1 Division of Hematology, Johns Hopkins University School of Medicine, Baltimore, MD 21205, USA
2 Biotechnology in Advanced Academic Programs, Krieger School of Arts & Sciences, Johns Hopkins University, Baltimore, MD 21218, USA
Abstract
Integrin signaling serves as a fundamental regulator in human pluripotent stem cell (hPSC) biology, mediating adhesion, survival, and pluripotency through interactions with extracellular matrix (ECM) components. Specific integrins, including α6β1, αvβ5, and α3β1, engage ECM ligands such as laminin-511/521 and vitronectin (VTN) to sustain hPSC self-renewal. These engagements trigger essential downstream pathways, including PI3K/AKT, MAPK/ERK, focal adhesion kinase (FAK)-Src, and RhoA/Rho-associated protein kinase (ROCK), thereby maintaining the expression of pluripotency factors like OCT4, NANOG, and SOX2 while integrating mechanotransductive cues. FAK and Src convert ECM-derived mechanical signals into biochemical responses, regulating cytoskeletal reorganization, YAP/TAZ nuclear translocation, and context-dependent gene expression. For scalable, xeno-free culture, recombinant substrates such as truncated vitronectin (VTN-N) and laminin-511/521 E8 fragments, paired with defined media (e.g., Essential 8 or mTeSR1), support robust hPSC expansion under good manufacturing practice (GMP) conditions. Extending to differentiation, integrin-ECM crosstalk directs lineage commitment across diverse fates, including hematopoietic, cardiovascular, neural, hepatic, epithelial, endodermal, and oligodendroglial lineages, by fine-tuning signaling specificity and ECM composition. This review focuses on recent advances in the mechanistic interplay between integrin signaling and ECM proteins in hPSC maintenance, mechanotransduction, and lineage-directed differentiation, emphasizing defined culture systems and their translational potential in regenerative medicine.
Keywords
- human pluripotent stem cells
- integrins
- extracellular matrix (ECM)
- pluripotency
- differentiation
- mechanotransduction
Integrin–extracellular matrix (ECM) interactions are central to realizing the translational potential of human pluripotent stem cells (hPSCs) in regenerative medicine, disease modeling, and cell-based therapy. By providing biochemical and biomechanical cues that regulate adhesion, survival, and lineage specification, integrin signaling enables the development of defined culture systems that support large-scale expansion and controlled differentiation of hPSCs into clinically relevant cell types [1]. These integrin-guided processes are essential for generating functional derivatives for cell replacement therapies, constructing physiologically relevant in vitro disease models, and advancing drug discovery platforms. However, key challenges remain for clinical translation, including reducing cultivation costs, eliminating animal-derived components, and optimizing xeno-free ECMs and media formulations compatible with good manufacturing practice (GMP) standards. Addressing these challenges through a deeper mechanistic understanding of integrin–ECM signaling will be critical for achieving reproducible, safe, and scalable hPSC-based therapies.
The goal of this review is to elucidate how integrin signaling orchestrates hPSC behavior, from adhesion and survival to self-renewal and lineage specification, through dynamic interactions with defined ECM components. We aim to summarize current understanding of integrin–ECM-mediated signaling pathways, including PI3K/AKT, MAPK/ERK, focal adhesion kinase (FAK)–Src, and RhoA/Rho-associated protein kinase (ROCK), and their integration with mechanotransductive regulators such as YAP/TAZ. By focusing on how specific integrin–ligand pairs govern hPSC pluripotency and differentiation, we seek to define the molecular principles that enable xeno-free, scalable hPSC culture and guide lineage-specific differentiation. Ultimately, this review aims to advance strategies for engineering ECM-based microenvironments that enhance the safety, efficiency, and translational potential of hPSC-derived cell therapies.
In hPSCs, highly expressed integrins, such as
hPSCs also are highly sensitive to ECM stiffness, with FAK-Src signaling tuned
to softer substrates (e.g., laminin-based matrices) compared to MSCs, which
respond to a broader range of stiffness via
Integrin signaling governs hPSC behavior by transducing ECM-derived cues into
intracellular responses that regulate adhesion, survival, pluripotency, and
differentiation. Integrins are transmembrane heterodimers composed of one
| Category | Examples | Binding specificity |
| RGD-binding integrins | Recognize the arginine-glycine-aspartic acid (RGD) motif found in many extracellular matrix (ECM) proteins, such as fibronectin (FN), vitronectin (VTN), and fibrinogen. | |
| Leukocyte cell-adhesion integrins | Primarily expressed on white blood cells and mediate cell-to-cell adhesion by binding to intercellular adhesion molecules (ICAMs) and other counter-receptors on endothelial cells. The | |
| Collagen-binding integrins | Recognize specific GFOGER (O = hydroxyproline) sequences in collagen, the most abundant protein in the ECM. | |
| Laminin-binding integrins | Mediate cell adhesion to laminin, a major component of the basement membrane. Some of these integrins, like |
The functional versatility of integrins arises from their ability to pair with
multiple subunits, bind diverse ligands, and activate overlapping intracellular
pathways. Some integrins occupy more than one functional group or have
overlapping ligand specificities. For example,
Integrins are crucial mediators of mechanotransduction, the process by which
cells sense and respond to mechanical forces from their ECM and neighboring cells
[25]. In hPSCs, integrins bind to ECM components to create a physical and
biochemical link between internal cellular cytoskeleton and its external
surroundings, and act as mechano-sensors, detecting cues like substrate
stiffness, shear stress, or ECM topography. These signals are transduced through
pathways such as FAK and Src, modulating cytoskeletal organization, gene
expression, and cell fate decisions (Fig. 1) [26, 27, 28]. For example, integrins
(e.g.,
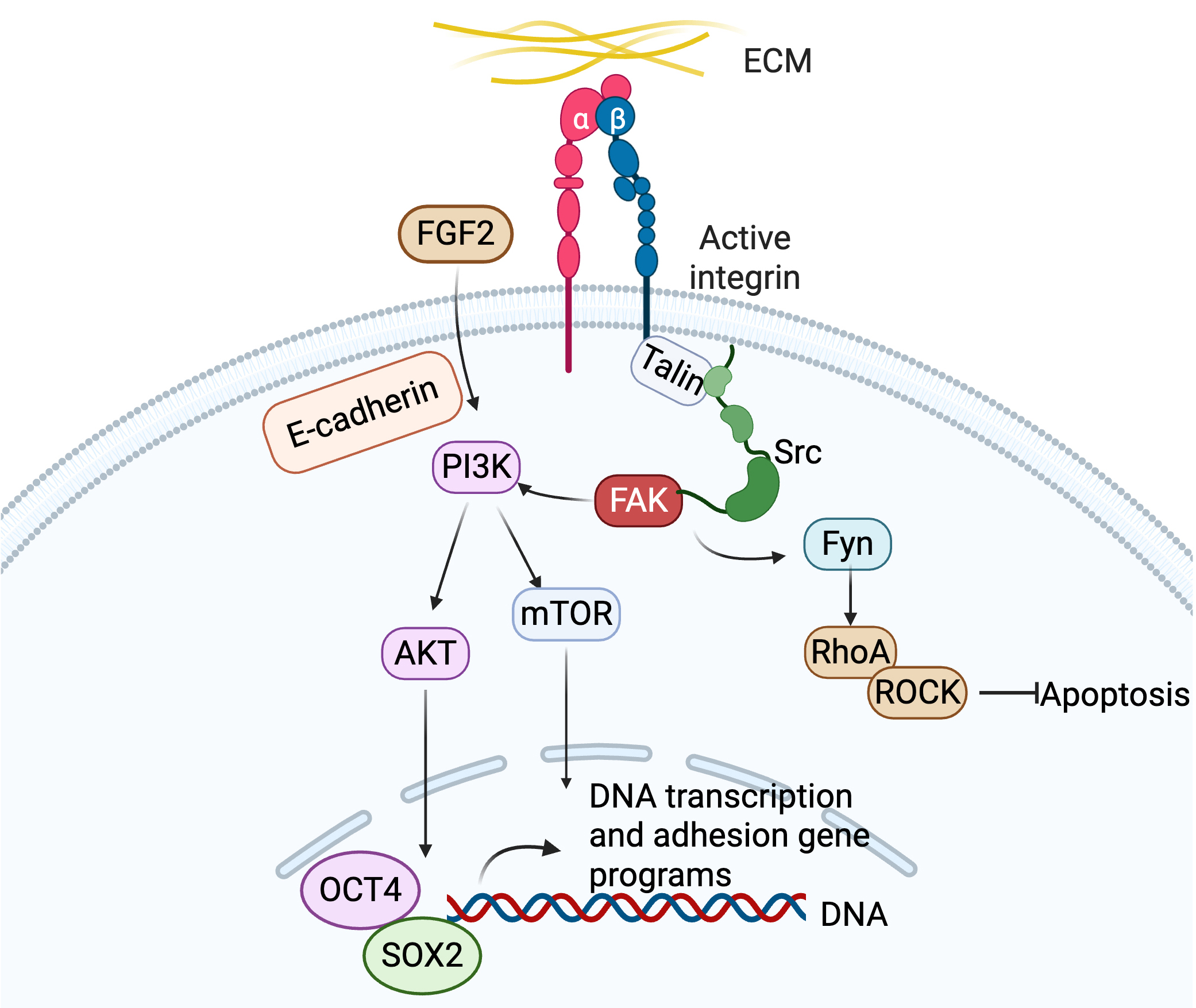 Fig. 1.
Fig. 1.
Activation of integrin signaling by ECM proteins to regulate hPSC adhesion, pluripotency and differentiation. ECM, extracellular matrix; hPSC, human pluripotent stem cell. Created in BioRender (https://BioRender.com/b1mqg26).
FAK activation is one of the major integrin-mediated signaling events.
Mechanical forces or ECM binding induces integrin clustering, recruiting FAK to
focal adhesions through interactions with talin and paxillin. FAK undergoes
autophosphorylation at tyrosine 397 (Y397), creating a docking site for blinding
Src Homology 2 (SH2) domain of Src family [28, 30]. Src binding to FAK-Y397
disrupts autoinhibitory conformation of Src, enabling phosphorylation of
additional FAK sites (e.g., Y576/Y577) for full kinase activation. The FAK-Src
complex recruits downstream effectors like Grb2, PI3K, and p130Cas, amplifying
downstream signaling [31, 32, 33, 34]. In hPSCs,
The FAK-Src complex activates RhoA, promoting actin polymerization and
actomyosin contractility via ROCK-mediated myosin light chain phosphorylation.
This enhances cytoskeletal tension, critical for cell migration and adhesion
maturation. In hPSCs,
Cytoskeletal tension through the engagement of
FAK activation protects cells from anoikic [44], making integrins natural targets for regulating anoikis resistance. This resistance is essential for hPSC survival during single-cell passaging and differentiation [45, 46]. Anoikis, a form of apoptosis induced by detachment from ECM, is a major contributor to low post-transplantation survival rates in stem cell therapy. For example, anoikis limits the efficiency of hESC-derived cardiomyocyte (hESC-CM) engraftment in cardiac repair [47]. Co-transplantation with biocompatible ECM materials can mitigate anoikis by providing structural and biochemical cues that support cell survival [48].
FAK-Src signaling drives cytoskeletal remodeling, focal adhesion turnover, and
directional motility. FAK phosphorylation is required for MSC differentiation
[49], while in cancer stem cells, FAK promotes migration and
epithelial-to-mesenchymal transition (EMT), enhancing invasive potential [50]. In
hPSCs, integrin-mediated activation of
Mouse embryonic fibroblasts (MEFs) were initially used as feeder cells for derivation and maintenance of hESCs [55]. Human foreskin fibroblasts later provided a xeno-free alternative culturing system to replace MEFs [56]. Matrigel, derived from mouse sarcoma tumors, served as feeder-free system for hESC growt [2, 57]. However, those feeders and Matrigel are allogeneic or xenogeneic origin with potential source of variability and viral or bacterial contamination. Matrigel is a complex and undefined mixture of ECM proteins, including laminins, collagen IV, fibronectin, and other basement membrane proteins, triggering complex integrin signaling. The undefined composition of Matrigel complicates standardization of hPSC culture and differentiation.
Integrin-ECM interactions are essential for hPSC maintenance and lineage
specification (Table 2, Ref. [3, 4, 58, 59, 60, 61, 62, 63, 64, 65]). High levels of
integrin
| Integrin receptor | ECM ligands | hPSC pluripotency | hPSC differentiation | signaling | References |
| Laminin-511, Laminin-521, Laminin-111 | Major role | Neural, cardiac, hepatocyte | PI3K/Akt, FAK and ILK | [3, 58, 61] | |
| VTN (via RGD sequence) | Major role | Hemogenic endothelial, endothelial, hematopoietic | FAK and ILK | [4, 59, 60] | |
| VTN, FN (RGD) | Secondary role | Hemogenic endothelial, hematopoietic | PI3K and FAK | [59, 60] | |
| FN (RGD) | Less effective | Mesenchymal, endothelial, cardiac | MAPK/PI3K | [59, 63] | |
| Laminin-511, -521, -111 | Low affinity | Neural, epithelial, hepatocyte | [58, 61, 62] | ||
| VCAM-1, FN (CS-1 region) | Minimal role | Hematopoietic, endothelial | MAPK/PI3K | [64, 65] | |
| Laminin-111, -211 | Minor role | Myogenic, neural, hepatocyte | [58, 61, 62] |
Laminin isoforms are widely used for the maintenance of hPSC pluripotency,
particularly in defined, feeder-free culture systems. Laminin-511 and laminin-521
are heterotrimeric glycoproteins, composed of
E8 fragments of laminin-511 and laminin-521, containing the C-terminal E8 region
(LG1-3 domains of the
VTN, a monomeric glycoprotein (75 kDa in ECM, or 65 kDa and 10 kDa fragments in
plasma), contains an RGD (Arg-Gly-Asp) motif in its somatomedin B domain for
integrin binding, and domains for binding heparin, collagen, and plasminogen
activator inhibitor-1 (PAI-1). The RGD motif of VTN primarily binds to
mTeSR1 contains BSA, which acts as a carrier for lipids and other components and is known to bind to non-specific surfaces, whereas Essential 8 (E8) medium does not contain BSA. E8 medium is specifically designed as a simplified, defined alternative to mTeSR1, with the goal of reducing variability by removing undefined components like BSA. However, a study comparing two feeder-free culture systems for deriving hiPSCs from fibroblasts found the Matrigel/mTeSR1 combination to be significantly more efficient than the Vitronectin/E8 system in generating hiPSC colonies and expressing pluripotency markers [72]. It provides a key insight into how specific culture conditions can influence the efficiency of generating and maintaining high-quality hiPSCs, which is crucial for their eventual use in therapeutic applications and disease modeling.
Laminin-511 and laminin-521 are the most abundant laminin subtype expressed in hPSCs [73, 74, 75, 76]. The crosstalk between laminins and hPSCs not only supports hPSC pluripotency, but also hPSC differentiation since differentiation protocols of hPSCs often require single-cell dissociation, which can induce apoptosis in hPSCs [77]. Laminin are expressed in early stage of embryos, suggesting a role of guiding the earliest steps of embryonic development [78, 79, 80, 81]. During hPSC differentiation, the expression pattern of laminin isoforms changes. The expression of laminin-511, but not laminin-521, is increased by retinoic acid (RA)-induced mesodermal and endodermal differentiation in hESCs, indicating that laminin isoforms are involved in lineage differentiation [82]. Other Integrin ligands, such as VTN and FN, also have profound influences on differentiation when combined with specific growth factors (Table 3, Ref. [60, 61, 62, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107]).
| Lineage | Key integrins | Role | Key pathways | References |
| Cardiac | Facilitate cardiomyocyte differentiation; FN promotes mesoderm via |
PI3K/AKT (ILK inhibition reduces AKT phosphorylation). | [83, 84, 85, 86, 107] | |
| Endothelial | Drive endothelial commitment with VTN; Laminin-411 and Collagen IV enhance CD31 expression; FN inhibits via TGF |
VEGF via PI3K/AKT and MAPK/ERK. | [87, 88, 89] | |
| Hematopoietic | Promote hematopoietic transition with VTN; |
PI3K/AKT and MAPK/ERK enhance VEGF signaling. | [60, 90, 91] | |
| Neural | Support dopaminergic neurons with laminin-521/-511; mediate progenitor adhesion/migration; aid Schwann cell differentiation and myelination. | PI3K and MAPK. | [92, 93, 94, 95] | |
| Hepatic and Cholangiocyte | Drive hepatocytes and definitive endoderm with laminin-521/-511; laminin-411 E8 promotes cholangiocytes. | PI3K/AKT and MAPK regulate growth. | [61, 62, 96, 99, 103, 104] | |
| Corneal Epithelial | Enhance adhesion with laminin-332 E8. | Supports wound healing and mechanotransduction. | [97, 98] | |
| Definitive Endoderm | Enhance endoderm transition with RGD peptides and matrix stiffness; |
MEK-ERK and PI3-kinase regulated by integrin-ECM. | [99, 100, 101, 102] | |
| Oligodendrocyte | Support maturation with VTN and laminin; aid myelination. | PI3K, MAPK, Fyn kinase, Cdc42/Rac1. | [105, 106] |
The interaction of specific ECM proteins with integrins such as
In human cardiac tissue, laminins (e.g., laminin-211, -221, -411, -421, -511,
and -521) are highly expressed during fetal cardiac development and in adult
cardiac tissue [108]. Their interactions with
Laminin-411 is important for increasing hPSC-derived endothelial differentiation
via the activation of multiple signaling pathways, including Focal Adhesion
Kinase (FAK), Integrin-Linked Kinase (ILK), Notch, and
VTN-integrin signaling directs hPSC differentiation toward hematopoietic
lineages. As in embryonic development, hematopoietic differentiation from hPSCs
proceeds through three major developmental stages: (1) mesodermal induction from
hPSCs, (2) specification of hemogenic endothelium from mesodermal progenitors,
and (3) endothelial-to-hematopoietic transition (EHT) to generate hematopoietic
stem and progenitor cells. VTN promotes the development of hematopoietic-fated
mesoderm and hemogenic endothelial cells, upregulating key hematopoietic
transcriptional factors, including GATA2, PU.1, GATA1, GFI1B, and KLF1 [60].
Synergistic
Integrins also modulate ECM organization to regulate cell signaling during
hematopoiesis, with
Beyond
The ectoderm is the outermost of the three primary germ layers in early embryogenesis. During development, it differentiates into neuroectoderm and non-neural ectoderm. Given the difficulty of isolating adult stem cells from neural tissues, generating neuroectodermal lineages from hPSCs has become a central focus in current ectoderm differentiation studies.
Both laminin-511 and laminin-521 provide essential signals that support the
survival and self-renewal of hPSCs while priming them for subsequent
differentiation. These laminins also facilitate the differentiation of hPSCs into
dopaminergic neurons [92]. The laminin
Laminin-511, in particular, promotes the survival and differentiation of
midbrain dopaminergic neurons by engaging integrin
The interaction between laminin-521 and integrin
The surface ectoderm gives rise to the corneal epithelium, conjunctival
epithelium, lens, and the epidermis of the eyelids. Among ECM components,
laminin-332 is an essential constituent of the basement membrane of corneal
epithelial cells and plays a central role in supporting their growth.
Specifically, E8 fragments from laminin-332 markedly enhance adhesion and
expansion of iPSC-derived corneal epithelial cells compared with fragments from
other laminins. This difference in adhesive properties across laminin substrates
enables the selective enrichment of iPSC-derived corneal epithelial cells over
other ocular cell types [97]. Integrin
Definitive endoderm is an epithelial cell layer formed during early embryonic development that gives rise to multiple internal organs and tissues, including the digestive tract (stomach, intestines, liver, pancreas), respiratory system (lungs, trachea, bronchi) and thyroid gland. Definitive endodermal progenitors are characterized by the expression of key transcription factors, such as SOX17, GATA4, and FOXA2 [129].
Integrin signaling, in coordination with other pathways, plays a crucial role in
guiding the differentiation of definitive endoderm into specific organ lineages.
FN and VTN are major ECM components that promote definitive endoderm
specification by interacting with integrins
Synthetic PEG-based hydrogel functionalized with cyclic RGD peptides have been
shown to support hPSC attachment and promote transition to definitive endoderm.
In this context, matrix stiffness emerges as a key mechanical cue, with FAK
signaling serving as a downstream intracellular mediator of integrin-ECM
interactions [102]. During the early stages of definitive endoderm
differentiation, integrin-mediated traction forces are required for SMAD2
phosphorylation and nuclear translocation, which enhances the expression of
definitive endoderm markers, such as SOX17, thereby linking mechanical signaling
to TGF-
Laminin-521 and -511, interacting with primarily
VTN-integrin signaling, combining with signaling molecules sonic hedgehog (Shh),
retinoic acid (RA), and noggin, supports hPSC differentiation into
oligodendrocytes, mediated by
Besides VTN, other ECM proteins may interact with integrins in oligodendrocytes
to initiate intracellular signaling cascades, influencing crucial functions like
proliferation, survival, and maturation that are essential for CNS myelination
[137, 138]. Laminin-2, found on the surface of axons, binds to the
Transitioning from conventional 2D monolayer cultures to 3D systems profoundly alters integrin signaling in hPSCs, particularly during lineage differentiation, such as organoid formation. Enhanced cell–cell and cell–ECM interactions in 3D systems create a more physiologically relevant microenvironment than 2D cultures, thereby enhancing the fidelity of hPSC differentiation and tissue morphogenesis [140]. For example, cardiomyocytes derived from 2D hPSC cultures fail to fully recapitulate the electrophysiological and contractile properties of native heart tissue [141, 142, 143]. In contrast, 3D organoid systems have been successfully established to generate diverse tissue types from hPSCs, including the brain, eyes, kidney, lung, stomach, intestine, inner ear, skin, thyroid, and liver [144, 145, 146, 147, 148, 149, 150, 151, 152, 153].
Synthetic 3D matrices, such as polyethylene glycol (PEG)-based hydrogels, offer defined and tunable microenvironments that improve reproducibility and control compared to conventional organoid methods. For instance, Chow et al. [154] demonstrated that encapsulating hPSC-CMs in a 3D PEG hydrogel improved cardiac function and tissue integrity following myocardial infarction in rats, suggesting indirect paracrine or remodeling effect despite limited long-term cell survival. Within such systems, integrins mediate cell–matrix attachment and transduce essential “outside-in” signals that regulate survival, proliferation, and mechanosensation. Functionalization of hydrogels with integrin-binding peptides, particularly RGD motif, enhances cell adhesion [155]. Through RGD–integrin interactions, cells form focal adhesions that physically couple the cytoskeleton with the ECM, promoting dynamic cell–matrix crosstalk and activating intracellular signaling pathways that govern adhesion, spreading, proliferation, migration, differentiation, and mechanotransduction. During vascular differentiation of iPSCs, 3D PEG hydrogels have been shown to upregulate ECM components and integrins relative to 2D cultures, facilitating the formation of functional vascular networks [156].
Despite these advances, fully recapitulating human physiology in 3D hPSC cultures remains challenging, largely due to the absence of a vascular network capable of delivering oxygen and nutrients while removing metabolic waste. Nevertheless, the integration of 3D culture systems with defined xeno-free ECMs controlled mechanical properties represents a promising direction for future studies aimed at modeling human development and disease and advancing hPSC-based tissue engineering and therapeutic applications.
Integrin-ECM interactions are fundamental to hPSC maintenance and lineage
specification. The transition from xenogeneic feeders such as MEFs and Matrigel
to defined substrates like laminin-511/521 and VTN has enabled xeno-free,
reproducible culture systems suitable for clinical translation. In the
pluripotent state,
hPSCs exhibit distinct integrin profiles and heightened mechanosensitivity
compared to somatic and adult stem cells, relying primarily on
Despite these advances, challenges remain in translating integrin–ECM biology into fully defined, GMP-compliant hPSC platforms. Current recombinant ECM proteins remain costly and variable, and the complexity of integrin signaling networks complicates the dissection of ligand-specific effects on pluripotency and lineage choice. Furthermore, conventional 2D culture systems often fail to capture the spatial, mechanical, and topographical cues that regulate integrin clustering and focal adhesion dynamics in vivo.
Future research should focus on engineering synthetic ECMs with tunable
biochemical and biomechanical properties to systematically dissect
integrin-mediated mechanisms under controlled conditions. Integration of
bioengineered hydrogels, 3D microenvironments, and advanced imaging and omics
approaches will enable high-resolution mapping of integrin signaling networks.
Additionally, combining these systems with CRISPR-based perturbation and
mechanobiological modeling may reveal how integrin signaling integrates with
pathways such as Wnt, Notch, and TGF-
Ultimately, the development of cost-effective, scalable, and xeno-free ECM platforms, together with a deeper mechanistic understanding of integrin–ECM crosstalk, will be essential for achieving reproducible, safe, and efficient hPSC-based applications in regenerative medicine, disease modeling, and cell therapy manufacturing.
TW: Contributed to drafting the manuscript and preparing the figures. ZZW: Involved in study design, manuscript writing, and final approval of the manuscript. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by Maryland Stem Cell Research Fund (2024-MSCRFD-6400).
The authors declare no conflict of interest.
During manuscript preparation, Google Gemini 3 and OpenAI GPT-4.1 were used for language editing and grammar checking. The authors reviewed and edited all text and take full responsibility for the content of the published work.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

