1 Department of Otorhinolaryngology, Head and Neck Surgery, The First Affiliated Hospital of Zhengzhou University, 450052 Zhengzhou, Henan, China
2 Department of Otorhinolaryngology, Head and Neck Surgery, The Second Affiliated Hospital of Zhengzhou University, 450000 Zhengzhou, Henan, China
Abstract
Disruption of the blood labyrinth barrier (BLB) is considered a pathological cause of diverse hearing impairments. Perivascular-resident macrophage-like melanocytes (PVM/Ms) play a critical role in maintaining inner ear homeostasis and BLB integrity. Activation of PVM/Ms leads to decreased production of pigment epithelium-derived factor (PEDF), contributing to BLB breakdown. This study investigated the role of the adenosine A2A receptor (A2AR) pathway in lipopolysaccharide (LPS)-induced inflammation in PVM/Ms and elucidated the underlying mechanisms.
The anti-inflammatory effects of adenosine and its specific receptor A2AR were evaluated in LPS-induced PVM/Ms. The levels of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and tissue inhibitor of matrix metalloproteinase 1 (TIMP-1) were measured by quantitative real-time PCR (qRT-PCR) and enzyme linked immunosorbent assay (ELISA). Additionally, matrix metalloproteinase-9 (MMP-9) and PEDF were quantified using western blot and ELISA, respectively. An endothelial cell (EC)-PVM/M co-culture model exposed to LPS was established and treated with adenosine and SCH58261 to assess effects on BLB permeability.
LPS treatment significantly changed the production of inflammatory factors, including IL-6 and TNF-α, as well as MMP-9, TIMP-1, and PEDF. These changes were abrogated by adenosine, which also reduced the production of reactive oxygen species (ROS) and inhibited the activation of PVM/Ms. SCH58261 partially reversed the effects of adenosine following LPS treatment. The p38 MAPK pathway was found to be involved in adenosine regulation of LPS-induced PVM/Ms.
Adenosine attenuates the inflammatory activation of PVM/Ms and enhances their ability to maintain endothelial barrier integrity by binding to A2AR. The findings support adenosine and A2AR as potential therapeutic targets for treating hearing impairments.
Keywords
- adenosine
- adenosine A2A
- lipopolysaccharides
- stria vascularis
- cochlea
The cochlear blood labyrinth barrier (BLB) tightly regulates the cochlear micro-environment for auditory function [1, 2, 3, 4, 5]. Disruption of the BLB is strongly associated with a number of hearing disorders, including noise-induced hearing loss and age-related hearing loss [1]. Progress has recently been made in detailing the structural complexity of this barrier. This has highlighted the critical role of accessory cells in BLB integrity, such as perivascular-resident macrophage-like melanocytes (PVM/Ms) [1, 2]. The authors previously demonstrated that PVM/Ms are activated in animals exposed to loud sound, in aged animals, and in animals with induced inflammation [3, 4, 5]. Previous research has also demonstrated that lipopolysaccharide (LPS)-induced PVM/M activation, through its induction of cytokine production, affects the expression of tight junction-associated proteins (zonula occludens-1 [ZO-1], occludin, and vascular endothelial cadherin) and is one of the main causes of BLB disruption [5].
LPS-induced damage of the blood brain barrier (BBB) is associated with the
expression of pro- and anti-inflammatory cytokines, including interleukin-1
(IL-1), reactive oxygen species (ROS), and tumor necrosis factor-
Adenosine is an effective endogenous immunomodulator and plays a key role in the functional regulation of various cells and molecules in the central nervous system (CNS) [14]. Adenosine receptors consist of four subtypes: adenosine A1 receptor, adenosine A2A receptor (A2AR), adenosine 2b receptor (A2BR) and adenosine A3 receptor, all of which belong to the G protein coupled receptor family [15, 16]. Adenosine regulates various cellular functions, such as inflammation, angiogenesis, and wound healing, by interacting with corresponding adenosine receptors on the cell surface. Among these, the balance of A2AR is also considered to be necessary for oxidative stress in the cochlea, which is the most common underlying mechanism of inner ear pathology [15, 17]. In recent years, the potent anti-inflammatory effects of adenosine have attracted increasing attention. Activation of A2AR has been shown to play a critical anti-inflammatory role in maintaining blood retinal barrier homeostasis by decreasing microglial activation and reducing inflammatory cytokine levels [18]. However, to the best of our knowledge, the role of adenosine in regulating the integrity of the BLB has yet to be investigated.
The present study demonstrates that adenosine plays a central role in LPS-induced BLB dysfunction. We examined the effect of adenosine on PVM/Ms in LPS-induced BLB injury, as well as the underlying mechanism. The findings presented herein may inform more suitable drug therapeutic strategies for the management of hearing loss.
C57BL/6 mice aged 10–15 days were obtained from inbred C57BL/6J mice purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). The protocol for the care and use of these animals was approved by the Ethics Committee of the First Affiliated Hospital of Zhengzhou University (2024-KY-0035-002).
Mice were anesthetized by intraperitoneal injection of tribromoethanol (0.2 mL/10 g body weight of 1.25% solution, TG-Avertin-M, Tigergene, Wuxi, Jiangsu, China) and then euthanized by cervical dislocation. The protocols for generating the cell lines used in these experiments were reported previously [2, 19]. All cell lines were validated by STR profiling and tested negative for mycoplasma. Purified primary PVM/Ms or endothelial cells (ECs) were pretreated with 1 µM adenosine (T0853, TopScience, Shanghai, China) for 1 h, then with 1 µg/mL LPS (L4391, Sigma-Aldrich, St. Louis, MO, USA) for 24 h. PVM/Ms or ECs were treated with 1 µM SCH58261 (T6979, TopScience, China) for 1 h to inhibit A2AR activity. For the suppression of p38 mitogen-activated protein kinases (MAPK) activity, PVM/Ms or ECs were treated with 10 µM SB203580 (S1076, Selleck, Houston, TX, USA) for 30 min.
The cytotoxicity of 1 µM adenosine (with or without 1 µM SCH58261) for PVM/Ms was measured within 48 h of treatment by cell counting kit-8 assay (CX001S, Cellorlab™, Shanghai, China). PVM/Ms were seeded into 96-well plates and treated with LPS (1 µg/mL) for different times, then incubated with 10 µL of CCK-8 solution at 37 °C and 5% CO2 for 3 h. The absorbance of surviving cells was measured at 450 nm using a microplate reader.
The procedure was reported previously [20]. Briefly, total RNA (300–500 ng) was extracted from different groups of PVM/Ms using an RNA extraction kit (RNAiso Plus 9108, Takara, Kusatsu, Japan) and quantified with an ND-2000 NanoDrop spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA). Total RNA (500 ng) from each sample was then reverse-transcribed using an all-in-one, 1st-strand cDNA synthesis supermix (E047, Novoprotein, Beijing, China). Following cDNA synthesis, quantitative real-time PCR (qRT-PCR) was performed using SYBR qPCR Supermix Plus (E096, Novoprotein, China). The real-time PCR cycling conditions were: 50 °C for 2 min, 95 °C for 10 min, 40 cycles of 95 °C for 15 s, and 60 °C for 1 min. Triplicate measurements were made for each gene. GAPDH was used as the internal reference gene, and the 2-ΔΔCT method was used to calculate the relative expression of mRNA. The primer sequences are shown in Table 1.
| Gene | Sequence (5′-3′) |
| IL-6 | F: TTCTTGGGACTGATGCTG |
| R: CTGGCTTTGTCTTTCTTGTT | |
| TNF- |
F: ACGGCATGGATCTCAAAGAC |
| R: AGATAGCAAATCGGCTGACG | |
| TIMP-1 | F: TCTGGCATCCTCTTGTTGCTAT |
| R: CCACAGCGTCGAATCCTT | |
| PEDF | F: TCGCATAGACCTTCAGGAGATTA |
| R: ATCAGAGTCCAAGCCATATCGTA | |
| GAPDH | F: GTGAGGCCGGTGCTGAGTAT |
| R: GTGCAGGATGCATTGCTGAC |
qRT-PCR, quantitative real-time PCR; IL-6, interleukin-6; TNF-
Total protein was extracted from PVM/Ms in different groups using RIPA lysis buffer (PC101, Epizyme, Shanghai, China) and subjected to Western blot as previously described by our group [2]. PVDF (WJ001, Epizyme, China) membranes were incubated overnight at 4 °C with the following primary antibodies: MMP-9 rabbit polyclonal antibody (1:1000, A0289, ABclonal, Wuhan, Hubei, China), p38 MAPK rabbit antibody (1:1000, A0227, ABclonal, China), p-p38 MAPK rabbit antibody (1:1000, AP0526, ABclonal, China), and GAPDH (1:5000, K200057M, Solarbio, Beijing, China) as the loading control. After washing three times with TBST (G0001, Servicebio, Wuhan, Hubei, China), the membranes were incubated with corresponding secondary antibodies for 1 h at room temperature. The protein bands were visualized using a chemiluminescence reagent (SQ202, Epizyme, China), with some images equivalently enhanced for ease of visualization.
The concentrations of specific proteins in the culture medium of PVM/Ms were
measured with the following commercial ELISA kits according to the manufacturer’s
instructions: pigment epithelial-derived factor (PEDF) (CSB-E08820, CUSABIO,
Wuhan, Hubei, China), interleukin-6 (IL-6) (RK00008, ABclonal, China),
TNF-
ROS in PVM/Ms was evaluated with a ROS assay kit (S0033S, Beyotime, Shanghai, China). PVM/Ms were seeded into 6-well plates and incubated with LPS (1 µg/mL) for 24 h after different pretreatments. Subsequently, the cells were incubated at 37 °C in the dark for 30 min with 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA) at a final concentration of 10 µM. Cells were subsequently treated with trypsin and washed three times with PBS. The production of ROS was measured by flow cytometer (BD Biosciences, San Jose, CA, USA) at emission and excitation wavelengths of 535 and 488 nm, respectively.
Purified primary PVM/Ms (passage 3~4) were seeded into glass well dishes and treated with LPS (1 µg/mL) for 24 h after different pretreatments. The PVM/Ms were subsequently fixed in 4% PFA (Solarbio, P1110, China) for 15 min, permeabilized in 0.25% Triton X-100 (Solarbio, T8200, China) in PBS for 5 min, and blocked with 10% goat serum. They were then incubated overnight at 4 °C with rat monoclonal antibody F4/80 (14-4801-85, eBioscience, San Diego, CA, USA) diluted 1:50 in 1% PBS-bovine serum albumin. After three washes in PBS, the samples were incubated with 4′,6-diamidino-2-phenylindole (C0065, Solarbio, China) and with the secondary antibodies Dylight 488 goat anti-rat IgG (A23240, Abbkine, Wuhan, Hubei, China) at 1:200 and Griffonia simplicifolia-IB4 (GS-IB4) conjugated to alexa fluor 568 (I21412, Life Technologies, Eugene, OR, USA) at 1:100 for 1 h.
ECs were seeded onto polyethylene terephthalate membranes of transwell inserts (353095, Corning Falcon, Corning, NY, USA) and cultured for 2–3 days to form an EC-only monolayer. For coculture models, PVM/Ms were seeded in advance in the lower well surface of the transwell receiver trays for 1–2 days, and then co-cultured with ECs that had formed a confluent monolayer. Chambers were examined microscopically for EC monolayer confluence, integrity, and uniformity. The permeability of EC monolayers was assessed by quantifying the flux of FITC-dextran (FD10S, Sigma-Aldrich, St. Louis, MO, USA) in the upper and lower chambers. FITC-dextran was added to the upper chamber at a final concentration of 1 mg/mL. After 20 min incubation in darkness, 100 µL of medium was collected from the receiver tray and transferred to 96-well plates for measurement. The permeability of EC monolayers was assessed by the detection of FITC-dextran fluorescence using a plate reader (SpectraMax M5, Molecular Devices, San Jose, CA, USA), with filters for 485 nm excitation and 535 nm emission.
SPSS 26.0 software (SPSS Inc., Chicago, IL, USA) was used to perform all
statistical analyses. Data were presented as the mean
The levels of inflammatory factors IL-6 and TNF-
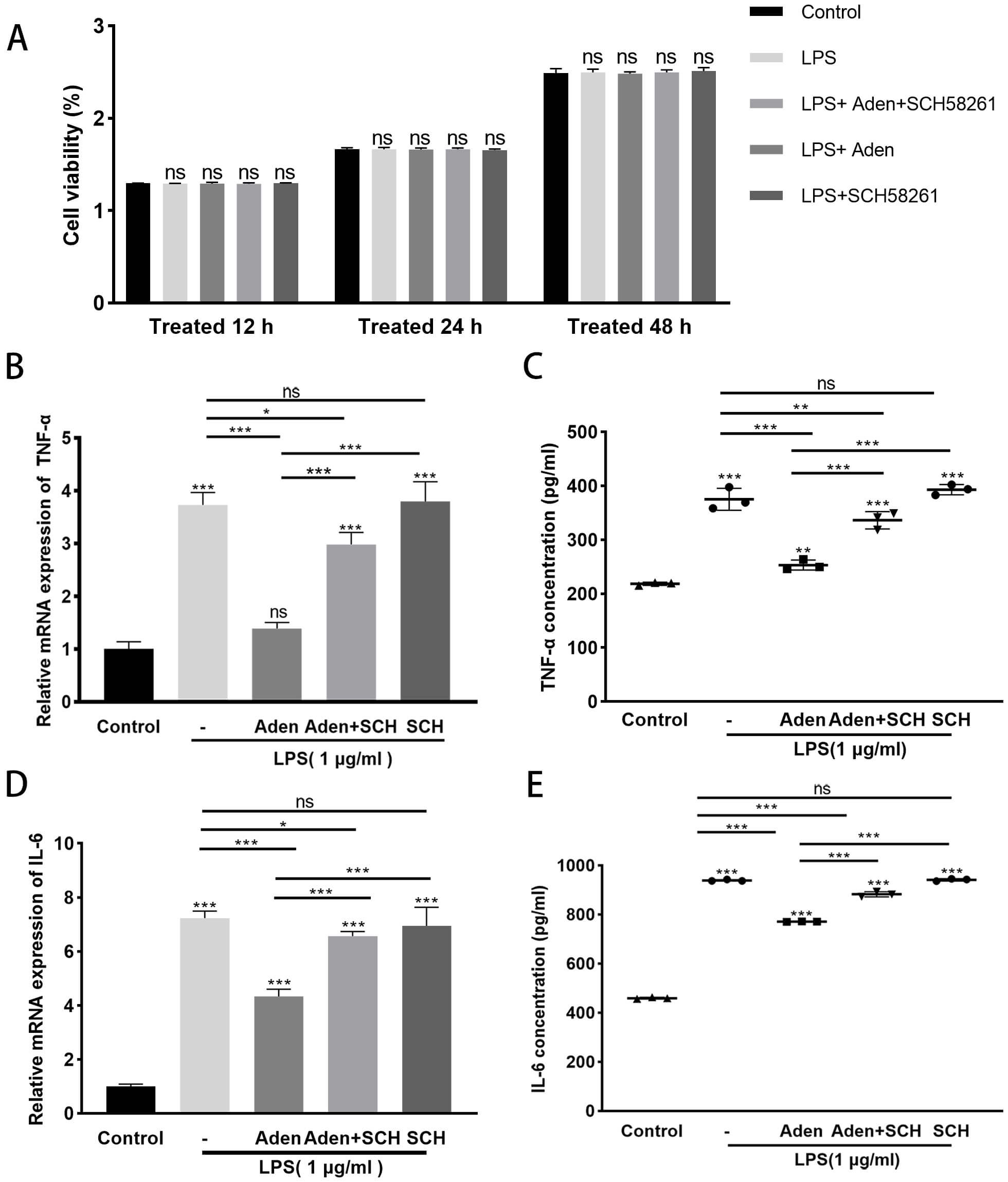 Fig. 1.
Fig. 1.
Adenosine decreases expression of the inflammatory factors IL-6
and TNF-
The overproduction of ROS is closely associated with disruption of the BBB [18, 25]. Furthermore, several studies have reported that tissue damage is closely related to an increased level of MMP-9 [26, 27, 28, 29]. The present study investigated the effects of adenosine on ROS and MMP-9 levels in PVM/Ms. ROS levels were found to be markedly increased following LPS treatment of PVM/Ms. Adenosine treatment reduced the level of ROS, but this effect was reversed by treatment with the A2AR antagonist SCH58261 (Fig. 2A). In addition, Western blot, qRT-PCR and ELISA assays revealed that MMP-9 expression in PVM/Ms increased following LPS treatment, whereas TIMP-1 expression decreased. These changes were prevented by adenosine treatment, while SCH58261 partially counteracted the effects of adenosine (Fig. 2B–E). In summary, adenosine decreased ROS levels and MMP-9 protein expression, and increased TIMP-1 expression in LPS-treated PVM/Ms. These effects appear to be mediated via A2AR.
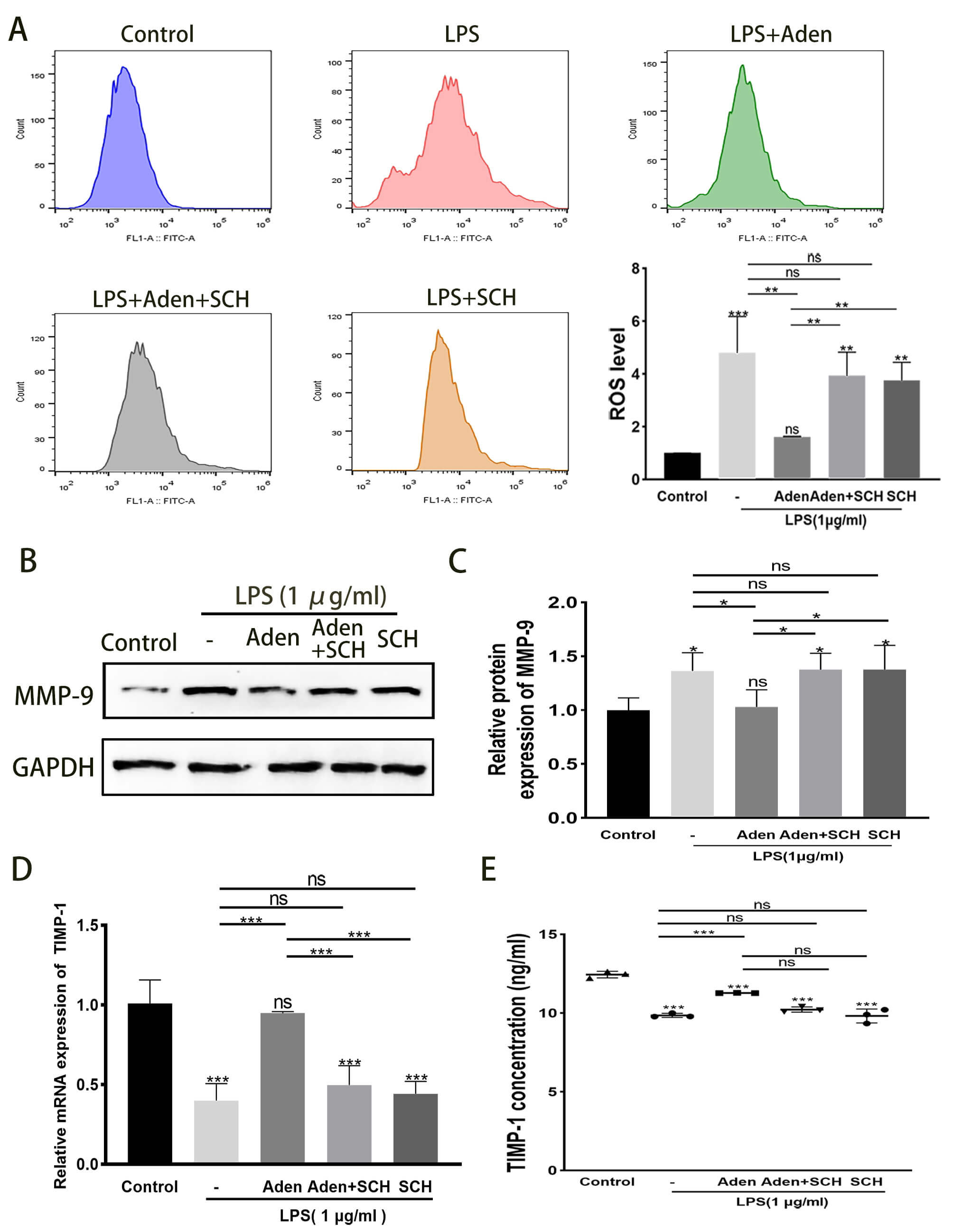 Fig. 2.
Fig. 2.
Adenosine decreases ROS levels and MMP-9 protein expression and
increases TIMP-1 expression in LPS-treated PVM/Ms by activating A2AR. (A) ROS
levels were measured by flow cytometry. Adenosine decreases ROS levels in
LPS-treated PVM/Ms by activating A2AR. (B–E) Changes in mRNA and protein
expression of MMP-9 and TIMP-1 in PVM/Ms. Values shown are the mean
PEDF, a key signaling molecule secreted by PVM/Ms, regulates barrier integrity by modulating the expression of tight junction-related proteins [2, 3]. Inflammatory damage to cochlear BLB was shown to be closely related to the activation of PVM/Ms [3, 5]. Zhang et al. [5] previously reported that LPS activated PVM/Ms, resulting in significant morphological changes and positive GS-IB4 staining. In agreement with these earlier results, the present study also found that LPS indeed activated PVM/Ms. For example, these showed altered morphology, with some becoming flattened and amoeboid in shape. Moreover, the dendrites of PVM/Ms were observed to be significantly shortened (Fig. 3A,B). A significant decrease in the activation of PVM/Ms was observed following treatment with adenosine, with the dendritic shape becoming slightly longer than in the LPS group. The aforementioned changes were significantly mitigated by inhibition of A2AR (Fig. 3C–E). Similarly, adenosine treatment resulted in the upregulation of PEDF expression in LPS-treated PVM/Ms, which could be prevented by SCH58261 treatment (Fig. 3F,G). The above results indicate that adenosine attenuates LPS-induced PVM/Ms activation and increases PEDF secretion by activating A2AR.
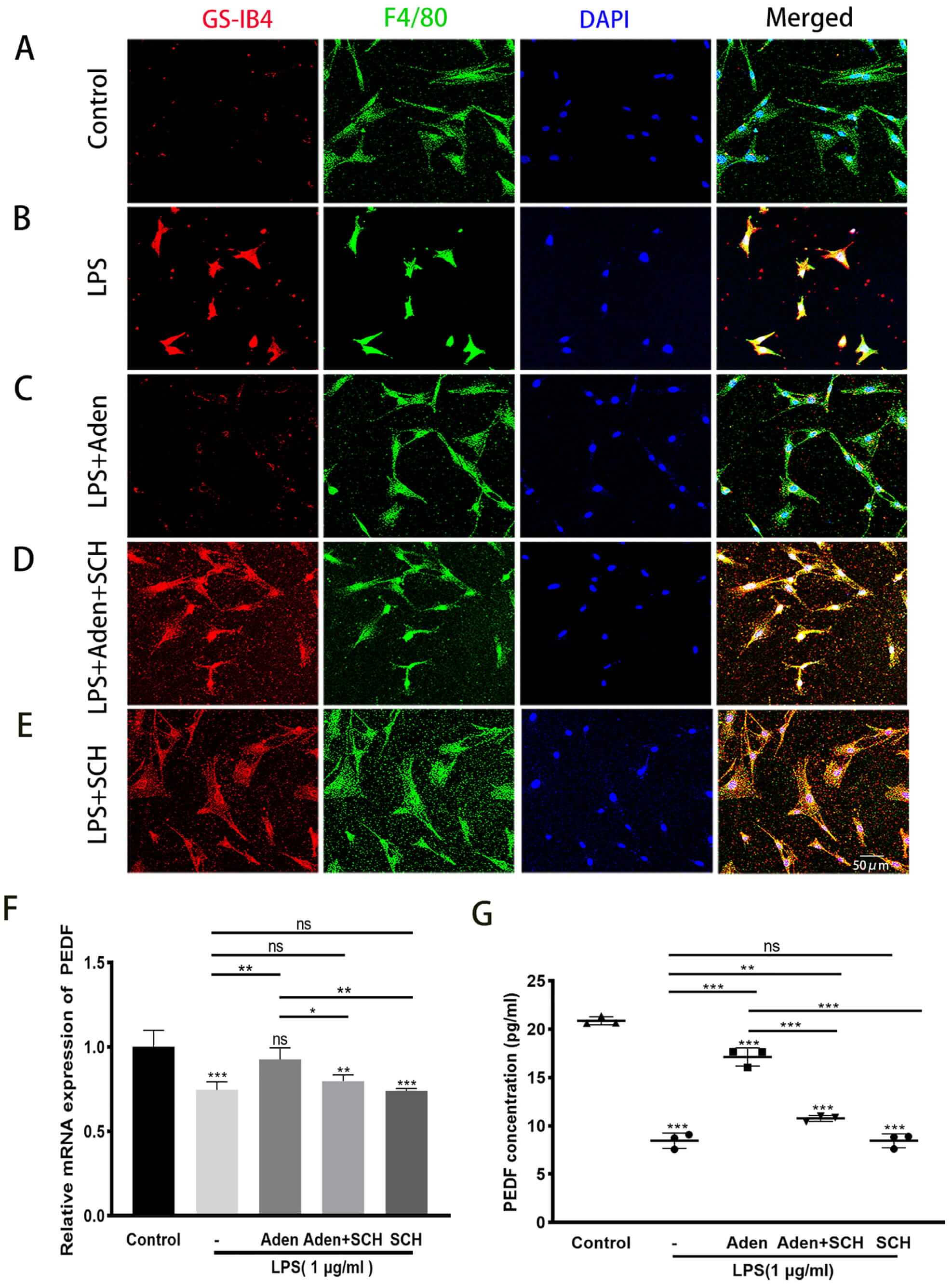 Fig. 3.
Fig. 3.
Adenosine attenuates LPS-treated PVM/M activation and increases
PEDF secretion by activating A2AR. (A–E) Adenosine inhibited the inflammatory
activation of PVM/Ms and increased dendritic length compared to the LPS group.
These changes were significantly attenuated following inhibition of A2AR. PVM/Ms
were labeled with an antibody for F4/80 (green), while the nucleus was labeled
with DAPI (blue). Activated PVM/Ms show red fluorescence due to the presence of a
terminal galactopyranosyl group on the membrane surface which binds the lectin
GS-IB4. Scale bar = 50 µm. (F,G) mRNA and protein expression of PEDF in the
PVM/Ms. Adenosine increased the expression of PEDF in LPS-induced PVM/Ms. This
effect was reversed by SCH (SCH58261). Values shown are the mean
Previous research has demonstrated that anti-inflammatory drug DMH-CBD activated
macrophage A2AR targets NF-
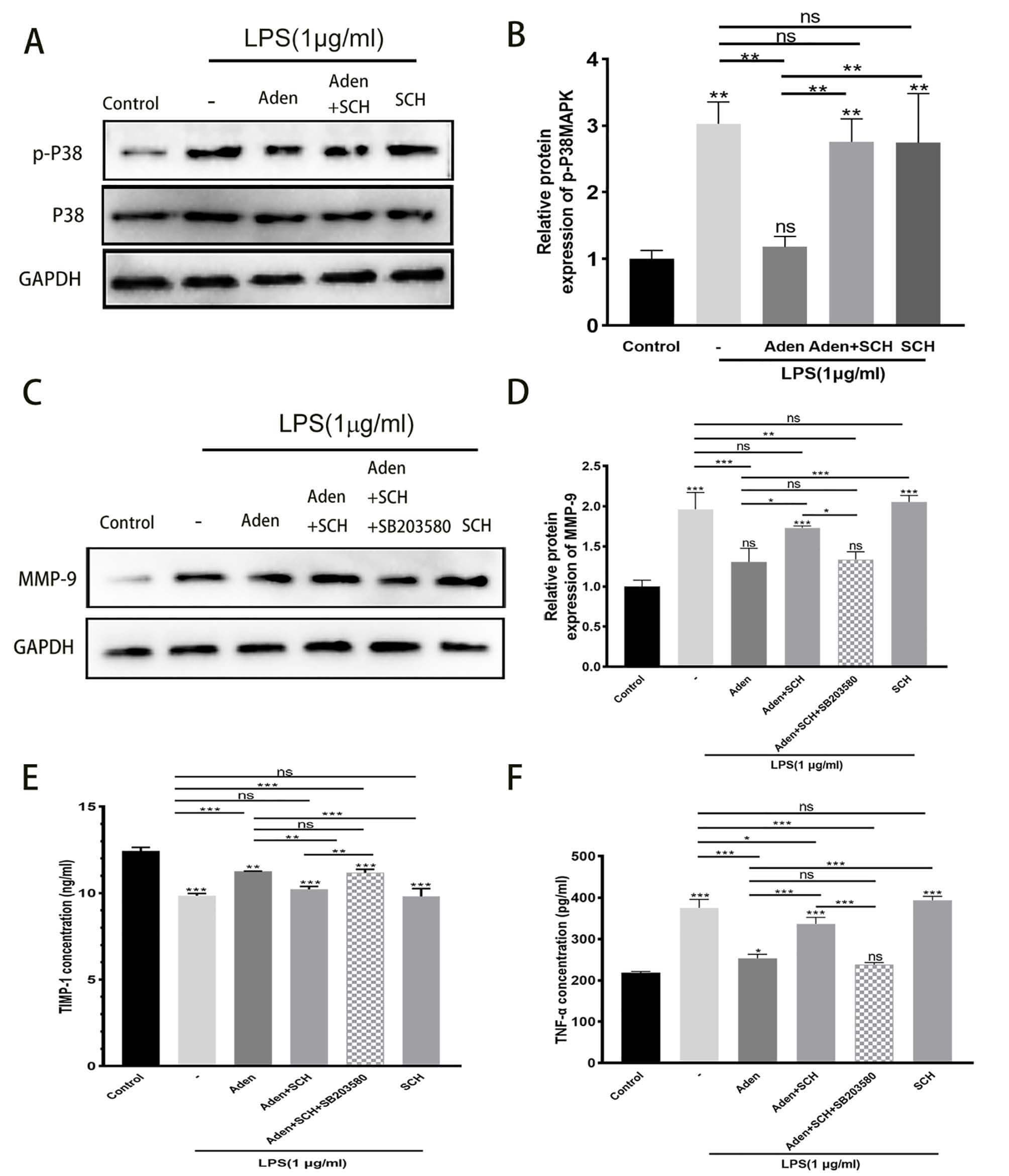 Fig. 4.
Fig. 4.
Adenosine reverses the increase in MMP-9 and TNF-
We next established in vitro models of EC monolayers and EC-PVM/M co-culture to further examine the effects of adenosine on BLB integrity. Whereas previous research demonstrated protective effects of adenosine on ECs [20], the present study examined its effects on PVM/Ms. A schematic diagram of the BLB model and in vitro experimental setup is illustrated in Fig. 5A. The permeability of the EC monolayer barrier was determined by evaluating the fluorescence intensity of FITC-dextran crossing the cell monolayer paracellularly into the inferior chamber of a Transwell. FITC-dextran leakage was observed to be lower in the co-culture group than in the EC monolayer group (Fig. 5B), suggesting that PVM/Ms play a beneficial role in maintaining the integrity of the endothelial barrier, in accordance with the findings by Zhang et al. [2]. However, following LPS-treatment the EC monolayer barrier was found to be more permeable than the EC-PVM/M co-culture (one-way ANOVA). Pre-treatment with adenosine significantly decreased the LPS-induced FITC-dextran leakage in both groups, which was abrogated by treatment with SCH58261. These in vitro findings demonstrate that adenosine reduces the permeability of the BLB following LPS treatment by activating A2AR, with a positive effect on PVM/Ms as well as ECs.
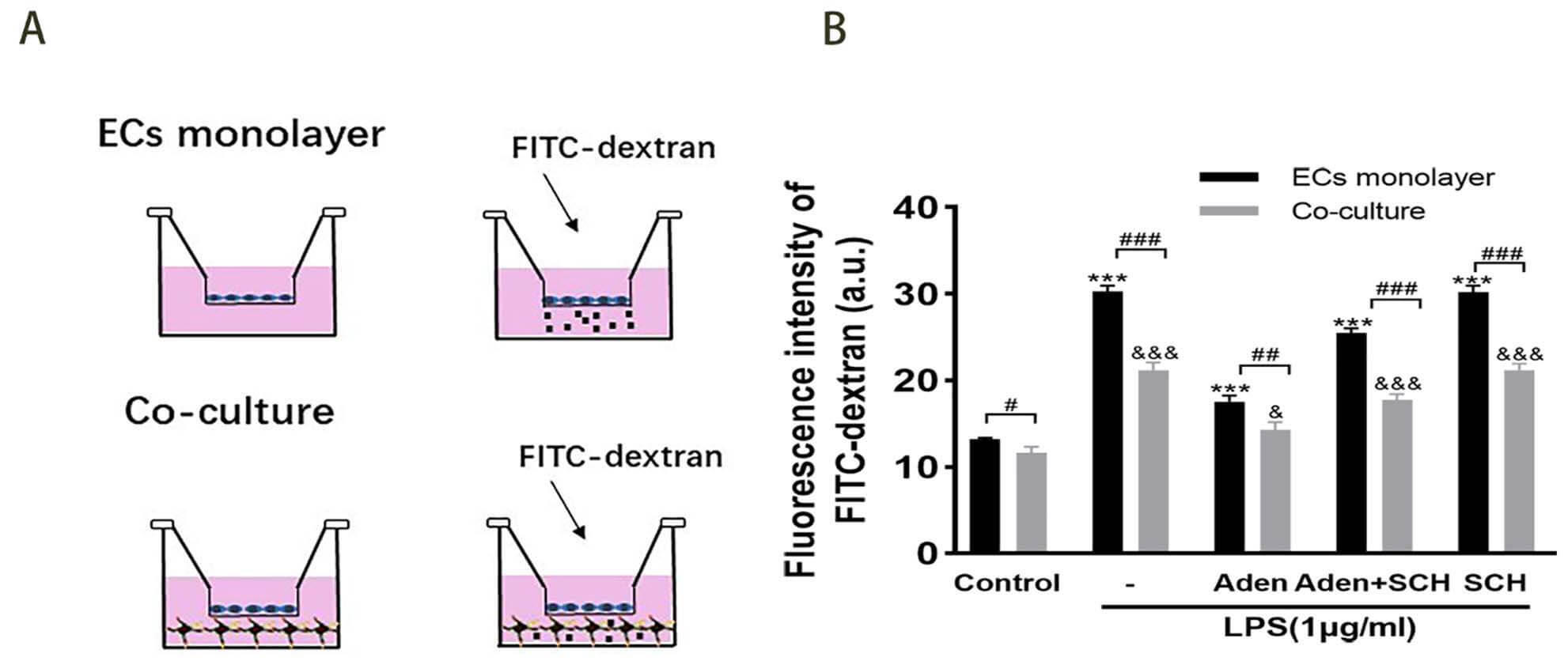 Fig. 5.
Fig. 5.
Adenosine reduces the permeability of the BLB following LPS
treatment by activating A2AR. (A) Schematics of the BLB models and in
vitro experimental setup to detect paracellular permeability of the EC monolayer
using a Transwell chamber. The arrows indicate FITC-dextran dropped in the upper
chamber. (B) Quantitative analysis of FITC-dextran in the lower transwell chamber
in the presence or absence of PVM/Ms. There was significantly less leakage of
FITC-dextran in the adenosine-treated group than in the LPS-treated group. SCH
was able to reverse this change. Values shown are the mean
The authors have previously demonstrated that PVM/Ms, the resident macrophages of the stria vascularis, play a critical role in maintaining tissue homeostasis and BLB integrity [2]. PVM/Ms are activated in animals exposed to loud sound or in aged animals, and are closely associated with BLB dysfunction [3, 4]. In the present study, LPS treatment of PVM/Ms was used to mimic the inflammatory response in vivo. Adenosine was found to attenuate LPS-induced PVM/M activation and to increase PEDF secretion by activating A2AR. It is well known that LPS can provoke macrophages to differentiate into the inflammatory phenotype via the p38 MAPK pathway [32]. The present study found that adenosine attenuated the phosphorylation of p38 MAPK in PVM/Ms, and this effect was reversed by an A2AR inhibitor.
Dysfunction of the BLB is associated with a number of hearing disorders [1]. The essential role of PVM/Ms in regulating BLB integrity is mainly due to their communication with ECs in the capillary network skeleton that control microvascular permeability [2]. In another previous study, the authors reported that LPS activation of PVM/Ms resulted in significant morphological changes, as well as the disruption of BLB integrity by reducing the expression of tight junction proteins between ECs, including ZO-1 and occludin [5]. Adenosine is an ancient extracellular signalling molecule that can regulate almost all aspects of tissue function [32, 33]. A number of studies have shown that A2AR plays a favorable role in brain barrier integrity [10, 21]. The potential protective role of adenosine in the auditory system was also discussed recently, although this was limited to sensory hair cells [34]. The extensive range of neuroprotective action suggests that adenosine may also exert a protective effect in the cochlea. The authors previously demonstrated that adenosine partially reversed the LPS-induced decrease in tight junction-related proteins (ZO-1 and occludin) to protect BLB integrity in ECs by acting on A2AR [20]. The present study further demonstrated that LPS induced the activation of PVM/Ms, thereby losing the capacity to produce PEDF. These changes were significantly mitigated following the inhibition of A2AR. PVM/Ms are known to regulate the integrity of the BLB through secretion of the signaling molecule PEDF [2], which upregulates the expression of tight junction-associated proteins between ECs [2]. The present study found that adenosine increased the expression of PEDF in LPS-treated PVM/Ms, and this effect was blocked by the adenosine A2AR antagonist SCH58261 (Fig. 3F,G). These results indicate that adenosine attenuates LPS-induced PVM/M activation and increases PEDF secretion by activating A2AR. The integrity of the barrier is critical for maintaining inner ear homeostasis [1, 35], suggesting that adenosine may be a promising target for hearing impairment caused by BLB dysfunction.
MAPKs are involved in the ERK, p38 and JNK pathways, with p38 MAPK being a key
enzyme in cell signaling pathways responsible for the production of inflammatory
cytokines [36]. The activated macrophage A2A receptor was recently reported to
target NF-
In the brain, adenosine inhibits inflammatory cytokine production by activated
primary murine microglia via A2A receptors [42]. Similarly, adenosine was found
to target PVM/M activation, thereby decreasing the release of inflammatory
cytokines such as IL-6, TNF-
In conclusion, the present study demonstrates that adenosine acts via A2AR to attenuate LPS-induced PVM/M injury. This maintains integrity of the endothelial barrier by modulating the inflammatory activation of PVM/Ms and promoting PEDF expression. Inhibition of p38 phosphorylation in the MAPK pathway may be one of the underlying mechanisms. These findings suggest potential targets for the development of adenosine-based therapeutics for hearing disorders associated with PVM/M activation.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
WF and JL contributed equally to the study. Specifically, WF, JL, ZX, and LN did the immunohistochemistry, qRT-PCR and WB. BC and BZ were involved in data analysis. JZ designed the research study, analyzed the experimental results and wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All experimental procedures involving animals in this study complied with the NIH Guide for the Care and Use of Laboratory Animals and were approved by the Ethics Committee of First Affiliated Hospital of Zhengzhou University (2024-KY-0035-002).
Not applicable.
This study was supported by the Natural Science Foundation of Henan Province of China (Grant no. 242300420078), the Henan Province Higher Education Institutions Key Scientific Research Project Plan (Grant no. 26A320041), funds from the Science Foundation for The Excellent Youth Scholars of Zhengzhou University (Grant no. JC23862072).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





