1 Oncology Department, Hunan Provincial People’s Hospital, The First Affiliated Hospital, Hunan Normal University, 410002 Changsha, Hunan, China
2 Key Laboratory of Epigenetics and Oncology, The Research Center for Preclinical Medicine, Southwest Medical University, 646000 Luzhou, Sichuan, China
3 Oncology Department, The Second Xiangya Hospital, Central South University, 410012 Changsha, Hunan, China
Abstract
In recent years, immunotherapy has gained increasing prominence in the treatment of hepatocellular carcinoma (HCC). However, effective immune-related biomarkers for HCC remain limited. In this study, both transcriptomic data and clinical information on HCC were obtained from The Cancer Genome Atlas (TCGA) database.
The TIMER and GEPIA databases were used to validate the association between WDR4 expression and immune infiltration. Additionally, clinical and pathological data from patients who underwent single-agent immunotherapy for HCC were collected from Hunan Provincial People’s Hospital (The First Affiliated Hospital of Hunan Normal University). The relationship between WDR4 expression levels, clinical pathological data, and patient prognosis was assessed using the Kruskal–Wallis test and Kaplan–Meier survival curve analysis. Spearman’s correlation analysis was utilized to confirm the relationship between WDR4, CD68, and PD-L1 in HCC tissue.
WDR4 was significantly upregulated in HCC tissues compared to para-carcinoma tissues (p < 0.001) and exhibited strong diagnostic potential. WDR4 expression showed significant associations with various immune cells, including macrophages (p < 0.001). Kaplan–Meier survival analysis revealed that patients with high WDR4 expression had shorter postoperative progression-free survival in the context of immunotherapy. Data from 37 patients who underwent postoperative single-agent immunotherapy for HCC demonstrated a significant correlation between WDR4 expression levels and disease-free survival (DFS), with strong statistical significance (log-rank p < 0.001).
WDR4 shows elevated expression in HCC tissues and is associated with immune infiltration, establishing it as a prognostic biomarker in HCC. Furthermore, the positive correlation observed between WDR4 and CD68, as well as PD-L1 (CD274), underscores its potential as a guiding factor in immunotherapeutic approaches for HCC.
Keywords
- hepatocellular carcinoma
- TCGA
- the WDR4 gene
- clinical prognosis
- immunotherapy
Primary liver cancer ranks as the fifth most prevalent malignancy globally and second in male mortality rates [1], with hepatocellular carcinoma (HCC) representing its predominant pathological subtype. Surgical resection remains historically effective for early-stage HCC [2], yet post-curative resection five-year recurrence exceeds 60% [3]. For advanced HCC, significant benefits have been achieved with targeted immunotherapies [4, 5, 6]. The REFLECT study [7] and the RATIONALE-301 study [8], for instance, have demonstrated promising clinical outcomes with lenvatinib targeted therapy and tislelizumab immunotherapy in the treatment of HCC. Despite molecular targeting and immunotherapy emerging as first-line treatment modalities for advanced HCC, overall survival and drug response rates remain dismally low. The high metastatic potential of HCC and its poor overall treatment outcomes remain major hurdles [9]. Additionally, the development of drug resistance significantly compromised patient prognoses [10, 11]. Currently, clinically utilized immune checkpoint blockade (ICB) drugs primarily encompass anti-CTLA-4 and anti-PD-1/PD-L1 antibodies [12, 13]. However, unlike squamous cell lung cancer, PD-L1 lacks validation as an immunotherapy biomarker in HCC. Asian HCC patients exhibit ~15% objective response rates (ORR) to ICB monotherapy, constrained by an immunosuppressive microenvironment enabling immune evasion [12, 14]. Consequently, HCC lacks validated diagnostic/prognostic biomarkers and immunotherapy assessment criteria [15], necessitating exploration of molecular targets governing HCC pathogenesis, prognosis, and immune infiltration.
The WD repeat domain 4 (WDR4) gene, located at human chromosome 21q22.3, encodes a WD-repeat protein family member primarily implicated in cell cycle regulation, signal transduction, apoptosis, and gene expression modulation [16, 17]. Elevated WDR4 expression occurs in multiple malignancies including hepatocellular carcinoma (HCC), lung cancer, and esophageal cancer [18, 19, 20]. Functioning as a core m7G modification subunit, WDR4 critically regulates transcription, mRNA splicing/translation, and immune microenvironment remodeling [21, 22, 23, 24]. The MYC-targeted WDR4 pathway induces CCNB1 translation, promoting HCC proliferation, metastasis, and sorafenib resistance [25]. Moreover, METTL1/WDR4-mediated tRNA m7G modification confers lenvatinib resistance in HCC [26]. Despite these findings, research remains predominantly focused on METTL1 with limited investigation of WDR4 [27]. Here, HCC datasets from TCGA were analyzed to elucidate WDR4’s significance. Utilizing R software (https://cran.r-project.org/bin/windows/Rtools/) and online databases, we assessed correlations between WDR4 expression and clinicopathological features alongside immune cell infiltration, validating differential WDR4 expression in cancerous versus paracancerous tissues via immunohistochemistry alongside PD-L1 and CD68 correlations.
The TCGA data portal (https://portal.gdc.cancer.gov/) provided HCC RNA-seq data and clinical information focused on WDR4 expression [28]. Using R software, we integrated and deduplicated WDR4 mRNA expression with clinical data, yielding three hundred sixty-nine HCC samples and fifty adjacent tissue samples. Concurrently, clinical data were collected from thirty-seven patients at Hunan Provincial People’s Hospital (The First Affiliated Hospital of Hunan Normal University) who underwent HCC resection between January 2019 and December 2022 and received postoperative anti-PD-1 monotherapy. Inclusion criteria stipulated: ① All cases were diagnosed with primary HCC based on pathological examination; All cases underwent curative resection for HCC, followed by postoperative monotherapy (PD-1) immunotherapy. ② No targeted therapy, chemotherapy, or radiotherapy was administered before or after surgery; ③ No significant underlying comorbidities, secondary primary tumors, or immune-related diseases were present; ④ The medical records were complete, and follow-up data were comprehensive. Exclusion criteria: ① Patients diagnosed with other types of liver cancer (e.g., intrahepatic cholangiocarcinoma or mixed types); ② Hepatic metastases; ③ Patients who received preoperative antitumor treatments such as radiotherapy, chemotherapy, or local interventions were excluded; ④ Patients who did not receive monotherapy with immunotherapy after surgery, for example, those who received combined immunotherapy and targeted therapy or other antitumor treatments, were also excluded; ⑤ Patients with underlying diseases that severely affected their health were excluded; ⑥ Patients with immune-related underlying diseases were also excluded.
Utilizing HCC WDR4 mRNA and clinical data from TCGA, differential
expression analysis was conducted via R’s “Limma” package [29], where genes
with
The TIMER2.0 database (http://timer.cistrome.org/) generated graphical representations of relationships between WDR4 expression levels and various immune cells, cumulative survival rates, and immune-related cell interactions [30, 31, 32]. Kaplan-Meier survival curve analysis evaluated associations between immune cell infiltration levels and HCC patient prognosis. The GEPIA database [33] (http://gepia.cancer-pku.cn/) analyzed relationships between WDR4 and immune cell molecular markers, including PD-1 (PDCD1: The gene name for PD-1), PD-L1, CTLA-4 and mismatch repair genes MLH1, MSH2, MSH, PMS2.
The WDR4 RNA sequencing data from HCC patients in the TCGA database
were filtered and analyzed using R programming packages. The results were
visualized and explored to investigate the various functions and signaling
pathways associated with WDR4 co-expressed genes in cancer [34]. R
Under informed consent, specimens from 37 postoperative HCC patients receiving
single-agent immunotherapy were collected, including cancerous and adjacent
tissues, along with clinical data. Immunohistochemical staining [25, 36] was
performed to assess the expression of WDR4, PD-L1, and CD68 in both
cancerous and adjacent tissues. The scoring criteria for WDR4 and CD68
were based on staining intensity: a score of 1 (
The clinical data were statistically analyzed using R software version 4.2,
GraphPad Prism 8.0 (https://www.ddooo.com/softdown/157257.htm), and Xiantao
Academic (https://www.xiantaozi.com). The Wilcoxon rank-sum test was employed to
assess the difference in WDR4 expression between the two sample groups.
An AUC value greater than 0.70 was considered indicative of significant
diagnostic value. The Log-rank test was employed to validate the prognostic
differences associated with WDR4 in HCC patients, where
In this study, TIMER2.0 database analysis (Fig. 1A) revealed significant
WDR4 upregulation in multiple cancers, including bladder, breast,
cervical squamous cell carcinoma, cholangiocarcinoma, colon, esophageal, head and
neck squamous cell carcinoma, NK/T-cell lymphoma, HCC, lung adenocarcinoma, lung
squamous cell carcinoma, pheochromocytoma, prostate, rectal adenocarcinoma,
gastric adenocarcinoma, and endometrial carcinoma, with downregulation observed
only in thyroid carcinoma. Further validation using TCGA data from 369 HCC and 50
para-carcinoma tissues demonstrated significant WDR4 overexpression in
HCC versus para-carcinoma tissues (p
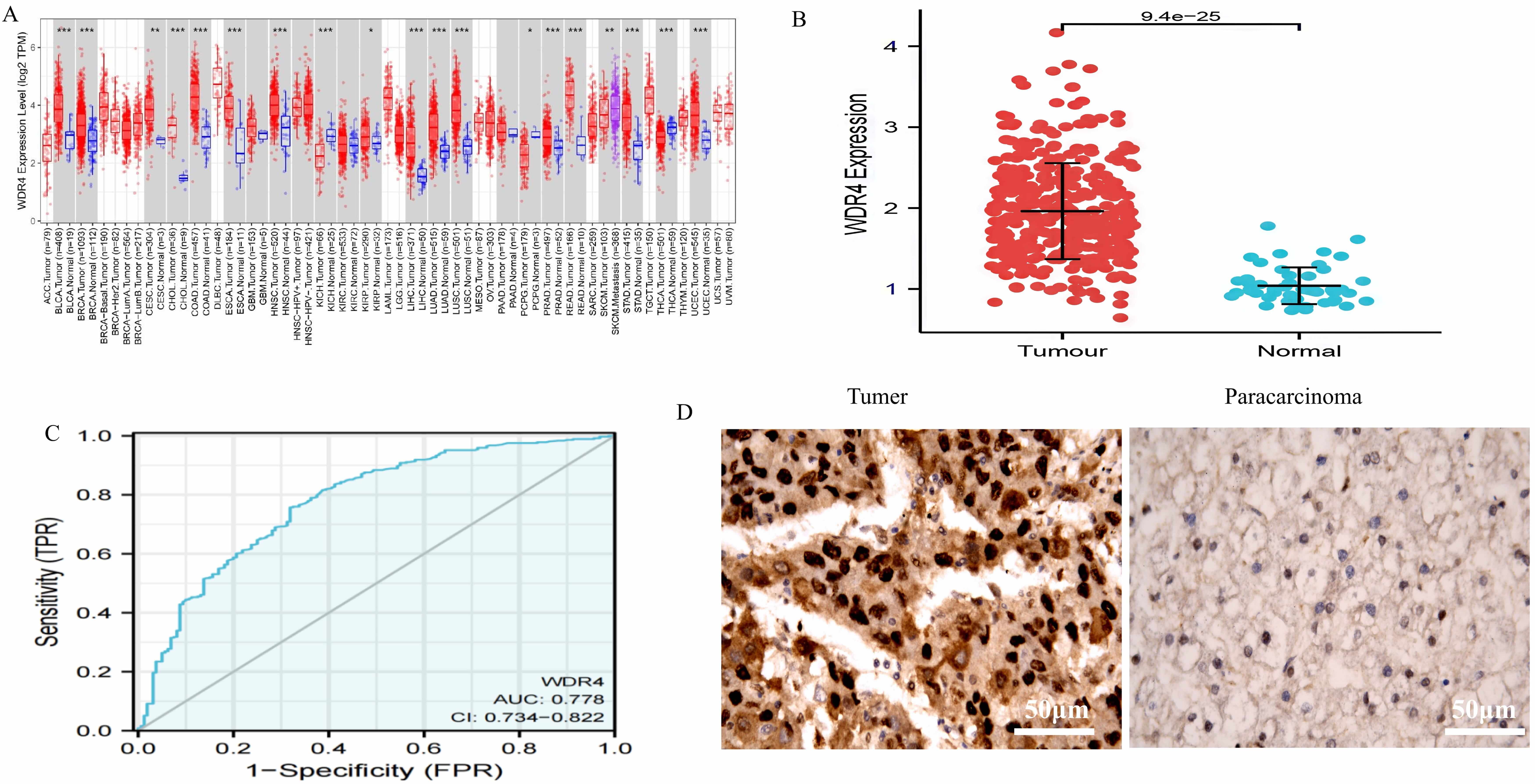 Fig. 1.
Fig. 1.
The expression of WDR4 in various cancers in the TIMER2.0 and
the expression and ROC curve analysis of WDR4 in cancers including HCC.
(A) The differential expression of WDR4 in various tumor tissues and
para-carcinoma tissues was analyzed using the TIMER2.0 database (***, p
Furthermore, immunohistochemical analysis confirmed the nuclear localization of
WDR4, as illustrated in Fig. 1D. Notably, WDR4 exhibited high
expression in HCC tissues while demonstrating low expression in para-carcinoma
tissues. The expression patterns of WDR4 in tumors and adjacent
non-cancerous tissues from 37 HCC patients are summarized in Table 1. In HCC
tissues, 10 cases (27.03%) exhibited low expression, while 27 cases (72.97%)
showed high expression. In para-carcinoma tissues, 1 case (2.70%) displayed high
expression, and 36 cases (97.30%) exhibited low expression. These results were
statistically significant (
| WDR4 expression level | HCC tissues | Para-carcinoma tissue | p-value | |||
| Number (n) | Percent (%) | Number (n) | Percent (%) | |||
| Low | 10 | 27.03% | 36 | 97.30% | 38.84 | |
| High | 27 | 72.97% | 1 | 2.70% | ||
HCC occurrence and progression involve regulation by multiple genes and diverse
factors; to elucidate the relationship between WDR4 and HCC development,
Kruskal-Wallis testing analyzed correlations between WDR4 expression
levels and patient clinical-pathological data (Fig. 2), revealing significantly
increased WDR4 expression in stage III versus stage II and I patients
(Fig. 2A), higher levels in T2 and T3 versus T1 stages (Fig. 2B), elevated
expression in pathological grades G3 and G4 compared to G1 and G2 (Fig. 2C),
increased WDR4 in patients with vascular invasion versus without (Fig. 2D), and higher expression associated with AFP levels
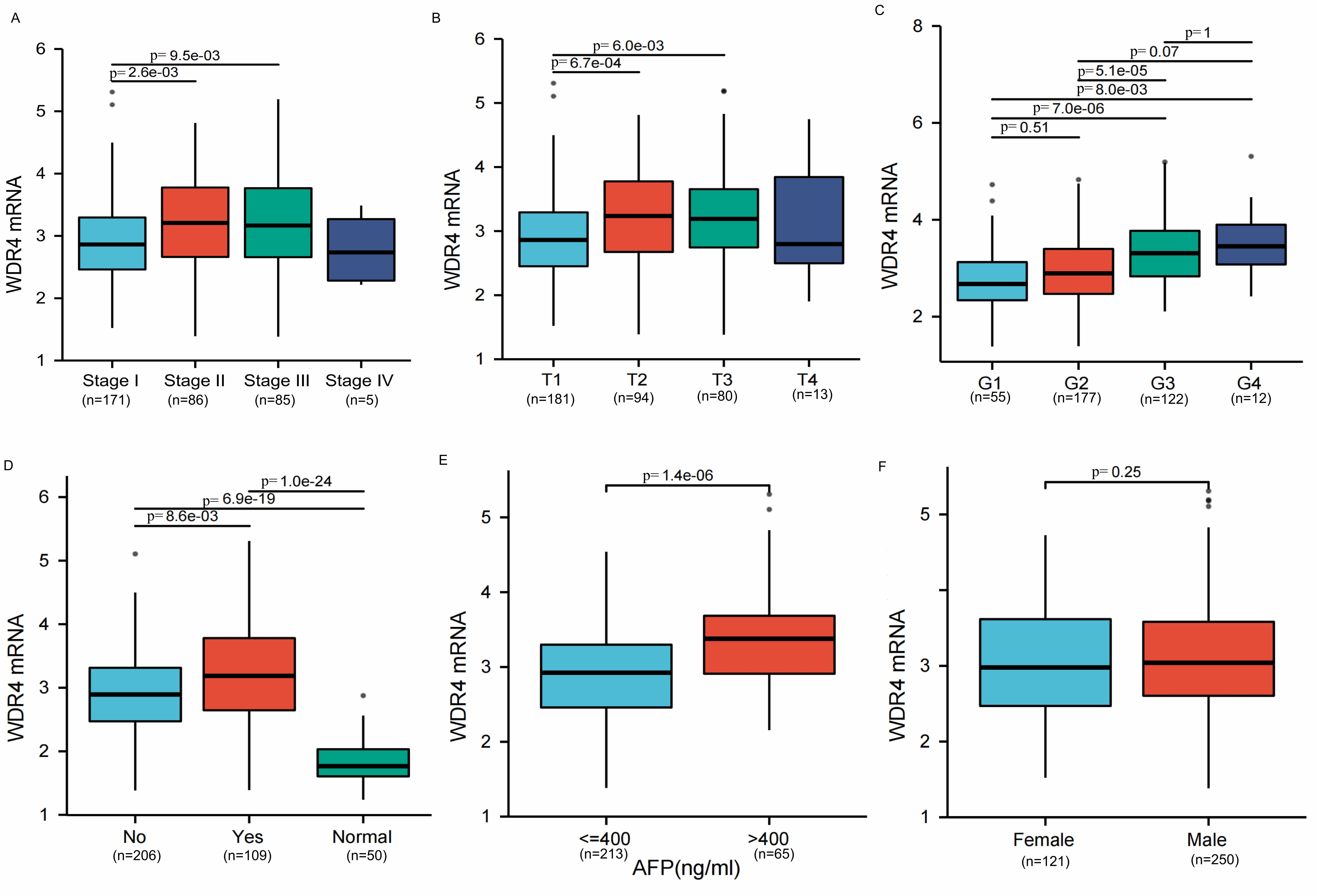 Fig. 2.
Fig. 2.
The relationship between WDR4 mRNA expression levels and the clinical stage, grade, and gender of HCC patients. (A) The relationship between WDR4 mRNA expression levels and the clinical stage of HCC patients. (B) The association between WDR4 mRNA expression levels and the T-stage classification of HCC patients. (C) The relationship between WDR4 mRNA expression levels and the pathological grading of HCC patients. (D) The relationship between WDR4 mRNA expression levels and vascular invasion in HCC patients (NO: representing no vascular invasion; YES: indicating presence of vascular invasion; Normal: para-carcinoma tissue). (E) The correlation between WDR4 mRNA expression levels and AFP values in HCC patients. (F) The relationship between WDR4 mRNA expression levels and gender in HCC patients.
Clinical-pathological data from 37 postoperative HCC patients treated with
single-agent immunotherapy were analyzed to validate the relationship between
WDR4 and clinical-pathological features. The results, as shown in Table 2, indicated a significant correlation between higher WDR4 expression
levels and higher pathological grades (p
| Clinical feature | N | WDR4 expression level | p-value | ||
| Low | High | ||||
| Gender | 0.412 | ||||
| Male | 32 | 8 | 24 | ||
| Female | 5 | 2 | 3 | ||
| Age | 0.395 | ||||
| 18 | 4 | 14 | |||
| 19 | 6 | 13 | |||
| Pathological grade | |||||
| Medium-high differentiation | 11 | 2 | 9 | ||
| Medium differentiation | 22 | 8 | 14 | ||
| Low-medium differentiation | 4 | 0 | 4 | ||
| Stage staging | 0.350 | ||||
| I | 14 | 6 | 8 | ||
| II | 2 | 0 | 2 | ||
| III | 17 | 3 | 14 | ||
| IV | 4 | 1 | 3 | ||
| T stage | 0.407 | ||||
| T1 | 15 | 6 | 9 | ||
| T2 | 2 | 0 | 2 | ||
| T3 | 8 | 1 | 7 | ||
| T4 | 12 | 3 | 9 | ||
| Vascular invasion | 0.503 | ||||
| No | 24 | 7 | 17 | ||
| Yes | 13 | 3 | 10 | ||
Using R software, 490 genes co-expressed with WDR4 were identified and analyzed through Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment. Results revealed significant roles for WDR4 in hormone metabolism, vitamin D metabolism, and organic acid transport, as well as involvement in apical plasma membrane, basement membrane, and synaptic membrane formation (Fig. 3A). Molecular function analysis indicated primary activities in channel functions, passive transmembrane transporter activities, and signaling receptor activator activities. KEGG analysis demonstrated enrichment in neuroactive ligand-receptor interactions, retinol metabolism, cytochrome P450-mediated drug metabolism pathways, and chemical carcinogenesis-DNA adducts (Fig. 3B).
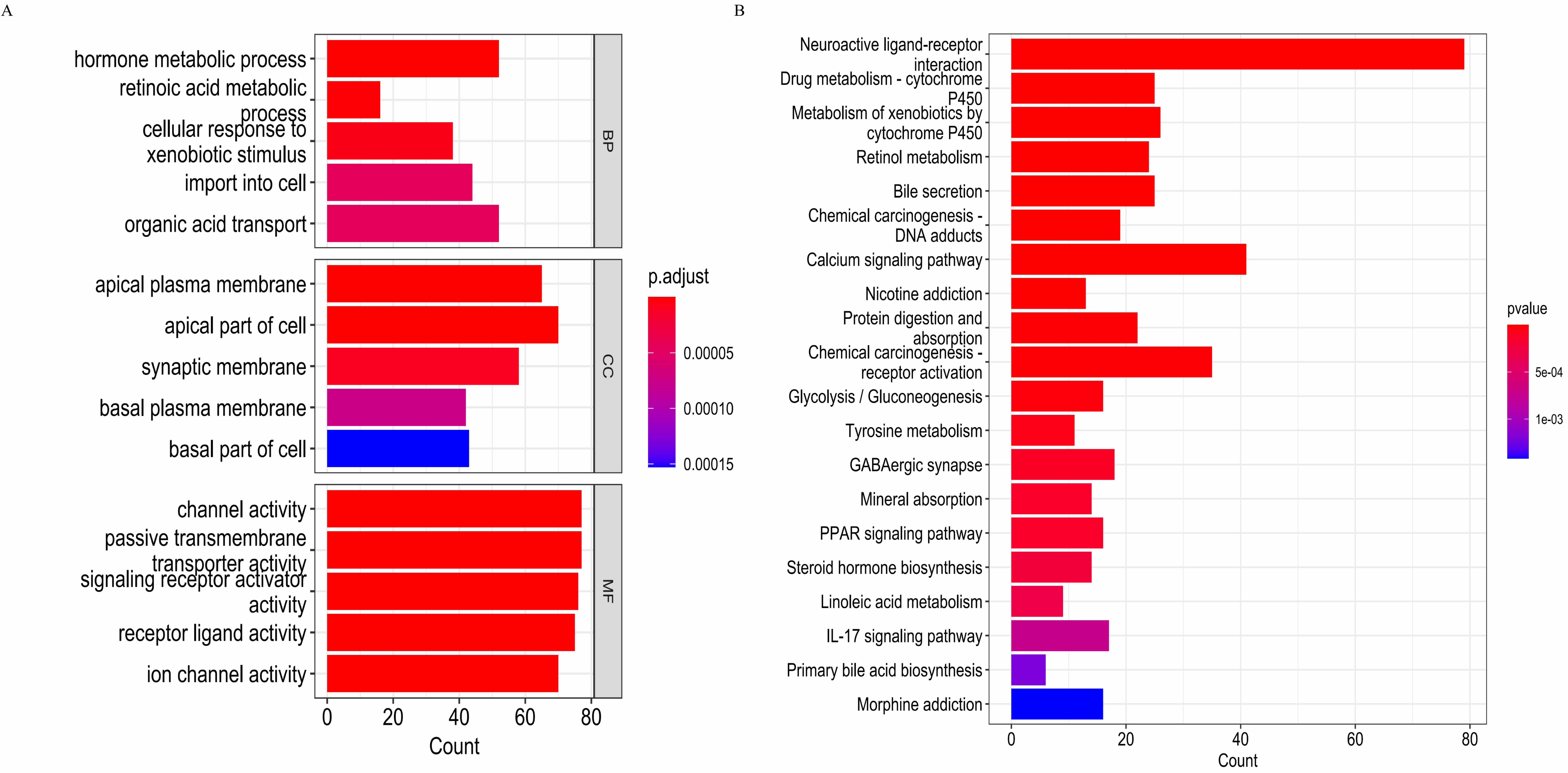 Fig. 3.
Fig. 3.
GO and KEGG of WDR4. (A) GO functional enrichment analysis of WDR4. (B) KEGG functional enrichment analysis of WDR4.
To investigate the relationship between WDR4 and immune infiltration,
the TIMER database was used to analyze correlations between WDR4 expression and immune cell infiltration in HCC (Fig. 4A). The results revealed a
positive correlation between WDR4 expression in HCC and the infiltration
levels of CD4+ T cells, B cells, macrophages, dendritic cells, lymphoid
progenitor cells, and myeloid-derived suppressor cells (p
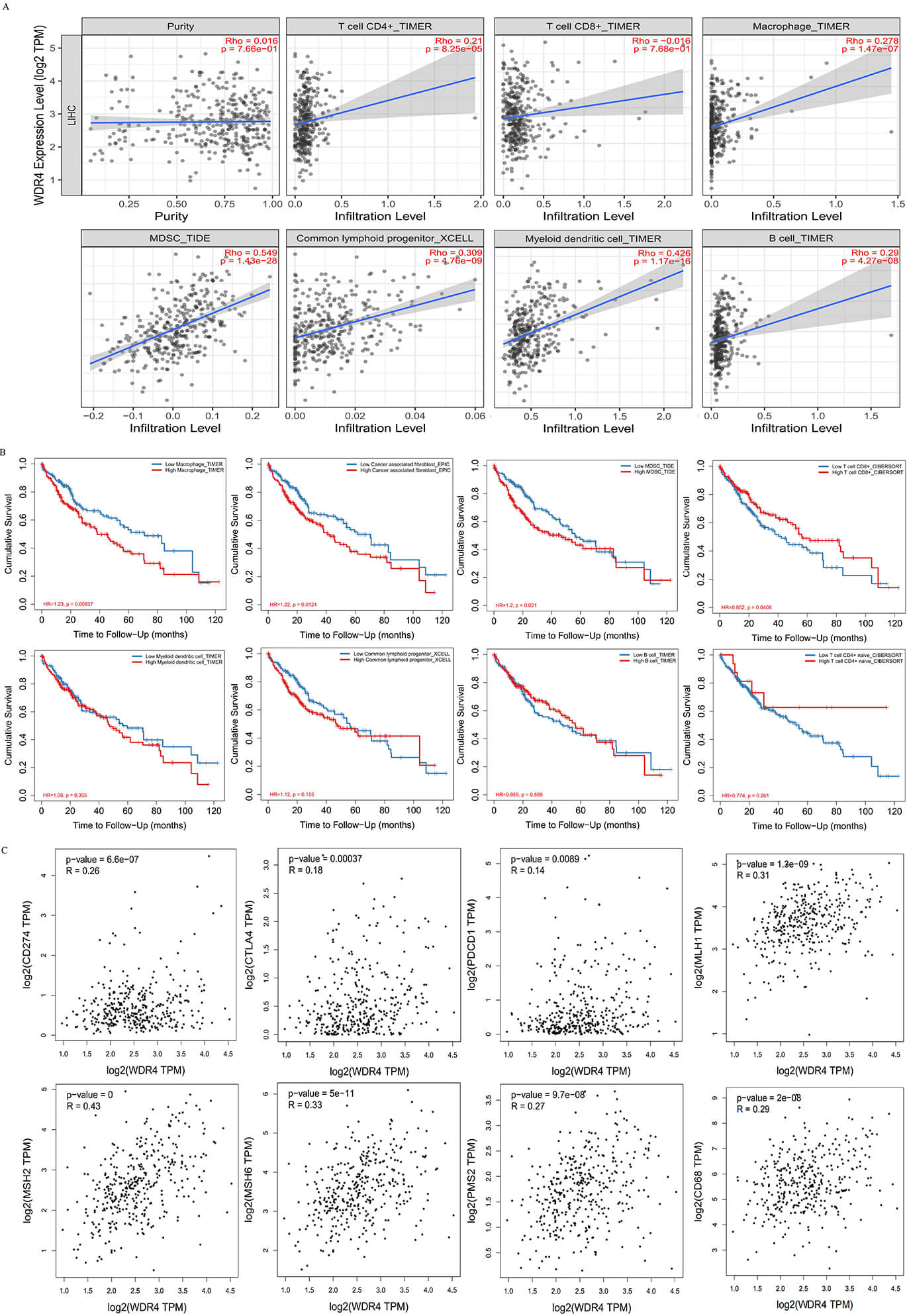 Fig. 4.
Fig. 4.
Illustration of a compelling association between WDR4 expression in HCC patients and various immune-related markers. (A) Plots showing the relationship between WDR4 expression levels and CD4+ T cells, CD8+ T cells, macrophages, myeloid inhibitor cells, lymphoid progenitor cells, dendritic cells, and B cells. (B) The cumulative survival rate of HCC patients is correlated with the degree of infiltration of macrophages, tumor-associated fibroblasts, dendritic cells, CD8+T cells, lymphoid progenitor cells, B cells, CD4+ T cells, and myeloid progenitors. (C) Correlations between WDR4 expression Levels in HCC and PD-L1, CTLA-4, PD-1, MLH1, MSH2, MSH6, PMS2, CD68 in the GEPIA Database.
In pursuit of a deeper understanding of the influence of immune cell
infiltration on cumulative survival rates in HCC, this study delved into the
relationship between immune-related cells and patients’ overall survival using
the TIMER database. Through meticulous analysis, we uncovered intricate
connections between immune cell infiltrates and the cumulative survival outcomes
of patients afflicted with HCC. As depicted in Fig. 4B, significant correlations
between immune cell infiltration and the 120-month cumulative survival rates of
HCC patients were revealed. The cumulative survival (120) was positively
correlated with CD8+ T cells (p = 0.0408) and the infiltration of
macrophages (p
We utilized the GEPIA database to investigate the interplay between
WDR4 and macrophages, as well as clinically relevant immune molecular
markers. Comprehensive analysis of WDR4 expression in HCC patients
revealed associations with immune cell-related molecular markers. Table 3 shows
significant correlations between WDR4 expression in HCC tissues and
specific macrophage-related markers. Notably, CCL2 (a tumor-associated
macrophage marker) exhibited significant relevance (p = 0.012), while
IRF5 (an M1 macrophage marker) showed substantial positive correlation
(p
| Cell type | Cell markers | Hepatocellular carcinoma | |||
| Tumour | Nontumorous | ||||
| r | p | r | p | ||
| Tumor-associated macrophages | CCL2 | 0.130 | 0.012 | 0.340 | 0.015 |
| M1 type macrophages | INOS(NOS2) | –0.055 | 0.290 | 0.032 | 0.820 |
| IRF5 | 0.250 | 0.490 | |||
| COX2(PTGS2) | 0.066 | 0.210 | 0.290 | 0.042 | |
| M2 type macrophages | CD163 | 0.230 | 0.550 | ||
| MS4A4A | 0.230 | 0.480 | |||
Our findings demonstrate a significant correlation between WDR4 expression and immune responses in HCC, particularly its substantial association with immune cells like macrophages and clinically relevant immune markers, underscoring WDR4’s pivotal role in HCC immune infiltration and immunological landscape.
To delve deeper into the intricate interplay between WDR4, immune-related markers, and macrophages, we conducted a rigorous Spearman correlation analysis on 37 HCC tissue samples. Clinical and pathological data from patients who underwent single-agent immunotherapy for HCC were collected from Hunan Provincial People’s Hospital (The First Affiliated Hospital of Hunan Normal University). The results unveiled a significant positive correlation between WDR4 expression levels and key macrophage markers, including CD68 and PD-L1. As illustrated in Table 4 and Fig. 5, the expression levels of CD68 and PD-L1 exhibited a similar trend to that of WDR4, in both patients with high and low WDR4 expression. These findings strongly suggest that WDR4 may indeed be positively associated with macrophages and PD-L1. This intricate relationship underscores the potential immunomodulatory role of WDR4 in the context of HCC.
| Statistical results | WDR4 | |
| CD68 | Spearman correlation analysis | 0.528 |
| Significance (double-tailed) | ||
| N | 37 | |
| PD-L1 | Spearman correlation analysis | 0.509 |
| Significance (double-tailed) | ||
| N | 37 |
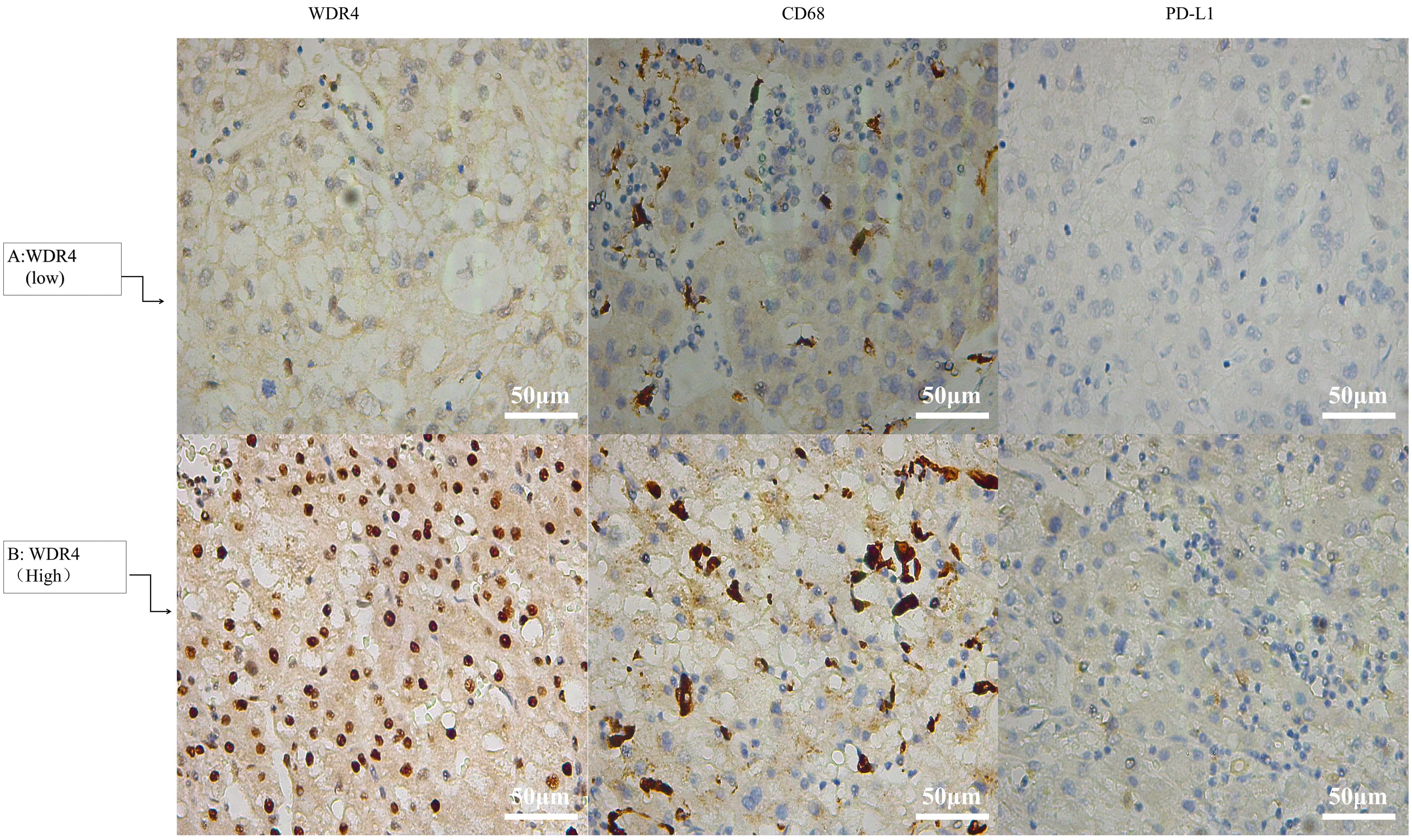 Fig. 5.
Fig. 5.
Expression levels of WDR4, CD68, and PD-L1 in HCC
tissues (400
To investigate the relationship between WDR4 expression and HCC
prognosis, we analyzed overall survival using R software, with Kaplan-Meier
curves demonstrating significantly shorter survival in
high-WDR4-expression patients versus low-expression counterparts
(n = 371, p
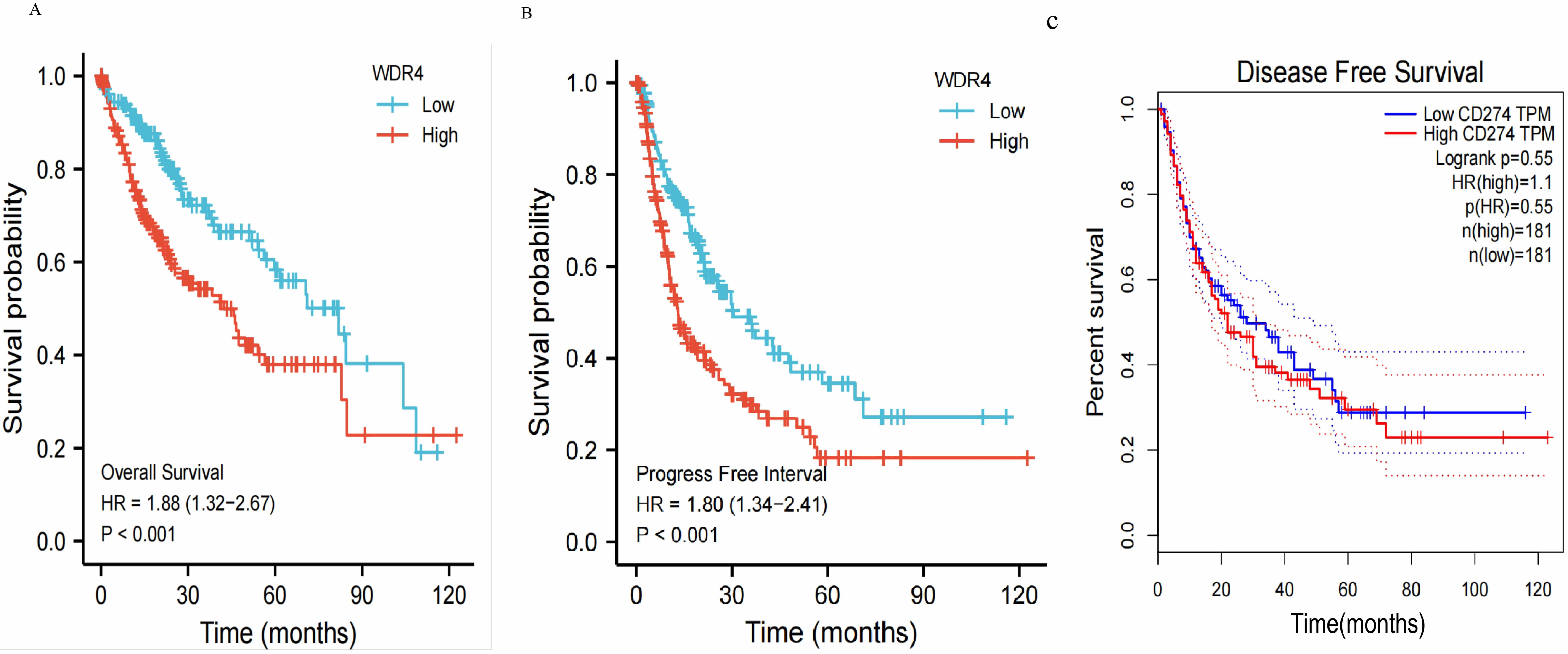 Fig. 6.
Fig. 6.
Elevated WDR4 expression correlates with poor OS and PFS clinical prognosis in HCC patients, but the expression of PD-L1 has no significant correlation with the DFS of HCC patients. (A) Kaplan-Meier survival curves for OS of WDR4 high and low expression groups versus HCC patients in the TCGA database. (B) Kaplan-Meier survival curve for PFS of WDR4 expression in HCC patients from TCGA database. (C) Kaplan-Meier survival curve for DFS of PD-L1 expression in HCC patients from GEPIA database.
In the final validation step, we analyzed data from 37 patients
post-immunotherapy with PD-1 inhibitors, where Kaplan-Meier survival analysis of
disease-free survival revealed a significant correlation between WDR4
expression levels and patient outcomes after single-agent immunotherapy (Log-rank
p
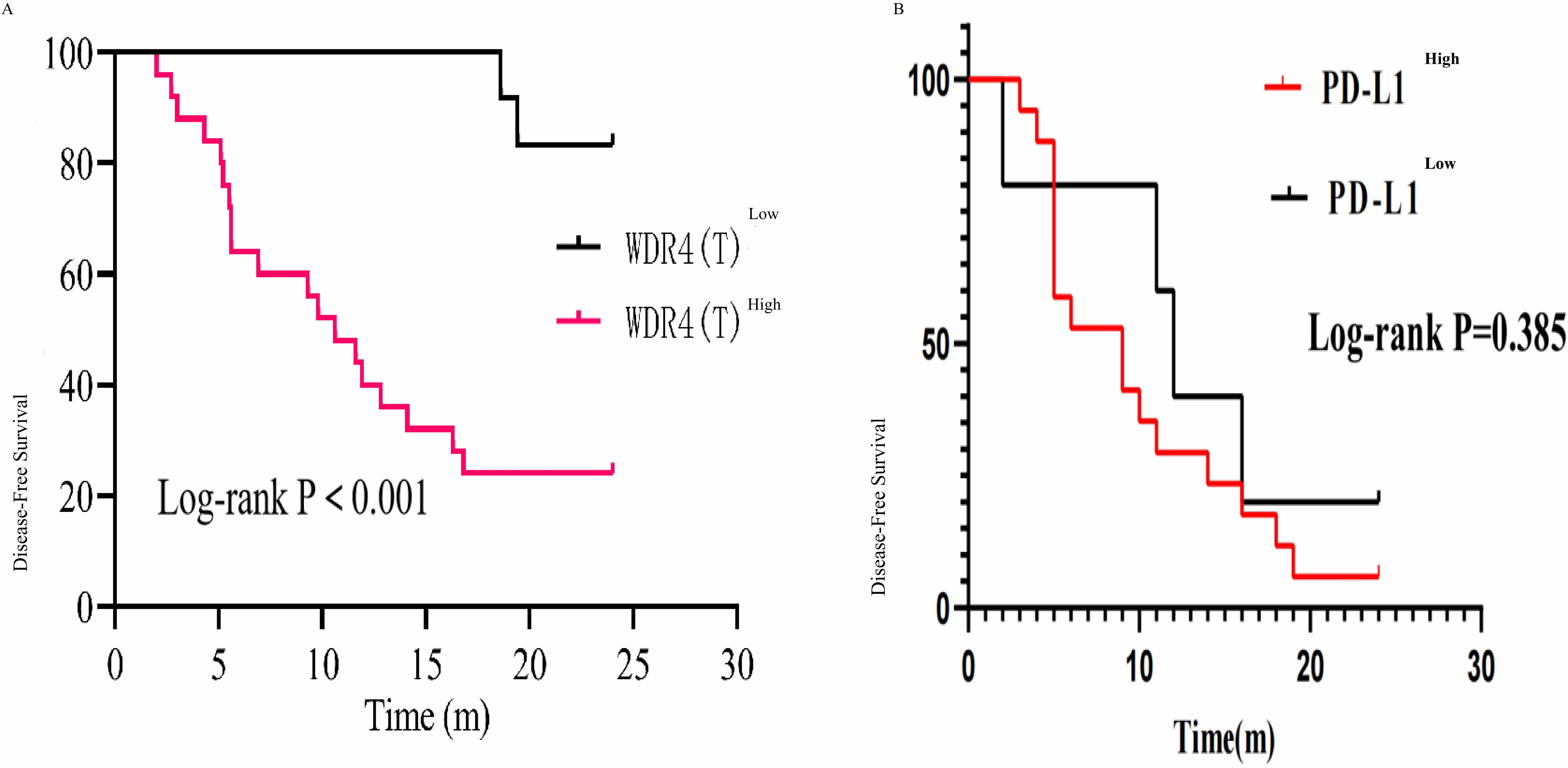 Fig. 7.
Fig. 7.
Elevated WDR4 or PD-L1 expression correlates with Disease-Free Survival (DFS) clinical prognosis in HCC patients. (A) Kaplan-Meier survival curves of high and low WDR4 expression groups and DFS in HCC tissues. (B) Kaplan-Meier survival curves for high-and low PD-L1 expression groups and DFS in HCC tissues. Data from 37 HCC samples that underwent single-agent immunotherapy were collected from Hunan Provincial People’s Hospital (The First Affiliated Hospital of Hunan Normal University).
For its highly malignant nature, insidious progression and frequent recurrence, hepatocellular carcinoma (HCC) demonstrates poor treatment outcomes, underscoring the critical importance of identifying effective diagnostic indicators and therapeutic biomarkers [37]. While alpha-fetoprotein (AFP) represents the most common serological HCC diagnostic marker [38], approximately 30–40% of patients lack AFP elevation. Conversely, nonmalignant conditions including chronic liver disease and reproductive system tumors can elevate AFP levels, limiting its diagnostic specificity [39]. Although studies link AFP to HCC immunotherapy response [40], immune tolerance and other factors preclude its utility as a reliable immunotherapy biomarker [38]. WDR4, a WD repeat protein family member, exhibits overexpression in hepatoblastoma, lung cancer, pancreatic cancer, and head/neck malignancies, implicating it in cancer initiation, progression, metastasis and adverse prognosis [18, 19, 20, 41]. WDR4 cooperates with METTL1 to maintain m7G methyltransferase activity [42], with METTL1/WDR4-mediated m7G tRNA methylation proving pivotal for mouse embryonic stem cell self-renewal and differentiation [17]. WDR4 potentially engages in protein-protein interactions and ligand binding, with triplication in trisomy 21 potentially contributing to Down syndrome phenotypes [16]. Additionally, defects in the WDR4-ARHGAP17-Rac1 signaling pathway may associate with Spearson cerebellar developmental disorders [43]. Ma et al. [19] demonstrated that WDR4/METTL1 knockdown impairs m7G tRNA modification, suppressing proliferation and invasion, whereas overexpression promotes tumorigenesis. This study reveals significantly elevated WDR4 expression in HCC versus adjacent tissues, correlating with metastasis and poor prognosis, positioning WDR4 as an oncogene with diagnostic and adverse prognostic biomarker potential in HCC.
Cancer development is recognized as an evolutionary ecological process [44]. The tumor microenvironment (TME), constituting the milieu for tumor growth, is pivotal in tumor initiation and progression. The TME comprises all non-cancerous host cells and non-cellular components, including fibroblasts and immune cells [45]. Recent studies [46, 47] highlight crucial roles for macrophages and stromal fibroblasts in tumor initiation, with the TME intricately linked to tumor formation, sustenance, and metastasis. Research on the HCC immune microenvironment is advancing, with breakthroughs in targeted angiogenic drugs; combined targeted immunotherapy outperforms sorafenib [48]. Nevertheless, the HCC drug arsenal remains limited, and drug resistance compromises overall treatment efficacy. Immunoinfiltration plays a pivotal role in tumor initiation, progression, metastasis, and drug resistance [18, 19, 20, 41, 49]. WDR4 contributes to renal cancer’s immune microenvironment, where its knockout inhibits proliferation and enhances sunitinib/sorafenib sensitivity in 786-0 and Caki-1 cells [24]. Additionally, WDR4 negatively regulates Promyelocytic Leukemia (PML) via ubiquitination, promoting immune suppression and shaping the TME to facilitate lung cancer progression [49]. Here, we observed positive correlations between WDR4 expression in HCC and infiltration levels of macrophages, tumor-associated fibroblasts, and other immune cells. Further analysis linked immune cell infiltration to cumulative 120-month survival, revealing a negative correlation for macrophages. These findings associate WDR4 with macrophage, tumor-associated fibroblast, and myeloid-derived suppressor cell infiltration in liver cancer, ultimately affecting prognosis.
Immunotherapy has become the fourth pillar of cancer treatment alongside surgery, chemotherapy and radiation [50]. Immune checkpoint inhibitors targeting PD-1, PD-L1, CTLA-4 and mismatch repair genes (MLH1, MSH2, MSH6, PMS2) represent pivotal benchmarks in cancer immunotherapy. However, monotherapy immunotherapy shows inferior efficacy to targeted therapy in first-line HCC treatment [51]. Unlike other solid tumors, studies show no established correlation between tumor cell PD-L1 expression and anti-PD-1 inhibitor response in HCC [12], potentially due to HCC’s complex immune microenvironment. The liver harbors approximately 80% of body macrophages that scan and infiltrate the vasculature [52]. Tumor-associated macrophages (TAMs), the most abundant immune infiltrates in the TME, critically influence HCC through phenotypic diversity [53]. As integral components of the immune microenvironment, macrophages contribute indispensably to innate and adaptive immunity [54]. Although typically tumoricidal, macrophages also exhibit tumor-promoting effects: CD68⁺ MI macrophage-infiltrated hepatoma cells induce PD-L1 overexpression [55], and TAMs induce immunosuppression within the HCC TME [56]. Despite expanding research on HCC macrophages, effective immunotherapy biomarkers remain elusive. Recent pan-cancer analyses reveal aberrant WDR4 expression correlating with immune cell infiltration across tumors [57], while Li et al. [58] found that high expression of m7G core genes is associated with poor prognosis in HCC. Additionally, by constructing an immune escape-related protein-protein interaction (PPI) network, they discovered that (METTL1, WDR4) and 19 mRNA risk signature genes are related to immune escape. This study identifies significant associations between WDR4 and macrophages in HCC, particularly TAMs. Immunohistochemistry confirms significant positive correlations between WDR4 expression and both PD-L1 and CD68 levels. Notably, PD-L1 expression showed no significant correlation with DFS under postoperative immunotherapy monotherapy, whereas elevated WDR4 expression corresponded to shorter DFS in immune monotherapy recipients, suggesting WDR4 as a potential HCC immunotherapy indicator.
WDR4 exhibits elevated expression levels within HCC tissues and is associated with immune infiltration, which establishes it as a prognostic biomarker in HCC. Furthermore, the positive correlation observed between WDR4, CD68, and PD-L1 underscores its potential as a guiding factor in immunotherapeutic approaches for HCC.
The datasets supporting the findings of this study are available from the corresponding author upon reasonable request. Any further inquiries regarding methods or analysis details will also be provided by the authors upon request.
SSF and LYY conceived and designed the study. SSF and LYY as well as acquisition and management funds. LYY, SSF, ML, JW, YZ, MM, JJF captured, collected, and extracted the data contained in this analysis. SSF and JJW analysis data. LYY originally drafted the paper. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
In this study, 37 tissue samples (including tumor and adjacent tissues) and clinical data from cancer patients (HCC) who received postoperative anti-PD-1 monotherapy were approved by the Ethics Committee of Hunan Provincial People’s Hospital (The First Affiliated Hospital of Hunan Normal University) (Ethics Approval No. 2019-19). This study was conducted in accordance with the guidelines of the Declaration of Helsinki, and informed consent was obtained from all patients. Furthermore, immunohistochemical staining to evaluate the expression of PD-L1 and CD68 in cancer tissues and adjacent tissues within WDR4 has been approved by the Ethics Committee of Hunan Provincial People’s Hospital (The First Affiliated Hospital of Hunan Normal University) (Ethics Approval No. 2024-402).
We would like to express our gratitude to all those who helped us during the writing of this manuscript, and thanks to all the peer reviewers for their opinions and suggestions.
This work was supported by the Hunan Province department unit fund (2023JJ60019).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







