1 Goethe University Frankfurt, University Hospital, Institute of Neuroradiology, D-60528 Frankfurt am Main, Germany
Abstract
The pathogenesis of multiple sclerosis (MS) is a complex topic that involves many individual aspects, several MS risk factors and various alterations in the immune system. In addition to the aspects still being researched in MS today, such as the activation of pro-inflammatory lymphocytes and macrophages, a particular scientific interest refers to the dysregulation of iron and to ferroptosis as an important form of programmed cell death (PCD), which is closely associated with this dysregulation. These two aspects are particularly significant because the new McDonald criteria include the diagnostic determination of so-called “paramagnetic rim lesions” (PRLs) in MS, which are areas of abnormal iron accumulation visible on magnetic resonance imaging (MRI) scans. Along with other diagnostic criteria, these PRLs are an important criterion for the diagnosis of MS. This review begins with an overall description of pathogenic risk factors and the dysregulation of the immune system. However, the priority of the review is the dysregulation of the cerebral iron balance and ferroptosis, as well as other forms of programmed cell death, which have also been demonstrated in MS: apoptosis, pyroptosis, necroptosis and NETosis. One of the main current findings on the forms of PCD in MS is their non-physiological occurrence in the brain’s own cells such as oligodendrocytes and neurons, and the extensive protection of pro-inflammatory cells from the occurrence of PCD. Emerging evidence indicates that ferroptosis occurs at an early stage in MS and appears to play a role in its pathogenesis. This review will present the distinct characteristics of the various PCDs in MS, alongside other pathological mechanisms, genetic factors, and current experimental approaches of therapeutic concepts that affect especially ferroptosis and pyroptosis. The aim is to provide an overview on iron dysregulation and forms of PCD as key aspects for understanding the pathogenesis of MS.
Keywords
- multiple sclerosis
- immunology
- iron metabolism
- ferroptosis
- pyroptosis
- apoptosis
- necroptosis
- NETosis
The etiopathogenesis of multiple sclerosis (MS) has been the subject of uninterrupted and intensive research since the 19th century. After MS was first described independently by Robert Carswell and Jean Cruveilhier in the years 1838 to 1841 [1], it was subsequently regarded as a multifactorial autoimmune disease. Despite recent advances, the pathogenesis of MS remains incompletely understood. The currently increasing prevalence of MS worldwide is an additional factor that emphasizes the need for continued research into all known pathophysiological factors. Based on a comparison of epidemiological data from 81 countries from the years 2013 and 2020, a marked increase in the global prevalence of MS from 29.26 to 43.95 cases per 100,000 people was observed between 2013 and 2020. This increase in prevalence affects all regions of the world [2, 3].
In addition to the long-known risk factors such as vitamin D deficiency or genetic profiles, as well as pathophysiological alterations in the immune system, the dysregulation of the iron balance has currently become a major focus of MS research. Ferroptosis, a type of cell death associated with iron imbalance and first identified by Dixon et al. in 2012 [4], is also relevant here. Therefore, a review of ferroptosis in MS is needed, but should be preceded by an overview of MS risk factors and pathophysiology to provide context. This review also describes other forms of cell death, namely apoptosis and autophagic cell death. Although known for a long time, the role of apoptosis, autophagy and autophagic cell death in MS is still not fully understood. Pyroptosis, first described in 2001 by Cookson and co-workers [5], is another form of cell death that is also attracting increasing scientific interest in MS, which also applies to other forms of programmed cell death such as necroptosis and NETosis. This review will present and discuss the current knowledge on all these aspects, including genetic factors and current strategies to new therapeutic options that could influence various forms of cell death in MS.
Approximately 2.9 million people worldwide suffer from MS, the prevalence is particularly high in the countries of North America and Europe, but lower in Latin America, Africa, East Asia and generally in countries near the equator. Statistically, the prevalence in the various countries correlates with the distance from the equator to the north and south, whereby the reduced endogenous vitamin D production as a result of less sunlight exposure is considered to be an important factor [2, 6]. Vitamin D is involved in neuroprotection through various signaling pathways, such as the stimulation of anti-inflammatory T regulatory cells (Tregs), the suppression of pro-inflammatory Th17 cells and M1 microglia, the promotion of myelin and neuronal acid synthesis in oligodendrocytes, and the downregulation of the human leukocyte antigen (HLA) risk allele HLA-DRB1*15:01. The totality of these effects of vitamin D also explains the increased risk of developing MS as a result of vitamin D deficiency [7, 8]. In addition, over 200 genetic risk factors for MS, mostly specific alleles or single-nucleotide polymorphisms, have been identified and mainly influence immune cell pathways. Despite the sex-specific ratio of 3:1 between women and men, the role of sex hormones and the importance of the X chromosome in the development of MS is controversially discussed [9, 10, 11]. An important indication of the significance of genetic factors is an increased risk of disease of 20–30% in identical twins of MS patients and 2–5% in fraternal twins [12]. The risk alleles of the HLA-II genes, such as HLA-DRB1*15:01, which have been known for decades, are important examples of MS risk factors. There is now a consensus that not the HLA genes and their alleles alone, but only a polygenic risk score, taking into account numerous other genes, shows a significant correlation with the risk of MS [13, 14, 15]. Important examples are risk alleles of the gene for the vitamin D receptor, as well as risk alleles of genes that are directly associated with the regulation of immune cells, such as the forkhead box P3 (FOXP3) gene, which is an important marker of T-regulatory cells [16, 17]. Epigenetic changes such as aberrant methylation status of HLA genes and aberrant expression of several micro RNAs (miRNAs) are also discussed as risk factors and biomarkers of MS [18, 19]. Current evidence suggests that considering both genetic and environmental or lifestyle factors provides a better correlation with MS risk than genetics alone. One example is the increased risk of MS in smokers in combination with the presence of HLA risk genes [20]. However, even without considering genetic findings, nicotine consumption, lack of physical activity and obesity are considered risk factors for MS [21, 22]. Dysbiosis of the bacterial intestinal flora is also a significant etiological factor due to the detection of a reduction in anti-inflammatory species such as bacteroides or parabacteroides, as well as an increased presence of pro-inflammatory species such as Akkermansia in MS patients. The detection of reduced immune globulin A (IgA) coating of individual bacterial species is also considered pathogenetically significant. The authors emphasize the need for further research to better understand the range of species with pro- and anti-inflammatory effects, as well as the underlying pathomechanisms [23, 24, 25]. Infection with the Epstein-Barr virus (EBV), which is seropositive in the cerebrospinal fluid (CSF) of almost 100% of all MS patients, is considered one of the most important environmental factors. A variety of signaling pathways have been described for the pathological influence of EBV on the immune system in MS. A major influence of the EBV is the immortalization of infected B-cells by means of the so-called latent member protein 1 (LMP1), as well as the induction of continued antigen presentation by the B-cells with production of cytotoxic autoantibodies and cytokines [9, 26, 27, 28]. A causative association with MS is also discussed for infections with other virus species such as human herpesvirus-6 (HHV-6) and human endogenous retroviruses (HERVs). In contrast to EBV, not all MS patients are seropositive for these viruses [29, 30, 31].
The cumulative influence of all the risk factors mentioned above promotes the
peripheral development of CD4+ Th1 and Th17 T cells, which increasingly express
pro-inflammatory cytokines such as interferon-
Macrophages are generally present in MS lesions and are recruited from
peripheral monocyte-derived macrophages, which infiltrate the central nervous
system (CNS) at an early stage of MS, as well as from resident mononuclear
phagocytes of the CNS, which include microglia and CNS border-associated
macrophages (BAMs). These BAMs are prominently localized perivascularly, in the
meninges, and in the area of the choroid plexus. Before infiltrating the CNS, the
peripheral macrophages together with dendritic T cells are involved in the
presentation of myelin-specific antigens to the immunoreactive Th1 and Th17 cells
that subsequently infiltrate the CNS. These T cells are also stimulated by the
expression of IL-1
Evidence of iron dysregulation and regional redistribution in MS has primarily
been obtained through 3T or 7T brain magnetic resonance imaging (MRI) scans,
including echo-planar diffusion-weighted (DWI) and multi-planar
susceptibility-weighted (SWI) MR sequences. A key finding is an increased iron
concentration in the deep grey matter in early stages of MS compared to control
collectives, as well as in patients with clinically isolated syndrome (CIS)
[40, 41, 42]. In later stages, an increased iron concentration was found at the edges
of the MS foci with a reduced concentration in the core area of the lesions [37].
The early increase of iron concentration in the deep grey matter has been
reported to correlate with the severity of neurological deterioration, but no
differences were found between healthy individuals and MS patients with regard to
the iron content in the entire cerebrum [40]. Furthermore, a reduced iron content
in the unaffected white matter of MS patients has been reported in comparison
with healthy persons [43]. Oligodendrocytes have the highest iron content of all
CNS cells, as it is an important cofactor in the synthesis and homeostasis of
myelin [44]. Current evidence suggests that inflammation-related damage to myelin
and oligodendrocytes is a main cause of increased local iron levels in MS [45].
Microglia are already found in the early stages of MS lesions and, together with
the peripherally infiltrated macrophages, are significantly involved in the
pro-inflammatory activity and the subsequent uptake of myelin detritus and iron
(Fig. 1, Ref. [7, 9, 14, 19, 21, 23, 26, 30, 33, 34, 37, 39, 45, 46, 47, 48, 49]) [46]. The increased iron accumulation radiologically
detectable in the form of the so-called “paramagnetic rim lesions” at the edge
of MS lesions corresponds to a neuropathologically confirmed accumulation in
activated microglia [50, 51]. This intracellular excess of iron damages in
particular the mitochondrial membranes and the iron-sulphur clusters with
increased release of reduced Fe2+ and the generation of free radicals and
oxidative stress, which further increases the damage to myelin and
oligodendrocytes (Fig. 1) [47]. Furthermore, astrocytes also play a role in the
increased uptake of iron under pathological conditions by increasingly expressing
the receptor for the iron transport protein transferrin in demyelinating
diseases. It is discussed that an increased intracellular iron concentration in
astrocytes could have a pro-inflammatory effect [45, 48, 52]. The significance of
increased iron accumulation in neurons and lymphocytes is also the subject of
current scientific interest. Increased iron in neurons impairs their function, a
central cause appears to be the increased expression of hepcidin by astrocytes
(Fig. 1). Hepcidin is a negative iron regulator that leads to the degradation of
the iron exporter ferroportin1 and thus to increased intracellular iron
accumulation. A role of polymorphisms of the hepcidin gene and other
hepcidin-regulating genes is also being discussed as a possible cause of
this dysregulation [45, 49, 53]. In T and B lymphocytes, higher intracellular
iron levels are thought to promote proliferation and increased production of
pro-inflammatory cytokines such as IL-2, IFN
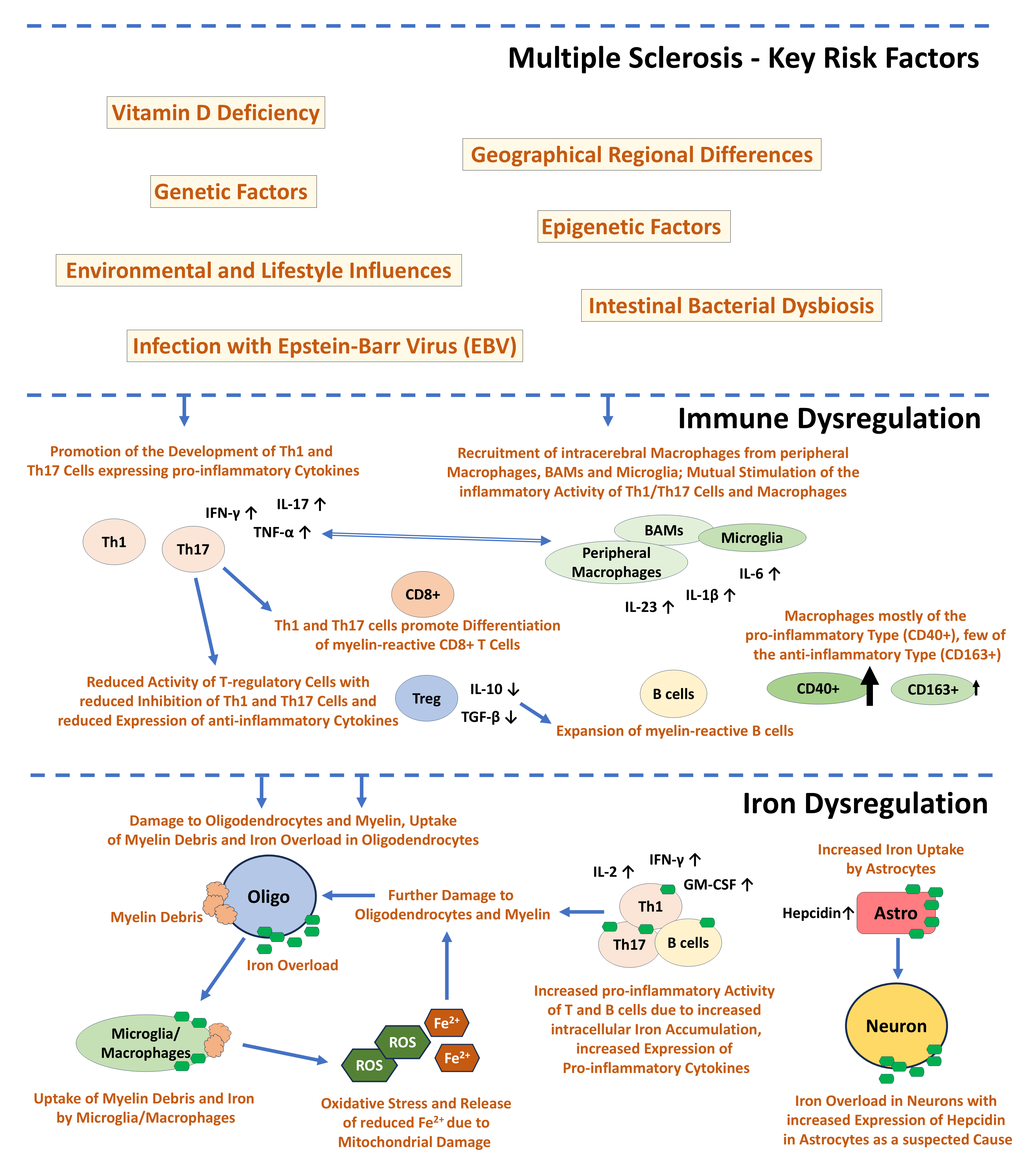 Fig. 1.
Fig. 1.
Multiple Sclerosis - key factors of disease risk [7, 9, 14, 19, 21, 23, 26, 30], immune
dysregulation [9, 33, 34, 37, 39], and iron dysregulation [45, 46, 47, 48, 49] as described in the
text. IFN-
Ferroptosis is a form of programmed cell death first described by Dixon and colleagues in 2012 [4], which differs completely from apoptosis in terms of morphology and pathobiochemistry. In contrast to apoptosis, there is no disintegration of the chromatin and the cell nucleus, but there is damage to the cell membranes and the mitochondria with rupture of their membrane and degeneration of the mitochondrial cristae [55]. It is now known that ferroptosis frequently occurs in MS and plays a role in the development of the disease [56]. A key promoter of ferroptotic cell death is increased iron accumulation in the cytosol with additional reduction of the trivalent iron Fe3+ by the ferric reductase six-transmembrane epithelial antigen of prostate 3 (STEAP3) into the reactive and cytotoxic bivalent iron Fe2+. This reduction can occur intracellularly, but Fe2+ can also be taken up across the cell membrane by the divalent metal transporter 1 (DMT1), and it can occur after degradation of the redox-inactive iron storage protein ferritin by autophagosomes (ferritinophagy) [57]. This excess of divalent iron, also known as the “labile iron pool” (LIP), also triggers the Fenton reaction, in which Fe2+ is oxidized again by hydrogen peroxide, releasing hydroxyl radicals and hydroxide ions (Fig. 2, Ref. [55, 56, 57, 58, 59, 60, 61]) [57, 58]. An additional effect of LIP is the catalysis of the peroxidation of lipids, in particular polyunsaturated fatty acids (PUFAs). This peroxidation occurs mainly through the conversion of free long-chain fatty acids into fatty acyl-CoA esters by acyl-CoA synthetase long-chain family member 4 (ACSL4). The subsequent oxidation by iron leads to the formation of toxic phospholipid hydro-peroxides, which destroy the cell membranes and lead to ferroptosis [56, 59]. The expression of the pro-ferroptotic protein ACSL4 is significantly increased in MS [60]. The triggering of ferroptosis is further promoted by a reduced expression of the cystine/glutamate antiporter system (XcT), which leads to a reduced cellular uptake of cystine, which is involved in the biosynthesis of the antioxidant glutathione (GSH). The reduced availability of cysteine and GSH results in lower levels and activity of the anti-ferroptotic enzyme glutathione peroxidase 4 (GPX4), which reduces the inhibition of lipid oxidation and ultimately leads to ferroptosis (Fig. 2) [61]. Furthermore, ferroptosis in MS is triggered by increased proinflammatory T-cell activity and associated myelin degradation. It also accelerates lipid peroxidation and inflammation by releasing danger-associated molecular patterns (DAMPs), which further activate inflammatory T cells (Fig. 2) [55, 57]. The release of DAMPs is accompanied by an increase in by-products of lipid peroxidation such as 4-hydroxynonenal (4-HNE), which itself arises from the peroxidation of PUFAs and is often used as an immunohistological marker in the context of histological detection of ferroptosis (Fig. 2) [55, 57]. Ongoing research continues to add new insights into the molecular cascade involved in ferroptosis. Based on experimental analyses and RNA expression analysis in MS patients, there is evidence that ferroptosis is a very early event in MS and experimental autoimmune encephalomyelitis (EAE), and occurs earlier than T-cell activation. It is hypothesized that ferroptosis may promote T-cell activation through T-cell receptor (TCR) signaling in vitro and in vivo [60]. Another key finding relates to the endoplasmic protein stimulator of interferon genes (STING), which contributes to the release of interferon 1 (IFN1) in microbial infections through canonical activation. In MS, non-canonical activation of STING has been described, which leads to degradation of the anti-ferroptotic GPX4 by autophagy. In EAE, increase in the phosphorylated activated form of STING has been described with increasing exacerbation of EAE [62]. The protein ninjurin-1 (NINJ1), which plays no role in the early phase of ferroptosis in the context of lipid peroxidation, has been shown to be involved in the late phase. It is involved in the rupture of the plasma membrane and the release of DAMPs, and it has also been shown that its reduced expression in macrophages protects these cells from ferroptosis [63]. Another newly described inducer of ferroptosis in MS and EAE is the histone methyltransferase G9a, which leads to a repression of anti-ferroptotic genes and contributes to the reduction of intracellular glutathione levels and thus to the initiation of ferroptosis [64].
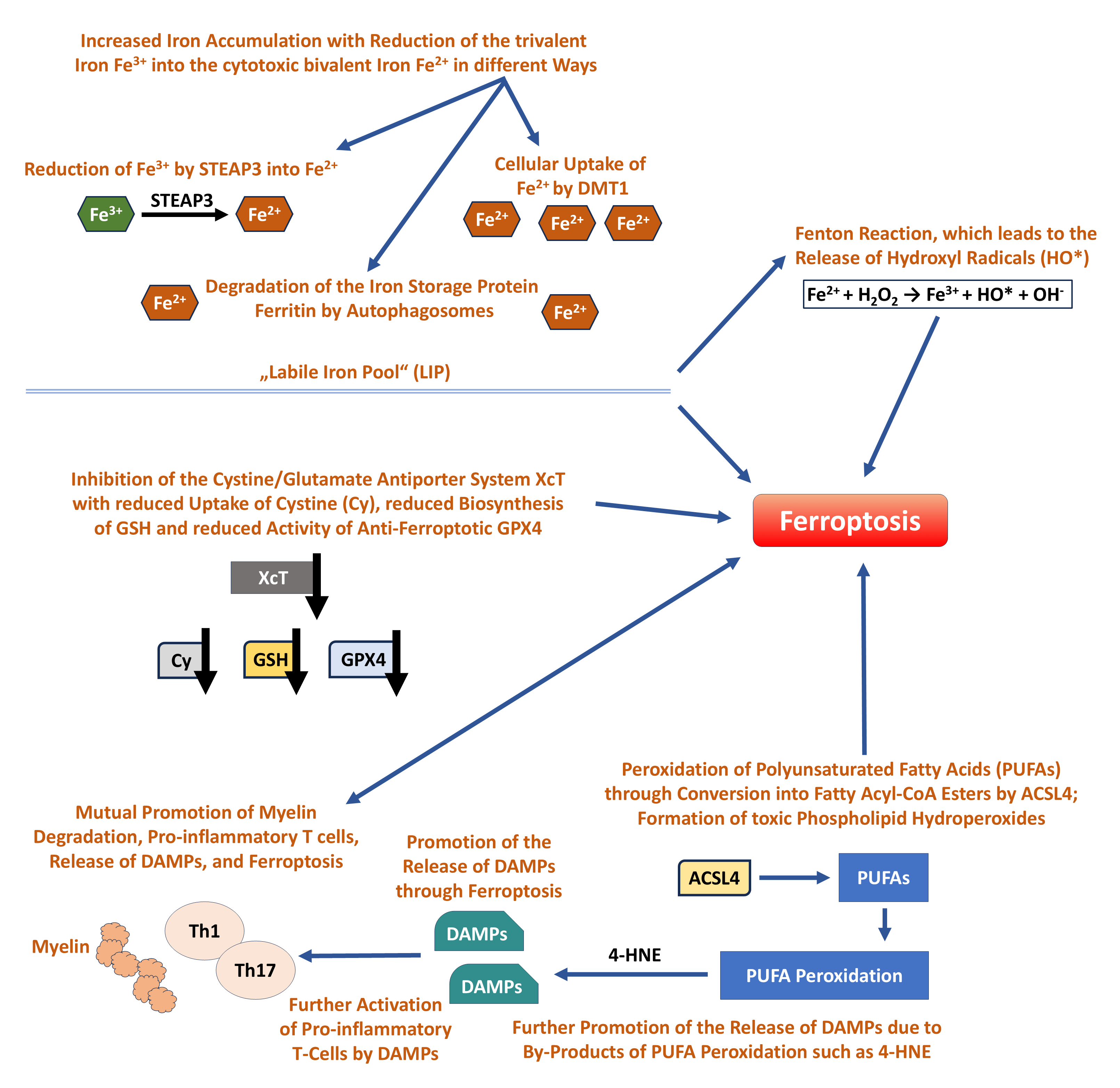 Fig. 2.
Fig. 2.
Ferroptosis in Multiple Sclerosis - the main changes and pathways and their cumulative effect on initiation of ferroptosis [55, 56, 57, 58, 59, 60, 61] as described in the text. STEAP3, six-transmembrane epithelial antigen of prostate 3; DMT1, divalent metal transporter 1; GSH, glutathione; GPX4, glutathione peroxidase 4; DAMPs, danger-associated molecular patterns; 4-HNE, 4-hydroxynonenal; PUFAs, polyunsaturated fatty acids; ACSL4, acyl-CoA synthetase long-chain family member 4.
Research on cell-type-specific ferroptosis in MS is still in its early stages, with genetic and expression studies providing new insights. Single cell analyses in defined regions of the central nervous system of MS patients and control cases showed a particularly pronounced change in the expression of ferroptosis-relevant genes (ferroptosis score) at the edges of active MS lesions, mainly affecting the phagocytizing microglia and macrophages. Compared to these peripherally localized macrophages, the macrophages in the center of the lesions showed a lower ferroptosis score. In contrast, a significantly higher ferroptosis score was detected in the neurons of the grey matter [65]. Single cell analysis revealed that ferroptosis occurs in all primary cell types of the CNS, with evidence of reduced expression of several ferroptosis-inhibiting proteins such as glutathione peroxidase 4 (GPX4) or ferritin heavy chain 1 (FTH1). Anti-ferroptotic gene expression was reduced in neurons, oligodendrocytes, oligodendrocyte precursor cells and astrocytes of MS patients compared to controls [66]. Further studies have confirmed ferroptosis in all these cell types, and reduced expression of anti-ferroptotic proteins such as GSH and GPX4 has been identified as a significant contributing cause [48, 61, 67]. The situation is different for CD4+ T lymphocytes, which show a high expression of the anti-ferroptotic protein GPX4 in MS patients and are therefore largely protected from ferroptosis. This observation was verified by an experimental knockout of GPX4, which resulted in improved symptoms in mice with EAE [68]. Single cell analysis of human cells from MS patients also showed that the ferroptosis score is low in both T and B lymphocytes, confirming that the high ferroptosis score in MS lesions affects macrophages in contrast to lymphocytes [65]. Genetic analyses are usually based on a large data set of many genes and their changes in certain diseases. Methods such as “weighted gene co-expression network analysis” and “receiver operating characteristic curve analysis” can be used for that purpose. These are data analysis methods for detecting changes in specific genes in defined diseases, in this case: changes concerning ferroptosis-related genes in MS [69]. These ferroptosis-related genes are involved in numerous functions such as mitochondrial translation, microglia activation, inflammation and oxidation, but the need is emphasized that the exact involvement of individual genes in MS has yet to be investigated [69]. In addition to the already mentioned reduced expression of anti-ferroptotic genes such as GPX4, the downregulation of the gene of the anti-inflammatory hemoxigenase1 (HMOX1) should be emphasized [69]. Ferroptosis-associated genes that are overexpressed in MS include the genes of arachidonate lipoxygenases 5 and 12 (ALOX5, ALOX12), which are involved in the lipid peroxidation of PUFAs [70], as well as the mitochondrial protein voltage-dependent anion-selective channel protein 3 (VDAC3). This protein is involved in the interaction with pro-apoptotic proteins, and has a number of other functions that are still poorly understood and which are evidently related to PUFAs [70].
Apoptosis can be induced via two main pathways: the extrinsic pathway, mediated by receptors of the tumor necrosis factor (TNF) family and by the effector caspases 3, 6 and 7; and the intrinsic (mitochondrial) pathway, initiated by pro-apoptotic proteins like Bcl-2-associated X protein (BAX), Bcl-2 homologous antagonist/killer (BAK), caspase 3 and the release of cytochrome C. Other factors may be involved in the induction of apoptosis, in particular the increased expression of p53 and complement-mediated apoptosis [71, 72]. The role of defective apoptosis in multiple sclerosis was already suspected at the turn of the millennium. Studies revealed that expression of anti-apoptotic proteins such as myeloid cell leukemia sequence 1 (MCL1) was increased in T-cells, while pro-apoptotic proteins such as BAK were reduced [73, 74]. Inspired by these results, increased expression of histone deacetylase 3 (HDAC3) was discovered in MS patients. HDAC3 is involved in various processes of cell proliferation and inhibition of apoptosis, whereby the downregulation of p53 appears to play a role. In vitro inhibition of HDAC3 resulted in an increased expression of p53 in T-cells from healthy individuals. In contrast, T-cells from MS patients showed no increased expression of p53, but instead an increased resistance to apoptosis. Therefore, a role of HDAC3 in the prevention of apoptosis of autoreactive T-cells and thus an involvement in the promotion of autoimmunity is suspected [71]. The Fas ligand (CD95) is a member of the TNF family and is involved in the initiation of apoptosis. In MS, its expression on the surface of CD4+ T-cells is reduced. Moreover, in mice with Fas gene knockout, maintenance of EAE due to the lack of Fas expression was confirmed experimentally [75]. Expression of the Fas ligand (Fas-L) by astrocytes is necessary for the induction of apoptosis by Fas, which was demonstrated in knockout mice without expression of astrocytic Fas-L by the lack of expression of caspase 3 and the absence of apoptosis of autoreactive CD4+ T-cells [76]. The nuclear receptor 4A1 (NR4A1) is involved in cellular proliferation, DNA repair, the regulation of T-cell-mediated immunity and the initiation of apoptosis. When comparing MS patients with a mild clinical course and patients with active relapsing-remitting MS, a reduced expression of NR4A1 with suppression of apoptosis of activated T-cells was found in the latter group, as well as a suppression of regulatory T-cell activity. With regard to cell type specificity, it is noteworthy that when comparing the transcriptional profiles of both patient groups, 75% of the genes were linked to CD4+ T-cells, but not to CD8+ T-cells, CD14+ macrophages or CD20+ B-cells. The authors conclude that these mechanisms collectively contribute to an augmented autoimmune response and increased disease activity, and that targeting the NR4A1-pathway in reactive T-cells could be a viable therapeutic strategy to prevent MS progression [77].
Apoptosis in MS and in EAE has been demonstrated in oligodendrocytes (OL),
although it has been pointed out that their cell death in MS can also be caused
by other factors such as metabolic stress or ferroptosis. It must therefore still
be emphasized today, that the exact mechanism responsible for OL cell death in
human MS remains elusive [78]. Apoptosis has been detected in oligodendrocytes of
MS brains within morphologically intact brain areas, in the grey matter near
intact neurons, and at the edge of areas with active demyelination. It is
therefore hypothesized that apoptosis of oligodendrocytes precedes or even
induces myelin damage in MS [79]. Pathological changes in mouse models with
induced apoptosis of oligodendrocytes revealed demyelination and migration of
microglia, which were similar to the changes in human MS [80, 81]. Apoptosis of
cerebral neurons in a mouse model with defective autophagy induced by suppressed
autophagy-related protein 5 (Atg5) also demonstrated the role of
oligodendrocytes. The defective autophagy in oligodendroglia led to myelin
defects in the animals with reduced expression of basic myelin protein (MBP) and
cyclic nucleotide phosphodiesterase (CNP), cognitive impairment in the context of
behavioral tasks, as well as degeneration of axons and apoptosis of neurons with
increased expression of apoptosis markers such as caspase 3 [82]. To date, the
contribution of neuronal apoptosis to the clinical course of MS is controversial.
However, there is evidence, that microglia trigger neuronal apoptosis by
increased expression of cytotoxic molecules such as TNF
This form of cell death was described by Cookson and co-workers in 2001 [5]; the
name refers to the detection in macrophages in the context of a Salmonella
infection. Pyroptosis represents an independent form of PCD, characterized by its
own signaling pathways. It has been reported in various inflammatory reactions,
but also in several neurodegenerative diseases and tumors. The typical activation
occurs through the contact of danger-associated molecular patterns (DAMPs) and/or
pattern-associated molecular patterns (PAMPs) with the inflammasome. The
inflammasome is a protein complex of the innate immune system. This protein
complex consists of sensor complexes such as the protein family “nucleotide
binding oligomerization domain leucine rich repeat and pyrin domain containing”
(NLRP). These NLRPs recognize DAMPs and PAMPs mediated by Toll-like receptors
(TLR) and subsequently lead to activation of the “nuclear factor
kappa-light-chain-enhancer of activated B-cell complex” (NF-
Microglia and neurons were found to be the main cell types in which pyroptosis occurs in MS [92, 93]. More and more activation pathways of pyroptosis affecting these cell types were discovered. The protein “absent in melanoma 2” (AIM2) is a sensor for double-stranded DNA and is involved in the activation of the inflammasome and caspase-1 after recognition of aberrant DNA. In cortical neurons, activated AIM2 not only triggers caspase 1 but also increases the expression of Il-1ß and mediates pyroptosis [93]. Application of IFN-ß prevents pyroptosis through the downregulation of AIM-2, NLRP1 and NLRP3, so that IFN-ß is considered a potential therapeutic agent in MS regarding the prevention of this form of PCD [86, 93]. GSDMD-initiated pyroptosis was demonstrated by in vitro analysis in microglia from MS patients and from an EAE mouse model; experimental inhibition of caspase-1 using the small-molecule inhibitor VX-765 demonstrated a suppression of pyroptosis in each case [94]. Overexpression of caspases 3 and 7 in GSDMD-immunopositive microglia was also described as an additional signaling pathway in microglia from MS patients and from EAE transgenic mice. Here, as well, a decline in pyroptosis was observed in the form of a decrease in typical GSDMD-positive cytological changes with rounding and fragmentation of the cell membrane following experimental inhibition of caspases 3 and 7 [92]. Caspases 3 and 7 are also activated by caspase 1 and are then in turn involved in the activation of GSDMD, whereby in particular a co-localization of activated caspase 3 and activated GSDMD in the microglia from the MS lesions has been demonstrated [92]. Irrespective of pyroptosis and inflammatory diseases such as MS, extensive research into the inflammasome and its components, particularly NLRPs, should be noted. This research complex is all the more important for microglia in MS, as the mere activation of NLRPs does not necessarily result in pyroptosis [95]. Of the 14 NLRP proteins, NLRP1, NLRP3 and NLRP4 in particular have been described as being involved in the activation of an aberrant immune response in autoimmune diseases, as well as in the activation of pyroptosis [93, 95]. The best-researched protein, NLRP3, has multiple roles. In addition to activating inflammatory reactions including pyroptosis, it is involved in the recruitment of activated T-cells and in the prevention of cell death of Th17 cells. In microglia, it is involved in promoting the expression of inflammatory cytokines and in the initiation of pyroptosis [65, 86, 88]. The expression of NLRP1 and AIM-2 was detected in neurons, but no neuronal expression of NLRP3 was observed [96]. NLRP1 also activates caspase-1, but it is pointed out that its exact cascade of action is still poorly understood [88, 96]. NLRP4, like NLRP3, is present in microglia and is increasingly expressed in MS foci. Therefore, NLRP3 and NLRP4 are considered to be the actual regulators of inflammatory activity and also of pyroptosis in activated microglia in MS [88].
In addition to the three most commonly studied forms of cell death in MS (ferroptosis, apoptosis, pyroptosis), there are already findings on necroptosis and NETosis. The role of autophagy in MS is also still the subject of research. The special case of autophagic cell death is subject to a strict definition with the need to exclude all other forms of cell death. Proof of the occurrence of autophagic cell death in MS is still pending.
Degterev and co-workers first described necroptosis in a mouse model with
cerebral ischemia. It was given its name because of its cytological similarity to
necrosis with rupture of the plasma membrane [97, 98]. Central and diagnostically
relevant molecules in the activation cascade are receptor-interacting protein
kinase 1 and 3 (RIPK1/3) and the protein mixed lineage kinase domain-like (MLKL),
which is activated by RIPK1 and RIPK3 and subsequently induces necroptosis as a
result of oligomerization. The receptor-interacting protein kinases RIPK1 and
RIPK3 are serine/threonine protein kinases, which in turn are activated by
osmotic stress, tumor necrosis factor
The term “autophagy” refers to the self-preservation of a cell through the degradation of its own cellular components under physiological conditions. Since the first description of autophagy by Clark in 1957 and by Ashford and Porter in 1962 [105, 106], the molecular cascade has been investigated in detail. Of the three forms of autophagy, macroautophagy with the formation of an autophagosome surrounded by a double membrane is often used in the literature as a synonym for the term “autophagy”. Two further forms are microautophagy with typical changes in the lysosomal membrane such as protrusion or invagination, and chaperone-mediated autophagy with direct lysosomal uptake of the substances to be degraded as a result of the activity of the chaperone “heat shock protein family A member 8” (HSPA8) and the lysosome-associated membrane protein 2 (LAMP2) [107]. Macroautophagy, referred to below as “autophagy” in accordance with the literature, is essentially initiated by the activation of autophagy-related genes (ATGs). A central cascade consists of the activation of ATG8 and the Unc-51-like kinase complex (ULK) by AMP-activated protein kinase (AMPK), which, together with the activation of additional ATGs, leads to the formation of an autophagosome and, as a result of its fusion with lysosomes, to an autolysosome [87, 108]. In contrast to autophagy, much less is known about “autophagic cell death”. For its detection, genetic or experimental evidence is required that (1) it occurs as a result of activation of the autophagy cascade, and (2) that all other forms of programmed cell death can be excluded [107]. In MS, autophagy has a multifaceted significance regarding the promotion of other forms of cell death such as apoptosis or ferroptosis. This should be distinguished from autophagic cell death, whose role in MS has not yet been clearly established. Autophagy makes an important protective contribution in the CNS due to increased disposal of damaged mitochondria with consequently reduced production of ROS [108, 109, 110]. This applies in particular to astrocytes and oligodendrocytes with corresponding promotion of cellular survival of both cell types. It is still unclear whether autophagy is dysregulated in astrocytes or in neurons of MS patients [111]. ATG expression was reduced in oligodendrocytes of MS lesions, but increased in microglia. These findings indicate an inhibited protective effect of autophagy in oligodendrocytes, but an intensified cell-preserving function of autophagy in the case of pro-inflammatory microglia [108]. As with pro-inflammatory microglia, intensified autophagy plays an important role in the maintenance of pro-inflammatory T-cells, as well as B-lymphocytes, in which an activation of autophagy as a result of citrullination of peptides in the context of EBV infection is suspected [111]. Autophagy in microglia, in pro-inflammatory T-cells and in B-cells thus appears to be a central factor in the progression of MS [109, 111, 112]. Dysregulation of autophagy can also lead to the induction of various forms of cell death. Examples include the promotion of ferroptosis by degradation of the anti-ferroptotic protein GPX4 during autophagy [62], and indirectly by damage to myelin and neuronal axons due to reduced autophagic activity in oligodendrocytes with secondary induction of apoptosis of neurons [82]. As already mentioned, autophagic cell death must be distinguished from these examples of induction of other forms of cell death; a role for autophagic cell death in MS has not yet been conclusively confirmed.
NETosis is a form of cell death that affects neutrophil granulocytes (NGs) and, together with the general role of NGs, is attracting increasing scientific interest in the field of autoimmune diseases. With regard to NGs, it has been shown in MS that, in contrast to a control group of healthy volunteers, NGs exhibit a primed status, i.e., the status of primary activation as a result of the proinflammatory environment. The significant changes in the NGs were shown by higher expression of TLR-2, IL-8, CD43, and the formyl peptide receptor (fMLP-receptor), and also by enhanced degranulation and oxidative stress [113]. In addition, an increased proportion of so-called “low-density granulocytes” was detected in the peripheral blood of MS patients, which are characterized by a typical expression pattern with a lack of CD14-expression and increased CD15-expression. They occur more frequently in various pathologies such as inflammatory diseases or in patients with heart disease compared with healthy persons [114, 115]. It remains unclear whether this cell fraction of NGs plays a causal pathophysiological role or is more of an epiphenomenon [114]. Following contact of NGs with pathogens or as a result of diverse stimulation of NGs by immune complexes, cytokines or chemokines, the formation of so-called “neutrophil extracellular traps” (NETs) can occur. These NETs consist of various components such as nuclear proteins like histones, granules, cytoplasmic components like neutrophil elastase (NE), high-mobility-group protein B1 (HMGB1), myeloperoxidase (MPO), and proteinase-3 (PR3), which are released by the NGs into the extracellular space [116]. The first of the two basic forms of this “NETosis” is the “suicidal NETosis” in which lysis and thus cell death of the NGs occurs because of chromatin decondensation, increased calcium release, increased expression of protein kinase C (PKC), induction of the nicotinamide adenine dinucleotide phosphate oxidase (NADPH-oxidase) complex, and an increased production of reactive oxygen species (ROS). In “non-lytic NETosis”, NETs are formed without subsequent lysis of the NGs [116]. In the peripheral blood of MS-patients, significantly higher levels of NETs and increased concentrations of neutrophil-activating chemokines, cytokines, and neutrophil-derived enzymes were detected [113, 117]. Importantly, elevated plasma levels of several NET-associated proteins such as neutrophil elastase (NE) and myeloperoxidase (MPO) correlate with lesion burden and the extent of demyelination (Table 1, Ref. [55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 113, 114, 115, 116, 117, 118, 119, 120]) [117, 118]. In addition, NETs promote the activation of pro-inflammatory Th17 lymphocytes in autoimmune diseases [117, 119, 120]. Overall, previous experience confirms a significant role of NGs and NETs in promoting an inflammatory environment in various diseases and a potential role in MS, but it is also emphasized in the latest literature that research on NETs and NETosis is still at a very early stage [116, 117]. This also applies to the differences between the two forms of “vital non-lytic NETosis” and “suicidal NETosis” associated with cell death, which also still need to be investigated in MS and its experimental models.
| Type of PCD | TT: Typical Triggers | Major Components of the Molecular Cascade |
| MS: Major Impacts on MS and its Experimental Models | ||
| Ferroptosis [55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70] | TT: Increased iron accumulation, increase of ferrous iron (Fe2+); inhibition of the cellular antioxidant system; peroxidation of poly-unsaturated fatty acids | STEAP3, DMT1, PUFAs, ACSL4, GSH, GPX4 |
| MS: Early event in MS, preceding the activation of pro-inflammatory CD4+ T-cells; further acceleration of inflammation and demyelination through a release of danger-associated molecular patterns (DAMPs) | ||
| Apoptosis [71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85] | TT: Different triggers such as increased expression of receptors of the tumor necrosis factor (TNF) family, pro-apoptotic proteins such as BAX and BAK, p53, or complement-mediated activation | TNF- |
| MS: Dysregulation of apoptosis in reactive T-cells; evidence for a functional link between neuronal apoptosis and altered activation status of microglia | ||
| Pyroptosis [86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96] | TT: Contact between DAMPs and/or PAMPs with the inflammasome, detected by NLRPs; activation of GSDMD by ASC and Caspase-1 or by LPS and Caspases 4, 5 and 11 | DAMPs, PAMPs, NLRPs, ASC, GSDMD, LPS, AIM2, Caspases |
| MS: Cortical neurons are affected by pyroptosis due to activation of AIM2; pyroptosis can also occur in microglia | ||
| Necroptosis [97, 98, 99, 100, 101, 102, 103, 104] | TT: Inactivation of caspase 8 and overactivation of RIPK1/3 and MLKL in MS-lesions are an indication for the activation of the necroptotic machinery in MS | RIPK1/3, TNF- |
| MS: With additional consideration of experimental results, necroptosis was detected in all primary cell types of the CNS: neurons, oligodendrocytes, astrocytes, microglia; evidence for induction of necroptosis in oligodendrocytes and promotion of inflammation due to overexpression of RIPK1 in microglia | ||
| NETosis [113, 114, 115, 116, 117, 118, 119, 120] | TT: Contact between neutrophil granulocytes (NGs) and pathogens such as DAMPs and PAMPs leads to the expression of NETs consisting of numerous proinflammatory components such as NE or MPO | NETs, NE, MPO, HMGB1, PR3, PKC, ROS |
| MS: A higher concentration of NETs has been detected in the peripheral blood of MS patients; a significant pro-inflammatory role of NGs and NETs in the context of MS is suspected |
Abbreviations of molecules: - Ferroptosis: STEAP3, ferric reductase
six-transmembrane epithelial antigen of prostate 3; DMT1, divalent metal
transporter 1; PUFAs, polyunsaturated fatty acids; ACSL4, acyl-CoA synthetase
long-chain family member 4; GSH, glutathione; GPX4, glutathione peroxidase 4. -
Apoptosis: TNF-
Most of the risk factors for MS such as vitamin D deficiency and genetic influences have been well known for a long time. Currently, there is increasing interest in epigenetic factors in MS, including the methylation status of genes regulating myelin homeostasis [18], and the altered expression behavior of non-coding RNA molecules such as micro-RNAs (miRNAs) or circular RNAs (cRNAs) and their influence on the inflammatory activity of T and B-cells [19, 121]. Despite the known significance of EBV virus infection and pathological intestinal bacterial flora, the need for further research is also emphasized for these two aspects [23, 24, 26, 29]. The cumulative influence of all these risk factors contributes to cerebral immunological dysregulation; the scientific focus is still on the increased activity of pro-inflammatory CD4+ T-lymphocytes, peripheral monocyte-derived macrophages and resident mononuclear phagocytes of the central nervous system such as microglia, but also on B-lymphocytes (Fig. 1) [9, 33, 34, 35, 37]. An additional aspect of MS pathology is the dysregulation of the cerebral iron balance, mainly caused by inflammatory damage to the oligodendroglia and the iron-containing myelin [44, 45]. Increased uptake of iron by pro-inflammatory T-cells, B-cells and macrophages, and phagocytosis of myelin debris by macrophages seems to intensify pro-inflammatory activity, oxidative stress with damage to the mitochondria and increased production of cytotoxic Fe2+, causing damage to oligodendrocytes and macrophages [44, 45, 46, 47, 48]. Neuronal damage because of increased cellular uptake of iron has also been proven (Fig. 1) [45, 53]. The currently increased scientific interest in iron dysregulation in MS coincides with the forthcoming renewal of the McDonald criteria for the diagnosis of MS, especially the paramagnetic rim lesions (PRLs) in the brain. MRI scans will be a diagnostic criterion alongside other innovations such as the central vein sign or the detection of kappa free light chains as biomarkers. PRL is a paramagnetic effect detectable in T2*- and susceptibility-weighted MR sequences with central extinction of the MR signal due to iron-loaded macrophages at the edge of MS lesions. The sensitivity and specificity of this sign in MS is considered very high at 90% and therefore diagnostically significant [50, 51, 122]. Although it is currently repeatedly emphasized that the dysregulation of iron balance in MS is only incompletely understood, the imminent introduction of PRL as a diagnostic criterion represents an important step and ultimately proves that imaging evidence of dysregulation of iron balance in MS is diagnostically and clinically significant [50, 51, 122].
In addition to the direct promotion of pro-inflammatory activity of lymphocytes and macrophages, the promotion of ferroptosis is one of the most important aspects of focal iron overload in MS. The main findings on ferroptosis in MS are currently that it is a common form of programmed cell death (PCD), and that it occurs very early during MS. Ferroptosis plays a direct role in the development of MS and in the activation of pro-inflammatory lymphocytes, with ferroptosis and the pro-inflammatory process apparently promoting each other (Fig. 2) [56, 60]. Experimental and genetic analyses in MS brains revealed that ferroptosis occurs in all brain cell types: neurons, astrocytes, oligodendrocytes, and oligodendrocyte precursor cells [48, 61, 66, 67]. In contrast, pro-inflammatory T and B-lymphocytes showed a reduced ferroptosis score, and CD4+ T-cells additionally showed increased expression of the anti-ferroptotic protein GPX4, and thus extensive protection of these cells against ferroptosis [65, 68]. Very similar, inhibited apoptosis in T-cells of MS brains was found due to decreased expression of several pro-apoptotic proteins such as Fas-ligand (CD95) and “nuclear receptor subfamily 4 group A member 1” (NR4A1), as well as increased expression of anti-apoptotic proteins such as MCL1 and HDAC3 [71, 75, 76, 77]. In contrast to the reduced apoptosis of pro-inflammatory T-cells, a significant occurrence of apoptosis in oligodendrocytes (OLs) and in neurons has been demonstrated in MS, with apoptosis occurring very early and in morphologically intact brain areas, particularly in the case of OLs. Consequently, the hypothesis is being discussed that apoptosis of OLs contributes significantly to myelin damage at an early stage of the disease [78, 79]. In neurons, there is evidence for the occurrence of apoptosis in MS because of microglial activation with expression of inflammatory cytokines, as well as evidence for complement-activated apoptosis [72, 83, 84]. In MS, autophagy primarily makes a protective contribution as a result of the degradation of damaged mitochondria with the avoidance of increased oxidative stress. However, reduced expression of autophagy-regulating genes with a correspondingly reduced protective effect on endogenous brain cells has been demonstrated in MS lesions, whereas an increased expression in pro-inflammatory microglia with corresponding protection of these cells by autophagy was observed [108, 109, 111, 112]. The detection of autophagic cell death requires the reliable exclusion of other forms of programmed cell death, which has not yet been demonstrated in MS. In contrast, the occurrence of pyroptosis in MS has been described with certainty; it mainly affects microglia and neurons. However, the functions of both the gasdermins and the NLRPs as the main components of the inflammasome are still incompletely understood and the subject of research. According to current knowledge, NLRP1 is a component of the inflammasome in neurons, while NLRP3 and NLRP4 are involved in the regulation of the inflammatory activity of microglia [65, 86, 88, 96]. Like ferroptosis, necroptosis was also detected in autoptic and experimental studies in all primary cell types of the CNS in MS or EAE [99, 101, 102, 103, 104]. In addition, increased expression of the necroptosis-inducing protein RIPK1 in pro-inflammatory microglia was detected in MS, which led to necroptosis in oligodendrocytes [101]. Based on previous experience, the occurrence of necroptosis in MS can be considered certain, although experience in this regard is still relatively limited compared to the proven significance of ferroptosis (Table 1).
Concerning other forms of PCD, recent findings include the importance of neutrophil granulocytes and their formation of “neutrophil extracellular traps” (NETs) in MS, whose concentration in the serum of MS patients is significantly increased and correlates with the severity of the disease [113, 117, 118]. Further investigation is needed to determine whether this expression of NETs in MS occurs as a result of vital or suicidal NETosis, and whether the increase of low-density granulocytes is an epiphenomenon or a causal factor in the pathogenesis of MS [113, 114, 117, 118]. Parthanatos, another type of PCD, has already been shown to play a role in other neurodegenerative diseases such as amyotrophic lateral sclerosis or Parkinson’s disease, but also in EAE and MS [123, 124]. The central regulatory protein of parthanatos, poly-ADP-ribose polymerase-1 (PARP-1), leads to cellular protection when moderately activated, but to cell death as a result of nicotinamide adenine dinucleotide (NAD) depletion when more strongly activated. Therefore, it is currently unclear whether therapeutic manipulation of PARP-1 is beneficial or detrimental to disease progression [125, 126]. Panoptosis is a form of cell death in which key enzymes of apoptosis, necroptosis and pyroptosis and thus all three forms of PCD are activated. The exact mechanism of panoptosis has not yet been clarified. A panoptosome is postulated, which leads to the initiation of cell death in a similar way to the inflammasome and apparently contains various components of the inflammasome such as NLRP2 [127]. With regard to the interaction of different forms of PCD, panoptosis is therefore an important example due to the involvement of key molecular components of apoptosis, pyroptosis, and necroptosis. It is believed that the panoptosome plays a central role in this interaction due to its simultaneous activation of the molecular components of these PCDs. While research into panoptosis in tumors is already at an advanced stage, there is also some experience with neurodegenerative diseases such as Alzheimer’s disease; research into panoptosis in the context of MS is still pending [87, 127, 128, 129, 130]. Another example for the interaction of different PCDs is the role of caspase 8, which is an obligate promoter of apoptosis but, in the event of its inactivation and the simultaneous activation of RIPK1/3, enables the initiation of necroptosis [99, 100]. Research into the interaction of different PCDs is more advanced in the case of tumors such as gliomas, an important finding concerns cysteine as a cofactor of necroptosis and, at the same time, as an inhibitory factor of ferroptosis [87, 131]. However, it is explicitly pointed out that findings on the role of PCDs and their interactions are only specific to the respective disease or group of diseases and cannot necessarily be transferred to other diseases [87, 131]. In MS, in-depth research into these interactions between different PCDs is still in its infancy.
The same holds for the question concerning the involvement of different PCDs in
different cell types. As explained in the previous chapters, the current state of
knowledge is that ferroptosis and necroptosis can occur in all primary cell types
of the CNS in the context of MS and its models: neurons, oligodendrocytes,
astrocytes, and microglia [48, 61, 66, 67, 99, 100, 101, 102, 103, 104]. The occurrence of apoptosis
has been described primarily in oligodendrocytes and neurons, whereas neurons and
microglia were found to be the main cell types in which pyroptosis occurs in MS
[78, 79, 80, 81, 82, 83, 84, 92, 93]. It should be noted that in the case of microglia, their
significance in the induction of neuronal apoptosis in MS is suspected to be due
to increased expression of inflammatory cytokines [83, 84]. Another important
aspect mentioned in this context is the extensive prevention of PCD of
pro-inflammatory lymphocytes, particularly concerning ferroptosis and apoptosis
[65, 68, 76, 77]. When considering the significance of individual cell types in
PCDs, it must be taken into account that the current literature often highlights
the as yet incomplete but constantly growing knowledge for almost all cell types,
for example, the important aspect of the occurrence of apoptosis in
oligodendrocytes [78, 79]. It is important to note that the complex topics of
iron dysregulation, PCDs and their molecular components, as well as the cell
types involved in MS, is still dominated by an ever-growing number of individual
observations. One example of new research findings in this area is the
experimental blocking of MLKL, which leads to an improvement in clinical symptoms
and, as expected, to the blocking of necroptosis. In addition, however, it has
been shown that MLKL, apart from its importance as a necroptosis activator, has
additional significance in the development of an inflammatory response as a
result of the activation of Th1 and Th17 cells, as well as in the activation of
several signaling pathways such as the nuclear factor kappa-light-chain-enhancer
of activated B-cells (NF-
Another important aspect of research into PCDs in MS will be the role of
non-coding RNA molecules (ncRNAs). In the case of micro-RNAs, there are
approaches to understanding their role in the pathogenesis of MS and in
influencing certain forms of PCDs such as ferroptosis [136, 137], as well as in
the suppression of ferroptosis in the EAE model using micro-RNA miR-367-3b
delivered by stem-cell derived exosomes [138]. Investigation of the influence of
ncRNAs is therefore an essential aspect of current experimental research into
potential therapeutic approaches targeting forms of PCD (Fig. 3). Also, all other
aspects mentioned with regard to the forms of PCD point to a variety of
possibilities for experimental concepts for the development of new treatment
modalities for patients with MS. Current studies mainly relate to the
experimental targeting of ferroptosis or pyroptosis in the mouse model of EAE and
show remarkable results on clinical and morphological improvements due to the
inhibition of these PCDs (Table 2, Ref. [56, 138, 139, 140, 141, 142, 143, 144, 145]). In the case of
ferroptosis, in addition to the aforementioned application of microRNAs, current
therapeutic approaches using ferrostatin analogues and iron chelators are also
noteworthy [56, 138, 139, 140]. It should be emphasized that differences in in-vivo
success have been reported in the context of the use of various ferroptosis
inhibitors [56]. All of these approaches resulted in the inhibition of
ferroptosis, mainly due to the promotion of the expression of anti-ferroptotic
proteins such as GSH and GPX4. Histologically, reduced gliosis and a significant
attenuation of demyelination were demonstrated, as well as reduced loss of
oligodendrocytes, together with reduced iron deposition (Table 2) [56, 138, 139, 140].
Pyroptosis was targeted experimentally by influencing pro-inflammatory and
pyroptosis-promoting signaling pathways such as the HMGB1/TLR4/NF-
 Fig. 3.
Fig. 3.
Additional important aspects of research into various forms of PCD in multiple sclerosis, following on from research priorities already initiated: Neutrophil granulocytes and neutrophil extracellular traps (NETs), parthanatos, panoptosis, epigenetic research concepts, and special considerations for therapeutic concepts targeting PCDs.
| Type of targeted PCD | First Author, Year | Name and Type of the Agent |
| Ferroptosis | Van San 2023 [56] | UAMC-3203 (Ferrostatin-analogue) |
| Treatment lowered the overall clinical disease score with amelioration of disease progression and significant reduction in demyelination; this effect was more pronounced when compared with the use of other ferroptosis inhibitors | ||
| Ferroptosis | Fan 2023 [138] | miR-367-3p (micro-RNA) |
| miR-367-3p delivered by stem cell-derived exosomes led to alleviation of EAE by suppressing ferroptosis via activation of the antioxidant system cystine/glutamate antiporter system (XcT, SLC7A11) with activation of GPX4; the extent of inflammation and demyelination was significantly reduced | ||
| Ferroptosis | Jhelum 2023 [139] | UAMC-3203 (Ferrostatin-analogue) |
| Treated mice showed clinical improvement with reduced paralysis; histology showed reduction in myelin loss and lesion size, as well as increased protein levels of anti-ferroptotic proteins from the antioxidant system (XcT, GSH, GPX4) | ||
| Ferroptosis | Rayatpour 2022 [140] | Deferiprone (Iron chelator) |
| Reduction of demyelination, astrogliosis, and microgliosis in the optic nerve of EAE mice; reduction of axonal damage and ganglion cell loss in the retina; increased expression of the anti-ferroptotic protein GPX4; the iron deposition in the optic nerve was significantly diminished according to histological confirmation | ||
| Pyroptosis | Feng 2025 [141] | Sh-HMGB1 and Sh-NLRP3 (short hairpin RNA vectors targeting HMGB1 and NLRP3) |
| Application of the vectors via lentivirus infection led to inhibition of the HMGB1/TLR4/NF- | ||
| Pyroptosis | Song 2022 [142] | Liraglutide (GLP-1R agonist) |
| The glucagon-like peptide-1 receptor (GLP-1R) agonist is known to have neuroprotective capacity; after application, the disease score was ameliorated with reduction of inflammation and demyelination, the expression of the pyroptosis-associated proteins caspase-1, NLRP3 and GSDMD was also reduced | ||
| Pyroptosis | Wang 2025 [143] | Forsythoside B (natural phenylethanoid glycoside) |
| The severity of the clinical course was attenuated; the degree of demyelination and inflammation was also reduced with lowered amount of inflammatory CD4+ cells and glial activity; the expression of NLRP3 and GSDMD was decreased | ||
| Pyroptosis | Kiasalari 2021 [144] | Ellagic acid (natural polyphenol) |
| Severity of EAE was significantly reduced after treatment, as well as the expression of pro-inflammatory cytokines (TNF- | ||
| Pyroptosis | Zhao 2025 [145] | Evodiamine (natural tryptamine indole alkaloid) |
| Remyelination was enhanced as a result of upregulation of microglial SLC2A4-regulated autophagy; likewise, downregulation of NLRP3 led to inhibition of microglial pyroptosis, which was confirmed in vitro in a microglial cell line | ||
Dysregulation of the iron balance and different types of programmed cell death (PCD), in particular ferroptosis, apoptosis and pyroptosis, play a major role in the pathophysiology of MS. An important aspect of the forms of cell death is their ambivalent role in this disease due to their non-physiological occurrence in the brain’s own cells and their widespread absence in pro-inflammatory cells. Research of different forms of PCD in MS is at the beginning, and approaches to understand NETosis, parthanatos, and the role of individual micro-RNAs will bring further insights in the near future. Experimental results on ferroptosis and pyroptosis in animal models encourage a further intensive search for potential therapeutic approaches in MS. In addition, iron dysregulation has become increasingly important in neuroradiological imaging diagnostics, and the new diagnostic McDonald criteria will include the imaging correlate of iron dysregulation as a sign of MS disease. Based on current knowledge, it must be emphasized that both the dysregulation of iron metabolism and the individual forms of PCD in MS are not yet fully understood. This also applies to the diverse functions of the various molecular components of PCDs, as well as to the interactions between the individual forms of PCD and their occurrence and significance in the different cell types of the brain and in immune cells. Current studies show that more and more details are being discovered about all these aspects and that it is still not possible to comprehensively or even definitively explain the precise role and mechanism of these individual aspects in MS, such as the dysregulation of iron metabolism. In addition to the aspects mentioned above, the central tasks for future research include investigating additional forms of PCDs, of which NETosis and parthanatos have already been described in MS, while research into other forms such as panoptosis is still pending. The increasing importance of epigenetics in medical research is also becoming apparent in the context of MS and its models in relation to the forms of PCDs. In particular, it seems important here to investigate the significance of individual non-coding RNA molecules in the different forms of PCD and thus their role as potential therapeutic targets or even as therapeutic agents. Finally, it is likely that future research on forms of PCD will also focus on cell type specificity, for example, with regard to therapeutic targeting (Fig. 3).
RN and EH designed the review. RN performed the literature research. RN and EH wrote the initial draft of the article. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
Funding was provided by the Open-Access Publication Fund of Goethe University Frankfurt, administered by the University Library - Johann Christian Senckenberg (Central Library, Freimannplatz 1, 60325 Frankfurt am Main, Germany) [Grant/Award Number: 3100320061].
The authors declare there are no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



