1 Blue Mountains World Interdisciplinary Innovation Institute, Blue Mountains National Park, NSW 2782, Australia
Abstract
In the current perspective and review article, we address the human body as a living ecosystem with collecting watersheds and draining hydrosheds; we integrate our discoveries over the past quarter of a century and pose the critical open research questions to be addressed going forward, with the aim to improve cell, tissue, organ and organismal health. First, we address the flow of fluid through the tissues of the musculoskeletal system, after which we describe the interactions of the fluid, at multiple lengths and time scales, with the molecular to macroscopic non-fluid tissue components, discussing bone and tissues in the context of “living” chromatography and/or electrophoresis columns. Thereafter, we discuss the implications of functional barrier integrity, and the effects of cytokines on active barrier function and molecular transport between organ systems, tissue compartments, and within tissues. In addition, we address the fluid and its flow and the multi-physics implications thereof for the living inhabitants of tissues, i.e., the cells. Finally, we describe the implications of the solid and fluid components and the cellular inhabitants on ecosystem health, where the tissues and organs comprise the organism form interacting ecosystems throughout life and in the context of health and disease. By taking convergent approaches to understanding musculoskeletal, human and environmental health (which themselves are interdependent), we hope to pave new paths of innovation and discovery, to improve the lives of our worlds’ inhabitants, from the worlds of our bone and joints and bodies to the interacting ecosystems of our Earth to unknown worlds beyond our current understanding.
Keywords
- musculoskeletal health
- fluid flow
- synovial joint
- time scale
- length scale
- physiological systems
- ecosystems
- convergent approach
A quarter of a century ago, the nascent field of “bone fluid flow” emerged and thereafter grew into an ongoing, burgeoning area of research and discovery. Throughout the twenty-five-year period, parallel and combined, multi- time and length scale theoretical (typically computational) and experimental approaches advanced the field, across musculoskeletal tissues and organ systems (e.g., circulatory - lymphatic, vascular, immune, nervous, etc.) [1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21]. The current perspective review article highlights and integrates the resulting discoveries from my laboratory in context of the vibrant research arena; this is not intended to diminish in any way others’ significant contributions to the field which are wholeheartedly acknowledged (see References and Acknowledgements).
The origin and physiological implications of bone fluid flow trace back to a hypothesis first posed by Piekarski and Munro in 1977 [22], that mechanical loading of bone induces fluid flow through the network of periosteocytic interstitial fluid canals (the lacunocanalicular system, LCS), thereby augmenting via convection molecular transport to bone’s inhabitant cells. Twenty years ago, empathizing with the resident cells of our bones, we posed the osteocyte-centric research question, “Whither flows the fluid?” [4]. More recently, using molecular tracer tracking methods developed and described in our earliest papers [2, 3, 4, 5, 6, 23, 24, 25, 26, 27], combined with state-of-the-art organ-to-cell-scale cryo- and multimodal imaging methods [28, 29, 30, 31, 32], we have begun to answer the “whither” question in context of cellular inhabitants of not only bone but also different tissues comprising synovial joints of the musculoskeletal system. Our recent approaches expand our range of scientific query to the interplay between the cardiovascular and musculoskeletal systems [29, 30], as well as interface tissues of the mesoderm, including periosteum, ligament, interosseous membrane, and myofascial tissues [31, 32].
The current perspective and review article integrates the resulting discoveries from my laboratory and delineates the critical open research questions to be addressed going forward, with the aim to improve cell, tissue, organ and organismal health in context of the human body as a living ecosystem. To that end, we draw analogies between the ecosystem of the human body and those of environmental ecosystems such as the Amazon River basin [23] and the hydrosheds (analogous to a watershed, with the emphasis on the multiscale fluid flow network rather than the land drainage) of the greater Sydney basin. Insodoing, we use a convergent approach to crack a currently intractable challenge; as defined by the U.S. National Academies of Science, “[c]onvergence is an approach to problem solving that integrates expertise from life sciences with physical [referring to physics, chemistry, materials science, mathematical, and computational] sciences, medicine, and engineering to form comprehensive synthetic frameworks that merge areas of knowledge from multiple fields to address specific challenges. Convergence builds on fundamental progress made within individual disciplines but represents a way of thinking about the process of research and the types of strategies that enable it as emerging scientific and societal challenges cut across disciplinary boundaries in these fields. The concept of convergence … is thus meant to capture two dimensions: the convergence of the subsets of expertise necessary to address a set of research problems, and the formation of the web of partnerships involved in supporting such scientific investigations and enabling the resulting advances to be translated into new forms of innovation and new products.” [33].
When one describes the tissues and organs of the body in context of environmental biosystems or household objects such as kitchen sponges, one can better visualize the concept of interstitial fluid flow and load-induced fluid flow. Indeed, ecosystems such as swamps derive their name from the German Schwamm or Middle English Swamm which itself means “sponge”, originally describing an organism with a soft, porous skeleton capable of imbibing and retaining water even long after the organism has died, e.g., when the soft “sponge” skeleton found use as a bathing sponge in Ancient Greece [34].
Bones and cartilage are like swamps that sequester and hold water for entire tissue and organ ecosystems, providing sustenance for inhabitants of entire watersheds through vagaries of the weather and catastrophic events such as bushfires [35]. In contrast to the range and spatial distribution of porosities within, e.g., vertebrate tissues (nm to µm scales, as described in detail in [4]), the water holding pores of the hydroshed swamp “sponge” range from nm to µm, and even to m, if one considers the respective water holding pores of plants, the soil, the rock fissures and crevasses holding the ground water that wells to the surface via springs, and the vast network of streams, creeks, rivers and river basins that make up the hydrosheds supplying watersheds across the globe [23, 36].
Fluid flow through the human body and the tissues of the musculoskeletal system exhibits analogies to ecological hydrosheds, with multi length scale pores and conduits and diverse flow patterns described quantitatively by the dimensionless Reynolds number (Re). Defined by the ratio of inertial to viscous forces to characterize flow through pipes, Reynolds number is applied across diverse fields of use to compare flow patterns in different e.g., biosystems, where a high Reynolds number (generally greater than 2200) describes turbulent, i.e., irregular flow path with mixing (e.g., white water rapids), flow and a low Reynolds number (generally below 1100) is described as laminar, i.e., smooth path with minimal mixing (steady slow stream), flow. Inertial forces refer to the intrinsic resistance to acceleration (equal and opposite to accelerating force times the mass of the fluid) and viscous forces refer to the fluid’s capacity to resist relative motion between layers of the fluid. In most non-freezing ecological hydrosheds, the water viscosity remains roughly constant, while fluids of the human body can exhibit vastly different viscosities and can also invalidate continuum assumptions, e.g., at small length scales when plasma skimming or reduced rate of red blood cell migration or the formation of a red blood cell free layer occurs in the vicinity of vessel branches (bifurcations) [37, 38].
Within tissues of the musculoskeletal system, and within the biosystems of
tissue compartments comprising our synovial joints (knees, hips, etc.), most
flows, outside of the large nutrient vessels inserting from the cardiovascular
system, would be expected to exhibit laminar to creeping (Re
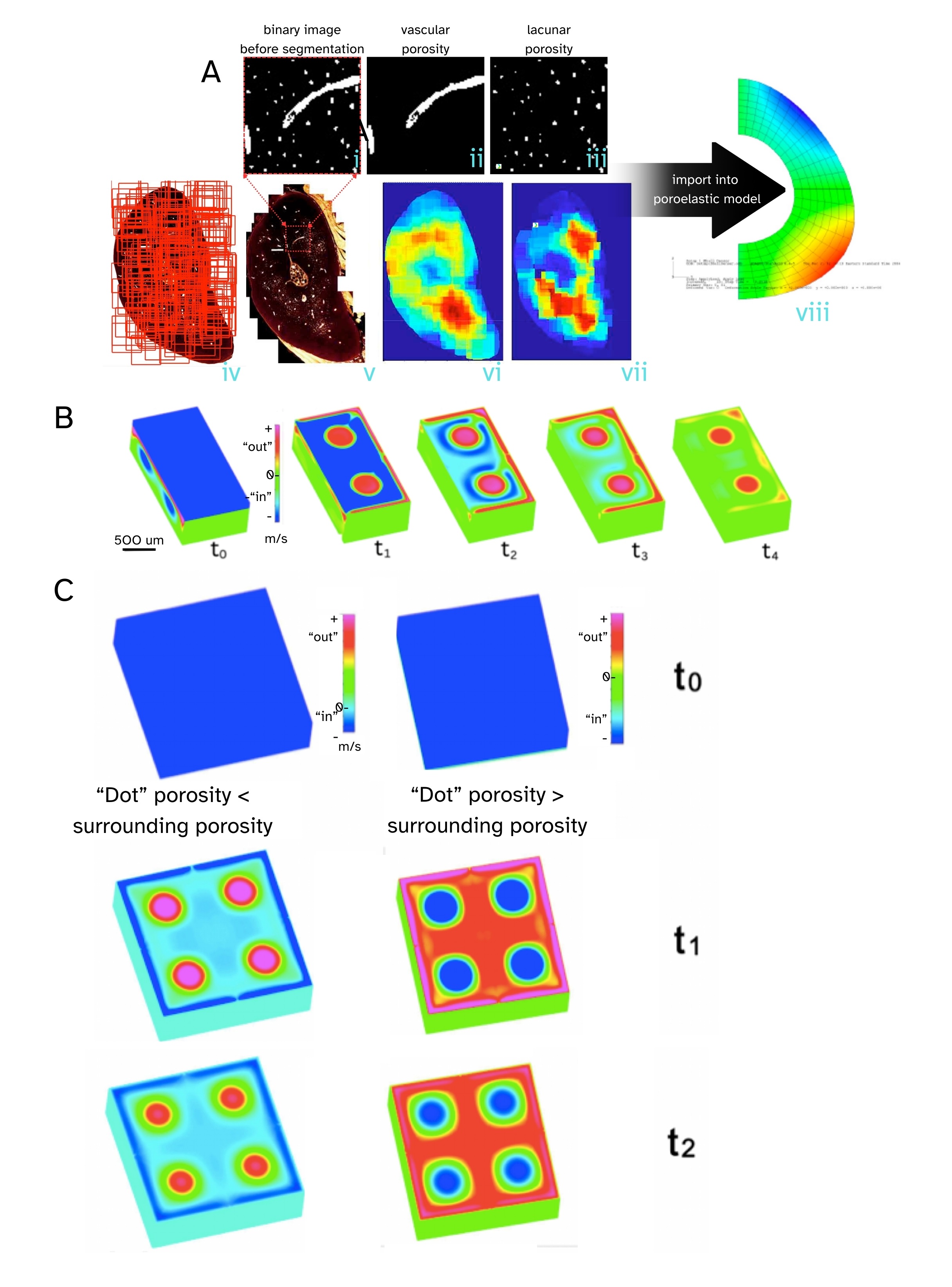
Bones and cartilage are like sponges, in that they are porous, and both imbibe as well as exude water, but their mechanisms of action differ. Interestingly, some areas of bone exhibit nonintuitive sponge like behavior, due to a unique combination of pore size/distribution and local mechanical stiffness (Fig. 1, Ref. [40, 41]) [40]. Namely, we showed experimentally, and probed effects using computational models, to discover and describe how “fortuitous combinations of anisotropic stiffness and permeability coefficients in [the] poroelastic structure [of] bone result in counterintuitive flow”, i.e., when the bone “sponge” is subject to compression (squeeze), bone imbibes fluid, whereas under tension (pull) bone egresses fluid. These “fortuitous combinations” of properties appear to be more prevalent in areas of bone that are less vascularized, providing compensatory mechanisms for molecular transport [40]. Just as ecological hydrosheds exhibit myriad fractal like networks at diverse length scales, bones exhibit both hierarchical and fractal-like vascular transport networks that fully anastomose with the hierarchical fluid porosity networks comprising the lacunocanalicular system (LCS) and bone nano- to microporosity [40, 41, 42, 43].
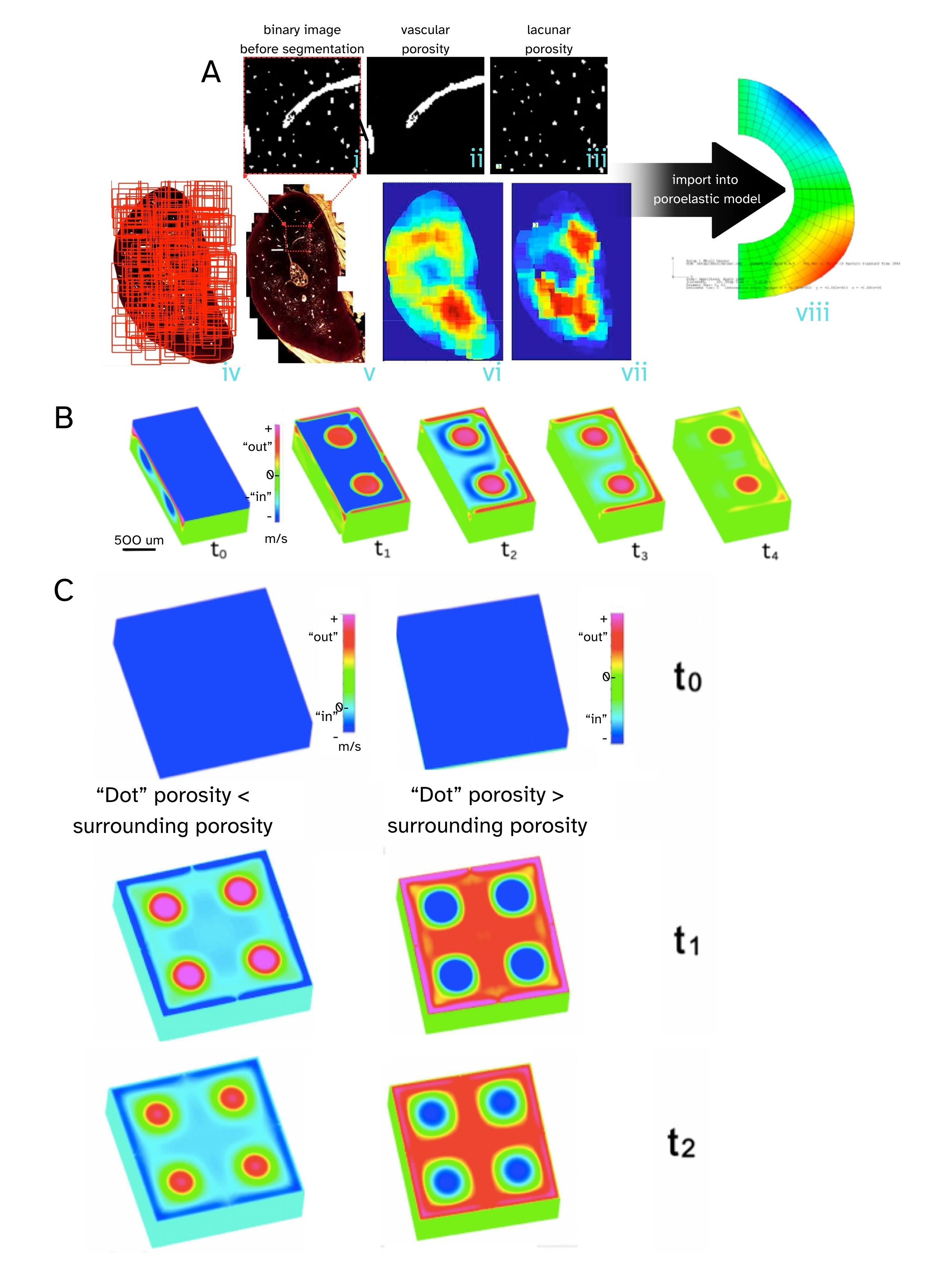 Fig. 1.
Fig. 1.
Bone, the “smart sponge”. Tracking high resolution spatial distribution of bone pores, including vascular porosity and lacunar porosity in the rat ulna cross section, together with poroelastic finite element modeling (FEM) and computational fluid dynamics (CFD) simulations, reveals counterintuitive flow (imbibement under compression and exudation under tension, opposite to that of a typical kitchen sponge) under specific pore distributions. (A) Thousands of high-resolution confocal images (iv) are stitched together to create a single high resolution image of the ulna cross section showing both vascular and lacunar pores (v). The black and white (binary) images (i) are separated into the respective porosities, including lacunar (ii) and vascular (iii), with density heat maps depicting their respective spatial distributions (vi, vii - warm colors indicate higher densities). Poroelastic FEM gave first predictions of counterintuitive behavior, with flow going into the bone under compression unlike typical sponge behavior, where squeezing a sponge squeezes the fluid out. (B,C) Using CFD we created a modular model system, akin to a miniature (500 micron on edge) computer keyboard key with four dots extending through the volume, on a computer keyboard, to model a tiny section of bone. (B) In cut through images, the depth of the “key” can be visualized. (C) Depending on whether the “dot” porosity was smaller (left column) or respectively greater (right column) than the porosity of the surrounding material, compressing the key resulted in either exudation at the dots and imbibement at the surrounding surface, respectively imbibement at the dots and exudation at the surrounding surface. Pressing on the key (compression) commenced at t0 and flow was followed along sequential steps, t1, t2, t3, etc. Outward (+) flows are red and pink, and inward (-) flows are turquoise and blue. Reproduced and adapted with permission from M.L.K.Tate [40, 41].
In addition to their swamp-like (longer length scale of tissue to organ) and sponge-like (length scale of tissues and sub tissue volumes) properties, bone and other tissue compartments of the musculoskeletal system exhibit properties of “living” chromatography columns. Chromatography is a laboratory technique in analytic chemistry designed to separate a mixture into its components, where a fluid mobile phase carries the mixture through a e.g., column or plate carrying a stationary phase; due to differential partitioning between both phases, the mixture constituents separate. Interestingly, flow rate impacts the resolution of e.g., size separation chromatography, where high flow rates shorten the run time but decrease resolution and slow flow rates increase resolution, i.e., prevents peak dispersion, which is the basis of high-pressure liquid chromatography for size separation [44].
Using bone tissue as an example, when biologically and chemically inert, fluorescent-tagged dextran molecules of increasing molecular weight (300–2,000,000 Da) are injected via the tail vein into anaesthetized rats, the differentially sized pores of the bone tissue sieve the molecules according to size [24]. The 300 Da probe penetrates the mineral matrix porosity which impedes permeation of larger tracer molecules. The larger pericellular spaces of the lacunocanalicular system (LCS) permit permeation of larger molecules up to 10 kDa. Without mechanical load-induced convective transport, transport of molecules above 10 kDa is ineffective through the lacunocanalicular space. With mechanical load-induced transport, probes up to 70 kDa penetrate the LCS. Beyond 70 kDa molecular tracers are impeded from bone porosity, irrespective of loading. Hence, “bone acts as a molecular sieve” and “mechanical loading modulates transport of solutes through the pericellular space that links osteocytes deep within the tissue to the blood supply and to osteoblasts and osteoclasts on respective bone forming and resorbing surfaces” [24].
Remarkably, in a study carried out using the same technique albeit in guinea pigs with and without naturally occurring osteoarthritis, and with a bolus injection of mixed size fluorescent-tagged molecules via the heart, we demonstrated that the tissues of all compartments of the knee joint exhibit molecular sieving, effectively separating out the bolus of two differently sized (respective green—10 kDa and red—70 kDa) fluorescent tagged dextran tracers [29] within five minutes’ circulation time. Using state of the art episcopic cryoimaging, we measured volume (voxels) of red and green tracers which equate to relative tracer concentrations if all imaging parameters are controlled. Using this experimental approach, we observed that aged animals with naturally occurring osteoarthritis exhibited tracer concentrations lower than those of the younger cohort and that the younger cohort exhibited significantly higher concentrations of the 10 kDa (green) compared to the 70 kDa (red) molecular tracer. We were surprised to observe the dearth of fluorescence indicating a lack of tracer permeation in the muscle tissue of either cohort, although muscle fasciae did exhibit bright fluorescence. Tissues of the meniscus, ligament and tendon fluoresced strongly with the 10 kDa (green) tracer but articular cartilage did not. Bone tissue demonstrated colocalization of both the 10 kDa and 70 kDa tracers. The 70 kDa tracer appeared to be excluded from the bounding, interface tissues of periosteum, the growth plate, and cartilage, yet was abundant in the bone marrow compartment. In younger animals, small caliber channels through the articular cartilage fluoresced green with the 10 kDa tracer but not in the older cohort [29]. Based on these studies, the size selective sieving properties of bone and other musculoskeletal tissues change depending on age and health status, respectively disease state.
Like chromatography, electrophoresis is a technique to separates molecules based on their size; in addition, electrophoresis can separate molecules based on their charge. Electrophoresis uses an electric current rather than fluid flow or pressure to move the molecules through a “column” comprising a gel or matrix [45]. It is typically carried out in an aqueous environment, as the separation mechanism harnesses the difference in migration rates of charged ions, molecules or particles in an electric field. “Most charged species are fairly soluble in aqueous media and thus water is the most obvious solvent for electrophoresis” [45]. Ions, molecules or particles “with a difference in their charge” to size ratio exhibit different migration rates [45].
Saturated (wet) bone tissue represents a model electrophoresis system, if one considers the evidence for streaming potentials with load-induced fluid flow in bone as presented experimentally, respectively computationally three decades ago by Guzelsu and Walsh [46, 47], as well as Zeng, Weinbaum and Cowin [48]. Streaming potentials derive from the mechanical force-induced motion of “ion carrying extracellular fluid in the bone matrix” [46], where the slope of the “streaming potential versus pressure [force/area of application]” “…[relate] to the electrokinetic (zeta) potential” and is linear in the low-pressure region. Furthermore, similar trends have been reported in comparing “estimated zeta potentials from streaming potentials with existing data obtained by particle electrophoresis…” [46].
In follow on experiments Walsh and Guzelsu [47] probed the unique contributions of the inorganic, exposed mineralized matrix and the organic, protein lined channels of the vascular system to the calculated zeta potentials from intact streaming potentials and postulated that the organic vessel lining “limits potential-determining ions’ access to the mineralized matrix”. At the time of Walsh and Guzelsu’s studies (1990–1991) [46, 47], my lab had not yet demonstrated that collagen lines the canalicular channels as well [49], which would further limit potential-determining ions’ access to the mineralized matrix, placing the dominant mechanism for streaming potential development onto the flow of fluid through the nano- and microporosity of bone itself.
Finally, Walsh and Guzelsu [50] examined effects of osmotic gradient induced flows (in the absence of mechanical loading) on streaming potentials in saturated bone exposed to high ionic strength (0.75) NaCl solutions, observing “flow-dependent streaming potentials in the absence of mechanical deformation”, indicative of how changes in the ionic concentration of the fluid phase of bone resulting from e.g., trauma and/or health conditions, may also impact flow through bone and physiology of the cellular inhabitants of bone tissue.
Early studies (early 2000s) from my group in collaboration with Schaffler and Nasser [51], where we developed a fatigue fracture model in the forelimb of the rat [51], showed a systemic increase in the small molecular weight (615 Da) fluorescent intravital tracer, Procion Red, permeability, indicated by increased tracer fluorescence in the fractured ulna as well as the uninjured contralateral ulna of the control side compared to healthy, uninjured control ulnae [52]. At the time we postulated that immunomodulatory cytokines, released in response to the localized fracture, exerted a systemic effect on bone tissue permeability.
In the subsequent years, research groups determined across a variety of tissues, from brain to lung, that cytokines, secreted by cells of the immune system in response to inflammatory events as diverse as flu or trauma, modulate molecular transport and molecular barrier function across tissue interfaces. In 2013 in collaboration with Docheva and Richter et al. [53], we demonstrated for the first time that human periosteum, the outer bounding membrane of bone, expresses zonula occludens 1 (ZO-1), “a tight junction membrane protein conferring epithelial barrier membrane properties” to periosteum. The implication of this functional barrier membrane property covering all nonarticular surfaces of bones was significant, as it provided a putative molecular mechanism [54], together with Sharpey’s fibers “velcro-ing” periosteum to bone [55, 56], to “zip-lock” and “unzip” in a controlled manner the barrier function on the outer surface of bone, via immunomodulatory control.
Fifteen years after our initial observation of systemic permeability changes
with trauma (2001) [51, 52], we tested the hypothesis “that two common cytokines,
with multifaceted roles in the etiology of osteoarthritis as well as immune state
in general, modulate the barrier function properties of joint tissue
interfaces”. We delivered fluorescent tagged 70 kDa dextran tracers in a single
bolus with one of two immunomodulatory cytokines, transforming growth factor-
In health, the interstitial fluid flow cycle sustains the inhabitant cells of the respective tissue compartments of the e.g., synovial joint ecosystem by ensuring nutrient and waste transport. Any changes resulting in less efficient transport of either nutrients or waste would be expected to impact adversely on the cellular inhabitants of the respective ecosystems, as we measured using classical experimental fluid mechanics methods in scaled-up, 3D-printed volumetric renderings of actual LCS image stacks from healthy and diseases subjects [61] (Fig. 2, Ref. [23, 62, 63, 64, 65]). This circles back to Piekarski and Munro’s original hypothesis of 1977 [22], where they postulated but could not yet prove that mechanical loading of osteons, with their concentric arrangement of lamellae, would promote convective transport as a means to augment less efficient diffusional molecular transport through the LCS in the mineralized matrix of bone, thereby sustaining the cells (osteocytes) firmly “rooted” (non-motile) within the bone matrix. In context of the historical development of this research area, it is interesting to note that Biot first developed the theory of poroelasticity, since applied to fields of use as diverse as soil mechanics and hydrology to biological tissues, in 1941 [66]! Around the same time of Piekarski and Munro’s original postulate in 1977 [22], Carter and Hayes [67] published the first in a series of papers treating bone as a two-phase porous structure to probe its behavior under compression and Lakes and Katz [68] reported on the viscoelastic properties of wet cortical bone.
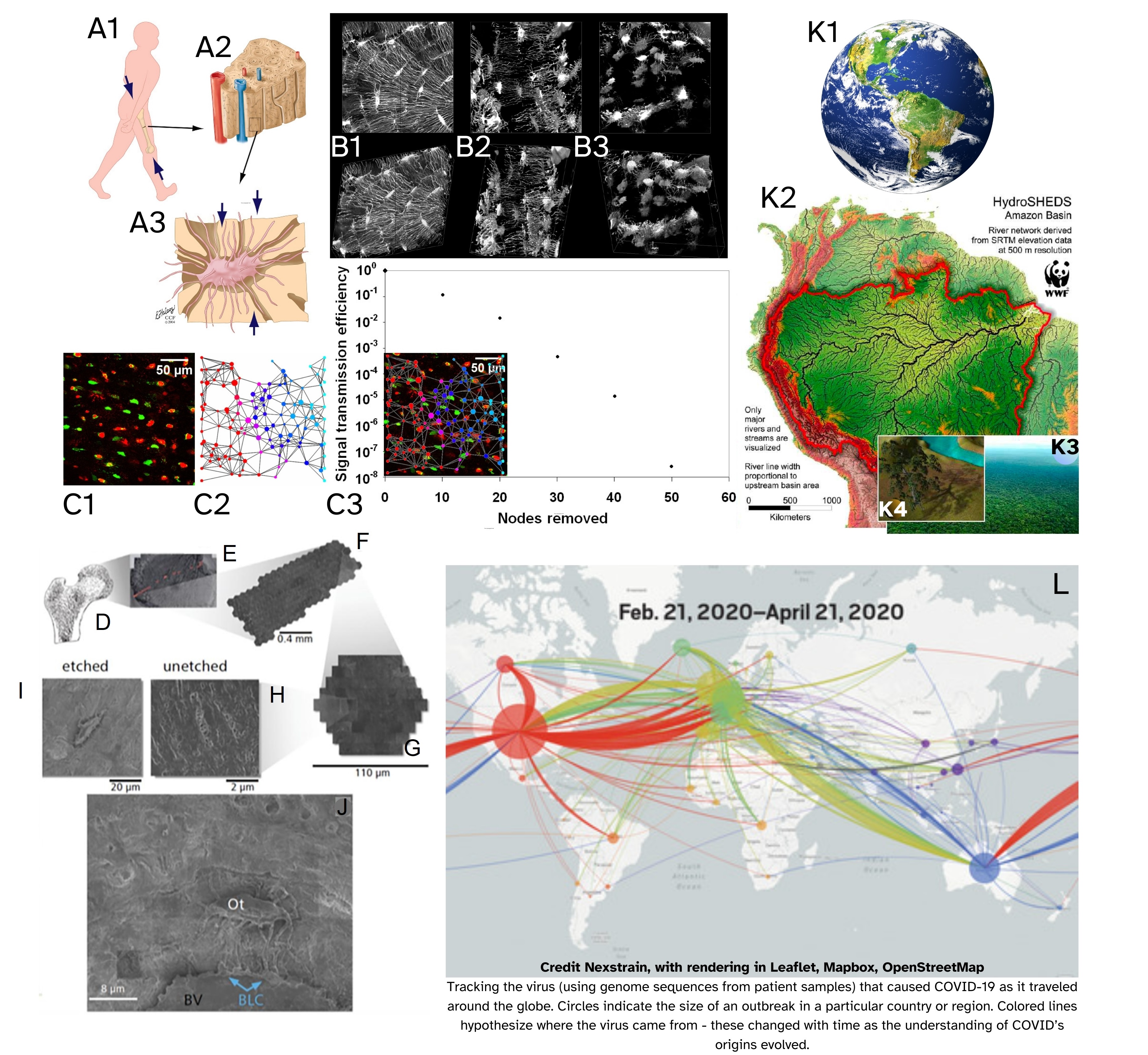 Fig. 2.
Fig. 2.
Cross scale approaches and cross domain synergies aid in predicting and managing emergence of diseases in diverse ecosystems of e.g., the human body and the Earth. Approaching diverse organ systems of the human body as interacting biosystems, with relevant hydro- and watersheds, and using geospatial and epidemiological approaches, offers unique utility to understand disease outbreaks and management in individual patients and populations of patients. New, ultra-high (time and length scale) resolution imaging modalities will in the future enable the identification and, thereafter, the prediction of disease emergence. This in turn is expected to enable a new era of healthcare where diseases are treated at earliest stages of emergence, before significant tissue degeneration occurs, ultimately paving a path for prevention. (A1–A3) Impact of physiological loading of the tissues of the musculoskeletal system on molecular traffic to/from inhabitant cells, (A1) e.g., the stiff, saturated “sponge” of bone tissue (A2), results in interstitial fluid flow through the lacunocanalicular system (A3) in which osteocytes are anchored. (B1–B3) High resolution volumetric microscopy of the osteocytic network demonstrates changes relevant to the cellular and pericellular networks of the living inhabitants of bone, (B1) in health, and disease, e.g., (B2) early osteoporosis, (B3) advanced osteoporosis. (C1–C3) Osteocyte network connectivity and its impact on molecular traffic, i.e., signal transmission efficiency (e.g., nutrient transfer) through the network: (C1) Red-green viability assay (ethidium bromide) indicates viable (green) and nonviable (red) osteocytes in situ in slice of bone tissue. (C2) The original cell network (C1) is recreated to probe effect of node removal from network on signal transmission through the network (C3). This provides a means to probe diverse effects of disease and trauma on signal transmission, both forward (prospective) and backward (retrospective) in time. (D–J) Advances in imaging have enabled google-maps navigation and study of human hip joints resected in the normal course of hip replacement surgery, with the capacity to zoom in from the entire hip to single cells and collagen fibrils inhabiting the joint. The technique allows for unprecedented length scale resolution but not yet temporal resolution of disease processes. Fig. 2 (A–J) is reproduced and adapted with permission from M.L.K.Tate [62, 63, 64]. (K1–K4) This convergent approach integrates advanced microscopy with geospatial, computational and machine learning (AI) approaches to enable a new era of epidemiology within the worlds of individual’s tissues, populations of individuals, and environmental health which is interdependent on human health. (K1) The Earth’s ecosystems include e.g., the Amazon River Basin (K2) including forest populations (K3) comprised of individual trees (K4). Adapted and used with permission [23]. (L) The geonavigational approach described in (D–J) was applied to track the outbreak and spread of COVID during the pandemic. Adapted and used with permission [65].
The essential nature of fluid flow to tissue health extends not only throughout the life cycle of organisms but also plays a requisite role at the earliest stages of life, i.e., in the patterning of the embryo and the emergence of complex, multiscale flow networks through development of the cardiovascular system and musculoskeletal system [69, 70]. As an example, the etiology of hypoplastic left heart syndrome, “a life-threatening congenital heart disease” in which left heart structures fail to develop correctly or completely, relates to disturbances in blood flow within the developing heart [71]. Mineralization of the bone templates (Anlagen) in utero is itself modulated by mechanical loading of the poroelastic cartilaginous Anlage [72, 73, 74], though the degree to which flow fields per se modulate the process has not yet been fully described. Even the transport of calcium, predominantly from the mother via the placenta through the fetal circulation to the skeleton is flow dependent [75] and in a relatively closed loop until birth; “the fetal kidneys filter the blood and excrete mineral into urine, which in turn makes up much of the volume of amniotic fluid, [which] is swallowed, and its mineral content can be absorbed by the fetal intestines, thereby restoring it to the circulation” ([75], see [76, 77, 78, 79] for further descriptions of blood flow in bone patho-/physiology). Indeed the flow of fluid is essential across lifeforms and length and time scales; the “flow of life” is just as important for nascency of coral reefs [80] and ancient mountain ranges [81, 82] as it is for human beings and for the diverse biological organisms supported through Earth’s own hydrosheds.
At some point in the life cycle of the organism, the balance of sustainability and health tips and degradation processes outpace growth and repair processes. This tipping point is a natural consequence of the collective effects of acute trauma, fatigue damage (wear and tear, under the threshold for acute failure, over many cycles) throughout life, lifestyle (diet, exercise) and age-related tissue degeneration which manifests in middle age, across all tissues of the human body. In addition, common musculoskeletal (osteoarthritis – for a detailed, recent review of transport related data related to the osteoarthritic joint, refer to [83], osteoporosis, osteomalacia) as well as other organ system-wide diseases, such as diabetes and cardiovascular disease, manifest not only with immunomodulatory changes but also with physical and chemical and flow pattern changes to both the “water- and hydroshed networks” of organisms, organs and tissues (Table 1, Ref. [23, 84, 85]). Such changes become evident, e.g., through denudation of native forests, where air and water flow patterns are changed and exert significant impacts on individual trees as well as the entire ecosystem; e.g., physical changes in boundary conditions via logging which exposes trees to higher gusts, increasing the potential for branch breakage and uprooting [23]. Spillage of chemicals and/or use of pesticides may affect viability of plants and organisms in exposed areas, changing the viability of the ecosystem not only in directly affected regions but potentially with far-flung effects given the interdependence of species within the ecosystem habitat [86].
| System - subcell | Effect on Flow, Transport | Example Disease States | Ecosystem Analogies | |
| 1 Cardiovascular | |||
| System | Cardiomyopathy, congestive heart failure | Changing systemic weather conditions or human interventions reducing wind, water tables | ||
| Meso | Edema, dehydration | Flooding, drought | ||
| Micro | Occlusions, local damage | Atherosclerosis, aneurysms, phlebitis | Damming, land slips, sediment pollution | |
| Peripheral vascular disease | Xylem vessel occlusion, insect (e.g., borer) damage | |||
| Tissue necrosis, gangrene | Xylem and root tissue disease: root rot, branch dieback | |||
| Nano | Opening, closing of cell-cell adhesions | Closing tight junctions in blood-brain-barrier promotes edema after infarct or stroke [84] | Closing or occlusion of plasmodesmata prevents exchange of cytoplasm between plant cells [85] | |
| 2 Immune | Diverse, e.g., Autoimmune conditions, most diseases and trauma involve inflammation | ||
| System | Systemic cytokine release, which modulates cytokines and tight junction permeability | Destruction of hydrosheds by humans and/or natural disasters, insect infestation, effects of pesticides; denuding of forest changes boudary conditions and hydrosheds, with trickle down changes at smaller length scales (e.g., plasmodesmata), affecting health and immune function of individual trees [85] | |
| Meso | |||
| Micro | |||
| Nano | |||
| 3 EM protein aggregation (Nano) | Diabetes, neurodegenerative conditions (e.g. tau proteins in dementia) | | ||
| Note: Effects at NANO-MICROSCALE scale up to matrix and tissue scale (MESOSCALE) | Protein aggregation, crosslinking modulates mechanical properties of matrix/associated tissues, including vascular networks, | MICRO to MESOSCALE - changes in algal and fungal populations, insect populations, scaling up to plant and animal populations | |
| MESOSCALE = Over/undergrowth of rainforest canopy (e.g. invasive vines overgrowing native plant species) – lack of balance of plant life due to pollution (pesticides), ageing of ecosystem or lack of renewal due to internal/external factors, deforestation/forest denudation/development (disruption to natural flow paths of air and water [23]) | |||
| Diminishment of network connectivity between cells = |
Changes in network connectivity, e.g. of syncytial plants and their communities | ||
1 and 2 take a systems-centric perspective on effect of changes to specific organ systems, and 3 takes a disease-centric view, up-scaling the nano-scale effect of age-related degenerative diseases, e.g., EM protein aggregation (dementia, diabetes), across systems. Botanical analogies are in italics.
Such physical changes range from changes in lacunar and canalicular size and shape, changes in network connectivity, to changes in mineralization and associated microporosity of the matrix and/or increased cross linking of organic matrix molecules. Associated changes to bone and musculoskeletal tissue compartment fluid biochemistry and/or ionic properties are less well described in context of flow and transport.
We are in an exciting era of science, where physical and algorithmic computing power has the potential, when used responsibly, to push other technological developments, such as in imaging, pharmaceuticals, theranostics, wearables, next generation implants cum bionics, exercise and physiotherapy as health adjuvants (like tooth brushing), to new heights with associated anticipated benefits for human and environmental health. Given increasing scarcity of R&D resources it will be important to both set priorities as well as to use convergent, interdisciplinary and multiscale approaches to ask and decipher the hardest, most compelling research questions that will also have impacts across disciplines, maximizing benefit for human and environmental health, e.g.,
- Understanding (bio)mineralization with development of integrated chemical engineering models of bone transport and balances across kingdoms and length-/timescales of life, from coccoliths to corals to bones to mountains [75, 80, 81, 82, 87];
- Deciphering synergies and parallels between sponge and tissue development, given that sponges are the earliest ancestors of most genuses and sponge evolution may give clues to processes relevant to human health, from endochondral ossification to age-related degeneration;
- Tying changes in network connectivity/permeability/signal transmission at the smallest length scale, e.g., tight junctions, to tissue and organ level molecular permeability, transport, and signaling underpinning healthy ecosystems and/or disease emergence [62, 63, 87].
Using convergent approaches such as those implemented for environmental protection and sustenance, one can envision a future with a more integrated understanding of musculoskeletal health in context of systemic wellness, and management of injury or disease using multifaceted approaches, from
- physical therapy and rehabilitation to
- implementation of wearables and implants cum bionics for augmented performance and continuous enhancement of cellular healing, to
- targeted cytokine control of functional barrier interfaces to modulate permeability direction and magnitude, with the aim to
- achieve guided transport of nutriceuticals or bioactive molecules to sites of healing and/or tissue neogenesis.
Interestingly, the recent albeit painstaking discovery of “dark oxygen” presents a compelling example of the power of convergent thinking, where lack of convergent thinking likely slowed the process of discovery, even though curiosity and persistence ultimately prevailed. Namely, it has long been assumed that all oxygen derives from photosynthesis, by plants and algae, with far reaching implications for the origin of biological life on Earth [88]. “Dark oxygen” refers to oxygen found in depths of the ocean (4000 m depth) where no sunlight can penetrate. Reported by Smith et al. [88] recently, “anomalous oxygen readings” in the depths of their ocean exploration site were originally thought to be attributed to defective sensors; over subsequent years of exploration, Sweetman and his team discovered that electrolysis, i.e., the separation of (sea)water into oxygen and hydrogen, via an electrical current, appears to occur naturally on the sea floor in the presence of rare metals called “polymetallic nodules”. Ironically, these metals have caught the attention of the mining industry for their battery-like properties.
Whether this groundbreaking discovery could have been made sooner through intentional convergent approaches cannot be tested, but the application of convergent research practices may provide an additional tool for transdisciplinary research teams to make new discoveries “hidden at the interface” between scientific and technological disciplines. The U.S. National Science Foundation describes convergence research as a “means of solving vexing research problems, especially those focusing on societal needs, ….driven by a specific and compelling problem [regardless of whether posed via “deep scientific questions or pressing societal needs”… [with a] deep integration across disciplines…intentionally bring[ing] together intellectually diverse researchers to develop effective ways of communicating across disciplines, …[causing] their knowledge, theories, methods, data and research communities [to] intermingle” [89].
By taking convergent approaches to understand musculoskeletal, human and environmental health (which themselves are interdependent) we hope to pave new paths of innovation and discovery, to improve the lives of our worlds’ inhabitants, from the worlds of our bone and joints and bodies to the interacting ecosystems of our Earth to unknown worlds beyond our current understanding.
LCS, Lacuno Canalicular System; Re, Reynolds number; Pe, Peclet number; Da, Dalton; kDa, kilo Dalton; TGF, Transforming Growth Factor; TNF, Tumor Necrosis Factor; ZO-1, Zonula Occludens 1.
The single author (MLKT) was responsible for the conception and writing and revision of the entire manuscript.
Not applicable.
My professional acknowledgement goes to my community of scientists and engineers, buttressed by previous and future communities, who have explored the world of our bodies and our bones, with each new discovery raising a multitude of new research questions. While the current perspective paper emphasizes discoveries from my own lab, our work would not have been possible without the discoveries and insights that came before and with our own; a review and integration of these would “fill a book”, the writing of which would be a worthwhile endeavor outside of the scope of the current perspective.
In addition, my heartfelt and deepest thanks go to my mentors, emeritus Professor Dennis Carter (Stanford University), emeritus Professor Peter Niederer (ETH Zürich), the late Professor Stephan Perren (AO Institute Davos), emeritus Distinguished Professor Clare Rimnac (Case Western Reserve University), emeritus Distinguished Professor Hunter Peckham (Case Western Reserve University), and Professor Chris Roberts (University of New South Wales), who have supported me throughout my career and without whose support I would have never had the opportunity to continue to push boundaries and ask the hardest research questions. I have also been privileged to have extraordinary colleagues with whom I have had the delight to engage in regular scientific discussions and critical thought experiments throughout the course of my career, including the late emeritus Professor John Currey (University of York), Dr. Dirk Zeidler (Zeiss Semiconductor Division, Oberkochen), Professor Vittorio Sansalone (University of Paris Est Creteil), Professor Stefan Milz (Ludwig Maximilians University Munich), Professor Iwona Jasiuk (University of Illinois Champaign-Urbana), Professor Thomas Bauer (Hospital for Special Surgery, New York), Professor Roy Aaron (Brown University), as well as my entire research team, many of whom started with me as protegés and with time became colleagues and collaborators. In addition, I am eternally grateful for my long-standing partner in scientific discovery and life, Dr. Ulf Knothe, who contributed to our lab’s discoveries in so many unique ways, ranging from microsurgical experiments - e.g., ex vivo sheep forelimb vascular and molecular transport studies (transplantation medicine), novel in vivo surgical methods to generate tissue in situ (regenerative medicine), bee brain extractions (neurophysiology) - to constant support of the MechBio R&D program through our successes as well as most challenging times.
Finally, I would like to express my gratitude to the anonymous donors and their families who so selflessly gave their tissues for scientific study after joint replacements (tissues removed in the course of replacing the joint, IRB approved) or after their passing (cadaveric tissue donation), thereby contributing to the future through generation of new knowledge and discoveries destined to promote human health throughout life.
The research program was supported in part through Swiss National Science Foundation Grants (823A-056609, 3200-049796.96), a Swiss Med Tech Initiative of the Commission for Technology and Innovation grant (3895.1, MedTech 536), AO Foundation Grants (99-K56,00-K49, 02-K83, 04-K3, 04-S4, 07-99K), U.S. National Science Foundation Grants (CMMI-0826435, 0335539), a U.S. NIH National Institute of Dental and Craniofacial Research Grant (R01-DE13740), U.S. NIH National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health Grants (R21 AR049351-01, R13 AR050594-01, 5 T32 AR 007505-20), grants from the Alexander von Humboldt Foundation, a Whitaker Foundation grant (RG-02-0527), NASA John Glenn Biomedical Engineering Consortium grants (JGBEC NCC3-1000, JGBEC NCC3-1008), grants from the Wallace H. Coulter Foundation, an Australian National Health and Medical Research Council Development Grant (APP1119636), and the Paul Trainor Chair of Biomedical Engineering endowment.
The author declares no conflict of interest with respect to the review. For full disclosure, Prof Knothe Tate serves as a science and technology advisory on a number of corporate and academic advisory boards, as an editor or member of the editorial board on a number of biomedical journals, and she has financial interests in start-up companies she founded; none of these activities intersects directly with the subject of the current scientific perspective which is fundamental in nature.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.