1 Department of Laboratory Medicine, The Affiliated Guangdong Second Provincial General Hospital of Jinan University, 510317 Guangzhou, Guangdong, China
2 The Fourth Clinical Medical College, Jinan University, 510317 Guangzhou, Guangdong, China
3 The Second Clinical Medical College, Guangdong Medical University, 510317 Guangzhou, Guangdong, China
4 The Second School of Clinical Medicine, Southern Medical University, 510317 Guangzhou, Guangdong, China
†These authors contributed equally.
Abstract
Liver fibrosis, the end-stage pathological state of many liver diseases, is primarily driven by the activation of hepatic stellate cells (HSCs) and collagen deposition resulting from various pathogenic causes. Thrombospondin-2 (THBS2), a secreted extracellular matrix glycoprotein encoded by the TSP gene family, has been found to activate the TLR4-transforming growth factor-β (TGF-β)/FAK signaling axis and HSCs through autocrine signalling, thereby contributing to the development of liver fibrosis. Latexin (LXN), the only known zinc-dependent metallocarboxypeptidase inhibitor in humans, has not yet been studied for its role in liver fibrosis is yet to be studied.
In this study, we used adeno-associated virus 9 (AAV9) to generate a mouse model of liver fibrosis with LXN knockdown and used siLXN to knock down the LXN gene in the human hepatic stellate cell line LX-2. The mechanisms underlying the association between LXN and hepatic fibrosis progression were investigated using quantitative polymerase chain reaction, western blot, immunohistochemistry, and immunofluorescence staining.
LXN knockdown reduced carbon tetrachloride (CCl4)-induced liver injury and suppressed activation of hepatic stellate cells, while also inhibiting the expression of α-SMA and collagen I. Furthermore, LXN demonstrates a substantial positive correlation with THBS2, and LXN knockdown was capable of downregulating THBS2.
The LXN-THBS2 signaling axis may promote liver fibrosis progression by inducing the activation of HSCs.
Keywords
- cirrhosis
- liver fibrosis
- hepatic stellate cells
- THBS2
- LXN
Liver fibrosis is the primary pathological process underlying the development of
chronic liver disease worldwide, and it is primarily caused by various hepatitis
viruses (such as HBV and HCV), metabolic dysfunction-associated steatohepatitis
(MASLD, formerly NAFLD), alcoholic liver disease (ALD), and other conditions [1].
This condition is marked by the build-up of a substance called the ‘extracellular
matrix’ (ECM) [2], ultimately leading to cirrhosis, liver failure, and
hepatocellular carcinoma (HCC). The primary source of the ECM is a ctivated
hepatic stellate cells (HSCs), which play a pivotal role in the development and
progression of liver fibrosis [3, 4]. Transforming growth factor-
Latexin (LXN) is the sole endogenous inhibitor of carboxypeptidase A in humans. It is vital for protein turnover and transport, maintaining the number of hematopoietic stem cells, inflammation, and tumors [8, 9]. There are reports that LXN is a novel regulator of endothelial cell (EC) morphology maintenance, and LXN knockout can markedly improves vascular permeability, vasodilation, and atherosclerosis in mice [10]. Additionally, LXN is a critical positive modulator of adipocyte differentiation. Mice lacking LXN show resistance to obesity, glucose tolerance, insulin tolerance, and hepatic steatosis induced by high-fat diet (HFD) [7, 11]. LXN expression is associated with joint cartilage mineralization during the progression of traumatic osteoarthritis in rats [12]. In a mouse model of DSS-induced colitis, LXN deficiency resulted in more severe colitis. Recent studies have demonstrated that a deficiency in LXN expression is negatively correlated with the prognosis of solid tumors, including prostate cancer, pancreatic cancer, melanoma, thyroid cancer, and HCC, and that it plays a vital role in tumor cell migration and invasion [13, 14, 15, 16, 17, 18, 19]. However, there are no relevant studies on its role and mechanism in liver fibrosis.
Thrombospondin-2 (THBS2) is a secreted extracellular matrix glycoprotein encoded
by the TSP gene family that plays multiple roles in tissue development,
angiogenesis, immune regulation, and disease progression. In NAFLD patients,
THBS2 is a useful biomarker for the diagnosis of advanced fibrosis [20].
Zhang et al. [21] found that THBS2 can initiate the
TLR4-TGF-
As stated in the Ishak scoring system [22], liver tissue samples were collected from four subjects. Patients with liver fibrosis at stages S2, S3, S3/S4, and S4 at the Second People’s Hospital of Guangdong Province were included. We strictly adhered to ethical guidelines during the acquisition process to ensure informed consent from the patients. Record clinical information, including patient age, gender, aetiology(including viral hepatitis type, alcohol consumption history, and metabolic syndrome-related indicators, etc.), disease course, liver function indicators (alanine aminotransferase (ALT), aspartate aminotransferase (AST), bilirubin, albumin, and globulin, etc.), imaging examination results (liver stiffness values, ultrasound findings, etc.), and treatment history. All experiments involving human tissue were conducted in accordance with the ethical policies and procedures approved by the Medical Ethics Committee of The Affiliated Guangdong Second Provincial General Hospital of Jinan University. The ethical approval number for this study is 2023-KY-SB-37.
24 SPF-grade C57BL/6 J mice, aged 6–8 weeks with a body mass of 19–23 g, were procured from Zhuhai Baishitong Biotechnology Co., Ltd., Guangdong Province, China. A Total of 24 mice were housed in a constant temperature environment (22–23 °C) and were subjected to a standard 12-hour light-dark cycle, with sufficient food and water provided. All animal experiments were conducted in accordance with the ethical guidelines and policies approved by the Laboratory Animal Ethics Committee of The Affiliated Guangdong Second Provincial General Hospital of Jinan University, under approval number 2023-DW-SB-48.
The mice were randomly assigned to four groups with six mice in each group (n = 6): control group (injected with adeno-associated virus 9 (AAV9)-GFP virus), CCl4-treated model group (injected with AAV9-GFP virus), CCl4-treated model group (injected with AAV9-LXN shRNA), and control group (injected with AAV9-LXN shRNA). The model group used a CCl4-induced liver fibrosis model, mixing CCl4 with olive oil at a ratio of 1:3 to 1:5. The mixture was administered via intraperitoneal injection based on mouse body weight (0.1 mL per 5 g of body weight), the control group received 5 µL/g of corn oil, three times per week for a period of eight weeks.
shRNA sequences targeting LXN were designed and synthesized based on the mouse
LXN gene sequence. After annealing to form double strands, the strands were
connected to the AAV9 vector. The constructed vector contained promoters, shRNA
sequences, and poly(A) tails. This section was constructed to Cyagen Biosciences.
The constructed AAV9-LXN shRNA vector and the auxiliary plasmid were
co-transfected into HEK293 cells. Following cell culture and virus packaging
processes, cell culture supernatants were collected and purified by
ultrafiltration and ultracentrifugation to obtain the AAV9-LXN shRNA virus. After
determining the virus titer, the virus was stored at –80 °C. Starting from week
3, AAV9-GFP virus and AAV9-LXN shRNA virus (at a dose of 1
The blood collected from mice was centrifuged at 3000 rpm at room temperature for 15 minutes, the supernatant was collected and stored at –80 °C for subsequent experimental analysis. Serum ALT, AST, alkaline phosphatase (ALP), albumin (ALB), total bilirubin (TBIL), total bile acids (TBA), cholesterol (CHO), and triglycerides (TG) levels were measured using an automatic biochemical analyzer (Hitachi 7020, Japan).
After removing the liver tissue, a portion was treated with 4% formaldehyde for 36 hours, then embedded in paraffin and sectioned (4–6 µm thick). The morphology of hepatocytes and the presence of inflammatory cell infiltration were observed using HE staining, and the severity of liver fibrosis was assessed using Sirius red staining.
For immunohistochemical staining of Alpha-Smooth Muscle Actin (
An Animal Tissue Total RNA Extraction Kit (PK10021, Proteintech) was used to extract total RNA from the liver tissue. A spectrophotometer was used to detect the RNA concentration and purity.
The accepted range for the A260/A280 ratio is 1.8–2.0. The reverse
transcription process was executed by means of a reverse transcription kit
(B639252-0100, Sangon Biotech), and RT-qPCR was conducted using the TB
Green® Premix Ex TaqTM II kit (RR420A, TAKARA) on an ABI7500 PCR
instrument. We used
| Gene | Forward primer | Reverse primer |
| TGTCCACCTTCCAGCAGATGT | AGCTCAGTAACAGTCCGCCTAGA | |
| COL1 | GAGGGCCAAGACGAAGACATC | CAGATCACGTCATCGCACAAC |
| TCGGATACTTCAGCGTCA | GGGAGTAATGGTTGGAATG | |
| LXN | GCGGTTATGTAATGTGGCAG | GTGGTAAGATGAGGTGCTGTAA |
COL1, collagen I;
LX-2 cells were procured from the Shanghai Institute of Cell Biology, Chinese
Academy of Sciences. All cell lines used in this study were authenticated by
short tandem repeat (STR) profiling and tested negative for mycoplasma
contamination. After thawing from liquid nitrogen, the cells were resuspended in
DMEM (Gibco, USA) supplemented with 10% fetal bovine serum (Gibco, USA) and 100
U/mL penicillin and streptomycin (Solarbio, Guangzhou, China). Cells were then
maintained in a 5% CO2 incubator for subsequent experiments. The cells were then
cultured in six-well plates and grouped as follows: (1) Control group, cultured
under standard conditions. (2) TGF-
| Primer name | Sequence (5′-3′) | Length | Tm °C |
| LXN-siRNA1 | CCAGAAGUCAACUUCACAUUUTT | 23 | 50.2 |
| AAAUGUGAAGUUGACUUCUGGTT | 23 | 50.2 | |
| LXN-siRNA2 | GAAGUCAACUUCACAUUUGAATT | 23 | 48.4 |
| UUCAAAUGUGAAGUUGACUUCTT | 23 | 48.4 | |
| LXN-siRNA3 | CAUCCACAAUACGGCACUAAATT | 23 | 51.9 |
| UUUAGUGCCGUAUUGUGGAUGTT | 23 | 51.9 | |
| NC-siRNA | UUCUCCGAACGUGUCACGUTT | 21 | 52.4 |
| ACGUGACACGUUCGGAGAATT | 21 | 52.4 |
For liver tissue paraffin sections, fixation with 10% neutral buffered formalin, boil in sodium citrate buffer for 15 minutes, and incubation at room temperature for one hour after blocking with 5% bovine serum albumin (BSA), followed by incubation with the appropriate primary antibody at 37 °C overnight. After thorough washing with PBS, sections were incubated with FITC-labelled goat anti-rabbit IgG at 20 °C for 1 h. Then, the sections were counterstained with 4,6-diamidino-2-phenylindole (DAPI), washed, and mounted using an anti-fluorescence quenching mounting medium (Prolong® Gold). The cells were observed under a fluorescent microscope (OLYMPUS IX83, Tokyo, Japan). The following antibodies were utilised for immunofluorescence staining: anti-LXN (1:3500; Bioss, bs-1971R) and anti-THBS2 (1:2000; CUSABIO, P35442).
Total protein was extracted from the liver tissue using a dedicated kit for
animal tissues. Lysate proteins from LX-2 cells were prepared using 1:100 PMSF
and RIPA buffer, followed by SDS-PAGE to separate 30 micrograms of protein and
polyvinylidene difluoride (PVDF) membrane was used to transfer it. For 1 hour at
20 °C, the membrane was blocked with 5% bovine serum albumin (BSA), the primary
antibody was incubated with the PVDF membrane at 4 °C overnight, then the PVDF
membrane was thoroughly washed with TBST, and incubated with goat anti-rabbit
secondary antibody (IgG) (ZK0623) at 20 °C for 70 minutes, followed by ECL
chemiluminescence imaging to detect the bands. For the purpose of visual analysis
of proteins, ImageJ (NIH, Bethesda, MD, USA) was employed. The primary antibodies
utilized for western blotting are listed below: anti-LXN antibody (1:300, Bioss,
bs-1971R), anti-
The statistical analyses were conducted using the GraphPad Prism v8.00 software (La Jolla, CA, USA). The experimental data were then compared using Student’s t-test or analysis of variance. The statistical significance of the results was determined by applying a p-value cutoff of 0.05.
We obtained three sets of transcriptome data related to human and mouse liver
fibrosis from the GEO database (GSE25583, GSE55747, and GSE84044), from which we
screened for differentially expressed genes (
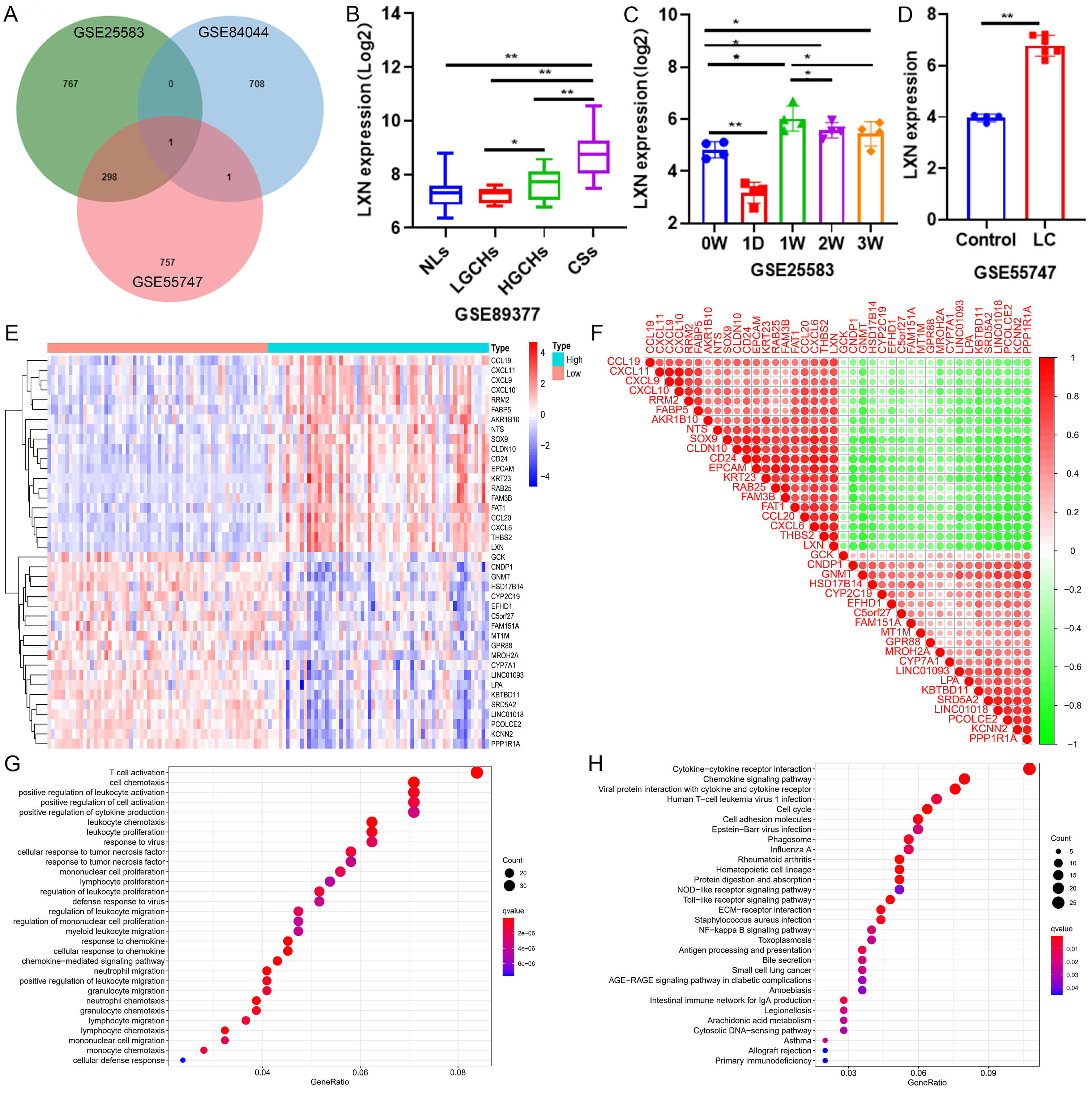 Fig. 1.
Fig. 1.
Latexin (LXN) is upregulated in liver fibrosis and highly
correlated with Thrombospondin-2 (THBS2). (A) The Venn diagram illustrates the
correlations among differentially expressed genes across three liver fibrosis
datasets. (B) LXN expression in four distinct pathological states within the
human liver tissue dataset. NLs, Normal liver tissue; LGCHs, Low-grade chronic
hepatitis; HGCHs, High-grade chronic hepatitis; CSs, Cirrhosis. * p
After analyzing the expression profiles of 124 patients with different stages of liver fibrosis (GSE84044), it was established that there was a gradual increase in LXN expression concomitant with the progression of inflammatory activity grading (G0–G4) and fibrosis staging (S0–S4) (Fig. 2A,B). To validate this result, we collected liver tissue samples from four patients with liver fibrosis stages S2, S3, S3/S4, and S4, and performed HE staining, Sirius red staining, and LXN immunohistochemical staining. HE staining revealed that as the stage of liver fibrosis increased, the portal areas gradually expanded, forming fibrous septa, and the hepatic lobule structure became disordered with increased inflammatory cell infiltration. Sirius Red staining showed that collagen deposition gradually increased and fibrous septa formed as liver fibrosis progressed. The immunohistochemical staining results for LXN were consistent with our expectations, with LXN expression increasing as the stage of liver fibrosis progressed (Fig. 2C, Supplementary Fig. 1A,B).
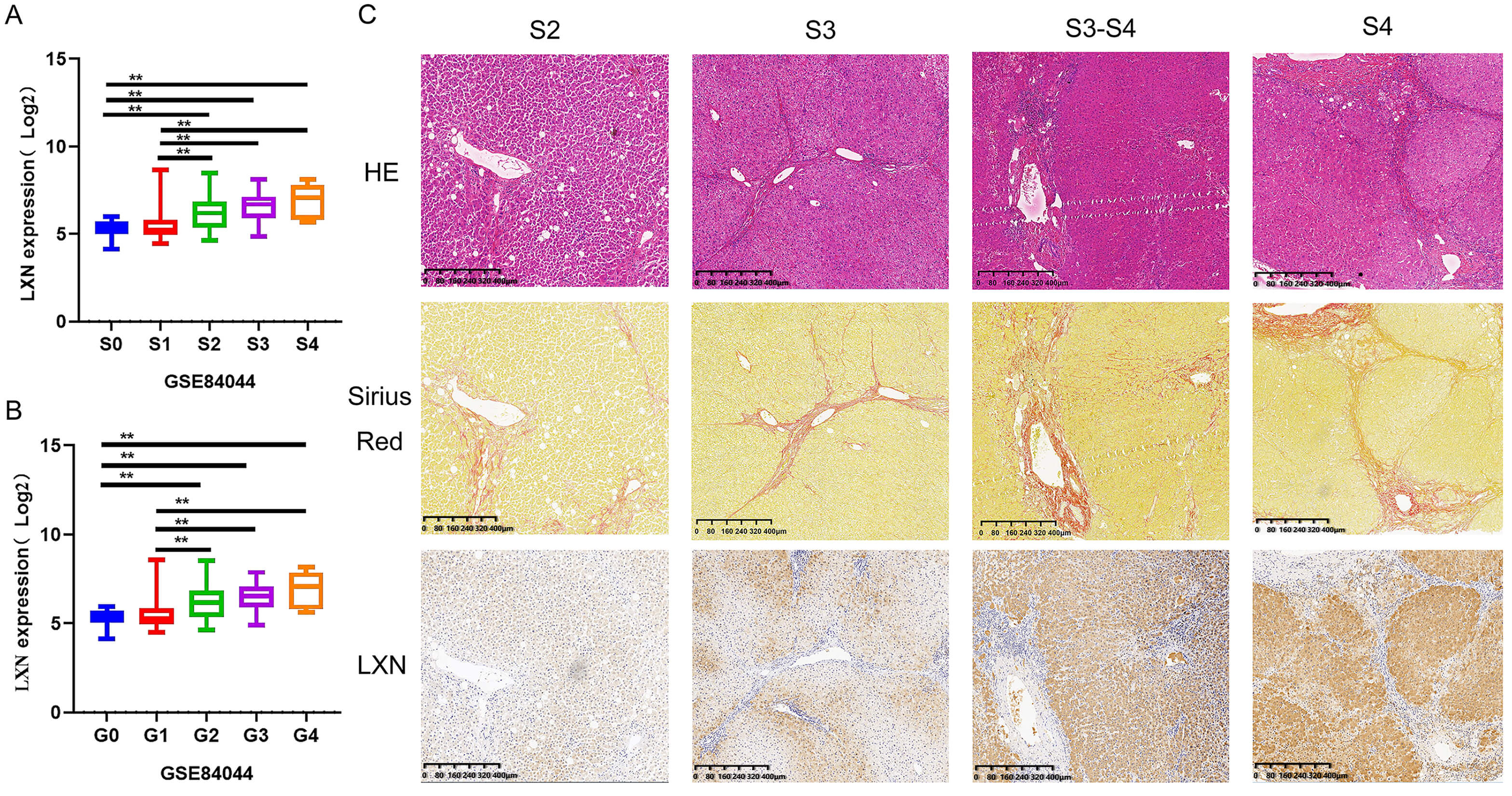 Fig. 2.
Fig. 2.
Expression of LXN in human liver tissue increases with the
progression of liver fibrosis. All experiments were performed in triplicate (n =
3). (A,B) In a dataset comprising 124 patients with distinct stages of hepatic
fibrosis, the expression of LXN correlates with both the grading and staging of
fibrosis. ** p
This study used a CCl4-induced liver fibrosis model in which mice received intraperitoneal injections of CCl4 for eight weeks. To determine whether LXN knockout could alleviate CCl4-induced liver fibrosis in mice, AAV9-LXN-shRNA or control (GFP) was injected into C57BL/6 mice starting at week 3 (Fig. 3A). After the experiment, the mice were euthanised and whole blood and liver tissue were collected. The body weight and ALT, AST, ALP, ALB, TBIL, TBA, CHO, and TG levels of the mice were measured (Fig. 3B–J). The results showed that murine subjects in the CCl4-modelling group exhibited a reduced mean body mass compared with the control group, and indicators such as TBIL, TBA, ALT, AST and ALP were all elevated, indicating that CCl4 treatment damaged the liver function of mice. Following LXN knockdown using AAV9-LXN-shRNA, the levels of TBIL, TBA, ALT, AST, and ALP were reduced compared to the model group. TBA is a sensitive marker of liver damage and can directly participate in the fibrotic process through mechanisms such as HSC activation and oxidative stress. This finding suggests that suppression of LXN may lead to a reduction in HSC activation and oxidative stress, consequently alleviating CCl4-induced liver injury and mitigating liver fibrosis.
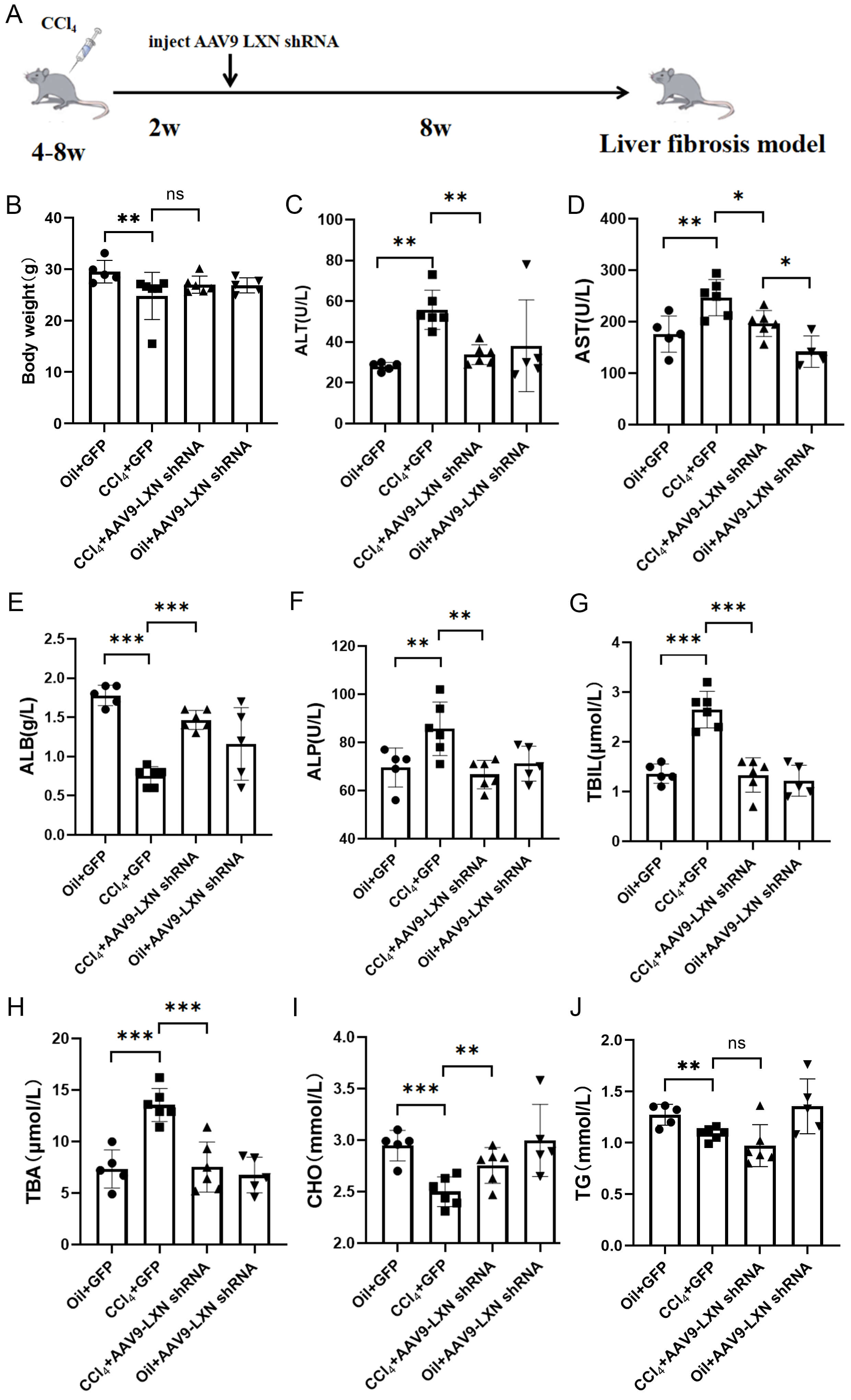 Fig. 3.
Fig. 3.
Weight and blood biochemical parameters in the four groups of
mice. (A) Schematic diagram of the treatment protocol for the CCl4-induced
adeno-associated virus 9 (AAV9)-LXN-shRNA mouse model. (B–J) Body weight and
Serum Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST), alkaline
phosphatase (ALP), albumin (ALB), total bilirubin (TBIL), total bile acids (TBA),
cholesterol (CHO), and triglycerides (TG) levels in the four mouse groups. * p
Subsequently, the present study investigated the potential of AAV9-LXN shRNA to
reduce liver fibrosis and protect the liver from the harmful effects of
CCl4. Photographs of the isolated livers were taken, and HE and Sirius red
staining were utilised in order to ascertain the histopathological status of the
mouse liver tissue and collagen deposition. Immunohistochemical staining for
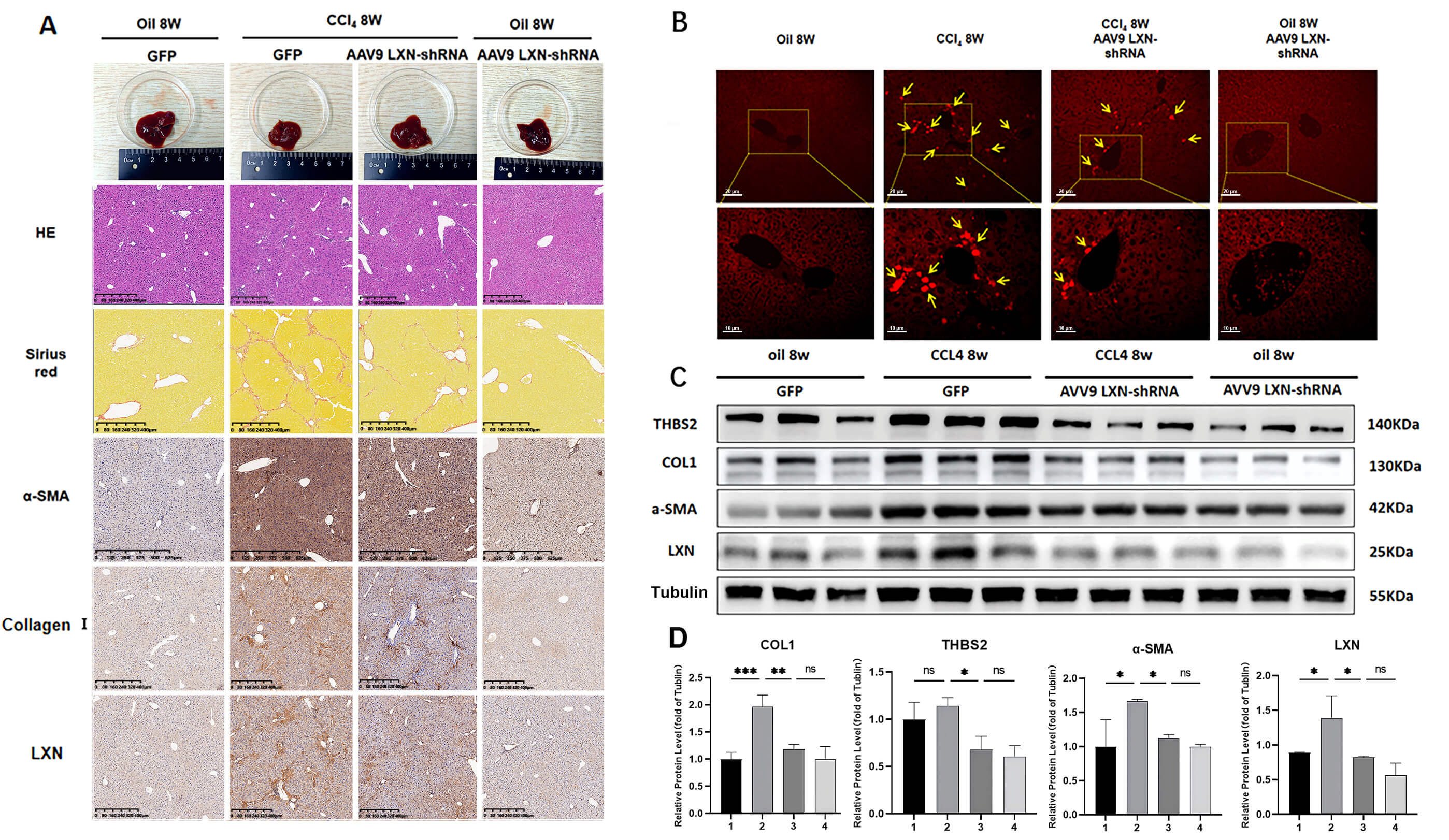 Fig. 4.
Fig. 4.
AAV9-LXN shRNA reverses CCl4-induced liver fibrosis. All
experiments were performed in triplicate (n = 3). (A) Histological examination of
liver tissue from oil 8w, CCl4 8w-induced model, CCl4 8w AAV9 LXN-shRNA,
and oil 8w AAV9 LXN-shRNA-treated mice via hematoxylin and eosin staining,
Sirius red staining, and immunohistochemical staining for
Additionally, RT-qPCR and Western blot analyses further confirmed these results, showing that the CCl4-treated groups exhibited significantly increased LXN expression and markedly elevated HSC activation indices, whereas LXN-silenced mice in the reversal group demonstrated reduced HSC activation indices. Furthermore, the expression of these genes was significantly reduced following LXN knockdown (Supplementary Fig. 2 and Fig. 4C,D). In summary, liver fibrosis upregulates LXN expression, and LXN knockdown can reverse and regress liver fibrosis.
The LX-2 cells were subjected to a treatment with TGF-
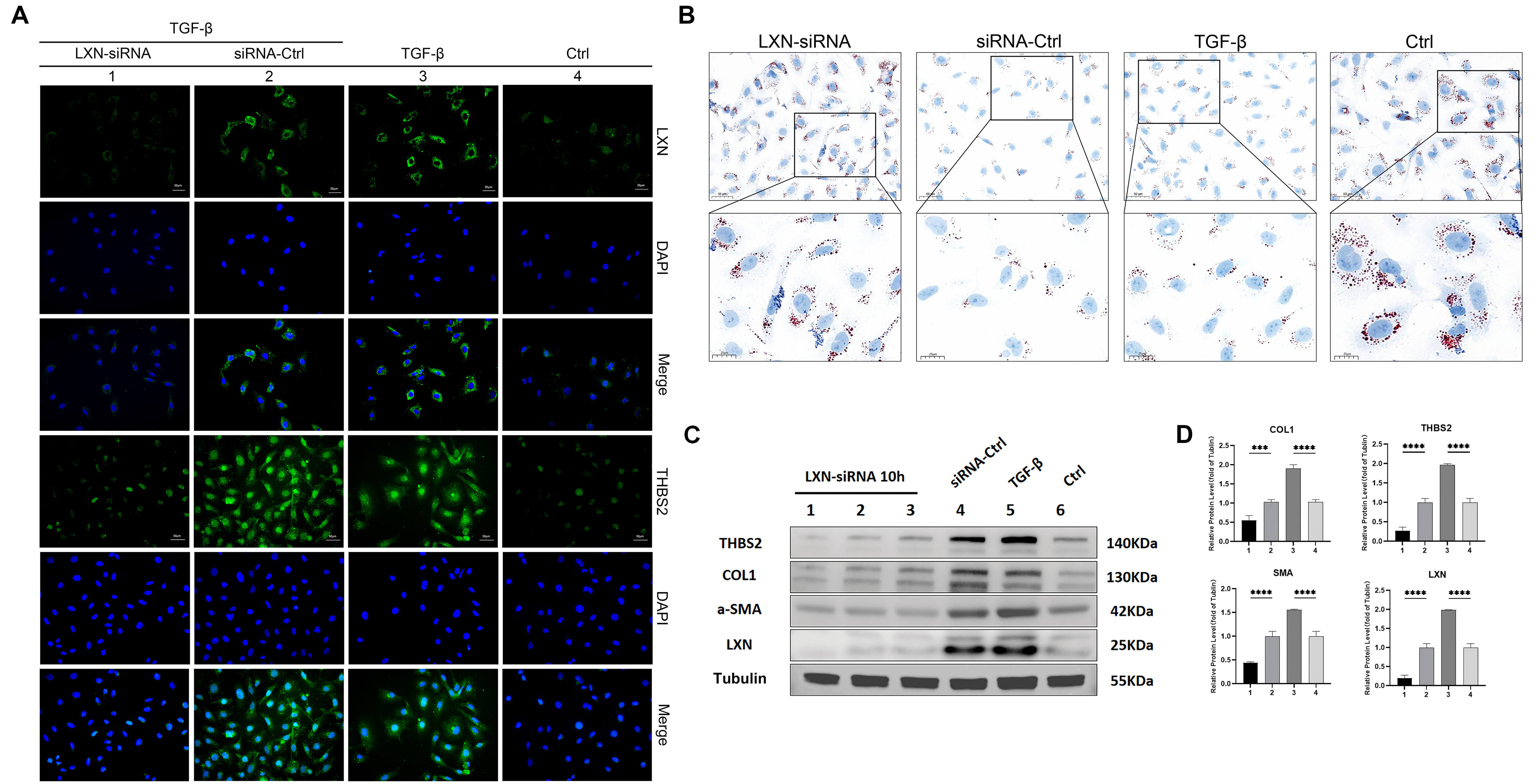 Fig. 5.
Fig. 5.
LXN siRNA interference reduces the activation of hepatic
stellate cells (HSCs). All experiments were performed in triplicate (n = 3). (A)
Immunofluorescence staining for LXN and THBS2 in cells from the control group,
transforming growth factor-
Single-cell sequencing data of liver tissues during hepatic fibrosis, retrieved from the GEO database (GSE174748), was further analyzed. Unsupervised clustering of the qualified single-cell dataset identified 10 distinct cell clusters (Supplementary Fig. 3A). Subsequently, a panel of classical HSC marker genes (DCN, HGF, ACTA2, and ADAMTSL2) was utilized to validate and annotate each cell cluster (Supplementary Fig. 3B). Via this marker gene-mediated identification strategy, the HSC cluster was successfully localized and defined within the heterogeneous cellular population of liver tissues (Supplementary Fig. 3C). Further differential expression analysis demonstrated that the LXN gene was specifically highly expressed in HSCs, with negligible expression detected in other cell clusters (Supplementary Fig. 3D).
Excessive ECM deposition and HSC activation in liver tissue are significant factors in fibrosis and profibrotic cytokine formation [23, 24]. Liver fibrosis is reversible, unlike other serious liver conditions such as cirrhosis and HCC. Liver fibrosis can be reversed or eliminated by inhibiting HSC activation through certain pathways [25].
Using the Gene Expression Omnibus (GEO) database, we found that LXN is
upregulated in advanced liver fibrosis, with increased expression as the stage
progresses, and increased LXN expression in primary HSCs, suggesting that it is
closely related to liver fibrosis. Current research indicates that LXN plays a
role in protein turnover, maintaining hematopoietic stem cell numbers,
inflammation, and tumorigenesis. However, its role and underlying mechanisms in
liver fibrosis remain unclear. CCl4-induced liver fibrosis in animals is
widely used to simulate the pathogenesis of liver fibrosis in humans [26].
CCl4 induces liver fibrosis in C57BL/6 mice. AAV9-LXN shRNA was used for LXN
knockdown. By measuring the levels of liver injury markers, such as ALT, AST, and
TBA, we found that LXN knockdown reduced CCl4-induced liver injury.
Additionally, the results demonstrated inhibition of
At the same time, analysis of the GEO database revealed a positive correlation
between LXN and THBS2. Previous studies have reported that LXN regulates THBS1 to
form a signaling cascade that plays a pivotal role in regulating the function of
hematopoietic stem cells, stress responses, and cancer development under
steady-state conditions [30, 31]. THBS2 has been shown to activate the
TLR4-TGF-
Nevertheless, this study had some limitations. For example, it is unclear how
LXN regulates THBS2 expression. Research has identified that LXN regulates THBS1
transcription through the Rps3-NF-
Moreover, only knockdown experiments were conducted for LXN, omitting the need for overexpression experiments. Further studies are needed to determine how increased LXN expression causes liver fibrosis. Further investigation is necessary to elucidate the mechanism by which THBS2 activates HSCs and to determine whether other signaling pathways are involved in this process.
In summary, this study demonstrates that knocking out LXN downregulates THBS2
expression and reverses CCl4-induced mouse liver fibrosis and
TGF-
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
HYW, HYJ, and YCY devised the research plan; DLC provided academic guidance and technical support for this research. HYJ, SYW conducted data analysis, HYW, HYJ, SYW, XRL and XT conducted experimental research, XT, YCY, DLC provided advice for the research, and HYW authored the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study has been approved by the Institutional Review Board (IRB) of The Affiliated Guangdong Second Provincial General Hospital of Jinan University, approval number [2023-KY-SB-37]. All participants were provided with information regarding the study and gave their written informed consent prior to participation. This study was conducted in compliance with the Declaration of Helsinki and all applicable ethical guidelines. The animal study protocol was approved by the Animal Care and Use Committee (ACUC) of The Affiliated Guangdong Second Provincial General Hospital of Jinan University, protocol number [2023-DW-SB-48]. The study adhered to the guidelines set by the committee.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL46251.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





