1 School of Pharmacy, Faculty of Medicine and Health, University of Sydney, Sydney, NSW 2006, Australia
2 Department of Internal Medicine I, University Hospital Regensburg, 93053 Regensburg, Germany
Abstract
Sepsis represents a life-threatening organ dysfunction due to a compromised host response caused by bacterial and viral infections. Although progress has been made to unravel the underlying pathophysiology, sepsis remains a very serious condition. Adiponectin is an adipokine with multiple beneficial activities relevant to glucose and lipid metabolism whose serum levels are low in obesity and metabolic diseases. In addition, several immunoregulatory activities of adiponectin have been described and circulating adiponectin levels have been positively associated with inflammation in autoimmune diseases such as rheumatoid arthritis and type 1 diabetes. Although an understanding of physiological consequences is still limited, blood adiponectin levels are also elevated in chronic kidney disease and liver cirrhosis, possibly due to impaired biliary and renal excretion. Natriuretic peptides, which increase adiponectin synthesis in adipocytes and are induced in autoimmune diseases and cirrhosis and decreased in obesity, appear to contribute to altered systemic adiponectin levels. In patients with sepsis, data on circulating adiponectin levels are not concordant, probably due to the large diversity in cohorts analyzed, often including patients with a higher risk for severe sepsis due to chronic metabolic disease or other comorbidities. This review article summarizes the main physiological activities of adiponectin and describes its role in inflammation and experimental sepsis. Adiponectin levels obtained in observational studies from serum or plasma of patients with critical illness are discussed. From this analysis, we conclude that circulating adiponectin levels are reduced in sepsis and septic shock, suggesting the potential of adiponectin receptor agonists as an option for sepsis therapy.
Keywords
- adiponectin
- bacterial infection
- inflammation
- SARS-CoV-2
- obesity
- sepsis
Sepsis is a very serious health condition characterized by organ dysfunction
that is predominantly (
Along these lines, the high blood levels of pro- as well as anti-inflammatory cytokines and chemokines commonly found in sepsis patients implicated their potential for diagnosis and therapy. However, early approaches to treat sepsis using agents targeting hyperinflammation failed and despite the success of anti-inflammatory therapies such as cytokine blockers for severe coronavirus disease 2019 (COVID-19), sepsis-specific treatments are currently lacking [4, 5, 6]. Yet, it has become clear that novel immunomodulatory sepsis therapies need to address hyperinflammation, but also the immunosuppressive phase of sepsis [4]. In addition, monitoring the very complex sepsis patient response and stratification of patients for disease severity and immune characteristics may become an option for appropriate and patient-specific therapies [4]. The development and diagnostic value of specific biomarkers is highlighted by several examples. For instance, despite the heterogeneity of COVID-19 symptoms [7], SARS-CoV-2 induces an innate immune reaction involving neutrophils, monocytes and macrophages, which subsequently prime an adaptive immune response in lymphocytes. Infection is cleared in almost 80% of cases but in patients developing sepsis upon SARS-CoV-2 infection, an exaggerated production of inflammatory cytokines such as interleukin-6 (IL-6) contributes to tissue injury and organ failure [8]. Here the identification of molecules that can help predict disease outcome and benefit the evaluation of disease pathophysiology could classify novel drug targets. In the case of infections with methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci, other challenges in sepsis therapy become apparent. As the identification and characterization of bacteria responsible for the infection commonly takes several days, antibiotics are initially given by chance. Hence, biomarkers for early detection of the bacterial strain causing the infection or the identification of antimicrobial resistances that can trigger sepsis are of utmost importance but have yet to be established [9, 10].
We have recently addressed the relationship between adiponectin, a multifunctional adipokine almost exclusively produced by adipocytes, in patient cohorts of severe diseases, identifying opportunities for the prognosis and management of sepsis [11, 12]. In this review, we summarize the multiple pro- and anti-inflammatory activities of adiponectin and their effects in sepsis models and aim to provide an association of adiponectin levels with sepsis severity and outcome.
Adiponectin is produced by adipocytes from white and brown adipose tissues [13, 14]. Circulating levels of adiponectin are much higher than other common adipocyte-derived adipokines such as leptin and exist as low molecular weight (LMW) trimer, medium molecular weight (MMW) hexamer and high molecular weight (HMW) multimer complexes [15, 16, 17]. Among the circulating adiponectin isomers, LMW and HMW make up the majority of adiponectin, while MMW hexamers are found in lower concentrations [15, 16, 17]. In addition, globular adiponectin, a proteolytic cleavage product of adiponectin (17 kDa), may circulate at very low levels in the blood [18, 19].
Adiponectin conveys its diverse biological activities through the binding to the
Adiponectin receptor 1 (AdipoR1) and AdipoR2 at the surface of target cells,
leading to the activation of different signaling pathways [20, 21]. Downstream of
AdipoR1 and AdipoR2, the signaling pathways regulated by adiponectin still have
to be fully resolved, but adiponectin binding to AdipoR1 activates adenosine
monophosphate-activated protein kinase, the fuel-sensing enzyme conserved in all
mammalian cells. Further benefiting energy homeostasis, AdipoR2 activation
increases the activity of the transcription factor peroxisome-proliferator
activated receptor (PPAR)
Given the strong association of alterations in adipokine secretion with metabolic disease, a substantial number of studies have investigated the influence of gender, age, body mass, metabolic and other chronic diseases and their pharmacological treatments with plasma levels of adipokines, including adiponectin. Along these lines, females were found to exhibit higher circulating adiponectin levels than males. Furthermore, females displayed a higher proportion and absolute amount of HMW adiponectin, and demonstrated a robust inverse correlation between body fat measures and the proportion of HMW species [26].
As listed below, there is a common trend that systemic adiponectin levels are reduced in obesity, metabolic disease and cardiovascular disease [24, 27, 28]. Patients with type 2 diabetes, hypertension and non-alcoholic fatty liver disease also revealed low blood adiponectin levels [21, 24, 27, 29, 30]. These reduced systemic adiponectin levels [24, 27, 28] are generally associated with low-grade systemic inflammation and elevated C-reactive protein (CRP) levels, the most commonly used clinical inflammation marker [31], implicating an interconnection of inflammation and adipokine production, secretion and plasma levels. In support of this, pro-inflammatory cytokines such as tumour necrosis factor (TNF) suppressed adiponectin synthesis in adipocytes to contribute to low circulating adiponectin levels [32]. Vice versa, low serum adiponectin concentrations in turn exacerbated metabolic dysfunction and inflammation, suggesting a positive feedback loop that enables adiponectin to self-reinforce hypoadiponectinemia [33].
Based on the various protective activities of adiponectin in metabolic and cardiovascular complications, drugs that selectively activate adiponectin receptor signaling have been developed. This includes AdipoRon, an orally active agonist for AdipoR1 and AdipoR2 that protects from diabetes, kidney injury and cardiac hypertrophy in several animal models [34]. In a murine model of systemic sclerosis, AdipoRon acted in an anti-inflammatory and anti-fibrotic manner in three fundamental pathomechanisms underlying the clinical manifestations of systemic sclerosis, (i) the activation of fibroblasts, (ii) the transdifferentiation of adipocytes to myofibroblasts, and (iii) T-helper cell polarisation to the Th2/Th17 type [35]. In spinal cord injury and neuroinflammation of mice, enhanced adenosine triphosphate-binding cassette transporter A1-dependent lipid efflux mediated the anti-inflammatory activity of AdipoRon [36].
These multiple beneficial metabolic effects of adiponectin listed above may also contribute to protecting against inflammatory-driven oncogenic malignancies. However, these discoveries may not apply to liver cancers as serum adiponectin concentrations of patients with hepatocellular carcinoma (HCC) were increased [37]. Notably, with circulating adiponectin levels being elevated in severe liver dysfunction and cholestasis, this suggests that higher adiponectin amounts in HCC are caused by an underlying liver disease. The source of higher adiponectin concentrations in liver disease is still unclear and as abundant amounts of adiponectin in urine and bile implicate renal as well as biliary excretion [38], an impaired hepatic uptake and biliary excretion of adiponectin may contribute to elevated levels in HCC.
Circulating adiponectin levels were also elevated in chronic kidney disease [39], leading to studies assessing the diagnostic value of urinary adiponectin in patients with renal dysfunction. Urinary adiponectin amounts were elevated in diabetic patients compared to normal weight and obese controls and may be used as a biomarker to assess the risk for diabetic nephropathy [40].
Several other studies detected high systemic adiponectin levels in patients with autoimmune diseases such as type 1 diabetes and rheumatoid arthritis [41, 42] (Fig. 1). Monoclonal antibodies against MMW adiponectin showed therapeutic potential and improved symptoms in a collagen-induced arthritis mouse model, indicating that specific adiponectin isoforms mediate deleterious effects in this autoimmune disease [43]. Notably, total and HMW adiponectin levels were positively related to all-cause mortality across several clinical studies and only a few studies reported an inverse relationship [44, 45]. Brain natriuretic peptides (BNP) and atrial NP (ANP) increased adiponectin expression and secretion of primary human adipocytes [46] and the association of adiponectin with mortality rate was confounded after considering these paracrine/hormone factors [45]. NP hormones derived from the heart serve a variety of purposes and are indicators of vascular remodeling following ventricular or atrial wall stretching. Additionally, some evidence supports inflammation to increase NP production [47]. While serum NP levels are elevated in liver cirrhosis [48], chronic kidney disease [49] and autoimmune diseases [47, 50], reduced NP levels have been linked to obesity [51, 52], non-alcoholic fatty liver disease [48], and insulin resistance [53] (Fig. 1). Therefore, it is reasonable to assume that NP plays a critical role in the regulation of serum adiponectin levels.
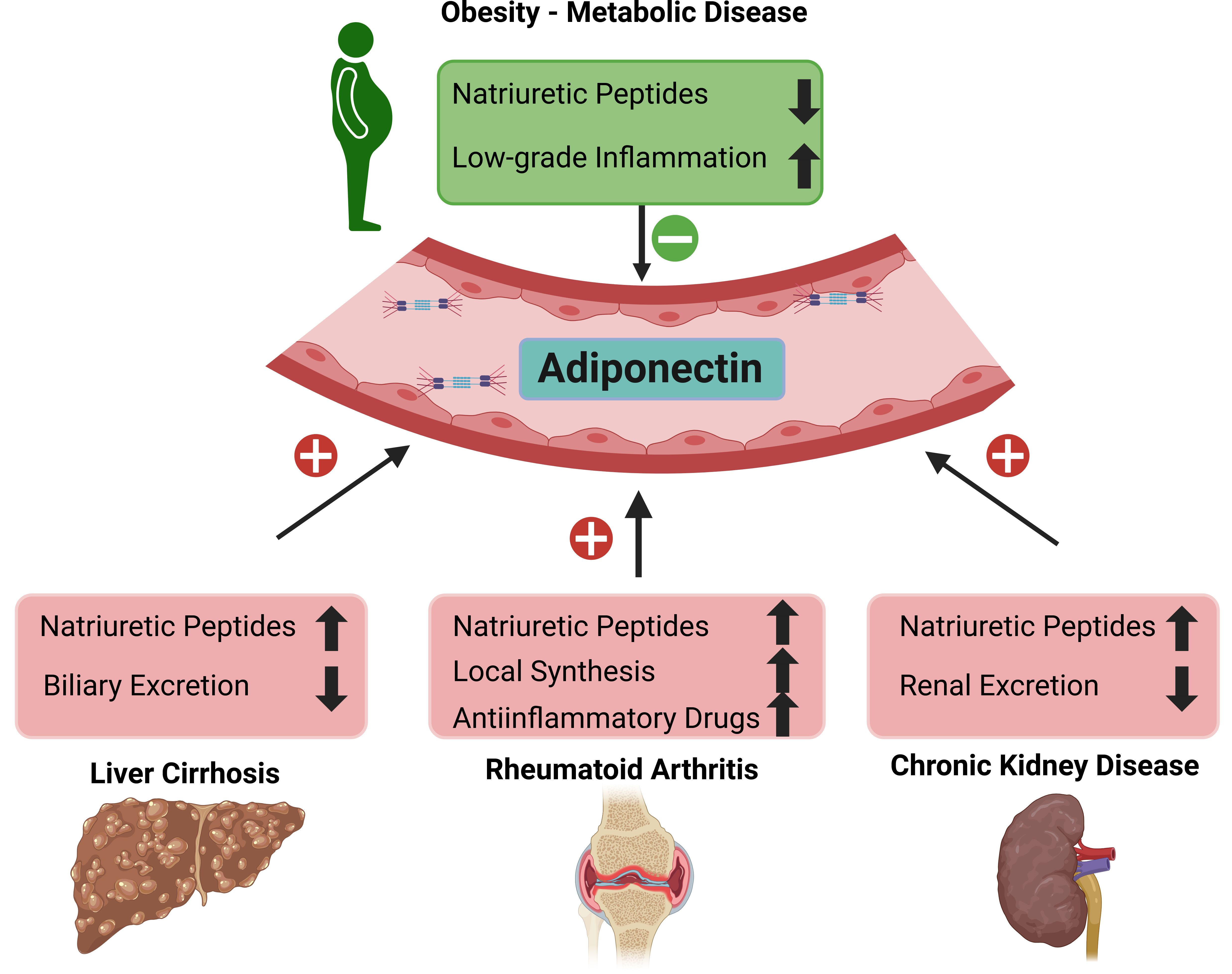 Fig. 1.
Fig. 1.
Systemic adiponectin levels in relation to different diseases.
Liver cirrhosis, chronic kidney and autoimmune diseases such as rheumatoid
arthritis are related to high systemic adiponectin levels. In contrast, obesity
and associated metabolic diseases are characterized by low amounts of systemic
adiponectin levels. Factors which are increased in the respective diseases
Other causes of high serum adiponectin levels in autoimmune diseases include drugs such as anti-IL receptor antibodies or glucocorticoids, which are associated with increased adiponectin levels [54] (Fig. 1). In rheumatoid arthritis patients, elevated serum levels of adiponectin are a result of higher local adiponectin synthesis in articular adipocytes and synovial membrane explants [54] (Fig. 1).
Taken together, efforts to utilize circulating adiponectin levels as a diagnostic and/or prognostic tool in sepsis has to consider various confounding factors such as gender and metabolic diseases as well as medications and underlying diseases.
The majority of studies investigating pro- and anti-inflammatory effects of
adiponectin focussed on the analysis of monocytes/macrophages or myeloid cell
lines, such as THP-1, U937 or RAW264.7. Full-length and globular adiponectin were
shown to induce IL-10 expression in lipopolysaccharide (LPS)-activated Kupffer
cells and reduce their pro-inflammatory response [55]. Incubation of THP-1 foam
cells for 24 h with human adiponectin derived from HEK293 cells suppressed TNF
and CC chemokine ligand 2 (CCL2) mRNA expression [56]. Adiponectin preincubation
for 24 h, to mimic the mode of action of circulating adiponectin prior to an
inflammatory response, lowered the pro-inflammatory effect of LPS in porcine
macrophages, and was accompanied by IL-6 downregulation, while IL-10 expression
increased [57]. LMW adiponectin produced in insect cells also exerted
anti-inflammatory activities in LPS-incubated THP-1 cells [58]. HMW adiponectin
preincubation conveyed potent anti-inflammatory activities in macrophages and
suppressed IL-1
Further evidence for the anti-inflammatory roles of globular adiponectin, which circulates only in lower amounts compared to the other three adiponectin isoforms [18, 19], for macrophages exist. Globular adiponectin was found to reduce production of inflammatory cytokines [41] and inhibited LPS-induced inflammasome activation in macrophages by induction of autophagy [62]. Globular adiponectin also reduced TNF expression and induced IL-10 expression in rat Kupffer cells exposed to LPS [55]. Furthermore, globular adiponectin impaired C-reactive protein (CRP) synthesis in human aortic endothelial cells and rodent hepatocytes [63], consistent with an inverse correlation between systemic adiponectin and CRP levels in patients with coronary artery disease [64].
M1 and M2 polarization states of macrophages critically influence pro- and anti-inflammatory conditions. While M1 macrophages are mostly responsible for pro-inflammatory responses, M2 macrophages are generally considered to facilitate anti-inflammatory activities. These differential pro- and anti-inflammatory responses of M1 and M2 macrophages are also reflected in their response to adiponectin exposure. For instance, after the classical generation of activated M1 macrophages upon incubation with LPS and interferon-gamma, these cells showed a pro-inflammatory response when challenged with full-length adiponectin. On the other hand, M2 macrophages obtained by IL-4 or IL-10 stimulation produced higher levels of IL-10 when incubated with adiponectin [61].
Yet, despite the anti-inflammatory responses triggered by globular adiponectin described above, others reported activation of the transcription factor nuclear-factor kappa B (NF-kappaB) by globular adiponectin in myeloid cell lines [65, 66]. Globular adiponectin also increased N-formylmethionyl-leucyl-phenylalanine-induced production of reactive oxygen species by human phagocytes [18] and TNF and IL-6 expression in macrophages and THP-1 cells [67]. Interestingly, in the latter studies, pre-incubation of THP-1 cells with globular adiponectin inhibited its ability to promote TNF or IL-6 secretion upon re-stimulation. It was proposed that chronic abundance of globular adiponectin may desensitize monocytes and macrophages for further activation by adiponectin (Fig. 2). Markedly, pre-incubation with globular adiponectin rendered THP-1 cells also incapable of mediating the pro-inflammatory effects of LPS [67].
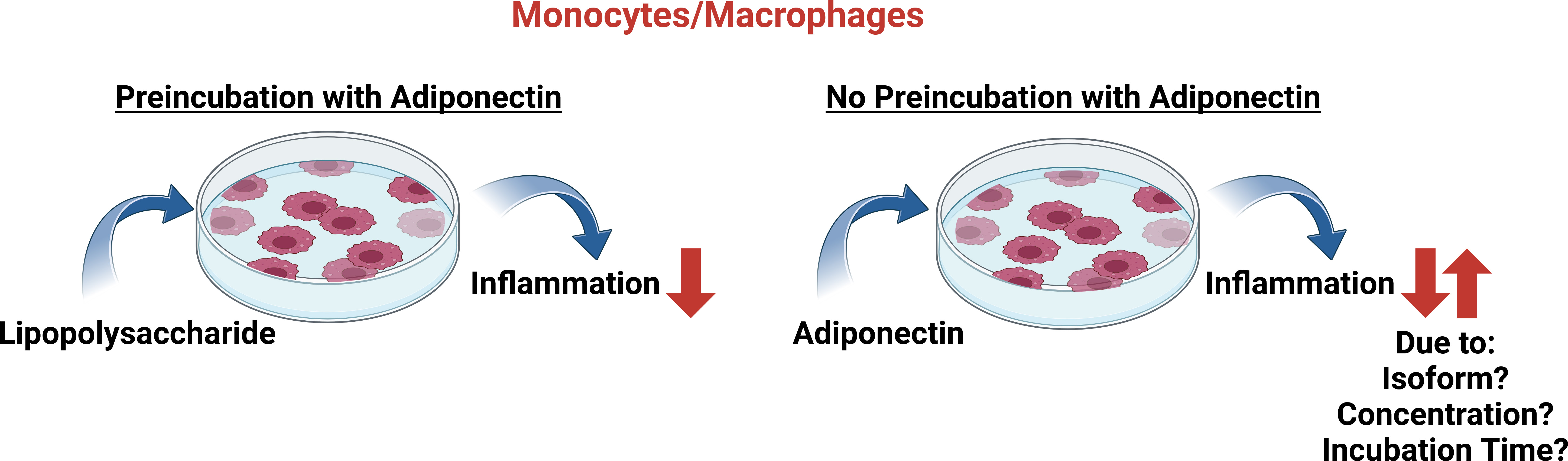 Fig. 2.
Fig. 2.
In vitro effects of adiponectin exposure on the inflammatory response of monocytes/macrophages. Preincubation of monocytes/macrophages with adiponectin lowers the pro-inflammatory effects of lipopolysaccharide treatment. On the other hand, the cultivation of cells with adiponectin in the absence of lipopolysaccharide is capable of conferring pro- as well as anti-inflammatory responses. It should be noted that different experimental conditions (e.g., incubation times, activation procedures, adiponectin concentrations and isoform utilization) and cell models used in the various studies also need to be considered (see text for details). Created in https://BioRender.com.
In line with these observations, in the obese, low adiponectin levels may prime macrophages to an exacerbated response to inflammatory metabolites [24, 33]. It should be noted that the studies listed above [65, 66, 67] used 5–10 µg/mL globular adiponectin in their experimental setup. Although these amounts are equivalent to total adiponectin levels found in the circulation, these concentrations are much higher than physiological concentrations of globular adiponectin [18, 19]. However, as macrophages express elastase, which can cleave full-length adiponectin, the levels of globular adiponectin may indeed be a lot higher in the microenvironment of these cells compared to values obtained for globular adiponectin amounts in the circulation [68]. Future research will have to address the potentially different systemic and local adiponectin levels and their impact on individual cells in tissue microenvironments.
In addition to globular adiponectin (see above), HMW adiponectin has also been reported to modulate inflammatory activities via NF-kappaB activation. HMW adiponectin was found to increase NF-kappaB activity as well as CCL2, CCL20, CXCL8 and IL-6 levels in monocytes/macrophages. Preincubation with native adiponectin (forming HMW, MMW and LMW complexes) reduced the activation of NF-kappaB by TNF and LPS [42, 66]. In non-stimulated primary monocytes, overnight exposure with HMW, but not LMW adiponectin induced IL-6 expression [58]. On the other hand, adiponectin unable to form HMW isoforms failed to exert pro-inflammatory effects in U937 monocytes. Yet, in this study, globular adiponectin increased NF-kappaB activity, which correlated with increased mRNA and protein levels of inflammation marker genes [66].
From the studies on the role of adiponectin in macrophage activation and production of inflammatory cytokines and chemokines, it became apparent that it remained challenging to unravel opposite outcomes of adiponectin in inflammation, often possibly compounded by the different experimental models and conditions used (e.g., globular vs. full-length adiponectin; incubation periods) (Fig. 2). Overall, current models favour adiponectin-related effects to depend on the cellular activation status, the distinct signaling potential of the different adiponectin isoforms, their local concentrations and the receptor repertoire being expressed by the various cell types [69, 70]. The time course of disease progression also needs to be considered (Fig. 2). For instance, in human endotoxemia, the initial increase in pro-inflammatory cytokines such as TNF was followed by the release of IL-6 and subsequently anti-inflammatory cytokines such as IL-10 [71]. Also, incubation periods chosen to expose cells with LPS as well as adiponectin may make a difference in experimental outcomes. Yet, despite the difficulties in aligning experimental settings, there is consensus that at least in macrophages, preincubation with adiponectin results in the downregulation of inflammatory cytokine production when cells are subsequently exposed to LPS (Fig. 2).
While numerous animal studies analysed the role of adiponectin in metabolic complications, data on its effect in animal models for infectious diseases and sepsis are rare [25, 30, 72, 73].
Obese mice having low blood adiponectin levels as well as adiponectin null mice had an impaired clearance of Listeria monocytogenes, which was restored by supplementation with adiponectin [74]. Infection of murine adipocytes with Listeria monocytogenes causes a transient increase of CCL2 and this was followed by higher expression of adiponectin. Adiponectin deficiency abrogated infection-mediated induction of CCL2, which is essential for macrophage recruitment and clearance of microbiota [75]. As Listeria monocytogenes is a pathogenic Gram-positive bacterium causing listeriosis that may result in septicemia [76], this could indicate high adiponectin levels to protect from the development of critical illness triggered by this bacterium. This hypothesis of adiponectin supplementation to act in an anti-inflammatory manner is supported by other findings. For instance, in a murine sepsis model caused by cecal ligation and puncture, repeated dosing of globular adiponectin (produced in Escherichia coli) prior to the sepsis-inducing procedure, normalized leukocyte/platelet adhesion in the cerebral microcirculation of normal diet-fed and diet-induced obese mice [24]. Also, in a rat model of abdominal sepsis, adiponectin treatment lowered the serum levels of TNF, soluble intercellular adhesion molecule 1, IL-6, and macrophage inhibitory factor, while IL-10 levels were elevated. Although the adiponectin isoform used for these experiments was not provided, these findings correlated with better survival compared to controls [77]. In line with these observations, adiponectin protected from severe lung injury in a rat sepsis model. In bronchoalveolar lavage neutrophil infiltration, levels of inflammatory cytokines and myeloperoxidase activity improved upon adiponectin therapy [78]. A substantial number of control animals, but not adiponectin-deficient mice survived cecal ligation and puncture. This was associated with a more severe renal and hepatic injury with higher infiltration of leukocytes in the adiponectin null animals [79].
Besides these findings from adiponectin supplementation studies and animal models lacking adiponectin, the determination of adiponectin levels supports a protective role for adiponectin in sepsis and inflammation. In an endotoxemia model and in cecal ligation and puncture, plasma adiponectin concentrations and its expression in epididymal fat were decreased [79]. Notably, these findings might not be specific to the induction of sepsis, as similar results were obtained from sham-operated animals [79]. Reduced adiponectin mRNA expression in epididymal, perirenal and subcutaneous fat of mice challenged with LPS was also reported [80]. LPS injection to mimic murine sepsis caused a downregulation of AdiopR1 expression in muscle and perirenal adipose tissue while AdipoR2 levels were reduced in the liver, perirenal fat and subcutaneous fat. Interestingly, AdipoR2 expression levels normalized within 24 h in all of these tissues. AdipoR1 mRNA levels increased in muscle tissue and returned towards normal in perirenal fat, indicating an immediate transient response to LPS exposure, followed by normalization of mRNA expression [81]. Furthermore, AdipoR1 surface levels of lung leukocytes were also reduced when animals were challenged with chitin, yet lung inflammation nevertheless improved by exogenous adiponectin [82]. To date, a detailed knowledge on AdipoR1 and AdipoR2 expression patterns in different cells and tissues in human sepsis is still lacking. It is important to note that the levels of adiponectin receptors may be low in sepsis, which presents a challenge in the development of adiponectin receptor agonists for the treatment of sepsis.
Several studies addressed the therapeutic potential of adiponectin receptor agonists in animal and cell models for sepsis [83, 84]. Notably, the 10-amino acid peptide analogue of adiponectin, ALY688, effectively reduced IL-6, CCL2, type 1 interferons and CXCL10 levels in a murine sublethal LPS endotoxemia model. ALY688 was found to increase transforming growth factor-beta expression in mice with and without LPS challenge [83], yet did not alter inflammatory cytokine and chemokine levels in control animals. An increase of circulating T-cell number was noticed in controls while T-cells decreased in endotoxemic mice. Neutrophil and monocyte counts were not modified by ALY688 treatment [83]. In addition, the globular adiponectin-derived peptide KS23 improved endotoxin-induced uveitis in Lewis rats. KS23 also decreased TNF and IL-6 levels in LPS-stimulated RAW264.7 cells by blocking nuclear translocation of NF-kappaB [84].
Taken together, all animal studies have reported protective effects of adiponectin and adiponectin receptor agonists in models of inflammation and sepsis. However, several other drugs that have shown anti-inflammatory effects in rodents have not been successful in patients with sepsis [85]. These observations could point at additional, yet to be identified beneficial activities mediated by adiponectin that other anti-inflammatories are lacking. It remains to be seen whether adiponectin-related approaches will be more successful.
In addition to the various animal models and patient cohorts described in the previous sections, several studies determined serum/plasma adiponectin levels in patients with sepsis, yet results varied depending on the cohort analyzed [86]. Most reports were based on a limited number of sepsis patients and as outlined earlier (section 2.2), sepsis pathophysiology differs between genders, with male patients having a higher risk for critical illness and fatal outcomes [87, 88]. Moreover, serum adiponectin levels of females are higher in comparison to males [25, 69, 73], but most observational studies did not account for gender differences. One gender-specific report found significantly lower plasma adiponectin levels only in female, but not male patients with systemic inflammatory response syndrome (SIRS)/sepsis compared to female controls. Infection of sepsis patients with either Gram-negative or Gram-positive bacteria exhibited negligible impacts on plasma adiponectin levels in both genders (Fig. 3) [12].
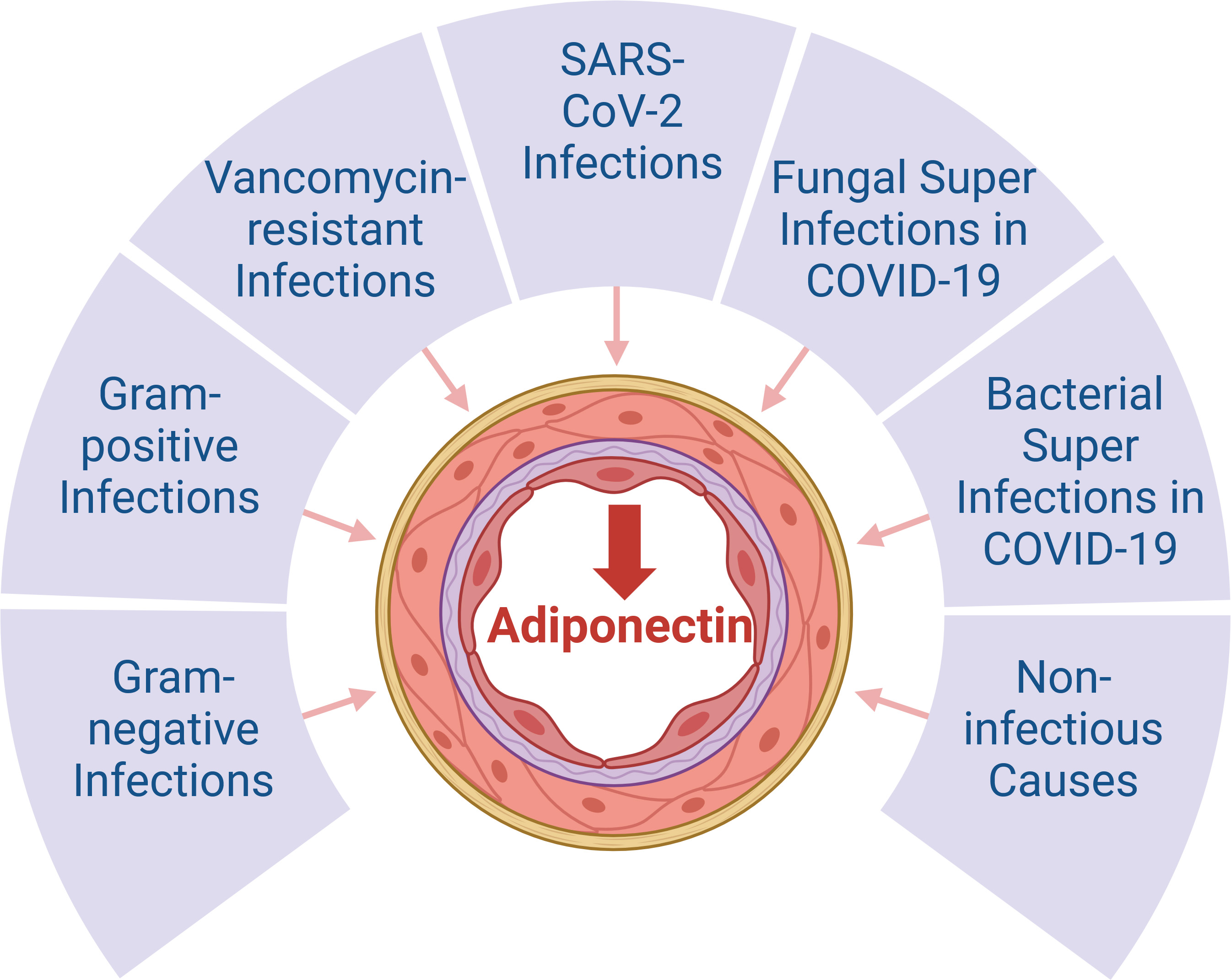 Fig. 3.
Fig. 3.
Systemic adiponectin levels in patients with sepsis. There is
consensus that systemic adiponectin concentrations are low in patients with
sepsis (
Differential outcomes from several other studies make an overall interpretation
challenging. For instance, 41 mechanically ventilated patients with sepsis showed
significantly higher adiponectin levels compared to 40 healthy controls (10.9
SARS-CoV-2 infection is a more recent cause of sepsis [98, 99] and most studies observed lower serum adiponectin levels in COVID-19 patients compared to healthy controls, which further declined with disease severity [11, 100, 101, 102, 103] (Fig. 3). Yet, others reported similar adiponectin amounts in COVID-19 patients and controls [104, 105, 106]. These opposing findings might in part be explained by the diversity of COVID-19 severity and associated complications, which may impact on adiponectin levels. In particular, the infection of adipocytes and adipose-tissue-localised macrophages by SARS-CoV-2 induces inflammation in adipose tissues, which is associated with a reduction in adiponectin synthesis [107, 108] (Fig. 3). While this implicates low serum adiponectin levels in patients infected with SARS-CoV-2, Spirina et al. [109] found higher adiponectin levels in 92 COVID patients experiencing pneumonia. Adiponectin levels were not elevated any further in severe cases developing acute respiratory distress syndrome. Diabetes and arterial hypertension were also not associated with lower adiponectin levels in this cohort. Conversely, infection with SARS-CoV-2 resulted in a reduction of adiponectin levels solely in females [12].
The diversity in symptoms associated with COVID-19 differentially affecting plasma adiponectin levels is further reflected in SARS-CoV-2 infected patients with intestinal lesions, which showed strongly elevated adiponectin levels. On the other hand, this was not observed in SARS-CoV-2 patients with hepatitis [109].
Notably, glucocorticoid therapy was associated with lower adiponectin levels [109]. This is in accordance with downregulation of adiponectin in fat tissues of mice after glucocorticoid administration [110, 111]. In patients with rheumatoid arthritis glucocorticoids were related to increased adiponectin levels [54] and the association of this medication with systemic adiponectin levels in sepsis needs further study. Overall, the diverse outcomes of adiponectin levels in SARS-CoV-2 infected patients, often influenced by a variety of comorbidities, indicate that COVID-19 sepsis is not generally associated with grossly altered serum adiponectin levels in comparison to sepsis because of other causes.
Moreover, patients with COVID-19 pneumonia and patients with bacterial community-acquired pneumonia, who were comparably ill, had similar adiponectin levels [112] (Fig. 3). These findings are in accordance with observations reported by others [100, 113]. Nevertheless, it is pertinent to highlight that patients with COVID-19-related respiratory failure exhibited significantly lower plasma adiponectin levels compared to patients with respiratory failure unrelated to SARS-CoV-2 [101]. Thus, circulating adiponectin levels generally appear to decrease with severe disease, regardless of SARS-CoV-2 infection [98] (Fig. 3). However, adiponectin levels of patients with severe sepsis were not associated with a higher risk for acute respiratory distress syndrome [114].
Superinfection with bacteria occurs in approximately 25% of COVID-19 patients [115, 116]. In non-hospitalized and hospitalized patients with COVID-19, 20% of cases had bacterial superinfections [117]. The meta-analysis revealed fungal and viral superinfections to occur in 8% and 4% of COVID-19 patients, respectively [117]. Superinfections were more prevalent in patients with severe COVID-19 and associated with a more severe clinical course and poorer outcomes [117, 118, 119]. Despite bacterial infections generally worsening outcomes, the presence of bacterial superinfection did not result in a change in serum adiponectin levels with severe COVID-19 (Fig. 3). Conversely, in vancomycin-resistant enterococci bacteremia, a significant further reduction in serum adiponectin levels was observed [11]. Yet, these findings were obtained from a rather small cohort and need further confirmation (Fig. 3). Since the initial isolation of vancomycin-resistant enterococci in England in 1988, these bacteria have disseminated at an alarming rate, becoming the predominant cause of nosocomial infections worldwide. Over time, enterococci have acquired genes that render them resistant to a multitude of antibiotics, including vancomycin [120, 121]. Low plasma adiponectin levels were also proposed to predict infections in patients with gastric surgeries [122]. The association of serum adiponectin concentrations with bacterial, viral and fungal infections is currently understudied, and further analysis is required.
The association of serum adiponectin levels and mortality in severely ill patients is a further unresolved issue. The adverse outcome was not related to altered adiponectin levels in two studies [11, 109]. On the other hand, an association between higher plasma adiponectin concentrations and mortality was observed in patients with acute respiratory failure [123] and in patients with sepsis at the time of enrollment and at day 7 [90]. Another study found an association between low adiponectin levels in sepsis and mortality [124]. In a cohort of patients admitted to the intensive care unit, low adiponectin levels were found to predict overall survival [93]. Additionally, a gender-specific analysis identified a trend towards higher plasma adiponectin amounts in male non-survivors [12]. The relationship between serum adiponectin levels and survival is partly influenced by the inclusion of severely ill and less severely ill patients, with the latter cohort having higher adiponectin levels and better survival. However, most studies did not report a significant correlation between adiponectin levels and mortality [11, 100, 102].
The majority of studies have employed a single measurement of serum adiponectin levels at the time of hospital admission. Hillenbrand et al. [97] conducted a longitudinal analysis of adiponectin levels during the hospital stay, demonstrating a slight increase in adiponectin levels in non-survivors. Notably, in this cohort, the body mass index of survivors was 30 kg/m2, which was higher than the BMI of non-survivors (25 kg/m2) [97]. A high BMI is a risk factor for severe COVID-19 and sepsis, but was also supposed to be protective in critical illness [125]. The fluctuations of adiponectin levels during sepsis may indicate a compensatory anti-inflammatory mechanism that counteracts the initial systemic inflammation, thereby facilitating the resolution of sepsis and promoting recovery. Conversely, an early pronounced response of adiponectin followed by a failure to sustain further increases could be indicative of a poor prognosis [126].
Sepsis is a state of an uncontrolled immune response [127, 128]. Adiponectin has been shown to reduce inflammation in activated monocytes, increasing anti-inflammatory pathways and blocking pro-inflammatory effects [57, 65, 129]. In addition, adiponectin improves metabolic health, which may contribute to a better outcome [130]. Insulin response, hepatic steatosis and fibrosis are associated with low adiponectin levels [69]. A recent review summarised the increasing evidence pointing at multiple beneficial anti-inflammatory effects of adiponectin in the lung. Several lung cell types, including immune, epithelial and endothelial cells, express adiponectin receptors. While both pro-inflammatory and anti-inflammatory properties have been documented, the anti-inflammatory role of adiponectin has been predominantly noted in lung cells [131]. However, further analysis is required to define the optimal timing of anti-inflammatory therapy in sepsis, as early blockade of inflammation may exacerbate disease severity [132]. Given the possibility that adiponectin receptors could be abundant in immune cells as well as peripheral cells such as pancreatic beta cells and hepatocytes, an adiponectin receptor agonist may improve sepsis severity and outcome.
The current data indicate that systemic adiponectin levels are reduced in patients with sepsis. Experimental models have demonstrated that adiponectin exerts a protective effect against sepsis, and preliminary data suggest a protective role in Gram-positive bacterial infection [70, 73, 75, 79]. The in vitro effects of adiponectin on monocytes/macrophages include both pro- and anti-inflammatory effects and are best explained by a model proposing that adiponectin activates these cells, rendering them resistant to further inflammatory stimuli. Another question to be addressed in future studies is the underlying mechanisms causing low adiponectin levels in sepsis. Inflammatory cytokines induced by viral and bacterial infections in sepsis cause reduced adiponectin synthesis in fat tissue. In addition, SARS-CoV-2 can infect adipocytes, thereby also lowering adiponectin expression (Fig. 4). NP levels in sepsis are commonly high [133] and should contribute to an increase of adiponectin production (Fig. 4). However, BNP resistance due to the lowering of BNP receptors and their concomitant desensitization has been described in heart failure [134, 135]. BNP levels in sepsis are more than 8-fold higher than in controls [136, 137] and increase about 5-fold in patients with liver cirrhosis [48], but whether BNP resistance specifically emerges in sepsis is unknown. Glucocorticoids such as prednisolone and dexamethasone are common therapies for these severely ill patients [138], which may also decrease serum adiponectin amounts (Fig. 4). In conclusion, the effects of adiponectin in sepsis and the pathways that contribute to low adiponectin levels in severe disease are incompletely understood. Current data suggest that adiponectin receptor agonists may have multiple beneficial effects in critically ill patients, but clinical trials have yet to be initiated.
 Fig. 4.
Fig. 4.
Association of adiponectin and inflammation in sepsis. Infection with SARS-CoV-2 or bacteria increases systemic inflammation, and inflammatory cytokines lower adiponectin production of adipocytes. SARS-CoV-2 can infect adipocytes, which also contributes to reduce adiponectin production. It is yet unclear if high levels of natriuretic peptide increase adiponectin amounts in sepsis. Glucocorticoid therapy is likely to lower adiponectin levels, but the improvement in inflammation with this approach may also contribute to an increase in adiponectin levels. Low adiponectin in sepsis may affect the function of immune cells in the blood and peripheral tissues. Created in https://BioRender.com.
CB and TG performed literature search and wrote the first draft of this review article. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




