1 BK21 Graduate Training Program of Korean Medical Therapeutics for Healthy Aging, Pusan National University, 50612 Yangsan, Republic of Korea
2 Department of Korean Medical Science, School of Korean Medicine, Pusan National University, 50612 Yangsan, Republic of Korea
3 Department of Rehabilitation Medicine, School of Medicine, Pusan National University, 50612 Yangsan, Republic of Korea
4 Department of Sasang Constitutional Medicine, Division of Clinical Medicine 4, School of Korean Medicine, Pusan National University, 50612 Yangsan, Republic of Korea
Abstract
Stroke is a leading cause of long-term disability, and many patients fail to achieve complete recovery following cerebral injury. Therefore, post-stroke rehabilitation is essential to restore impaired function. Transcranial electrical stimulation (tES), transcranial direct current stimulation (tDCS), and transcranial alternating current stimulation (tACS) have emerged as promising neuromodulation approaches to enhance post-stroke recovery. These treatments have therapeutic effects to restore impaired function by modulating cortical excitability and reorganizing brain tissue through electrical stimulation. However, the fundamental mechanisms underlying these therapeutic effects remain poorly understood. This review focused on the neurobiological mechanisms underlying tES that extend beyond cortical excitability and encompass long-term neuroplasticity, cerebral blood flow, neurometabolism, and neuroinflammatory modulation. Our summary provides a comprehensive understanding of tES processes and plays a vital role in the advancement of improved treatments. Additionally, our review promotes enhanced clinical outcomes through interactions with various stroke rehabilitation strategies.
Keywords
- stroke
- transcranial electrical stimulation
- excitability
- neuroplasticity
- cerebral blood flow
- neurometabolism
- neuroinflammation
Patients who experience stroke frequently fail to achieve full neurological recovery, resulting in long-term impairment that markedly diminishes quality of life and remains a leading cause of chronic disability [1, 2]. Despite advances in stroke management, no targeted therapy currently exists to repair damaged neural tissue and fully restore function, leaving most post-stroke patients dependent upon rehabilitation interventions [3, 4, 5]. Non-invasive brain stimulation encompasses electrical, magnetic, optical, and ultrasound modalities, with electrical stimulation being the most applicable [6, 7, 8].
Transcranial electrical stimulation (tES) for stroke rehabilitation is an emerging therapeutic approach that modulates cortical excitability and induces long-lasting effects, with minimal side effects [6, 9, 10]. tES includes two major modalities, direct current stimulation (tDCS) and transcranial alternating current stimulation (tACS), with the tDCS method predominantly utilized in clinical practice as a standard [11].
tDCS involves the administration of a constant weak direct current via two scalp electrodes to influence cortical excitability in a targeted cortical area [6, 12, 13, 14]. Early investigations of tDCS have focused on the modulation of cortical excitability influenced by polarity, producing contrasting effects on neuronal excitability [12, 13, 14]. Anodal tDCS increases cortical excitability, whereas cathodal tDCS decreases it [6, 12, 13].
tACS applies a mild sinusoidal alternating electric current at a designated frequency across the scalp to modulate brain activity and function as neural oscillation entrainment [15, 16, 17]. tACS is sometimes regarded as a variant of transcranial magnetic stimulation delivered at a predetermined frequency [17]. Cortical excitability, according to anodal and cathodal currents, can be clearly distinguished in tDCS; however, in tACS, the effects on cortical excitability are intricate because of the interplay between cortical excitation and inhibition [12, 13, 15, 18].
Despite many promising clinical trials of tES for stroke rehabilitation, either alone or in combination with other therapies, the number of study showing minimal or different effects remains a concern for its clinical application [7]. Moreover, the mechanisms underlying its beneficial effects remain to be elucidated. tDCS and tACS resemble electrical stimulation; however, because the electricity used differs between direct and alternating currents, the underlying mechanisms in the brain may manifest differently. tES may have therapeutic effects not only through cortical excitability induced by electrical stimulation but also via various biological mechanisms or processes. Elucidating these putative pathways is essential for the development of clinical interventions and therapeutic strategies. A literature search was conducted on PubMed using the terms “tES”, “tDCS”, and “tACS” alongside “stroke” to provide the transparency and reproducibility necessary for this mechanism-oriented review. Exclusion criteria were rigorously enforced for literature that solely presented clinical outcome measurements without significant mechanistic analysis or research that predominantly concentrated on non-electrical brain stimulation methods unless they elucidated a fundamental tES mechanism. Therefore, this study aimed to review the mechanisms of tDCS and tACS, with a focus on stroke rehabilitation.
Following a unilateral stroke, the inhibitory interaction between the cerebral hemispheres is impaired through the transcallosal pathway [19, 20]. The lesioned ipsilateral hemisphere exhibits reduced excitability, whereas the undamaged contralateral hemisphere becomes hyperexcitable, disturbing the normal balance of interhemispheric inhibition via an excessive influence on the stroke-affected side. This phenomenon is referred to as interhemispheric imbalance, or the interhemispheric competition model [19, 21, 22]. The ipsilesional hemisphere remains inactive owing to continuous inhibition of the symmetrical healthy hemisphere, potentially worsening functional impairment for the patient [5, 19, 22].
Direct current electrical stimulation can modulate cortical excitability by modifying the membrane potential threshold through the involvement of N-methyl-d-aspartate (NMDA) receptors [12, 23, 24]. Clinically, anodal stimulation leads to neuronal depolarization, whereas cathodal stimulation leads to hyperpolarization [6, 12, 13]. Cortical excitability is crucial to restore brain function, with an equilibrium between excitatory glutamatergic and inhibitory gamma-aminobutyric acid-expressing (GABAergic) neurons [7, 23, 25]. Following stroke, neuronal inhibition increases in the peri-infarct region, which is mediated by gamma-aminobutyric acid (GABA) A receptors, resulting from impaired GABA transporter function [7, 26, 27]. This indicates that the interhemispheric imbalance is not solely a decrease in neuronal excitability but rather a neurochemical mechanism involving dysregulation of GABA tone. To correct this asymmetry, anodal tDCS is applied to the ipsilesional hemisphere to enhance cortical excitability, whereas cathodal tDCS is administered to the contralesional hemisphere to suppress excitability, with both approaches potentially combined through simultaneous bihemispheric stimulation [6, 21, 28, 29]. These configurations reduce the inhibitory influence of the intact hemisphere on the lesioned hemisphere.
To optimize the alleviation of interhemispheric imbalance, the integration of anodal tDCS and bihemispheric tDCS with constraint-induced movement therapy is utilized [30, 31, 32]. tDCS stimulation in each hemisphere varies according to polarity; nonetheless, a reduction in GABA levels has been noted [33], and the influence of tDCS on motor performance is negatively correlated with GABA concentration [34]. tACS in both hemispheres can phase-specifically modulate connections between brain regions within the corresponding frequency band; however, no studies have applied this to stroke [35].
The interhemispheric competition model serves as a valid mechanistic basis for the efficacy of tES treatment post-stroke by readjusting the balance of interhemispheric inhibition. However, the treatment of stroke with tES needs to be performed cautiously, considering the polarity, dose, and configuration of the electrodes based on the pathophysiological framework of stroke, as these factors yield varying clinical outcomes [10, 36, 37]. The application of anodal stimulation in the early stages of stroke considers the inflammatory response of the brain, which adversely affects clinical outcomes [10, 38], whereas cathodal tDCS facilitates superior clinical recovery in this stage [10, 39, 40]. Moreover, excessive stimulation of GABA signaling during stroke recovery diminishes neuronal excitability and impedes recovery [27, 41]. If tES is applied uniformly in this model without considering the pathological frameworks at different stages after stroke, it is likely to yield different clinical outcomes [7].
A novel neural rehabilitation model, termed the bimodal balance-recovery model, was introduced to address the limitations of the oversimplified interhemispheric imbalance model, incorporating interhemispheric inhibition and structural reserve post-stroke [7]. When the structural reserve is high, the affected hemisphere can restore normal interhemispheric balance. However, when the structural reserve is low, the contribution of the contralesional hemisphere may be beneficial; this mechanism is referred to as vicariation or interhemispheric compensation [42]. Intact residual networks can compensate for the injured hemisphere through brain reorganization; hence, activation of the healthy hemisphere may represent an effective therapeutic strategy to promote functional recovery post-stroke [7, 43, 44]. The proposed model facilitates a patient-specific approach to brain stimulation tailored to the neural architecture of individual patients [7].
Clinical evidence from transcranial magnetic stimulation has questioned the ability of the interhemispheric competition model to modulate abnormal transcallosal inhibition, based on the assumption that all patients belong to a single cohort [45, 46, 47]. There were no signs of changes in the interhemispheric imbalance or contralesional hemisphere hyperexcitability [45, 47, 48, 49]. Interhemispheric inhibition and baseline impairments exhibit bimodality, allowing for a criterion impairment level for patient classification based on the bimodal effects of the contralateral brain [49].
In patients with moderate-to-severe impairment, enhancing contralesional excitability with anodal tDCS or bilateral stimulation can improve motor performance [50, 51]. Ipsilesional excitability initially diminishes and then increases, correlating with impairment and function; however, interhemispheric inhibition remains symmetrical [45]. In a rodent stroke model, the contralesional cortex exhibited anatomical adaptation in response to stroke size [52]. Patients with severe stroke primarily relied on the undamaged hemisphere for functional recovery, whereas those with minor stroke did not [53]. Application of anodal tDCS to the contralesional cortex shows markedly enhanced recovery relative to ipsilesional stimulation. Therefore, the intact hemisphere can facilitate the recovery of impaired function [54, 55]. These findings demonstrate that the role of the contralesional hemisphere in healing increases with lesion severity. To restore interhemispheric activity, tES therapeutic options are selected according to the degree of individual damage.
The brain inherently displays rhythmic activity at various frequencies—waves
Externally applied electric fields modify the evoked network activity, and entrainment-based tACS can be synchronized to normalize endogenous oscillation patterns at a relevant frequency [15, 60, 61]. Periodic electrical stimulation of the tACS interacts with normal oscillations in the brain, synchronizing neuronal firing rhythms to the corresponding frequency [15, 16, 62]. tDCS, measured by electroencephalography, modulates oscillatory activity, which is a marker and facilitator of neuroplasticity [63, 64]. tDCS does not directly enforce the inherent frequency in the brain; rather, it indirectly modifies endogenous oscillations in the brain through long-term alterations in neuronal spike rates and synaptic activity [44, 65, 66].
Clinical studies support the potential of tES in altering neuronal networks via synchronization and coordination mechanisms [67, 68, 69]. In animal studies, tACS corrects atypical neuronal firing patterns by resynchronizing neurons to a healthy rhythm [70, 71]. The tDCS-induced interhemispheric brain coherence in the low gamma frequency range promotes functional recovery after stroke [72]. Therefore, the therapeutic benefits of administering tES are related to the recovery of healthy brain oscillations and the restoration of interregional coherence within cortical networks.
Neuroplasticity is the unique capacity of the brain to create new synapses and reorganize neural networks in response to stimuli. Following a stroke, this mechanism is essential for the continuous regeneration of injured brain tissue. The basic theoretical premise of tES, which underlies its significant therapeutic efficacy, indicates the reorganization of brain circuits [17]. Long-term therapeutic benefits are produced via polarity-specific long-term potentiation (LTP), which is triggered by tES-induced intracellular calcium levels and forms connections with various downstream pathways [29, 54, 55, 73, 74].
Moreover, successive tES stimulation causes initial cortical excitability, which is then transmitted to subcortical areas by activating NMDA receptors, intracellular calcium cascades, and other pathways [13, 14, 75]. Connection-based diffusion from the cerebral cortex to the subcortical areas following tES may be correlated with the therapeutic outcomes of functional recovery in stroke. Similar to neuroplasticity in the healthy brain, a time-limited window exists post-stroke to enhance functional recovery, which can be strengthened and reorganized through training and appropriate stimuli [5, 44].
tES promotes neuronal pathways that improve neuroplasticity and functional reorganization in injured brains [76]. Furthermore, combinations with other rehabilitation programs have a synergistic effect and improve therapies based on Hebbian plasticity [76, 77, 78, 79]. In animal studies, tDCS exerts neuroprotective effects by maintaining neuroplasticity by modifying genes linked to plasticity, neuroplastic biomarkers, and brain circuits [80, 81, 82]. Brain-derived neurotrophic factor (BDNF)-tropomyosin-related kinase receptor B (TrkB) signaling is involved in the therapeutic effects of tDCS on functional recovery and neuroplasticity [83, 84].
Neuroplasticity, a key feature of functional recovery after stroke, results from processes that facilitate the reorganization of brain networks. Thus, by providing stimulation in response to brain activity, tES-induced neuroplasticity may be similar to activity-dependent therapy [85, 86, 87]. tES augments the neuroplasticity of the brain, facilitating the recovery of functions in neural circuits impaired by stroke via the formation of new synapses. When combined with rehabilitative therapy, it promotes restoration of various functions, restoring and compensating for pre-injury functions through additional processes, including Hebbian plasticity, homeostatic metaplasticity, and vicariation.
Hebbian plasticity, proposed by Donald Hebb in 1949, is founded on the premise that “neurons that fire together, wire together” [88]. This theory offers a conceptual framework to elucidate how experience and learning facilitate the adaptation and enhancement of neural networks [89, 90]. Post-stroke, patients reacquire motor learning with repetitive practice similar to that of healthy individuals, indicating synaptic reinforcement at the cerebral level within the motor network based on Hebbian plasticity [91]. Synaptic connections are reinforced by the simultaneous activity of two neurons, which encompass coactivation, LTP, and long-term depression (LTD). Similar to LTP and LTD, repeated tES sessions cause long-lasting changes in synaptic efficacy via NMDA receptor-mediated calcium dynamics, resulting in the insertion or removal of synaptic receptors [92, 93, 94].
Clinical tDCS enhances Hebbian plasticity during task performance, particularly when administered in a task-specific manner that interacts with endogenous plasticity [95, 96]. Simultaneous stimulation of the motor cortex and peripheral nerves using paired associative tACS improves the corticospinal excitability of the motor neurons, eliciting an LTP-like response [97]. Providing temporally synchronized stimulation tailored to a patient’s movements or cerebral activity enhances the synaptic connections of the remaining neural pathways and intensifies learning effects. Therefore, the integration of other therapies, such as consistent exercise with tES, may facilitate the reorganization of the cerebral cortex and markedly improve functional recovery in patients with stroke.
Although Hebbian plasticity is a fundamental component of brain learning and memory, it can destabilize neural circuits if operating excessively or irregularly [98]. Homeostatic metaplasticity is a self-regulatory mechanism that modulates LTP or LTD in response to prior synaptic activity, thereby preventing excessive excitation or inhibition and maintaining brain activity within the physiological range [98, 99]. When numerous reinforcement processes occur, they exhibit a reduced sensitivity to further reinforcement.
The Bienenstock–Cooper–Munro (BCM)-like plasticity model, which dynamically adjusts the plasticity threshold according to the activation history, has been elucidated [100, 101, 102]. This mechanism can improve recovery outcomes after stroke by efficiently regulating synaptic responses based on the brain’s previous activity state and tES intervention. To prevent metaplastic inhibition, excitatory inputs must be modulated or delayed after the completion of a high-intensity task. In contrast, short-term tDCS before training can increase plasticity of the motor cortex, which magnifies synaptic changes during subsequent practice. Homeostatic metaplasticity offers a theoretical framework for the significance of the timing, dosage, and sequencing of tES and other training [102, 103]. Preconditioning the brain with cathodal tDCS enhances alterations in neuronal excitability caused by anodal tDCS or intermittent theta burst stimulation [103, 104]. Following cathodal tDCS, mesh glove stimulation can be used to modify motor cortex excitability in metaplastic modulation [105]. The concurrent application of anodal tDCS and neuromuscular electrical stimulation failed to yield the anticipated synergistic effect, which was potentially attributable to the influence of homeostatic plasticity [106]. However, tDCS does not significantly improve motor fatigue or excitability regulation in healthy adults, nor does it affect neuroexcitability, indicating a limited influence of metaplasticity on implicit motor learning [107, 108, 109].
The tES treatment of stroke promotes neuroplasticity in the injured brain by utilizing the principles of Hebbian plasticity and homeostatic metaplasticity. tES enables the simultaneous activation of neurons, reinforcement of activated neurons to restore functional connections, or reduction of excessive excitability to preserve the stability of the neural network. These mechanisms may alter the timing and intensity of tES to optimize neuroplasticity.
tES facilitates motor recovery following stroke by stimulating the release of growth and neurotrophic factors that activate neuroplasticity, neuronal survival, and brain reorganization [29, 54, 83, 110]. tDCS activates calcium channels, which trigger the activation of downstream cascades that promote the production of growth factors [13, 29, 83, 111]. The brain contains numerous pathways composed of axonal fibers that connect various cerebral regions [112]. Thus, tES elevates intracellular calcium levels in the non-stimulated cortical hemisphere, indicating that calcium may trigger the release of neuroprotective factors at non-stimulated sites [29, 54, 73, 113].
tDCS increases neuroplasticity within the motor cortex through BDNF secretion and TrkB activation, which are crucial mechanisms to improve neurological function after stroke [29, 114]. The BDNF genotype in patients with stroke affects motor learning capability, as people with the atypical BDNF genotype show reduced efficacy in enhancing motor learning by tDCS [115, 116]. However, the therapeutic benefits of tDCS may differ according to catechol-O-methyltransferase genotype polymorphism, whereas those of BDNF may not [117]. A reduction in BDNF levels may have positive effects on recovery, highlighting the need for electrode positioning, polarity adjustments, and personalized intensity [70].
tDCS facilitates BDNF-TrkB signaling and functional recovery, with multifaceted biological mechanisms contributing to brain recovery and regeneration beyond simple electrical stimulation [29, 83, 84, 110]. tDCS provides a molecular basis for neuroplasticity by modifying neuroplasticity-related genes, such as BDNF, cyclic adenosine monophosphate (cAMP) response element-binding protein, synapsin I, and calcium/calmodulin-dependent protein kinase II [80, 81]. tDCS elevates BDNF levels and indicators of axonal regeneration in the peri-infarct cortex, along with neuroprotection and neuroplasticity facilitated by the BDNF/TrkB–phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt) pathways [118, 119]. Moreover, the continuous use of tDCS upregulates the expression of activity-dependent growth factors, such as BDNF, growth/differentiation factor 5 (GDF5), and platelet-derived growth factor subunit A (PDGFA) [54, 118]. These factors govern the proliferation and differentiation of oligodendrocyte progenitors, thereby facilitating functional recovery [54, 120, 121]. tDCS/tACS treatment regulates inflammatory cytokines in microglia as well as the activation of neurotrophic factors, such as BDNF and fibroblast growth factor 9 [70].
Neurotrophic factors stimulate the proliferation, differentiation, and survival of neural stem cells, facilitating neuronal formation and maturation [122, 123]. From a regenerative medicine perspective, tDCS activates adult neurogenesis regardless of its polarity; cathodal tDCS sometimes has an even greater effect [110, 124, 125]. Additionally, tDCS induces the differentiation of neural stem cells by inhibiting Notch1 signaling [126]. Furthermore, anodal tDCS treatment promotes cell proliferation in the ipsilesional striatum, subventricular zone, and corpus callosum [54].
One of the main mechanisms by which tES promotes neuroplasticity in stroke rehabilitation is the upregulation of the expression of growth factors, specifically BDNF. This highlights the essential functions of tES in neuronal survival, synapse formation and reformation, axonal growth, and restoration of connections between neurons, thus facilitating the reorganization and functional recovery of impaired brain circuits.
Diaschisis is a condition characterized by reduced brain activity in regions not directly affected by injury following stroke. It is reversible and gradually recovers over time and primarily occurs in regions that are structurally and functionally linked to the affected area owing to the loss of input and rapid neurochemical alterations [127, 128]. Vicariation, the functional reorganization of the brain in which diaschisis is irreversible and permanent, occurs when a permanently impaired part of the brain is functionally replaced by another area [129, 130]. The adult brain undergoes considerable structural and functional reorganization after injury, which is crucial for recovery [131, 132].
Diaschisis and vicariation during stroke recovery, and alterations such as disruption or compensation within functional networks, are closely related to cerebral blood flow (CBF) and neurometabolism, including the rates of oxygen and glucose consumption. The diaschitic region exhibits a reduction in CBF linked to the metabolic rate with no physical damage, whereas neuronal activity and blood flow serve as crucial indicators of functional reorganization of the brain [127, 129]. This entails the synchronized function of neurons, astrocytes, and vascular cells; however, neurovascular coupling (NVC) is impaired after a stroke [133]. The NVC regulates neuronal activity and CBF via coordinated signaling across many cells [134]. During the recovery phase, rehabilitation training or tES influences CBF, which, in turn, affects neuroplasticity and neuronal reorganization, leading to functional recovery.
Clinical trials have been conducted to investigate the interaction between nerves and blood vessels through tES because it may affect CBF velocity [51, 135]. tDCS modulates CBF and intracranial compliance, alleviating hemodynamic load on the affected hemisphere [136, 137]. In contrast, tACS alters cerebral hemodynamics, including enhanced blood flow velocity and diminished blood flow resistance [138]. The response of blood flow velocity to tDCS differs among individuals, which is attributable to variations in baseline neuronal excitability and cerebral blood velocity [135]. The integration of tDCS and computer-aided training effectively modulates CBF in patients with stroke [139]. Ischemia results in an imbalance in cerebral oxygen saturation, which is strongly correlated with EEG readings; however, this imbalance, resulting from cerebral vasomotor reactivity, is restored by tDCS [136, 140, 141, 142, 143]. In animal studies, tDCS reduced both the incidence and severity of cerebral vasospasms, with alterations in blood flow depending on polarity [144, 145, 146]. tACS affects CBF, leading to increased blood flow at specific frequencies [147, 148].
These results indicate that tDCS may elicit functional CBF responses by enhancing vasodilation, and tACS indirectly influences CBF via frequency-dependent modulation of neural rhythms. tES modulates CBF during stroke rehabilitation and is linked to neurometabolic activity that enhances the delivery of oxygen and nutrients to the affected areas or other regions. The augmentation of CBF may be associated with the restoration of NVC and the enhancement of neuroplasticity, which play a role in the remodeling of functional networks post-stroke.
Stroke markedly disrupts neurometabolism in the brain. Hence, cerebral reorganization or functional recovery refers to the restoration of the brain’s energy system [149, 150]. Restoration of ATP synthesis, mitochondrial activity, and metabolic conditions, together with NVC, is crucial to maintain the functionality of the recovered neural network [134, 151, 152]. Therefore, therapeutic strategies for stroke should employ a comprehensive approach that integrates functional induction with metabolic support, including the optimization of mitochondrial activities. The tES approach may enhance energy utilization and recovery in neuronal cells; however, data on its impact on mitochondrial activation and dynamics are limited.
The administration of tDCS to healthy individuals elevates cerebral energy
consumption, leading to a temporary state of energy depletion, which causes
changes in systemic glucose metabolism and related hormones [153]. Nonetheless,
tDCS therapy in patients with stroke does not alter brain metabolite
concentrations [154]. In preclinical animal study, tDCS decreases mitochondrial
damage and preserves mitochondrial quality control signaling in neurons [155].
This indicates that tACS exhibits mitochondrial activity, which is linked to
therapeutic advantages [151, 152, 156]. This suggests that tACS, similar to
exercise therapy, affects neurotrophic factors, such as BDNF signaling, perhaps
improving mitochondrial complex subunits and function through the peroxisome
proliferator-activated receptor gamma co-activator (PGC-1
Restoring and stabilizing the metabolic environment is essential for the efficacy of stroke rehabilitation and is necessary to optimize these effects from the early stages of stroke until recovery [149, 150]. Employing tES to restore and regulate brain metabolism may facilitate functional recovery by enhancing neuroplasticity and reorganization. This function is likely associated with enhanced blood flow metabolism, and further investigation is required.
Following ischemic stroke, a severe reduction in CBF leads to an infarct zone characterized by a damaged ischemic core, resulting in irreversible necrosis along the surrounding penumbra [159]. Clinical research supports the prevention of neuronal death by positioning the penumbra/peri-infarct region, which is defined by a partially preserved blood supply and temporary neuronal viability, and is a pivotal concept in stroke neuroprotection [160, 161, 162]. Major research targets in stroke, including excitotoxicity, oxidative stress, apoptosis, and inflammation, are all related to neuronal survival in the penumbra [163, 164]. Cathodal tDCS therapy in patients with stroke exhibits a potentially beneficial effect by reducing the extent of ischemic and infarct regions through the inhibition of excitotoxic effects and penumbral salvage [39, 40].
Preclinical animal studies on changes in cerebral infarction through the regulation of inflammation and neural death are more diverse than clinical studies. tDCS enhances activated microglia in both anodal and cathodal configurations, eliciting an innate immune response [165]; however, cathodal tDCS facilitates superior recovery by modulating neuroinflammatory responses and apoptosis [10, 166, 167]. The polarity of tDCS, especially that of cathodal tDCS, reduces cell death through the alleviation of inflammatory markers and oxidative stress and the regulation of NMDA receptors [168, 169]. tDCS promotes neuronal survival via the Cezanne-SIRT6-DNA damage regulation pathway or via DNA fragmentation [170, 171]. The anti-inflammatory response and enhancement of nerve plasticity and angiogenesis associated with tDCS are facilitated by the cyclic guanosine monophosphate-adenosine monophosphate (cGMP-AMP) synthase stimulator of the interferon gene pathway [172]. Cathodal tDCS suppresses ferroptosis through the dual mechanisms of mitophagy inhibition and nuclear factor erythroid 2-related factor 2 (Nrf2) activation [167].
Electrical stimulation at 20 Hz diminishes neuroinflammation and apoptosis-related markers via the BDNF/TrkB and PI3K/Akt/mTOR pathways [173], whereas 20 Hz tACS modulates 11 genes typically associated with apoptosis and inflammatory responses, along with tDCS [70]. Research on the effects of tDCS on detrimental factors in stroke has demonstrated its neuroprotective effects via the regulation of propionic acid [174], 3-oxo-lithocholic acid [175], isoleucine [176], and surplus locus protein 4 [177]. The effects of tDCS vary according to the polarity, and its application during the ischemia and reperfusion stages can markedly improve neuroprotection against cerebral injury [169, 171].
tDCS treatment exerts therapeutic effects by inhibiting neuronal damage and
reducing the levels of inflammatory factors via the NMDA-mediated sterol
regulatory element-binding protein 1 (SREBP1) pathway [55]. tDCS/tACS treatments
also commonly regulate stroke brain immune-related targets in response to
damage-associated molecular patterns (DAMPs), including DAMPs (S100
calcium-binding protein A9 (s100a9)) and its receptor (toll-like receptor 4
(Tlr4) and myeloid differentiation primary response 88 (Myd88)), phagocytosis
receptor (triggering receptor expressed on myeloid cells 2 (Trem2)), signaling
cascade (signal transducer and activator of transcription 1 (Stat1) and nuclear
factor kappa-light-chain-enhancer of activated B cells (NFkB)), and cytokines
(interleukin (IL)-6 (IL-6), IL-1
Cathodal tDCS demonstrates superior neuroprotection in relevant stroke models. This may be due to the modulation of neuroinflammatory pathways that provide neuroprotective effects, particularly during the acute and subacute periods after ischemia. Following a stroke, tES modulates inflammation-related mechanisms to mitigate excessive responses and safeguard the surrounding tissues. Furthermore, its anti-inflammatory effects enhance neuronal survival by regulating various apoptotic and survival-related factors. The anti-neuroinflammatory properties of tES are important in preventing brain injury and promoting functional recovery during rehabilitation.
tES focuses on neuroplasticity and network reorganization by modulating cortical excitability and related processes; however, the possibility of indirect involvement of the peripheral nervous system (PNS) has been raised. In humans and rodents, tES accounts for approximately 75% of the current applied to the scalp, owing to the soft tissue and the skull, making it insufficient to entrain neural activity [178, 179]. To stimulate the cortex, either a higher intensity current than conventional tES is employed [178], or different high-frequency currents are delivered to the brain to create temporal interference at different frequencies, selectively stimulating the deep brain [180]. Because of the transcranial and transcutaneous stimulation, the effect of tACS on the motor system can be elicited solely by transcutaneous stimulation [179].
Involvement of the PNS may be an important consideration in the design of tES therapy. The effects of tES can be modulated or enhanced by the PNS, and synergistic outcomes can be achieved by combining central and PNS stimulations [181, 182]. Similar to cortical excitability in the tES, calcium channels involving NMDA receptors are important signaling components that represent the therapeutic advantages of PNS [70, 183]. Recent studies have indicated the possible indirect effects of tDCS via the trigeminal nerve [184, 185] and have highlighted the role of the vagus nerve in the outcomes of tDCS [186].
Clinical and physiological parameters improve when tDCS and PNS are applied simultaneously, proving to be more effective than when used in isolation [181, 187, 188]. Nonetheless, it is equally accurate that combined stimulation does not invariably exhibit additional effects or consistently reveals enhanced efficacy [108, 189]. Electrical stimulation techniques, including tDCS, tACS, and electroacupuncture, positively influence motor function in a stroke model via a shared mechanism that modulates neuronal cell death, regardless of whether the stimulation targets the cerebral cortex or the peripheral somatosensory input [70]. Therefore, these therapeutic interventions can induce both bottom-up and top-down bidirectional neural activation, leading to synergistic advantages [182].
We investigated the therapeutic effects of tES in stroke rehabilitation, including cortical excitability, neuroplasticity, neurovascular and neurometabolic stability, inflammatory regulation, and the role of peripheral nerves. These categories of molecular mechanisms indicating therapeutic effects interact interdependently, going beyond simple cortical excitability. This dynamic interaction forms a complex loop that enhances its therapeutic benefits (Fig. 1A). tDCS normalizes cortical connections disrupted by stroke through neural rewiring, leading to the recovery of motor function [190]. Enhancements in motor and cognitive functions correlate with elevated expression of growth factors, including GDF5 and PDGFA, along with neurogenesis in the subventricular zone [54]. Motor function recovery includes activation of the BDNF/TrkB pathway, synaptic remodeling, restoration of brain connections, and potential enhancement of angiogenesis [83]. For recovery of motor and cognitive functions, NMDA-dependent SREBP1 signaling and related inflammatory factors, such as cyclooxygenase-2 (COX-2) and Akt, are involved [55]. Therefore, the modulation of neuronal excitability elicited by tES facilitates neuroplasticity and the secretion of neurotrophic factors, such as BDNF, thereby maintaining long-term functional recovery. They also diminish neuroplasticity deficits along with anti-neuroinflammation and reorganization through neurovascular and neurometabolic stability.
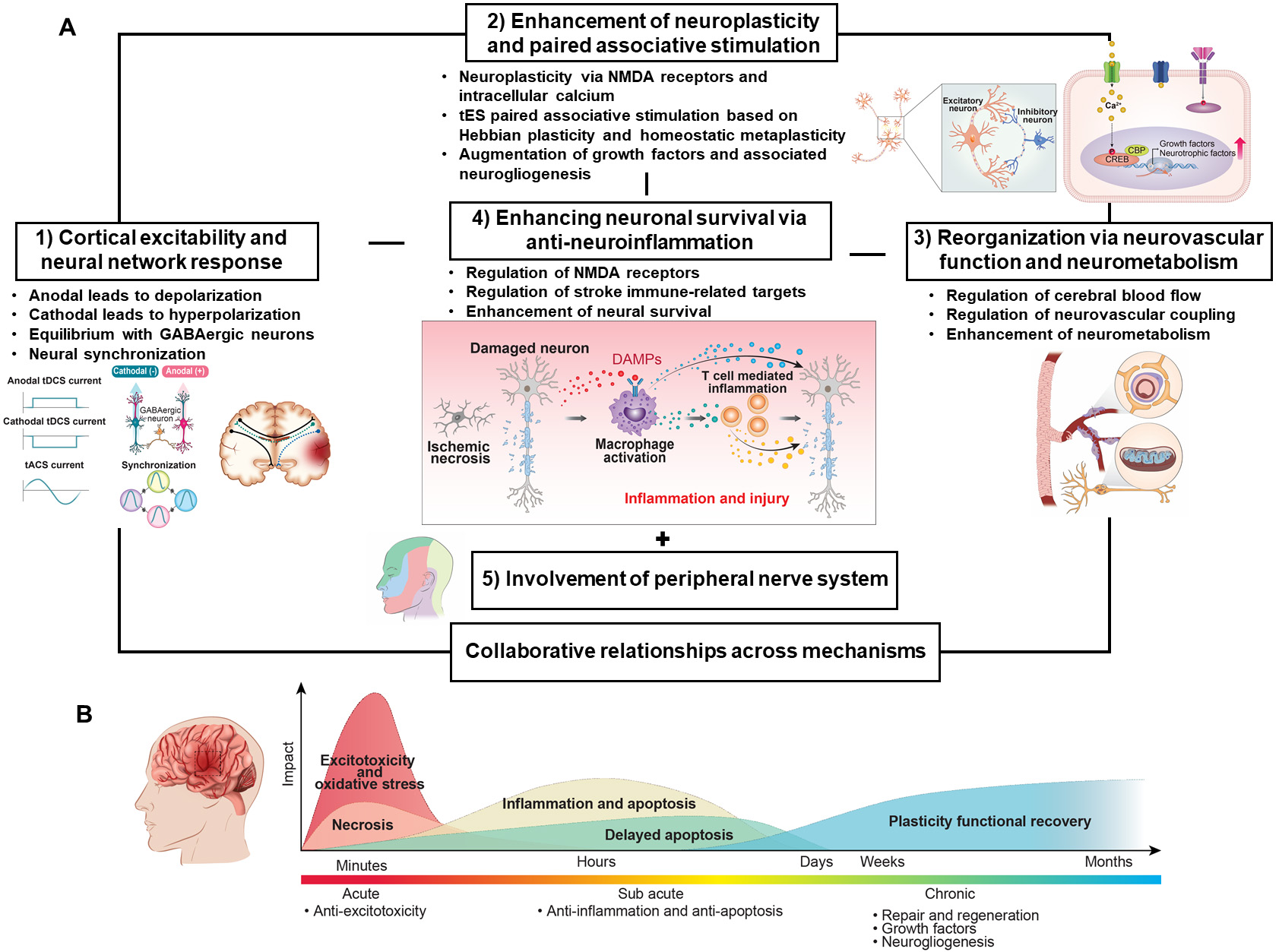 Fig. 1.
Fig. 1.
Therapeutic mechanisms of tES in stroke rehabilitation. (A) Therapeutic mechanisms of tES in stroke rehabilitation. (1) Cortical excitability, a crucial factor in brain function recovery through NMDA receptors, is induced by anodal and cathodal stimulation, leading to neural depolarization and hyperpolarization, respectively. This is influenced by the balance between excitatory glutamatergic and inhibitory GABAergic neurons. Depending on the severity and stage of the stroke, it is applied to either alleviate interhemispheric imbalance or promote interhemispheric compensation. Additionally, synchronizing the unique brain rhythmic activity that appears as the firing timing of neuron groups is also an essential mechanism for brain function recovery. (2) Successive tES stimulation induces long-lasting neuroplasticity by activating NMDA receptors and the intracellular calcium cascade, thereby facilitating the reorganization of brain networks. The tES can be influenced by Hebbian plasticity and homeostatic metaplasticity to optimize neuroplasticity. tES regulates the expression of neuroplasticity-related genes, as well as growth factors and neurotropic factors, through intracellular calcium signaling and the downstream cascade. (3) Vicariation in stroke, a functional reorganization of the brain, is associated with the regulation of CBF and neurometabolism by tES. CBF regulation, which responds to the energy required for brain, is controlled by NVC, and blood flow is associated with increased neurometabolic activity. (4) tES promotes cell survival by regulating stroke brain immune-related targets, including NMDA receptors, DAMPs and their receptors, phagocytic response, and inflammatory cytokines. The enhancement of neuronal survival through anti-inflammatory mechanisms, particularly in the penumbra/peri-infarct area, is crucial. (5) Due to the fact that most of the current from tES is lost as it passes through the skull, the therapeutic effect may be accomplished by stimulating the trigeminal nerve, a peripheral nerve, on the scalp. (B) The therapeutic effects of tES in stroke rehabilitation are linked to multiple processes. Different strategies are required for functional rehabilitation depending on the stroke stage. Owing to the potential for diverse clinical outcomes from the application of varying tES parameters, such as polarity, frequency, intensity, duration, electrode configuration, stimulation target, and session frequency, a careful design is required. tES, transcranial electrical stimulation; NMDA, N-methyl-d-aspartate; GABAergic, gamma-aminobutyric acid-expressing; CBF, cerebral blood flow; NVC, neurovascular coupling; DAMPs, damage-associated molecular patterns.
There are still gaps in the knowledge regarding the stimulation protocols, parameters, long-term effects, and basic mechanisms of tES in relation to stroke rehabilitation. This highlights the inapplicability of animal models in humans and their disconnection from therapeutic practices. However, there are questions regarding whether tES can activate the cortex by passing through the skull. Rodent studies have used higher intensities than human studies, even when considering head size, complicating the direct application to clinical scenarios [191, 192, 193, 194]. Clinically, field intensities higher than those employed in traditional tES guarantee consistent cortical excitability in the human brain [178, 193].
tES treatment requires enhanced precision in current administration and spatial focality in the cerebral cortex. The traditional large rectangular pad induces nonfocal current diffusion [195, 196] thus, small ring-based high-definition (HD) electrodes are employed to overcome this [197, 198]. The modified HD electrode with integrated needles enhances the focus relative to traditional HD electrodes; however, it exhibits limitations in accurate stimulation [55]. Moreover, newly developed electrodes and excessive stimulus intensity intended to enhance cortical activity and local stimulation may induce uncomfortable sensations such as edge effects; therefore, further clinical and animal studies are needed to address this problem [199, 200].
The goal of post-stroke treatment to date has been to reduce the initial impact and complications and to maximize functional ability through comprehensive physical therapy, employing a strategy aimed at activating a limited window of neuroplasticity during the early stages [5, 7, 44]. However, depending on the stage after stroke, functional recovery is achieved through stage-specific modulation of inflammation, neuroplasticity, NVC, network reorganization, and structural remodeling [44, 83, 201]. The design approach for tES needs to be varied based on the stage of the stroke because it may produce different outcomes depending on parameters, such as polarity, frequency, intensity, duration, electrode configuration, stimulation target, and number of sessions (Fig. 1B). tES is non-invasive and is expected to promote functional recovery in patients with stroke through various mechanisms with repeated applications. However, careful prescription design of tES is required, considering its parameters and the post-stroke stage. Along with the standardization of each parameter, analysis of the long-term effects of repeated applications and personalized stimulation combined with other treatments will lead to improved tES strategies.
All authors contributed significantly to this work. HJL, JHK, and BTC concept and design; HJL, HKS, YIS, JHK, and BTC search and critical review of literature; HKS and YIS critical review of the manuscript; BTC drafting of the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by a 2-Year Research Grant of Pusan National University.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

