1 Laboratory of Pharmacology, Zaozhuang Thoracic Hospital, 277500 Zaozhuang, Shandong, China
2 School of Basic Medicine, Inner Mongolia Medical University, 010107 Hohhot, Inner Mongolia, China
3 School of Pharmacy, Inner Mongolia Medical University, 010107 Hohhot, Inner Mongolia, China
†These authors contributed equally.
Abstract
An appropriate animal model that can simulate the pathological process of atherosclerosis is urgently needed to improve treatment strategies. This study aimed to develop a new atherosclerosis model using ApoE-/- mice and to characterize lipidomics, gut microbiota profiles, and phenotypic alterations in adipose tissue using this model.
After a 14- or 18-week high-fat diet (HFD), male ApoE-/- mice were randomly divided into four groups and treated separately with or without short-term and strong co-stimulation, including ice water bath and intraperitoneal injection of lipopolysaccharide and phenylephrine. As a control group, C57BL/6 mice were fed with conventional chow. The serum lipid levels, aortic arch pathology, adipose tissue phenotypic changes, plasma lipidomics, and 16S rDNA gene sequencing of colon feces were investigated.
The serum lipid levels were significantly lowered following extended HFD feeding for four weeks. However, co-stimulation increased serum interleukin (IL)-1β levels but did not affect serum lipid profiles. Co-stimulation revealed typical vulnerable atherosclerotic plaque characteristics and defective adipose hypertrophy associated with peroxisome proliferator-activated receptor γ (PPARγ) regulation in adipose tissue and a reduction in mitochondrial uncoupling protein 1 (UCP1) within brown adipose tissue. Plasma lipidomic analysis showed that sphingomyelin (SM), ceramide (Cer), and monohexosylceramide (HexCer) levels in plasma were significantly elevated by HFD feeding, whereas co-stimulation further elevated HexCer levels. Additionally, glycerophosphocholines (16:0/16:0, 18:2/20:4, 18:1/18:1) and HexCer (C12:1, C16:0), Cer (d18:1/16:0), and SM (C16:0) were the most sensitive to co-stimulation. Combined co-stimulation and HFD-fed increased the abundance of Firmicutes, the abundance of f_Erysipelotrichaceae, and the Firmicutes/Bacteroidota ratio but decreased the abundance of microflora promoting bile acid metabolism and short-chain fatty acids (SCFAs) in mouse feces. The results were consistent with the findings of epidemiologic atherosclerotic cardiovascular disease studies.
This study established an ApoE-/- mouse atherosclerotic vulnerable plaque model using a multi-index evaluation method. Adipogenic disorders, dysregulation of lipid metabolism at the molecular level, and increasing harmful gut microbiota are significant risk factors for vulnerable plaques, with sphingolipid metabolism receiving the most attention.
Keywords
- atherosclerosis
- vulnerable plaque
- sphingolipid
- gut microbiota
- ApoE-/- mouse
Atherosclerotic cardiovascular disease (ACVD) is a chronic, multi-factorial condition involving lipid metabolic disorders, chronic inflammation, vulnerable plaques, and a proclivity for vascular thrombosis, all of which are risk factors for the condition [1]. Continuous endothelial cell injury cause lipid deposition, platelet adhesion, and arterial smooth muscle cell proliferation, while vascular turbulence and low temperature damage accelerate plaque progression [2]. The initial stage of atherosclerosis involves lipid deposition in the arterial endothelium, ranging from lipid spots to lipid streaks. Smooth muscle cells then proliferate and migrate, and the number of circulating Ly6chi monocytes rises. Most of these cells are drawn into the plaque, becoming foam cells upon the additional absorption of oxidatively damaged lipoproteins [3, 4]. In addition, bacteria-host interactions are closely linked to the initiation, progression, and repeated aggravation of atherosclerotic lesions, which may eventually result in thrombus formation, leading to acute coronary syndromes or stroke [5]. Pathological and imaging studies indicate that fragile plaques in human arteries show large necrotic cores, thinner or fractured fiber caps, more macrophages, spotted calcium, and positive remodeling [1, 6, 7].
Dysregulated lipid metabolism and gut microbiota dysbiosis are two established risk factors for ACVD [8, 9, 10]. Recent advances in coronary artery disease lipidomics have demonstrated that particular lipids, including oxidized lipids, sphingomyelin (SM) with its long saturated fatty acyl chains, glycerophosphocholines (PCs) with its saturated and monounsaturated heavy acyl chains, ceramide (Cer), and monohexosylceramide (HexCer, a subclass of glucosylceramide), are linked to the clinical classification and features of coronary artery disease [8, 9, 11, 12, 13]. In addition, lipopolysaccharides (LPSs) are known to be integral proteins of gram-negative bacteria that regulate the development of atherosclerosis. In one study, 516 middle-aged people participated in a five-year epidemiological study, and those with plasma LPS concentrations of 50 g/mL or higher had a three-fold greater risk of developing atherosclerosis than those with lower levels [8]. The catecholamine neurotransmitter phenylephrine is a vasoconstrictor that significantly increases plaque rupture rates [10]. Furthermore, numerous studies have demonstrated a connection between the gut microbiota and the development of atherosclerosis. A high-fat diet (HFD) is known to disrupt the balance of the gut microbiota, contributing to the progression of cardiovascular diseases by increasing the abundance of Erysipelotrichaceae, facultative anaerobes, and opportunistic pathogens [14].
White, beige, and brown adipocytes moderate metabolic health in humans. Accompanied by adipocytes and immune cells (such as B cells, T cells, and macrophages), white adipose tissue (WAT) stores excess energy and regulates endocrine functions. Brown adipose tissue (BAT), a type of thermogenic adipose tissue, is multilocular, is enriched in mitochondria, and exhibits high levels of the mitochondrial uncoupling protein 1 (UCP1), which controls thermogenesis and energy metabolism. Adipose tissue dysfunction associated with chronic inflammation has been identified as a cause of atherosclerosis. Gutiérrez-Cuevas et al. (2021) [15] reported that visceral WAT dysfunction increases the levels of pro-inflammatory adipokines, oxidative stress, and the renin-angiotensin-aldosterone system, leading to myocardial fibrosis and cardiac dysfunction. In another study, the association between BAT and cardiometabolic diseases was retrospectively analyzed in 52,487 study participants, and individuals with higher BAT/weight ratios independently correlated with lower odds of coronary artery disease, cerebrovascular disease, and other metabolic diseases, supported by lower values of blood glucose, triglycerides, and high-density lipoprotein levels [16].
Numerous atherosclerosis (AS) models have been constructed to induce the
formation of atherosclerotic plaques using different methods [17], including
physical injury [18], pharmacological action, and gene knockout approaches [19, 20]. However, these atherosclerosis models pathologically represent some, but not
all, features of ruptured human plaques [17, 18, 21]. For both primary and
clinical research, a suitable animal model of human ruptured plaques is
essential, considering the limits of clinical studies on mechanism mining.
Therefore, this study was conducted to establish an ApoE-/- murine
model of atherosclerotic plaque instability to investigate alterations during the
intermediate and advanced stages of atherosclerosis. As triggers for the ruptured
plaques, a HFD and short-term co-stimulation (including LPS, phenylephrine
intraperitoneal injection, and cold exposure) were employed. A set of indices for
model evaluation was developed based on serum biochemical indicators and
descriptions of pathomorphological alterations in plaques. Plasma lipidomics, gut
microbiota composition, and adipose tissue phenotype were evaluated after 14
weeks and 18 weeks of HFD-fed with or without co-stimulation. Furthermore, levels
of peroxisome proliferator-activated receptor
Male ApoE-/- (8 weeks old; 20–24 g) and wildtype C57BL/6 (8 weeks old, 22–25 g) mice were purchased from SiPeiFu BioTech Co., Ltd. (Beijing, China) and housed in an environment with a 12-h light cycle, controlled temperature (21–24 ℃), and free access to water and food. After ApoE-/- mice were fed an HFD (BEIJING KEAO XIELI Co., Ltd., # D12079B, Beijing, China) containing 21% fat, 50% carbohydrate, and 20% protein for 14 or 18 weeks, a modified mixture of short-term stimuli at the end of the experiment was used to activate unstable plaques, as reported by Wang et al. [19]. The mice undergoing short-term co-stimulation were divided into Model B-14wks (n = 12) and Model B-18wks (n = 12) groups. Specifically, after 14 or 18 weeks of HFD, mice in Model B groups were treated with short-term stimuli, including an iced water bath (0 °C) for 5–10 min on day 1 and 2, intraperitoneal injection with 1 µg/kg/day of LPS on day 1 and 2, and then intraperitoneal injection with 8 µg/kg of phenylephrine on day 3 (Fig. 1A). The ApoE-/- mice fed with HFD without co-stimulation were divided into “Model A-14wks” (n = 10) and “Model A-18wks” (n = 9). C57BL/6 mice (n = 10) served as the control group and were fed a standard chow diet (3% calories from fat). Body weight and food intake were recorded weekly for all mice.
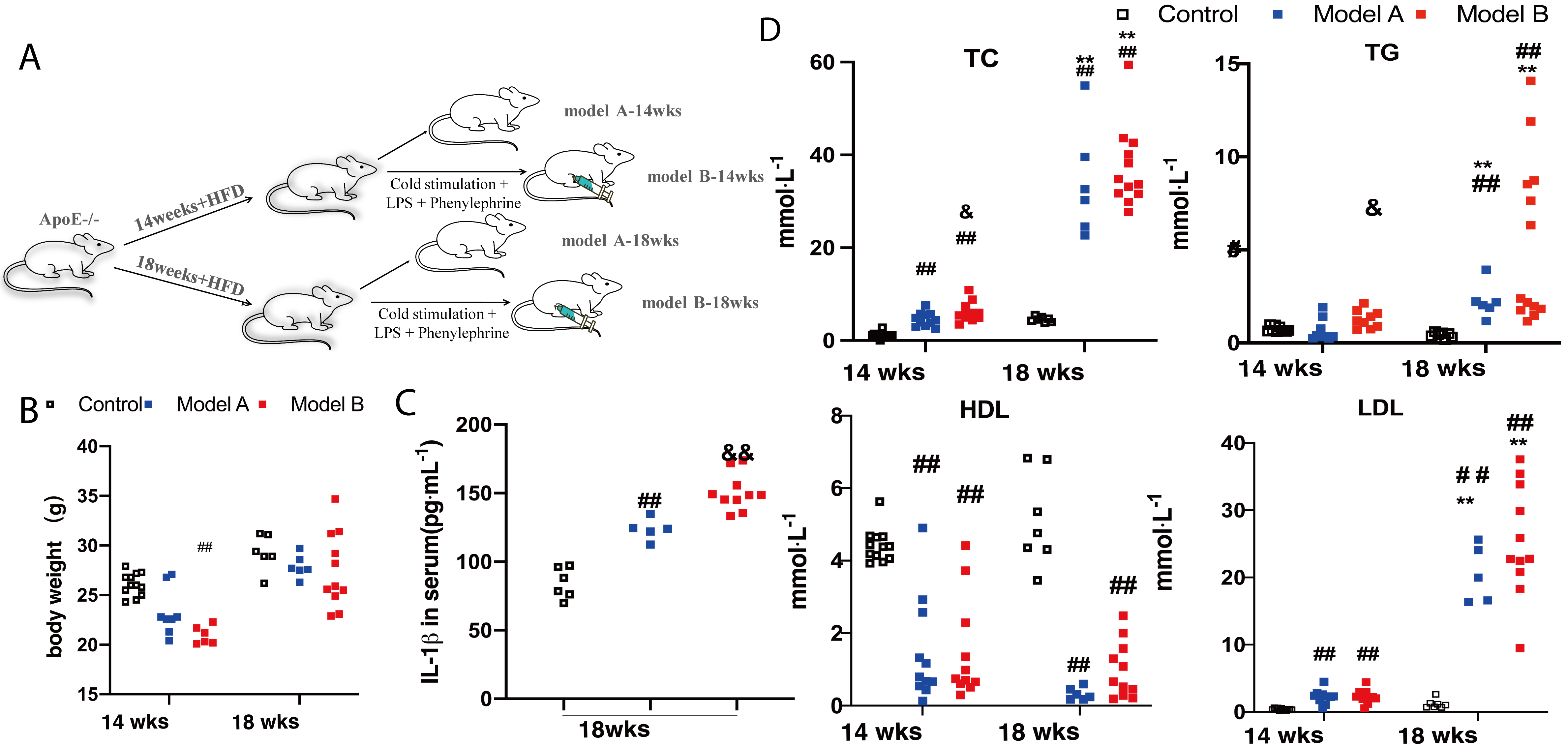 Fig. 1.
Fig. 1.
Body weight and biochemical parameters of mice. (A)
Experimental procedure. (B) Body weight of mice at 14 and 18 weeks. (C) Serum
interleukin (IL)-1
After 12 h of fasting, the mice were sacrificed under 5% isoflurane inhalation anesthesia and blood samples, the heart’s aortic root, the entire aorta, BAT on the scapular surface, and epididymal white adipose tissue (eWAT) were harvested for follow-up analysis.
Serum total triglyceride (TG), total cholesterol (TC), low-density lipoprotein
cholesterol (LDL), and high-density lipoprotein cholesterol (HDL) levels were
measured using commercially accessible kits (Nanjing Jiancheng Bioengineering
Institute, A110-1-1, A111-1-1, A113-1-1, A112-1-1, Nanjing, Jiangsu, China).
Mouse IL-1
Necrotic core formation is a hallmark of unstable atherosclerotic plaques. The
unstable necrotic core is a pit of hypocellular plaque lacking collagen and
containing thin fibrous caps, necrotic debris, and cholesterol clefts. During the
progression of atherosclerosis, the major arterial vessels undergo outward
remodeling, thereby increasing the lumen area. When evaluating atheroma lesions,
the area occupied by atherosclerotic lesions was measured via en face Oil Red O
staining of the aorta, and the degree of severity was quantified as the
percentage of atherosclerotic lesion area corrected by the total aortic volume.
Following a gradient of 10%, 20%, and 30% sucrose-saturated solutions for
dehydration, the heart’s aortic root embedded in the optimal cutting temperature
(OCT) medium was cut into 7-µm serial sections. Hematoxylin and eosin
(H&E) staining was used to analyze the areas of plaques, necrotic cores, and
vascular cross-sections [22]. The plaque
compositions of lipids, collagens, smooth muscle cells (SMC),
and macrophages were evaluated using Oil Red O (Service, #G2013, Wuhan,
China) staining, Masson’s three-color stain (Solarbio, #G1340, Beijing, China),
immunohistochemistry for the anti-
The adipose tissue was embedded in paraffin wax, sliced to 5-µm thickness, stained using a H&E kit (Service bio, G1076, Wuhan, China), and then photographed under an optical microscope (Leica, DM3000, Wetzlar, Germany).
Liquid-liquid-mass spectrometry (LC-MS) technology is the foundation of
lipidomic research [24, 25]. Lipidomic analysis was analyzed at Novogene
Technology Co., Ltd. (Beijing, China). Vortex treatment was performed on a 100
µL plasma sample (IAS) mixed with 750 µL methanol. Subsequently, at
room temperature (21–24 ℃), 2.5 mL of methyl tertiary butyl ether (MTBE) was
added, and the mixture was shaken for 1 h. Phase separation was induced by adding
0.625 mL of MS-grade water, followed by a 10-min incubation. The samples were
then centrifuged for 10 min at 1000 g. The upper phase was collected,
while the lower phase underwent a second extraction using 1 mL of a solvent
mixture (MTBE:methanol:water = 10:3:2.5, v/v/v). The organic phases from both
extractions were combined, dried, and stored at –80 °C for no longer
than 3 months. Before analysis, the dried residue was re-dissolved in 100
µL of isopropanol. Quality control (QC) samples were prepared by pooling
and equilibrating aliquots from each plasma sample. Plasma lipidomic
ultra-high-performance liquid chromatography coupled to tandem mass spectrometry
(UHPLC-MS/MS) analysis was conducted using a high-field quadrupole orbitrap (Q
Exactive™ HF-X) mass spectrometer system (Thermo Fisher Scientific Inc.,
Dreieich, Germany), coupled with a Thermo Vanquish™ UHPLC system (Thermo
Fisher Scientific Inc., Dreieich, Germany), equipped with a heated electrospray
ionization interface (ESI) (UHPLC-Q-Exactive-MS/MS analysis). Chromatographic
separation was performed on a Thermo Accucore C30 (2.1
Fecal samples for 16S rDNA gene sequencing were analyzed at Novogene
Technology Co., Ltd. (Beijing, China). Community DNA fragments underwent
paired-end sequencing using the Illumina HiSeq PE250 platform (NovaSeq6000, San
Diego, CA, USA). The bioinformatics pipeline for microbiome analysis was
implemented in QIIME 2 (https://qiime2.org). Amplification of the V3–V4 regions
of the 16S rRNA gene was achieved with the specific primers
F515 (5′-CACGGTCGKCGGCGCCATT-3′) and R806
(5′-GGACTACHVGGGTWTCTAAT-3′), using the gg_13 database
(https://www.arb-silva.de/) and DADA2 (https://benjjneb.github.io/dada2/) as the primary
denoising method. Experimental data reliability was evaluated through rarefaction
and species accumulation curves. Species diversity within samples was assessed
with
Frozen adipose tissue samples were homogenized for 30 min with
radioimmunoprecipitation buffer (1% phenylmethylsulfonyl fluoride (PMSF)). In
addition, protein expression levels of PPAR
Image-Pro Plus6.0 software (Media Cybernetics, Rockville, MD, USA) was used to
analyze histopathological images. Statistical analyses were conducted with
GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA), and the results were
expressed as mean
The weights of the mice and serum
biochemical parameters were analyzed to determine the effect of the combined
intervention and HFD on serum lipid levels. In contrast to the weight of mice in
the control group at 14 weeks, the weight of mice in the Model B-14wks group was
reduced (Fig. 1B) (p
Notably, at 18 weeks, the serum IL-1
The results of Oil Red O staining of the en face of the whole aorta are shown in
Fig. 2A. With short-term co-stimulation, the ratio of plaque area to the aorta
(the positive region of Oil Red O staining) was significantly higher in the two
model groups at 18 weeks than in the corresponding groups at 14 weeks (p
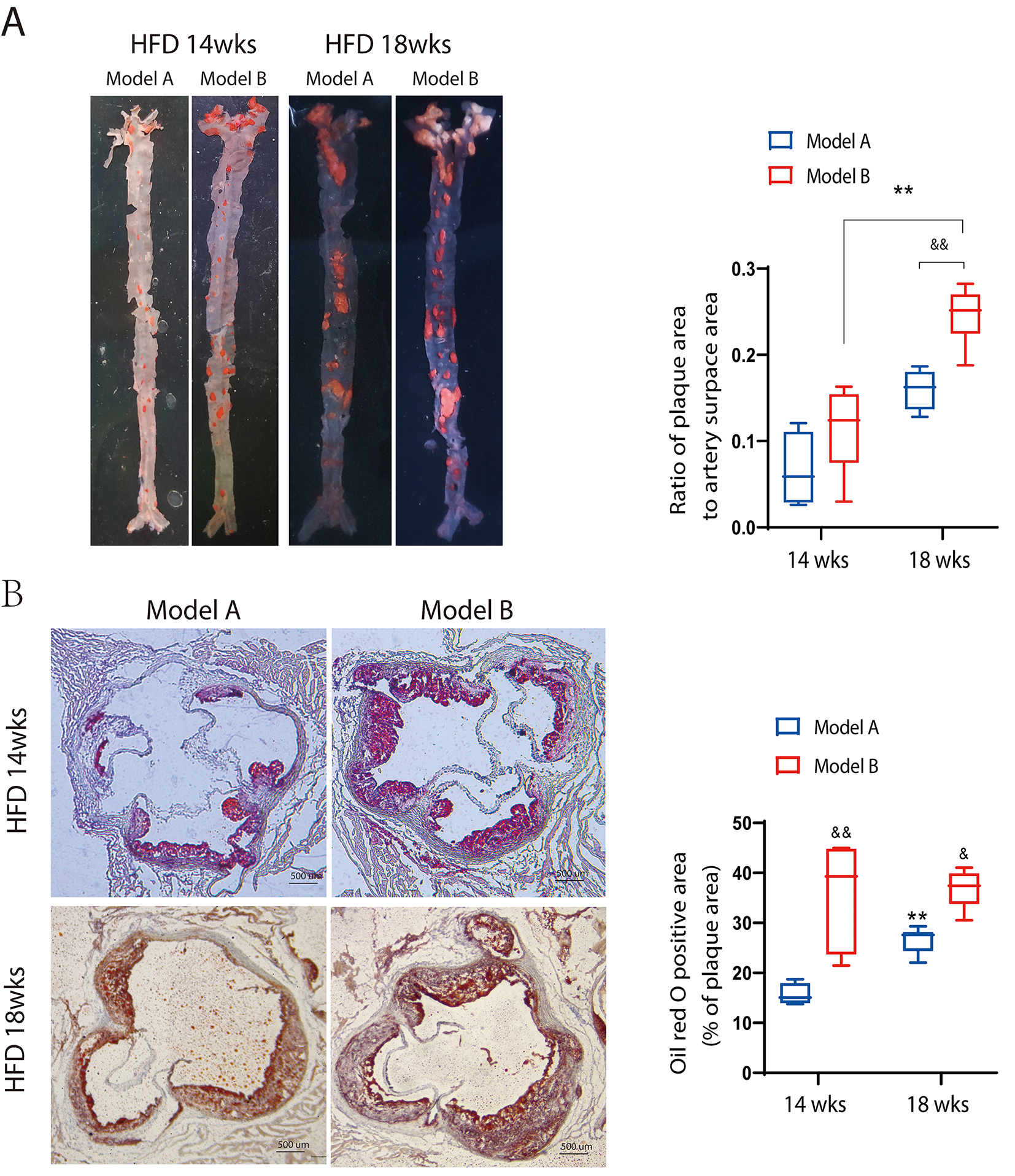 Fig. 2.
Fig. 2.
Oil red O staining of the face of the whole aorta and
cross-sections of the aortic sinus. (A) Oil
red O staining of the whole aorta. (B) Oil red O staining of the aortic sinus.
Scale bar: 500 µm. **p
H&E staining of the transverse aortic sinus was utilized to assess the
cross-sectional vessel area, the plaque-to-vessel area ratio, and the necrotic
core-to-plaque area ratio. The collagen content was evaluated using Masson’s
trichrome staining. A damaged fibrous cap and erythrocyte accumulation layer
indicated previous rupture and healing processes [11]. As shown in Fig. 3A,B,
when ApoE-/- mice were fed the HFD and co-stimulated, a
substantially greater plaque-to-vascular area ratio was observed in the Model B
group than in the Model A group at the same period (p
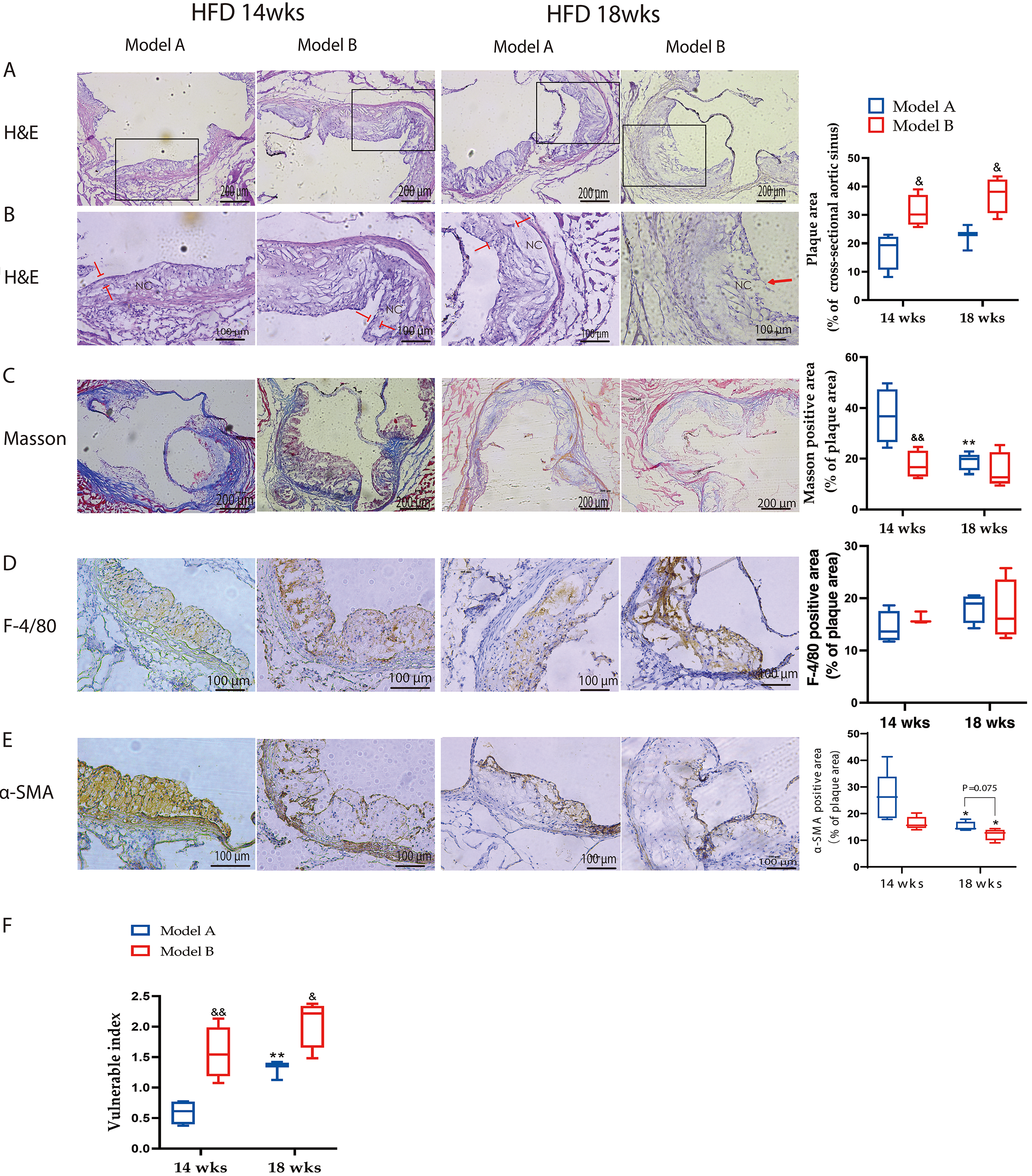 Fig. 3.
Fig. 3.
Plaque characteristics of ApoE-/- mice after combined
stimulation. (A) H&E staining of the aortic
sinus of Model A and B mice fed with an HFD after 14 or 18 weeks. The box
shows a substantially greater plaque-to-vascular area ratio was observed in the
Model B group than in the Model A group at the same period (p
To quantify the contents of macrophages and smooth muscle cells in aortic
plaques, immunohistochemical staining was performed using F4/80 and
These results suggest that a prolonged HFD promotes abnormal arterial vessel dilation and the rupture of elastic fibers. Furthermore, short-term co-stimulation accelerated lipid deposition onto the vessels, forming sparser, more extensive, and vulnerable plaques in the short term. The plaque phenotype observed in Model B-18wks mice was similar to that of unstable human plaques [27], including a huge plaque size, necrotic core, thin fibrous cap, decrease in entirety collagen, and positive remodeling, that is, a significant increase in vessel area. As the HFD time increased, these characteristics became more pronounced.
In the case of an excess energy supply, WAT
causes adipocyte hypertrophy (increase in volume) or hyperplasia (increase in
number). During adipose tissue remodeling, hyperplasia of WAT is generally more
advantageous than adipocyte hypertrophy. This is because small adipocytes exhibit
increased insulin sensitivity compared with large adipocytes, whereas large
adipocytes often indicate metabolic derangements [28, 29]. In this study, the
adipocyte phenotype in mice was assessed (Fig. 4). With co-stimulation, the size
of eWAT adipocytes in Model B-18wks mice was severely increased compared to that
of Model B-14wks mice (Fig. 4A), and the BAT adipocytes in Model B mice showed
widespread hypertrophy both at 14 and 18 weeks in comparison to those of Model A
at the same period (Fig. 4B). In addition, the ratio of both eWAT and BAT to body
weight (mg/g) of model mice at 18 weeks was higher than that of the corresponding
mice at 14 weeks (p
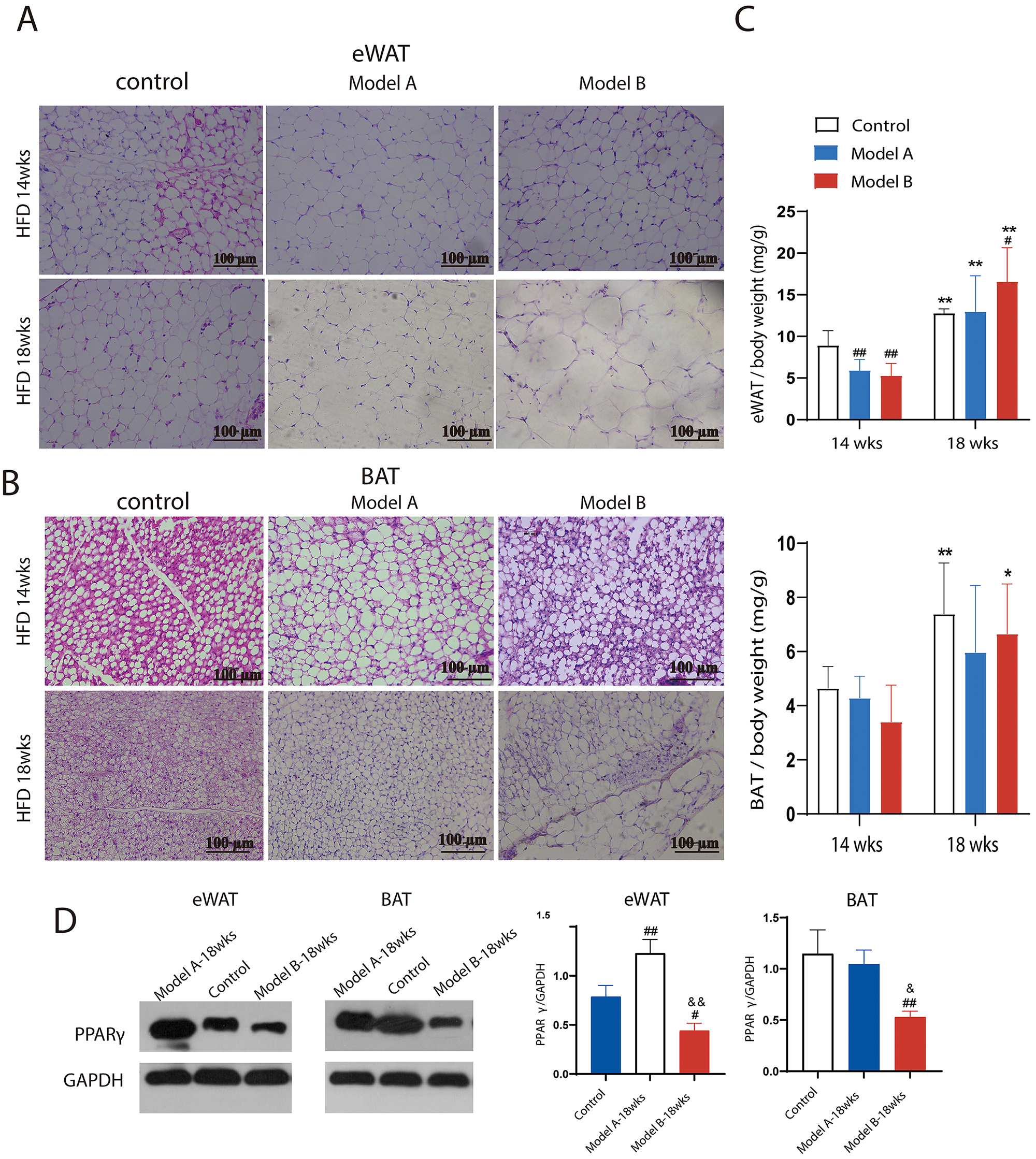 Fig. 4.
Fig. 4.
Combined treatment altered
adipose tissue phenotype. (A) H&E staining of white epididymal tissue
(eWAT), n = 4–6. (B) H&E staining of brown adipose tissue (BAT), n = 4–6. (C)
Ratio (mg/g) of BAT and eWAT to body weight, n = 10. (D) Peroxisome
proliferator-activated receptor
PPAR
Plasma samples from each group at 18 weeks were analyzed using UHPLC-Q-Exactive-MS/MS under both electrospray lonization (ESI) positive and negative ion modes. To maintain data reliability, QC samples were included in the sequence, which was run after every seven injections. The Pearson correlation coefficient between QC samples was between 0.99 and 1, indicating that the method was stable and appropriate for these samples throughout the testing process. Typical base peak ion chromatograms are shown in Supplementary Fig. 2. To minimize false positives, we further screened the data according toSkotland et al. [31]. A total of 988 lipid molecules was identified and analyzed, and these were mainly grouped into 16 subclasses, including fatty acyl (FA), triacylglycerols, PC, phosphatidylethanolamine, phosphatidylserine, phosphatidylglycerol, phosphatidic acid, phosphatidylinositol, SM, Cer, and HexCer. Detailed information about the identified lipids is presented in Supplementary Fig. 3.
Heatmaps of HCA and PLS-DA plots were used to visualize the cluster overview of
all plasma samples from the Model A-18wks, Model B-18wks, and control groups
having significantly different lipid compositions (Fig. 5A and
Supplementary Fig. 4). The volcano plot analysis results revealed that
the plasma lipid compositions of Models B and A at 18 weeks were notably altered
compared to those of the control group; that is, more than 400 molecules were
upregulated, and 30 molecules were downregulated in both groups
(Supplementary Fig. 5). The apparent separation between Model A-18wks
and Model B-18wks is shown in Fig. 5B, with good model quality (R2: 0.69 and Q2:
–0.78 in positive ion modes; R2: 0.76 and Q2: –0.68 in negative ion modes,
respectively; p
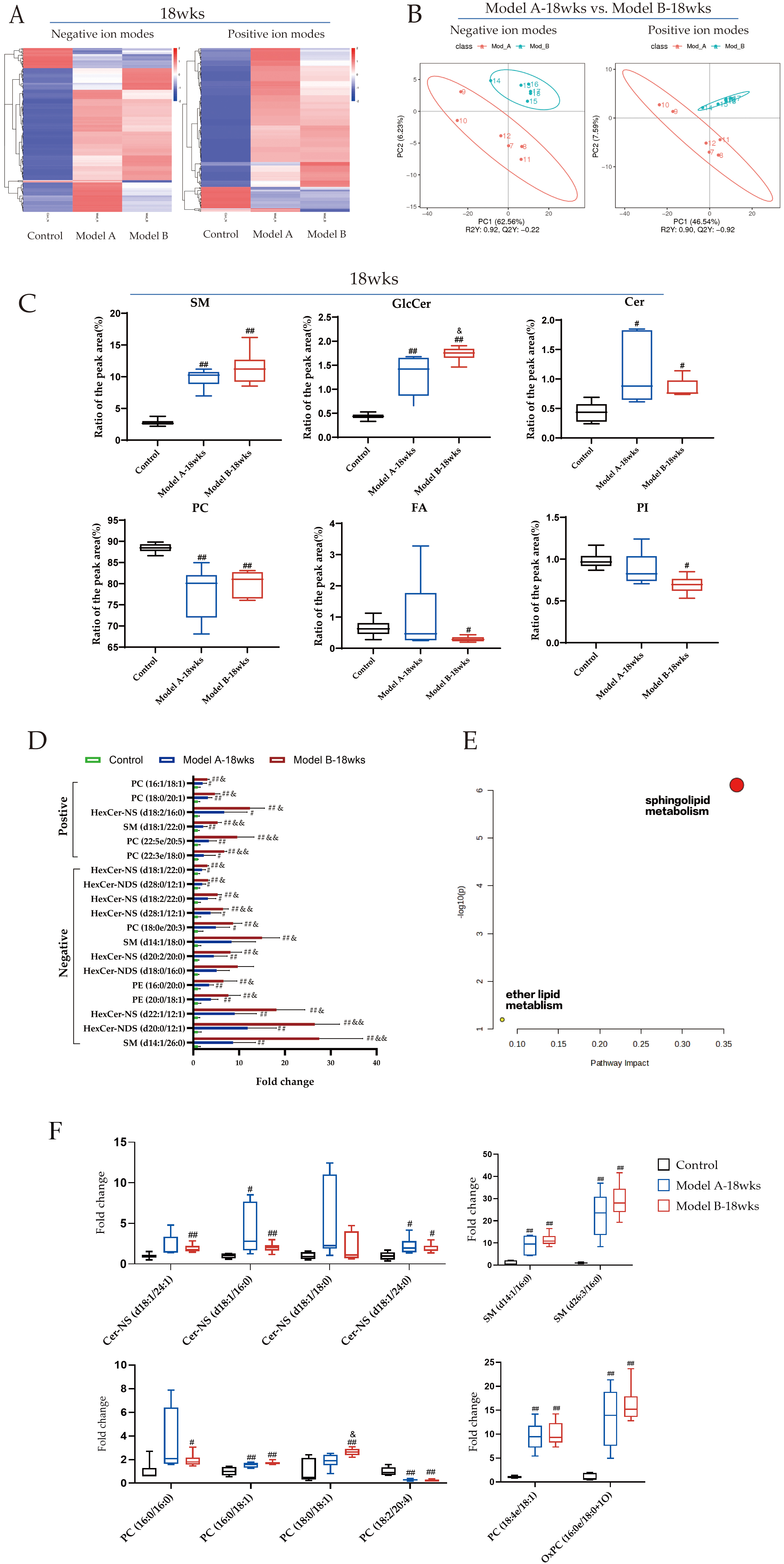 Fig. 5.
Fig. 5.
Co-stimulation altered plasma lipidomic profiles.
(A) Clustering heat maps of hierarchical
clustering analysis (HCA) between three groups. (B) Partial least squares
discriminant analysis (PLS-DA) score plot of Model A-18wks vs. Model B-18wks
group. (C) Ratio of the peak area of lipid subclasses to the plasma lipid
(%). (D)
Potential lipid markers associated with
plaque vulnerability were assessed using the thresholds of variable importance in
projection (VIP)
In line with recent studies [7, 8, 9, 10], the statistical significance of the plasma
lipid variables was determined using PLS-DA based on the criteria of VIP
To determine the potential lipid markers
linked to AS plaque vulnerability, individual molecules showing significant
differences were analyzed. Compared with those in the control and Model A-18wks
group, 19 lipid components were significantly elevated in the Model B-18wks group
(p
Several clinical cohort trials [9, 13] have confirmed that multiple individual
lipid components, including Cer (d18:1/16:0, d18:1/18:0, and d18:1/24:1), HexCer
(d18:1, d18:2, C16–C18, C20–C26), SM-16, and PC (16:0/16:0, 16:0/18:1,
18:0/16:1), are positively associated with the risk of cardiovascular disease
(CVD) outcomes, while lysophosphatidylcholine (LPC) (16:0, 18:0, 18:1, 18:2, and
20:4) is inversely correlated [34]. Additionally, Chen et al. [35]
analyzed plasma lipidomics in C57BL/6J mice on a normal diet, C57BL/6J mice, and
ApoE-/- mice on a 16-week HFD; after controlling for the effects of
the HFD, they found that reductions in C16 sphingosine and PC (17:1/22:6) levels
and increases in PC (18:2/20:4), PC (16:0/16:0), PC (18:0/16:1), SM (d18:1/24:1),
SM (d16:0/28:5), and SM (d18:1/16:0) levels were positively associated with AS.
Therefore, we further assessed these reported cardiovascular risk markers.
Notably, 17 lipid species were consistent with the clinical studies, given that
Cer-NS (d18:1/16:0, d18:1/24:1, and d18:1/24:0), HexCer-NS (d18:1/16:0), SM
(d14:1/16:0 and d26:3/16:0), and PC (16:0/16:0, 16:0/18:1, 18:0/18:1, 18:1/18:1,
and 18:4e/18:1) levels were significantly higher, while LPC (18:2, 18:0, 16:0,
20:4) and PC (18:2/20:4) levels were significantly lower in the two model groups
than in the control group (p
Using 16S rRNA sequencing of the colon contents, we examined the effects of
long-term HFD and short-term co-stimulation on the intestinal microbiota of
ApoE-/- mice. The results are shown in Fig. 6A, where the
operational taxonomic unit rarefaction curve is seen to gradually flatten as the
sequencing volume increases, indicating adequate sequencing data. Compared with
the control group, the results of
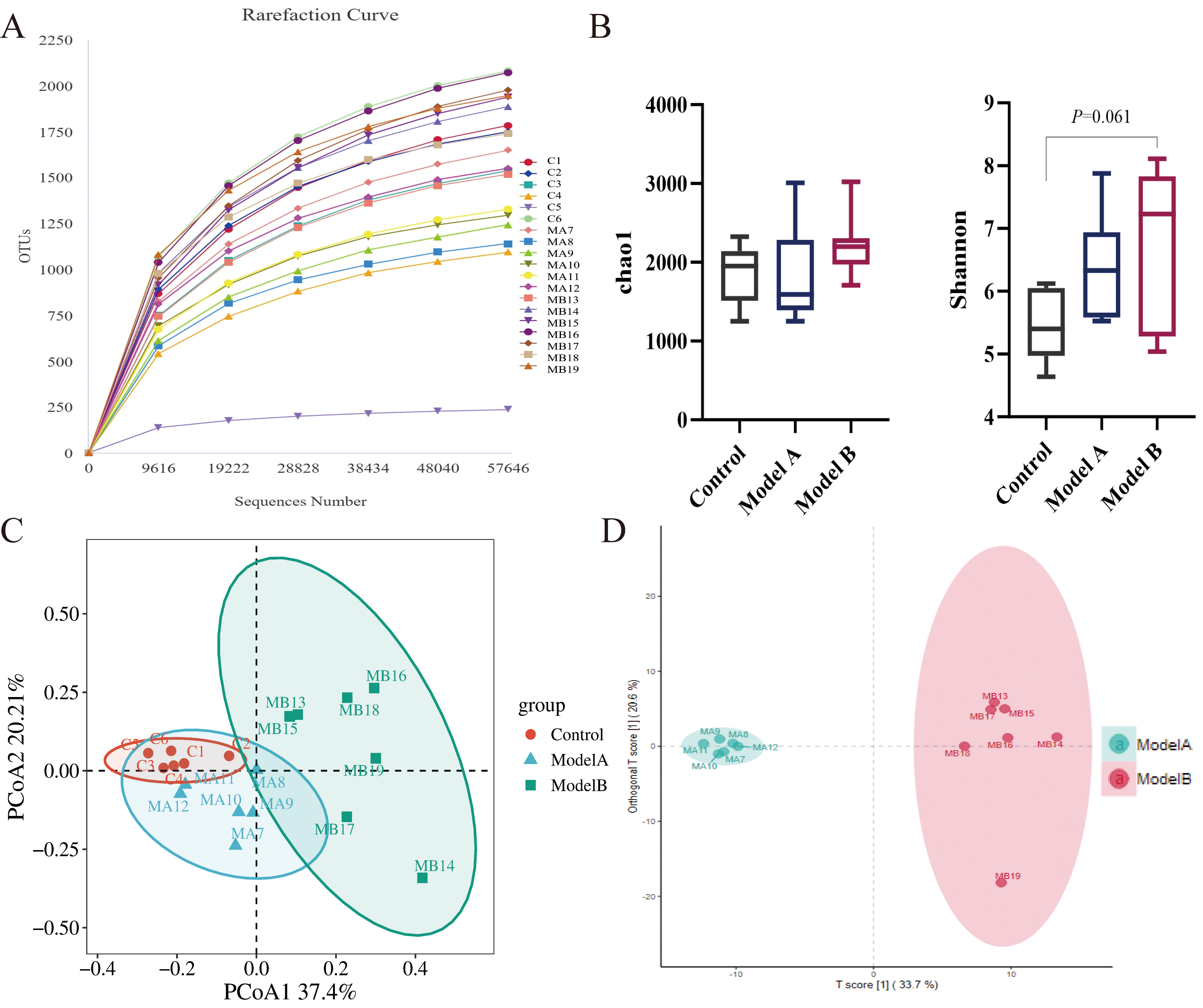 Fig. 6.
Fig. 6.
Combined stimulation increases intestinal microflora disorders.
(A) Operational taxonomic unit rarefaction curve. (B) Chao1 and Shannon indices
reflect the
Subsequently, the community composition and species abundance were examined
across various taxonomic levels. At the phylum level (Fig. 7A,B), the Model
B-18wks group had significantly greater levels of dangerous bacteria
(p_Firmicutes) and significantly lower relative abundances of
beneficial bacteria (p_Bacteroidota) compared to the Model A-18wks
groups (p
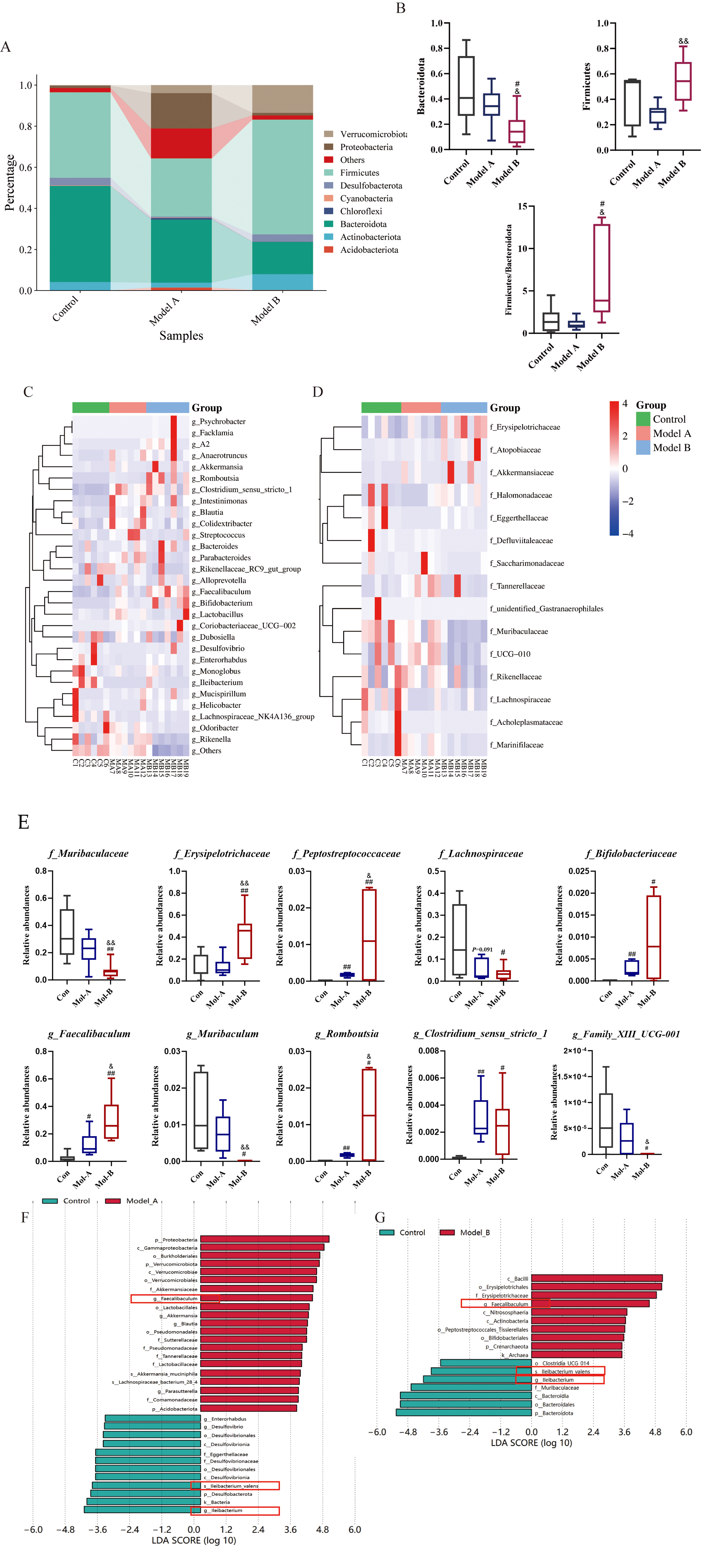 Fig. 7.
Fig. 7.
Combined stimulation alters the dominant microflora composition
from the phylum to the species level. (A) Stacked chart of microbial flora at
the phylum level. (B) Relative abundance of Firmicutes and
Bacteroidota and the ratio of Firmiciutes to
Bacteroidota (F/B). Data are presented as mean
To reproduce the risk of coronary artery disease in humans, we employed short-term stimulation (with a combination of LPS, phenylephrine injection, and cold) to trigger atherosclerotic plaque rupture in HFD-fed ApoE-/- mice. HDF-fed ApoE-/- mice with stimulation had higher TC, TG, and LDL levels, lower HDL levels, and enhanced atherosclerotic plaque growth than HDF-fed ApoE-/- ones. Although brief co-stimulation did not appreciably affect blood lipid levels, it resulted in significant vulnerability to atherosclerotic plaques. Specifically, the model involving co-stimulation and a HFD for 18 weeks exhibited many vulnerable plaque characteristics [41], including an increased vessel area, large plaque size, necrotic core, increased lipid accumulation, a thin or disrupted/buried fibrous cap, decreased total collagen, increased expansion of macrophage cells, decreased SMCs, and increased plaque vulnerability index. Simultaneously, Multilayered subsurface fibrous caps and erythrocyte extravasation were observed in some samples, suggesting prior plaque rupture and subsequent neointimal healing.
Lipid metabolic abnormalities are a hallmark of atherosclerosis, which is a
chronic illness. Adipose tissue dysfunction can induce dyslipidemia, activate
vascular inflammation, and promote atherosclerosis development [42]. Our study
revealed that a HFD markedly increased the adipose tissue-to-body weight ratio
(mg/g), adipocyte hyperplasia, and high PPAR
Lipidomics analysis has the potential for false positives, due to the complexities involved in lipid extraction and characterization. Variability in sample preparation and ionization efficiency can lead to misidentification of lipid species, potentially skewing results. Experts in the field, Skotland et al. (2024) [31], suggested some simple guidelines for assessing the likelihood of lipid species being present. These guidelines for mammalian samples include ① Major lipid species should not contain fatty acyl chains or long-chain bases with an odd number of carbon atoms; ② The major fatty acyl chains in phospholipids and glycerolipids contain 16–22 carbon atoms with minor amounts of C14; ③ Fatty acyl chains of sphingolipids typically contain 14–26 carbon atoms with no or only one double bond; ④ In glycosphingolipids, the double bond is found mainly in fatty acyl chains with 22 carbons or higher. In this study, the data was chosen and examined using the aforementioned standards.
Sphingolipids consist primarily of the most abundant SM (approximately 87%), complex HexCer (9–10%), and Cer (approximately 3%) in human plasma. In a retrospective subcohort (n = 5991) of the LIPID study, higher total Hex-Cers were associated with a significantly higher CVD risk, with HexCer (d18:1/16:0) and HexCer (d18:1/18:0) as high-risk markers [34]. Another study [13] analyzed the plasma lipids of 2627 Singaporeans of Chinese heritage and found that total Cer levels were not associated with CVD risk. However, higher levels of circulating HexCer, total long chain sphingolipids (C16–C18), and complete 18:1 sphingolipids were associated with an increased CVD risk. A few plaque samples from patients who died from atherosclerosis revealed a significant increase in HexCer levels in the intima tissue where the plaque grows compared with those in areas without plaque [43]. These findings reveal that HexCer, similar to Cer, is a high-risk lipid marker for CVDs. Certain sphingolipid species are associated with atherogenesis. Particular attention has been paid to the association between phospholipids and CVD. Accumulation of Cer species is observed in atherosclerotic lesions and obesity and is strongly correlated with diabetes; certain types of Cer species have emerged as predictive biomarkers for future cardiovascular adverse events [44]. In this study, the lipidomics results revealed an apparent elevation of SM, Cer, and HexCer levels induced by HFD, and additional upregulation of HexCer in the lipid composition caused by co-stimulation, revealing the most vital relationship between HexCer species and plaque vulnerability. In addition, analysis of the individual lipid molecules revealed that PC (16:0/16:0, 18:2/20:4, 18:1/18:1), HexCer (C12:1, C16:0), Cer (d18:1/16:0), SM (C16:0), and SM containing long saturated fatty acyl chains may be risk markers for vulnerable plaques.
Mounting evidence suggests that plaque stability may be greatly impacted by the
complex crosstalk between HexCer, PPAR signaling, SCFA metabolism, and adipose
tissue dysfunction [45, 46]. Elevated levels of HexCer have been associated with
insulin resistance and chronic inflammation in adipose tissue, factors that are
pivotal in the progression of metabolic disorders and cardiovascular diseases
[47]. Furthermore, Jang et al. [48] reported that HexCer increased
rosiglitazone-induced PPAR
As bacteria metabolites, SCFAs are essential for adipose tissue metabolism and
anti-atherosclerosis. By controlling the gut microbiota, SCFAs trigger fatty acid
oxidation and glycolysis [49]. Yi et al. [50] reported that SCFAs can
significantly inhibit oxidized low-density lipoprotein (Ox-LDL)-induced
macrophage activation and inflammatory damage by modulating the NOD-, LRR- and
pyrin domain-containing protein 3 (NLRP3) Caspase-1 signaling pathway, suggesting
that Exogenous SCFAs, by regulating immune responses and cellular metabolism, may
provide new therapeutic targets for atherosclerosis. Wang et al. [51]
reported that SCFAs altered the distribution of the lipid in serum and inhibited
adipogenic differentiation by suppressing the function of Cers and further
decreasing PPAR
Increased amounts of circulating lipids deposited in non-adipose tissues, such
as the heart and blood vessel walls, result from metabolic abnormalities in
adipose tissue that impact circulating lipid metabolism [55]. A compelling body
of evidence suggests that Cers may be responsible for a significant portion of
tissue damage in various cardiometabolic disorders. Furthermore, HexCer
accumulation is positively correlated with the number of macrophages in plaques,
and it induces the release of pro-inflammatory cytokines such as tumor necrosis
factor (TNF)-
A novel ApoE-/- model was developed using short-term co-stimulation plus 18 weeks of HFD. The results demonstrated that atherosclerotic plaque vulnerability, adipogenesis disorders, lipid metabolic inflexibility, and harmful microflora increases were similar to those in middle- to late-stage atherosclerosis. This study lays the groundwork for further scientific research and the development of associated drugs. It is evident that Cer and HexCer metabolism can aggravate atherosclerosis progression and increase inflammatory responses; however, to elucidate the underlying mechanisms, further research is required to determine how HexCer is linked to adipose tissue dysfunction, SCFA metabolism, and the PPAR pathway.
HFD, high-fat diet; Cer, ceramide; HexCer, monohexosylceramide; LPC, lysophosphatidylcholine; SM, sphingomyelin; CVD, cardiovascular disease; IL, interleukin; TC, total cholesterol; TG, triglyceride; SCFA, short-chain fatty acid; WAT, white adipose tissue; BAT, brown adipose tissue; QC, quality control; PLS-DA, partial least square discriminant analysis; SMA, smooth actin; LDL, low-density lipoprotein; HDL, high-density lipoprotein; LPS, lipopolysaccharide; HCA, hierarchical clustering analysis; FC, fold change; LEfSe, linear discriminant analysis effect size; H&E, hematoxylin and eosin; FA, fatty acyl; PPAR
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Conceptualisation, JL, and LB; Data curation, GY, QZ, and FL; Funding acquisition, JL; Methodology, JL, YW, and GY; Investigation and Validation, PZ, and XZ; Visualization, GY, QZ; Writing an original draft, GY; Writing review and data curation, XT. All authors contributed to editorial changes in the manuscript. All authors have read and agreed to the published version of the manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The animal study was reviewed and approved by the Biomedical Ethics Committee of Inner Mongolia Medical University (No. YKD2019146). All experiments were performed following the Animal Ethics Code and Operational Guidelines. All efforts were made to minimize animal suffering and discomfort in compliance with the 3R principles.
In addition to acknowledging peer reviewers’ contributions for their thoughts and recommendations, we would like to thank everyone who assisted us in writing this paper.
This research was funded by the National Natural Science Foundation of China (grant no.81960757, 82260813).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/FBL27236.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.







