1 Institute of Acupuncture and Moxibustion, China Academy of Chinese Medical Sciences, 100700 Beijing, China
2 Institute of Basic Theory for Chinese Medicine, China Academy of Chinese Medical Sciences, 100700 Beijing, China
†These authors contributed equally.
Abstract
Acupuncture has been shown to promote gastrointestinal motility. This study explores whether cutaneous transient receptor potential vanilloid 1 (TRPV1)+ fibers at Zusanli (ST36) acupoint can mediate the multimodal effects of acupuncture on colorectal motility, as well as examining their mechanistic role.
C57BL/6 mice were subjected to electroacupuncture (EA), manual acupuncture (MA), 46 °C thermal stimulation, and 1% capsaicin at the ST36 acupoint. Colon motility was quantified via the area under the curve (AUC) and contraction amplitude. Immunofluorescent co-localization of TRPV1 with CGRP, NF200, peripherin, and tyrosine hydroxylase (TH) was conducted in TrpV1Cre mice to determine neural phenotypic subtypes. Furthermore, TrpV1ChR2-eYFP and TrpV1NpHR-eYFP transgenic mice that underwent optogenetic activation or silencing of local TRPV1+ fibers at ST36 were evaluated for acupuncture-like stimulation effects on colorectal AUC and amplitude.
All applied stimuli in C57BL/6 mice significantly increased colorectal motility parameters (AUC and amplitude, p < 0.05) compared to baseline. TRPV1+ somatosensory neurons in the dorsal root ganglion (DRG) predominantly co-expressed with peripherin (46.76%) and CGRP (27%), which are markers of unmyelinated peptidergic fibers, but rarely with NF200 (6%) or TH (< 1%). Optogenetic activation (30 mW blue light) of TRPV1+ fibers in TrpV1ChR2-eYFP mice mimicked acupuncture-like stimuli, with significantly enhanced colorectal AUC and amplitude (p < 0.05). In contrast, optogenetic silencing of TRPV1+ fibers with yellow light abolished acupuncture-like stimulation of colorectal motility in TrpV1NpHR-eYFP mice (p < 0.05).
Through the use of spatiotemporally precise optogenetic control, our study revealed that TRPV1+ sensory fibers at ST36 are the major convergent pathway for multimodal (electrical/mechanical/thermal/chemical) enhancement of colorectal motility by acupuncture.
Keywords
- dorsal root ganglion
- TRPV1
- colon
- acupuncture
Acupuncture represents a somatosensory neuromodulatory intervention and exerts its therapeutic effects through mechanotransduction in cutaneous and subcutaneous tissues. This is mediated by peripheral primary sensory afferent fibers. Acupuncture is also considered a mainstream therapeutic modality for functional gastrointestinal disorders (FGIDs), with many randomized clinical trials (RCTs) published in high-impact journals demonstrating statistically significant improvements in global symptom relief [1, 2, 3, 4, 5]. As one kind of somatic stimulation, acupuncture can transmit stimuli through the dorsal root ganglion (DRG) and spinal cord level to achieve an analgesic effect on visceral pain and modulate gastrointestinal motility [6, 7, 8, 9]. However, it remains unclear which specific subtypes of DRG neurons mediate the effects of acupuncture.
DRG serves as a critical relay station for peripheral-to-central nervous system signaling [10]. Emerging evidence highlights the functional specificity of small-to-medium diameter DRG neurons in mediating somatosensory stimulation-induced modulation of visceral homeostasis through somatovisceral regulatory pathways, particularly through highly-expressed transient receptor potential vanilloid 1 (TRPV1) receptors [10, 11]. Indeed, TRPV1 is anatomically positioned as a core signaling mediator in acupuncture-induced somato-visceral integration, localizing to cutaneous nociceptive terminals, small-to-medium diameter DRG neurons, spinal cord dorsal horn nuclei, and visceral afferent networks [12]. In vivo constitutive TRPV1-deficient models exhibit attenuated acupuncture-mediated gastric modulation [13, 14], while electrophysiological evidence shows that TRPV1-dependent colorectal stimulation in Vil-ChR2 mice elicits behavioral responses that was similar to that seen with balloon distension of the colon [15]. Collectively, this suggests that TRPV1-expressing, small-to-medium DRG neurons are the leading candidate subtype mediating the effects of acupuncture on visceral function.
Despite this compelling association, the proportional mechanistic contribution of TRPV1 specifically within acupuncture-mediated visceromotor regulation remains unknown. This knowledge gap is compounded by the systemic pleiotropy of TRPV1 expression across multiple physiological axes, thereby complicating interpretation in traditional global knockout models. To address this limitation, the present study employed optogenetic interrogation for spatiotemporally precise activation and inhibition of TRPV1+ neural circuits at the ST36 (Zusanli) acupoint. This targeted strategy circumvents the interpretative limitations inherent to global knockout models. Moreover, it enables mechanistic dissection of TRPV1-dependent gut-brain signaling in acupuncture-evoked colorectal motility regulation, as well as quantitative determination of its modality-specific contributions to colorectal neuromodulation.
A total of 21 adult mice (22–25 g body weight) were used in this study, comprising 6 C57BL/6 wild-type mice, 2 TrpV1𝐶𝑟𝑒 mice, 8 TrpV1ChR2-eYFP mice (expressing ChR2-EYFP), and 5 TrpV1NpHR-eYFP mice (expressing NpHR3.0-EYFP). The sex of the animals was not distinguished in the experimental design. C57BL/6 mice (22–25 g) were obtained from SPF Biotechnology Co., Ltd. (Beijing, China). The TrpV1𝐶𝑟𝑒 mouse line (Stock No. 017769) was generously provided by Dr. Shenbin Liu’s laboratory at Fudan University. Optogenetic mouse lines, including ChR2 (Ai32; Stock No. 024109) and NpHR (Ai39; Stock No. 014539), were acquired from The Jackson Laboratory (Bar Harbor, ME, USA). Transgenic TrpV1ChR2-eYFP and TrpV1NpHR-eYFP mice (22–25 g) were generated and maintained by Shanghai Model Organisms Center, Inc. (Shanghai, China).
All animals were housed in standard polycarbonate cages (maximum 5 mice per
cage) with ad libitum access to autoclaved water and standard rodent
chow. The vivarium was maintained at 22
The fabricated polyethylene balloon catheters used here were described previously [13]. Catheter integrity was validated prior to experimentation via underwater pressure testing (1 mL syringe inflation) to confirm the balloon membrane integrity and leak-proof tubing connections. Following 4-h fasting (ad libitum water access), mice were anesthetized with 4% isoflurane in oxygen for induction and 2% isoflurane via nose cone for maintenance. The lubricated balloons were inserted 2 cm beyond the anal verge.
Following surgical preparation, a three-way stopcock was connected to a calibrated pressure transducer (Biopac Systems) pre-filled with distilled, deionized water. The system was meticulously purged of air bubbles to ensure signal fidelity, and the recording apparatus was then initialized and calibrated. This comprised a NeuroLog NL108A amplifier (Digitimer Ltd., Letchworth, UK), Micro1401 data acquisition interface (Cambridge Electronic Design, Cambridge, UK), and Spike2 software (v7.03, Cambridge Electronic Design, Cambridge, UK).
The stopcock configuration was adjusted to establish atmospheric pressure equilibration within the balloon system. Baseline calibration was performed to establish a 0 cmH₂O reference. A precise volume of distilled water (50–100 µL) was injected into the balloon using a calibrated microsyringe (Hamilton Company, Bonaduz, Switzerland). The spherical water-filled balloon, positioned within the colorectal lumen, transduced intraluminal pressure changes, which were transmitted hydrostatically through the polyethylene catheter to the pressure transducer. The analog pressure signal was amplified (NeuroLog NL108A) and digitized (Micro1401) for acquisition. Real-time signal visualization and recording were performed using Spike2 (v7.03, Cambridge Electronic Design Limited, British) with appropriate filtering.
Following stabilization of the colorectal motility patterns (typically 15–30
minutes post-surgical preparation), baseline recordings were acquired for 1
minute prior to stimulation. Each stimulation protocol was administered for
precisely 1 minute, followed by a minimum 5-minute washout period to allow
complete recovery of motility parameters to baseline levels, as confirmed by
real-time monitoring of pressure traces. This inter-stimulus interval was
maintained to prevent carryover effects between experimental conditions.
Comparative waveform analysis between pre-stimulation baseline and stimulation
periods was performed using two key parameters: Area Under Curve (AUC), and
Contractile Amplitude. AUC, pressure-time integral, represents the total
propulsive effort (cmH2O
The ST36 acupoint was localized according to standardized anatomical landmarks described in the Experimental Acupuncture reference text. It was identified as 3 mm lateral to the tibial crest at the knee joint level, corresponding to the classical anatomical position in murine models. Stimulation duration was maintained for 1 minute.
EA was administered using acupuncture needles connected to a Constant Current
Isolated Stimulator (DS3; Digi-timer Ltd., Welwyn Garden City, UK). Stimulation
parameters were set at 1 mA intensity, 10 Hz frequency, and 1 ms pulse width. The
needle was inserted vertically to a depth of 5 mm at the ST36 acupoint. Manual
Acupuncture (MA) was performed by a single trained practitioner to ensure
consistency. Following vertical needle insertion, manual stimulation was applied
using a standardized technique involving rapid bidirectional rotation
(180° clockwise and counterclockwise) at a 2 Hz frequency for 1 minute.
A calibrated temperature probe was heated to 47
As illustrated in Fig. 1A, four different types of stimulation were applied sequentially to the ST36 acupoint of C57BL/6 mice: EA, MA, 46 °C, and CAP. The waveforms were subsequently compared before and during the stimulation.
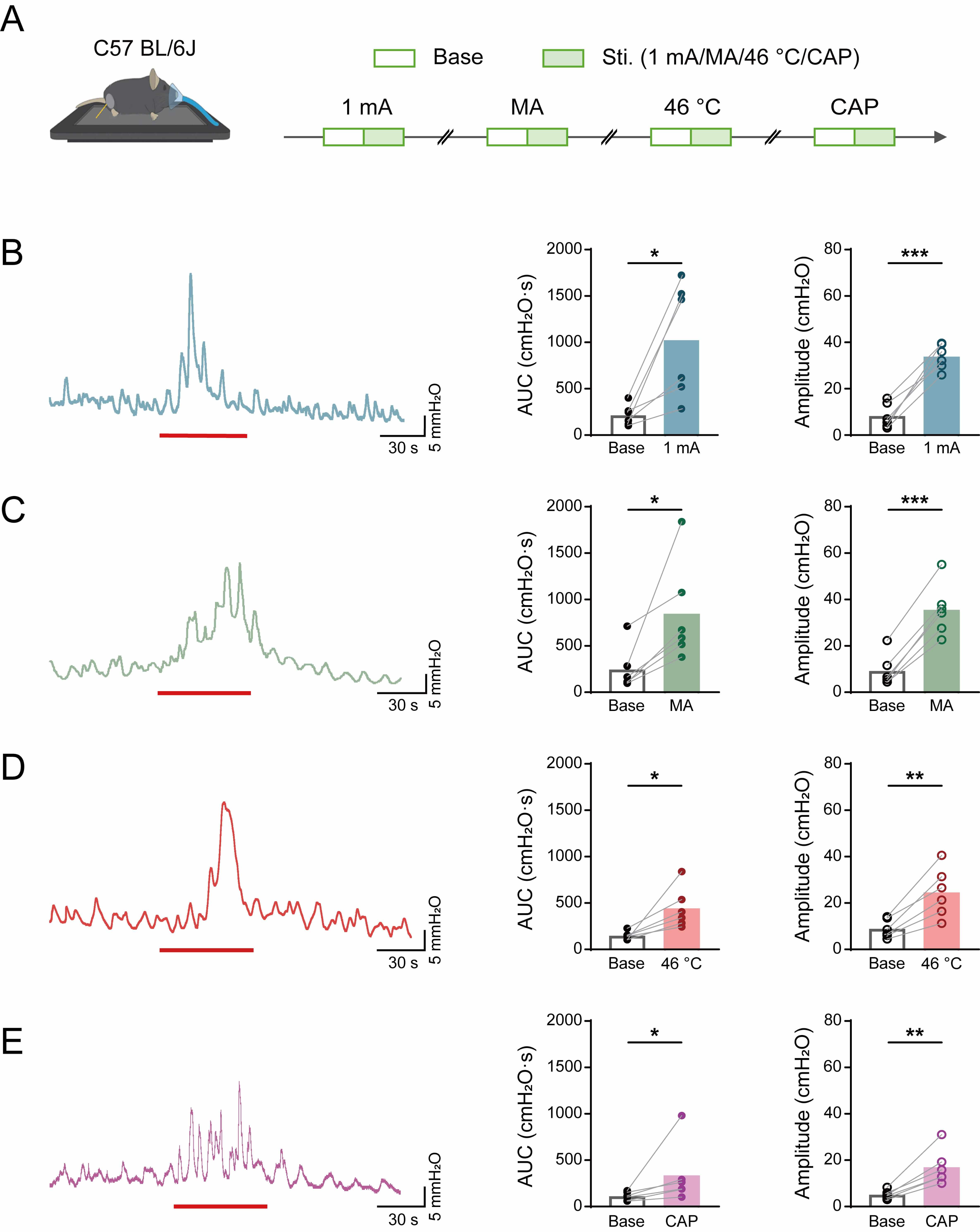 Fig. 1.
Fig. 1.
Effects of diverse acupuncture-like stimuli at ST36 on
colorectal motility in C57 mice. (A) Experimental flowchart for acupuncture-like
stimulation at ST36 in C57 mice. Mouse image was modified from Heaster TM et al. (https://doi.org/10.3389/fbioe.2021.644648). (B–E) Representative example graphs of
colorectal motility (left), and statistical graphs of AUC (middle) and amplitude
(right) during EA (B), MA (C), 46 °C heat (D), and capsaicin (E) stimulation of
C57 mice at ST36. Length of each red trace refers to 1 min. n = 6, *p
Light (473/593 nm) for stimulation was delivered through a 200 µm diameter optical fiber (RWD, R-FC-L-N2-200-L1) positioned perpendicular to the skin and affixed tightly against it. The fiber tip was placed directly above the ST36 acupoint. Prior to each experiment, laser power output from the fiber optic cable was measured using a photometer (PM100D, Thor Labs, Newton, NJ, USA).
The optogenetic activation protocols shown in Fig. 3A employed identical motility recording and analysis pipelines as those established in prior experiments, with light parameters optimized to achieve suprathreshold TRPV1+ fiber activation (473 nm, 30 mW, 10 Hz).
For yellow light inhibition stimulation, after the colorectal motility recordings in mice had stabilized, a 1-minute recording segment was selected as the baseline for comparison. This was followed by a 1-minute EA (1 mA, 10 Hz) stimulation. Once the colorectal motility trace returned to a stable state, another 1-minute baseline was selected. Yellow light stimulation (20 mW, 10 Hz) was then applied for 30 seconds in advance, followed by a combined stimulation of yellow light (20 mW, 10 Hz) and EA (1 mA, 10 Hz) for 1 minute. The process was repeated with increasing intensities of yellow light, as shown in Fig. 6A. Changes in the AUC and amplitude of colorectal motility induced by EA alone and by the combination of yellow light and EA were compared. As shown in Fig. 7A, after determining the effective intensity of yellow light (30 mW, 10 Hz), different peripheral acupuncture-like stimuli were sequentially applied, with the same operational procedures and statistical parameters as described previously.
After recording of colorectal motility, TrpV1ChR2-eYFP and TrpV1NpHR-eYFP mice were weighed and anesthetized with 1.25% tribromoethanol (Avertin; 2 mL/100 g body weight, i.p.). TrpV1𝐶𝑟𝑒 mice, which did not undergo motility recording, were weighed and anesthetized directly (Avertin; 2 mL/100 g body weight, i.p.). Following depilation of the ST36 acupoint region using electric clippers, thoracotomy was performed to expose the cardiac apex. Systemic perfusion was initiated with 40 mL of ice-cold phosphate-buffered saline (PBS; 0.9%, pH 7.4) followed by 40 mL of freshly prepared 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer (PB; pH 7.4), delivered at a rate of 5 mL/min using a peristaltic pump. Following perfusion, bilateral DRG from lumbosacral (L6–S2) segments were dissected and post-fixed in 4% PFA for 3 h at 4 °C. Tissues were then cryoprotected through sequential sucrose gradients (15% and 30% in 0.1 M PB) at 4 °C until equilibrium (typically 24–48 h), as determined by tissue sedimentation. Cryoprotected samples were stored in 30% sucrose solution at 4 °C until further processing.
Following complete cryoprotection, tissue samples were embedded in optimal cutting temperature (OCT) compound (Epredia Richard-Allan NEG 50, Kalamazoo, MI, USA) and rapidly frozen on a cryostat microtome freezing chamber (Leica CM1950, Wetzlar, Germany). Serial coronal sections (20 µm thickness) were obtained using a cryostat microtome maintained at –20 °C. Sections were systematically collected and mounted onto positively charged glass slides (Superfrost Plus, Thermo Fisher Scientific, Waltham, MA, USA). To ensure tissue integrity and minimize freeze-thaw artifacts, sections were air-dried for 30 minutes at room temperature.
Tissue sections were rehydrated in 0.1 M PB (pH 7.4) for 10 minutes to remove
residual OCT compound. Non-specific binding sites were blocked with a solution
containing 5% normal goat serum (Jackson ImmunoResearch, West Grove, CA, USA),
0.5% Triton X-100 (Sigma-Aldrich, Saint Louis, MO, USA), and 0.1 M PB for 1 h at
room temperature (22
The following primary antibodies were diluted in antibody dilution buffer (3% normal goat serum, 0.5% Triton X-100 in 0.1 M PB) and applied to sections: TRPV1 (1:600, GP14100, NEUROM), CGRP (1:1500, 24112, IMMUNOSTAR), NF200 (1:600, Ab207176, Abcam), PER (1:1000, AVES, NEUROM), TH (1:600, ab112, Abcam), and GFP (1:1000, GFP-1010, Aves Labs). Incubation was performed in a humidified chamber at 4 °C for 16–18 h. After three 10-minute washes in 0.1 M PB, the following species-specific secondary antibodies conjugated with Alexa Fluor dyes (Thermo Fisher Scientific, Waltham, MA, USA) were applied at a 1:500 dilution in antibody dilution buffer and incubated for 2 h at room temperature in darkness: goat anti-guinea pig 594 (A-11076, ThermoFisher, Waltham, MA, USA), goat anti-chicken 488 (A-11039, ThermoFisher), and goat anti-rabbit 488 (A32731, ThermoFisher, Waltham, MA, USA). Following three washes, sections were then coverslipped using anti-fade mounting medium containing 50% glycerol (Vector Laboratories, San Francisco, CA, USA) and sealed with clear nail polish.
Fluorescence imaging was performed using an epifluorescence microscope (Olympus
FV1200, Tokyo, Japan) equipped with appropriate filter sets and a confocal laser
scanning microscope (Zeiss LSM 880, Oberkochen, Germany) with 20
All statistical analyses were performed using GraphPad Prism software (version
8.0, GraphPad Software Inc., San Diego, CA, USA). Data are expressed as the mean
We first systematically characterized the effects of diverse acupuncture-like stimuli modalities on colorectal motility patterns in C57BL/6 mice, focusing on the ST36 acupoint. The stimulation paradigm incorporated four distinct interventions, as illustrated in Fig. 1A.
Quantitative analysis of colorectal motility parameters (Fig. 1B–E) revealed
that all stimulation modalities significantly enhanced motor function compared to
baseline recordings. Specifically, marked increases were observed in the AUC (EA:
from 214.8
In order to demonstrate the effectiveness of the TRPV1 channel in
acupuncture-like stimuli, we conducted detailed immunohistochemical
characterization of TRPV1-expressing neurons in DRG sections from
TrpV1𝐶𝑟𝑒 mice, focusing especially on the homosegemental level
(L6–S2, Fig. 2). Quantitative analysis demonstrated that TRPV1 predominantly
co-localized with markers of unmyelinated peptidergic neurons (CGRP: 27.39
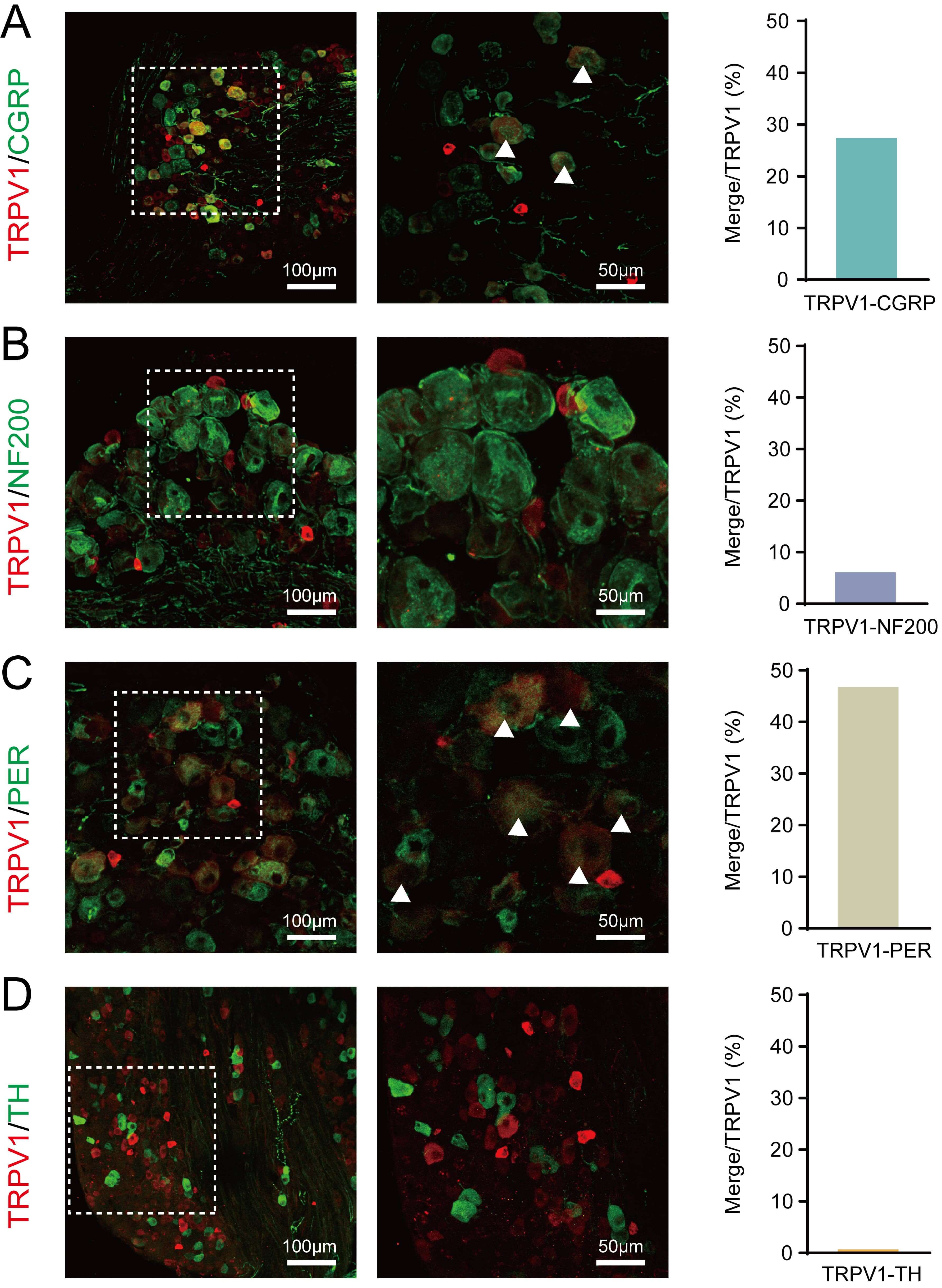 Fig. 2.
Fig. 2.
Molecular characterization of TRPV1+ neurons in
TrpV1𝐶𝑟𝑒 mice DRG. (A–D) Representative immunofluorescence images showing
the co-expression of TRPV1 (red) with: (A) CGRP (green), (B) NF200 (green), (C)
Peripherin (green), and (D) Tyrosine hydroxylase (TH; green). Scale bar: left 100
µm, right 50 µm. Right panels: Quantification of
co-localization percentages (mean
To determine whether TRPV1+ neurons are the best candidates for acupuncture effects, we generated TrpV1ChR2-eYFP mice in which cutaneous TRPV1 sensory neurons can be selectively activated with precise temporal and spatial control using an external source of blue light. TrpV1ChR2-eYFP mice were generated by breeding Ai32 mice [16] with mice expressing Cre recombinase under the control of the TRPV1 promoter [17]. ChR2-expressing neurons in DRG can be visualized using immunofluorescence microscopy based on their expression of eYFP. As expected, TRPV1+ neurons in the DRG of TrpV1ChR2-eYFP mice co-expressed eYFP (Fig. 3A).
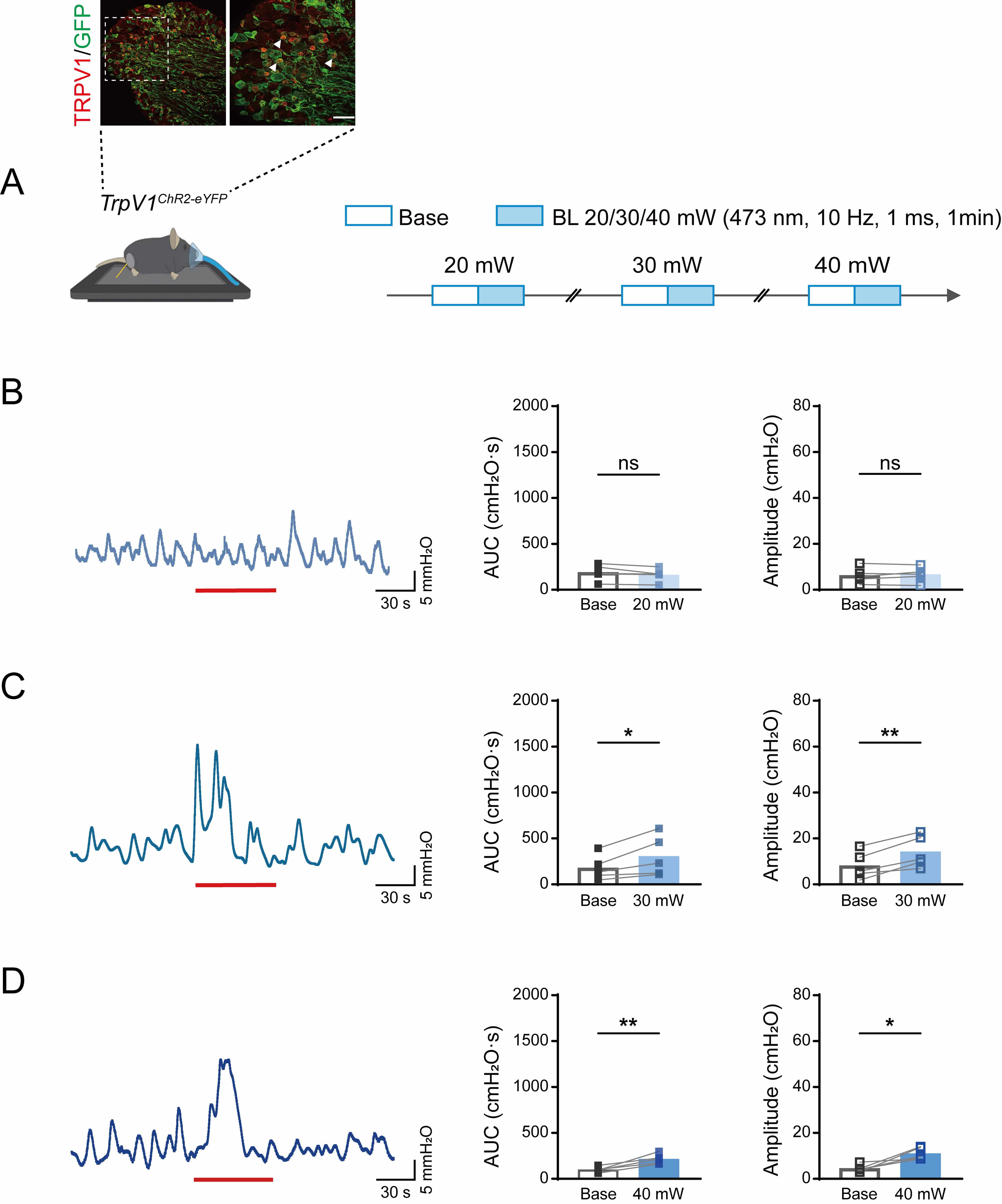 Fig. 3.
Fig. 3.
Effects of different intensities of blue light stimulation at
ST36 on colorectal motility in TrpV1ChR2-eYFP mice. (A)
Immunofluorescent visualization of TrpV1ChR2-eYFP DRG neurons,
showing the expression of ChR2/eYFP (green) and TRPV1 (red) (top). Scale bar, 50
µm. Experimental flowchart for different intensities of blue light
stimulation at ST36 in TrpV1ChR2-eYFP mice (bottom). Arrowheads
indicate double stained neurons. Mouse image was modified from Heaster TM et al. (https://doi.org/10.3389/fbioe.2021.644648). (B–D) Representative example graphs of
colorectal motility (left), and statistical graphs of AUC (middle) and amplitude
(right) during 20 mW (B), 30 mW (C), and 40 mW (D) blue light stimulation in
TrpV1ChR2-eYFP mice at ST36. Length of each red trace refers to 1
min. n = 5, *p
To determine the optimal optical stimulation parameters, we systematically
evaluated the intensity of blue light (473 nm) on colorectal motility patterns in
TrpV1ChR2-eYFP transgenic mice using a graded intensity protocol
ranging from subthreshold to suprathreshold levels (20–40 mW). As shown in Fig. 3B–D, optical stimulation was achieved at 473 nm with the parameters of 10 Hz
frequency, 1 ms pulse width, and 1 min illumination duration. This elicited a
significant increase in colorectal motility, as quantified by both AUC and
contraction amplitude. Notably, light intensities of 30 mW and 40 mW produced
robust and statistically significant responses (AUC: 20 mW, from 189.2
To examine the effects of the TrpV1 receptor during acupuncture-like stimulation, we next compared optogenetic stimulation (30 mW) with multiple acupuncture-like stimuli in TrpV1ChR2-eYFP mice, as illustrated in Fig. 4A. Quantitative assessment of colorectal motility parameters (Fig. 4B–F) revealed that both optical stimulation and acupuncture-like interventions elicited significant potentiation of contractile activity, as evidenced by increased AUC and contraction amplitude. The remarkable similarity in response profiles provides compelling evidence that TRPV1-expressing sensory neurons mediate the visceromotor effects of acupuncture-like stimuli.
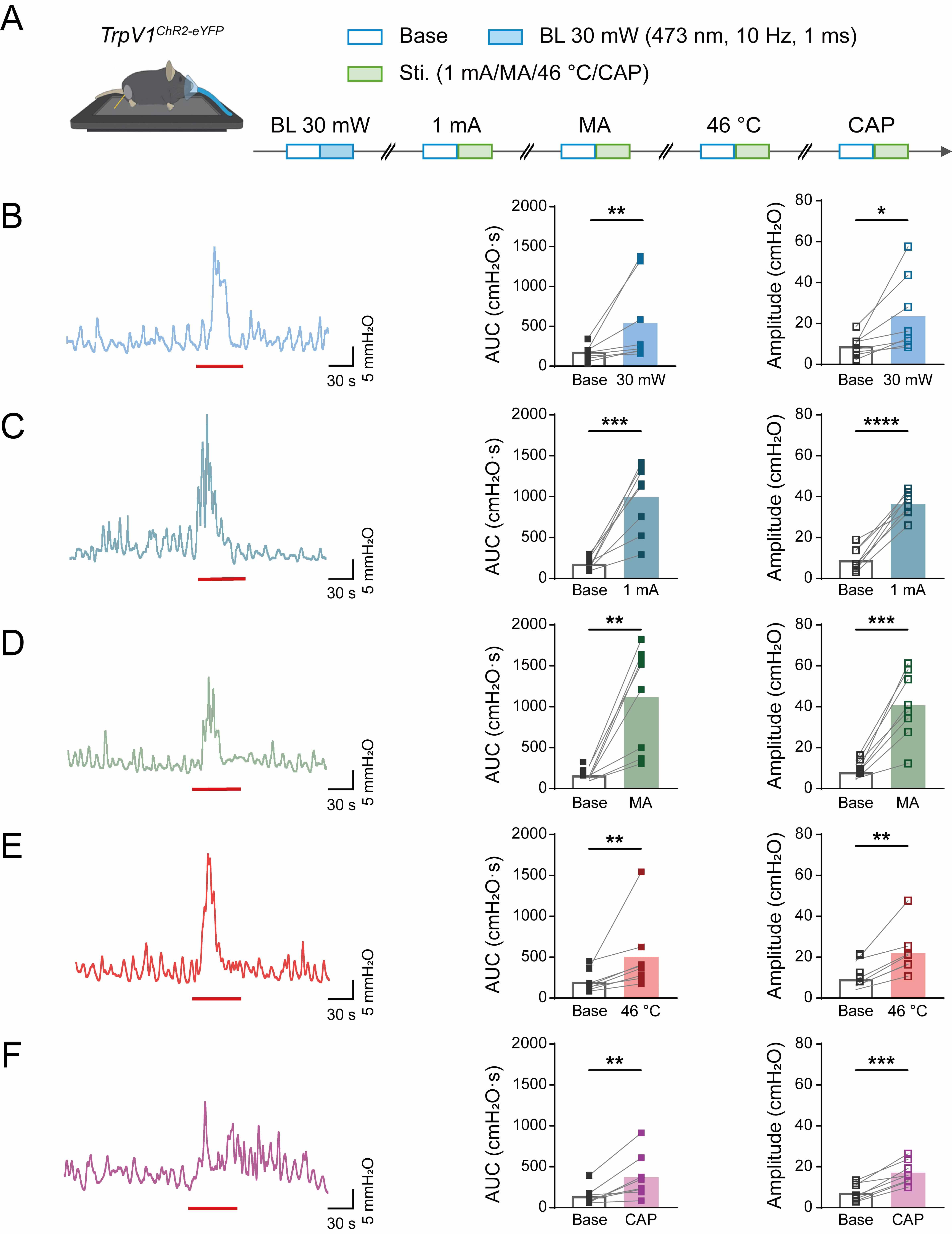 Fig. 4.
Fig. 4.
Colorectal motility of TrpV1ChR2-eYFP mice in
response to different acupuncture-like stimuli at ST36. (A) Experimental
flowchart for different intensities of blue light stimulation at ST36 in
TrpV1ChR2-eYFP mice. Mouse image was modified from Heaster TM et al. (https://doi.org/10.3389/fbioe.2021.644648). (B–F) Representative example graphs
of colorectal motility (left), and statistical graphs of AUC (middle) and
amplitude (right) during stimulation of TrpV1ChR2-eYFP mice at ST36
with 30 mW blue light (B), EA (C), MA (D), 46 ℃ heat (E), and CAP (F). Length of
each red trace refers to 1 min. n = 8, *p
Quantitative analysis also revealed distinct efficacy profiles among the
stimulation modalities (Fig. 5). EA and MA interventions demonstrated superior
modulation of colorectal motility function compared to 30 mW optogenetic
stimulation (EA: 511.3
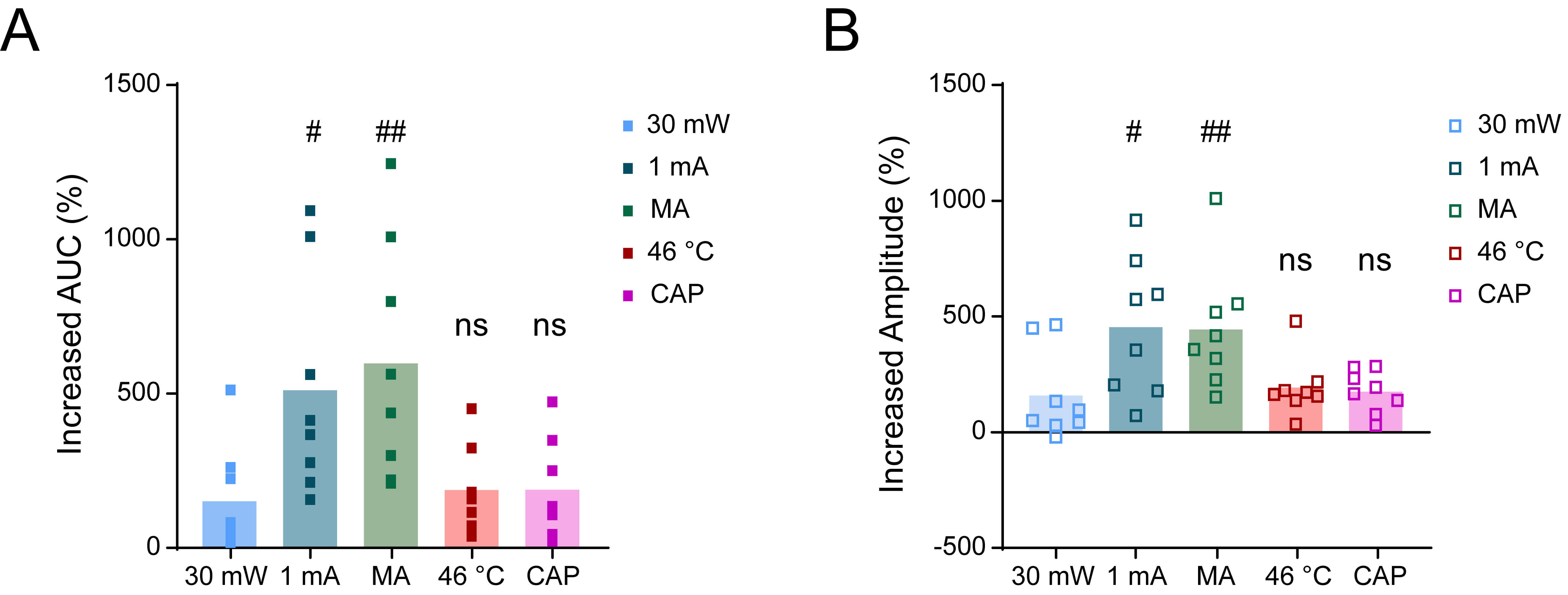 Fig. 5.
Fig. 5.
The extent of increased colorectal motility in
TrpV1ChR2-eYFP mice induced by different acupuncture-like stimuli
at ST36. (A) Proportion of increase in the AUC of colorectal motility in
TrpV1ChR2-eYFP mice during stimulation at ST36 with blue light at
30 mW, EA, MA, 46 °C, and CAP, compared with the respective baselines.
(B) Proportion of increase in the amplitude of colorectal motility in
TrpV1ChR2-eYFP mice during stimulation at ST36 with blue light at
30 mW, EA, MA, 46 °C, and CAP, compared with the respective baselines. n
= 8, #p
We next employed an optogenetic loss-of-function strategy to investigate the contribution of TRPV1+ sensory pathways to acupuncture-evoked colorectal modulation. TrpV1NpHR-eYFP enables cell-type-specific silencing of TRPV1+ afferents through yellow light (593 nm) activation of halorhodopsin. Immunohistochemical analysis of DRG sections (L6–S2) revealed precise co-localization of eNpHR3.0-eYFP expression with endogenous TRPV1 immunoreactivity, as shown in Fig. 6A.
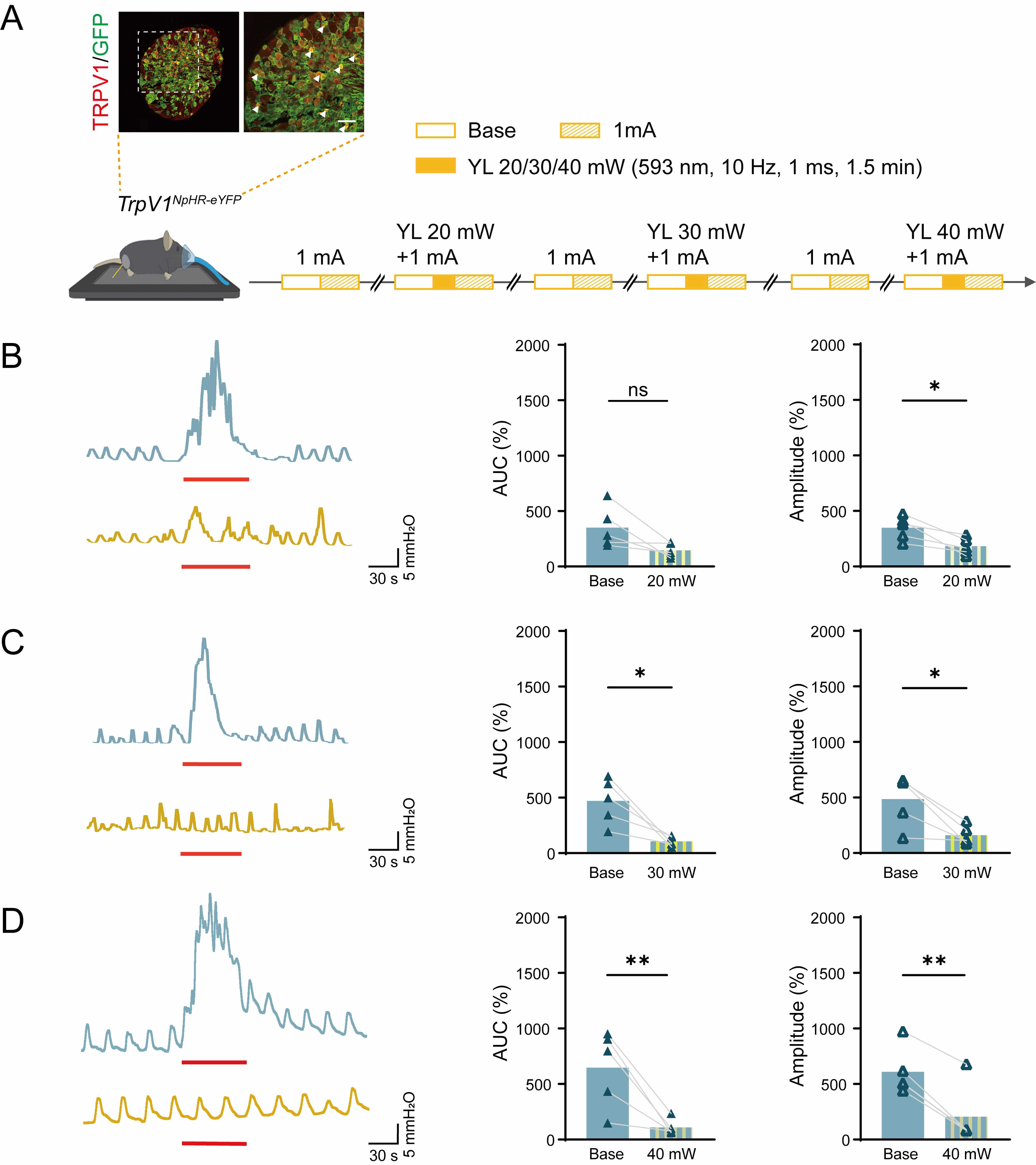 Fig. 6.
Fig. 6.
Effects of EA and EA with different intensities of yellow light
stimulation at ST36 on colorectal motility in TrpV1NpHR-eYFP mice.
(A) Immunofluorescent visualization of TrpV1NpHR-eYFP DRG neurons
showing the expression of NpHR/eYFP (green) and TRPV1 (red) (top). Scale bar, 50
µm. Experimental flowchart for different intensities of yellow light
stimulation of ST36 in TrpV1NpHR-eYFP mice (bottom). Arrowheads
indicate double stained neurons. Mouse image was modified from Heaster TM et al. (https://doi.org/10.3389/fbioe.2021.644648). (B–D) Representative example graphs of
colorectal motility (left) and statistical graphs of AUC (middle) and amplitude
(right) during EA stimulation and EA with 20 mW (B), 30 mW (C) and 40 mW (D) of
yellow light stimulation at ST36 in TrpV1NpHR-eYFP mice. Length of
each red trace refers to 1 min. n = 5, *p
To establish the optimal parameters for optogenetic inhibition, we
systematically characterized the ideal intensity for yellow light (593 nm)
stimulation of colorectal motility in TrpV1NpHR-eYFP mice. As shown
in Fig. 6B–D, quantitative analysis revealed that both 30 mW and 40 mW
irradiance levels significantly suppressed colorectal motility function. This was
evidenced by reduced AUC (30 mW: from 471
To further illustrate the inhibitory effect of TrpV1 during yellow light
stimulation, we combined optogenetic inhibition with four distinct somatic
acupuncture-like interventions in TrpV1NpHR-eYFP mice (Fig. 7A).
Detailed experimental procedures are described in the Methods section. As shown
schematically in Fig. 7B–E and Fig. 8A,B, quantitative analysis demonstrated
that optogenetic silencing of TRPV1+ afferents produced modality-specific
attenuation of colorectal motility parameters. For AUC measurements, significant
reductions in efficacy were observed across all stimulation conditions, as shown
in Fig. 7 (EA: from 695.1
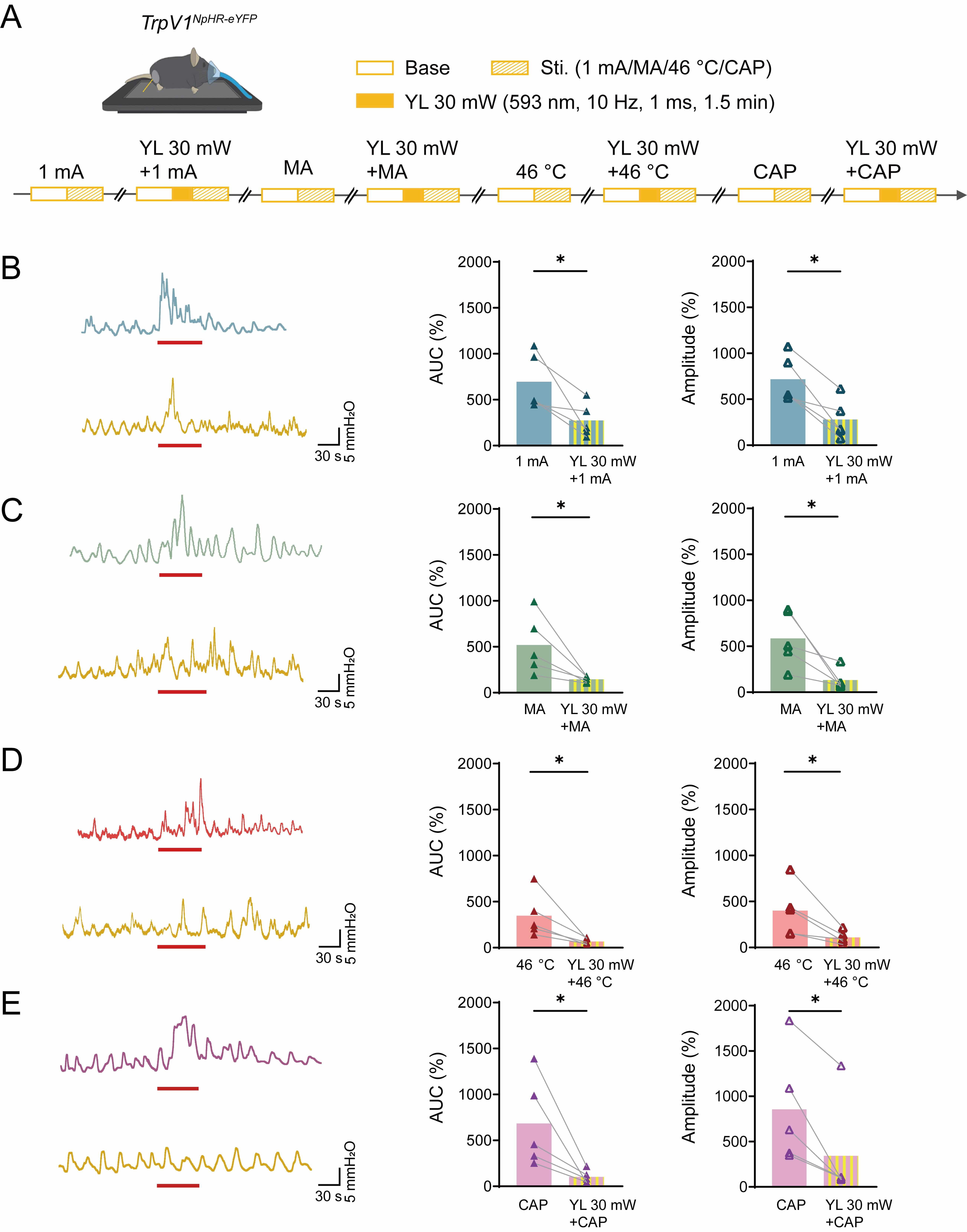 Fig. 7.
Fig. 7.
Effects of acupuncture-like stimuli and acupuncture-like stimuli
combined with yellow light stimulation at ST36 on colorectal motility in
TrpV1NpHR-eYFP mice. (A) Experimental flowchart for four different
stimuli and stimuli with 30 mW yellow light stimulation at ST36 in
TrpV1NpHR-eYFP mice. Mouse image was modified from Heaster TM et al. (https://doi.org/10.3389/fbioe.2021.644648). (B–E) Representative example graphs of
colorectal motility (left), and statistical graphs of AUC (middle) and amplitude
(right) of acupuncture-like stimuli: EA (B), MA (C), 46 ℃ heat (D), and CAP (E),
and acupuncture-like stimuli with 30 mW yellow light stimulation in
TrpV1NpHR-eYFP mice at ST36. Length of each red trace refers to 1
min. n = 5, *p
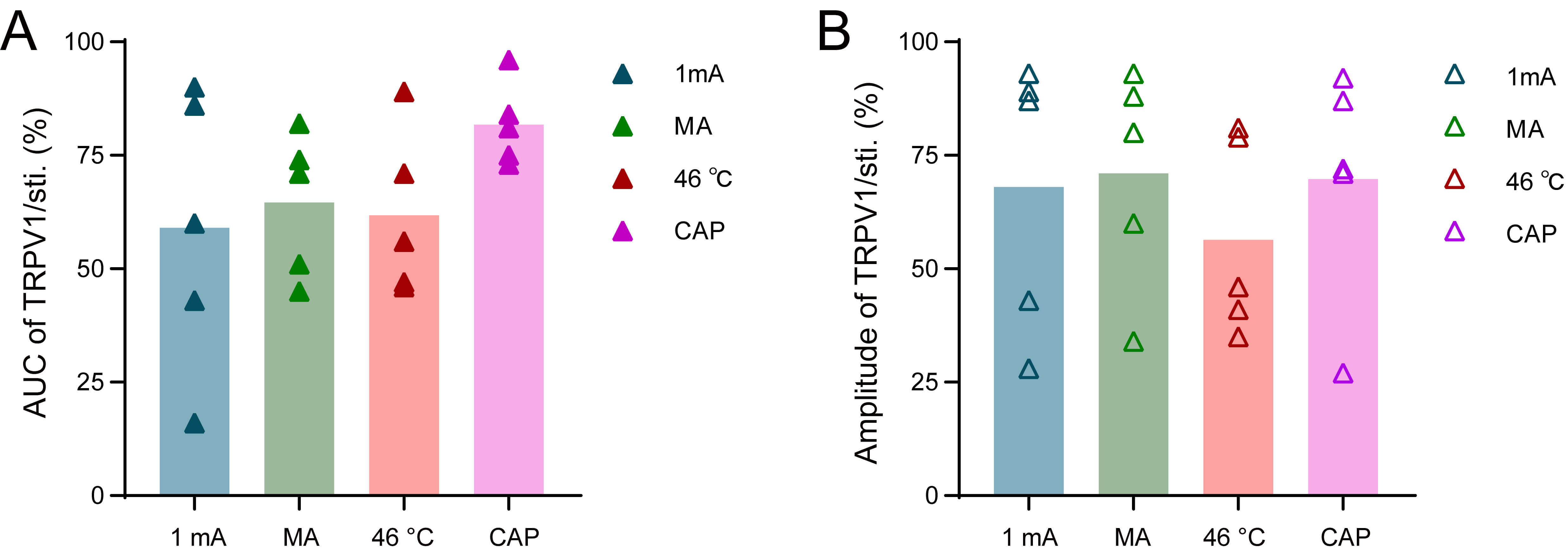 Fig. 8.
Fig. 8.
The proportion of contribution of TRPV1 to the facilitation of colorectal motility under various acupuncture-like stimuli at ST36. (A) Statistical chart of the proportion of TRPV1’s effect on the AUC of colorectal motility in TrpV1NpHR-eYFP mice induced by EA, MA, 46 °C heat, and CAP stimulation at ST36. (B) Statistical chart of the proportional effect of TRPV1 on the amplitude of colorectal motility in TrpV1NpHR-eYFP mice induced by EA, MA, 46 °C heat, and CAP stimulation at ST36. n = 5.
Further analysis revealed that TRPV1-expressing fibers at ST36 differentially
mediate the effects of acupuncture-like stimuli modalities on colorectal motor
parameters. EA: 64.97
The two key questions addressed in this study were: (1) whether TRPV1 mediated the modulation of colorectal motility by heterogeneous acupoint stimulation, and (2) what the relative contribution of TRPV1 signaling was to these neuromodulatory effects. We functionally dissected these mechanisms by characterizing colorectal motor patterns in genetically distinct mouse strains in response to region-specific light modulation of TRPV1-expressing afferents at the ST36 acupoint.
Building on established neurophysiological principles in which
A
TRPV1 serves as a polymodal transducer essential for peripheral nociception, and
is therefore predominantly expressed in small- and middle-diameter peripheral
neurons [19, 20]. Research has shown that its expression is enriched in acupoints and
can be dynamically upregulated by EA [19]. In our model, TRPV1 showed
preferential co-expression with CGRP+ nociceptors (20–25% overlap), but
minimal co-localization with NF200+ neurons (
Specific optogenetic activation of TRPV1+ afferents with blue light (473 nm) enhanced colorectal motility in TrpV1ChR2-eYFP mice. The effect was comparable in amplitude to that induced by 46 °C heat or capsaicin interventions, but was less potent than EA or MA (Fig. 5). This mechanistic divergence arises from differential nociceptor engagement profiles, with 46 °C heat, capsaicin and optogenetic stimulation primarily activating cutaneous TRPV1+ fibers, whereas EA and MA concurrently engage multiple nociceptor subtypes (e.g., TRPV1+, ASIC3+, P2X3+ populations) across cutaneous and muscular compartments, generating synergistic neuromodulatory effects through spatial summation in spinal cord projection neurons [23].
Conversely, optogenetic inhibition of TRPV1+ afferents during EA at ST36 with yellow light (589 nm) in TrpV1NpHR-eYFP mice significantly attenuated the stimulation-induced potentiation of colorectal motility by various modalities (Figs. 6,7). Consistent with previous findings in TRPV1 KO mice, our results support the crucial role of TRPV1 in mediating acupuncture-like stimuli effects [24]. Notably, Dong et al. [25] recently provided direct functional evidence by demonstrating that specific pharmacological inhibition of deep TRPV1+ sensory fibers innervating the ST36 region prevented EA at ST36 from effectively triggering gastric reflexes. This finding suggests that deep TRPV1+ sensory fibers innervating the ST36 fascial compartment are one of the key initial components in the cascade of EA-promoted gastric motility [25]. As shown in Fig. 8, we observed variations in the relative contribution of TRPV1-mediated signaling to colorectal motility regulation depending on the stimulus. This indicates that TRPV1 plays a complex role in mediating the regulatory effects of acupuncture-like stimuli on colorectal motility, with variable contributions depending on the type of stimulation and the specific metrics measured. While optogenetic silencing of TRPV1+ fibers at the ST36 acupoint was utilized in this study, future validation should incorporate orthogonal approaches. This could include topical application of selective antagonists, and chemogenetic inhibition via DREADD-expressing models to establish modality-independent causality.
A key consideration is how activation of TRPV1+ afferents in the hindlimb (ST36) influences distant visceral motility. The somato-visceral pathway recruited by ST36 stimulation likely involves both spinal and vagal mechanisms. Afferents from the ST36 region (tibialis anterior muscle) primarily originate from lumbar DRGs (L4–L6) and project to the corresponding spinal segments [26]. Following initial integration in the spinal cord, this information may influence colorectal motility via a spino-bulbo-vagal pathway [24], reaching brainstem nuclei such as the nucleus tractus solitarius and the dorsal motor nucleus of the vagus, ultimately modulating gastrointestinal function through vagal efferents. Supporting vagal involvement, optogenetic stimulation of ST36 has been shown to robustly potentiate cervical vagal efferent activity. Thus, the observed modulation of colorectal motility may result from activation of TRPV1+ lumbar afferents at ST36 and their subsequent integration within this spino-vagal axis.
While optogenetics provides cell-type-specific causality, several limitations should be considered. (1) The penetration depth of transdermal optical stimulation is limited, likely affecting cutaneous and superficial subcutaneous fibers [27, 28]. (2) Experiments were conducted under isoflurane anesthesia, which may influence autonomic tone and baseline colonic motility [29]. (3) While our motility data support effective inhibition by yellow light, we lack in vivo electrophysiological recordings (e.g., from the sciatic nerve or dorsal roots) to directly quantify the silencing efficiency of TRPV1+ fibers during NpHR activation. (4) Sample sizes were small for some experimental groups. Future studies should employ orthogonal approaches—such as topical application of selective antagonists and chemogenetic inhibition—for validation, and directly investigate the complete neural circuitry in awake, behaving models.
Our research has provided novel insights into the regulatory role of TRPV1 receptors in colorectal motility when activated in an acupoint-targeted manner, with the elucidation of key mechanisms in this physiological process. This enhances our understanding of the role of TRPV1 in regulating gastrointestinal function, as well as having significant implications for clinical practice. Specifically, by considering the degree of TRPV1 involvement as a key biomarker, we anticipate substantial improvements in the design of therapeutic interventions, leading to more effective improvement of neuroregulatory functions in the gastrointestinal tract. The present work establishes a theoretical basis for developing novel therapeutic strategies to address common conditions such as functional gastrointestinal disorders, ultimately improving clinical outcomes and patient quality of life.
FGIDs, Functional gastrointestinal disorders; DRG, Dorsal root ganglion; ST36, Zusanli acupoint; EA, Electroacupuncture; MA, Manual acupuncture; AUC, Area under the curve; TH, Tyrosine hydroxylase; RCTs, Randomized clinical trials; TRPV1, Transient receptor potential vanilloid 1; OCT, Optimal cutting temperature; SEM, Standard error of the mean; ANOVA, Analysis of variance.
Data will be made available upon request from the corresponding author.
NZ: Methodology, Validation, Formal analysis, Investigation, Data curation, Writing—original draft, Writing—review and editing, Supervision, Project administration. HC: Investigation, Writing—review and editing, Visualization. XG: Conceptualization, Investigation, Resources, Supervision, Project administration. SL: Methodology, Formal analysis, Investigation, Data curation. WL: Methodology, Formal analysis, Investigation. XL: Validation, Formal analysis, Investigation. KL: Validation, Formal analysis, Investigation. SW: Conceptualization, Resources, Supervision, Project administration, Funding acquisition. BZ: Conceptualization, Resources, Supervision, Project administration, Funding acquisition. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript for publication. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All experimental procedures were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Experimental Animal Ethics Committee of the China Academy of Chinese Medical Sciences (No. D2024-01-26-07).
Not applicable.
This study was supported by National Natural Science Foundation of China (82230123, 82174281), and the Fundamental Research Funds for the Central Public Welfare Research Institutes (ZZ14-YQ-033).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








