1 Department of Infectious Disease, Beijing Friendship Hospital, Capital Medical University, 100050 Beijing, China
Abstract
The activation of adenosine-monophosphate-activated protein kinase (AMPK) by berberine (BBR) benefits various inflammatory diseases. Conversely, high mobility group box-1 (HMGB1), a prototypical damage-associated molecular pattern (DAMP), typically exerts opposing effects. This research aims to investigate the relationship between AMPK and HMGB1, elucidating the functions and underlying mechanisms by which BBR alleviates acute lung injury (ALI) caused by lipopolysaccharide (LPS).
Male C57BL/6J mice were intragastrically administered BBR twice daily for three days with a total of 25 and 100 mg/kg/day. On day four, an intraperitoneal injection of 10 mg/kg LPS was administered, and BBR was given two hours before and six hours after this injection, respectively. Eighteen hours post-LPS administration, lung tissues and serum samples were collected to assess indicators of lung tissue injury, inflammation, oxidative stress, and apoptosis. The relationship between AMPK activation, HMGB1 release, and inflammatory activation was investigated in both mice and RAW264.7 cells using protein expression analysis, AMPK silencing, and exogenous HMGB1 introduction.
Our findings demonstrate that BBR activates AMPK and inhibits HMGB1 expression, translocation, and release in LPS-induced ALI, resulting in reduced histopathological lung injuries, decreased expression of inflammatory cytokine genes, and diminished oxidative stress and apoptosis. Mechanistic studies revealed that BBR decreases extracellular HMGB1 in LPS-stimulated RAW264.7 cells and inhibits HMGB1-stimulated nuclear factor Kappa B (NF-κB) activation. Concurrently, silencing the activation of AMPK by siRNA and compound C reversed the BBR-reduced extracellular HMGB1 level in LPS-stimulated RAW264.7 cells.
Based on these findings, we conclude that BBR effectively inhibits inflammation, oxidative stress, and apoptosis in LPS-induced ALI by modulating the AMPK-HMGB1-NF-κB axis. Consequently, BBR and other AMPK activators may represent promising therapeutic options for managing systemic inflammation and injury during sepsis.
Keywords
- berberine
- sepsis
- acute lung injury
- AMP-activated protein kinase
- HMGB1 protein
Sepsis is a life-threatening impairment of organ function that arises from an unregulated systemic immune and inflammatory response by the host to an infection [1]. Among the various types of organ dysfunction, lung tissue injury is a pivotal contributor to the morbidity and mortality associated with sepsis [2, 3]. To date, traditional management strategies, such as early antibiotic administration and fluid resuscitation, have shown efficacy in mitigating early-stage sepsis to some degree. However, due to the significant host-mediated systemic inflammatory responses observed during sepsis, therapeutic approaches targeting specific signaling pathways and molecules warrant greater attention [1]. High mobility group box-1 (HMGB1), a multifunctional damage-associated molecular pattern (DAMP) and signaling molecule, has been identified as a crucial late-phase inflammatory mediator that contributes to endotoxin lethality in sepsis. Upon infection, the interaction between HMGB1 and TLR4/MD2, as well as the formation of HMGB1-lipopolysaccharide (LPS) complexes, plays a key role in triggering downstream inflammatory pathways, oxidative stress, apoptosis, and pyroptosis [4, 5, 6]. Consequently, targeting HMGB1 may lead to organ protection and potentially improve survival outcomes [1].
Berberine (BBR, Fig. 1A), a benzylisoquinoline alkaloid, is an active constituent present in various medicinal plants and exhibits multiple pharmacological properties, including antimicrobial, antiprotozoal, antidiarrheal, and antitrachoma activities. Recently, BBR has also been reported to possess therapeutic potential for metabolic, neurological, and cardiological diseases [7, 8, 9, 10, 11]. Among the various targets of BBR, adenosine-monophosphate-activated protein kinase (AMPK) activation is considered a pivotal target of BBR, mediating its diverse pharmacological effects. Previous studies have indicated that AMPK activation benefits the balance between oxidants and antioxidants during lung injury, whereas the release of HMGB1 triggers inflammation and subsequent apoptosis [4, 5, 12].
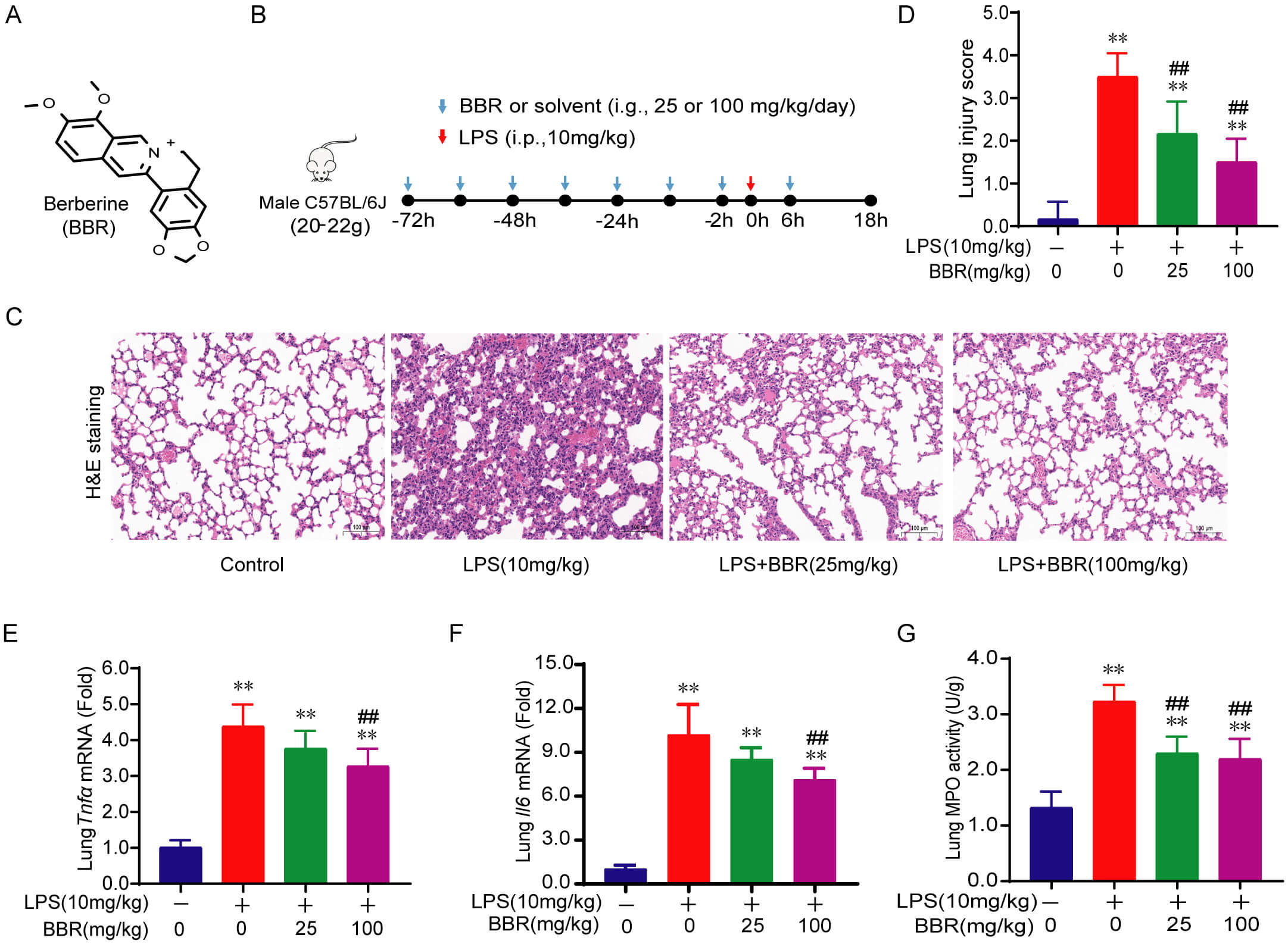 Fig. 1.
Fig. 1.
BBR ameliorates LPS-induced ALI and inflammatory
response in mice. (A,B) Chemical structure of BBR (A) and experimental scheme
(B). (C) H&E staining of lung tissues at 18 h after LPS stimulation
(200
Predictably, BBR may also activate AMPK during acute lung injury (ALI).
Nonetheless, the involvement of BBR and the activation of AMPK in the regulation
of lung injury, inflammation, oxidative stress, and apoptosis in the context of
sepsis, along with the role of HMGB1 in this process, remains inadequately
understood. This research aims to evaluate the impact of BBR on lung damage,
inflammatory responses, oxidative stress levels, and apoptosis in mice subjected
to LPS-induced sepsis. Furthermore, we elucidated the effect of BBR on AMPK
activation and HMGB1 levels during ALI, and the correlation between BBR-induced
AMPK activation and HMGB1 release in LPS-stimulated mouse monocyte/macrophage
cells, RAW264.7. Our results highlight the effectiveness of BBR in suppressing
inflammation, oxidative stress, and apoptosis in LPS-induced ALI, mediated
via modulation of the AMPK-HMGB1-NF-
Male C57BL/6J mice (6–8 weeks, 18–20 g) were sourced from SPF Biotechnology
Co., Ltd. (Beijing, China) and housed for one week under a 12-h light/dark cycle
in specific pathogen-free conditions before the experiment. To evaluate the
effect of BBR (A75865, InnoChem, Beijing, China; purity
Blood samples underwent centrifugation at 2500 g for a duration of 10 min to obtain serum. The levels of HMGB1 in serum and cell culture supernatants were measured using a mouse HMGB1 enzyme-linked immunosorbent assay (ELISA) kit (SEKM-0145, Solarbio, Beijing, China). Additionally, mouse lung tissues were homogenized, and commercial assay kits were utilized to quantify malondialdehyde (MDA) (S0131, Beyotime Biotechnology, shanghai, China), superoxide dismutase (SOD) (S0101, Beyotime Biotechnology, shanghai, China), and myeloperoxidase (MPO) (A044-1-1, Nanjing Jiancheng Biotechnology Co., Ltd., Nanjing, China) to assess oxidative stress in the lungs and infiltration of neutrophils.
Lung tissues that were fixed in paraffin, and sections measuring 5 µm were subjected to standard hematoxylin and eosin (H&E) staining to assess histopathological alterations using a light microscope. A semi-quantitative scoring system was utilized to evaluate the infiltration of neutrophils, the presence of pulmonary edema, disorganization within the lung parenchyma, and any hemorrhagic changes in the lung tissue. The scoring criteria were defined as follows: 0, no pathological changes observed in the visual field; 1, 2, 3, and 4 represent pathological changes present in less than 25%, 25%–50%, 51%–75%, and greater than 75% of the total visual field, respectively [18]. Apoptotic characteristics in lung tissues preserved with formaldehyde were identified using terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining, which employed the DAB (SA-HRP) (PR30010, Proteintech, Wuhan, China) TUNEL Cell Apoptosis Detection Kit (G1507, Servicebio, Wuhan, China). The proportion of TUNEL-positive cells was quantified using ImageJ software (NIH, Bethesda, MD, USA). Additionally, immunofluorescence was performed to detect HMGB1 in lung tissue, and its expression and intracellular distribution were evaluated.
Mitochondria were isolated from lung tissue using a mitochondrial isolation kit (C3606, Beyotime Biotechnology, Shanghai, China). In summary, the lung tissue underwent homogenization followed by centrifugation at 4 °C and 600 g for 10 min to eliminate cell nuclei. The resulting supernatant was centrifuged a second time at 11,000 g and 4 °C for 10 minutes to collect crude mitochondrial pellets. Next, the supernatant underwent centrifugation at 12,000 g for 10 minutes to separate cytoplasmic proteins. The concentration of proteins was assessed using a BCA protein assay kit (#23225, Thermo Fisher Scientific, Waltham, MA, USA), after which the proteins were heated and analyzed via Western blot.
RNA was extracted from the cells using the RaPure total RNA kit (R4011, Magen,
Shanghai, China), in accordance with the instructions provided by the
manufacturer. The mRNA expression levels of the inflammatory cytokine genes
Tnf
Lung homogenates or cell lysates were collected utilizing a protein extraction
reagent (78510, Thermo Fisher Scientific, Waltham, MA, USA) combined with
inhibitor cocktails for proteases and phosphatases (C0001 and C0004, TargetMol,
Boston, MA, USA). The concentration of protein was assessed using a BCA protein
assay kit. For Western blotting, protein samples were separated by 12% sodium
dodecyl sulfate-polyacrylamide gel electrophoresis, and subsequently transferred
to polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with 5%
fat-free milk and incubated overnight at 4 °C with antibodies against
ACTB (GB15001, Servicebio, Wuhan, China), HMGB1 (ET1601-2, HUABIO, Hangzhou,
China), AMPK
Mouse monocyte/macrophage cells RAW264.7 (CL-0190, Procell Life Science &
Technology, Wuhan, China) were maintained in a 5% CO2 atmosphere at 37
°C and cultured in DMEM supplemented with 10% FBS, 100 IU/mL
penicillin, and 100 µg/mL streptomycin. RAW264.7 cells were validated by
STR profiling and tested negative for mycoplasma (D101, Vazyme, Nanjing, China).
To investigate the critical role of HMGB1 in BBR-mediated NF-
To investigate whether AMPK activation by BBR contributes to the reduction of
extracellular HMGB1 levels in LPS-stimulated RAW264.7 cells, we inhibited AMPK
activation using RNA silencing or the inhibitor compound C (#T1977, TargetMol,
Boston, MA, USA). Briefly, either 50 nM negative control siRNA or siRNA targeting
AMPK
The data in this article are presented as the mean
To evaluate the effects of BBR (Fig. 1A) on LPS-induced ALI, mice were
administered BBR twice daily (a total of 25 or 100 mg/kg/day) for 3 days before
LPS induction, with an additional treatment at 6 hours post-LPS injection (Fig. 1B). 18 h following LPS stimulation, lung tissues stained with H&E from the
group treated solely with LPS displayed marked infiltration of inflammatory
cells, edema, increased interstitial congestion, and thickened alveolar walls.
Conversely, BBR treatment effectively alleviated these pathological changes (Fig. 1C), with the lung injury score corroborating the protective effect of BBR in ALI
(Fig. 1D). Inflammatory mediators play a vital role in the development of ALI
triggered by LPS. Additionally, our findings indicated that BBR markedly reduced
the overexpression of the inflammatory genes Tnf
ALI is characterized by an excessive inflammatory response that often exacerbates oxidative stress and apoptosis [22]. To investigate the effects of BBR on oxidative stress injury, we measured the activity of the antioxidant enzyme SOD and the levels of MDA in lung tissues. The results indicate that LPS significantly reduced SOD activity and increased MDA levels in lung tissues, whereas BBR treatment significantly reversed these alterations (Fig. 2A,B), emphasizing its potent antioxidant properties. To evaluate the effect of BBR on apoptosis, we conducted the TUNEL assay on lung tissues and analyzed the expression of apoptosis-related proteins. Our findings revealed a marked increase in the proportion of TUNEL-positive cells following LPS treatment, which was notably reduced by BBR (Fig. 2C,D). Additionally, BBR markedly suppressed the levels of apoptosis-related proteins, specifically cleaved caspase-3 and cleaved caspase-9, induced by LPS (Fig. 2E,F). Compared with the control group, the LPS group exhibited lower B cell lymphoma 2 (BCL2), levels and higher BCL2-Associated X protein (BAX) levels, and these abnormalities were also significantly attenuated by BBR treatment (Fig. 2E,F). The findings collectively affirm the antioxidative stress and anti-apoptotic properties of BBR in LPS-induced ALI, which correspond to its anti-inflammatory function.
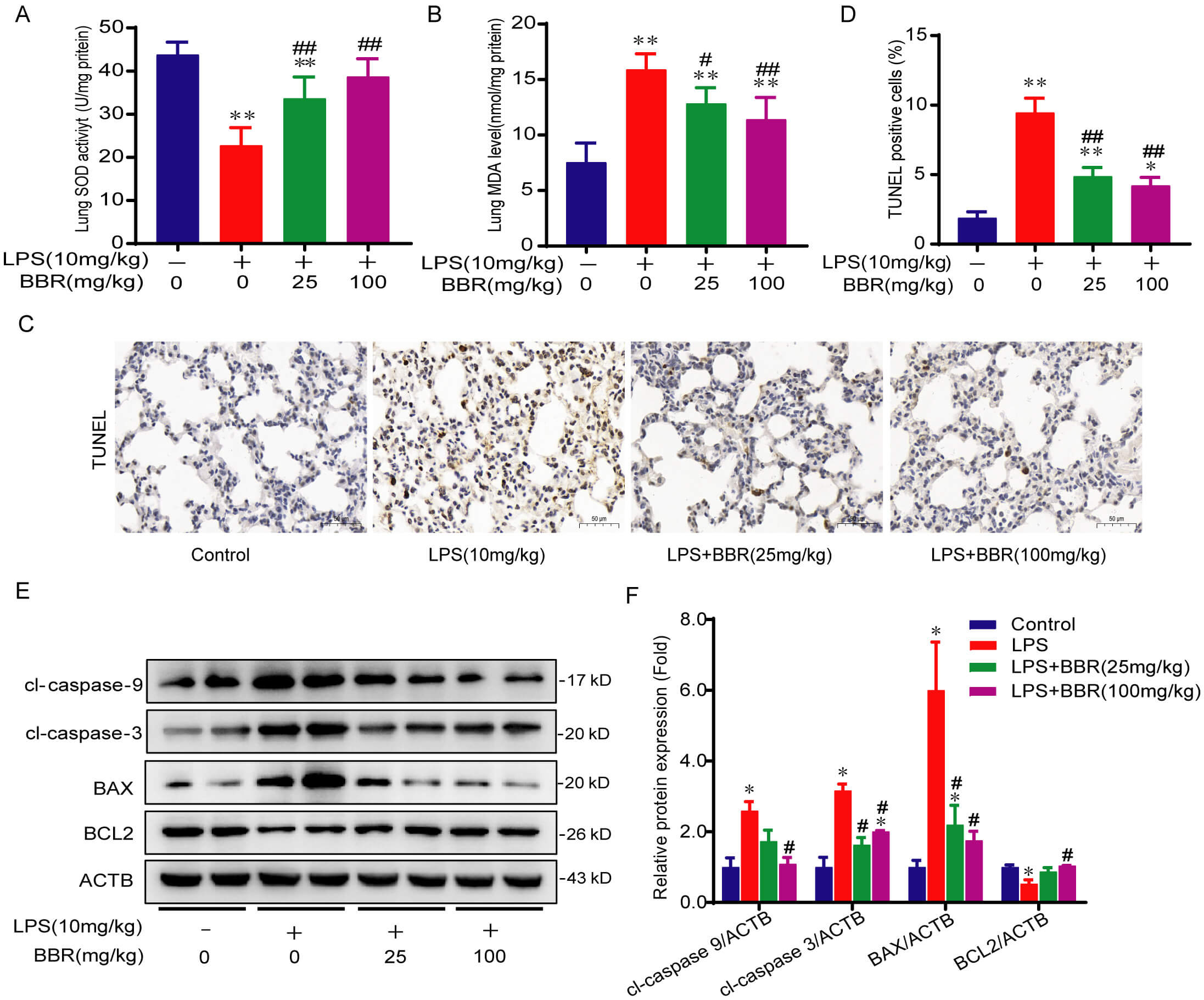 Fig. 2.
Fig. 2.
BBR inhibits oxidative stress and apoptosis during
LPS-induced ALI in mice. (A,B) MDA levels and SOD activity in lung tissues were
assessed as oxidative stress indicators at 18 h after LPS and/or BBR treatment.
(C,D) Representative staining (200
BBR has been widely reported to activate AMPK, which in turn contributes to the
maintenance of oxidant-antioxidant balance in various inflammatory conditions
[12, 23]. Conversely, excessive HMGB1 release triggers inflammatory responses and
subsequent apoptosis in sepsis [4, 5]. Consequently, we chose to examine the
roles of AMPK and HMGB1 in mediating the protective effects of BBR against
sepsis-induced inflammatory activation, a critical mechanism underpinning ALI.
The results indicate that LPS significantly reduced phosphorylated AMPK (p-AMPK)
levels and increased total HMGB1 protein levels, resulting in NF-
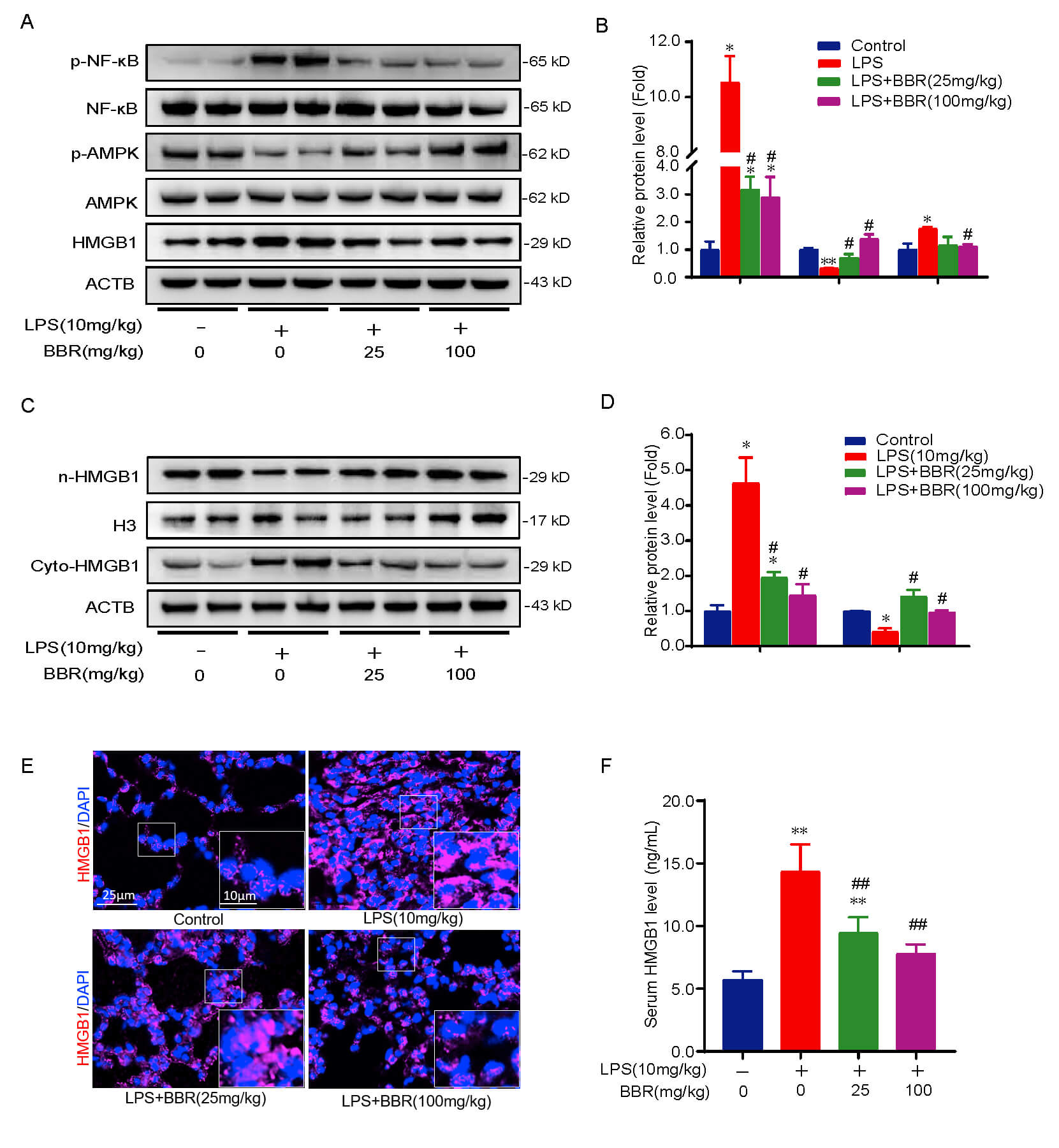 Fig. 3.
Fig. 3.
BBR activates AMPK and suppresses HMGB1-mediated NF-
The primary mechanism underlying sepsis is immune dysfunction, with macrophages
playing a pivotal role [25]. In this study, we utilized the mouse macrophage cell
line RAW264.7 to elucidate the role of HMGB1 in BBR-mediated inhibition of
NF-
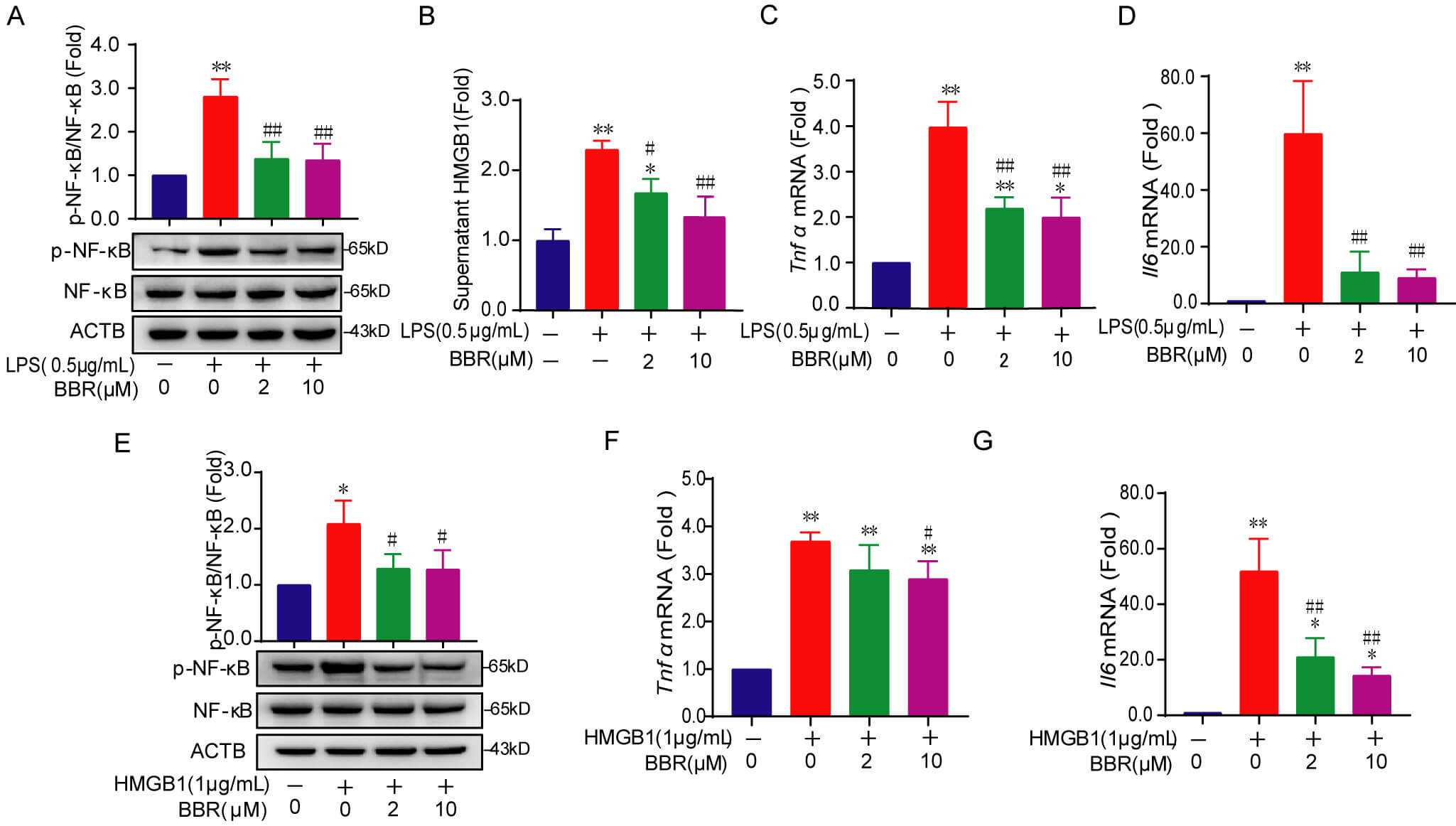 Fig. 4.
Fig. 4.
BBR decreases extracellular HMGB1 in LPS-induced
RAW264.7 cells and inhibits HMGB1-stimulated NF-
As extracellular HMGB1 serves as a final effector in the inflammatory activation
process, we further assessed whether BBR-induced decreases in extracellular HMGB1
levels are mediated through AMPK activation in LPS-stimulated RAW264.7 cells. For
this, we utilized AMPK
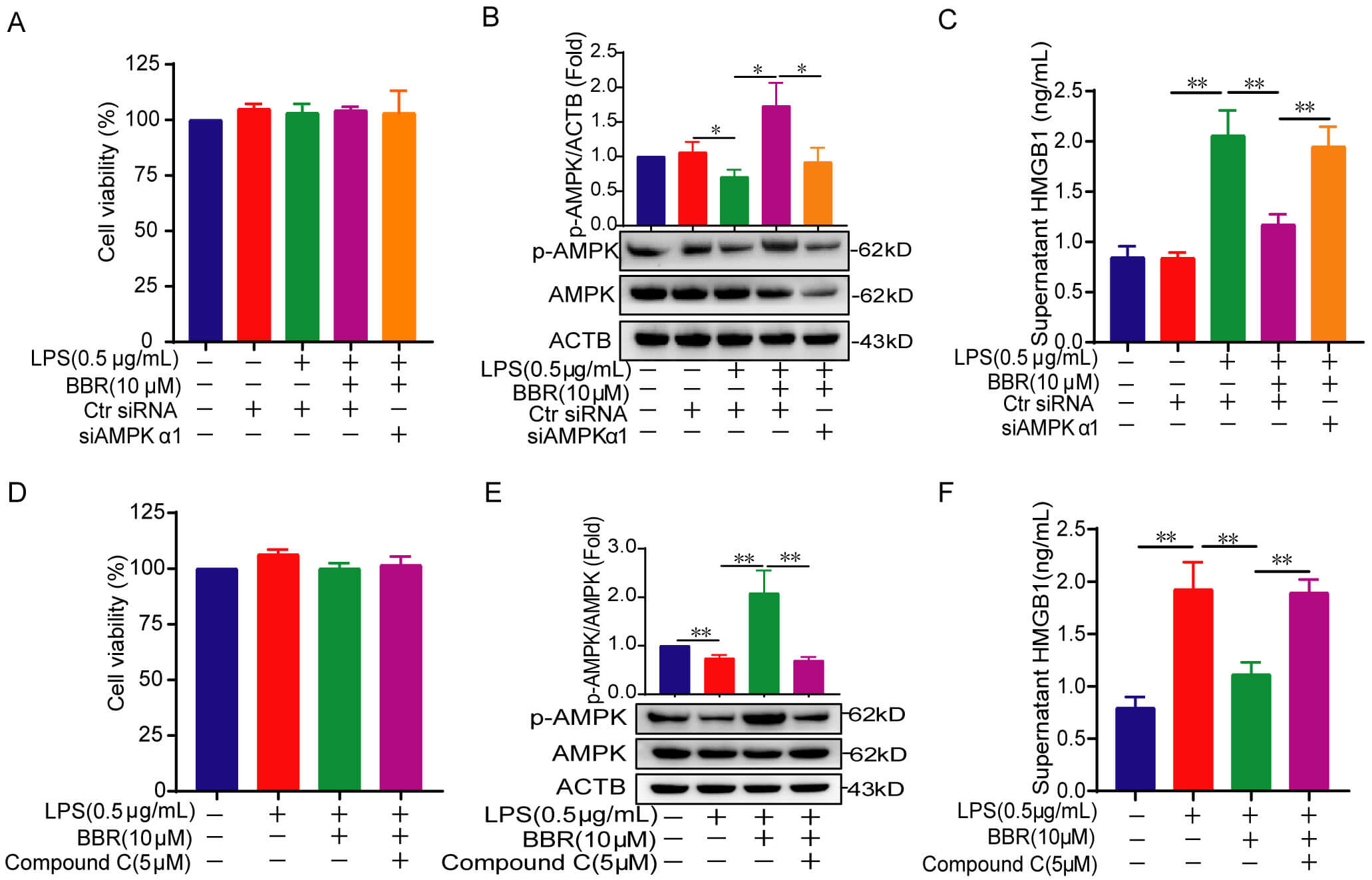 Fig. 5.
Fig. 5.
Silencing the activation of AMPK reverses BBR-reduced
extracellular HMGB1 level in LPS-stimulated RAW264.7 cells. (A,D) Cytotoxicity
detection in RAW264.7 cells after 24 h or 48 h treatment with LPS, BBR, siAMPK, or
compound C. (B,E) Western blot analysis of p-AMPK and AMPK protein levels. (C,F)
HMGB1 levels in cell culture supernatants detected via an ELISA method.
Data are presented as the mean
Targeting the signaling pathways and molecules that mediate systemic
inflammation represents a critical therapeutic strategy for alleviating organ
injury during sepsis [1]. We previously reviewed the protective effects of BBR
against multiple organ injuries, encompassing the intestine, liver, kidney, and
lung [29]. In this study, we further revealed that BBR significantly mitigates
LPS-ALI, as evidenced by reductions in histopathological damage, inflammatory
responses, oxidative stress, and apoptosis. Mechanistic insights reveal that BBR
activates AMPK, which subsequently diminishes serum and extracellular HMGB1
levels, ultimately inhibiting HMGB1-mediated NF-
To date, there remains a lack of effective treatments for irreversible septic
shock and multiple organ dysfunction syndromes, although extensive research has
been conducted on dysregulated signaling pathways and molecules underlying sepsis
pathogenesis [1]. Regarding signaling- and molecule-targeted anti-sepsis
strategies, BBR could alleviate LPS-induced lung injury through mechanisms
including inhibition of TNF-
Serum HMGB1 concentrations have been closely linked with mortality in animal sepsis models [35, 36]. Our study in a murine septic model revealed that BBR reduced serum HMGB1 concentrations, potentially by suppressing the total protein expression, nucleus-to-cytoplasm translocation, and extracellular release of HMGB1 (Fig. 3). These results reinforce the concept that HMGB1 translocation from the nucleus to the cytoplasm is a pivotal step preceding its extracellular release [37]. Although the reduced HMGB1 release might also be attributed to the enhanced HMGB1 deacetylation by SIRT3 or SIRT1, which are potentially regulated by BBR [10, 38, 39, 40], it might also stem from BBR’s potential modulation of the AMPK/mTOR and Nrf2/HO-1 signaling pathways [41, 42]. Collectively, the decrease in extracellular HMGB1—the functional form of HMGB1—contributes to the multifaceted regulation of inflammatory diseases by BBR, thereby reinforcing the idea that BBR is a multifunctional compound with diverse pharmacological activities [43].
Multiple cell types, such as alveolar epithelial cells, neutrophils, monocytes,
and macrophages, participate in the progression and prognosis of sepsis-related
lung injury. Among them, although alveolar epithelial cells initiate inflammation
via barrier damage, neutrophils drive injury through NETosis and
oxidative stress, and monocytes through infiltration and differentiation,
macrophages serve as central orchestrators by polarizing into pro-inflammatory
(M1) or reparative (M2) phenotypes, ultimately influencing the progression or
resolution of sepsis-induced lung injury across all phases [1, 44, 45].
Consequently, we investigated the anti-inflammatory mechanisms specifically in
RAW264.7 macrophages derived from mice. Exogenous HMGB1 accumulates on the
surface of macrophages, forming a complex with its receptors, which subsequently
activates the inflammatory pathway. As expected, our findings revealed that BBR’s
inhibition of NF-
Nonetheless, our study is subject to some limitations. For instance, we
primarily explored the causal or upstream-downstream relationship by which BBR
activates AMPK, reduces the release of HMGB1, and inhibits NF-
Taken together, the current study proposes that BBR markedly inhibits
inflammation, oxidative stress, and apoptosis via the
AMPK-HMGB1-NF-
ANOVA, One-way analysis of variance; ALI, acute lung injury; AMPK, AMP-activated protein kinase; BBR, Berberine; DAMP, damage-associated molecular pattern; ELISA, enzyme-linked immunosorbent assay; H&E, hematoxylin and eosin; HMGB1, High mobility group box-1; LPS, lipopolysaccharide; MDA, malondialdehyde; MPO, myeloperoxidase; p-AMPK, phosphorylated AMPK; PVDF, polyvinylidene fluoride; siRNA, small interfering RNA; SOD, superoxide dismutase; TUNEL, transferase dUTP nick end labeling.
The data associated with this paper are available upon request to the corresponding author.
TXL, CW, and WJQ designed the research study. TXL and CPZ extracted all data and performed analyses. YWZ, LH and GWH provided help and advice on methodology. TXL drafted the manuscript and performed the revisions. WJQ provided funding acquisition. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experiments were approved by the Ethics Committee of Beijing Friendship Hospital, Capital Medical University (Approval #24-2034) and conducted following the National Guidelines for Housing and Care of Laboratory Animals.
We sincerely thank professor Bangjiang Fang for his valuable suggestions on experimental design and professor Hu Li for giving operational guidance advice in the experiment.
This work was supported by High-level Public Health Technology Talent Construction Project (Academic Leader-02-29), the National key clinical specialty construction Project (2022) & University (No. yybsh2021007), Capital Medical University Natural Science Foundation (PYZ24071).
The authors declare no conflict of interest.
During the preparation of this work the authors used ChatGpt-3.5 in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





