1 Department of Internal Medicine, University of Genova, Viale Benedetto XV, 16132 Genova, Italy
2 Allergology and Clinical Immunology Unit, San Bartolomeo Hospital, 19038 Sarzana, Italy
3 Elderly and Disabeld Department, San Paolo Hospital, 17100 Savona, Italy
4 Department of Health Sciences (DISSAL), University of Genova, 16132 Genova, Italy
5 Hygiene Unit, San Martino Policlinico Hospital-IRCCS for Oncology and Neurosciences, 16132 Genova, Italy
6 Department of Clinical and Experimental Medicine, School and Operative Unit of Allergy and Clinical Immunology, University of Messina, 98125 Messina, Italy
Abstract
Type 2 immunity is represented by T helper 2 (Th2) lymphocytes and the cytokines produced downstream (Interleukin (IL)-4, IL-13, IL-31). They are increasingly recognized as pivotal mediators in the pathogenesis of immune-mediated dermatological conditions such as atopic dermatitis (AD) and psoriasis (Pso). In these disorders, they initiate and amplify immunological signaling cascades, promote cutaneous inflammation, and contribute to the induction of pruritus. In this context, IL-33 and IL-31 would be believed to be intrinsically linked and related to the acuity of the disease. The presence of an interleukin could in fact trigger the other, amplifying the inflammatory process of itchy skin disorders and therefore the extent of the symptoms. High levels of IL-31 may support the maintenance of a microenvironment that promotes both the growth and spread of solid tumors, as well as the development of cancer-associated pruritus. Given these premises, non-histaminergic mediators such as IL-31 and IL-33 could be explored as novel therapeutic targets for the treatment of pruritus in immune-mediated skin diseases and cancer, improving the QoL of patients. Finally, we briefly discussed the recent innovations in the field of monoclonal anti-IL-31 therapies.
Keywords
- IL-31
- IL-33
- IL-33/IL-31 axis
- IL-31 receptors
- cancer
Interleukin 31 (IL-31) belongs to the IL-6 superfamily, which includes among others IL-11, IL-21, IL-27, oncostatin M (OSM), leukemia inhibitory factor, and cardiotropin 1 [1]. This cytokine is mainly produced by type II helper (Th2) lymphocytes, monocytes/macrophages, mast cells, basophils, dendritic cells, fibroblasts and keratinocytes.
IL-31 plays a central role in innate and adaptive immune responses. The signaling
triggered by most of these cytokines is mediated through their interaction with a
novel cytokine receptor known as the gp130-like monocyte receptor or gp130-like
receptor (GPL), which is a member of the type I cytokine receptor family [2].
IL-31 receptor (IL-31R) consists of two subunits, the IL-31 receptor alpha
(IL-31RA) and OSM receptor
IL-31 controls the onset and maintenance of itching, mainly in atopic dermatitis (AD) and psoriasis (Pso) [4], favoring late onset of itching compared to histamine-induced itch [1, 4]. IL-31 through the phosphorylation of JAK1/2 and PI3K/AKT determines the activation of STAT3 and STAT5 inducing itching and Th-1-mediated skin keratinocyte cell proliferation, differentiation, apoptosis, and inflammation [1, 5]. Expression diversity is driven by microenvironment: autocrine expression of IL-4 activates Th1 cells to express IL-31 (Fig. 1). Various stimuli, including allergenic substances, infectious organisms, and UV radiation, can enhance the production of IL-31 [6].
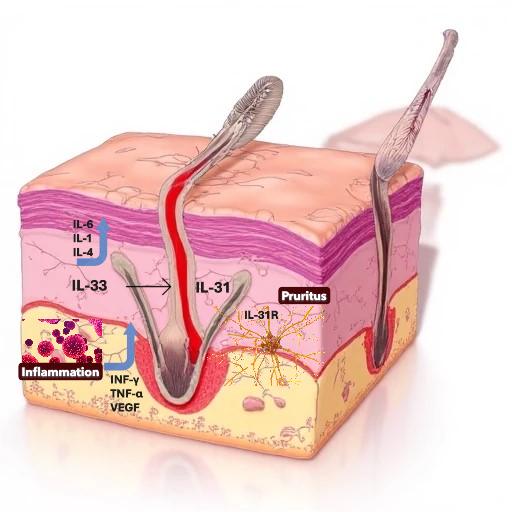 Fig. 1.
Fig. 1.
Role of the Interleukin (IL)-31/IL-33 axis in the pathogenesis of immune-mediated dermatological conditions and pruritus. Created with Microsoft Designer.
Murdaca et al. [4] proposed the emerging idea of an IL-31/IL-33
interaction network possibly playing a role in the progression of allergic
diseases, autoimmunity, and tumorigenesis. IL-33, a member of the “alarmins”
family, is a nuclear cytokine produced by endothelial cells, epithelial cells,
fibroblast-like cells, and myofibroblasts. It has been hypothesized that IL-33
may act both at an intracellular level as a nuclear factor regulating gene
expression and at an extracellular level as a cytokine of the IL-1 family [7]. IL-33
is composed of two distinct domains: an N-terminal nuclear domain and a
C-terminal IL-1-like cytokine domain, separated by a divergent central region.
The IL-1 homologous region of IL-33 allows it to interact with soluble interleukin 1 receptor-like 1 (ST2), a membrane
receptor classified within the Toll-like/IL-1 receptor group. This interaction
facilitates association with the IL-1 receptor accessory protein (IL-1RAcP),
which is shared among other IL-1 family members such as IL-1
Therefore, the involvement of the ‘IL-31/IL-33’ axis in allergic and autoimmune responses suggests a novel inflammatory mechanism, affirming IL-33’s role in initiating these responses [4, 9, 10, 11]. IL-31 activates a transcriptional program in sensory neurons that promotes neurite elongation and branching, thereby enhancing neuronal sensitivity to minimal stimuli and contributing to persistent pruritus [12].
Notably, itching is also a symptom of hematological tumorigenesis and cancers
[13, 14]. IL-31 is expressed differently across multiple tumor categories and has
the capacity to either promote or inhibit oncogenic advancement [15]. Akhtar
et al. [16] discuss the current knowledge on the role of IL-31 in the
pathogenesis of pruritus and in cancer onset and progression. They focus on the
key role of Th2 cytokine (IL-4, IL-13, IL-25, IL-31, IL-33 and thymic stromal
lymphopoietin) in mediating itch. In support of this theory, the authors point
out that the administration of dupilumab (an antibody against IL-4
On the other hand, itching is also considered a negative signal of the progression of the neoplastic disease. Ferretti et al. [19] confirmed first of all that the intense pruritus in patients with Hodgkin Lymphoma is caused by IL-31 released by tumor-infiltrating Th2 cells which is capable of supporting a microenvironment stimulating the growth and diffusion of Hodgkin cells expressing IL-31R. Moreover, elevated levels of IL-31 maintain a microenvironment also favorable to the growth and spread of solid tumors. Indeed, in lung cancer high levels of IL-31 favor disease progression and dermal rash, pruritus, and xeroderma occurring in patients treated with epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (erlotinib and gefitinib) [20]. Akhtar et al. [16] confirmed in breast, gastric, colon, endometrial and ovarian cancers the role of IL-31 in cancer progression and the appearance of itching.
Single-nucleotide polymorphisms (SNPs) of the IL-31 gene may represent important information for cancer progression and survival. Indeed, the SNP rs7977932 of the IL-31 gene is correlated with endometrial-cancer susceptibility and the SNP rs4758680 with bladder cancer [1].
The use of monoclonal antibodies targeting IL-31 may not only serve as an effective tool for controlling itching and improving quality of life, but also as a strategy to limit tumor progression by supporting the action of antineoplastic drugs. Among the most recent innovations in the field of monoclonal anti-IL-31 therapies, Nemolizumab and Vixarelimab are the two that have passed phase II studies [21, 22].
Nemolizumab is a subcutaneously administered humanized monoclonal antibody targeting the IL-31RA, developed by Chugai Pharmaceutical Co., Ltd. It is approved for the treatment of pruritus associated with atopic dermatitis (ADaP), prurigo nodularis (PN), chronic kidney disease-associated pruritus (CKDaP), and systemic sclerosis (SSc) [23, 24].
Vixarelimab is also a human monoclonal antibody that is currently being studied for the
treatment of PN alone. It acts at the level of the OSMR
It is plausible that further clinical studies including pharmacogenomics studies on SNPs will be able to confirm the role of the IL-31/IL-33 axis and pave the way for targeted therapies targeting IL-31 or IL-33.
GM: supervision, conceptualization, writing and editing; FP: conceptualization, writing and editing; AO, SG: designing the research study, supervision, writing and editing. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This research received no external funding.
Given his role as the Guest Editor and Editorial Board member, Giuseppe Murdaca had no involvement in the peer-review of this article and has no access to information regarding its peer review. Given her role as the Guest Editor, Francesca Paladin had no involvement in the peer-review of this article and has no access to information regarding its peer review. Given his role as the Guest Editor, Sebastiano Gangemi had no involvement in the peer-review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Graham Pawelec.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

