1 Department of Gastroenterology, Yichang Central People’s Hospital, The First College of Clinical Medical Science, China Three Gorges University, 443002 Yichang, Hubei, China
2 College of Traditional Chinese Medicine, Anhui University of Chinese Medicine, 230012 Hefei, Anhui, China
†These authors contributed equally.
Abstract
Autoimmune hepatitis (AIH) is a chronic immune-mediated inflammatory liver disease characterized by recurring immune-triggered hepatic injury. While scientists have yet to fully elucidate the precise triggers of AIH, contemporary research indicates that both gut microbiota and their metabolic products significantly influence AIH progression. These factors contribute to multiple mechanisms, including compromised intestinal barrier function, altered microbial and metabolite trafficking, and disrupted immune balance, leading to inflammatory responses. This review begins by exploring the intestinal microbial populations and their byproducts linked to AIH. It highlights how disrupted gut flora compromises intestinal immune defenses, enables bacterial migration from the gut to hepatic tissue, and induces liver inflammatory responses. Research validates that metabolic products from microbes, such as short-chain fatty acids (SCFAs), bile acids (BAs), and specific amino acids (glutamine, cysteine, tryptophan, and branched-chain variants, among others), interact with immune cell populations. These interactions, coupled with immune cell modifications, contribute to AIH progression. Our review identifies promising treatment strategies, including the use of probiotic supplementation, engineered prebiotic compounds, microbiota transfer procedures, and specific medications targeting gut microorganisms and their byproducts. These approaches could potentially reduce immune-triggered hepatic damage, offering potential new avenues for AIH management.
Keywords
- autoimmune hepatitis
- gut microbiome
- microbial metabolites transport
- gut-liver axis
- intestinal immune barrier
- immune-mediated liver injury
Autoimmune hepatitis (AIH) is a chronic immune-mediated inflammatory liver disease characterized by positive autoantibodies, elevated serum aminotransferase levels, and immunoglobulin G (IgG)/hypergammaglobulinemia [1]. Global pooled incidence and prevalence of AIH are estimated to be 1.28 and 15.65 cases per 100,000 inhabitants, respectively [2]. Moreover, AIH is more prevalent in females, adults aged over 65 years, North American populations, and high latitudes, with an increasing trend over time [2]. The etiology of AIH remains unclear and may involve genetic elements, environmental influences, and autoimmune responses [3, 4]. Most AIH patients show mild or even no symptoms and may develop cirrhosis or even liver failure in the later stages [5]. At present, medical treatment of AIH mainly involves two authorized medications: corticosteroids and azathioprine. Patients who are nonresponsive to initial treatment may develop subsequent liver complications or even die [6].
The human gut microbiota, which refers to a variety of microorganisms residing in the host intestinal tract, plays a crucial role in AIH development [7]. The main species of microbiota in the human gut include Firmicutes, Bacteroidetes, fungi, viruses, and phages [8]. Gut microbiota can influence liver disease progression through interactions along the gut-liver axis [9]. Increasing evidence has established a link between gut microbiota and AIH, which may be explained by the increased intestinal permeability and bacterial translocation in AIH patients [10, 11]. In addition, pathogen-associated molecular patterns (PAMPs) and lipopolysaccharide (LPS) are recognized by toll-like receptors (TLR) in the liver, which will trigger inflammatory and/or immunogenic cascades and produce reactive oxygen species (ROS) [12]. Persistent or abnormal inflammation and immune responses are the key basis for microbiome dysregulation in chronic liver inflammation and tissue damage, leading to AIH [13].
Furthermore, gut microbiota can produce a variety of metabolites with various bioactivities, including metabolites produced directly from diets, produced de novo, and generated by the host [14]. Among all the gut microbiota metabolites, the three most widely studied metabolites are short-chain fatty acids (SCFAs), bile acids (BAs), and amino acid-derived metabolites [15]. In addition to microbial metabolites, gut microbiota can also produce other broader byproducts, including endotoxins like LPS, generally referred to as microbial products. Gut microbiota and its microbial products may lead to decreased BAs, SCFAs, and polyamines, as well as increased LPS, branched-chain amino acids (BCAAs), tryptophan derivatives, and amino acids. These changes disturb immune balance by triggering various immune cells and signaling cascades, resulting in immune system dysregulation [16, 17, 18]. Changes in gut flora, microbial metabolites, bacterial translocation, and intestinal barrier breakdown all contribute to AIH progression [19, 20].
Both population-based epidemiological studies and animal studies have demonstrated a significant association between gut microbiota and AIH, which paves the way for further clinical trials focusing on site-specific molecular and cellular interventions for better disease management and prevention [21]. A comprehensive understanding of the cellular and molecular mechanisms underlying such an association may fuel efforts at therapeutic manipulation. By revealing the impact of gut microbiota on clinical aspects, these efforts can confer novel insights into better-targeted therapies with fewer side effects, thus addressing the current unmet clinical needs of AIH treatment [22]. A narrative overview examining the connection between microbiota and AIH development can unveil its molecular bases and offer fresh perspectives on potential AIH treatments. Therefore, we conducted the current review to systematically summarize how gut microbiota and microbial products affect various immune regulations, metabolic regulations, and cross-organ axis actions of AIH.
Gut microbiota significantly influences the onset and progression of AIH. In human studies, taxonomic analysis of the fecal microbiota from both healthy individuals and AIH patients reveal differences at the genus level [10, 16, 17, 19, 23, 24, 25, 26, 27]. Wei et al. [11] discovered that AIH patients had a reduction in specific obligate anaerobes and a rise in Porifera. Liwinski et al. [16] found a relatively lower level of anaerobic bacteria and a higher level of facultative anaerobic bacteria and lactic acid bacteria in AIH patients. However, some studies showed inconsistent results in the association between gut microbiota and AIH. For instance, Lou et al.’s study [18] showed increased Faecalibacterium and Lachospiraceae among AIH patients, while Liwinski et al.’s study [16] showed the opposite. Similarly, Manfredo Vieira et al.’s study [19] showed elevated E. gallinarum in AIH patients, while Wei et al.’s study [11] reported no significant association.
Multiple studies have also illustrated various biological mechanisms underlying the association between gut microbiota and AIH. Lin et al. [10] demonstrated that intestinal flora imbalance and compromised intestinal tight junction (TJ) integrity led to increased serum bacterial LPS concentrations [10]. Liwinski et al. [16] revealed that decreased Bifidobacteria was associated with treatment resistance among AIH patients [16]. This research also indicated that AIH can be used to differentiate AIH and primary biliary cholangitis (PBC) [16]. Furthermore, some studies have built diagnostic and prediction models based on gut microbiota. For instance, one study utilized the bacterial genera Bacteroides, Lachnospiraceae, Roseburia, and Ruminococcaceae to differentiate between AIH patients and healthy controls, achieving an area under curve (AUC) of 83.25% [18]. Another study combined Bacillus, Lactobacillus, Oscillatory helicobacter, and Clostridium to identify AIH patients, achieving an AUC of 78% [11, 17].
In animal models, numerous studies have also shown a significant association between gut microbiota and AIH. Yuksel et al. [23] developed a new AIH model based on immunization of HLA-DR3GR mice with DNA plasmids encoding human CYP2D6/FTCD fusion protein. The AIH mouse model demonstrated a rise in Akkermansiaceae and Lachospiraceae and a reduction in Lactobacillus, Bifidobacterium, and Rikenellaceae relative to the healthy controls [23]. Wang et al. [24] found a decrease in Peptostreptococcaceae under the phylum Firmicutes and an increase in Rikenellaceae under the phylum Bacteroidetes in another MRL/MpJ-Faslpr (MRL/Lpr) mouse model. In addition, Manfredo Vieira et al. [19] found that E. gallinarum increased in the germ-free C57BL/6 mouse model.
Although most of the existing evidence of microorganisms related to the pathogenesis of AIH tends to be consistent, there are also contradictions, which may be explained by the differences in the characteristics of disease microorganisms, experimental methods, and patients included. The composition of the gut microbiota in AIH is shown in Table 1 (Ref. [10, 11, 16, 17, 18, 19, 23, 24, 26, 27, 28]).
| Country | Study population | Increased in AIH | Decreased in AIH | Sequencing technique | Refs |
| China | 37AIH:78HC | phylum-level: | phylum-level: | 16S rRNA sequencing | [18] |
| Verrucomicrobia, | Alcaligenaceae, | ||||
| Lactobacillaceae, | Victivallaceae, | ||||
| Leptotrichiaceae, | Erysipelotrichaceae, | ||||
| Enterobacteriaceae, | Acidaminococcaceae, | ||||
| Veillonellaceae | Lachnospiraceae | ||||
| genus-level: | genus-level: | ||||
| Veillonella, | Pseudobutyrivibrio, | ||||
| Faecalibacterium, | Blautia, | ||||
| Klebsiella, | Lachnospira, | ||||
| Akkermansia, | Erysipelotrichaceae, | ||||
| Enterobacteriaceae-unclassified, | Ruminococcaceae, | ||||
| Megasphaera | Phascolarctobacterium | ||||
| China | 24AIH:8HC | Escherichia coli and Enterococcus | Bifidobacte rium, | 16S rRNA sequencing | [10] |
| were unchanged | Lactobacillus | ||||
| Japan | 39PBC:17AIH:15HC | Lactobacillales | Clostridium subcluster XIVa, | 16S rRNA sequencing | [27] |
| Veillonella | Streptococcus, Fusobacterium | ||||
| China | 20PBC:32AIH:20HC | Escherichiacoli | Bifidobacterium, | 16S rRNA sequencing | [26] |
| Lactobacillus, | |||||
| Bacteroides, | |||||
| C.leptum | |||||
| China | 91AIH:98HC | genus level: | genus-level: | 16S rRNA sequencing | [11] |
| Veillonella, | Clostridiales, | ||||
| Klebsiella, | RF39, | ||||
| Streptococcus, | Ruminococcaceae, | ||||
| Lactobacillus | Rikenellaceae, | ||||
| Oscillospira, | |||||
| Parabacteroides, | |||||
| Coprococcus | |||||
| Germany | 99PBC:72AIH:95HC | Vs. HC | Vs. HC | 16S rRNA sequencing | [16] |
| Streptococcus, | Lachnospiraceae, | V1-V2 | |||
| Lactobacillus, | ND3007, | ||||
| Veillonella | Intestinibacter, | ||||
| Vs PBC: | Erysipelotrichaceae, | ||||
| Faecalibacterium, | Bifidobacterium, | ||||
| Haemophilus, | Lachnispiraceae | ||||
| Ruminococcaceae | FCS020, | ||||
| Vs. UC: | Clostridiumfamily XIII, | ||||
| UBA 1819, | AD3011, | ||||
| Phascolarctobacterium | Faecalibacterium | ||||
| Ruminococcaceae, | Vs PBC: | ||||
| Odoribacter, | Bifidobacterium, | ||||
| Senegalimassilia, | Sellimonas, | ||||
| Subdoligranulum, | UBA89, | ||||
| Coprobacter, | Blautia, | ||||
| Lachnospiraceae, | Romboutsia, | ||||
| NK4A136, | Coprococcus, | ||||
| Parabacteroides, | Lachnoslostridium, | ||||
| Butyricimonas | Flavonifractor, | ||||
| DTU089, | |||||
| Butyricicoccus | |||||
| Vs. UC: | |||||
| Bifidobacterium, | |||||
| Blautia, | |||||
| Erysipeloclostridium, Intestinibacter, | |||||
| Phascolarctobacterium | |||||
| Egypt | 15AIH:10HC | phylum-level: | Prevotella, | 16S rRNA sequencing | [17] |
| Firmicutes, | Parabacteroides, | V3-V4 | |||
| Bacteroides, | Dilaster | ||||
| Proteobacteria | |||||
| penus-level: | |||||
| Faecalibacterium, | |||||
| Blautia, | |||||
| Streptococcus, | |||||
| Haemophilus, | |||||
| Bacteroides, | |||||
| Veillonella, | |||||
| Eubacterium, | |||||
| Lachnospiraceae, | |||||
| Butyricicoccus | |||||
| China | 32AIH:20 NAFLD:20 HC | Escherichia coli | Bifidobacterium, | 16S rRNA sequencing | [28] |
| Lactobacillus, | |||||
| Bacteroides, | |||||
| C. leptum | |||||
| America | Sterile C57BL/6 mice | E. gallinarum | - | 16S rRNA sequencing | [19] |
| (NZW × BXSB) hybrid mice | |||||
| America | NAC MRL/Lpr mice | Akkermansiaceae, | Rikenellaceae, | 16S rRNA sequencing | [24] |
| CON MRL/Lpr mice | Lachnospiraceae | Lactobacillaceae, | |||
| Bifidobacteriaceae | |||||
| America | WT NOD mice | Proteobacteria, | - | 16S rRNA sequencing | [23] |
| HLA-DR3NOD mice | Bacteriodetes | V1-V2 |
PBC, primary sclerosing cholangitis; AIH, autoimmune hepatitis; UC, ulcerative
colitis; NAFLD, nonalcoholic fatty liver disease; HLA, human leukocyte antigen;
NOD, nonobese-diabetic; WT, wild type; HC, healthy people; NZW
The changes in microbial metabolites in AIH patients are mainly reflected in the decreased levels of BAs, SCFAs, BCAAs, polyamines, arginine, and proline metabolism, as well as increased levels of tryptophan and butyrate [16, 17, 18]. The gut microbiota of AIH shows a shift towards more aerotolerant microorganisms, accompanied by a decrease in SCFA-producing obligate anaerobic taxa and reduced fecal SCFA abundance [16, 29]. Butyrate is the most important component of SCFAs, and it decreases in AIH patients [16]. The reason remains unclear, and several researchers discovered that the quantity of butyrate-producing bacteria diminished during the demonstration of reduced diversity within the order Clostridiales [30]. In a protein-fed farnesoid X receptor (FXR) knockout mouse model, butyrate supplementation reversed dysregulated bile acid synthesis and reduced liver lymphocyte infiltration, while FXR inactivation reduced butyrate-producing bacteria and colonic butyrate concentrations [31]. These findings suggest that decreased fecal SCFAs may lead to increased progression of AIH disease. BAs are also influenced by the metabolism of gut microbiota during immune metabolic processes [32]. Around 95% of BAs are absorbed back into the body, and primary BAs produced by the liver are converted into secondary BAs by gut bacteria [33, 34]. AIH patients are rich in Bacteroides, Clostridium, Klebsiella, and Fecal bacteria, which may affect BA metabolism [17]. The primary BAs are rapidly hydrolyzed by bile salt hydrolase (BSH), mainly found in Firmicutes, Bacteroidetes, and Actinomycetes [35, 36]. Secondary BAs are negatively correlated with Klebsiella and positively correlated with Fecal bacteria [37].
Additionally, the available evidence shows that the changes in specific amino acids are closely related to the pathogenesis of AIH. AIH is associated with increased metabolism of tryptophan, lysine, and glutathione (GSH) as well as decreased metabolism of arginine and proline [11]. Glutamine (Gln), the most prevalent amino acid utilized in the human body, serves as a nutrient that boosts immune functionality [38]. Wang et al. [39] developed a concanavalin A (ConA)-induced AIH mouse model [to explore the Protection of Gasdermin D (GSDMD) in ConA-induced AIH]. Mice in the ConA group were challenged with 15 mg/kg ConA (Sigma-Aldrich) through tail vein injection to establish AIH. The analyses included alanine aminotransferase (ALT) and aspartate aminotransferase (AST) assessment, histology analysis, serum cytokine assessment, RNA extraction and real-time PCR analysis, serum LBP analysis, 16S rRNA gene sequencing, fecal LC-MS metabolome assessment, and liver LC-MS metabolome assessment [39]. The results showed an increased level of L-glutamine and a decreased level of glycerophospholipid metabolites in the mouse model, which were correlated with liver damage and inflammatory indicators [39]. Microbiota-derived metabolites altered in AIH and their roles in the disease pathogenesis are shown in Table 2 (Ref. [16, 17, 29, 31, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55]).
| Metabolites | Abundance (vs HC) | Function | Potential treatment | Reference |
| SCFAs | Low | Anti-inflammation, immune regulation, protection of intestinal epithelial integrity, anti-cancer, cardiovascular protection, liver protection, and neuroprotective activities | B420 can increase butyrate in the EAH mice | [16, 29, 40, 41] |
| The fructose-6-phosphate phosphoketolase pathway of Bifidobacterium can produce SCFAs | ||||
| A high-fiber diet induces the production of SCFAs | ||||
| BAs | Low | Lipid absorption and metabolism regulate inflammation | Supplementation with butyrate can reverse BAs synthesis dysregulation and its associated hepatitis | [31, 42, 43, 44, 45] |
| The nonsteroidal FXR agonist PX20606 and OCA | ||||
| Trp | High | Colonic motility and secretory activity | Supplementing Trp has a protective effect against CCL4-induced liver damage | [46, 47, 48] |
| Gln | High | Rapidly proliferating cells, energy source for effector T cells | GLS antagonist: JHU083; COX-2 inhibitor | [39, 46, 49, 50] |
| BCAA | Low | Muscle tissue construction, protein synthesis, activate insulin secretion | Branched-chain amino acid supplementation in treatment of liver cirrhosis | [51, 52, 53] |
| Arg | Low | Regulate immune response, guide T cell activity | Intravenous Arg administration CD4+ T cell homeostasis mitigates liver inflammation in mice with polymicrobial sepsis | [17, 46, 54] |
| Pro | Low | Cell signaling, stress protection, and energy production | - | [17, 55] |
SCFAs, short-chain fatty acids; BAs, bile acids; Trp, tryptophan; Gln, glutamine; BCAA, branched-chain amino acid; B420, bifidobacterium animalis ssp. lactic acid 420; GLS, glutaminase; Arg, arginine; Pro, proline; FXR, farnesoid X receptor; CCL4, carbon tetrachloride; EAH, S100-induced experimental autoimmune hepatitis; OCA, obeticholic acid; COX-2, cyclooxygenase-2.
The gut mucosal barrier blocks damaging agents from infiltrating the tissues, organs, and the bloodstream in humans. Primarily, it consists of four kinds of defenses: the physical, chemical, microbial, and immune barriers [56]. The primary physical barrier consists of close connections among mucus and various epithelial cells (such as goblet cells, Paneth cells, and microfold cells), known as tight junctions (TJs), which are made up of transmembrane proteins [57]. The primary components of the chemical barrier include mucus from the epithelium of the intestinal mucosa and digestive juices like stomach acid and bile, along with digestive enzymes, lysozyme, and antimicrobial peptides (AMPs) [58]. The digestive fluid can degrade some microorganisms and antigens by destroying the cell walls of microorganisms through digestive enzymes and lysozyme [59].
A microbial barrier is defined as the usual gut parasitic bacteria that resist colonization by external strains, which include Bacteroidetes, Firmicutes, Proteobacteria, and Actinobacteria. These bacteria groups form interdependent and interacting micro-ecosystems with the microspatial structure of the host [60, 61]. The gut’s immune barrier includes both the innate immune system, featuring antimicrobial agents like mucin and AMP, and the adaptive immune system, which involves intestinal mucosal lymphoid tissues, such as mesenteric lymph nodes, Kupffer cells, and secretory immunoglobulin A (slgA) in the intestine [62, 63, 64]. Intestinal epithelial cells absorb, process, and display foreign antigens, which then trigger the activation of both T and B lymphocytes, resulting in adaptive immunity and mucosal immune responses. Dysbiosis of gut microbiota induces spontaneous autoimmunity in target organs mediated by CD4+ T lymphocytes (CD4+ T) and CD8+ T lymphocytes (CD8+ T) cells [65, 66]. The gut symbionts engage with the host’s mucosal immune system, resulting in an imbalance between regulatory T cells (Tregs) and T Helper 17 (Th17) cells [67].
Chronic barrier dysfunction leads to bacterial translocation, inflammatory response, and epithelial cell apoptosis. It can also regulate liver inflammation by affecting the intestinal mucosal barrier and microbial composition. The term “enterohepatic axis” describes the bidirectional interaction between the intestinal system and the liver, which is implicated in the development of various hepatic disorders, including AIH [68]. Manfredo Vieira et al. [19] discovered that Enterococcus played a role in the progression of AIH by spreading to the liver and mesenteric lymph nodes. Furthermore, a study found decreased expression of TJ protein and increased serum LPS in AIH patients [10]. The compromised integrity of TJ pertains to the diminished prevalence of Escherichia coli and Enterococcus, confirming that AIH is associated with bacterial ectopia and gut microbiota dysregulation [10]. The gut microbiota translocates to the liver through portal vein circulation of biosynthesized metabolites and PAMPs, regulating inflammatory cytokines [69, 70]. Pattern recognition receptors (PRRs) serve as detectors for microbe-associated molecular patterns (MAMPs) and initiate the innate immune response, leading to liver inflammation [70, 71, 72]. There is a close relationship between intestinal microbiome changes, intestinal leakage, and AIH. A disrupted balance of intestinal microbiota may result in a deteriorated gut barrier and an unstable immune system. Additionally, the gut microbiota and their byproducts may serve as continuous antigens that provoke the immune system in AIH. Consequently, the gut mucosal barrier could be considered a viable therapeutic target for AIH. Gut-liver axis barriers and immune cells involved in AIH are shown in Fig. 1.
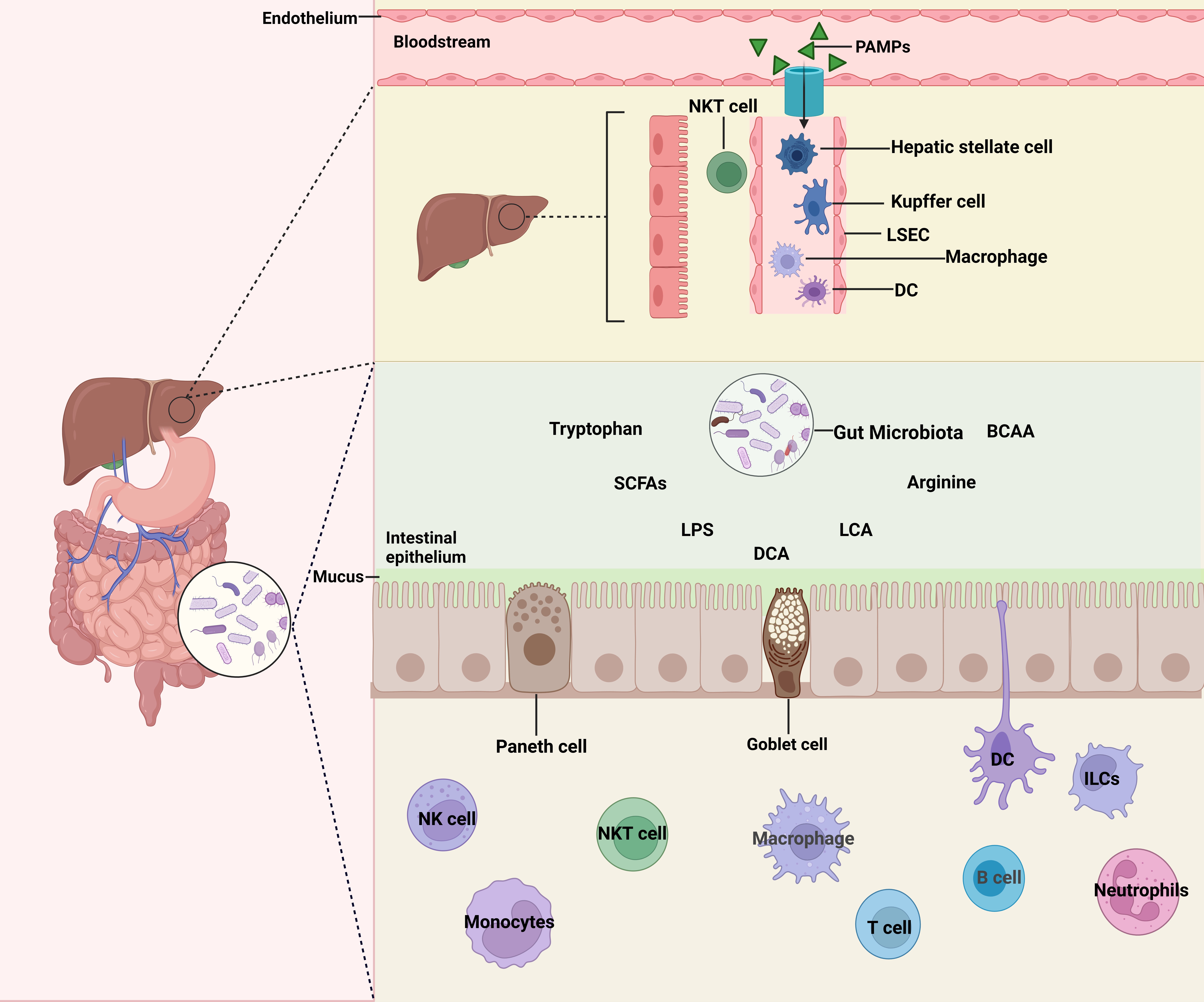 Fig. 1.
Fig. 1.
Gut-liver axis barriers and immune cells. The destruction of the intestinal barrier, along with pathogens produced by microorganisms and their metabolites, activates intestinal immune cells, leading to the generation of adaptive immunity. Abbreviations: PAMPs, pathogen-associated molecular patterns; NKT cell, natural killer T cell; LSEC, liver sinusoidal endothelial cell; DC, dendritic cell; BCAA, branched-chain amino acid; SCFAs, short-chain fatty acids; LPS, lipopolysaccharide; DCA, Desoxycholic acid; LCA, Lithocholic acid; ILCs, innate lymphoid cells. (Created with https://www.biorender.com/).
The liver is a vital immune organ rich in non-parenchymal hepatocytes and innate immune cells. Kupffer cells, hepatic sinus endothelial cells, and hepatic stellate cells represent the initial group of hepatocytes that encounter microbial products in cases of systemic infection or bacterial translocation [73, 74]. At present, various immune cells are known to participate in the pathogenesis of AIH, including dendritic cells (DCs), natural killer T cells (NKTs), liver macrophages, B cells, and monocytes [75, 76, 77, 78, 79, 80, 81].
Once the intestinal barrier is compromised, products from gut microbiota (like
LPS) along with gut-originated signaling molecules (such as PAMP and MAMP) pass
through the portal vein to the liver, where they activate the liver’s innate
immune receptors [82, 83, 84]. TLR expression is found in Kupffer cells, dendritic
cells, biliary epithelial cells, hepatic stellate cells, and hepatocytes [84]. In
Kupffer cells, the toll-like receptor 4–TIR-domain-containing adapter-inducing
interferon-
DCs and NKTs are important immune cells in AIH. Intestinal DCs can migrate to the liver via Peyer’s patches (PPs) [20]. Enteropathogenic Salmonella and streptococcus aggravate ConA-induced liver injury by inducing small intestinal DCs activation [10]. However, intestinal gram-negative bacteria can reduce ConA-induced liver injury by inhibiting NKT activation and DCs [90]. Damage to the intestinal barrier allows intestinal microbiota and their metabolites to pass into the liver, where they act as intestinal antigens. This transfer triggers the activation of hepatic DCs and NKTs, which are negatively regulated by gut microbiota [7]. However, antibiotic destruction of Syn bacteria causes overactivation of liver NKTs and accelerates immune-related liver damage [91]. An et al. [92] found that the intestinal microbe Bacteroides Fragilis could change the homeostasis of NKTs by supplementing the host’s endogenous lipid antigen environment with derived inhibitory sphingoid [93]. Sphingolipid can promote chemokine C-C motif ligand 5 (CCL5) signaling, thereby driving the expansion and activation of liver white blood cells [93].
The presentation of self-antigens to naive T cells can activate Th1, Th2, and Th17 pathogenic pathways and trigger AIH [94]. Tet2, a tet methylcytosine dioxygenase expressed in CD8+ T cells, serves as an epigenetic regulator linked to autoimmunity. The emergence of AIH-like conditions in Tet2 mice is associated with enhanced activity of Tc1 and Th1 cells in the liver. In the liver of Tet2 mice with AIH-like disease, bacteria that are ectopic to the liver can produce pathogens, triggering AIH pathology [95, 96].
In the liver,
AIH is characterized by chronic liver disease in which effector T cells,
macrophages, and plasma cells infiltrate the portal vein and periportal vein
along the interfacial liver [101]. Both SCFAs and BAs play essential roles in
immune regulation, with shared mechanisms via the activation of the signaling
pathways of G-protein-coupled receptors, such as G-protein
coupled receptor 109A (GPR109A), FXR, and the G protein-coupled bile acid
receptor 1 (GPBAR1) [9, 102, 103]. SCFAs can induce Tregs directly by inhibiting
histone deacetylases (HDACs) through solute carrier gene family 5a, member 8
(Slc5a8) [104] and indirectly by inducing Aldehyde dehydrogenase
(ALDH1A1) via GPR109A signaling [105]. Additionally, SCFAs
enhance the generation of Th1 cells and suppress the formation of Th17 cells
while stimulating T cells to produce IL-10 during the differentiation of both Th1
and Th17 cells [106]. Furthermore, SCFAs can also indirectly enhance
liver-resident NK (LrNK) cells function by acting on Kupffer cells and
hepatocytes through GPR109A [107] and control NK metabolism by inhibiting
mammalian target of rapamycin complex 1 (mTORC1), cellular
myelocytomatosis (c-Myc), and hypoxia-inducible factor-1 alpha
(HIF-1
NKT cells play a crucial role in the immune defense of the
liver and function as cellular detectors within the liver’s microcirculatory
system [111]. SCFAs can inhibit NKT cells from producing interferon
(IFN)-
Numerous amino acids play a role in the immune functions of the intestines and
liver and influence the progression of AIH. Lysine suppresses various
inflammatory mediators (encompassing TNF-
Among all amino acids present in humans, Gln ranks highest in quantity and
utilization. Yu et al. [50] developed a ConA-induced AIH mouse model to
explore the effect of Gln metabolism blocking on AIH. In this study, flow
cytometry analysis was used to evaluate the expression of T cell activation
markers (CD69 and CD25), enzyme-linked immunosorbent assay (ELISA) detected the
levels of IFN-
The interrelationships between immune cells, SCFAs, BA, and amino acids are shown in Figs. 2,3,4.
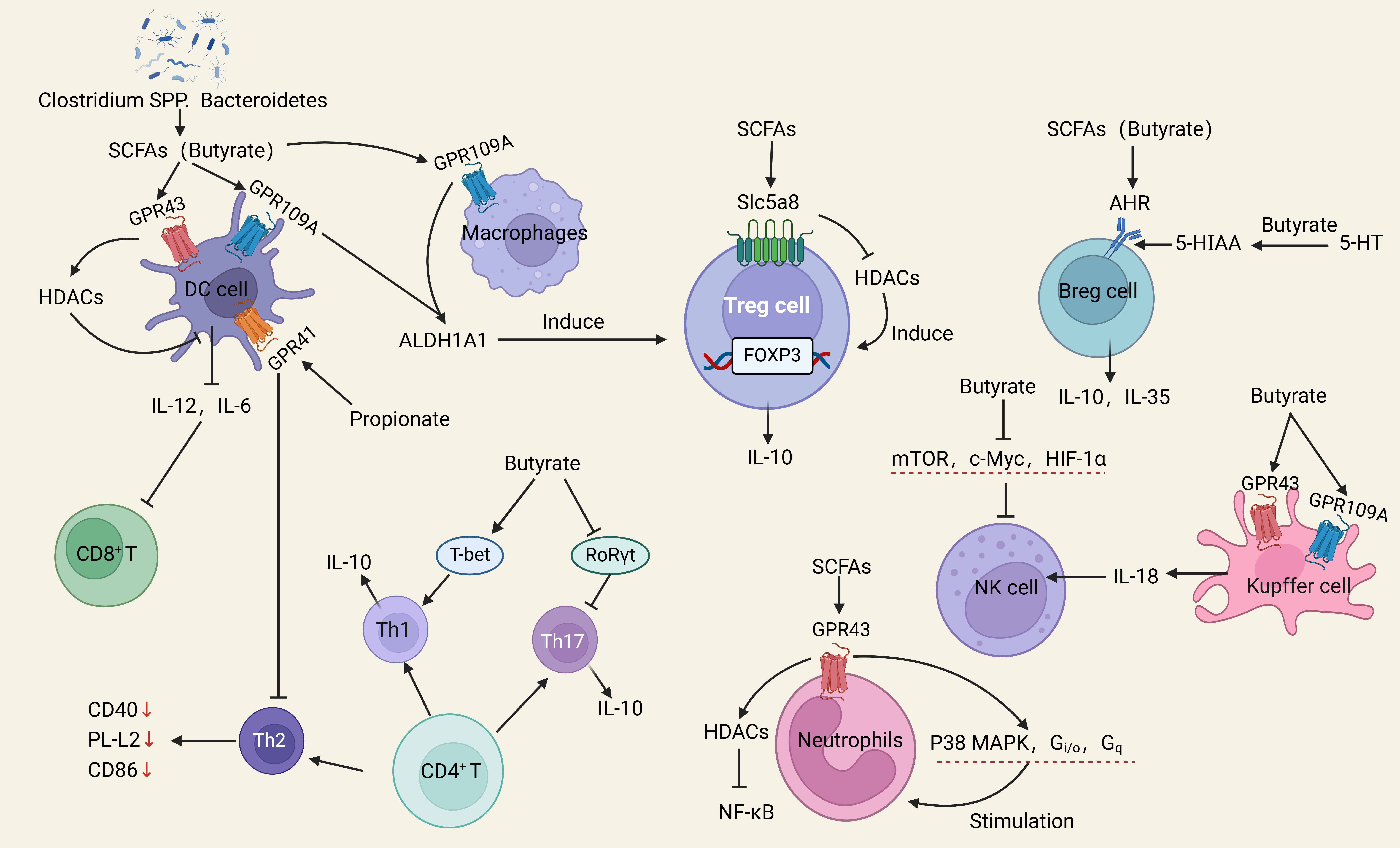 Fig. 2.
Fig. 2.
Schematic illustrations of the induction of various immune cells
by SCFAs. SCFAs induce Tregs and AhR ligands, regulate NK cell metabolism,
stimulate neutrophil migration, promote Th1 development, and inhibit Th2 and Th17
development. Abbreviations: Tregs, regulatory T-cells; DC, dendritic cells;
SCFAs, short-chain fatty acids; GPR41, G-protein-coupled receptor 41; GPR43,
G-protein-coupled receptor 43; ALDH1A1, Aldehyde dehydrogenase; HDACs, histone
deacetylases; mTOR, mechanistic target of rapamycin; MAPK, mitogen-activated
protein kinase; NF-
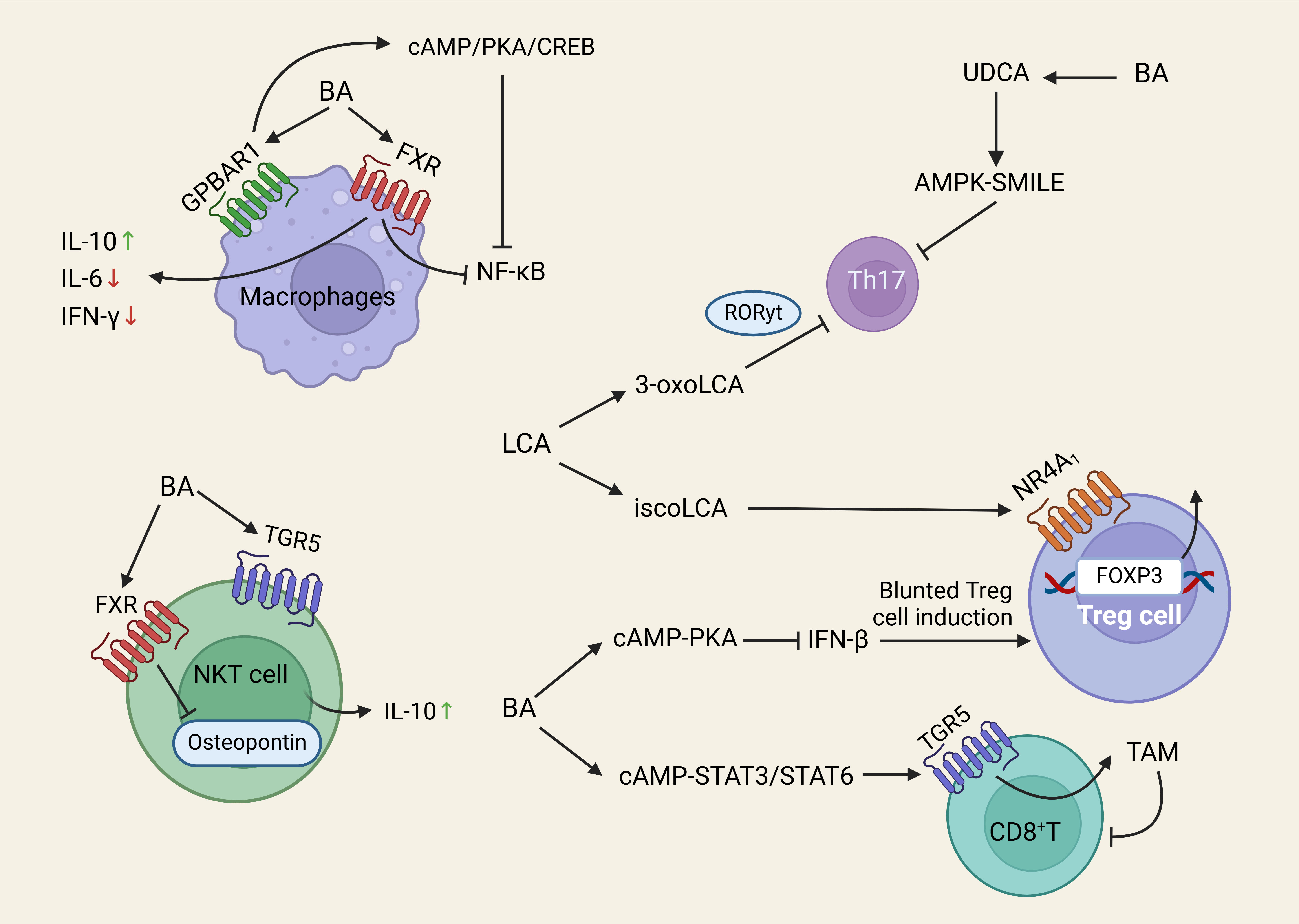 Fig. 3.
Fig. 3.
Schematic illustrations of the induction of various immune cells
by BA. BA in macrophages binds to GPBAR1. Inhibition of Th17 cell
differentiation, promotion of Treg cell differentiation, and reduction of Th17
cells. BA binds to TGR5 to activate the cAMP-STAT3/STAT6 signaling pathway.
Abbreviations: BA, bile acid; LCA, lithocholic acid; UDCA, ursodeoxycholic acid;
RoR
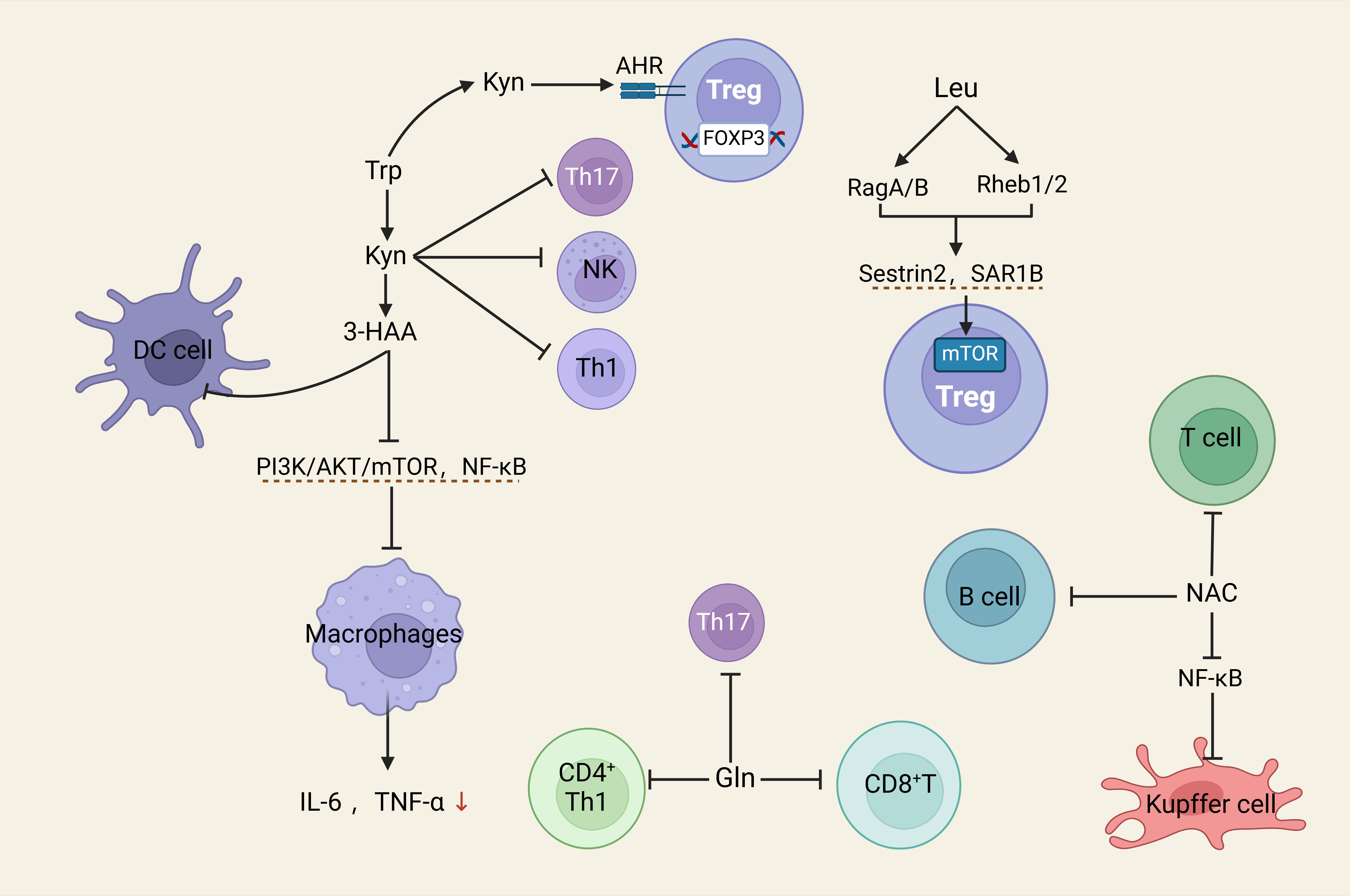 Fig. 4.
Fig. 4.
Schematic illustrations of the induction of various immune cells
by amino acid. Trp promotes the function of Tregs and inhibits the function of
Teff cells through the Kyn-AHR pathway. Abbreviations: Trp, Tryptophan; Teff cells, Effector T cells; Kyn,
kynurenine; Gln, glutamine; NAC, n-acetylcysteine; 3-HAA, 3-Hydroxyanthranilic
acid; NF-
At present, many studies have manifested that probiotics, prebiotics, and synthetic probiotics can effectively regulate intestinal flora. Typical probiotics are comprised of yeast, Clostridium butyricum, Lactobacillus, Bifidobacterium, and Actinomycetes [131]. Currently, available prebiotics typically consist of non-digestible oligosaccharides like oligofructose, fructooligosaccharides, and fructooligosaccharide-rich inulin, which promote the proliferation of advantageous bacteria strains, including Bifidobacterium and Lactobacillus [132, 133].
In an animal experiment, Bifidobacterium animalisssp. Lactic acid 420 (B420) alleviated S100-induced experimental AIH by regulating the intestinal microbiota configuration of the mouse model [29]. A study found that complex probiotics markedly decreased Th1 and Th17 cell differentiation while enhancing Tregs differentiation [134]. A recent study showed that Syn was superior to probiotics (Pro) or prebiotics (Pre) alone in ConA-induced acute liver injury [135]. In an acute immune-mediated liver damage mouse model, inulin-supplemented diet mice showed significant reductions in histology and serology of liver damage, demonstrating the benefits of prebiotics in the treatment of AIH [136]. These studies demonstrated that probiotics offer new hope for the treatment of AIH.
Noteworthily, most of the existing evidence regarding the beneficial effects of probiotics and prebiotics on AIH comes from preclinical research based on animal models. A previous clinical study found no significant effect of a probiotic cocktail therapy combining Lactobacillus and Bifidobacterium species in patients with autoimmune liver disease [137]. However, a meta-analysis involving 11 randomized controlled trials (RCTs) showed that probiotics, prebiotics, and synbiotics supplementation could potentially improve liver enzymes, lipid profiles, and liver steatosis in patients with non-alcoholic fatty liver disease [138]. Therefore, more clinical studies with larger sample sizes are warranted in the future to test the therapeutic use of probiotics and prebiotics on AIH in future clinical practice. In addition, it should be noted that various probiotics and prebiotics may produce different therapeutic effects in AIH treatment due to the different microbiome composition. At the same time, the supplemented bacterial strains also have the ability to positively alter the resident microbiota community. These microbiome-based variations could be used to predict patient responses to various therapies, determine the efficacy of probiotic use and its potential “personalized” effects as well as adjust treatment plans based on patients’ responses to achieve optimal outcomes. Several reviews have shown promising results from preclinical and clinical research underlining the emerging trend of a microbiome-based approach in AIH through improved understanding of the human microbiome and recent technological advances [139, 140]. Specifically, the detachment of microbiome research from the culture-based methods and the advancement of culture-independent techniques offered a new perspective to a more personalized microbiome-based treatment [139, 140].
FMT involves transferring healthy donor fecal bacteria into a patient’s intestinal tract [141]. FMT has been applied in treating numerous gastrointestinal disorders, indicating its promising therapeutic roles in autoimmune diseases. Research indicated that in a murine model of experimental AIH (EAH) mice, FMT could enhance liver health by reestablishing the gut microbiome balance and adjusting the TFR/TFH cell ratio via the TLR/MyD88 signaling route [26]. Furthermore, FMT treatment decreased bacterial translocation, mitigated hepatic injury, and partially normalized serum aminotransferases (ALT and AST) in CXCR5-/- EAH mice [28]. It also substantially enhanced the populations of Bifidobacteria and Lactobacillus and markedly reduced E. coli levels when compared to the control group [28].
FMT is emerging as a promising treatment for patients with autoimmune diseases, with well-established safety and efficacy in clinical study [142]. A systematic review and meta-analysis including 14 RCTs involving six types of autoimmune diseases showed that FMT was effective and relatively safe in treating a broad range of autoimmune diseases [143]. A meta-analysis based on 26 studies also found that FMT could significantly relieve ulcerative colitis and liver disease, with a good safety profile relative to the control group [144]. The therapeutic effects of FMT are reflected in various mechanisms, including restoration of altered gut microbiota composition, reconstruction of the intestinal microecosystem, and mediation of both innate and adaptive immune responses [142].
Clinical study has demonstrated that FMT is safe with low or nonsignificant adverse events, there is still a risk of infectious complications due to the impaired intestinal barrier integrity and dysfunctional immune responses of patients with autoimmune diseases [145]. In addition, the long-term effects of FMT for treating autoimmune disease are mixed, with some studies showing stable microbiome engraftment [146, 147] while others show nonsignificant effects [148]. Due to the limited data, small sample sizes, and high heterogeneity between studies, the safety profile and long-term efficacy of FMT in AIH remains unclear and warrants further research [142]. Furthermore, the applicability of FMT in the clinical setting may also encounter certain challenges, including finding a suitable donor, uncertain therapeutic effects due to complex fecal components and highly dynamic donor-specific microbiota, and the demand for highly specialized professionals, techniques, and conditions [142]. These challenges indicate the need for more evolutionary insights into the clinical application of FMT in the future.
Recent studies have identified two medications targeting the intestinal barrier
to reduce the symptoms of AIH: Berberine (BBR) and Pien Tze Huang (PTH). BBR
alleviates oxidative stress and regulates the intestinal microenvironment by
activating AMP-activated protein kinase signaling [149, 150]. It also stimulates
the growth of Akkermansia Muciniphila by enhancing mucin production in
the host intestine [149, 150]. In a mouse model, researchers also found that BBR
treatment increased beneficial bacteria in the intestine and reduced harmful
bacteria [149, 150]. PTH is a well-known traditional Chinese medicine that has
anti-inflammatory, neuroprotective, and immunomodulatory effects [151, 152].
Within the AIH mouse model’s liver, PTH decreased Th17 and multiple cytokines
while simultaneously enhancing beneficial bacterial populations and TLR2
signaling activation [153]. These actions increased IL-10-producing Treg/mTreg
cell numbers and suppressed the activation of both TLR4/NF-
The gut microbiome plays a vital role in maintaining immunological balance along the gut-liver axis. Nevertheless, current research examining the microbiome-immune-liver axis remains limited, and there is a lack of conclusive evidence of its therapeutic effects on AIH based on clinical trials. Although BBR has shown beneficial effects in alleviating concanavalin A-induced AIH in mice by modulating the gut microbiota [154], such effects have not been well demonstrated in clinical trials [155, 156]. Two meta-analyses showed that BBR treatment did not significantly reduce liver enzymes, including ALT and AST levels [155, 156]. Further RCTs with larger sample sizes and high quality are needed to examine the effects of BBR as a supplement for improving liver function in AIH patients. Moving forward, additional research focusing on the microbiome-immune-liver axis could provide insights into preventing and controlling AIH progression.
Treatments targeting microbial metabolites in AIH therapy mainly include SCFAs, BA, and amino acids-related metabolism. Sodium butyrate enhances the treatment of S100/FCA-induced AIH by altering the function of intestinal TJs and TLR pathways while also reducing the levels of pro-inflammatory cytokines [157]. A study found that both the high-fiber diet and sodium butyrate reduced liver damage and elevated ALT and AST levels in C57BL6 mice [41]. However, the positive therapeutic effects of butyrate and other SCFAs were predominantly based on animal studies, and more clinical trials are needed to test their clinical application and effectiveness in patients with AIH further. Notably, population studies have demonstrated that butyrate and other SCFAs are effective in regulating glucose metabolism and improving gut barrier function [158, 159]. Several RCTs showed that butyrate improved fatty liver index and plasma lipid patterns while attenuating inflammation in individuals with liver steatosis, metabolic syndrome, and diabetes [158, 159]. These findings suggest that SCFAs may be a promising therapy strategy for treating AIH, which warrants further clinical investigations.
Research shows that BAs signaling directly influence adaptive immunity [43, 160, 161]. OCA is a type of semi-synthetic BA and a steroid FXR agonist that reduces serum marker levels and improves the excretion of BAs from the liver [162]. FXR has hepatoprotective and anti-apoptotic effects in AIH [44]. In a rat model of CCL-induced cirrhosis, the nonsteroidal FXR agonist PX20606 can improve liver fibrosis and liver inflammation while reducing ALT and AST, indicating its potential to be used as adjuvant therapy for AIH [45]. Many AIH patients experience cholestasis, which can be effectively treated with ursodeoxycholic acid (UDCA) [163]. An RCT found that UDCA significantly decreased ALT levels, serum markers of fibrosis, and hepatic inflammation, with high safety and tolerability [164]. Another RCT showed that UDCA could modify the composition of the gut microbiota while not significantly affecting the microbial diversity [165]. A meta-analysis showed that UDCA treatment could significantly decrease ALT levels in patients with nonalcoholic fatty liver disease [166]. Another meta-analysis showed that UDCA therapy improved hepatic function by reducing liver enzymes, and its treatment effects vary across various subgroups [167]. Noteworthily, UDCA shows varying efficacy based on microbiome composition, with certain gut microbes influencing BA metabolism more than others. These findings suggest that a more nuanced understanding of the various roles of BAs in AIH treatment is needed, which can guide more effective and personalized therapy in the future.
Finally, various amino acids have also shown promising therapeutic effects in AIH treatment by enhancing the inflammatory response, reducing oxidative stress, and ameliorating immune dysfunction. Recent research indicates that AIH treatment involves mitigating mitochondrial oxidative stress, which could be reduced through the addition of antioxidants or GSH elements, including glycine, GSH, and NAC [168]. In a systematic review of 3687 patients, researchers found that corticosteroid therapy combined with NAC reduced fatal infection rates and short-term mortality [168]. In another meta-analysis of 672 patients, NAC improved transplant-free survival and overall length of hospital stay [169]. L-lysine supplementation in the liver has been shown to mitigate immune liver damage caused by CYP2D6 in mice, suggesting that L-lysine could be a potential supplementary treatment for AIH [51]. It should be noted that the clinical application of amino acids in AIH treatment is still in its infancy, with evidence predominantly coming from preclinical study [51]. More clinical trials testing the safety and efficacy of various amino acids in treating AIH are needed to provide more comprehensive clinical insights into their targeted and personalized clinical use in the future.
The gut microbiota constitutes a complex ecological network within the human
intestine, impacting numerous metabolic and immunological programs. An imbalance
in the gut microbiota leads to the disruption of the intestinal barrier,
facilitating the migration of microbiota to the liver and eliciting an
immunological response therein. This enables AIH progression through the
activation of relevant signaling pathways and modification of immune balance.
Furthermore, alterations in BA, SCFAs, specific amino acids, and elevated LPS
release stimulate signaling pathways, including TLR, NF-
A thorough elucidation of the precise pathways by which microbial-derived substances and bacteria promote AIH is still lacking, requiring further clinical and experimental research. Recent research on the gut microbiota of animals and humans primarily relies on fecal samples, which may not adequately represent the true condition and variations of gut bacteria. Moreover, the variability of gut microbiota and individual or ethnic variances can result in inconsistencies in sequencing outcomes using the widely employed 16S rRNA gene sequencing, rendering it inadequate for functional investigation. It is suggested that future studies should consider these variabilities and develop targeted and personalized therapeutical strategies based on the diversities in various characteristics. The existing clinical trials on the effectiveness of various treatment plans for AIH in human studies are limited by small sample size, considerable heterogeneity, and a lack of long-term follow-up. There is a need for high-quality RCTs with larger sample sizes and longer follow-ups to provide robust evidence of the effectiveness of AIH treatments.
Conceptualization, writing of original draft, reviewing, and editing (XZ); and conceptualization, reviewing, and editing (LW, YA and YZ). All authors contributed to editorial changes in the manuscript, read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
We would like to express our gratitude to all those who helped me during the writing of this manuscript and the peer reviewers for their constructive opinions and suggestions.
This work was supported by The Youth Talent Support Program of the Anhui Provincial Association for Science and Technology (RCTJ202429), National Natural Science Foundation of China Youth Project (8180050) and Technology project of China Three Gorges University (Z2023280/2023PTCM12).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




