1 The First Clinical Medical School, Shanxi Medical University, 030001 Taiyuan, Shanxi, China
2 Department of Pediatrics, First Hospital of Shanxi Medical University, 030001 Taiyuan, Shanxi, China
Abstract
Infection with respiratory syncytial virus (RSV) has the potential to exacerbate asthma by causing prolonged inflammation in the airways. Mounting evidence has revealed the significant involvement of programmed cell death protein-1 (PD-1) in the development of asthma. Although icariin (IC) has shown potential in improving airway remodeling in ovalbumin (OVA)-induced asthma, its impact and underlying mechanisms in cases of asthma aggravated by RSV infection are not thoroughly understood.
To explore the effect of IC on RSV-infected asthmatic mice and the mechanism involving PD-1.
A model of asthmatic mice infected with RSV was developed. To evaluate the effects of IC treatment, general behavioral characterization, histopathologic analysis, bronchoalveolar lavage fluid (BALF) analysis, and enzyme-linked immunosorbent assays (ELISA) were performed to assess the frequency of sneezing and nose scratching, the content of OVA-specific IgE, oxidative stress and airway inflammation in mice. Apoptosis was also assessed by terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL). Finally, the expression levels of apoptosis protein, oxidative stress-related protein, and PD-1 were assessed by western blot.
IC significantly ameliorated the sneezing and nose-scratching frequency (p < 0.001) and decreased OVA-specific IgE levels in asthmatic mice infected with RSV (p < 0.01). IC treatment remarkably reduced the infiltration of inflammatory cells around the alveoli and lowered the overall inflammation score. It also notably decreased the levels of inflammatory cytokines interleukin-4 (IL-4), IL-13, and IL-5, and decreased the numbers of neutrophils, eosinophils, and macrophages in the bronchoalveolar lavage fluid (BALF) (p < 0.001). IC ameliorated oxidative stress in RSV-infected asthmatic mice (p < 0.001). In addition, IC reduced apoptosis while increasing PD-1 expression in asthmatic mice infected with RSV (p < 0.001). Interestingly, si-PD-1 significantly reversed IC inhibition of inflammatory cytokines and apoptosis-related proteins, and promoted PD-1 protein expression (p < 0.01). The findings suggested that IC might be effective in alleviating asthma triggered by RSV in mice by regulating the expression of PD-1.
IC ameliorated RSV-induced asthma in mice by regulating PD-1 expression, and may hold promise as a potential therapeutic agent for RSV-induced asthma in mice. These findings provide valuable insights into the possibility of using IC as a treatment option for asthma caused by RSV.
Keywords
- asthma
- respiratory syncytial virus
- icariin
- PD-1
Asthma encompasses a diverse set of chronic respiratory conditions characterized
by varying causes and a range of clinical presentations. The primary symptoms
include coughing, tightness in the chest, difficulty breathing, wheezing, and
additional related signs [1]. In children, severe acute lower respiratory
infections are primarily caused by respiratory syncytial virus (RSV), which is an
enveloped, non-segmented RNA virus with a negative strand. This virus can result
in infant hospitalizations and, in severe cases, fatalities [2, 3]. Previous
research has verified that glucocorticoids and leukotriene modifiers, primarily
used for preventing and treating RSV-induced asthma, are effective in managing
asthma symptoms and decreasing the frequency of sudden flare-ups. Nonetheless,
these medications do not provide a complete cure for asthma and can cause a range
of side effects [4]. Furthermore, increasing evidence indicates that conciliatory
antiallergic decoction reduces pyroptosis in asthmatic mice infected with RSV and
Lipopolysaccharide (LPS)-stimulated 16HBE cells by suppressing the
TLR3/NLRP3/NF-
Icariin (IC), with a molecular formula of C33H40O15, is a monomer of pentenylated flavonoid glycoside derived from Epimedium, a traditional Chinese medicine. This compound, known as icariin, is considered one of the key pharmacologically active components in Epimedium [7]. It exhibits various effects including immune regulation, anti-inflammatory and antioxidative activities, and the suppression of cell apoptosis [8]. In studies related to lung cancer treatment, icariin was observed to impact the PI3K-Akt signaling pathway, decrease the phosphorylation levels of Bad, and other factors, and lower the mitochondrial membrane potential to facilitate tumor cell apoptosis [9]. Icariin, a flavonoid compound, shows significant resistance to oxidative stress when used to treat myocardial damage and heart failure [10]. Moreover, it has the capability to bond with inorganic substances like hydroxyapatite, creating a novel drug scaffold that enhances bone damage repair [11]. Furthermore, in a study with asthmatic mice induced by ovalbumin (OVA), conducted by Hu et al. [12], it was revealed that icariin intervention to significantly reduce the expression of interleukin-13 (IL-13), endothelin-1 (ET-1), and other vital cytokines involved in airway remodeling. This intervention also suppressed the activity of the MAPK/Erk signaling pathway, thereby inhibiting the proliferation of airway smooth muscle cells (AMSCs) and contributing to the enhancement of the airway remodeling process through the influence on multiple targets [12]. However, there is limited understanding of IC’s role in the exacerbation of RSV-induced asthma and the corresponding mechanisms.
Programmed cell death protein-1 (PD-1) serves as a crucial regulator of the immune response, playing a significant role in the effector functions of T cells [13]. PD-1 plays various roles in the regulation of host immunity, including the modulation of T cell activation, the priming of T cells, and the facilitation of effector functions during the initial phases of the T cell response [14, 15]. Research demonstrated that in neutrophilic asthma, PD-1 induction mainly occurs on T helper cells, and PD-1 knockdown led to exacerbated neutrophilic airway hyperreactivity (AHR) and lung inflammation [13]. Furthermore, Song et al. [16] demonstrated that IC exhibited anti-tumor properties by modifying the immune microenvironment in models of breast cancer. Specifically, IC reduced PD-L1 expression in 4T1 tumors [16]. PD-ligand 1 (PD-L1) is a negative regulatory molecule present on the surface of cancer cells. By binding to PD-1 on immune cells, it obstructs immune system activation, leading to a detrimental role. While earlier research has indicated potential protective effects of icariin against respiratory and immune system diseases [16, 17], but its specific role in modulating RSV-induced asthma through the PD-1 remains unclear.
Thus, we aim to investigate how IC impacts sneezing, nose scratching, airway inflammation, oxidative stress, apoptosis-related proteins, and PD-1 protein expression in mouse models of asthma induced by OVA and RSV. This research could enhance our understanding of how IC could be used to treat acute asthma exacerbated by RSV infection.
Thirty 6-week-old female BABL/c mice with SPF condition, weighing 20
The research found that ribavirin + budesonide has therapeutic efficacy for
asthma [5]. The BABL/c mice were randomly assigned to six groups, each consisting
of five mice: the asthma model (M) group, the normal control (NC) group, the
ribavirin + budesonide (PC) group serving as the positive control, and three
groups receiving different doses of IC (GN10278, GLPBIO, Montclair, CA, USA): low (IC-L), medium
(IC-M), and high (IC-H) dose. The asthma model in these mice was created based on
previously described methods with some modifications [18]. With the exception of
the NC group, all mice in the remaining groups were sensitized on day 1 and day
14 by intraperitoneal injection of 100 µg ovalbumin (OVA, S7951, Sigma,
Saint Louis, MO, USA) and 2 mg aluminum hydroxide (239186, Merck, Germany)
dissolved in 200 µL of physiological saline. During days 15–49, the mice
were exposed every other day with a 20-minute exposure to aerosolized 0.99% OVA
in saline. Additionally, on days 21, 35, and 49, the mice were intranasally given
50 µL RSV (1
Following euthanasia, blood samples from the abdominal aorta. The blood samples were spun in a centrifuge at a speed of 2700 g for 15 minutes at a temperature of 4 °C to separate and analyze the inflammatory factors present in the serum. Ultimately, the concentrations of OVA-specific IgE, malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH) were measured using ELISA kits (OVA-specific IgE, ml037618, Enzyme-linked Biotechnology Co., Ltd. Shanghai, China; MDA, SP30131; SOD, SP14390; CAT, SP14946; GSH, SP14856, Saipei Biotechnology Co., Ltd., Wuhan, China). The concentrations of IL-13, IL-4, and IL-5 were assessed using Beyotime provided kits (IL-13, PI539; IL-4, PI612; IL-5, PI620, Shanghai, China).
Lung tissues from each mouse were excised and weighed. The left lung tissues
were then fixed in a 4% paraformaldehyde solution (41678, Acros Organics,
Brussels, Belgium) for 24 hours before being processed in paraffin for further
processing. Subsequently, the preserved lung tissues were sliced into sections
that were 3 micrometers thick. These sections were then stained using hematoxylin
and eosin (HE, GF1010, Shanghai G
After euthanizing the mice, their lungs were rinsed with cold PBS. The resulting BALF was then spun at 2700 g for 10 minutes at 4 °C. The liquid part was separated and kept at –80 °C for later analysis. To count the cells, the samples were stained with Wright-Giemsa dye from Nanjing Jiancheng Bioengineering Institute (D010-1-1, Nanjing, China). The quantity of inflammatory cells, such as neutrophils, eosinophils, and lymphocytes, was then determined using a light microscope (XSP-17C, Leica, Germany).
Both the PD-1-specific siRNA and its corresponding control siRNA were designed and synthesized by HUAGENE (Shanghai, China). The sequences for si-PD-1 was 5′-CATTCACTTGGGCTGTGCTGCAGTT-3′. Lipofectamine 3000 (L3000015, Invitrogen, Mountain View, CA, USA) served as the transfection reagent, while si-NC acted as the control. The sequence of si-NC was 5′-CTACTTACTGCGTGGTTCGGCTTGA-3′. The si-NC and si-PD-1 were injected via the tail vein separately for 48 hours. The injection frequency was once every two days, and after three repetitions of siRNA treatment, PD-1 knockdown was assessed by reverse transcription quantitative polymerase chain reaction (RT-qPCR) and western blot.
To extract total protein from 100 mg of right lung tissue, the tissue was
treated with cold-RIPA lysis buffer. The amount of protein was then measured
using a bicinchoninic acid (BCA) reagent kit (P0010S, Beyotime, Shanghai, China).
Following this, protein samples with uniform concentrations were separated
through 10~15% SDS-PAGE (P0012A, Beyotime, Shanghai, China).
Upon transfer to the PVDF membranes (FFP19, Beyotime, Shanghai, China) provided
by Millipore in the USA, the membranes were blotted and subjected to overnight
incubation with the primary antibodies (Bax, ab53154, Abcam, 1:1000; Bcl-2,
ab194583, Abcam, 1:2000; Cleaved-Caspase 3, ab2302, Abcam, 1:2000; Nrf2, AF0639,
Affinity, 1:2000; HO-1, AF5393, Affinity, 1:1000; PD-1, ab89828, Abcam, 1:1000;
Lamin B1, ab65986, Abcam, 1:5000;
Following the manufacturer’s guidelines, the reaction system was generated using TB Green® Premix Ex TaqTM Kit (RR071A, BIOSCIENCE, Boston, MA, USA). The prepared mixture was reacted in the Real-Time PCR System (Roche, Shanghai, China) with the reaction program consisting of an initial denaturation at 97 °C for 4 minutes, followed by 38 cycles of denaturation at 97 °C for 4 seconds and annealing/extension at 60 °C for 30 seconds. All measurements were performed in triplicate to eliminate operational errors. The primers sequences listed in Table 1. The relative expression levels of the genes were calculated using the 2-ΔΔCt method.
| Primer names | Forward primer (5′-3′) | Reverse primer (5′-3′) |
| Nrf2 | ACCGGAGAATTCCTCCCAAT | AGCTCCTGCCAAACTTGCTC |
| HO-1 | AGACCGCCTTCCTGCTCAACAT | TCTGACGAAGTGACGCCATCTGT |
| Bax | AGGCCTCCTCTCCTACTTCG | CCTTTCCCCTTCCCCCATTC |
| Bcl-2 | AACATCGCCCTGTGGATGAC | AGAGTCTTCAGAGACAGCCAGGAG |
| Cleaved caspase-3 | CTCGCTCTGGTACGGATGTG | TCCCATAAATGACCCCTTCATCA |
| PD-1 | ATCTACCTCTGTGGGGCCAT | GAGTGTCGTCCTTGCTTCCA |
| b-actin | GCAGGAGTACGATGAGTCCG | ACGCAGCTCAGTAACAGTCC |
| Lamin B1 | AAGGCTCTCTACGAGACCGA | TCCTTCTTAGCATAATTGAGCAGC |
Note: Bax, Bcl-2 associated X Protein; Bcl-2, B-cell lymphoma-2; PD-1, programmed cell death 1; Nrf2, nuclear factor-erythropoietin 2-related factor 2; HO-1, heme oxygenase-1.
The data were presented as mean
The mice belonging to the M group exhibited elevated instances of sneezing and
nose-catching associated with asthma compared to mice in the NC group (Fig. 1A,B)
(p
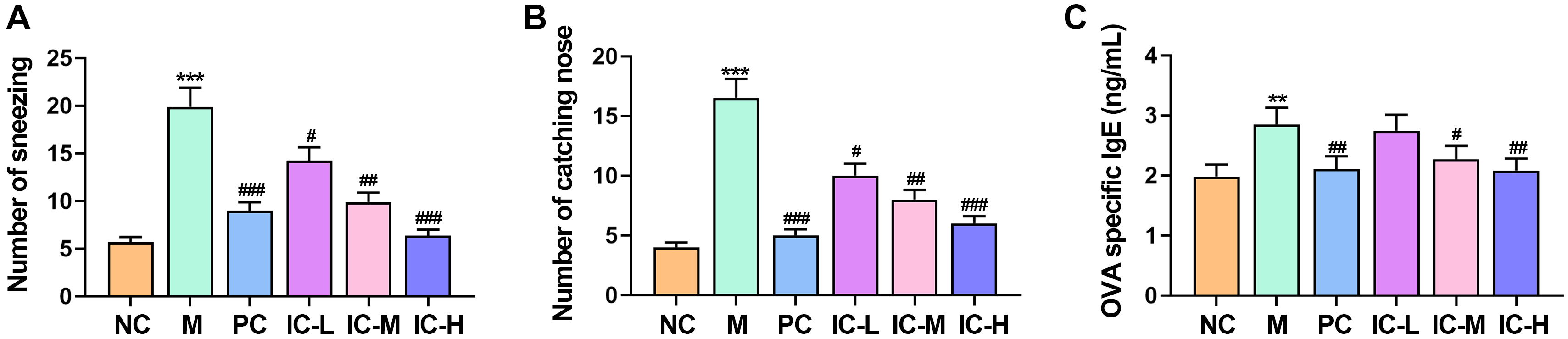 Fig. 1.
Fig. 1.
Effect of the icariin on the overall behavior of mice with
asthma induced by respiratory syncytial virus (RSV) was examined. The number of
sneezing (A), and catching nose (B) of the
mice. (C) The contents of ovalbumin (OVA)-specific IgE in serum were detected by
Enzyme-Linked Immunosorbent Assay (ELISA). n = 5. **p
A predominant clinical characteristic of asthma is the excessive accumulation of
inflammatory cells in lung tissue. Subsequently, lung tissue
from asthmatic mice was gathered for HE staining. HE staining showed a noticeable
increase in inflammatory cells around the alveolar walls compared to the NC group
(p
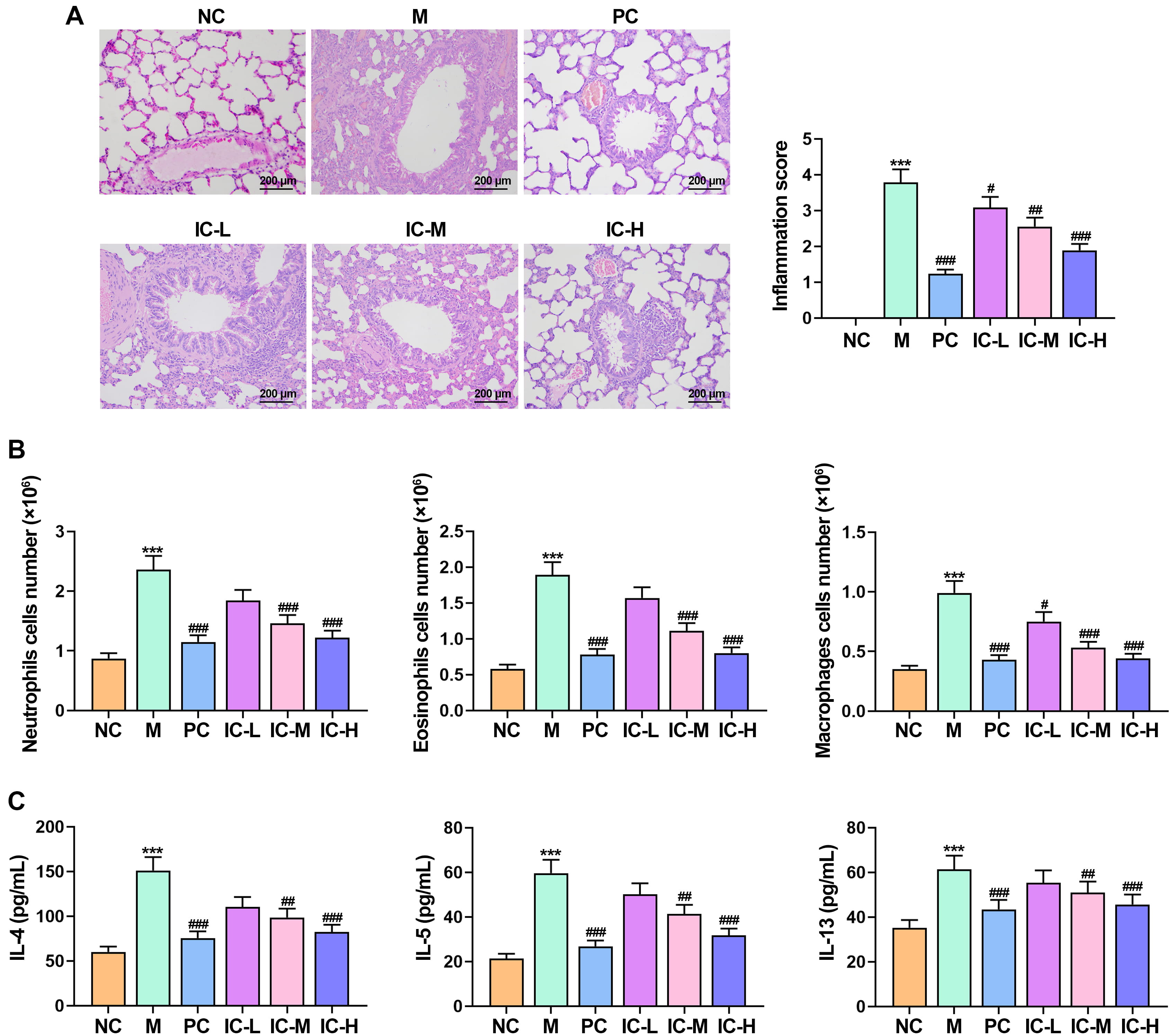 Fig. 2.
Fig. 2.
Effect of the icariin (IC) on inflammatory cells in RSV-infected
asthmatic mice. (A) Histopathological changes were assessed using HE staining of
mouse lung tissue. Bar = 200 µm. (B) The cell numbers of neutrophils,
eosinophils, and macrophages in bronchoalveolar lavage fluid (BALF) of mice in
each group. (C) Inflammatory cytokines IL-4, IL-13, and IL-5 in BALF
were measured using ELISA. n = 5. ***p
To further analyze the effect of IC on oxidative stress in RSV-induced asthma
mice, we examined the levels of MDA, GSH, SOD and CAT in lung homogenates. The
results from the ELISA analysis revealed that MDA level of RSV-induced asthma
mice was markedly elevated, and GSH, SOD and CAT was markedly reduced compared
with that in NC group (Fig. 3A) (p
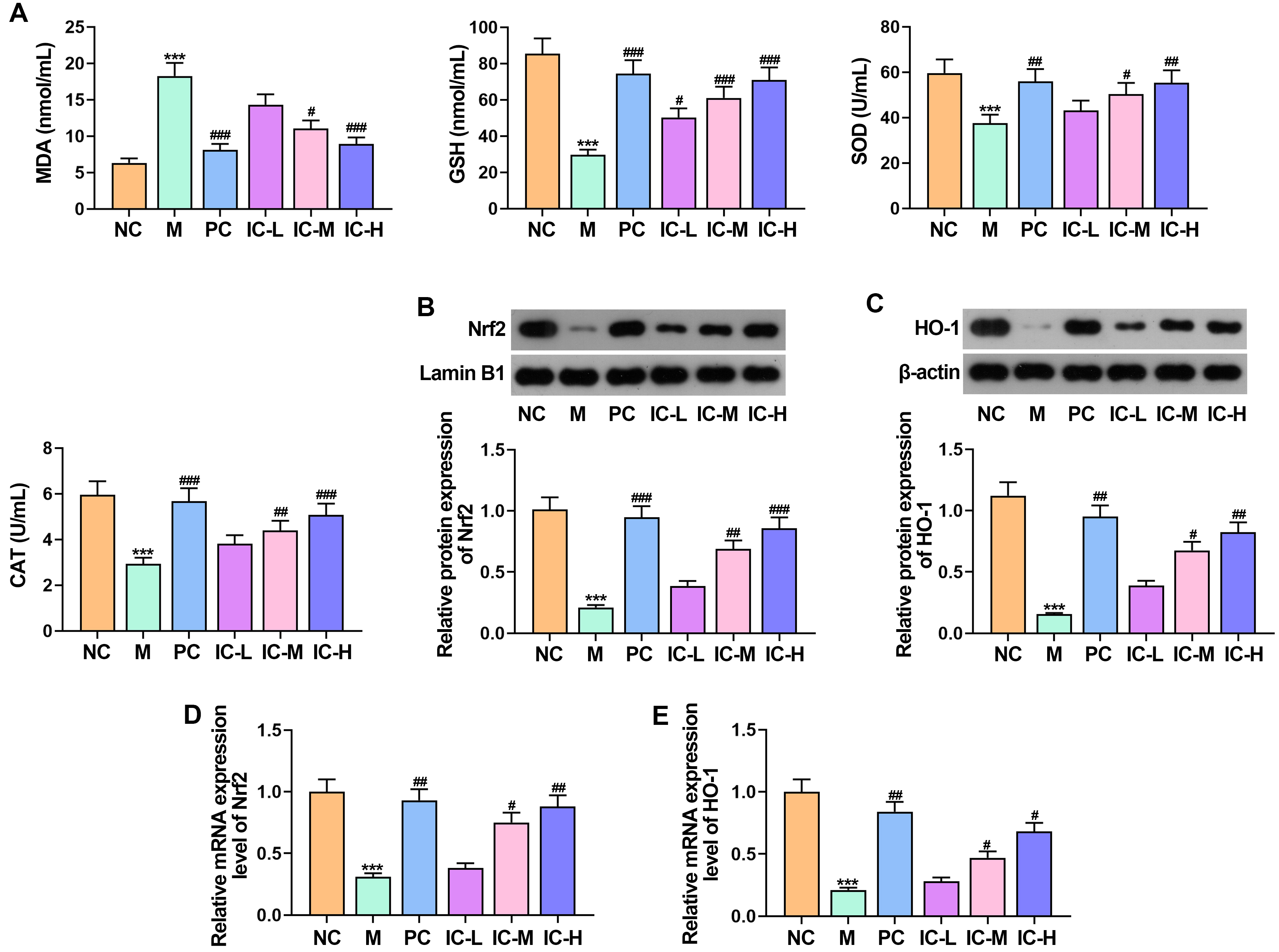 Fig. 3.
Fig. 3.
Effect of the IC on oxidative stress in asthmatic mice induced
by RSV. (A) The levels of MDA, SOD, CAT and GSH in lung tissue homogenates were
detected by ELISA. (B,C) Western blot was conducted to investigate the expression
levels of Nrf2 and HO-1. (D,E) RT-qPCR was used to assess the
mRNA levels of Nrf2 and HO-1. n = 5. ***p
In the control group of NC mice, a limited number of TUNEL-positive cells were
detected. Contrastingly, a notable rise in TUNEL-positive cells was observed in
the M group, which was substantially reduced by the treatment with IC or PC (Fig. 4A) (p
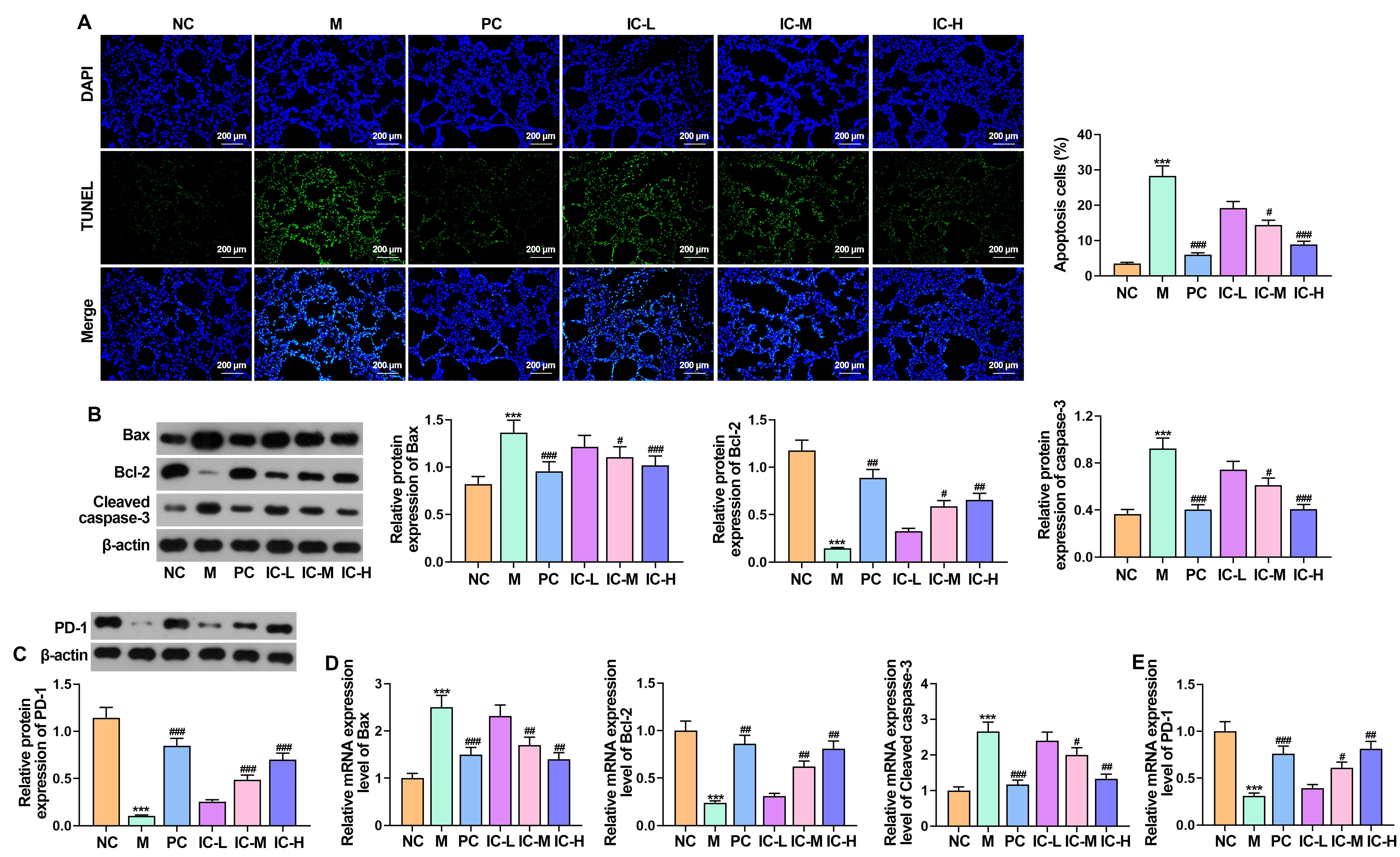 Fig. 4.
Fig. 4.
Effect of the IC on apoptosis and the expression of PD-1 in
asthmatic mice infected with RSV. (A) The number of apoptotic cells in lung
tissue by TUNEL assay. Bar = 200 µm. (B) The expression of Bax, Bcl-2 and
cleaved caspase-3 was detected by Western blot. (C) The expression of PD-1 in
lung tissue was detected by Western blot. (D,E) RT-qPCR was used to determine the
mRNA levels of Bax, Bcl-2, cleaved caspase-3 and PD-1. n = 5.
***p
PD-1 plays an important role in asthma, and its activation can reprogram T cells
to alleviate asthma. To this end, we further analyzed whether the improvement of
RSV-induced asthma by IC is related to PD-1 [13]. Compared with NC group, the
protein and mRNA expression of PD-1 was markedly reduced in the M group
(p
To further determine whether IC ameliorates RSV-induced asthma in mice by
modulating PD-1 expression. RT-qPCR results showed that the expression of si-PD-1
was significantly lower than that of si-NC, indicating that the si-PD-1 construct
was successful (Fig. 5A) (p
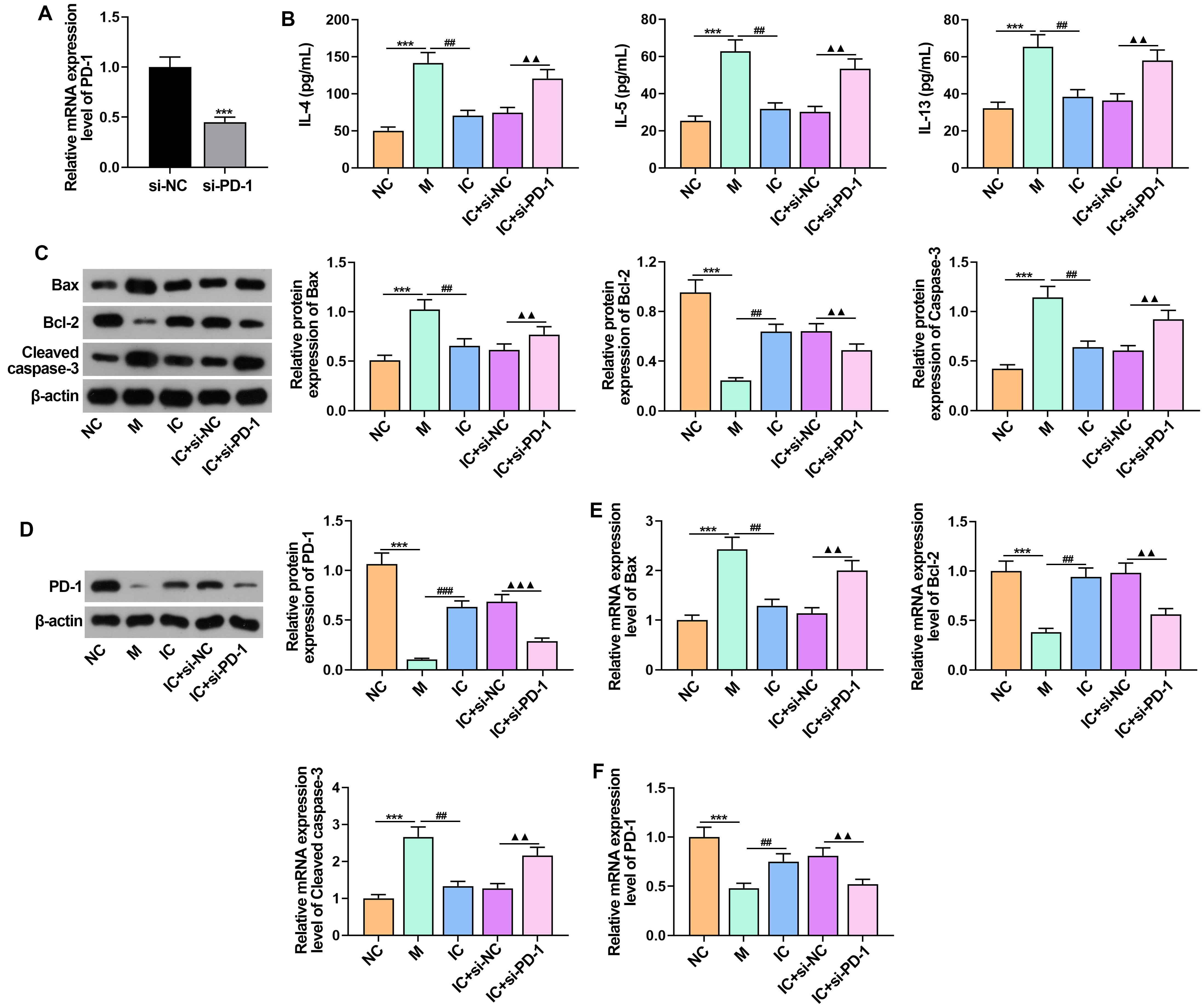 Fig. 5.
Fig. 5.
IC affects RSV-induced asthma in mice by modulating PD-1
expression. (A) Transfection efficiency of PD-1 knockdown was verified
using RT-qPCR. (B) Inflammatory cytokines IL-4, IL-5, and IL-13 in BALF were
determined by ELISA. (C) The expression of Bax, Bcl-2 and cleaved caspase-3 was
detected by Western blot. (D) Western blot detected the expression of PD-1 in
lung tissue. (E,F) The mRNA levels of Bax, Bcl-2, cleaved caspase-3 and
PD-1 was detected using RT-qPCR. n = 5. ***p
In addition, the mRNA and protein expression of cleaved caspase-3 and Bax was
markedly reduced and Bcl-2 expression was markedly increased after IC treatment
(Fig. 5C,E). Knockdown of PD-1 reversed the inhibitory effect of IC on Bax and
cleaved caspase-3, as well as the promotional effect on Bcl-2 (Fig. 5C,E).
Besides, the results showed that asthmatic mice infected with RSV had an
increased in PD-1 after IC treatment. Conversely, si-PD-1 treatment reversed PD-1
mRNA and protein levels in asthmatic mice (Fig. 5D,F) (p
A prevalent respiratory disease worldwide is asthma, which leads to substantial illness and death [21]. Icariin, derived from Epimedium, has garnered considerable attention for its clinical applications due to its high safety profile and its effectiveness in anti-inflammatory, antioxidant, and immune regulation [6]. Clinical research has confirmed that IC to control the expression of eosinophils and basophils, thereby delaying the progression to asthma [22, 23]. It is a beneficial treatment for asthmatic mice infected with RSV; however, its precise mechanism is still unknown. Our study demonstrates that IC can ameliorate asthmatic symptoms in mice by diminishing oxidative stress, airway inflammation, and apoptosis in asthmatic mice infected with RSV. Importantly, our research suggests that this effect might be associated with the modulation of PD-1 expression in lung tissue.
In this research, we explored the effect of IC on mice with asthma. Asthma mouse models were established through RSV nasal infusion and OVA sensitization. Subsequently, it was observed that IC enhanced the general conditions of the asthma model mice, as indicated by reductions in sneezing and nose rubbing incidents. Additionally, IC notably decreased the levels of OVA-specific IgE. pathological examination showed that the lung tissue of RSV-infected asthmatic mice had a large number of inflammatory cells, and their airway walls were thickened. Treatment with IC significantly reduced the thickening of the airway walls and the number of inflammatory cells around the airways, as confirmed by lower inflammation scores. Besides, the secretion of IL-13, IL-5, and IL-4 is pivotal in instigating and advancing chronic airway inflammation in asthma [24, 25]. It’s noteworthy that these cytokines are thought to play roles in attracting eosinophils, causing epithelial cell apoptosis, increasing mucus production, promoting the buildup of extracellular matrix, and causing an overgrowth of goblet cells [26, 27, 28]. In line with our earlier discoveries, the current results indicate a notable increase in inflammatory cells, especially neutrophils, lung inflammation and decreased the number of inflammatory cells in the BALF. Additionally, significant increased levels of IL-4, IL-13, and IL-5 in the BALF was observed in the serum of RSV-infected asthma mice.
Extensive evidence suggested that in asthma, certain antioxidants such as GSH,
CAT, and SOD levels are reduced, while the MDA level is elevated in the
hippocampus of asthmatic rats [29, 30, 31]. The administration of Myrtenol led to
increased SOD levels and reduced asthma-induced oxidative stress [30]. Earlier
research has proposed that icariin, as a flavonoid compound, demonstrates
significant capability in combating oxidative stress [10]. Of note, the impact of
Icariin on oxidative stress in asthma model rats has not been documented. This
study demonstrated a substantial reduction in tissue MDA levels following IC
treatment compared to the M group, while the levels of GSH, SOD, and CAT showed
significant increases. IC mitigated oxidative stress in RSV-induced asthmatic
mice. Nrf2, a transcription factor, is accountable for overseeing the synthesis
of internal antioxidants during oxidative stress [32]. Additionally, HO-1 serves
as the primary target protein of Nrf2 [33]. It has been documented that CRE
boosts the expression of antioxidant proteins such as HO-1 and Nrf2, offering
protection against OVA-induced asthmatic inflammation and oxidative stress [34].
Moreover, in a prior investigation, it was shown that IC controlled
carrageenan-induced paw edema and acute inflammation via the heme oxygenase-1
(HO-1)/Nrf2 and NF-
Apoptosis is a process that requires energy and involves the systematic breakdown of a cell. This process is characterized by the fragmentation of the cell’s nucleus, shrinking of the cell membrane, formation of small bulges or blebs on the plasma membrane, and the condensation of nuclear chromatin [36]. It is widely recognized that epithelial cell apoptosis may serve as a potential mechanism for asthma [37, 38]. To further investigate how IC affects apoptosis in lung tissue, a TUNEL staining analysis was performed on the lung tissues of mice with asthma. The results revealed that CAD treatment suppressed the number of apoptotic airway cells in the lung tissues compared to the untreated asthmatic mice, as indicated by the TUNEL staining. The signaling pathway involving cleaved caspase-3, Bax and Bcl-2 is well-known for its role in regulating cell death and survival in different diseases, including asthma [39]. Bcl-2 functions as an inhibitor of apoptosis, while Bax acts as a promoter of intrinsic programmed cell death, exerting a pivotal role in either inhibiting or promoting this process. Additionally, Caspase-3, a crucial protease, is closely associated with carrying out the final stage of apoptosis. A previous study has shown that, Zou et al. [40] observed that treating bronchial epithelial cells with Panax notoginseng saponins R1 significantly reduced the apoptosis caused by dexamethasone. Our current study is in accordance with previous research, as it demonstrates a notable decrease in the levels of Cleaved caspase-3 and Bax proteins and an increase in Bcl-2 expressions, as well as suppressed airway cell apoptosis in lung tissues in asthmatic mice following IC treatment.
PD-1 plays a vital part in regulating immune responses during viral infections and may represent a therapeutic target for augmenting antiviral immunity [41]. In this study, IC treatment increased the expression of PD-1 in asthmatic mice infected with RSV. The increase in PD-1 expression might contribute to the suppression of excessive immune responses and reduction of inflammation, which are critical in managing asthma exacerbations triggered by viral infections. The question of whether Icariin can alleviate RSV-induced asthma in mice by regulating PD-1 expression still lacks sufficient evidence and requires further investigation. Therefore, in this study, si-PD-1 significantly reversed the inhibitory effects of IC on inflammation and apoptosis, as well as the stimulatory effects on PD-1. These findings address the gap in our understanding of how icariin-mediated PD-1 modulation can improve RSV-induced asthma in mice.
In summary, our findings indicated that IC ameliorated RSV-induced asthma in mice by regulating PD-1 expression, and may hold promise as a potential therapeutic agent for RSV-induced asthma in mice. These findings provide valuable insights into the potential use of IC as a treatment strategy for RSV-induced asthma.
The data analyzed was available on the request from the corresponding author.
JYF and XHW designed the research study and wrote the first draft. JYF and XHW performed the research. JYF and XHW analyzed the data. Both authors contributed to editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All experimental protocols of this study were approved by ethics committee of Shanxi Medical University Ethics Committee (No: NO.DWYJ-2022-001). Animal experiments were conducted according to the “Guide for the Care and Use of Laboratory Animals” issued by the US National Institutes of Health as guidelines.
Not applicable.
This study is supported by the Basic Research Program (Free Exploration category) of Shanxi Province in 2023 (No. 202303021221215), and the National Natural Science Foundation Project (No. 82370024).
The authors declare no conflict of interest.
The deepL and Yodoto checking grammars are used in three parts of this manuscript, including 2.1, 2.2 and 2.4. During the preparation of this work, the authors used deepL and Yodoto to check spelling and grammar. After using this tool, the authors reviewed and edited the content as needed and took full responsibility for the content of the publication.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





