1 Research Center for High Altitude Medicine, State Key Laboratory of Plateau Ecology and Agriculture, Qinghai University Medical College, 810001 Xining, Qinghai, China
2 Hepatopancreatobiliary Surgery Department, The Research Key Laboratory for Echinococcosis of Qinghai Province, Qinghai University Affiliated Hospital, 810001 Xining, Qinghai, China
3 Center of Molecular Pathology, Qinghai Provincial People’s Hospital, 810001 Xining, Qinghai, China
4 Department of Infection Control, Qinghai Branch Hospital of the First Affiliated Hospital of Tianjin University of Traditional Chinese Medicine, 810001 Xining, Qinghai, China
Abstract
Alveolar echinococcosis (AE) is a serious zoonotic parasitic disease. This study aimed to investigate the mechanisms underlying the formation of the dense fibrotic band surrounding hepatic alveolar echinococcosis (HAE) lesions, which impedes chemotherapeutic drug penetration. Additionally, the roles of Interleukin-33 (IL-33) and eosinophils in the progression of fibrosis within this band were examined.
IL-33/suppression of tumorigenicity 2 (ST-2) expression levels were compared between patients with HAE and healthy controls, as well as between close to lesion tissues (CLT) and distant from the lesion tissues (DLT) using enzyme-linked immunosorbent assay (ELISA), western blot, and immunohistochemistry. Immunofluorescence co-localization analysis was performed to examine IL-33/ST-2 and eosinophil distribution. Masson’s trichrome staining was used to evaluate fibrosis in AE lesions. Cellular assays were carried out to assess the effects of IL-33 on eosinophil phagocytosis and migration, as well as its impact on α-smooth muscle actin (α-SMA) expression in hepatic stellate cells (HSCs).
ELISA findings indicated significantly elevated serum IL-33/ST-2 levels in patients with AE compared with healthy controls (p < 0.05). Immunohistochemistry and western blot analyses demonstrated higher IL-33/ST-2 expression in CLT than in DLT (p < 0.05), with IL-33/ST-2 and eosinophils exhibiting a highly consistent distribution within CLT. Masson’s trichrome staining confirmed increased fibrosis in CLT. Cellular assays showed that IL-33 enhanced eosinophil phagocytosis and migration, while IL-33 stimulation upregulated α-SMA expression on the HSC surface, with this effect being more pronounced in the presence of eosinophils.
IL-33 contributed to microenvironmental fibrosis within AE lesions via eosinophil-mediated mechanisms, highlighting a potential therapeutic target to improve chemotherapy efficacy in patients with AE.
Keywords
- alveolar echinococcosis
- eosinophils
- hepatic fibrosis
- IL-33/ST-2 signaling pathway
- lesion microenvironment
Alveolar echinococcosis (AE) is a zoonotic parasitic disease caused by the larval stage of Echinococcus multilocularis, predominantly found in the Northern Hemisphere, particularly in China, Central Asia, Russia, Europe, and North America [1, 2]. AE poses a significant threat to human health and has substantial implications for livestock development, primarily affecting the liver [3]. The primary management strategies for AE include imaging-based diagnosis and surgical removal of the parasitic lesion. Benzimidazole derivatives, particularly albendazole, are used as adjunctive therapy; however, their long-term administration is associated with adverse side effects [4, 5].
Immunological studies indicate a complex interaction between the host immune system and the parasite, influencing disease progression [6, 7]. AE research has found that there is a delicate balance between the host’s self-protection and the growth of the parasite body, in which the immunological regulatory mechanism plays an important role [6]. Eosinophils, as key innate immune cells, play diverse roles in parasitic infections and are regulated by interleukin-5 (IL-5) and the receptor siglec-8 [7, 8]. Interleukin-33 (IL-33) functions as both a nuclear factor and a cytokine involved in immune regulation. ST-2, as the receptor of IL-33, is widely distributed on the surface of eosinophils. The mRNA encoding ST-2 generates three splice variants: sST-2 (soluble form), ST-2L (transmembrane form), and ST-2V (variant form), with sST-2 functioning as a decoy receptor that downregulates IL-33 activity. A previous study has indicated that polymorphisms in the IL-33 and ST-2 genes correlate with increased eosinophil levels in humans [9].
In vivo studies have demonstrated that IL-33 induces eosinophilia by activating eosinophils. As primary effector cells in allergic and inflammatory diseases, eosinophils exhibit increased superoxide production, enhanced adhesion molecule expression, and prolonged survival [10, 11, 12]. Consequently, IL-33 plays a critical role in basophil- and eosinophil-mediated allergic diseases and inflammatory responses. Elevated serum IL-33 levels in acute schistosomiasis suggest its pro-inflammatory role during the acute phase, with levels closely correlating with eosinophil counts and disease progression [13]. Nine weeks following a single-dose praziquantel treatment, patients with schistosomiasis exhibit significantly elevated serum IL-33, along with elevated eosinophil counts, IL-13 levels, and enhanced ST-2 expression on eosinophils [14]. Studies have found that eosinophils are involved in the occurrence of allergic reactive diseases such as asthma and allergic conjunctivitis mainly through the binding of IL-33 to the ST-2 receptor on the cell membrane surface, thereby promoting the proliferation, activation and degranulation effect of eosinophils [15]. IL-33 was significantly elevated in the serum of patients with Schistosomiasis japonicum and infected mice. Moreover, studies have shown that eosinophil count and the course of schistosomiasis or other inflammatory diseases are closely related to IL-33 levels [13, 16, 17]. In a Schistosoma mansoni egg-induced mouse lung granuloma model, ST-2 deficiency resulted in reduced production of T-helper type 2 (Th2) cytokines, including interleukin-4 (IL-4) and interleukin-5 (IL-5), as well as decreased eosinophil infiltration within granulomas [18]. Study on filariasis have demonstrated that IL-33/ST-2 signaling is essential for the splenic clearance of microfilariae from peripheral blood [19]. From what has been discussed above, we know the IL-33/ST-2 signaling pathway thus exerts diverse biological effects across various infection models, tissues, and disease stages, thereby influencing disease progression and outcomes.
Hematoxylin-eosin (HE) and Masson’s trichrome staining of AE lesions reveals dense inflammatory fibrous bands that, while limiting lesion expansion, also hinder drug penetration, posing challenges for chemotherapy [20, 21, 22]. In the context of fibrosis, IL-33 has been identified as a key regulator of fibrosis development. Hepatic stellate cells (HSCs), the primary collagen-producing cells in liver injury, represent the main source of IL-33 [23]. Studies have indicated that IL-33 expression is upregulated in both mouse and human hepatic fibrosis [24, 25]. Research indicates that elevated IL-33 expression alone can drive extensive collagen fiber formation and extracellular matrix (ECM) deposition, whereas IL-33 gene-deficient mice exhibit significantly reduced hepatic fibrosis [26]. These findings highlight the crucial role of IL-33 in liver fibrosis.
Therefore, in order to explore how IL-33 induces peripheral fibrosis in AE lesions and verify whether IL-33 changes the fibrosis process of AE lesions by altering eosinophil behavior, we conducted the following experiments. enzyme-linked immunosorbent assay (ELISA), immunohistochemical staining, and Western blot analysis were performed to assess ST-2 and IL-33 protein expression in the tissue surrounding AE lesions. Multiple immunofluorescence co-localization analysis was conducted to examine the co-expression of eosinophils with these proteins, while Masson’s trichrome staining was used to quantify hepatic fibrosis within AE lesions. Additionally, in vitro cell experiments were conducted to investigate the relationship between IL-33, eosinophils, and fibrosis. These methodologies facilitated a detailed characterization of the ST-2/IL-33 signaling pathway in local immune responses and fibrotic progression in echinococcosis through eosinophil activation, providing a theoretical basis for potential advancements in prevention and treatment strategies.
The included subjects were all patients with hepatic alveolar echinococcosis
diagnosed for the first time, who met the requirements of surgery and did not
undergo drug chemotherapy. The tissues were mainly from the surgical specimens of
AE patients, and a total of 21 tissue specimens were collected. All 21 (12
females/9 males, mean age was 32 years) AE patients were initially diagnosed by
B-ultrasound and were eligible for surgery for the first time. The surgically
resected tissues of patients undergoing AE surgery have a complete structure,
including lesion tissues, close to lesion tissues (
50 mL of healthy volunteers’ venous blood was diluted with PBS 1:1, and the
diluent was slowly added into the centrifuge tube containing Percoll separation
solution (Solarbio, Beijing, China, density 1.130
The lesions of Echinococcus multilocularis were isolated from Mongolian gerbils infected with Echinococcus multilocularis preserved in our laboratory. Mongolian gerbils were sacrificed (After anesthesia with isoflurane (concentration: 1.5–2%, Continuous inhalation was maintained for 2–3 minutes to achieve adequate anesthesia prior to the procedure), euthanasia was performed by cervical dislocation (neck fracture) to reduce the suffering of experimental animals), and the metacestode tissues were isolated from the abdominal cavity, and an appropriate amount of sterile normal saline was added to wash the blood water on the cyst surface repeatedly. After being crushed by a tissue homogenizer (OSE-Y50, TIANGEN, Beijing, China), it was centrifuged at 4 °C and 12,000 rpm for 20 min. The supernatant was filtered through a sterile filter (0.22 µm, Cat. #: SLGP033RB, Merck Millipore, Darmstadt, Germany), and the protein was quantified to 20 mg/mL by BCA (Cat. #: A55860, Thermo Fisher, Waltham, MA, USA) and stored at –80 °C. E. multilocularis antigen (E.m-Ag) was prepared for intervention in vitro. All animal experimental procedures were also approved by the Ethics Committee of Qinghai University Affiliated Hospital (Approval No.: P-SL-2019039). All efforts were made to minimize animal suffering and reduce the number of animals used in accordance with the ARRIVE guidelines.
Blood samples were obtained from 12 individuals with hepatic alveolar echinococcosis (HAE) (From patients diagnosed with HAE by B-ultrasound) and 12 healthy control participants (Healthy volunteers from the physical examination center) after obtaining informed consent. Samples were collected using anticoagulated tubes and plasma was separated by centrifugation at 2300 g for 15 minutes. The plasma samples were then stored at –80 °C. ELISA kits (Cat. #: JL19282-48T/JL18366-48T, J&L Biological, Shanghai, China) were used to measure serum levels of secreted ST-2 and IL-33, with a standard curve correlation coefficient of R2 = 0.998.
Tissue samples (n = 15) were obtained from individuals with HAE, including close
to lesion tissues (
Surgically excised lesion tissues (n = 15) were fixed in 4% formaldehyde, dehydrated, embedded in paraffin, and sectioned into 3 µm slices. The sections were baked at 60 °C for 30 to 45 minutes, followed by sequential deparaffinization and rehydration using xylene I and II (15 minutes each), absolute ethanol I and II (5 minutes each), 95% ethanol I and II (5 minutes each), 90% ethanol I and II (5 minutes each), and 80% ethanol I and II (5 minutes each). Antigen retrieval was performed using high-pressure or microwave treatment for 10 to 15 minutes in 0.5M EDTA (pH 8.0) retrieval solution. Target areas were circled using a histochemical pen, followed by sequential incubation with an endogenous peroxidase blocker and a non-specific staining blocker (serum or 5% skimmed milk) for 10 minutes at room temperature. Primary antibody (antibody concentration: IL-33 (1:800), ST-2 (1:500)) incubation occurred overnight at 4 °C, followed by secondary antibody incubation at 37 °C for 1 hour after reaching room temperature. Streptavidin-peroxidase was applied for 10 minutes. Each step was followed by three washes with PBS for 3 minutes. DAB (Cat. #: YT075, Biolab, Beijing, China) was used for color development, followed by hematoxylin counterstaining, with staining duration monitored under a microscope (TISSUEFAXS SPECTRA, TissueGnostics, Vienna, Austria). Slides were rinsed with tap water for bluing, sealed, and examined. The following antibodies were utilized: anti-ST-2 antibody (ab25877) (1:500, Abcam, Cambridge, UK) and anti-IL-33 antibody (ab207737) (1:800, Abcam, Cambridge, UK).
Pathological tissue (n = 7) processing, deparaffinization, hydration, and antigen retrieval were conducted as described in the immunohistochemistry protocol. Following antigen retrieval, tissue sections were immersed in a blocking solution for 10 minutes. Primary antibody (antibody concentration: IL-33 (1:2000), ST-2 (1:50)) incubation was carried out for 1 hour at room temperature or overnight at 4 °C, followed by secondary antibody incubation for 10 minutes. Fluorescent dye was applied at room temperature for 10 minutes, followed by antigen retrieval. For multiple fluorescence staining, each staining round was verified using a fluorescence microscope before proceeding to the next step. After all staining procedures were completed, nuclear staining was performed with DAPI working solution at 37 °C for 5 minutes. Slides were mounted with an anti-fading fluorescence quenching mounting medium and examined under a fluorescence microscope (Cytation5, BioTek, Winooski, VT, USA). Eosinophils were defined as CD66⁺CD16⁻ cells. The following antibodies were used: anti-ST-2 antibody (ab25877), anti-IL-33 antibody (ab207737) (Abcam, Cambridge, UK), CD66 (Cat. #: MA5-48180, Thermo Fisher, MA, USA), and CD16 (Cat. #: MA5-36143, Thermo Fisher, MA, USA).
Masson’s trichrome staining was conducted using a staining kit (Cat. #: G1340, Solarbio, Beijing, China). Tissue (n = 15) processing, including hydration, was performed as described for immunohistochemistry. Sections were stained with Weigert’s iron hematoxylin for 5 to 10 minutes and differentiated with acidic ethanol for 1 to 2 seconds. Masson’s blue solution was applied for blue staining, followed by ponceau S staining for 5 to 10 minutes and immersion in phosphomolybdic acid solution for 1 to 2 minutes. After aniline blue staining for 1 to 2 minutes, sections were washed with a weak acid working solution for 1 minute. The sections were then dehydrated, cleared, mounted, and analyzed.
Eosinophils (EOL-1) were cultured in RPMI-1640 complete medium until
stabilization. After cell counting, a well-mixed cell suspension was transferred
to the upper chamber of Transwell plates (Cat. #: 140620, Thermo Fisher, USA)
(450 µL per well, 3
EOL-1 (5
Human hepatic stellate cells (HSCs) LX-2 (Cat. #: CL-0560, Procell Life Science
& Technology Co., Ltd., Wuhan, Hubei, China) were cultured in DMEM (Cat. #:
PM150210, Procell Life Science & Technology Co., Ltd., Wuhan, Hubei, China)
medium until stabilization. After digestion, cells (5
Immunohistochemistry images were analyzed using the TISSUE CONSTICS Analysis
System (version 7.1; StrataQuest, Austria, https://tissuegnostics.com), following
panoramic scanning. Multiple immunofluorescence and Masson’s trichrome staining
were analyzed using ImageJ (version 1.53; NIH, Bethesda, MD, USA,
https://imagej.nih.gov/ij/). Data are expressed as mean
Histopathological examination of HAE lesions indicated a distinctive
inflammatory fibrous band surrounding the lesion, forming a unique
microenvironment with diverse biological functions. To investigate the potential
role of IL-33 and ST-2 expression in this region, their expression levels were
assessed in tissue sample areas close to the lesion (CLT) (
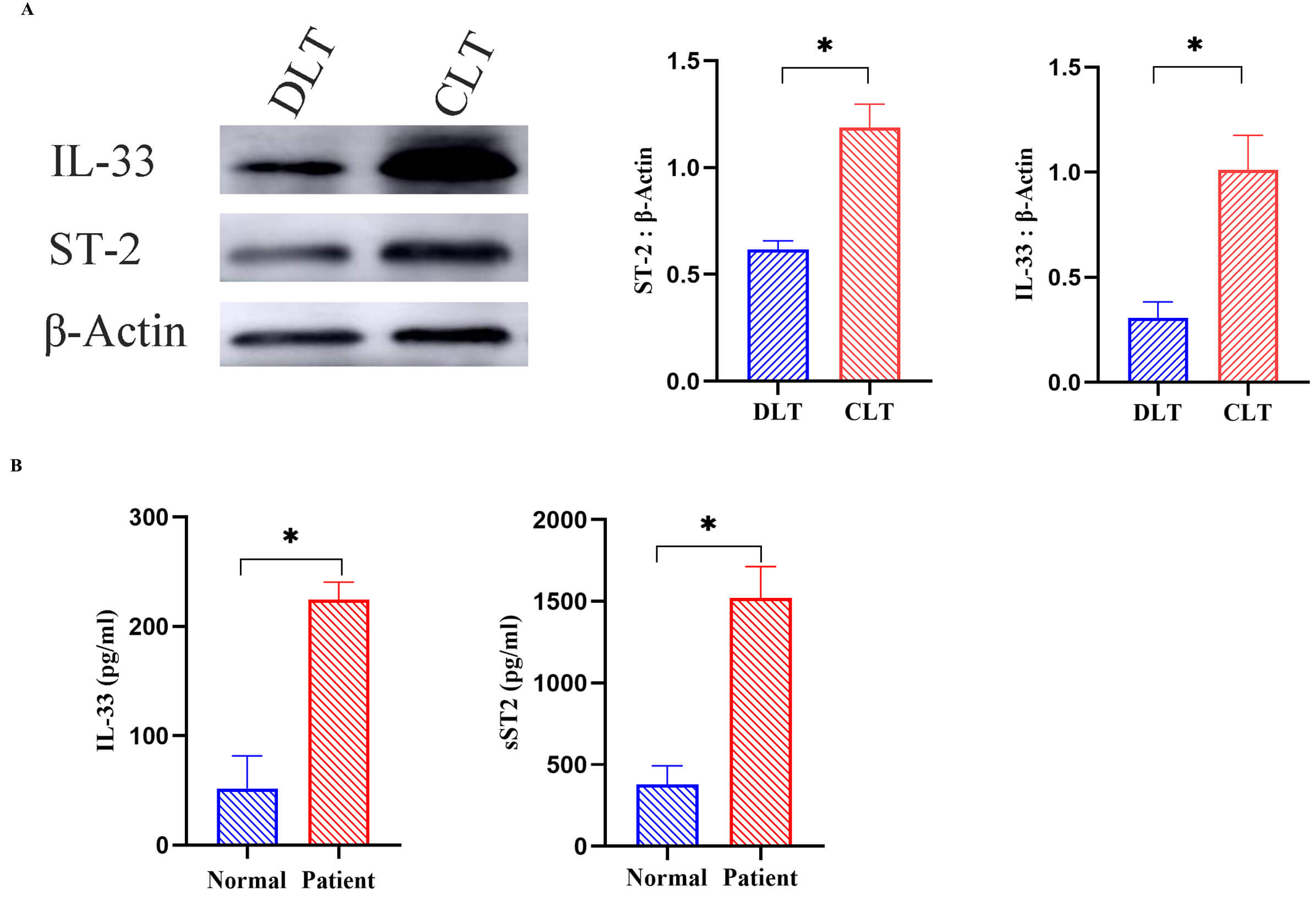 Fig. 1.
Fig. 1.
Western blot analysis of IL-33 and ST-2 expression in
CLT and DLT (n = 15), along with ELISA quantification of serum IL-33 and sST-2
levels in patients with AE and healthy controls (n = 12). (A) Western blot
analysis of IL-33 and ST-2 expression in CLT and DLT (n = 15) with grayscale
intensity quantified using ImageJ. (B) ELISA-based evaluation of serum IL-33 and
ST-2 levels in patients with AE and healthy controls (n = 12). Data are expressed
as mean
IL-33 is widely recognized as an “alarm” cytokine in inflammatory diseases. To
assess its systemic involvement in HAE, plasma levels of IL-33 and sST-2
expression were measured in patients with AE and compared to healthy individuals
using ELISA. As shown in Fig. 1B, plasma concentrations of sST-2 and IL-33 were
significantly higher in AE patients than in healthy individuals (p
Western blot analysis indicated elevated IL-33 and ST-2 expression in CLT. To
further investigate the spatial distribution of IL-33 and ST-2,
immunohistochemical staining was performed on tissue samples from both CLT and
distant from the lesion tissue (DLT) in AE patients. After panoramic scanning,
analysis was conducted using the TISSUE CONSTICS Analysis System (Fig. 2). The
software classified negative cells (green) and positive cells (red) in DLT and
CLT for statistical analysis. Results demonstrated that the proportion of IL-33
(Fig. 2B) and ST-2 (Fig. 2A) positive cells in CLT was significantly higher than
in DLT (p
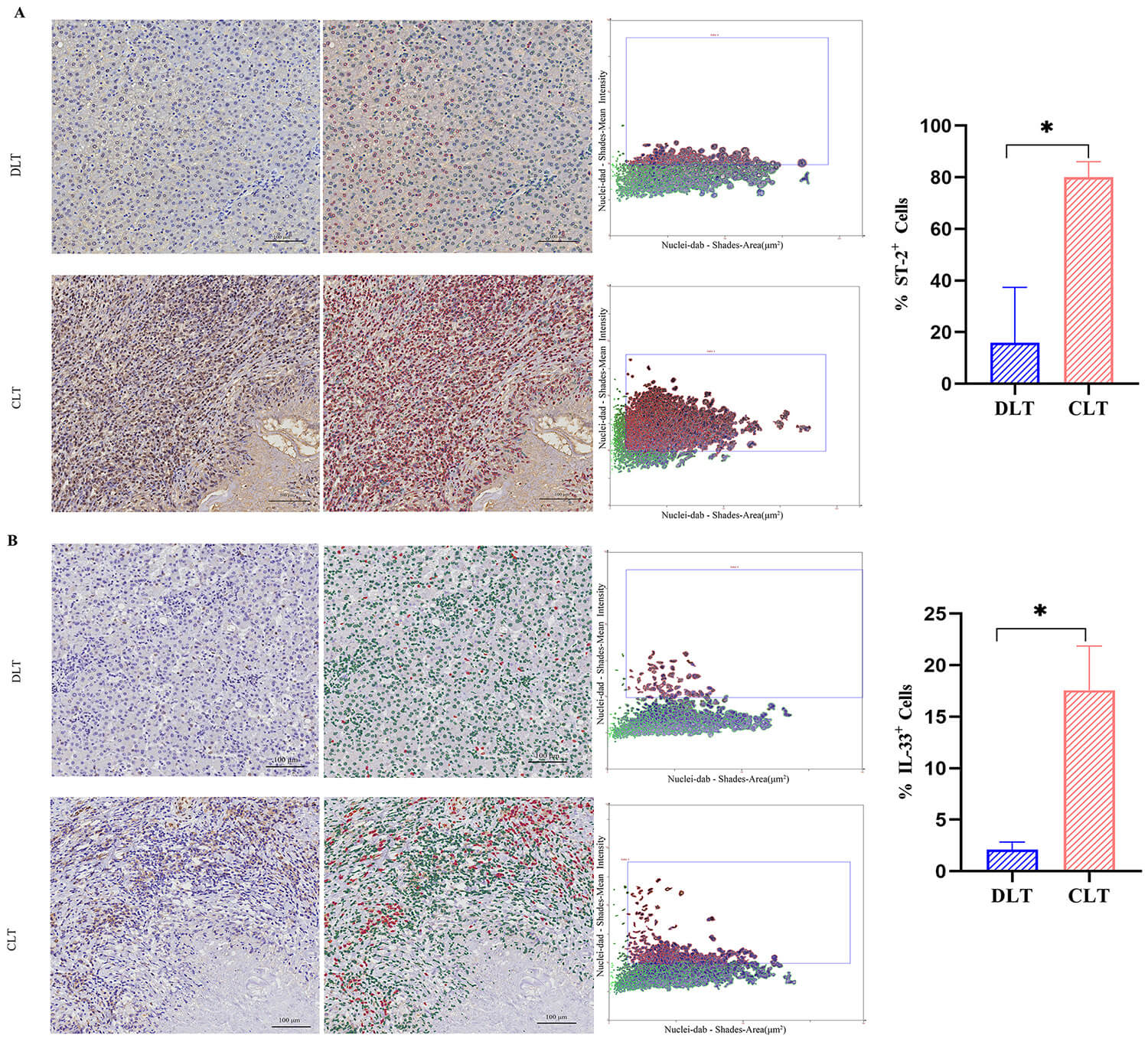 Fig. 2.
Fig. 2.
Immunohistochemical analysis of IL-33 and ST-2
expression in CLT and DLT (n = 15). (A) Representative immunohistochemical
images showing ST-2 expression in DLT and CLT. (B) Immunohistochemical images
depicting IL-33 expression in DLT and CLT. Quantification was performed using the
TISSUE CONSTICS Analysis System, which identified and measured negative cells
(green) and positive cells (red). Scale bar: 100 µm. Data are expressed as
mean
Eosinophils play a crucial role in various parasitic infections, yet their role
in AE remains unclear. Since ST-2, the receptor for IL-33, is widely expressed on
eosinophils, this study investigated the co-localization of IL-33 and ST-2 with
eosinophils within AE lesions. Immunofluorescence staining (Fig. 3) showed higher
IL-33 and ST-2 expression (green) in CLT compared to DLT, aligning with
immunohistochemistry findings (Fig. 2). Furthermore, analysis of IL-33 expression
and eosinophil distribution (Fig. 3A) (yellow) exhibited a strong correlation.
The color distribution in the deviation plot generated by ImageJ analysis was
relatively uniform (Fig. 3B), indicating a high degree of co-localization between
the two groups (R = 0.6835). This co-localized expression in CLT was
significantly higher than in DLT (p
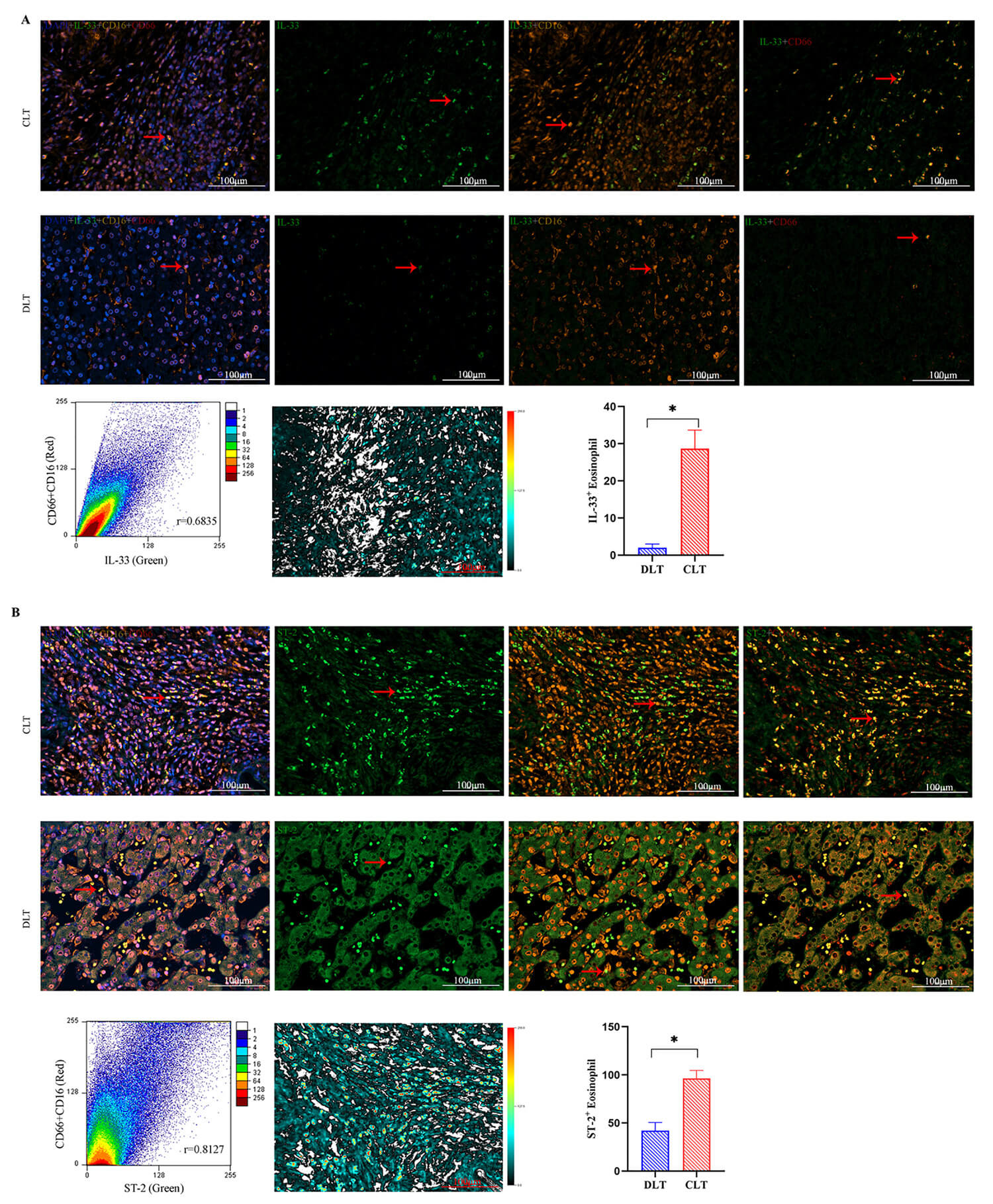 Fig. 3.
Fig. 3.
Immunofluorescence analysis of ST-2 and IL-33
expression in DLT and CLT and their co-localization with eosinophils (n = 7).
(A) Representative immunofluorescence images showing IL-33 and eosinophil
co-expression, with yellow indicating regions of overlap. (B) Immunofluorescence
images depicting ST-2 and eosinophil co-expression, with yellow indicating
co-localization. Quantification of ST-2 and IL-33 expression, along with their
co-expression with eosinophils, was performed using Image J. Error analysis plots
illustrate the extent of ST-2 and IL-33 co-localization with eosinophils. Scale
bar: 100 µm. Data are expressed as mean
IL-33 plays a key role in fibrosis development and was found to be highly
expressed in the tissues surrounding AE lesions (CLT). Given that HAE is
characterized by a dense inflammatory fibrous band, fibrosis formation in AE
lesions was analyzed. HE staining demonstrated a relatively preserved structure
in the DLT compared to CLT (Fig. 4). Masson’s trichrome staining further
demonstrated a significantly higher abundance of collagen fibers (blue) in CLT
than in DLT. Quantitative analysis using ImageJ software allowed for the
identification, isolation, and extraction of collagen fibers (blue) and tissue
(red), from which the collagen volume fraction (CVF) was calculated. The results
indicated a significantly higher CVF in CLT than in DLT (p
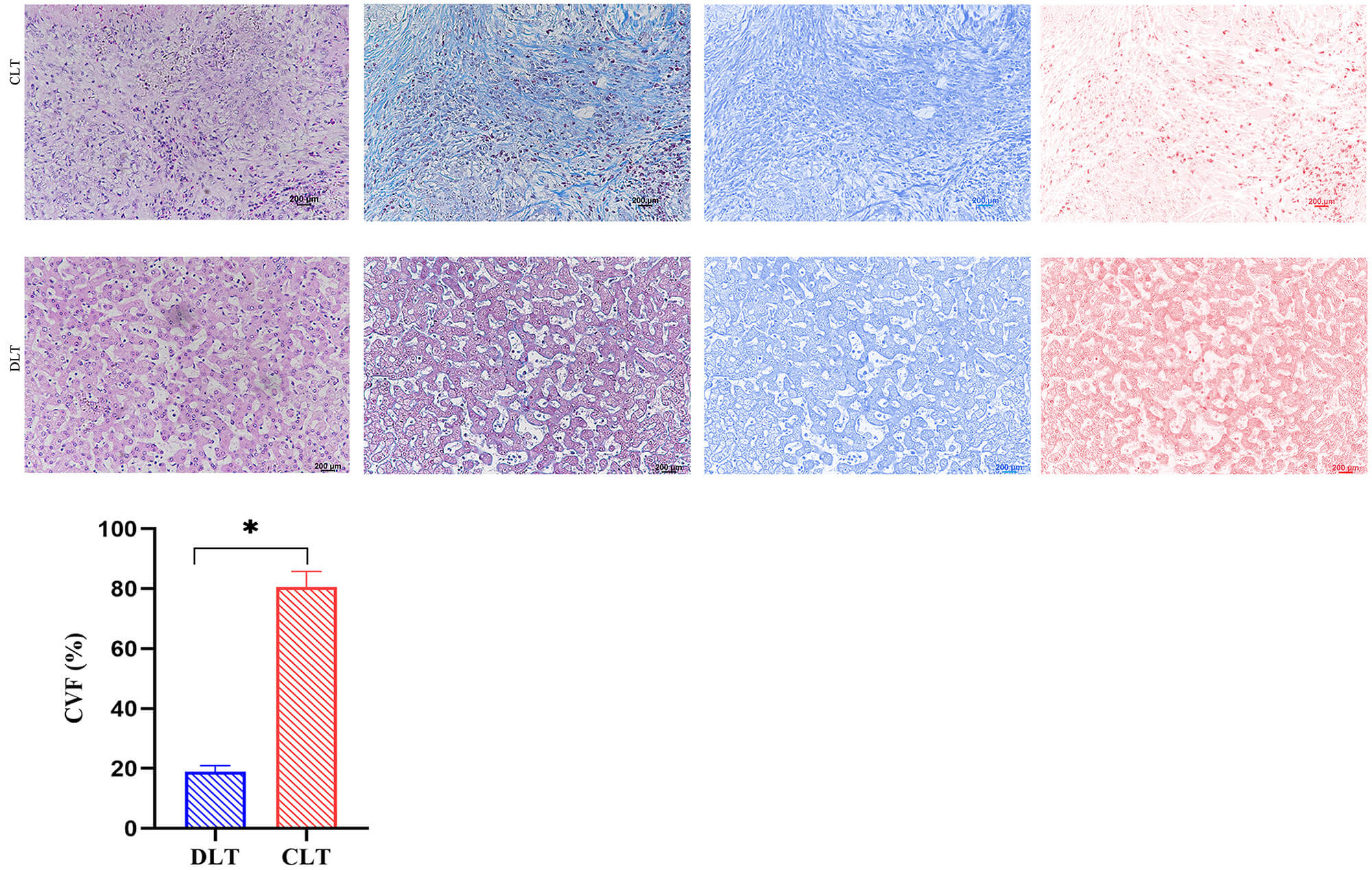 Fig. 4.
Fig. 4.
Hematoxylin-eosin (HE) and Masson’s trichrome staining
analysis of CLT and DLT (n = 15). Representative HE and Masson’s
trichrome-stained images of CLT and DLT. Quantitative analysis using Image J
software, which identified and extracted collagen fibers (blue) and tissue
structures (red). Fibrosis levels in CLT and DLT were quantified by calculating
the collagen volume fraction (CVF). Scale bar: 200 µm. Data are expressed
as mean
To assess the effect of IL-33 on eosinophil phagocytic function, eosinophils
were divided into three groups: blank control, E.m-Ag, and IL-33
intervention. Flow cytometry results (Fig. 5) indicated that eosinophil
phagocytosis of E. coli was significantly enhanced following stimulation
with IL-33 and E.m-Ag, with IL-33 inducing a more pronounced increase in
eosinophil phagocytic activity compared to the E.m-Ag stimulation
(p
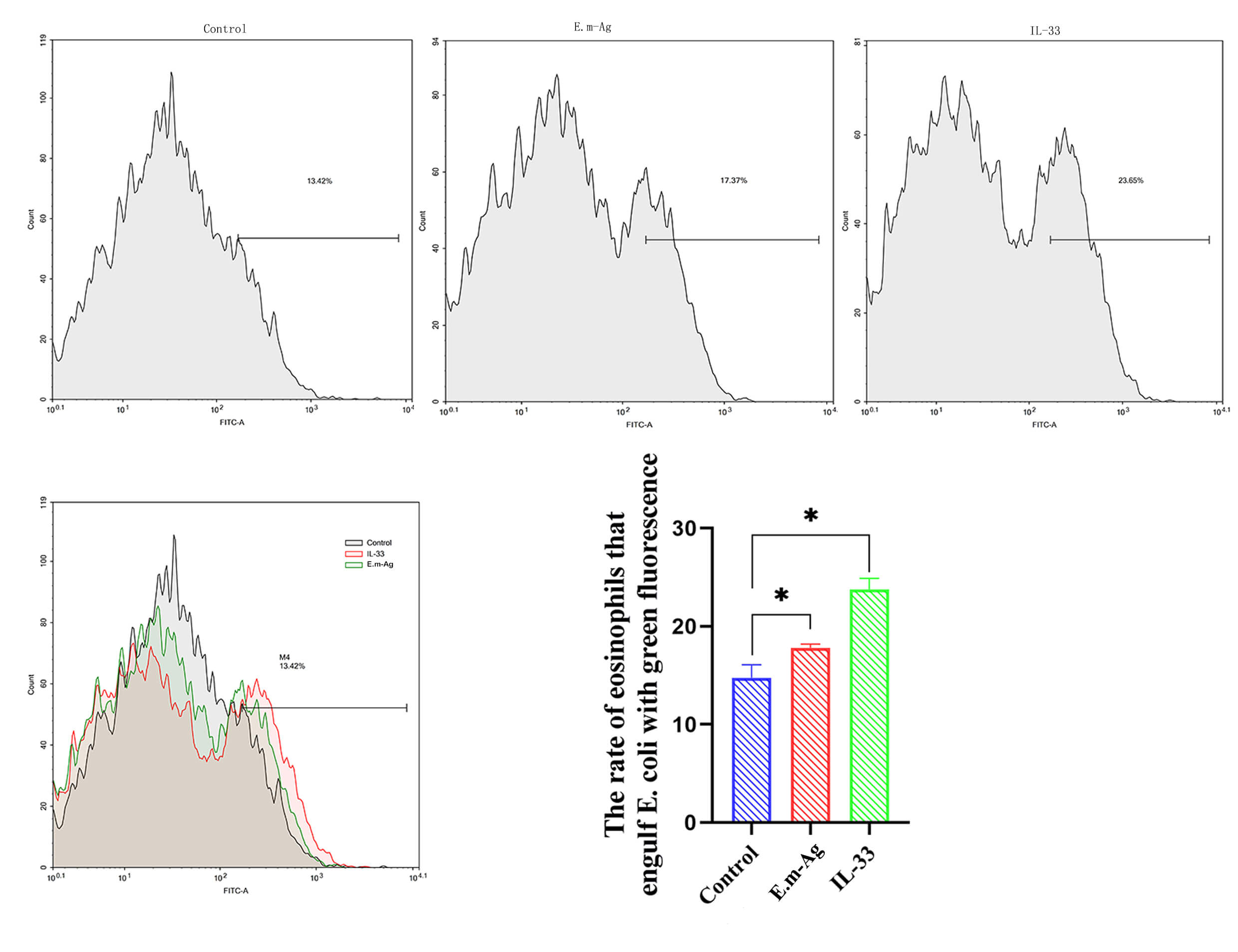 Fig. 5.
Fig. 5.
Eosinophil phagocytosis of green fluorescent E.
coli following different treatments. Phagocytosis assay results for the control
group (PBS, 50 µL/mL; n = 5), IL-33-treated group (3 ng/mL; n = 5), and
E.m-Ag-treated group (20 mg/mL; n = 5). Flow cytometry to detect GFP
signals in eosinophils. Data are expressed as mean
To evaluate the effect of IL-33 on eosinophil migration, a Transwell assay was
performed. The results demonstrated that IL-33 intervention significantly
increased eosinophil migration through the Transwell chamber compared to both the
blank control and E.m-Ag groups (p
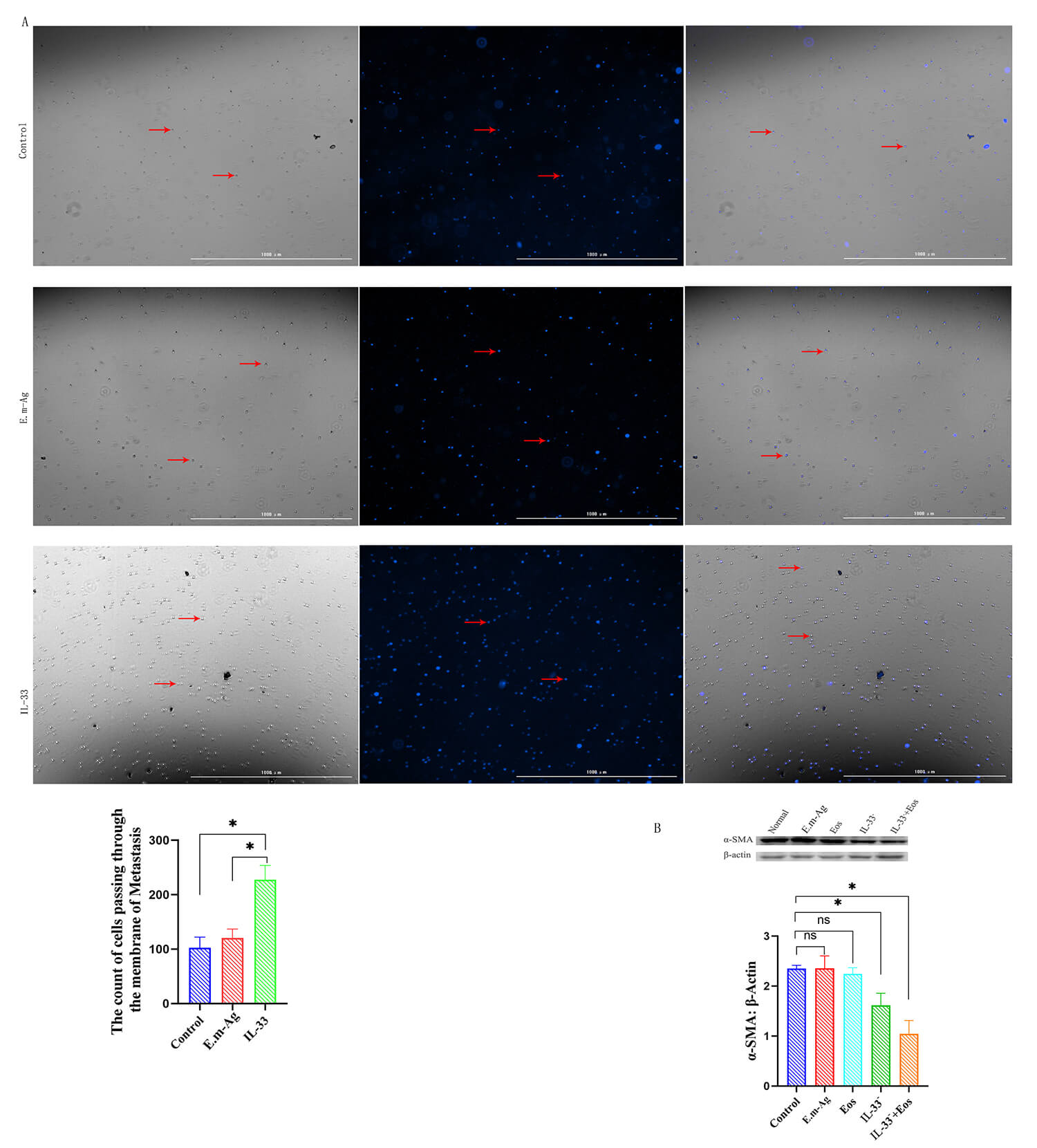 Fig. 6.
Fig. 6.
Eosinophil migration and
A dense fibrous band is a characteristic feature surrounding AE lesions, and
IL-33 has been implicated in fibrosis formation. To further investigate the
effect of IL-33 on lesion fibrosis during AE infection, five experimental groups
were established: blank control, E.m-Ag, eosinophil intervention, IL-33
inhibition, and eosinophil + IL-33 inhibition combined intervention. Following
various interventions on HSCs, Western blot analysis of
Increasing evidence highlights the critical role of the IL-33/ST-2 signaling pathway in infectious diseases, with its function influenced by factors such as pathogen type, affected organ, infection stage, and immune microenvironment. In this study, IL-33 was identified as a key mediator in the immune response to hepatic alveolar echinococcosis (AE). Findings demonstrated significantly elevated IL-33 and ST-2 levels in the peripheral blood of patients with AE compared to healthy individuals, which were somewhat different from the existing report, this might be more prominent because previous studies mainly focused on patients with AE rather than only on hepatic alveolar echinococcosis, underscoring its role in HAE progression [27]. This was further validated by Western blot and immunohistochemistry analyses, which showed increased IL-33/ST-2 expression in CLT relative to DLT. This discovery is similar to the reported study, reinforcing its involvement in local echinococcosis lesions [28]. IL-33, a member of the IL-1 family, exerts its biological effects by binding to its receptor ST-2 [29]. ST-2 is predominantly expressed on lymphocytes, including macrophages, Type 2 Innate Lymphoid Cells (ILC2), and eosinophils [24, 30, 31]. Given the well-established role of eosinophils in AE, their interaction with IL-33 was examined. Immunofluorescence analysis demonstrated a significant increase in eosinophils and IL-33 expression in CLT, with strong co-localization. In vitro studies confirmed that IL-33 intervention enhanced eosinophil phagocytosis and migration, demonstrating its role in activating and promoting the function of eosinophils.
Patients with AE develop a dense fibrous band surrounding their lesions,
creating a distinct immune microenvironment. While this fibrotic barrier limits
lesion expansion, it also obstructs chemotherapeutic drug penetration, reducing
antiparasitic efficacy and complicating treatment [19, 31]. Hepatic fibrosis in AE
arises primarily from HSC activation and an imbalance between extracellular
matrix (ECM) synthesis and degradation, leading to excessive ECM accumulation.
Notably, IL-33 expression correlates directly with fibrosis severity [22, 25, 32].
To explore IL-33’s role in AE-related fibrosis, in vitro HSC experiments
demonstrated that IL-33 inhibition significantly decreased
This study provides a comprehensive analysis demonstrating significantly
elevated IL-33/ST-2 expression within the lesion microenvironment of patients
with AE. In vitro experiments confirmed that IL-33 promotes
Thus, the IL-33/ST-2 signaling pathway plays a pivotal role in immune regulation during AE infection, contributing to fibrosis within the immune microenvironment of AE lesions. By modulating eosinophil activity, this pathway influences both disease progression and clinical outcomes. However, at the same time, we have to admit three limitations of our research. First, the peripheral blood eosinophil count of the subjects was not available. Because the level of blood eosinophils may vary greatly among individuals and affect local inflammatory infiltration, this is a potential confounding factor. Secondly, our research mainly focused on the lesion microenvironment, while the significant reduction of peripheral eosinophils in AE infection (Wang and Gottstein, 2016 [38]) as an immune escape mechanism has not been further studied. Finally, the obtained E.m-Ag antigen contains a trace of host blood components, which may have a potential impact on the experimental results. Further experiments are needed in the future to determine the relationship between global immunity and local immunity, peripheral and local eosinophil responses.
The expression of IL-33 and ST-2 and the distribution of eosinophils in the microenvironment of hepatic alveolar echinococcosis lesions increased abnormally. In vitro experiments showed that IL-33 could drive eosinophils to participate in the activation of hepatic stellate cells and promote the generation and expression of collagen fibers. Therefore, it is concluded that IL-33 promotes microenvironment fibrosis in AE lesions through the eosinophil mediated mechanism, highlighting the potential therapeutic targets for improving the efficacy of chemotherapy in AE patients.
AE, Alveolar echinococcosis; CVF, Collagenvolume Fraction; CLT, Close to the Lesion Tissue; DLT, Distant from the Lesion Tissue; HAE, Hepatic Alveolar Echinococcosis; EMC, Extracellularmatrix; Eos, Eosinophil; IHC, Immunohistochemistry; HSC, Hepatic Stellate Cell; IL, Interleukin; ELISA, EnzymeLinked Immunosorbent Assay; WB, Western Blotting; IF, Immunofluorescence;
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
SLC completed the design of the whole subject, completed all the experimental operations, and analyzed the experimental data. BJW and YXY made substantial contributions to the study design, experiment operation and assisted in data processing and statistical analysis. XMM participated in the conception and design of the study, coordinated data collection, and contributed to data analysis and interpretation and drafted the manuscript. YGZ gave guidance in the multiple immunofluorescence experiment and assisted in data processing and statistical analysis. HNF supervised and controlled the quality of the whole experiment and designed the experimental workflow, supervised the research process. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experimental procedures and the human sample experiments involved in this study were reviewed and approved by the Ethics Committee of Qinghai University Affiliated Hospital (Approval No.: PSL-2019039). The study was carried out in accordance with the guidelines of the Declaration of Helsinki. The samples referred to in the manuscript have obtained the informed consent of the the patients or their families/legal guardians/sample experiment. All efforts were made to minimize animal suffering and reduce the number of animals used in accordance with the ARRIVE guidelines.
We would like to acknowledge the hard and dedicated work of all the staff that implemented the intervention and evaluation components of the study.
National Science Foundation of China (81960576); Qinghai Science and Technology Department 2022 Science and Technology Project (Qinghai Provincial Key Laboratory of Hydatid Disease Research); National Clinical Key Specialty Construction Project of Hepatobiliary Surgery (Hydatid disease), Qinghai University Affiliated Hospital (No. 125, QingHealth Office).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






