abnormalities induced by DED, with specific benefits in reducing tear osmolarity and enhancing tear film stability.
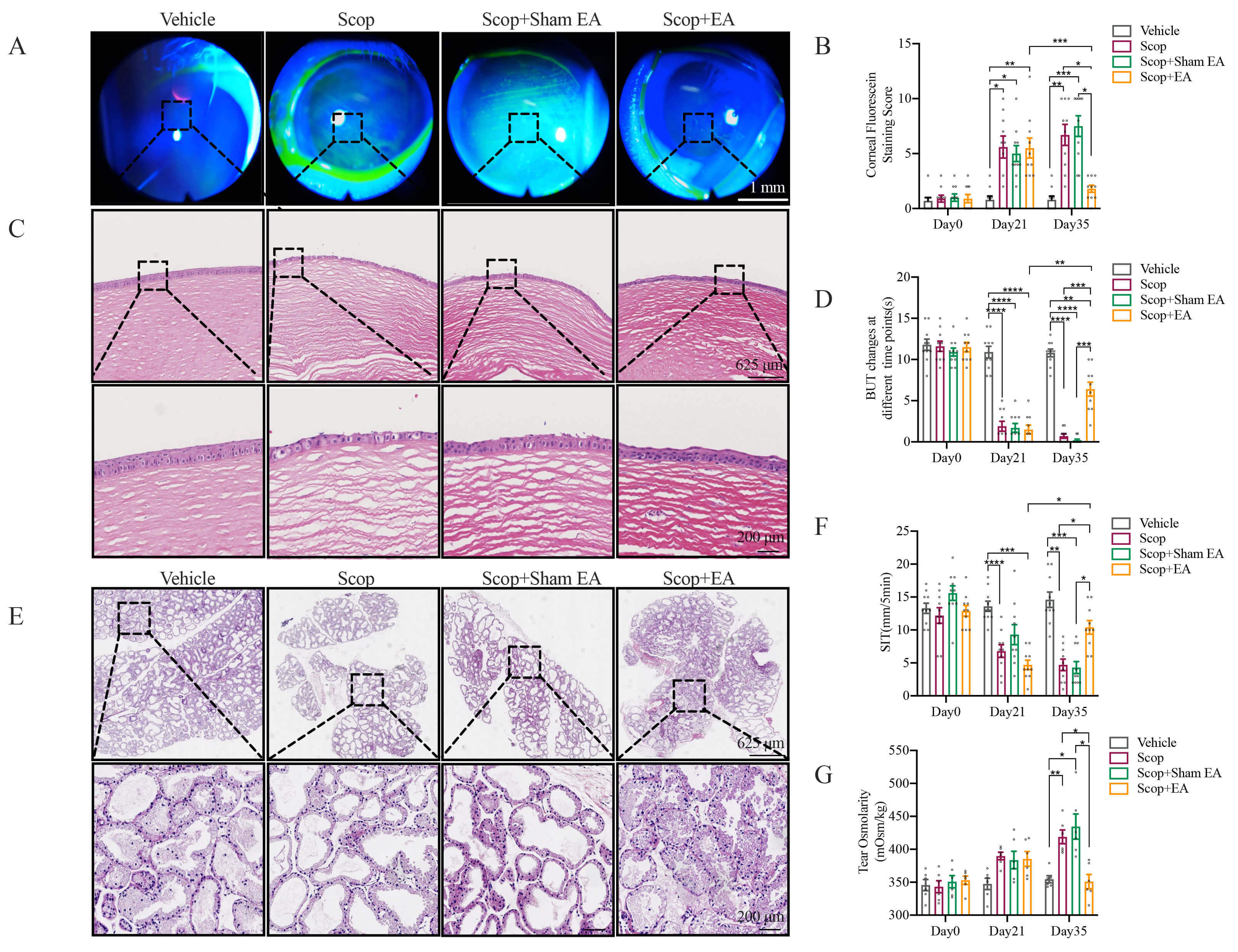 Fig. 2.
Fig. 2.
EA stimulation alleviates ocular surface tissue damage and
functional impairment in DED. (A) Representative images of corneal FL staining
on day 35 (The magnification of the slit lamp lens is 10
The impact of EA stimulation on the mitigation of ocular surface tissue damage caused by DED was assessed by histopathological analysis. After 5 weeks of Scop injection, the corneal epithelial layers were diminished with superficial squamous epithelial cells sloughing off, accompanied by edema in the stromal layer and visible corneal epithelial shedding in the Scop and Scop + Sham EA groups. EA stimulation notably improved the corneal damage, evidenced by an increase in corneal layers, a less disordered structure of the superficial squamous epithelium, and a reduction in corneal epithelial shedding and stromal edema (Fig. 2A,C). The lacrimal glands, which primarily secrete the aqueous component of tears, displayed acinar atrophy and increased cavity size compared to the Vehicle control group in the Scop and Scop + Sham EA groups. EA stimulation attenuated the acinar atrophy (Fig. 2E). The conjunctival epithelial cells showed disrupted layers and polarity, with infiltration of lymphocytes and neutrophils, and partial epithelial cell detachment in the Scop and Scop + Sham EA groups. EA stimulation similarly ameliorated the conjunctival epithelial cells, with a decrease in epithelial cell detachment and a reduction in lymphocytes (Fig. 3A). Conjunctival goblet cells secrete conjunctival mucin to form the mucin layer of the tear film, which is pivotal for maintaining tear film stability and ocular surface homeostasis [23, 24]. In comparison to the Vehicle control group, the Scop and Scop + Sham EA groups notably decreased the number of conjunctival goblet cells, whereas EA stimulation increased the number of goblet cells (Fig. 3B,C).
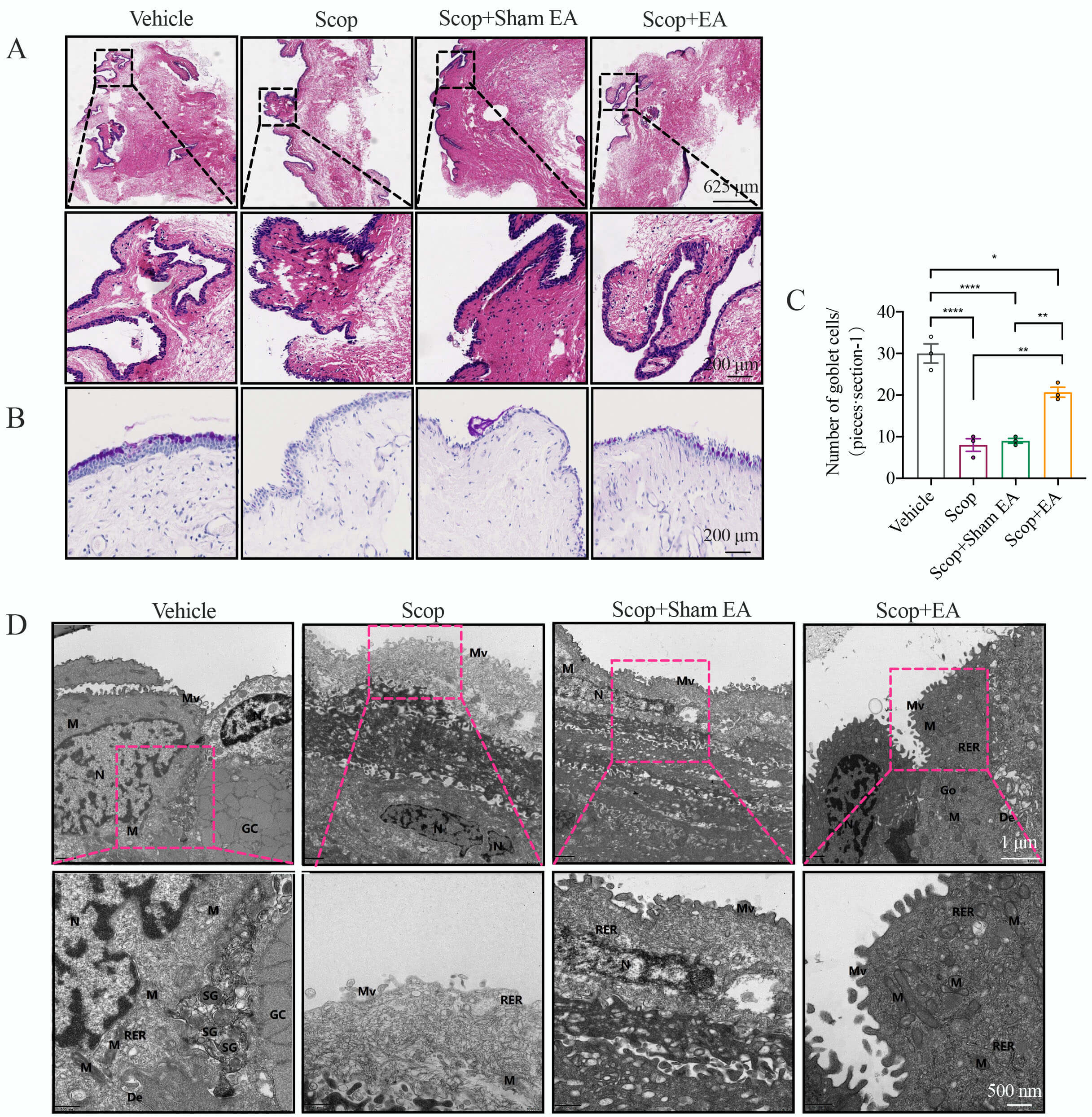 Fig. 3.
Fig. 3.
EA stimulation modulates conjunctival tissue and ultrastructure
in DED. (A) Conjunctival H&E staining on day 35 (n = 3). Scale bar = 625
µm. (B,C) Periodic-acid Schiff (PAS) staining of conjunctival goblet cells
and counting of goblet cells in each group (n = 3). Scale bar = 200 µm. (D)
Transmission electron microscopy of conjunctival epithelial cells, depicting
microvilli (Mv), mitochondria (M), RER, nuclei (N), nucleoli (Nu), desmosomes
(De), goblet cells (GCs), and secretory granules (SGs). Scale bars = 1 µm
or 500 nm. Quantitative data are presented as the mean
The ultrastructural integrity of conjunctival epithelial cells was assessed in the Vehicle control group, revealing a pattern of short-columnar arrangement with uniform cytoplasmic density, continuous cell membrane structures, and the presence of microvilli along the membranes. The mitochondrial membranes displayed elevated density, and the rough endoplasmic reticulum (RER) exhibited no signs of dilation, with ribosomes firmly anchored to its surface. Intercellular bridges were observed, and no significant widening of the intercellular spaces was noted. A profusion of secretory granules was evident adjacent to the goblet cells. By contrast, the Scop group displayed flattened cells with necrotic outermost layers. The cell membranes had microvilli, but the cytoplasm was sparse and disintegrating with a diminished number of organelles. Mitochondria were swollen, the membrane appeared blurred, and the matrix and cristae were slightly sparse. Intermediate layer cells were in a state of contraction, with a higher cytoplasmic density and increased intercellular space. The Scop + Sham EA group exhibited flattened cells, with microvilli attached to the membranes, and nuclei were elongated, with minimal accumulation of heterochromatin at the margins and a blurred nuclear membrane. Mitochondria were swollen, with a slightly sparse matrix and cristae, and the RER showed slight dilation with sparse ribosomes on its surface. Deeper-layer cells appeared contracted, with widened intercellular spaces. The Scop + EA group showed cells arranged in a short-columnar pattern, with uniform distribution of the cytoplasm. The cell membrane was intact, but microvilli were sparsely distributed. Nuclei showed a slight concavity, with heterochromatin aggregating in small patches and a clear nuclear membrane. Mitochondria were swollen, with intact membranes and slight dissolution of the matrix and cristae. The Golgi apparatus was not noticeably hypertrophied, and intercellular bridges were visible, with some local intercellular spaces slightly widened (Fig. 3D).
Previous experimental studies have established the pivotal role of
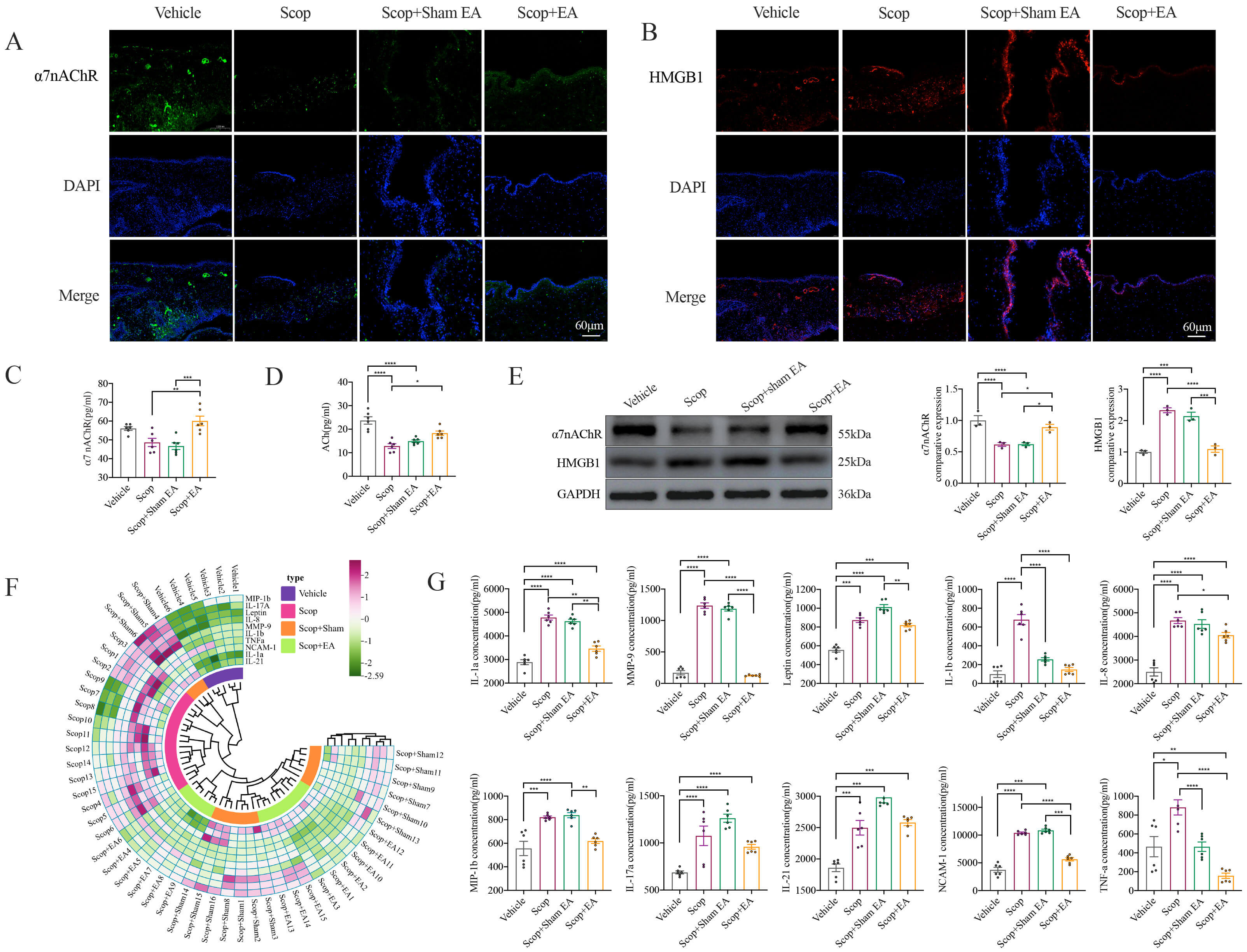 Fig. 4.
Fig. 4.
EA stimulation suppresses Scop-induced conjunctival inflammation
and activates
The conjunctiva represents the most metabolically active region of the ocular
surface, serving as a critical interface for the immune response to a wide array
of exogenous insults, including allergens, pathogens, and toxins. This mucosal
epithelium forms the first line of defense in the ocular surface’s innate and
adaptive immune systems [26]. To elucidate the mechanism by which EA stimulation
modulates inflammatory responses, we employed an antibody array to quantify the
expression of a panel of 10 cytokines and chemokines. Compared to the Vehicle
control group, the Scop-treated conjunctiva displayed pronounced upregulation of
IL-1
EA stimulation exerts its anti-inflammatory effects by stimulating the
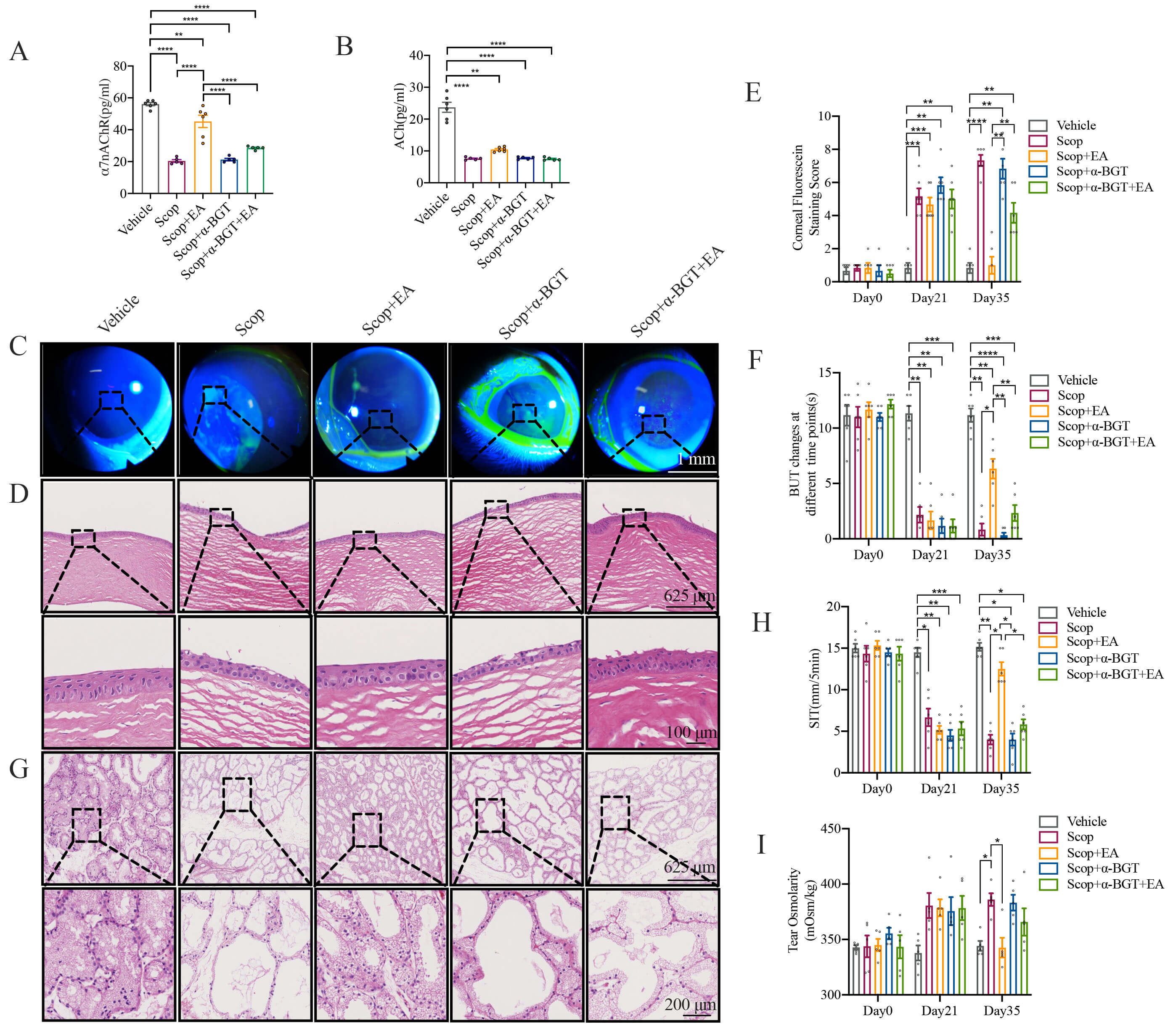 Fig. 5.
Fig. 5.
EA stimulation mediates its anti-inflammatory effects through
To elucidate the potential cholinergic anti-inflammatory mechanism underlying EA
stimulation, we examined whether the expression of HMGB1 and related
pro-inflammatory cytokines and chemokines was inhibited following
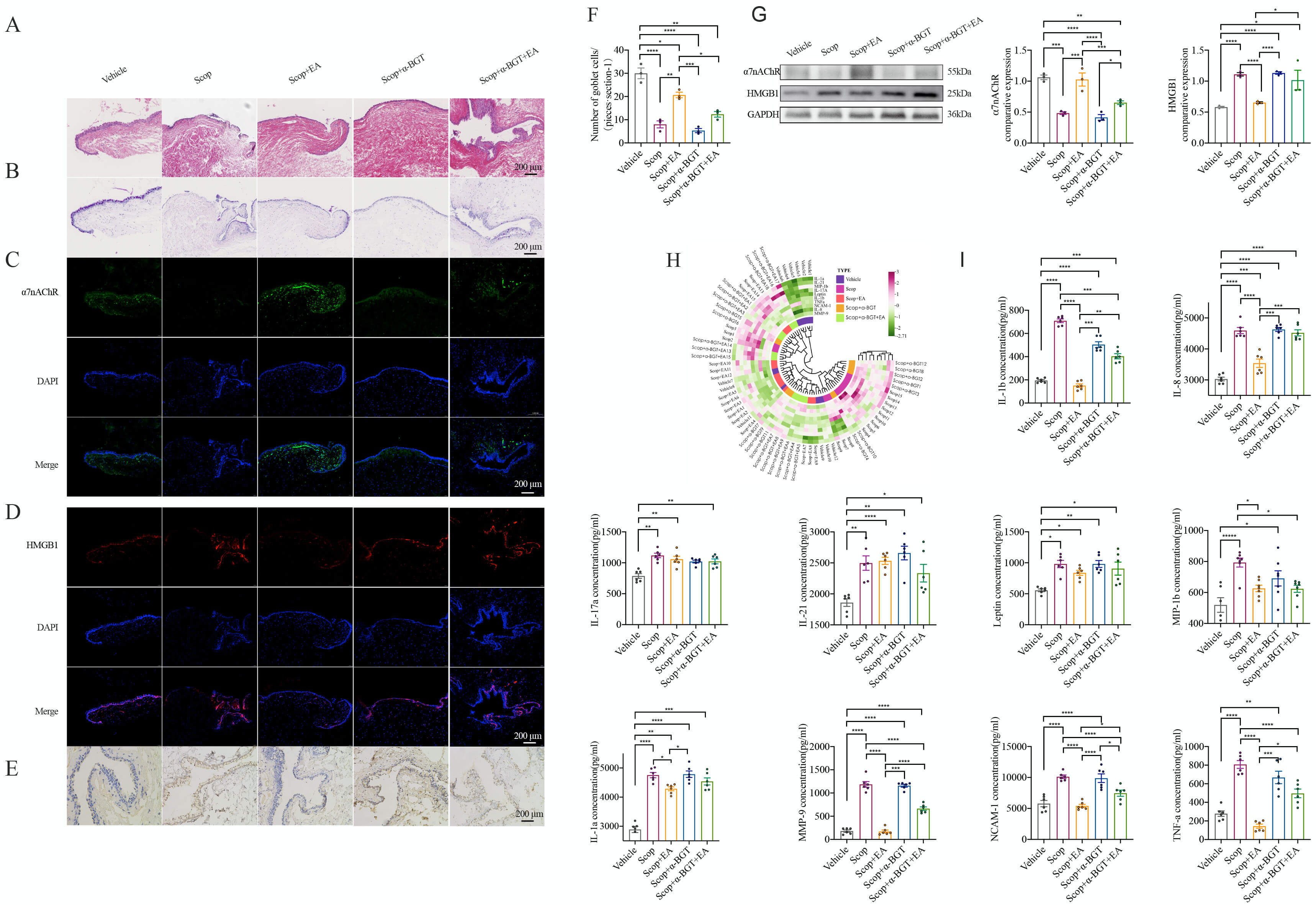 Fig. 6.
Fig. 6.
EA stimulation engages the
This study investigated the ocular surface protective effects of EA stimulation
and its potential mechanisms in a Scop-induced dry eye model. Our research
yielded two principal conclusions. First, EA was found to ameliorate ocular
surface functional damage and tissue injury in the cornea, lacrimal glands, and
conjunctiva induced by DED. Second, our data provide compelling evidence that
conjunctival damage in DED is associated with dysregulation of
DED is a multifactorial ocular surface disorder involving conjunctival goblet
cells, lacrimal glands, and meibomian glands. The coordinated function of these
structures is critical for maintaining the electrolyte, water, mucin, and lipid
composition of the tear film; dysfunction of these structures can lead to tear
film instability, hyperosmolarity, and inflammation [27]. Typically, the blinking
reflex refreshes the tear film at a rate of 5–10 times per minute, evenly
distributing the tear film across the ocular surface and promoting the release of
lipids from the meibomian glands. Prolonged interblink intervals or increased
tear evaporation due to environmental factors can result in elevated tear
osmolarity [28]. Elevated tear osmolarity is a hallmark of all forms of DED and
is positively correlated with tear film instability [29] and the severity of DED
[30, 31]. Tear hyperosmolarity interacts with inflammation, activating downstream
inflammatory cascades and exacerbating tear film instability [4, 24, 32]. The
present study demonstrated that Scop-induced DED is associated with significantly
elevated tear osmolarity, concurrent ocular surface functional damage, and tear
film instability. Furthermore, we detected upregulated protein expression of
HMGB1 and its downstream factors, including IL-1
According to traditional Chinese medicine theory, periocular acupuncture is
commonly used to treat various head and ocular conditions. Modern neuroanatomy
shows that the acupoints Jingming (BL-1), Cuanzhu (BL-2), Sizhukong (TE-23),
Tongziliao (GB-1), and Taiyang (EX-HN5) are located within the distribution of
the ophthalmic branch of the trigeminal nerve. Stimulation of these acupoints can
trigger excitatory signals in the brain via the trigeminal nerve, potentially
improving visual function [36]. Clinical evidence suggests that acupuncture is
more effective than artificial tears for DED treatment [37, 38]. Zhao et
al. [39] reported that periocular acupuncture modulates the serum levels of
TNF-
The
Additionally,
Several limitations of the current study should be noted. First, we used a
single pharmacologically induced DED model, which cannot fully recapitulate
autoimmune or age-related DED. Future studies should employ additional models
(e.g., benzalkonium chloride- or hormonal deficiency-induced DED) to validate the
generalizability of our findings. Second, using rabbits as experimental animals
has inherent limitations, particularly in antibody selection and validation for
molecular experiments. For example, RT-PCR was not used to verify results due to
gaps in species-specific gene databases—notably the absence of the rabbit
This study elucidated the effects and mechanisms of EA stimulation in
Scop-induced DED. The results showed that EA promotes corneal epithelial repair,
increases tear secretion, enhances goblet cell counts, and improves tear film
stability. Importantly,
The data presented in this study are available on request from the corresponding author.
ND and JZ contributed equally to this work as co–first authors. QBW and YCW conceived the study and designed the research protocol. ND and JZ drafted the manuscript. ND and JZ participated in the refinement of the research design (e.g., optimization of data presentation framework and experimental result interpretation logic) and drafted the manuscript. Data acquisition and analysis were performed by SJL, DDZ, XW, and MTH. Immunohistochemistry and immunofluorescence experiments were conducted by LZG and YFW. HXS and TYJ provided help and advice on the ELISA experiments. Figures and visualization were prepared by ND, JZ, and SJL. QBW and YCW critically revised the manuscript for important intellectual content. Figures and visualization were prepared by ND, JZ, and SJL, with ND and JZ involved in the analysis of experimental data presented in the figures to ensure accuracy and consistency with the research hypothesis. QBW and YCW supervised the project and approved the final version. All authors made substantial contributions to the study, reviewed the manuscript, and approved the final version. All authors contributed to editorial changes in the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal procedures were performed following the 3R principles and approved by the Ethics Committee of Nanjing University of Chinese Medicine (NO: 201809A018).
We thank Ejear English Editing Service for providing linguistic editing and proofreading services for this manuscript.
This work was supported by the Youth Program of the National Natural Science Foundation of China (No.82305377, 82205259), National Natural Science Foundation of jiangsu (No.BK20230455), the Foundation Project of Jiangsu Provincial Association of Traditional Chinese Medicine (CYTF2024002, CYTF2024001), Jiangsu Health Vocational College School-level Scientific Research Project (YIXT-YZ202401).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






