1 Department of Nephrology, The Second Affiliated Hospital of Qiqihar Medical University, 161006 Qiqihar, Heilongjiang, China
2 College of Basic Medical Sciences, Qiqihar Medical University, 161006 Qiqihar, Heilongjiang, China
3 Department of Laboratory Medicine, The Second Affiliated Hospital of Qiqihar Medical University, 161006 Qiqihar, Heilongjiang, China
Abstract
Chronic kidney disease (CKD), driven by progressive renal fibrosis, lacks effective therapeutic targets. This study investigates thrombospondin-4 (THBS4) as a novel mediator of CKD-related fibrosis and explores its mechanistic basis.
This study collected 100 patients diagnosed with chronic kidney disease and 30 healthy individuals. Enzyme-linked immunosorbent assay (ELISA) analysis was conducted to assess the expression of THBS4 in CKD patients. Mouse unilateral ureteral obstruction (UUO) renal fibrosis model and Human Kidney-2 (HK2) cell fibrosis model were constructed to analyze the expression changes of THBS4 in renal fibrosis. To examine the effects of inhibiting THBS4 expression on the process of renal fibrosis, these two models were analyzed using Sirius red staining, Masson staining, immunohistochemistry, real-time quantitative PCR (qPCR) and western blot methods.
The expression of THBS4 in the serum of CKD patients was found to be significantly higher (p < 0.05), and its concentration showed a negative correlation with the eGFR levels (r = –0.77, p < 0.05) and an increase corresponding to the progression of CKD stages (p < 0.05). THBS4 expression was dramatically increased in UUO-treated mouse kidneys as well as in TGF-β1-stimulated HK2 cells (p < 0.05). In vitro, the expression of renal fibrosis-associated proteins was also significantly reduced after interfering with THBS4 expression (p < 0.05). UUO-induced renal fibrosis and related protein expression were suppressed in THBS4 knockdown mice when compared to control mice (p < 0.05). The levels of p-AKT and p-PI3K exhibited a significant rise in conjunction with the onset of renal fibrosis (p < 0.05). The expression of p-AKT as well as p-PI3K showed a significant reduction upon inhibition of THBS4 expression (p < 0.05). Insulin-like growth factor 1 (IGF-1) treatment reversed these effects.
THBS4 was significantly overexpressed in CKD patients. By suppressing the expression of proteins associated with renal fibrosis and inhibiting the activation of the PI3K/AKT pathway, THBS4 has the potential to mitigate renal fibrosis.
Keywords
- chronic kidney disease
- thrombospondin-4
- renal fibrosis
- fibrosis-associated protein
- PI3K/AKT pathway
Chronic kidney disease (CKD) is a common disease in the global periphery. Epidemiologic studies have shown that the incidence and prevalence of CKD has risen by nearly 90% over the past 30 years [1]. This is expected to increase as the population ages. World Health Organization surveys have shown that the annual mortality rate attributed to CKD is estimated to range from 50 to 100 million deaths. It is estimated that by 2040, CKD will be the fifth leading cause of death worldwide [2]. The lack of disease awareness contributes significantly to the elevated morbidity and mortality rates associated with CKD. Since the early stages of CKD have no clinical symptoms, this stage is easily overlooked and undertreated. It is estimated that about 94% of patients with mild to moderate decline in renal function and about 48% of patients with severe renal insufficiency are undiagnosed [3, 4].
Renal fibrosis plays a crucial role in the advancement of CKD and is regarded as
the ultimate shared pathological alteration across various kidney diseases. CKD
is characterized by persistent inflammation, which initiates the fibrotic
process. The recruitment of inflammatory cells, including lymphocytes,
granulocytes, and macrophages, is a result of the inflammatory process. These
cells then release transforming growth factor
A secreted extracellular matrix protein, thrombospondin-4 (THBS4) belongs to the thrombospondin family [10]. As a crucial constituent of the extracellular milieu, THBS4 assumes a pivotal function in cellular attachment, movement, growth, and programmed cell death [11, 12]. Recent studies have shown that THBS4 plays a role in the process of wound healing and tissue restructuring through its regulation of extracellular matrix organization, repair, and remodeling [13, 14]. In addition, the mechanism of action of THBS4 is gradually being revealed in cardiovascular diseases, tumor biology, and neurodegenerative diseases [15, 16, 17].
While THBS4’s significance in other biological systems has been extensively investigated, little is known about its function in CKD, specifically with regard to the procession of renal fibrosis. Our assumption was that THBS4 during CKD could potentially regulate the progression of renal fibrosis by influencing the modulation of extracellular matrix and signaling pathways, including the phosphatidylinositol 3-kinase/serine-threonine kinase (PI3K/AKT) pathway. Further, we tested this hypothesis through clinical sample analysis and animal model studies and investigated the potential of THBS4 as a viable target for therapeutic intervention.
One hundred patients diagnosed with CKD from the Second Affiliated Hospital of
Qiqihar Medical University and 30 healthy people were collected for inclusion in
this study. Patient inclusion exclusion criteria: Patients attending our clinic
were identified as having chronic kidney disease after routine urinalysis and
renal function tests. Renal transplant recipients and patients with acute kidney
injury were excluded. The baseline data of the patients was recorded at the time
of diagnosis (Supplementary Table 1), and blood samples were collected
simultaneously. Based on the guidelines developed by Kidney Disease: Improving
Global Outcomes (KDIGO) [3], patients with CKD can be categorized into five
stages based on estimated glomerular filtration rate (eGFR): CKD1 (eGFR,
The study procured male C57BL/6 mice, aged 6–8 weeks, with an average weight of 25–30 grams, and THBS4-knockdown mice (shRNA-THBS4) were generated by intravenous injection of AAV9 particles expressing shRNA targeting THBS4 (Guangzhou Ruige Bio-Tech Co., Ltd., Guangzhou, China), while control mice (scramble) received AAV9-scramble-shRNA (Guangzhou Ruige Bio-Tech Co., Ltd., Guangzhou, China). A total of 12 mice were used in this study. All animal experiments comply with the international ethical standards for experimental animals and the regulations designated by the Second Affiliated Hospital of Qiqihar Medical University Ethics Committee (ethics number: [2022]0815-8-2). Human-derived renal tubular proximal epithelial cell line HK2 was purchased from Hunan Fenghui Biotechnology Co., Ltd. (CL0144, Changsha, Hunan, China).
Six 6–8 week-old healthy mice were prepared for each group. The mice were anesthetized by intraperitoneal injection of 2% sodium pentobarbital (4 mL/kg). In the experimental group, an incision was made on the dorsal side of the mice to reveal the left ureter. The ureter was then tied off using two 4.0-gauge silk sutures (0.35 mm diameter). The sham-operated group was operated only without ureteral ligation. 14 days later, mice were anesthetized as described above and then cervical dislocation was performed on the mice and kidney tissues were collected. Some kidneys were fixed with 4% formalin, followed by dehydration and paraffin embedding, and the remaining tissues were stored in a cryogenic liquid nitrogen environment for preservation [18].
HK2 cells were cultured in Dulbecco’s Modified Eagle Medium/Nutrient Mixture
F-12 (DMEM/F12; C11330500BT, Gibco, Grand Island, NY, USA) supplemented
with 10% fetal bovine serum (FBS; 10270106, Gibco, Grand Island, NY, USA)
and 1% penicillin/streptomycin (15140122, Gibco, Grand Island, NY, USA) at
37 °C under 5% CO2. For experimental interventions, cells were
divided into six groups: Control (untreated), si-NC (transfected with scramble
siRNA), si-THBS4 (transfected with THBS4-specific siRNA;
sequence: 5′-CGCCCUGAAUGAUCUCUAUTT-3′; L31520, Beyotime, Shanghai,
China), si-NC + TGF-
Sections were routinely deparaffinized and heat repaired in antigen repair solution for 35 min, and then immunohistochemical (IHC) assays were performed according to the instructions of IHC kit (36311ES50, Yeasen Biotechnology, Shanghai, China). The sections were treated with 3% H2O2 at a temperature of 4 °C for a duration of 10 minutes, followed by rinsing with phosphate belanced solution (PBS) for three cycles, each lasting 2 minutes; incubated with Avidin at room temperature for 15 min and rinsed with PBS for three cycles, each lasting 2 minutes/3 times; exposed to Biotin for a duration of 15 minutes at ambient temperature, followed by a 5-minute rinse with PBS; subsequently, the blocking solution was introduced and the sections were subjected to a 20-minute blocking process at ambient temperature. The blocking solution was removed, and the sections were exposed to the primary antibody (THBS4, 1:100, CSB-PA562708, Wuhan Huamei BIOTECH, Wuhan, China). for an extended period at a low temperature of 4 °C. Wash with PBS for a duration of 5 minutes, followed by incubation with the secondary antibody (Horseradish Peroxidase (HRP) Goat Anti-Rabbit, 1:2000, ab205718, abcam, Cambridge, UK) at ambient temperature for a period of 30 minutes. Rinse with PBS for 5 min, add 3-3′-diaminobenzidine (DAB) chromogenic solution for 5–10 min, rinse with water, re-stain, dehydrate, clear and seal. For the quantitative analysis of immunohistochemical staining, the intensity of staining was evaluated using the H-score method.
Paraffin sections were deparaffinized and subjected to PAS staining as described below. The sections were subjected to a 15-minute immersion in a 1% solution of periodic acid, followed by rinsing with water for 2–3 cycles. The sections underwent staining by being immersed in Schiff’s solution for a duration of 10–30 minutes, followed by rinsing with water 2–3 times. Stained with Haematoxylin for 1–2 min, washed 2–3 times in water. Immersion in 1% hydrochloric acid-ethanol for 5 sec, wash in water 2–3 times. Ammonia counterblue for 5–10 sec, rinsed in water, dehydrated, transparent, and sealed. Masson (ALP87019, Thermo Fisher Scientific, Waltham, MA, USA) and Picro Sirius Red (PSR) (36324ES60, Yeason Biotechnology, Shanghai, China) staining was performed according to the operating instructions of the kit. Staining results were quantitatively analyzed using Image-Pro Plus software (5.0.2, Media Cybernetics, Rockville Pike, MD, USA).
Patient serum was obtained by collecting patient whole blood and centrifuging the supernatant. The amount of THBS4 was detected based on the instructions of the ELISA kit (XG-E990828, Shanghai Sig Biotechnology Co., Ltd., Shanghai, China). The absorbance value of the test wells at 450 nm was read by an enzyme meter. The formula for calculating the concentration (C) of THBS4 in the samples based on the OD value (OD_sample) is as follows:
Where:
C is the concentration of THBS4 in the sample,
OD𝑠𝑎𝑚𝑝𝑙𝑒 is the optical density of the sample,
OD𝑏𝑙𝑎𝑛𝑘 is the optical density of the blank (background),
C𝑚𝑎𝑥 and C𝑚𝑖𝑛 are the maximum and minimum concentrations of the standard curve, respectively,
OD𝑚𝑎𝑥 and OD𝑚𝑖𝑛 are the corresponding optical densities of the standard curve.
Total RNA was extracted from tissues and cells using Trizol reagent (15596018, Invitrogen, Carlsbad, CA, USA). First strand DNA was synthesized by cDNA reverse transcription kit using RNA as a template. The reaction solution was prepared by SYBR Select Master Mix kit (4472919, ABI, Foster City, CA, USA) using DNA as a template. The reaction solution was used to complete quantitative fluorescence assay in a quantitative PCR instrument (Bio-Rad, Hercules, CA, USA) to accomplish quantitative fluorescence detection. The assay results were calculated using the 2-ΔΔCt method. The amplification primer sequences of genes are shown in Table 1 (Human) and Table 2 (mouse).
| Genes | Sequence (5′-3′) |
| COL1A1 | Forwards: GATTCCCTGGACCTAAAGGTGC |
| Reverse: AGCCTCTCCATCTTTGCCAGCA | |
| FN1 (encoding Fibronectin) | Forwards: ACAACACCGAGGTGACTGAGAC |
| Reverse: GGACACAACGATGCTTCCTGAG | |
| ACTA2 (encoding |
Forwards: CTATGCCTCTGGACGCACAACT |
| Reverse: CAGATCCAGACGCATGATGGCA | |
| THBS4 | Forwards: CCTTCTGAGACAGCAGGTTAAG |
| Reverse: GTCGGAGACTGAAACTTGAGAG | |
| GAPDH | Forwards: GGAGCGAGATCCCTCCAAAAT |
| Reverse: GGCTGTTGTCATACTTCTCATGG |
qPCR, real-time quantitative PCR; COL1A1, collagen type I alpha 1; FN1, fibronectin 1; ACTA2, actin alpha 2; THBS4, thrombospondin-4; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
| Genes | Sequence (5′-3′) |
| Col1a1 | Forwards: CGATGGATTCCCGTTCGAGT |
| Reverse: TTCGATGACTGTCTTGCCCC | |
| Fn1 (encoding Fibronectin) | Forwards: CGAGGTGACAGAGACCACAA |
| Reverse: CTGGAGTCAAGCCAGACACA | |
| Acta2 (encoding |
Forwards: GAGGCACCACTGAACCCTAA |
| Reverse: CATCTCCAGAGTCCAGCACA | |
| Thbs4 | Forwards: GACGGCTATGTCTGTGGAAAGG |
| Reverse: TCCGATGCCATCTCTGTCTGCA | |
| Gapdh | Forwards: GTGCTGAGTATGTCGTGGAG |
| Reverse: TCGTGGTTCACACCCATCAC |
The Radio Immunoprecipitation Assay (RIPA) lysis buffer was utilized to extract
the entire cellular proteins, while the protein concentration was determined
using the Bromocresol Green with Albumin (BCA) kit (A55864, Thermo Fisher
Scientific, Waltham, MA, USA). The separation of proteins was achieved
through the utilization of SDS-PAGE gel electrophoresis. The gel that underwent
electrophoresis was transferred onto a polyvinylidene fluoride (PVDF) mold and
sealed with 5% skimmed milk for 1 hour at ambient temperature. The gel was
subsequently subjected to overnight incubation at a temperature of 4 °C
with the primary antibody GAPDH (5174S; 1:1000), collagen type I
alpha 1 (COL1A1, 72026; 1:1000), Fibronectin (26836; 1:1000),
The statistical analysis of the experimental data was conducted utilizing IBM
SPSS statistics 26 software (IBM Corp., Armont, NY, USA). Continuous
variables are presented as mean
Serum was collected from 100 patients diagnosed with CKD as well as from 30
healthy people. ELISA assay showed significantly higher serum levels of
THBS4 in CKD patients compared to healthy control population (Fig. 1A;
p
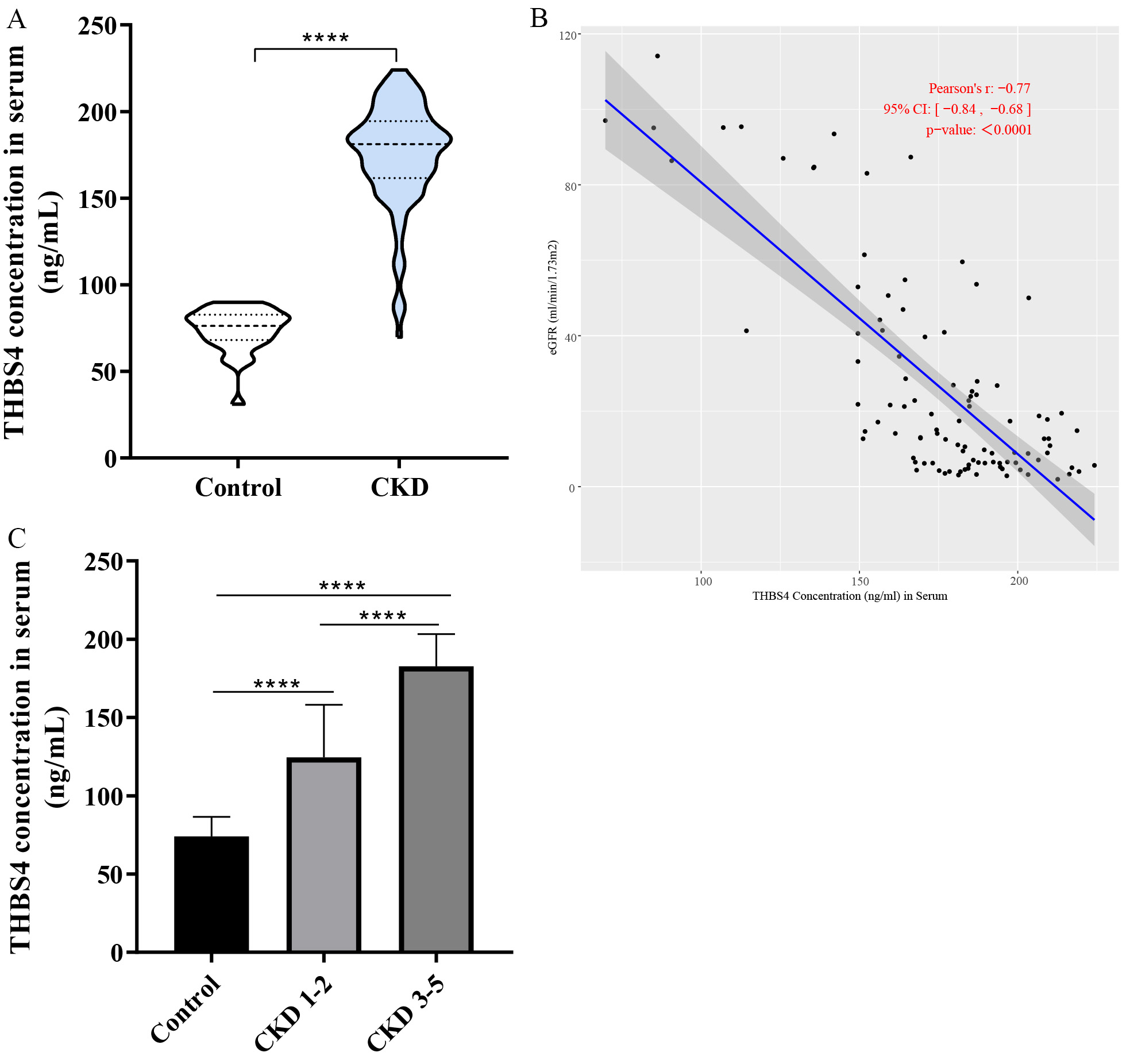 Fig. 1.
Fig. 1.
Expression analysis of THBS4 in clinical CKD patients.
(A) Expression analysis of THBS4 in serum of CKD patients and healthy
population. (B) Correlation analysis of THBS4 content with eGFR. (C)
Relationship between THBS4 content and staging of CKD patients. Control,
n = 30; CKD, n = 100. ****p
A mouse UUO model was constructed to reveal the expression of renal
fibrosis-related indexes. qPCR showed that COL1A1, encoding
Fibronectin (FN1), as well as encoding
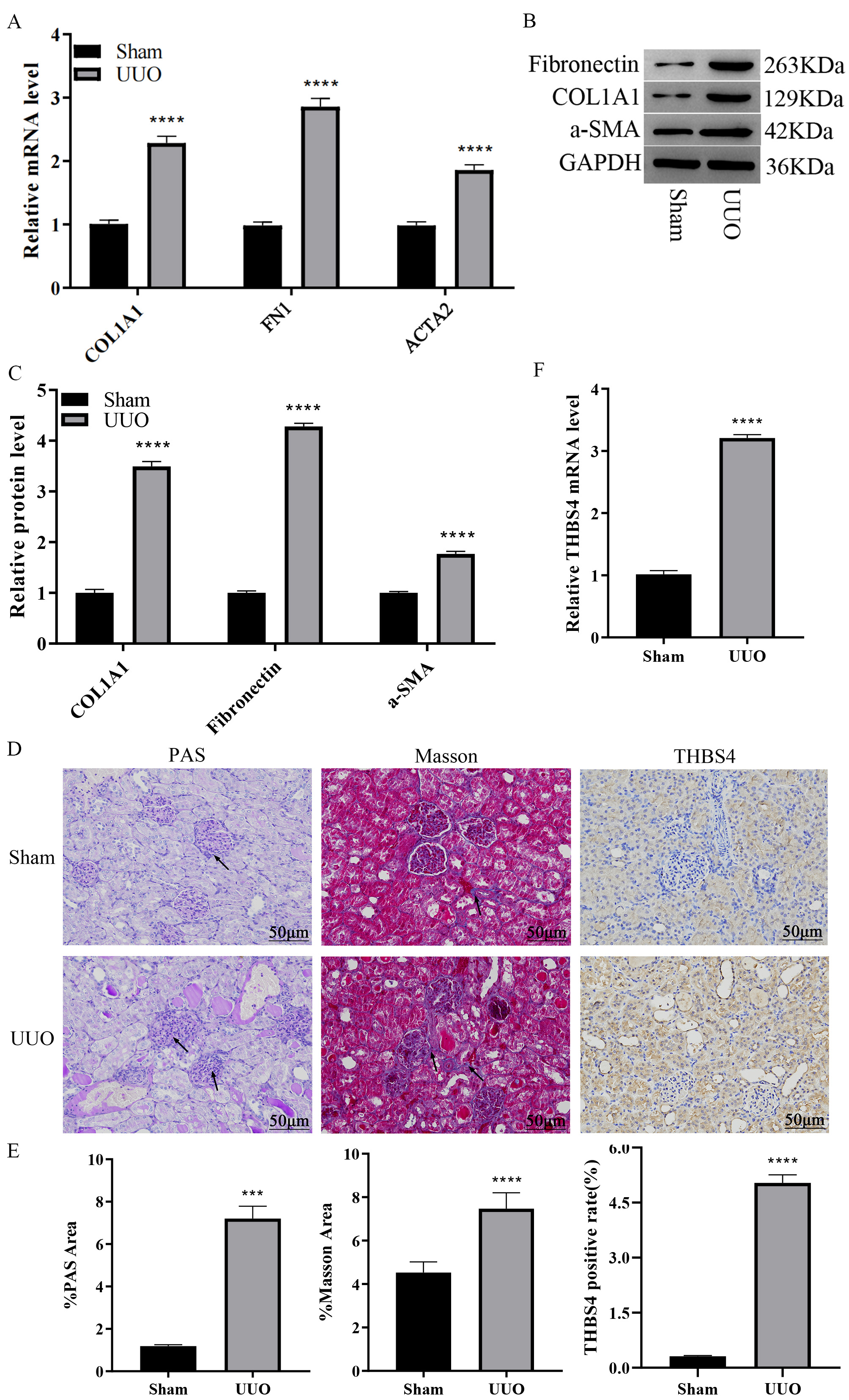 Fig. 2.
Fig. 2.
Expression analysis of THBS4 in the mouse UUO model.
(A) qPCR detection of mRNA expression changes of renal fibrosis indexes after
UUO. (B,C) WB detection of protein expression changes of renal fibrosis indexes
after UUO. (D) PAS, Masson staining, and IHC for detection of renal fibrotic
lesions; Magnification factor: 200
To ensure the efficacy of THBS4 knockdown, both protein and mRNA levels
were assessed. qPCR analysis showed a significant decrease in THBS4 mRNA
expression in the si-THBS4 group (Fig. 3A; p
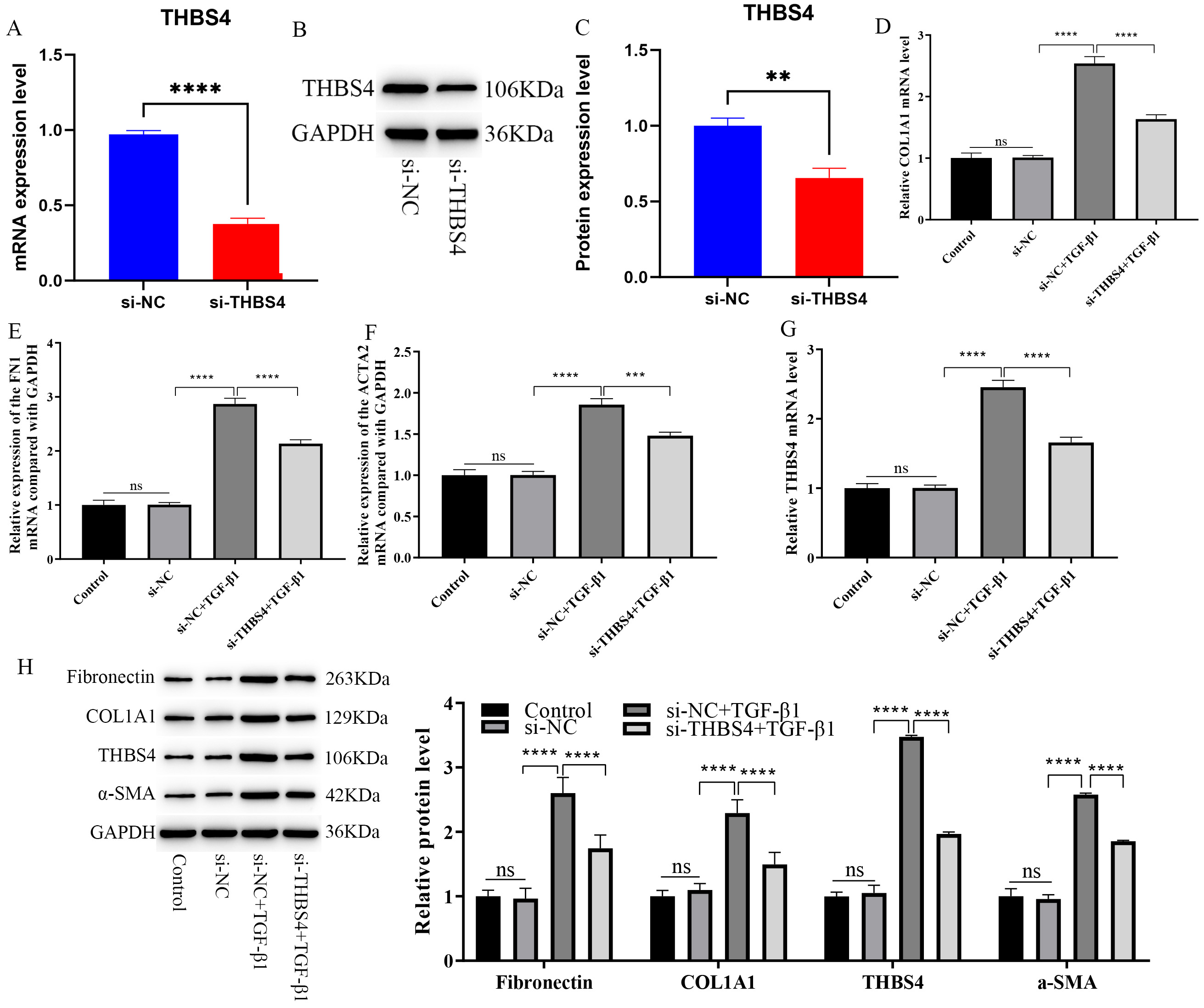 Fig. 3.
Fig. 3.
Effect of inhibiting THBS4 expression on HK2 cell
fibrosis. (A) qPCR validation of THBS4 mRNA knockdown. (B,C) WB
validation of THBS4 protein knockdown. (D–G) qPCR detection of the
effect of inhibiting THBS4 expression on TGF-
Control mice (scramble) and THBS4 knockdown mice (shRNA-THBS4)
were purchased. To confirm the efficacy of THBS4 knockdown in
vivo, both mRNA and protein levels were assessed in kidney tissues from control
mice and THBS4 knockdown mice. qPCR analysis revealed a significant
reduction in THBS4 mRNA expression in knockdown mice compared to control
mice (Fig. 4A; p
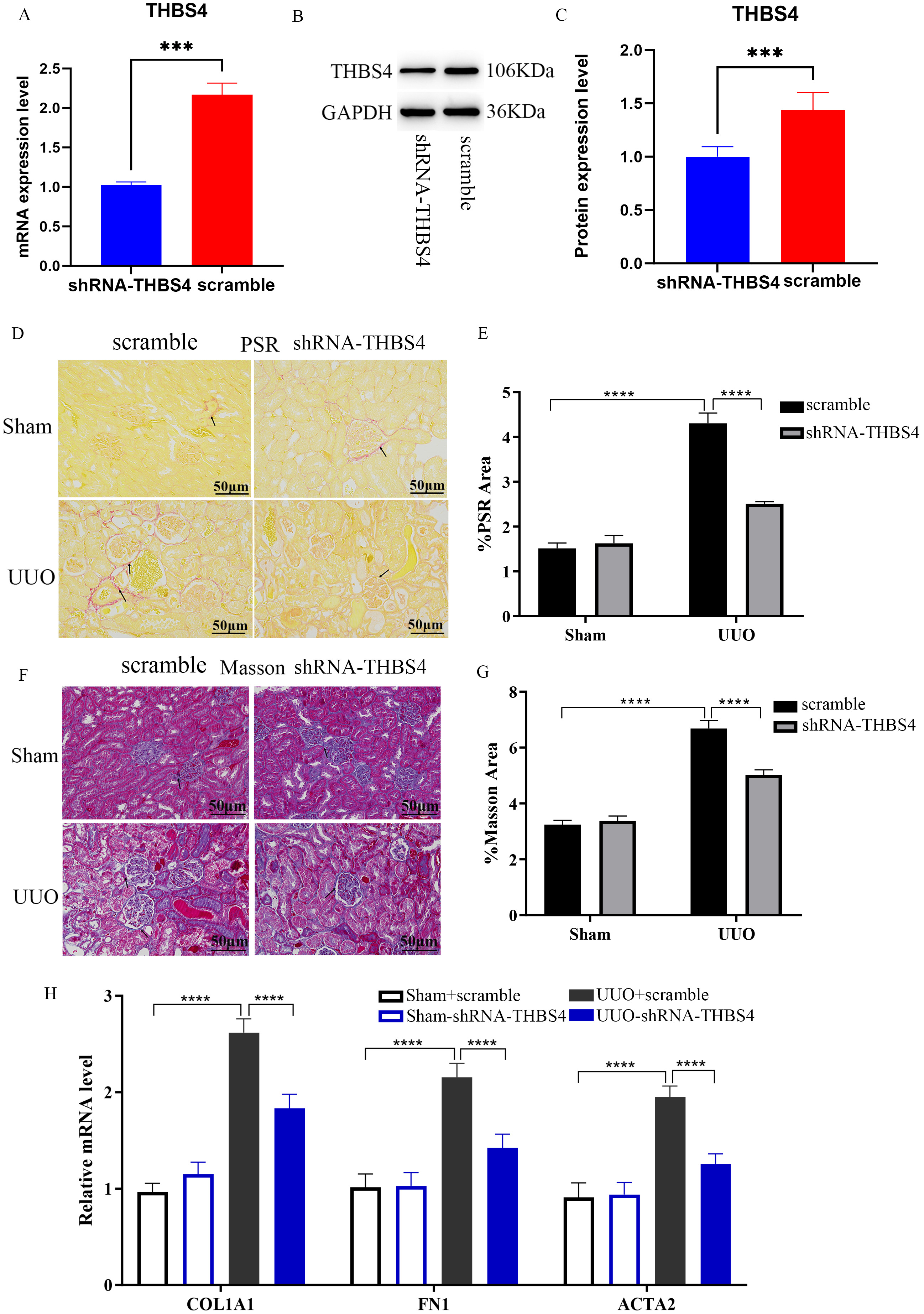 Fig. 4.
Fig. 4.
Mechanism study of THBS4 regulating the process of
renal fibrosis. (A) qPCR validation of THBS4 mRNA knockdown in kidney
tissues. (B,C) WB validation of THBS4 protein knockdown in kidney tissues. (D–G)
PSR and Masson staining to detect the fibrosis of mouse kidney after
THBS4 knockdown. Scale bar = 50 µm. (H) qPCR to detect the effect
of THBS4 knockdown on the expression of renal fibrosis indicators in mice. n = 6.
***p
The above experiments have confirmed that interfering with THBS4
expression can inhibit the renal fibrosis process. We next explored its mechanism
of action. In vitro, WB analysis showed that the protein expression
ratios of p-PI3K/PI3K and p-AKT/AKT were significantly
increased in HK2 cells following TGF-
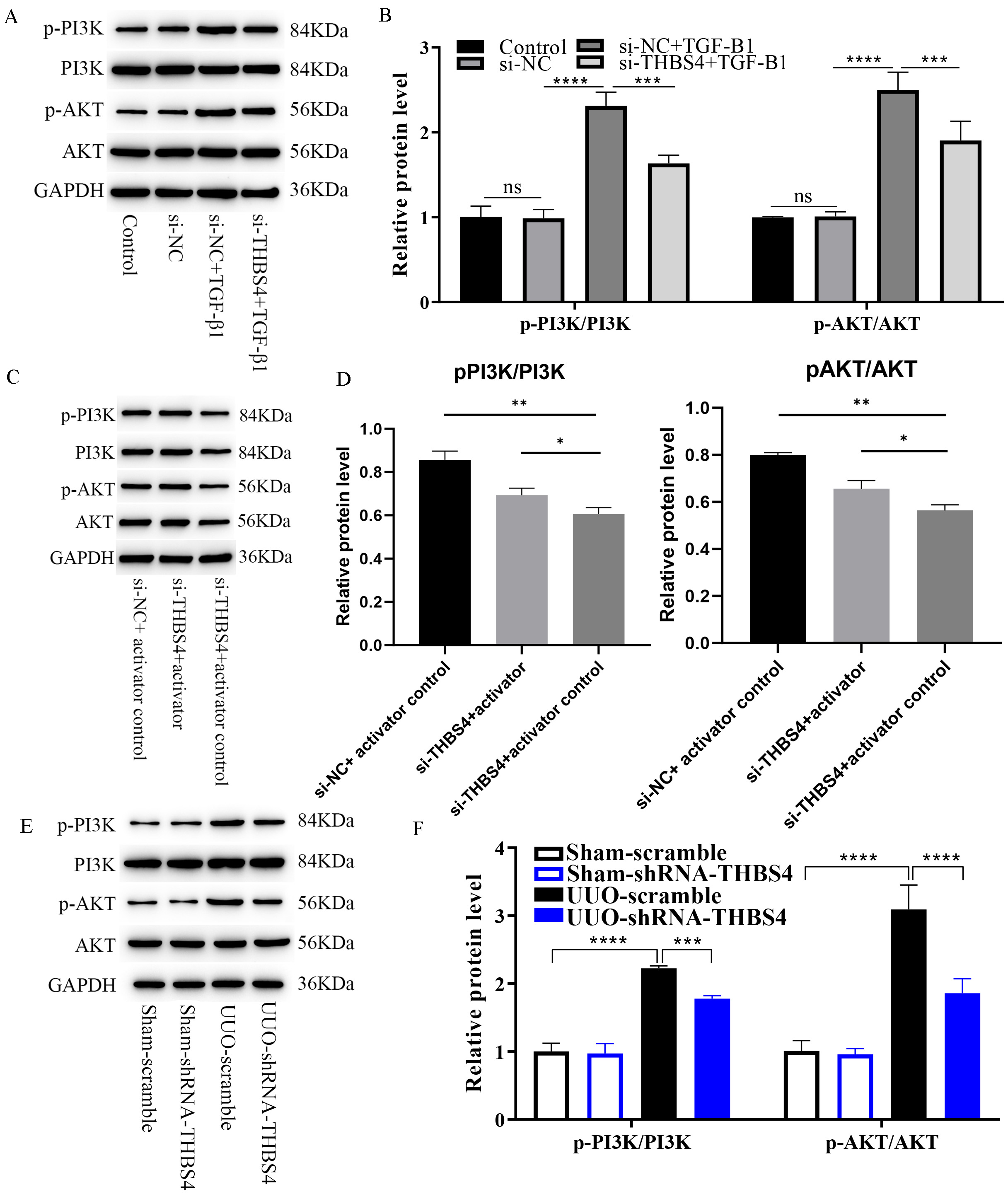 Fig. 5.
Fig. 5.
Reduced THBS4 expression inhibits PI3K/AKT pathway
activation. (A) WB detection of the effect of inhibition of THBS4
expression on PI3K/AKT pathway protein expression in cells, n = 3. (B)
Quantitative analysis of Fig. 5A. (C) WB analysis of IGF-1 activator-treated
cells with TGF-
In serum samples from clinical patients, we observed an inverse association
between eGFR and the expression of THBS4. The higher the CKD stage, the
higher the expression of THBS4. THBS4 expression was elevated
in TGF-
The role of THBS4 in a variety of other diseases has been reported.
THBS4 is overexpressed in tissues and cells of patients with
hepatocellular carcinoma, where it regulates epithelial-mesenchymal processes and
interacts with the integrin family to regulate the FAK/PI3K/AKT pathway, which in
turn regulates hepatocarcinogenesis [19]. In cases of gastric cancer, there was a
positive association observed between elevated THBS4 expression and
higher pathological stage as well as unfavorable prognosis [20]. Overexpression
of THBS4 suppresses programmed cell death in prostate cancer cells by
activating the PI3K/AKT pathway and promotes tumor growth in vivo [21].
Keloid (KL) is a common skin tumor characterized by significant fibrosis and
inflammatory response. THBS4 was found to be significantly overexpressed
in KL [22]. It has been demonstrated that the expression of THBS4 is
elevated in fibroblasts found in hypertrophic scars, which are subject to a
fibrotic response dependent on TGF-
CKD is a chronic kidney disease, and the common pathological features are
tubulointerstitial fibrosis will allow glomerulosclerosis. With the progression
of renal fibrosis, patients with CKD experience a gradual decline in their kidney
function and ultimately progress to end-stage renal disease [25]. TGF-
A pharmacological investigation revealed that the primary molecular mechanism by
which Fufang Shenhua tablet (SHT) intervenes in CKD involves suppressing the
activity of PI3K/AKT [28]. Suppression of mir-214-3-p leads to a decrease in
renal interstitial fibrosis through modulation of the PTEN/PI3K/AKT signaling
pathway [29]. Aloe rhodopsin also attenuated renal fibrosis by inhibiting the
PI3K/AKT/mTOR pathway [30]. A prior investigation demonstrated that the
upregulation of THBS1, a different constituent within the THBS family,
triggered the activation of the PI3K/AKT signaling pathway, thereby exacerbating
renal fibrosis and promoting certain epithelial-mesenchymal transition processes
[31]. These studies have shown that renal fibrosis can be attenuated in CKD
patients by inhibiting the PI3K/AKT pathway. Our study has demonstrated that
reduced THBS4 expression would can inhibit the process of renal
fibrosis. Moreover, reduced THBS4 expression inhibited the
TGF-
As an important component of the extracellular matrix, THBS4 can influence the process of renal fibrosis by regulating the deposition and degradation of extracellular matrix (ECM) [32]. The results of this study suggest that THBS4 expression is up-regulated in renal fibrosis models, and inhibition of THBS4 gene expression can reduce the degree of renal fibrosis. This suggests that THBS4 can be a potential target for early diagnosis and treatment of CKD. And unlike traditional therapies that focus on controlling risk factors for disease progression, therapies targeting THBS4 are directly aimed at the core mechanisms of renal fibrosis and thus may be more specific and effective.
Overall, based on clinical samples, cellular experiments and animal models, we found that THBS4 expression was upregulated in CKD and positively correlated with CKD patient stage and negatively correlated with eGFR. Inhibition of THBS4 can inhibit the secretion of fibrosis-related proteins and PI3K/AKT pathway activation to alleviate renal fibrosis. These results may contribute to the study of the molecular mechanisms of CKD disease. Despite this, there are still some limitations in this study, such as the sample size included in the study is not large enough, the mechanism of THBS4 regulating the PI3K/AKT pathway is not detailed enough, and there is a lack of direct role of THBS4 in the treatment of CKD. These are the focus of our further research in the future.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
XY and YZ designed the research study. XY and YY performed the research. KZ provided help and advice on the experiments. WS and LW analyzed the data. XY drafted the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was approved by the Second Affiliated Hospital of Qiqihar Medical University Ethics Committee (ethics number: [2022]0815-8-2) and patients or their families/legal guardians in the study gave informed consent. This study strictly adheres to the ethical principles and guidelines of the Declaration of Helsinki. All animal experiments comply with the international ethical standards for experimental animals and the regulations designated by the Second Affiliated Hospital of Qiqihar Medical University Ethics Committee (ethics number: [2022]0815-8-2).
Not applicable.
This work was supported by the Health Commission of Heilongjiang Province Scientific Research Topics (grant numbers 2022030350618).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL26076.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





