1 Department of Dermatology and Venereal Diseases, Affiliated Hospital of Qinghai University, Qinghai University, 810001 Xining, Qinghai, China
Abstract
Chronic skin diseases like psoriasis, atopic dermatitis, and vitiligo, characterized by long-term courses, frequent relapses, and complex management, severely affect patients’ lives. This review summarizes their epidemiology, pathogenesis, and therapeutic strategies, focusing on elucidating the core synergistic roles of cytokines including interleukin (IL)-17, tumor necrosis factor-α (TNF-α), and interferon-gamma (IFN-γ). Comparative analysis reveals overlapping genetic, immune, and environmental factors. Current therapeutic approaches have limitations, whereas targeted biologics, especially novel biologics developed using gene editing and cell therapy technologies to achieve precise immune modulation, demonstrate tremendous potential. Cross-disease immune investigations hold substantial value: (1) The identification of common targets to uncover shared immunoregulatory features, cross-regulatory patterns of key signaling pathways, and common disease targets amenable to drug repurposing. (2) The advancement of precision medicine through mechanism-based treatment approaches, such as broad-spectrum inhibitors and optimized combination therapies. (3) To guide drug development of individualized treatments using novel therapeutics by providing crucial insights into skin immunology. This research facilitates the shift from “disease-classification-based management” to “immune phenotype-directed therapeutics,” supporting the development of novel biologics and individualized strategies.
Keywords
- chronic skin diseases
- IL-17
- TNF-α
- IFN-γ
- targeted biologic therapy
- broad-spectrum pathway inhibitors
Chronic skin diseases (CSDs) are defined as persistent cutaneous disorders
characterized by prolonged duration (
Core features:
(1) Stubborn disease course: Alternating active and remission phases with limited curative potential through conventional therapies.
(2) Pathological accumulation: Chronic inflammation induces cumulative damage, including epidermal barrier disruption, aberrant keratinocyte differentiation, and dermal matrix remodeling.
(3) Systemic manifestations: Associations with metabolic dysregulation, psychological comorbidities (e.g., anxiety disorders), and multi-organ involvement.
CSDs arise from multifactorial interactions involving genetic predisposition, immune dysfunction, infections, environmental triggers (e.g., pollutants), and lifestyle factors. Common clinical presentations include erythema, papules, blisters, scales, lichenification, and pigmentary changes, frequently accompanied by pruritus, pain, or burning sensations.
The relapsing nature of CSDs necessitates stepwise therapeutic regimens involving acute-phase control, maintenance therapy, and preventive management. Interventions span pharmacotherapy (e.g., biologics, immunosuppressants), physical modalities (e.g., phototherapy), and surgical approaches. Patient education on skin care and lifestyle modifications is critical for disease control.
Major CSDs include Psoriasis, Atopic Dermatitis, Seborrheic Dermatitis, Vitiligo, Contact Dermatitis, Acne, etc. [2, 3, 4, 5]. These disorders share progressive epidermal barrier failure that disrupts cutaneous immune homeostasis, often leading to psychosocial burdens (e.g., anxiety and social withdrawal) and substantial socioeconomic costs.
CSDs affect diverse populations globally, with distinct demographic predispositions and risk factors across subtypes. Notably, atopic dermatitis (AD), psoriasis, and vitiligo exhibit epidemiological burdens, respectively, as summarized in Table 1 (Ref. [6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18]), imposing significant health and socioeconomic impacts, warranting urgent mechanistic and therapeutic investigations [1].
| Category | Psoriasis | AD | Vitiligo |
| Population | Adolescents & adults (15.30 years) | Children & adolescents | Children & adolescents |
| Gender | Males slightly |
Males slightly |
No significant difference |
| Incidence | 2%–3% | ~10% | 0.5%–2% |
| Temporal trend | Earlier age of onset | 1. Earlier age of onset | Earlier age of onset |
| 2. Rising incidence | |||
| 3. Gradual symptom worsening | |||
| Genetic contribution | 71.60% | 70%–80% | 30%–40% |
| Environmental triggers | 1. Infections triggering inflammation | 1. Climate/seasonal changes affecting skin barrier | 1. UV-induced melanocyte damage |
| 2. Psychological stress disrupting immune regulation | 2. Allergen exposure inducing immune activation | 2. Chemical exposure via immune-mediated pathways | |
| 3. Unhealthy lifestyles impairing immune homeostasis | 3. Unhealthy lifestyles | 3. Chronic stress & poor lifestyle habits | |
| Candidate genes | HLA-B, HLA-C IL-23, IL-17, TNF- |
FLG, STAT6, IL-4, IL-13, IL-31 | HLA-A, HLA-B, HLA-DR, CTLA-4, TYR |
| References | [11, 12] | [6, 13, 16, 17] | [7, 8, 9, 10, 14, 15, 18] |
AD, atopic dermatitis; UV, ultraviolet; IL, interleukin; HLA-B, Human Leukocyte
Antigen B locus; HLA-C, Human Leukocyte Antigen C locus; HLA-A, Human Leukocyte
Antigen A locus; TNF-
Psoriasis: Global prevalence of 2%–3%, peaking in young adults (15–30 years), with rising pediatric cases. Males show marginally higher susceptibility (Table 1) [19]. AD: Affects ~10% globally, with marked age disparity: 15%–30% in children vs. 2%–10% in adults. Increasing prevalence in low-income countries and early childhood populations (Table 1) [6]. Vitiligo: Prevalence of 0.5%–2%, age- and sex-independent, though lower in colder climates (Table 1) [7, 8, 9].
Psoriasis/Vitiligo: Higher incidence in cold/dry regions (e.g., Northern Europe, Russia) versus lower rates in warm/humid areas (e.g., Southeast Asia).
AD: Elevated prevalence in Africa/Oceania versus Northern/Eastern Europe. Urban areas in China report higher AD rates than rural regions, potentially linked to urban allergen exposure [3].
Psoriasis: Psoriasis can manifest at any age, with most patients exhibiting winter exacerbations or recurrences and summer remission. This seasonality is likely attributed to dry winter conditions that accelerate transepidermal water loss and compromise barrier integrity. The overall incidence has an upward trend, and the incidence rate of late-onset psoriasis in elderly women has increased.
AD: Peak incidence in childhood, then gradually decreased, and was consistent in the region after adolescence. Spring/summer flares correlate with allergen surges and ultraviolet (UV) exposure. Global prevalence increases due to pollution and lifestyle shifts [11, 20, 21].
Vitiligo: Vitiligo demonstrates a global prevalence of 0.5%–2% with no predominant age-or sex-specific clustering, though pediatric and adolescent cases are more prevalent. Most patients experience seasonal stabilization during autumn/winter and spring/summer exacerbations, particularly during seasonal transitions. This pattern likely reflects the combined effects of heightened UV radiation and metabolic acceleration in warmer months, where UV-induced oxidative stress may damage melanocytes and disrupt melanogenesis. Mirroring trends in other chronic dermatoses, vitiligo incidence shows a progressive rise globally, with notably increasing pediatric cases potentially linked to environmental triggers [7, 8, 9, 10].
Single nucleotide polymorphisms (SNPs), defined as genome-wide DNA sequence variations caused by single-nucleotide substitutions, serve as critical genetic markers for gene mapping and disease association studies. By analyzing SNP-disease correlations, researchers can pinpoint susceptibility loci linked to pathological mechanisms, providing actionable insights for diagnostic refinement and therapeutic development [11, 20]. Different chronic skin diseases have different SNPs (Table 1):
Psoriasis: Human Leukocyte Antigen C locus (HLA-C) (major susceptibility locus) [21];
AD: FLG (filaggrin loss-of-function), interleukin (IL)-4/IL-13 (Th2 polarization) [6, 13, 14];
Vitiligo: CTLA-4 (Cytotoxic T-lymphocyte-associated Antigen-4, immune checkpoint dysregulation), TYR (Tyrosinase gene, melanogenesis defects) [10, 15].
CSDs inflict profound physical and psychological burdens (e.g., chronic pruritus, depression, social stigmatization) while driving substantial healthcare costs. Advancing research into pathogenesis and targeted therapies remains imperative to alleviate patient suffering and socioeconomic strain.
Table 1 summarizes what has been mentioned above. It can be seen that there are many overlapping factors, such as temporal trend, gender, contribution among the three chronic skin diseases.
Inflammatory cytokines play central roles in chronic skin disorders by
initiating and sustaining pathological inflammation. Key mediators such as
IL-1
| Psoriasis | AD | Vitiligo | |
| Immune cells | T lymphocytes | T lymphocytes | T lymphocytes |
| B lymphocytes | B lymphocytes | Dendritic cells | |
| Dendritic cells | Dendritic cells | NK cells | |
| Macrophages | Mast cells | ILCs | |
| Neutrophils | Eosinophils | ||
| Non-immune cells | Keratinocyte | Keratinocyte | Keratinocyte |
| Fibroblast | Fibroblast | Endothelial cell | |
| Endothelial cell | Endothelial cell | Melanocyte | |
| Inflammatory factors | IL-17, IL-23, TNF- |
IL-4, IL-5, IL-13, IL-17, IL-31, IFN- |
IL-1, IL-6, IL-17, TNF- |
| References | [2, 22, 23, 27] | [14, 24, 26, 28] | [25, 29] |
IFN-
Psoriasis: The Th17/IL-17 axis dominates, where IL-23 activates Th17 cells to
secrete IL-17A/F, driving keratinocyte hyperproliferation (epidermal thickening,
scaling) and amplifying IL-6/TNF-
AD: Th2 polarization (IL-4, IL-13) suppresses barrier protein expression,
compromising skin integrity, while IL-31 mediates pruritus, perpetuating the
itch-scratch cycle [16, 24]; TNF-
Vitiligo: Interferon-gamma (IFN-
IL-17 (interleukin-17) can promote Th17 cell-mediated autoimmunity in some
patients (Table 2, Fig. 1), induce keratinocytes to secrete IL-6 and IL-8, and
recruit neutrophils and T cells [26]. Cytokines like IL-4/IL-13 impair
keratinocyte differentiation (barrier dysfunction, microbial colonization), while
transforming growth factor
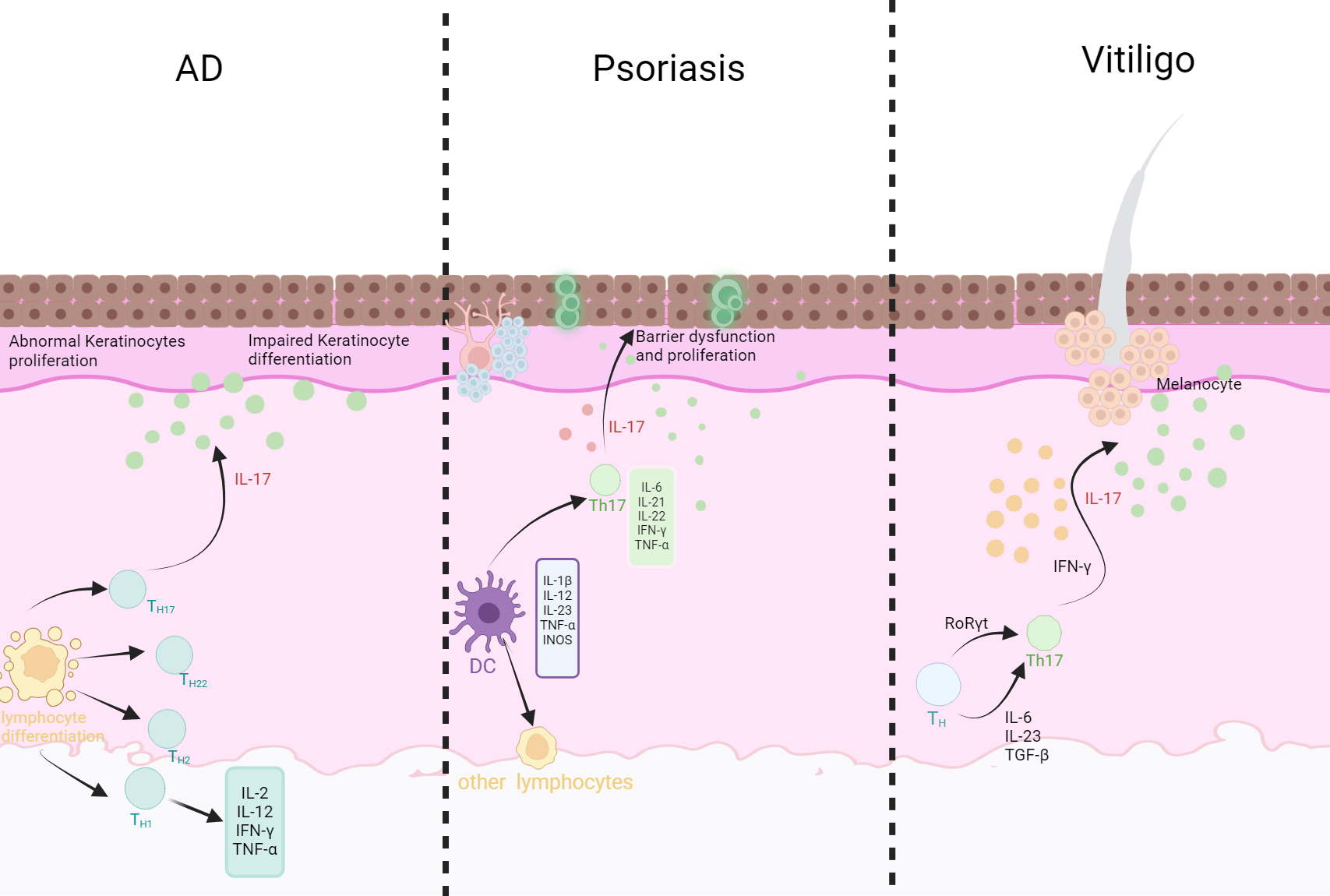 Fig. 1.
Fig. 1.
The pathogenesis of three chronic skin diseases. Atopic
Dermatitis (AD): Th1 cells secrete IFN-
These cells and inflammatory factors are involved in different chronic skin diseases, but they play different roles. Therefore, precise blockade of specific factors has become a key strategy for treating chronic skin diseases. The use of biologics targeting the above-mentioned inflammatory factors for the treatment of chronic skin diseases further confirms the core pathogenic mechanism of inflammatory factors.
Initiation refers to the initial phase where pathogenic factors breach host defenses. Progression involves dynamic pathological expansion from localized to systemic involvement, while maintenance denotes persistent chronicity with sustained pathological processes.
Initiation: Triggered by genetic predisposition and environmental interactions.
Progression: Driven by Th17-mediated inflammatory circuits (e.g., IL-23/IL-17 axis).
Maintenance: Sustained by immune memory and epigenetic dysregulation [2, 30, 31, 32].
Initiation: Originates from genetic barrier defects and environmental sensitization.
Progression: Dominated by Th2 polarization (IL-4/IL-13), with chronic
inflammation marked by IFN-
Maintenance: Linked to epigenetic alterations and immune memory, akin to psoriasis [6, 24].
Initiation: Genetic susceptibility and oxidative stress trigger melanocyte-directed autoimmunity.
Progression: Sustained by IFN-
Maintenance: Perpetuated by immune memory and melanocyte stem cell depletion, resulting in chronicity [7, 9, 19].
Disease staging guides therapeutic strategies: progression phases require inflammation control (e.g., biologics targeting IL-17/IL-23), while maintenance necessitates multimodal approaches (e.g., targeted therapies combined with lifestyle modifications), as summarized in Table 2 and Table 3 (Ref. [2, 7, 8, 9, 10, 13, 22, 24, 26, 27, 28, 31, 32, 33]). Precision in staging enhances treatment efficacy and long-term outcomes.
| Psoriasis | AD | Vitiligo | |
| Initiation | Genetics, immunity, environmental factors | Genetics, immunity, environmental factors, abnormal skin barrier function | Genetic, immune, environmental factors, melanocyte autoimmunity, micronutrient deficiencies |
| Progression | Immune-inflammatory response, neuroendocrine factors, abnormal keratinocyte proliferation | Immune-inflammatory response, neuroendocrine factors, and changes in the composition and structure of the skin surface microbiota | Immune-inflammatory response, neuroendocrine factors, melanocyte dysfunction |
| Maintenance | Complications, recurrent/chronic disease course | Complications, recurrent/chronic disease course | Complications, recurrent/chronic disease course, expansion of existing depigmented lesions and emergence of new ones |
| References | [2, 22, 27, 31, 32] | [13, 24, 26, 28] | [7, 8, 9, 10, 33] |
Comparison within this table reveals numerous shared factors in the pathogenesis of different chronic skin diseases, primarily involving immune and genetic components. Combined with the comparison in Table 2, it is evident that inflammatory factors and immune-genetic related factors play a dominant role in the pathogenesis of chronic skin diseases.
Having outlined the complex cellular and cytokine networks underlying psoriasis,
AD, and vitiligo (Section 3). As shown in the Venn diagram (Fig. 2), we now focus on the specific roles of the pivotal
inflammatory mediators that drive these shared pathways. This section delves into
the mechanistic contributions of IL-17, TNF-
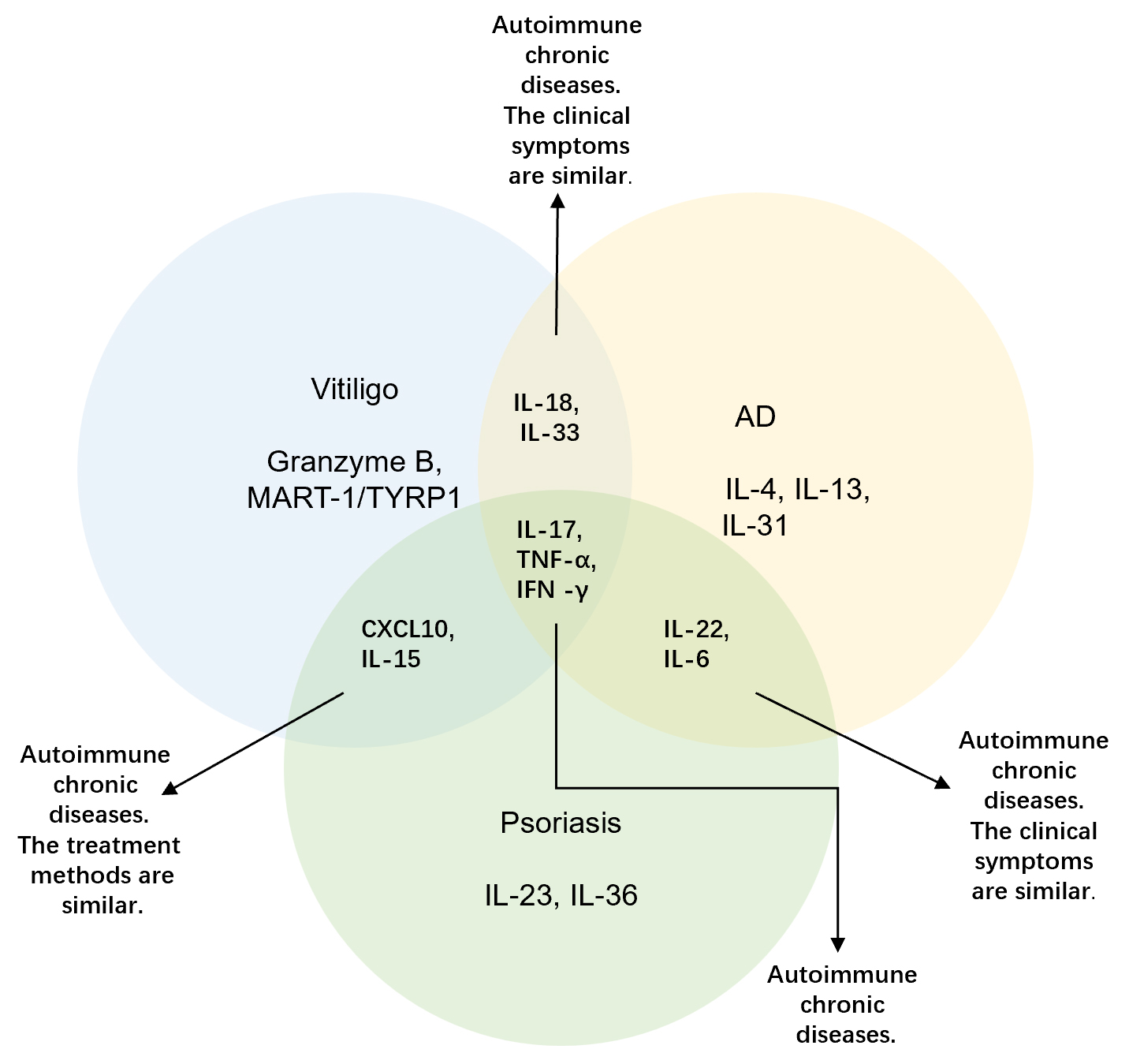 Fig. 2.
Fig. 2.
Venn diagram of three chronic skin diseases. Core Inflammatory
Networks in Chronic Skin Disorders: Vitiligo: Granzyme B-mediated damage and
T-cell responses to melanocyte antigens. Psoriasis: IL-23/IL-17 axis and
IL-36-driven hyperplasia. AD: Th2 cytokines (IL-4/13/31) causing barrier
dysfunction. Shared Pathways: • Vitiligo/Psoriasis: CXCL10
recruitment + IL-15 survival. • Vitiligo/AD: IL-18/33 alarmin
activation. • Psoriasis/AD: IL-6 acute-phase + IL-22 remodeling.
Universal Drivers: IL-17 (epidermal hyperplasia), TNF-
AD: Th17 cells contribute to pathogenesis via IL-17A, which activates keratinocyte inflammatory networks (e.g., STAT3 signaling), recruits’ neutrophils, and induces aberrant keratinocyte proliferation. This disrupts skin barrier function, exacerbating inflammation and perpetuating the itch-scratch cycle [34, 35, 36, 37].
Psoriasis: IL-17A binds keratinocyte receptors, triggering inflammatory cascades
(e.g., NF-
Vitiligo: Elevated IL-17 in lesional skin directly suppresses melanocyte proliferation, melanogenesis, and differentiation and enzymatic activity [41, 42]. Concurrently, it disrupts immune equilibrium, promoting autoimmune melanocyte destruction [43, 44].
AD & Psoriasis: Acting on keratinocytes, it can stimulate the release of
various inflammatory mediators, exacerbate inflammatory reactions, and also
affect the function of immune cells, exacerbating the recurrence of AD disease.
Similarly, TNF-
Vitiligo: TNF-
Psoriasis: Similar to TNF-
AD: Reduced IFN-
Vitiligo: IFN-
Given the above pathogenesis, the development of targeted therapy strategies has become crucial. Targeted therapy, by precisely intervening in the key pathways of diseases, not only breaks through the optimal therapeutic effects of traditional treatments but also reshapes the treatment pattern through molecular subtyping and individualized approaches. Its value is not only reflected in the improvement of clinical indicators, but more importantly, it reconstructs the treatment goals of chronic skin diseases from “symptom control” to “disease modification”, promotes comorbidity management, and achieves biomarker-stratified therapy. It is a strategic development direction for modern dermatology in treatment. There are currently few research reports on whether these diseases also have some commonalities in existing treatment methods. Below, we analyze therapeutic commonalities and distinctions across psoriasis, AD, and vitiligo:
Psoriasis can be treated with glucocorticoids, vitamin D3 derivatives, retinoids, calcineurin inhibitors, etc.; AD can be treated with glucocorticoids, calcineurin inhibitors, zinc oxide oil, calamine lotion, etc.; Vitiligo can be treated with glucocorticoids, calcineurin inhibitors, and vitamin D3 derivatives. It can be seen that there are many similarities in topical medications for them.
For psoriasis, methotrexate, cyclosporine, biologics such as TNF-
Psoriasis and vitiligo are mostly treated with narrow-band ultraviolet B (NB-UVB), 308 excimer laser, and UVA combined with psoralen (PUVA) [49]; AD treatment often uses narrow-band ultraviolet B (NB-UVB), UVA1, and pulsed dye laser (PDL) [50, 51, 52].
When the condition of patients with vitiligo is in the stable stage, there is no new lesion or expansion of the leukoderma for at least 6 to 12 months, or there is no repigmentation after at least 6 months of drug treatment and phototherapy, surgical treatment can be adopted. Common surgical methods include: Autologous Epidermal Transplantation (AET): a procedure involving the transplantation of non-cultured epidermal sheets or cell suspensions derived from the patient’s own healthy skin to the depigmented areas; Autologous Melanocyte Cell Transplantation (MCAT): specifically referring to the in vitro expansion of melanocytes isolated from a patient’s healthy skin biopsy, followed by their transplantation into the vitiligo lesions.
Commonly used prescriptions for psoriasis treatment in TCM include Xiaoyin Granules, Compound Indigo Naturalis Capsules, etc., [50, 53]. The commonly used prescriptions in AD are Xiaofeng San and Siwu Tang. Common prescriptions for vitiligo include Liuwei Dihuang Wan, Tongqiao Huoxue Tang, etc. In addition, traditional Chinese medicine fumigation, acupuncture, traditional Chinese medicine tinctures, or ointments can also be used for treatment [54].
Regarding the treatment methods and pathogenesis of these three chronic skin
diseases, there are many commonalities in topical drug treatment. For example,
glucocorticoid is the first-line drug for all three diseases: psoriasis (potent
glucocorticoids such as halometasone), AD (medium-potency glucocorticoids such as
hydrocortisone), and vitiligo (local block therapy). However, long-term use can
cause skin atrophy (with an incidence of about 20%–30%) [49]. Calcineurin
inhibitors (tacrolimus/pimecrolimus) are the first-line non-hormonal drugs for AD
(the first choice for children), the second-line treatment for vitiligo (with an
effective rate of 50%–60% on the face and neck), and are used in thin skin
areas for psoriasis patients [50]. Vitamin D3 derivatives (Calcipotriol) are the
first-line combination drugs for psoriasis (with a Psoriasis Area and Severity
Index (PASI) score improvement of
Current therapeutic approaches for chronic skin diseases offer diverse options for personalized treatment; however, all modalities carry inherent risks of adverse effects and disease recurrence. Long-term topical or systemic glucocorticoids use may lead to skin atrophy, telangiectasia, hyperpigmentation, Cushing’s syndrome, hyperglycemia, hypertension, and osteoporosis. Abrupt discontinuation can trigger rebound exacerbations. Other topical agents may cause skin irritation, dryness, desquamation, erythema, pruritus, or burning sensations. Systemic therapies, including oral medications, are associated with gastrointestinal disturbances, hepatorenal toxicity, bone marrow suppression, drowsiness, dizziness, xerostomia, and mucosal dryness. Phototherapy, commonly employed for these conditions, increases the risk of erythema, pruritus, blistering, and photocarcinogenesis [52]. Biologic therapies may induce injection-site reactions (redness, pain, itching), headaches, dizziness, hematologic abnormalities, elevated liver enzymes, gastrointestinal disturbances [51, 57, 58], and immunogenicity-driven drug resistance due to anti-drug antibody formation. Such resistance not only compromises disease control but also escalates treatment costs and limits therapeutic options.
The three chronic skin diseases discussed—psoriasis, AD, and vitiligo—share
overlapping pathogenic mechanisms mediated by IL-17, IFN-
(1) Induce keratinocytes and immune cells to release pro-inflammatory chemokines (e.g., IL-6, IL-8, CXCL10), recruiting neutrophils and T cells to amplify inflammation.
(2) Drive keratinocyte proliferation and secretion of inflammatory factors: Causing psoriasis epidermal hyperplasia and AD barrier disruption.
(3) Synergize in immune polarization: TNF-
IL-17 inhibitors (e.g., Secukinumab, Ixekizumab), validated for psoriasis. Then, whether this preparation can also be used in the severe AD subtype with high Th17 activity. Preclinical studies demonstrate that IL-17A blockade reduces melanocyte apoptosis and promotes repigmentation in vitiligo models [54], positioning IL-17 as a shared inflammatory hub across these diseases. Providing a theoretical basis for “one drug, multiple diseases”.
Current evidence indicates that IL-17 inhibitors demonstrate significant efficacy in psoriasis and show therapeutic potential for specific subtypes of AD and vitiligo. While large-scale clinical trials are still required for validation, future advancements in precision subtyping, combination therapies, and novel dual-target agent development hold promise for realizing the “one drug, multiple diseases” paradigm. Such strategies could enhance drug resistance mechanisms, reduce side effects of existing inhibitors, expand therapeutic scope, and advance individualized treatment.
The growing heterogeneity of chronic skin diseases has rendered traditional
clinical classification methods—based on lesion morphology or disease
staging—insufficient for precision medicine. Artificial intelligence (AI),
particularly machine learning and deep learning, offers innovative solutions by
integrating multi-omics data (genomic, transcriptomic, proteomic) with clinical
phenotypic information. AI not only overcomes the limitations of conventional
subtyping but also elucidates dynamic regulatory networks of central pathways
(e.g., IL-17, IFN-
Future interdisciplinary collaborations should integrate AI with liquid biopsy, real-time sensors, and other technologies to enable continuous monitoring of disease-related biomarkers and real-time acquisition of patients’ physiological and environmental data. This integration will facilitate the development of personalized treatment regimens. AI-driven dermoscopic image analysis systems could dynamically monitor skin disease progression and adjust therapeutic strategies in real time, optimizing outcomes and enabling dynamic precision management of chronic skin conditions.
Furthermore, establishing cross-disease multi-omics databases and deeply integrating AI-assisted decision systems with electronic health records will consolidate multimodal data. This approach will transition the “one drug, multiple diseases” concept from empirical medicine to precision science, maximizing the therapeutic breadth and depth of single agents. Data-driven decision-making will achieve the tripartite precision medicine goals of individualization, dynamic adaptation, and preventive care, ultimately revolutionizing the management of chronic skin diseases.
This review underscores that chronic skin conditions, including psoriasis, AD, and vitiligo, share common immunopathological mechanisms, centered on complex inflammatory cascades driven by overlapping cytokines and immune cell crosstalk.
(1) Shared Inflammatory Drivers: IL-17, TNF-
(2) Efficacy of Single-Agent, Multi-Disease Therapies: IL-17 axis inhibitors have demonstrated robust efficacy in psoriasis and emerging promise in AD or vitiligo. This approach enhances treatment efficiency, reduces adverse effects, and marks a pivotal shift from disease-specific care to mechanism-driven therapy.
(3) Future Directions: Integration of AI with multi-omics data will enable highly precise immune phenotyping, empowering clinicians to dynamically tailor treatments. This will expand the utility of single-agent therapies and transform chronic skin disease management into a personalized, data-driven practice.
By merging mechanistic insights with innovative technologies, we can develop more effective, scalable treatments—ultimately improving outcomes for patients with these debilitating conditions.
Conceptualization, RTM, WYM; writing—original draft preparation, RTM, WYM; writing—review and editing, RTM, WYM. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We sincerely appreciate the contributions of everyone who supported this manuscript’s development, including the peer reviewers for their invaluable insights and suggestions.
This research received no external funding.
The authors declare no conflict of interest.
During the preparation of this work, the authors used ChatGPT for language translation assistance. After using this tool, the authors thoroughly reviewed, revised, and edited the content as needed and take full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


